Abstract
Objective
Meniscus injuries in the inner avascular zone have weak intrinsic self-healing capacity and often progress to osteoarthritis. This study focused on evaluating the effects of polycaprolactone/silk fibroin/gelatin/ascorbic acid (PCL/SF/Gel/AA) composite scaffolds seeded with adipose-derived mesenchymal stem cells (ASCs), in the meniscus repair.
Design
To this end, composite scaffolds were cross-linked using N-hydroxysuccinimide and 1-ethyl-3-(3-dimethyl-aminopropyl)-1-carbodiimide hydrochloride. Scaffolds were then characterized by scanning electron microscope, mechanical tests, total antioxidant capacity, swelling, and toxicity tests.
Results
The PCL/SF/Gel/AA scaffolds exhibited suitable mechanical properties. Furthermore, vitamin C rendered them the highest antioxidant capacity. The PCL/SF/Gel/AA scaffolds also showed good biocompatibility and proliferation for chondrocytes. Moreover, the PCL/SF/Gel/AA scaffold seeded with allogeneic ASCs was engrafted in New Zealand rabbits who underwent unilateral punch defect in the medial meniscus of the right knee. After 2 months postimplantation, macroscopic and histologic studies for new meniscus cartilage were performed.
Conclusions
Our results indicated that the PCL/SF/Gel/AA composite scaffolds seeded with allogeneic ASCs could successfully improve meniscus healing in damaged rabbits.
Graphical Summary
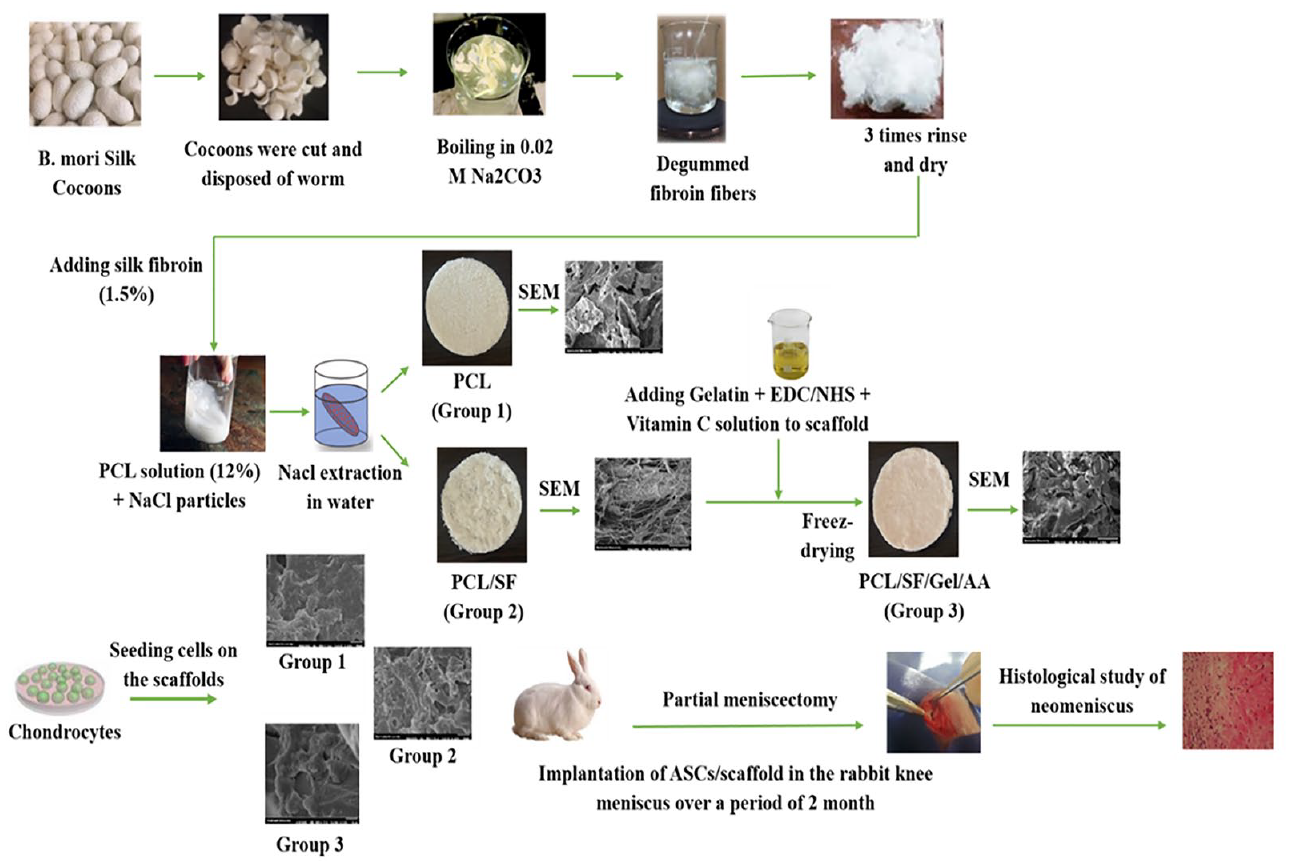
Graphical representation of the fibroin silk extraction, scaffold fabrication, ASCs culture, meniscectomy, grafting of the engineered meniscus scaffolds in the knee joint of the rabbit, and histological study of new meniscus tissue. PCL = polycaprolactone; Gel = gelatin; SF = silk fibroin; AA = ascorbic acid; ASCs = adipose-derived mesenchymal stem cells; NHS = N-hydroxysuccinimide; EDC = 1-ethyl-3-(3-dimethyl-aminopropyl)-1-carbodiimide hydrochloride.
Introduction
Meniscus injuries are common during age-related degeneration, heavy lifting, and sporting events. Normally, these injuries lack the self-healing ability in the white zone of the internal avascular. Because of the critical role of the meniscus in stabilization, buffering, and force distribution, partial or total meniscectomy may lead to imbalance and acceleration of articular cartilage degeneration.1 -3 Among the standard treatment methods, meniscal transplantation has several problems, including lack of donor, risk of infectious disease, anatomical mismatch, immunological rejection, as well as high storage and transportation costs.4,5 Several attempts have already been made to manufacture prostheses; however, they have not been successful, and there are still multiple problems such as synovitis, inadequate strength, wear, and damage to the articular cartilage. 6 Therefore, it seems that tissue engineering is the most appropriate option for meniscus reconstruction to substitute the injured area.
To regenerate the structure and function of the damaged meniscus, various natural macromolecules, synthetic materials, or composites, including collagen, 2 polyurethane, 7 poly lactic-co-glycolic acid (PLGA)-gelatin (Gel), 8 silk fibroin (SF)–polyvinyl alcohol (PVA), 9 Gel/chitosan, 10 polyurethane (PU)-polycaprolactone (PCL), 11 PVA-chitosan, 12 and silk13,14 have been used. However, studies are ongoing to achieve an ideal scaffold, which is more similar to the native tissue, and to regenerate damaged meniscus.
PCL is an aliphatic polyester that is semicrystalline. It is cost-effective, biocompatible, simply processed (its melting point is 60 °C), biodegradable, and has proper mechanical strength. Like other synthetic polymers, PCL has a hydrophobic surface and does not provide a suitable substrate for interaction and cell attachment. Studies have shown that several techniques, such as laser, plasma treatment, and incorporation of some natural polymers that are biologically active, help improve the biological properties of the surface of PCL.15-17 Owing to noted advantages, especially a high mechanical strength that is essential for meniscus tissue engineering, the application of PCL scaffold in combination with polymers such as PU 11 and silk 18 has been proved satisfactory for meniscus tissue engineering. In addition, due to its slow rate of degradation, PCL can provide adequate mechanical strength throughout the repair process of tissue. Although PCL scaffold can render a high mechanical strength, there is a risk of articular cartilage attrition. In addition, since PCL is a synthetic polymer with a hydrophobic surface, it is not a suitable substrate by itself for biological function and cell attachment. In this regard, Zong et al. 18 reported a combination of PCL with elastic SF sponge to improve shock absorption property and decrease the frictional force at the junction between articular cartilage and the scaffold.
Gelatin is a collagen-based biomacromolecule that is widely used in tissue engineering 19 . Gelatin has integrin-binding sites (such as RGD [arginine–glycine–aspartic acid]), and its application in the scaffold increases cell-scaffold interactions and biological function. 20 Gelatin is a hydrophilic polymer with high water absorption properties. Since the meniscus tissue has very few blood vessels, it is mainly nourished via synovial fluid absorption. Consequently, the application of polymers with water adsorption properties in the scaffolds will be helpful to mimic the native meniscus tissue.21,22 According to a report led by Peng, the bilayer scaffold composed of Gel/PLGA showed promising properties for meniscus repair, including good mechanical properties and cellular compatibility. Gelatin combined with other materials (e.g., chitosan 10 and PLGA 8 ) has shown desirable features for meniscus repair strategies. SF is a Food and Drug Administration–approved biopolymer. Fibrous silk is considered a suitable polymer for meniscus tissue engineering due to its high mechanical properties, biocompatibility, and support for the development of chondrogenic events. Also, the controlled degradability of silk biomacromolecule provides enough time for the repair process of tissue. 13 In a study by Pillai et al., 9 the application of SF-PVA constructs along with autoclaved eggshell membrane (AESM) powder, as a reinforcement, displayed the load-bearing ability and better in vitro and in vivo outcomes for meniscus tissue repair. Studies have shown that SF scaffolds, alone13,14 or in combination with PVA 9 and PCL, 18 present favorable stability and mechanical properties to form and function as a meniscus-like tissue.
Vitamins are essential micromolecules that are involved in the appropriate function of an organism. Ascorbic acid (AA) or vitamin C is a water-soluble vitamin that is not synthesized in the human body and is an essential nutrient for the body. AA has an essential function in the synthesis of collagen and some extracellular matrix compounds. Defect in the synthesis of collagen fibers results in the formation of the abnormal extracellular matrix. The human menisci contain 22% collagen, 72% water, and 0.8% glycosaminoglycans (GAGs), and therefore, collagen has a considerable share in the formation of extracellular matrix in the meniscus tissue. In 2002, Clark et al. 23 showed that AA at both transcriptional and post-transcriptional stages could enhance collagen and aggrecan synthesis in an articular cartilage explant system. 23 AA also plays a primary role in the wound healing process.24,25
The application of hybrid scaffolds made from several polymers is a desirable approach to bring together the properties of different polymers and overcome the limitations of a single polymer.26,27 High water absorption, excellent mechanical strength, and flexibility are among the characteristics of meniscus tissue. The current study assumes that the mixture of gelatin and silk with AA and PCL provides a better biomaterial with appropriate mechanical strength, flexibility, water absorption, biocompatibility, and biological interactions. Among all cell sources, mesenchymal stem cells (MSCs), because of their capability of differentiation to different mesenchymal lineages, anti-inflammatory, and immunomodulatory features28-34 are of particular interest. Adipose-derived mesenchymal stem cells (ASCs) obtained from the infrapatellar fat pad of the knee can differentiate into chondrocytes and produce proteoglycans and type II collagen.35,36
Materials and Methods
In this research, materials were bought from the Merck company, Germany, and Sigma-Aldrich, the United States, unless otherwise stated.
In this study, we applied solvent casting/particulate leaching (SCPL) and lyophilization methods to fabricate scaffolds. Mechanical properties (tensile and compression), biocompatibility, swelling rate, total antioxidant capacity (TAC), the morphology of scaffolds, and cell-scaffold interactions were determined in vitro. A punch defect was created in a rabbit model to induce the lesion and implant the scaffolds. The formation of new meniscus tissue at the lesion site was evaluated by histological studies 2 months after implantation.
Silk Fibroin Extraction
The extraction of SF from Bombyx mori was conducted according to a previously published method. 20 Briefly, cocoons were cut and disposed of the worm. Next, they were degummed by boiling for 30 minutes in a 0.02 M Na2CO3 solution. Then, fibers were cleaned thoroughly with hot (60 °C) and then cold distilled water (DW), each for 20 minutes. For the complete extraction of glue-like sericin proteins, this step was repeated 3 times. Finally, silk fibers were dried at room temperature.
Scaffold Preparation
For the first experimental group, the PCL solution was provided via dissolving 1.8 g of PCL in 15 mL of chloroform. The mixture was stirred for 1 hour at room temperature. Next, NaCl crystals (with diameters of over 400 μm) were added into the mix. The ultimate homogeneous solution was left overnight at room temperature to dry. Finally, scaffolds were immersed in DW, which was refreshed daily for 4 days to eliminate NaCl particles. In the second experimental group, 0.225 g of fibrous SF was added to the PCL solution, and the subsequent steps were followed as we did for the first group. The scaffold in the third group was made by modifying the second group as follows: 0.45 g gelatin and 0.15 g AA powder were introduced into 15 mL DW and stirred for 1 hour. After dissolving, the crosslinkers of N-hydroxysuccinimide (NHS)/1-ethyl-3-(3-dimethyl-aminopropyl)-1-carbodiimide hydrochloride (EDC) were added and then stirred further for 3 minutes at room temperature. The EDC/NHS/Gel ratio was adjusted to 1:1:8. This homogeneous solution was then poured into the container containing the dried PCL/SF scaffold and moved to the −20 °C freezer. After 1 day, the scaffolds were removed from the freezer and put into a freeze dryer (Alpha2-4LD, Germany) for 24 hours. Three types of fabricated scaffolds are referred to as PCL, PCL/SF, and PCL/SF/Gel/AA, respectively. In the end, the best scaffold, based on the results of in vitro studies, was selected for implantation in rabbits.
Scaffold Characterization
Scanning Electron Microscopy
The microstructures of scaffolds, such as interconnectivity between the pores and pore size, were detected by scanning electron microscopy (SEM, AIS2100, Seron Technology), in which the operating voltage was adjusted to 20 kV. Dry scaffolds were mounted on aluminum stubs and coated with a thin layer of gold.
Swelling Test
The mass of the dry samples (Wdry) in each experimental group was precisely determined. Then, the specimens were saturated at 25 °C in phosphate-buffered saline (PBS, pH 7.4). At the determined time points, scaffolds were eliminated from PBS and rubbed with a filter paper to remove extra water. Then the weight of scaffolds (Wwet) was measured. The PBS content in the swollen specimens was determined through the following equation: Swelling ratio (%) = [(Wwet-Wdry) / Wdry] × 100
Measurement of Total Antioxidant Capacity of Scaffolds
TAC was estimated using the Kiazist TAC kit (Kiazist, Iran), employing the cupric ion reducing antioxidant capacity (CUPRAC) assay.
Mechanical Properties of Scaffolds
Tensile and compression tests of scaffolds in all experimental groups were performed via a SANTAM STM-50 (ENG Design, Iran) tool in dry and wet conditions. For the wet state, the scaffolds were immersed in PBS for 30 minutes. The uniaxial compression and tension were exerted on the cube-shaped and dumbbell specimens, respectively, with a crosshead speed of 10 mm/min and 50 lb load cell, at 25 ± 2 °C, according to the ASTM D638-98 test method. The modulus was recorded and defined as the slope of the linear part of a stress-strain curve. The test was repeated 3 times for each scaffold.
Cell Culture Studies
Chondrocytes and ASCs
The C28/I2 cell line of human chondrocyte (NCBI Code, C620) originating from the coastal chondrocytes of the juvenile was bought from the Cell Bank of Pasteur Institute of Iran and used for in vitro experiments. The extraction of ACSs was conducted according to the protocol used in our previous study. 12 The cells were cultured in Ham’s/F12 culture medium (Gibco, USA) containing 1% penicillin/streptomycin (Pen-Strep, Gibco, USA) and 10% fetal bovine serum (FBS, Gibco, USA) in the condition of 95% humidity and 5% CO2 at 37 °C. The culture medium was refreshed every 2 days, and the passage of cells was done by 0.25% trypsin-EDTA solution (Gibco, USA).
Morphology of Cell-Loaded Scaffolds
The morphology of cell-loaded scaffolds was observed via an SEM (AIS2100, Seron Technology), and the voltage was adjusted to 20 kV. First, scaffolds were cut into circular shapes to fit into a 96-well plate. Then, the scaffolds were sterilized in 3 steps by a 10% PBS/streptomycin/penicillin solution, a 70% ethanol solution, and ultraviolet irradiation. Then, the samples were incubated at 37 °C with 5% CO2 and 90% humidity in Ham’s/F12 culture medium. 37 After 1 day, the culture medium was discarded. A suspension of 4 × 105 chondrocytes per cm2 was seeded on each scaffold. Next, the samples were immersed in the culture medium and incubated at 37 °C with 5% CO2 and 90% humidity. The medium was refreshed every 2 days. After 6 days, the scaffolds were rinsed with PBS and fixed with 4% paraformaldehyde (PF) for 90 minutes. After that, samples were dehydrated in a graded ethanol-water series to 100% ethanol and then allowed to completely dry at room temperature. Finally, all samples were coated with gold and assessed using SEM.
Determination of Cell Metabolic Activity and DAPI Staining
The viability rate of chondrocytes in the scaffolds was evaluated by an indirect MTT test (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide). For this, the sterilized scaffolds (~20 mg) were incubated in 10 mL PBS (Gibco, USA) at 37 °C and pH 7.4 for 1 month. After the period of incubation, these scaffold-degradation products were gathered and kept at 4 °C. Chondrocytes were cultured in 96-well microplates with a density of 3 × 103 cells per well. After 1 day of incubation, the culture medium was discarded and the supernatant (20 μL) of scaffolds and fresh medium (200 μL) were added into each well. This medium was refreshed every 2 days. On days 1, 4, and 6, the culture medium was removed, and then 100 μl MTT reagent was added onto every scaffold. After 3 hours of incubation at 37 °C in the dark, the MTT reagent was eliminated from the wells. Then, 100 μL DMSO (dimethyl sulfoxide) solvent was added into each well to solve unsolvable formazan crystals. In the end, the absorption of specimens was identified at 570 nm, employing a spectrophotometric microplate reader (ELISA, ICN, Switzerland). The MTT process of assessment was repeated 4 times for all specimens.
For qualitative evaluation, the chondrocytes were observed on fluorescent staining. On days 1 and 3, the culture medium was discarded, and cells were washed with PBS and fixed in a 4% PF solution (pH 7.4) in PBS for 30 minutes. Then, cells were washed with PBS and saturated in a 0.2% solution of Triton X-100 for 20 minutes at room temperature. After eliminating the Triton solution, the cells were washed with PBS, and then 4,6-diamidino-2-phenylindole (DAPI, 1:1000, Sigma-Aldrich, USA) was supplemented in each well for 5 min to color cell nuclei. Finally, cells were PBS-washed and observed by fluorescent microscope (UV-1800 240V, Japan).
Implantation of Constructs in Meniscal Defects: Rabbit Model
Five-month-old female New Zealand white rabbits were randomly divided into 3 categories: (a) the negative control group (empty defect), (b) the native group (positive control or no intervention was received by rabbits), and (c) the experimental group (defect implantation with PCL/SF/Gel/AA scaffold containing allogeneic ASCs; n = 7). Anesthesia was performed by the xylazine and ketamine mixture intramuscular injection. The medial meniscus position of the right knee was exposed by a medial parapatellar arthrotomy incision. Then, the medial meniscus, by displacement of soft and patella tissues, was exposed. A full-thickness meniscectomy with a diameter of 2 mm was performed in the avascular meniscus zone via a biopsy punch. One week before surgery, the scaffolds were sterilized as described above and immersed in a culture medium for 24 hours. Afterward, a suspension of ASCs was seeded on scaffolds. Next, samples were cultured and incubated at 37 °C with 5% CO2 and 90% humidity for 6 days. The medium was refreshed every 2 days. For implantation, the scaffold was cut exactly in the size of the lesion by biopsy punch and implanted at the lesion site ( Fig. 1 ). It was then fixed to the surrounding tissues by suture using 3-0 Monocryl. Tissues were then located at their place, and the skin was sutured by 3-0 resorbable nylon. To prevent infection, an intramuscular injection of cefazolin (100 mg/kg) was done 5 days after surgery. The rabbits had free movement and free access to water and food in their cages. Two months after implantation, the rabbits were anesthetized the same way as noted above. Then, the site of the lesion and the adjacent tissues were gathered and fixed in a 10% neutral buffered formalin. After tissue processing, tissue slides were stained with hematoxylin and eosin, and Masson’s trichrome and histological assessments were performed. A modified quantitative system of meniscus scoring was used for histological scoring that was published and developed for the evaluation of meniscal punch defects ( Table 1 ) 38 by 2 observers blinded to the treated groups.
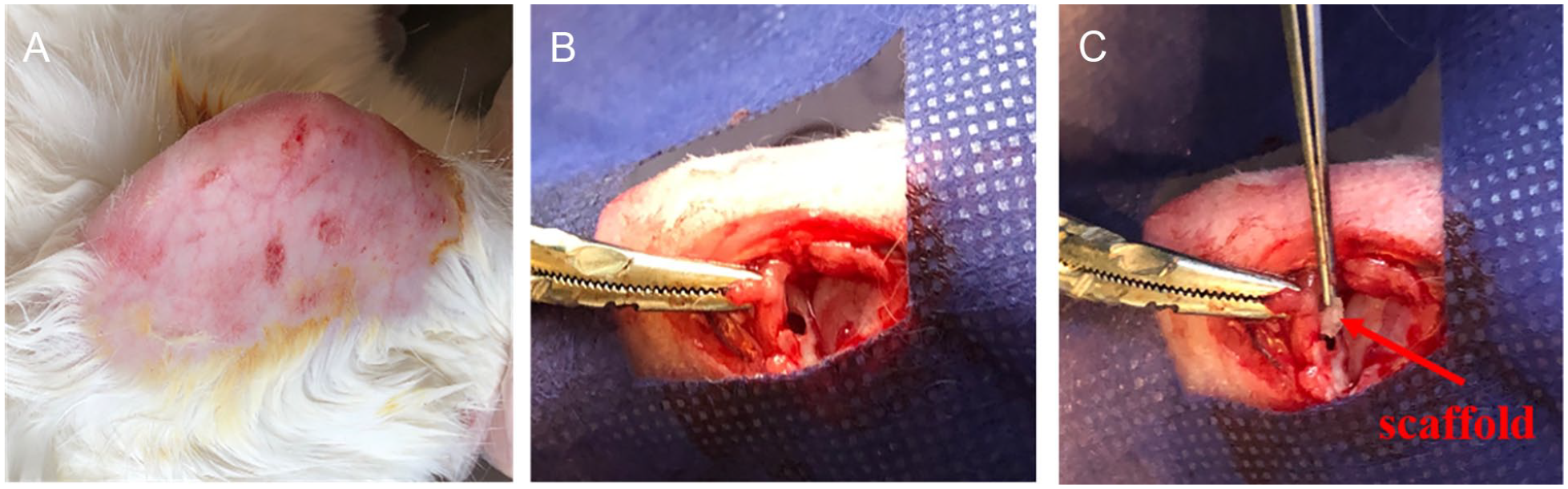
Macroscopic photographs of the surgical process: (
Histological Scoring to Evaluate the Quality of Tissue Healing. 38
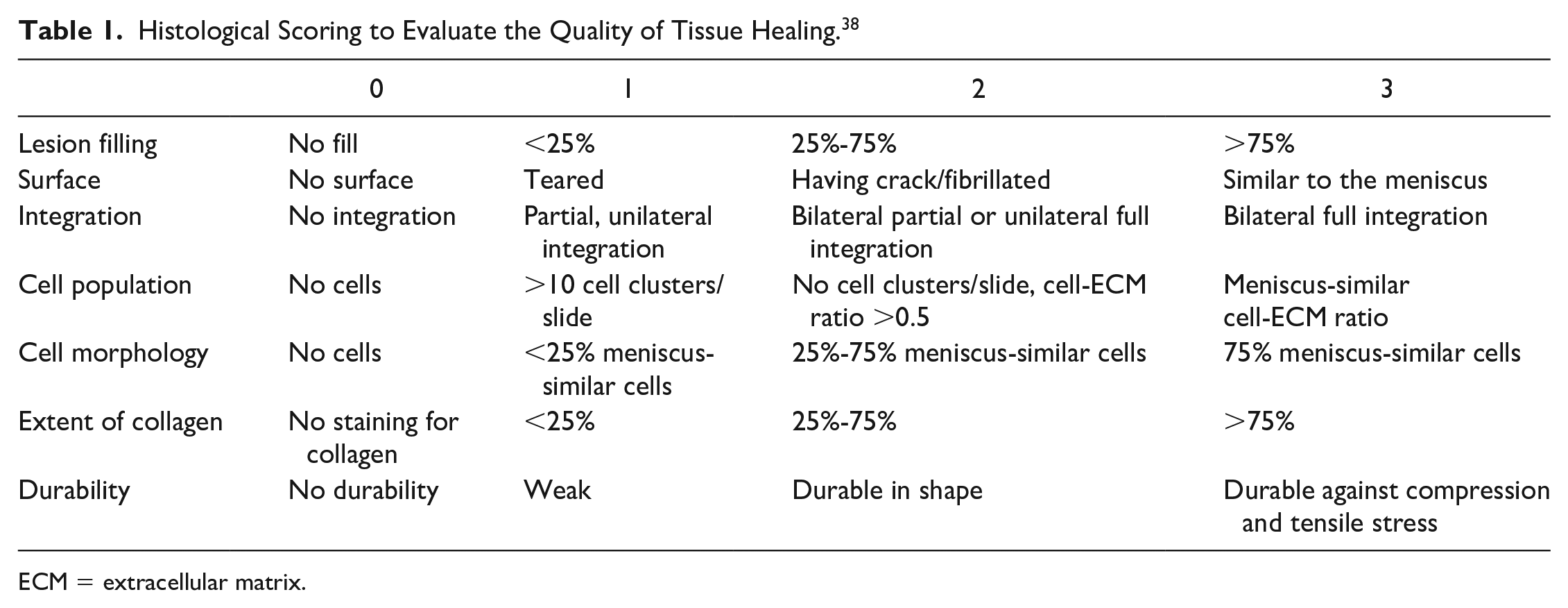
ECM = extracellular matrix.
Statistical Analysis
Information was evaluated as mean ± SD (standard deviation). One-way analysis of variance with Tukey’s test for post hoc analysis was performed using SPSS 21 software to measure differences among the groups, and P < 0.05 was considered statistically significant.
Results
In this research, we intended to construct 3-dimensional (3D) macroporous constructs by solvent-casting and particulate-leaching techniques as well as lyophilization method, which can simulate the repair of meniscus ECM. It was predicted that the 3D structure and also mechanical and biological properties of PCL/SF/Gel/AA scaffolds would improve desired cell-matrix interactions, resulting in meniscal tissue repair. Two other groups of scaffolds were fabricated as the control to evaluate the characteristic of the target scaffold. These control groups were made from pure PCL or PCL/SF composites.
Scanning Electron Microscopy
The macrostructure and microstructure of scaffolds in 3 groups are shown in Figure 2 . The structure of the pores is interconnected in all groups. In SEM images of the PCL/SF scaffold, the silk strand can be observed, while in PCL/SF/Gel/AA blend scaffolds, surface modification of the scaffold with gelatin biomacromolecule caused the silk fibers to be covered with gelatin. It seems that the surface modification of the PCL/SF/Gel/AA scaffold with gelatin biopolymer by freeze drying process decreased the scaffold pore size. The average diameter of SF fibers was measured by Image J software. The total average diameter of SF fibers in PCL/SF and PCL/SF/Gel/AA scaffolds was 9.66 ± 1.45 μm.
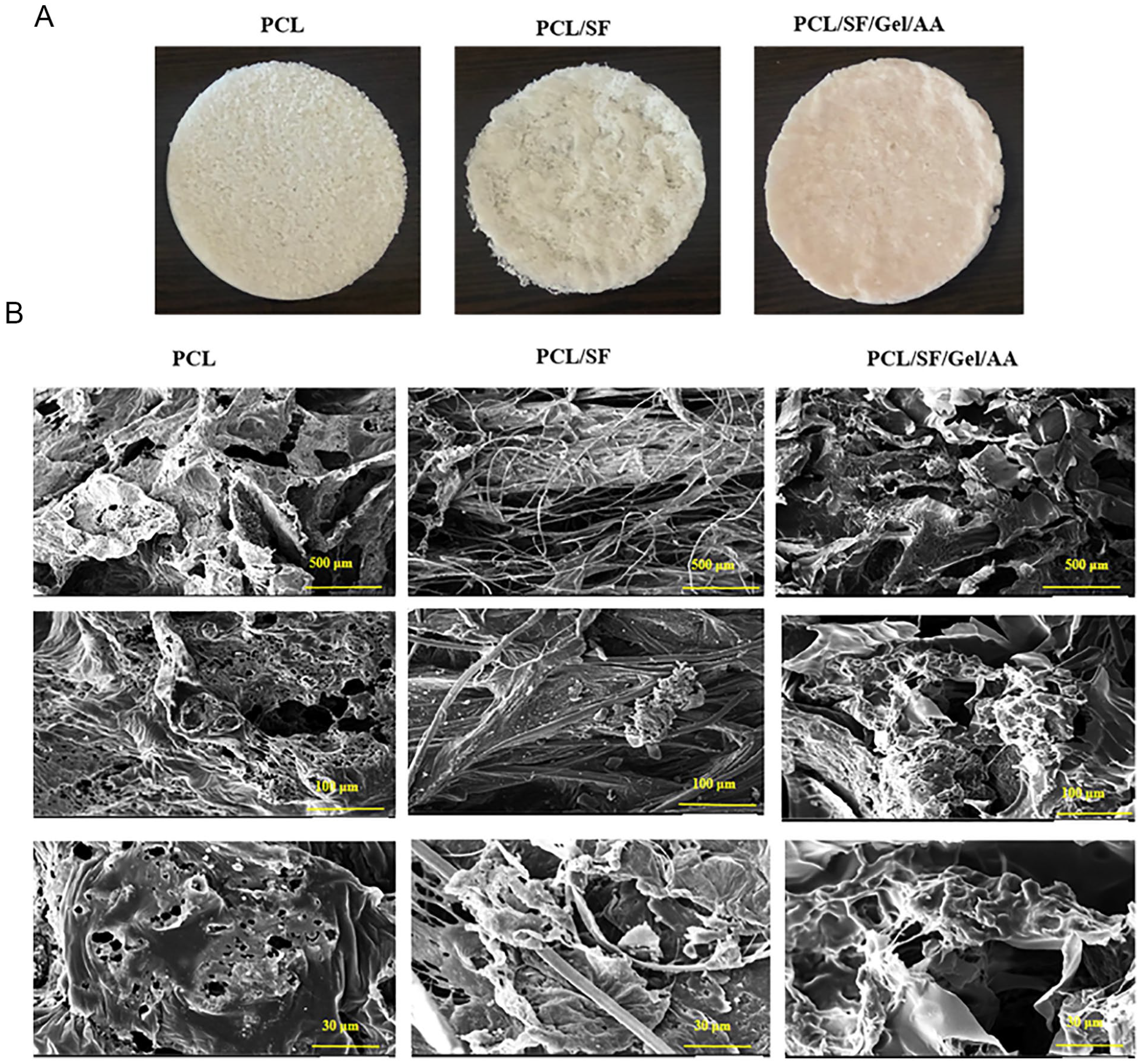
(
Swelling Test
The swelling features were obtained via the rate of water absorption, which is associated with bulk wettability. The scaffold swelling affects degradation, nutrition, and the rate of waste transfer in the scaffolds. 39 The results of the swelling test are provided in Figure 3 and Table 2 . Among the 3 scaffolds, the PCL and PCL/SF scaffolds displayed the highest and lowest swelling rate, respectively.
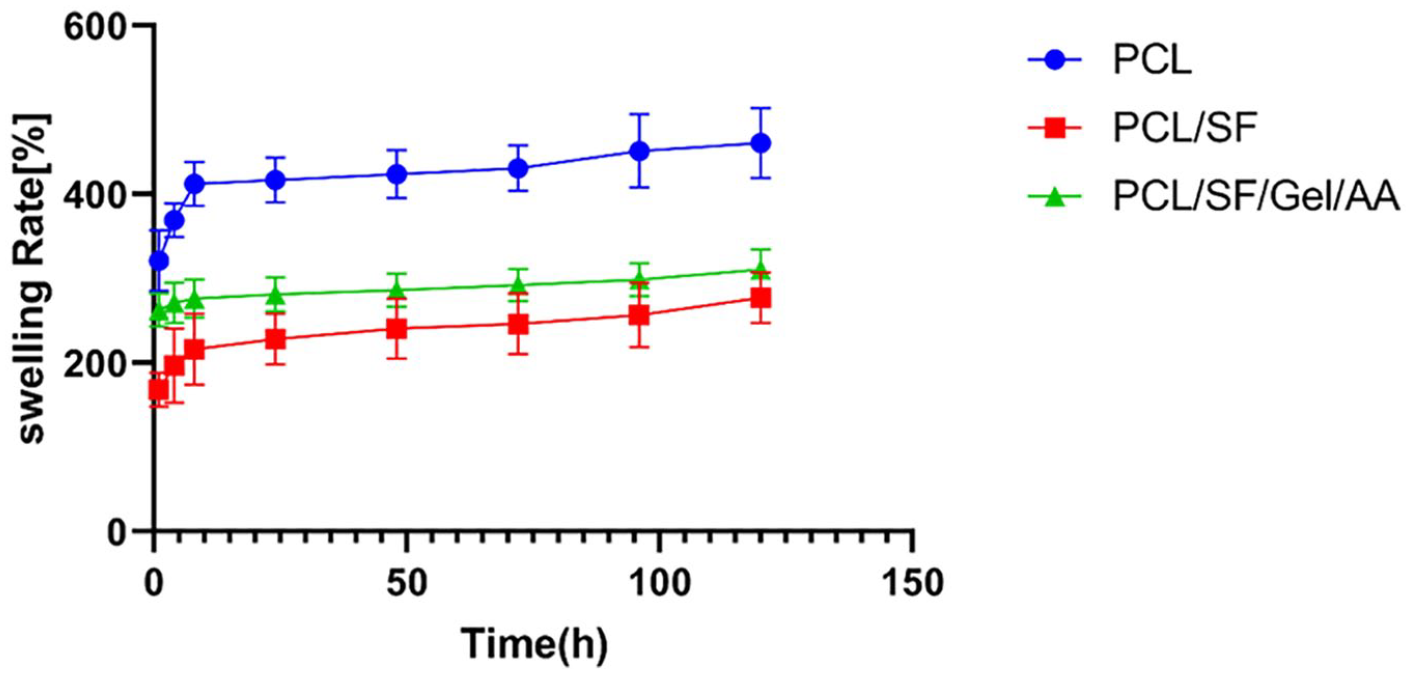
The swelling ratio of scaffolds.
The Swelling Ratio of the Constructs at Various Time Points. a
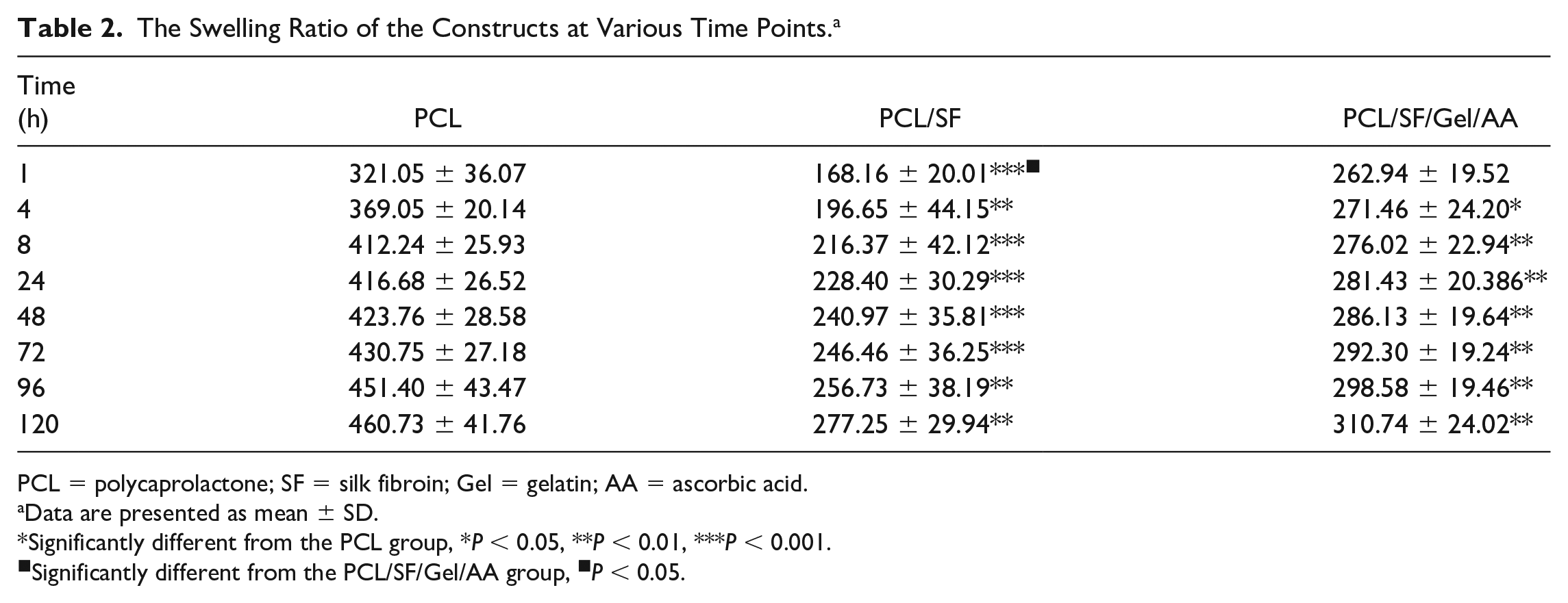
PCL = polycaprolactone; SF = silk fibroin; Gel = gelatin; AA = ascorbic acid.
Data are presented as mean ± SD.
Significantly different from the PCL group, *P < 0.05, **P < 0.01, ***P < 0.001.
Significantly different from the PCL/SF/Gel/AA group, ■P < 0.05.
Total Antioxidant Capacity of Scaffolds
The CUPRAC assay is based on the reduction of Cu+2 (cupric) to the Cu+1 (cuprous) in the presence of antioxidants, which creates color when exposed to chromogen. This color is visible at 450 nm. The absorption rate is directly related to the level of antioxidants in the sample. In this method, antioxidants such as thiol are measured while they are undetectable using FRAP, which works with iron. According to the data presented in Figure 4 and Table 3 , a significant increase was detected in the concentration of antioxidants in the PCL/SF/Gel/AA scaffolds, compared with other groups (P < 0.001). The PCL/SF/Gel/AA scaffold contained AA. Therefore, this construct exhibited the highest antioxidant concentration.
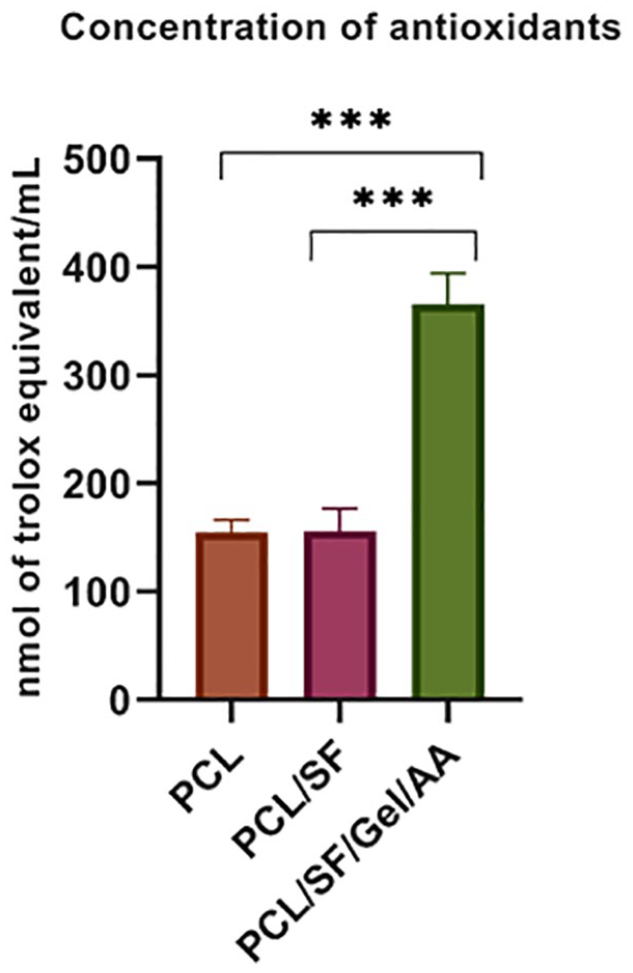
Total antioxidant capacity (TAC) of scaffolds according to the CUPRAC method. ***P < 0.001.
Total Antioxidant Capacity of Scaffolds. a

PCL = polycaprolactone; SF = silk fibroin; Gel = gelatin; AA = ascorbic acid.
Data are presented as mean ± SD. ***P = 0
Mechanical Characterization of the Scaffolds
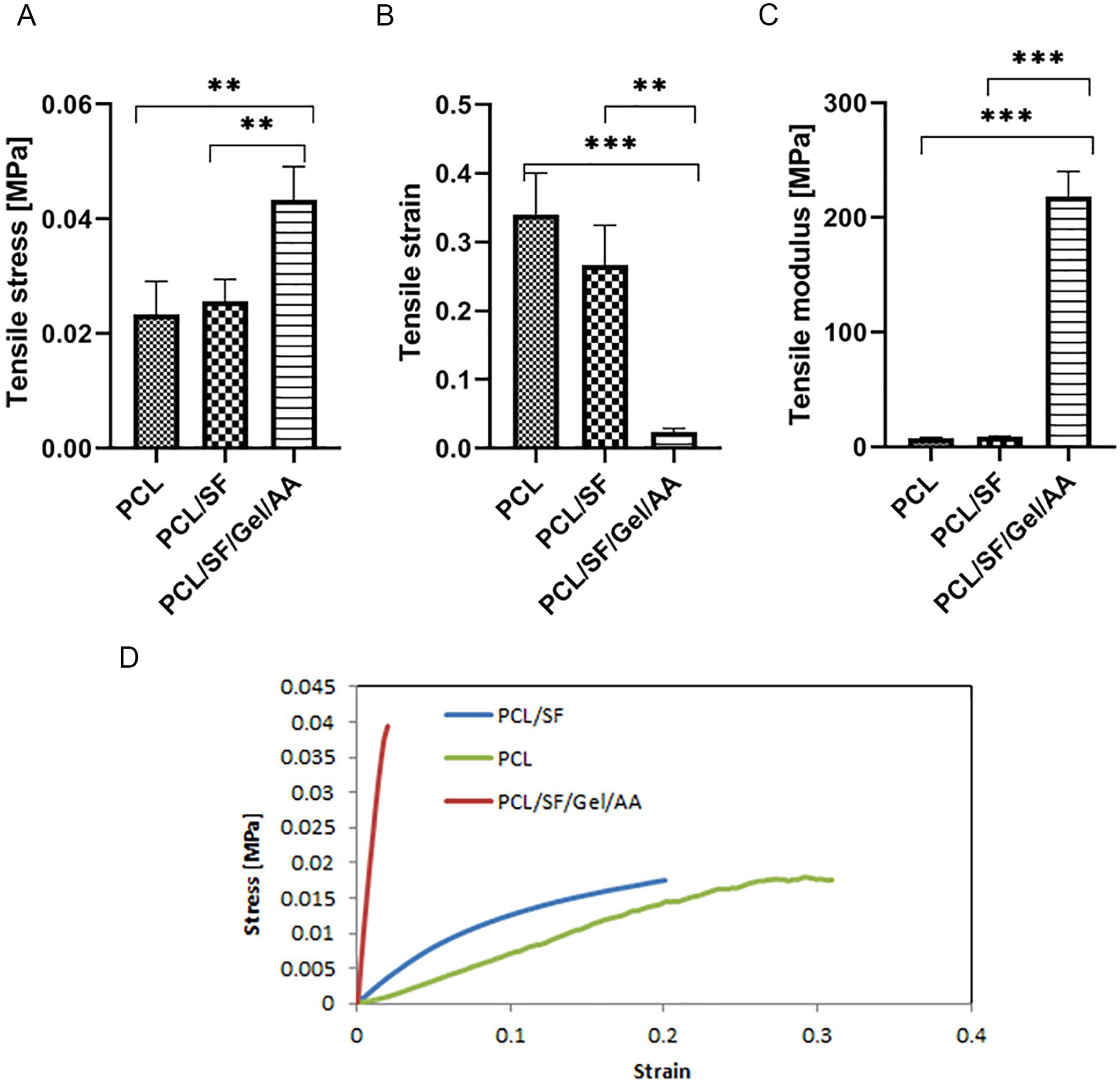
Tensile and compression mechanical tests were accomplished on the scaffolds in dry and wet conditions at room temperature. The stress-strain curves and other mechanical properties such as maximum stress, maximum strain, and Young’s modulus are illustrated in Figures 5 to 8 . The relevant values of the scaffolds in dry and wet conditions and human native meniscus are also presented in Tables 4 to 7 .
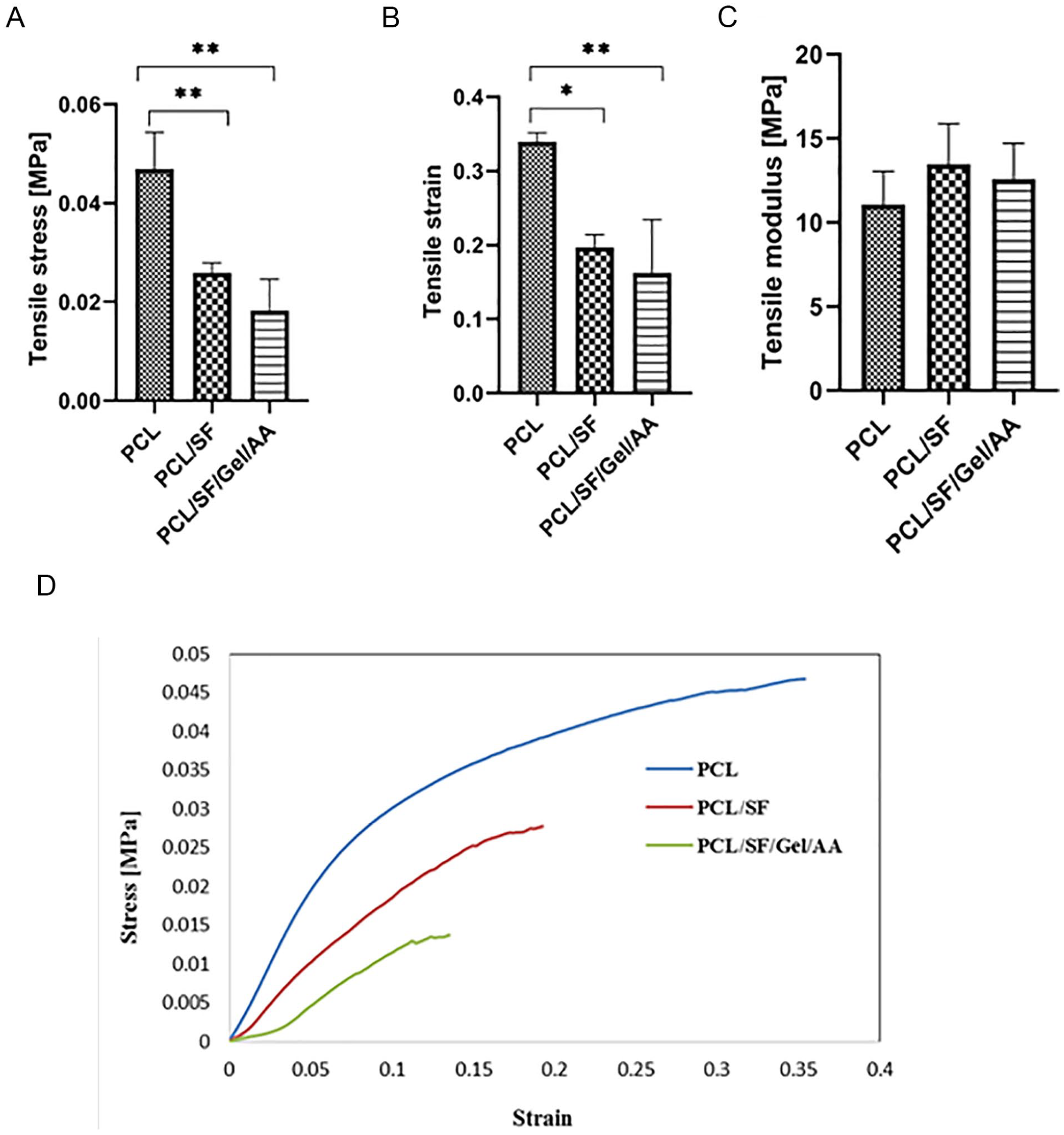
Tensile test of the scaffolds in dry condition. (
Tensile test of the scaffolds in wet condition. The samples were immersed in phosphate-buffered saline (PBS) for 30 minutes. (
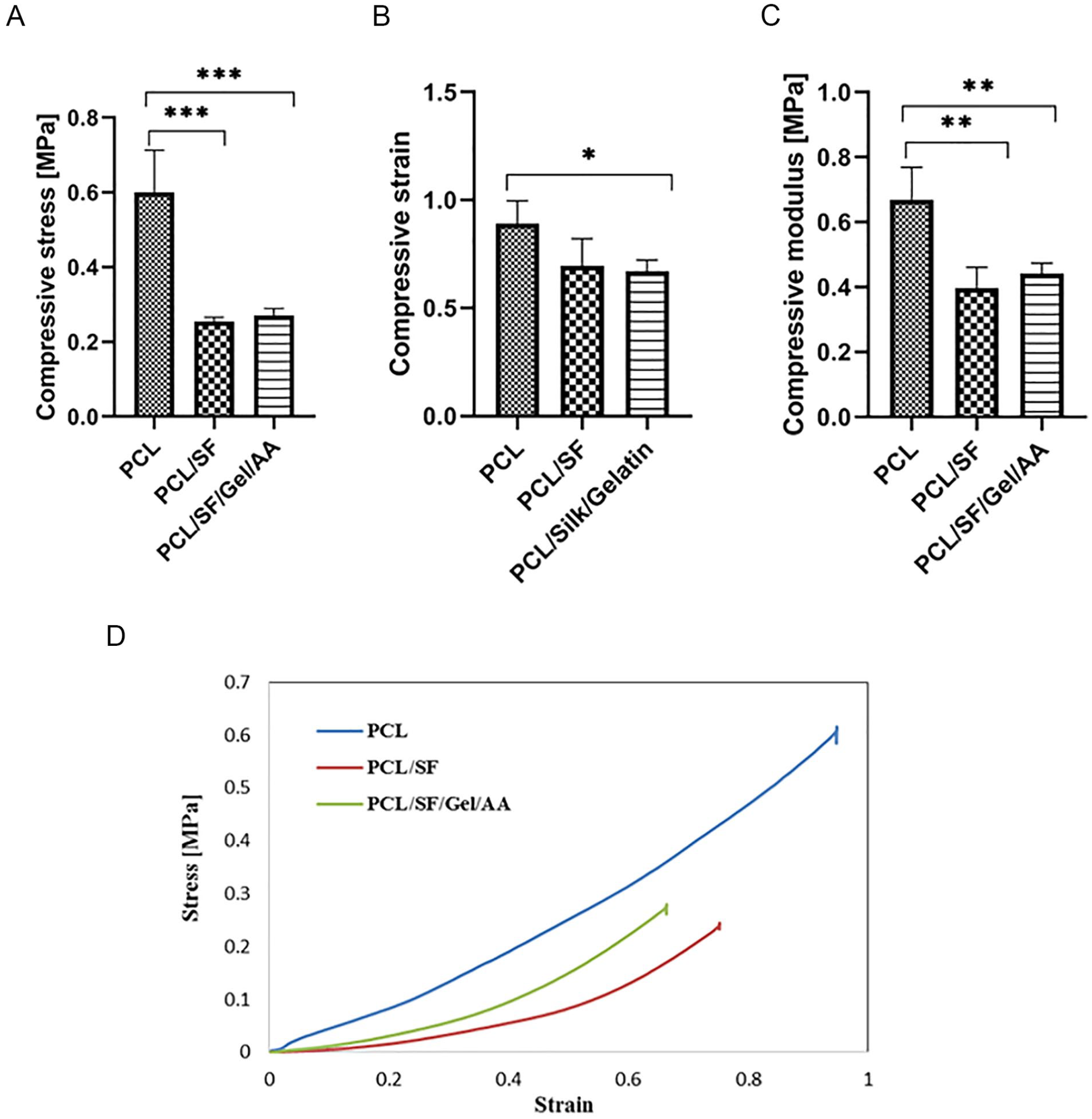
The compression test of the scaffolds in dry condition. (
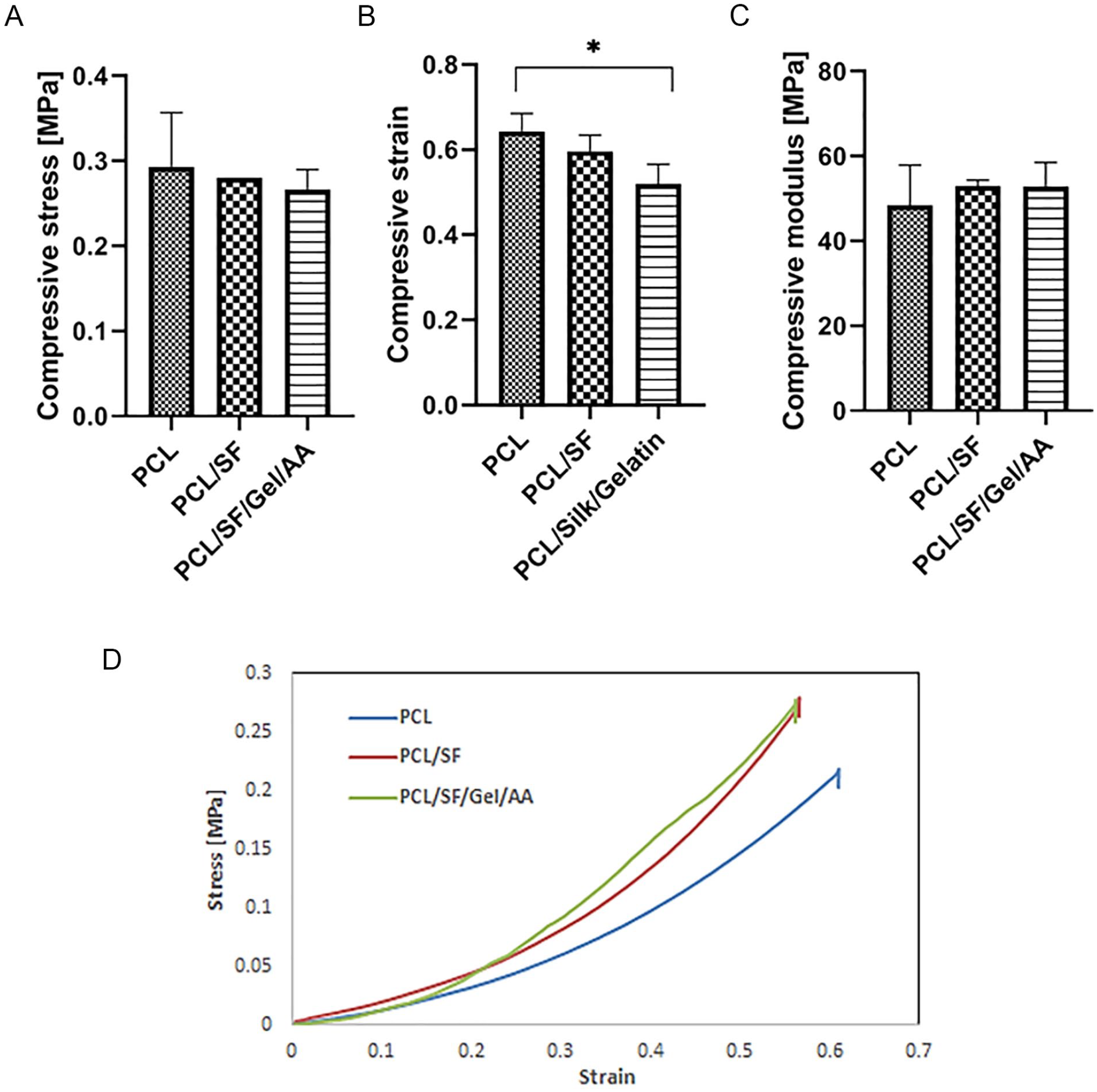
The compression test of the scaffolds in wet condition. The samples were soaked in phosphate-buffered saline (PBS) for 30 minutes. (
Tensile Test of the Engineered Meniscus Constructs in Dry Condition. a
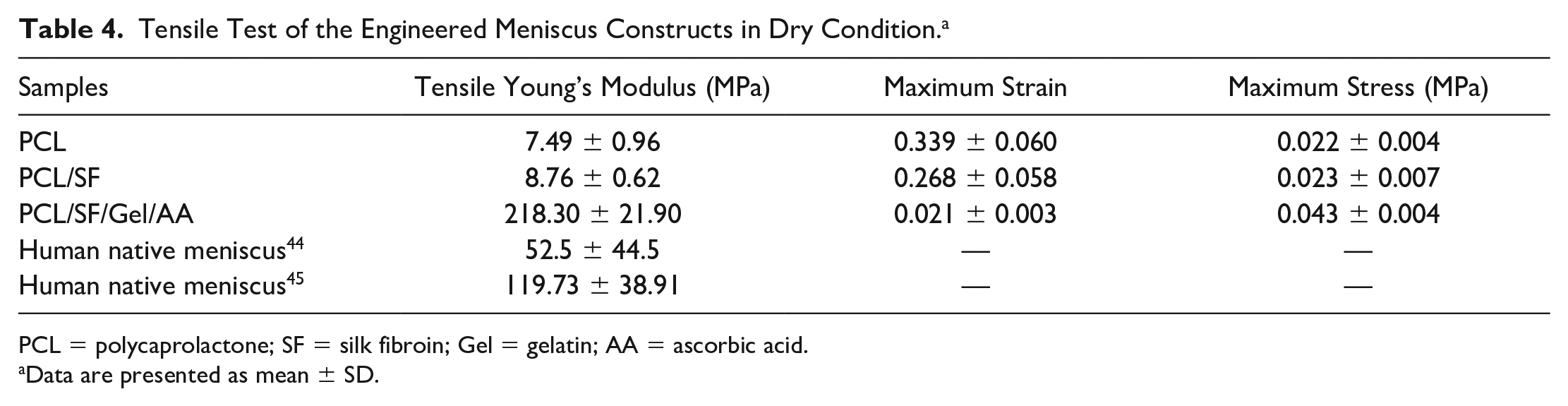
PCL = polycaprolactone; SF = silk fibroin; Gel = gelatin; AA = ascorbic acid.
Data are presented as mean ± SD.
Tensile Test of the Engineered Meniscus Constructs in Wet Condition. a
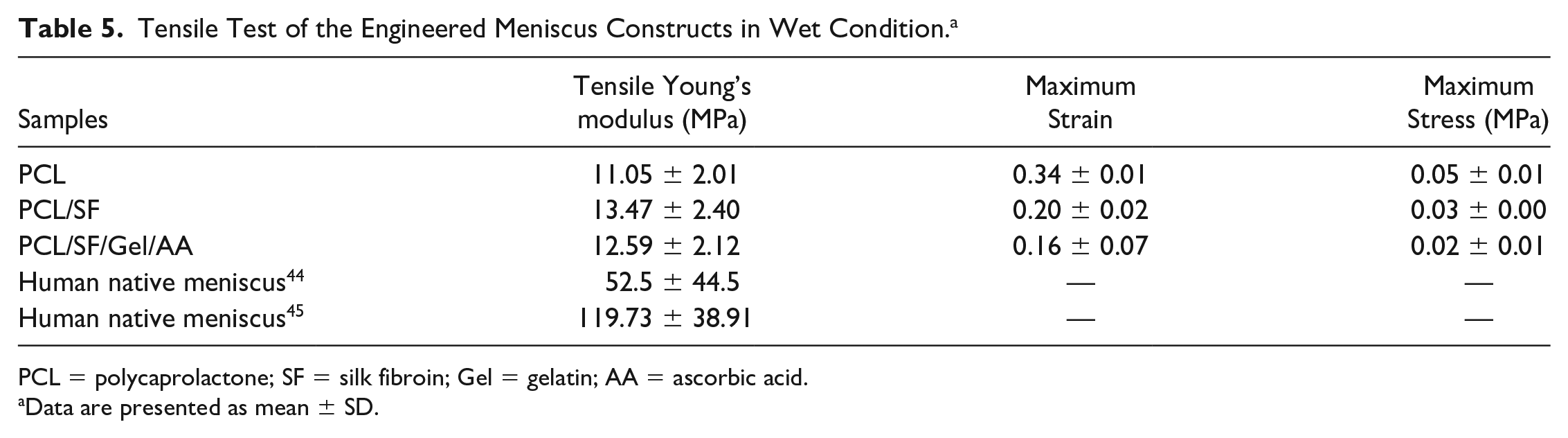
PCL = polycaprolactone; SF = silk fibroin; Gel = gelatin; AA = ascorbic acid.
Data are presented as mean ± SD.
The Compression Test of the Engineered Meniscus Constructs in Dry Condition. a

PCL = polycaprolactone; SF = silk fibroin; Gel = gelatin; AA = ascorbic acid.
Data are presented as mean ± SD.
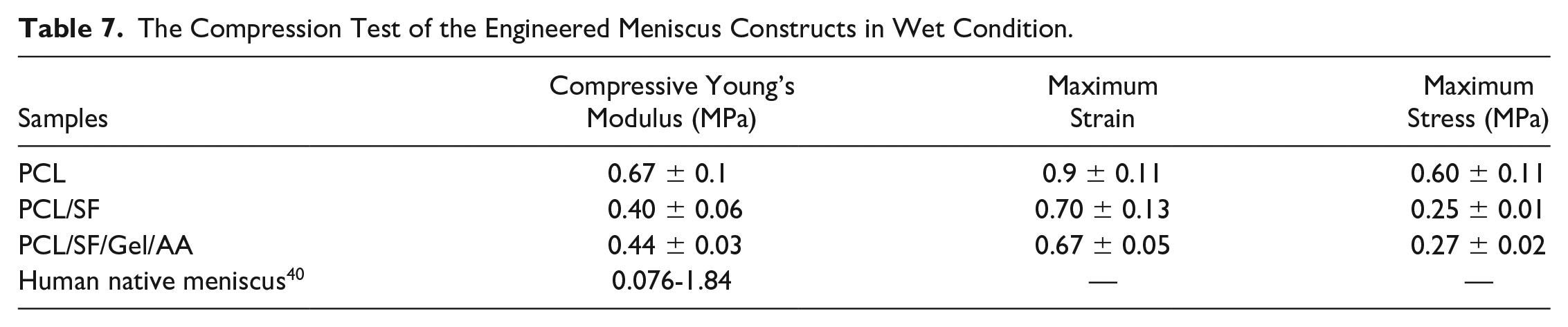
The Compression Test of the Engineered Meniscus Constructs in Wet Condition.
The result of the tensile test in dry condition showed that the elasticity of the PCL/SF/Gel/AA composite decreased significantly compared with other groups, while the stiffness in PCL/SF/Gel/AA blends experienced a significant rise. Thus, Young’s modulus in this blend increased significantly compared with other groups. In contrast, there was no significant difference in Young’s modulus between the groups in the wet condition. Furthermore, Young’s modulus in the PCL and PCL/SF scaffolds increased slightly compared with the dry state, but in the PCL/SF/Gel/AA scaffold, due to the presence of gelatin, water absorption was observed, and thus, Young’s modulus in this scaffold decreased significantly compared with dry condition and was almost similar to the PCL/SF scaffold.
The compression test results in dry conditions showed no significant difference in the maximum stress and Young’s modulus among the groups, while the maximum strain in PCL/SF/Gel/AA blends was significantly lower compared with the PCL scaffold. Therefore, in the dry condition, the application of gelatin biomacromolecule reduced the compressive strain in the PCL/SF/Gel/AA composite scaffold. In wet conditions, Young’s modulus and the maximum stress in the PCL/SF and PCL/SF/Gel/AA scaffold were significantly decreased compared with the PCL scaffold. Also, the maximum strain in the PCL/SF/Gel/AA scaffold was significantly reduced relative to the PCL scaffold.
In this study, a compression test was also performed for the bovine meniscus, in which the maximum stress, the maximum strain, and compressive modulus were 0.725 ± 0.015, 0.279 ± 0.030, and 2.95 ± 26.12 MPa, respectively.
Morphology of Cell-Loaded Scaffolds
In Figure 9 , which shows chondrocytes-loaded scaffolds on the sixth day, the expansion of cells on the surface of scaffolds can be observed, and cells fully covered the surface of the scaffolds such that chondrocytes on the PCL/SF and PCL/SF/Gel/AA composite scaffolds had high adhesion and expansion. This implies that these 2 scaffolds provided optimal conditions for cell growth and proliferation.
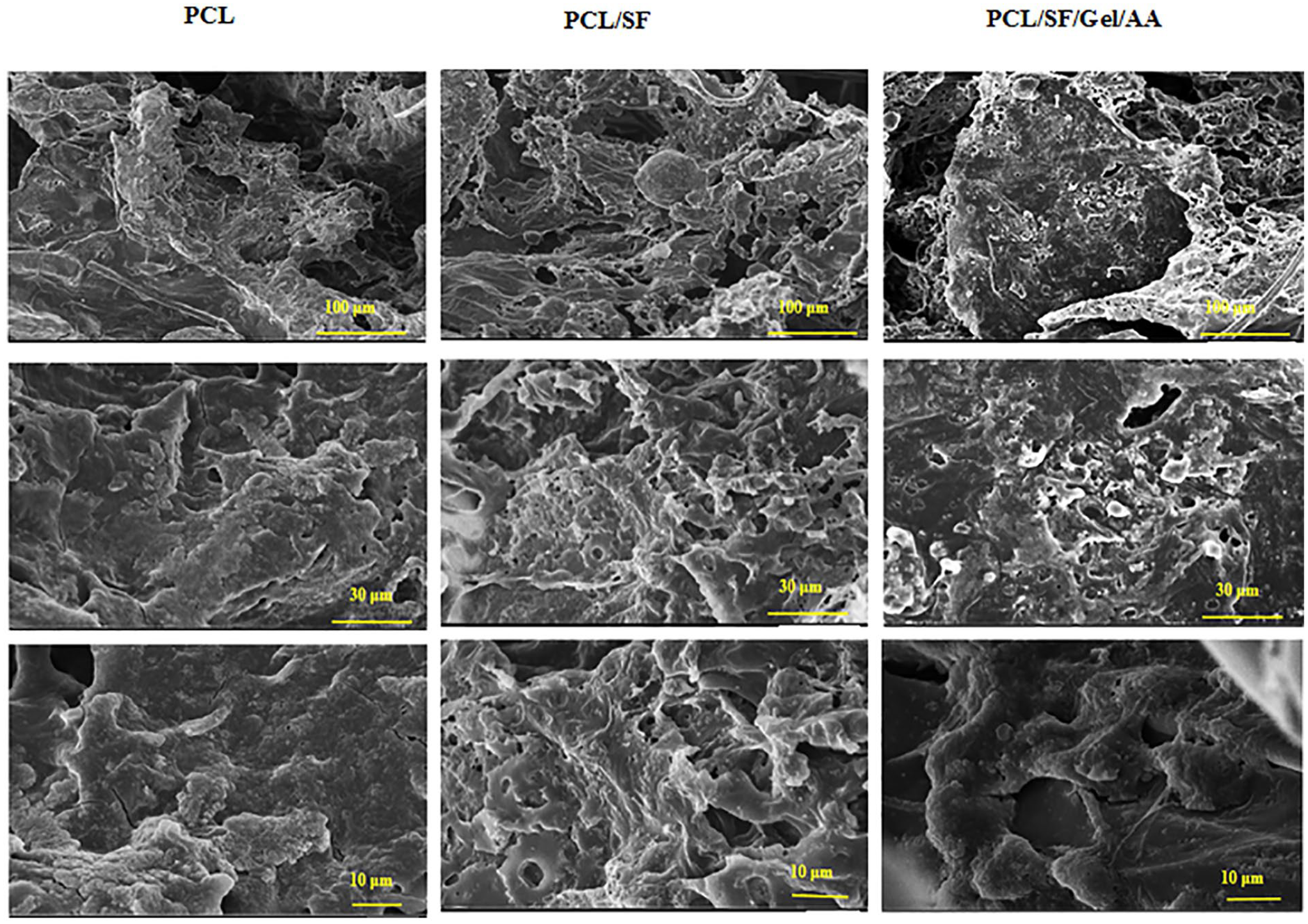
Scanning electron micrograph of chondrocytes-seeded scaffolds with magnifications of 100, 30, and 10 μm.
Determination of Cell Metabolic Activity and DAPI Staining
Any advances in meniscus tissue engineering depend on the development of scaffolds, cultured by essential cells, that display the meniscus tissue features. Thus, we assessed the influence of the scaffold on metabolic activity and viability of chondrocytes via indirect MTT test during 1, 4, and 6 days. The results of the indirect MTT assay illustrated that chondrocytes were viable and proliferated after 6 days ( Fig. 10 ). Therefore, our findings demonstrated that the tissue-engineered scaffolds had no toxicity and could be employed as a suitable substrate for the adhesion and growth of chondrocytes.
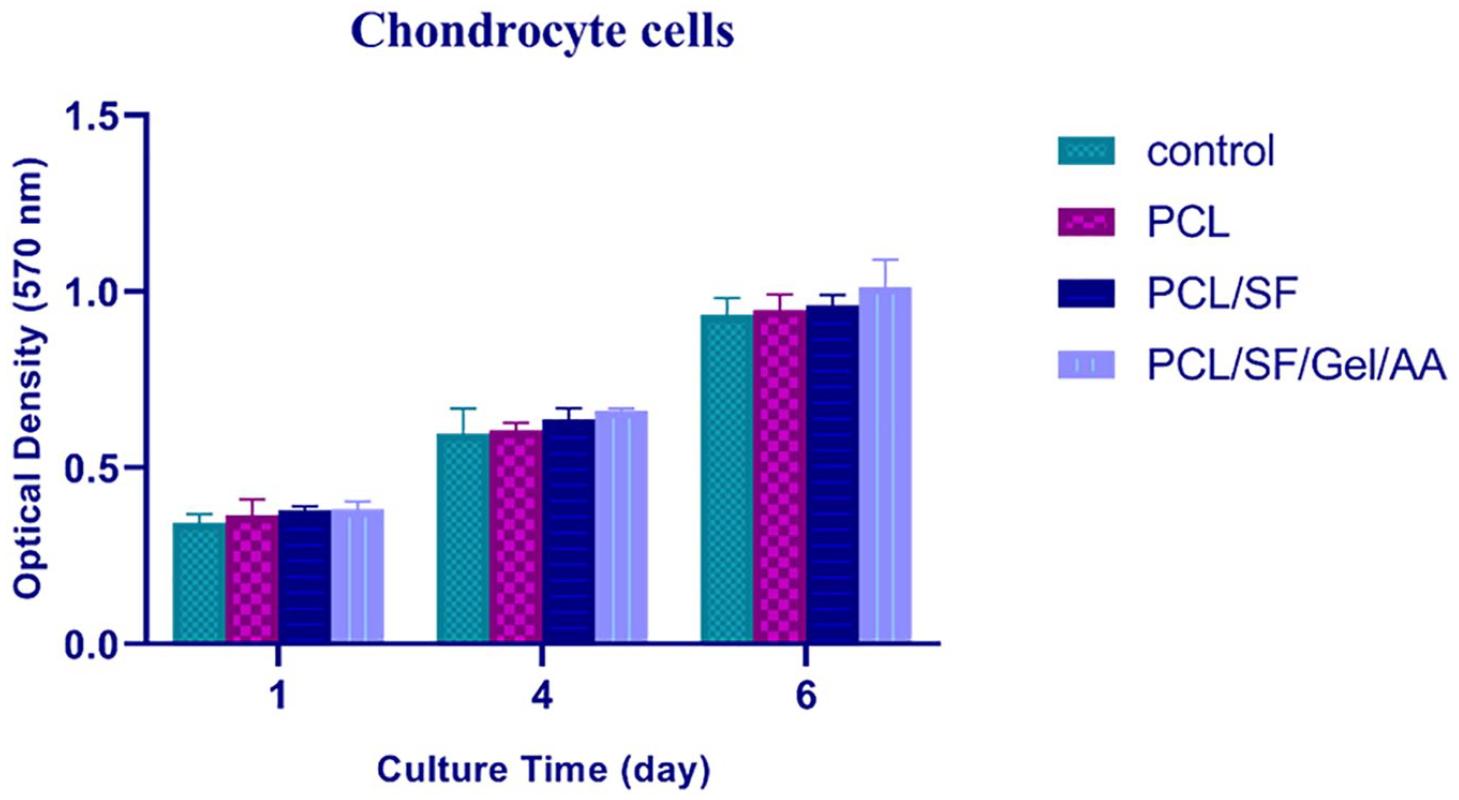
Indirect MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) test of the scaffolds, 1, 4, and 6 days after cell seeding. The results demonstrated that the scaffolds were biocompatible for chondrocytes (n = 3).
In addition, for a qualitative evaluation, the nucleus of chondrocytes was screened by DAPI staining. 41 In line with MTT results, DAPI staining verified that the cells firmly adhered to the plate surface, and therefore, the scaffolds were nontoxic ( Fig. 11 ). It also appeared that cell proliferation, compared with other groups, was higher in the PCL/SF/Gel/AA scaffold.
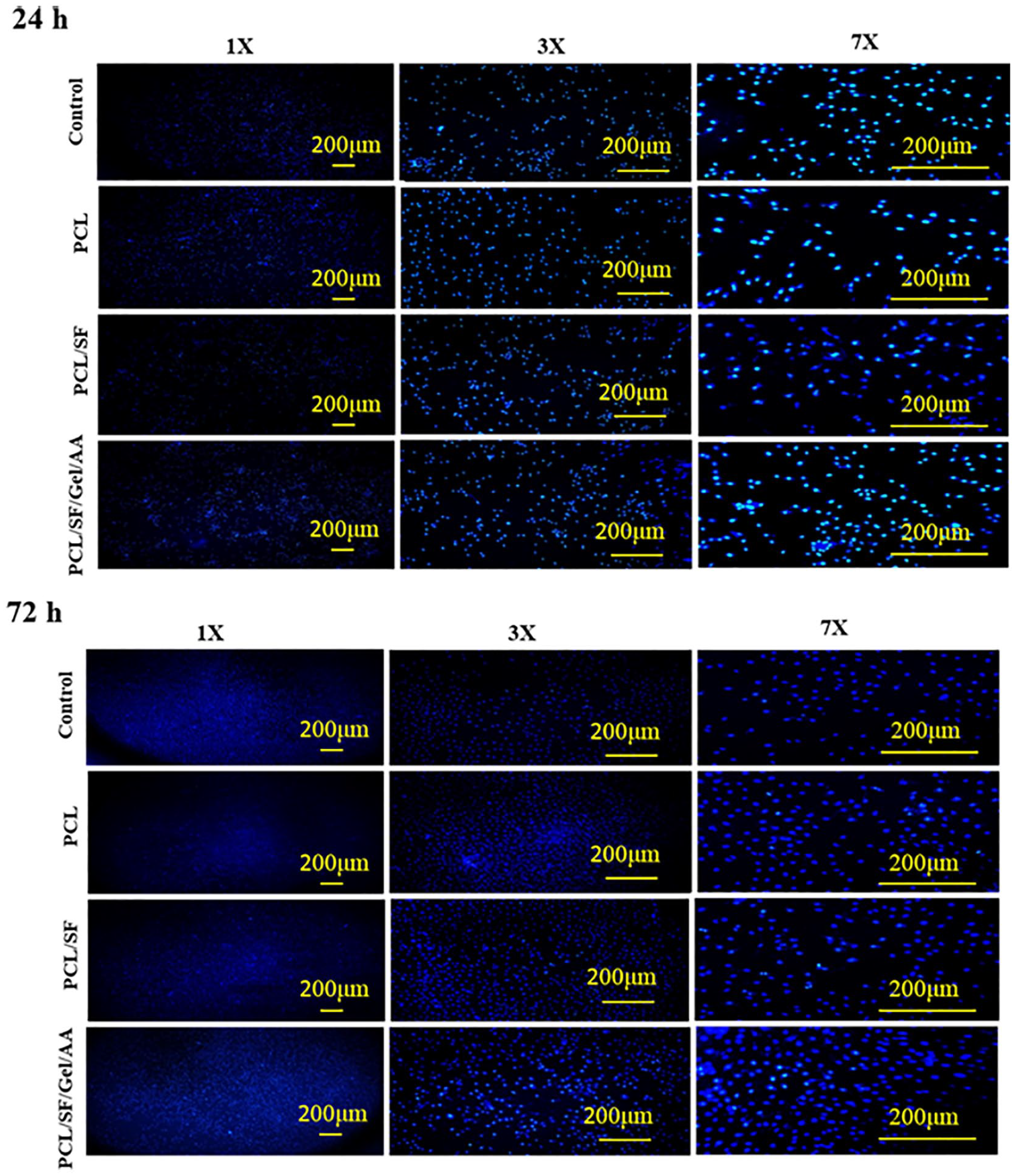
The illustration of chondrocytes 24 and 48 hours after cell seeding and staining by DAPI (4,6-diamidino-2-phenylindole). The results implied that chondrocytes proliferated properly, and thus, scaffold-degradation products were nontoxic for chondrocytes, 1 and 3 days after cell seeding.
Macroscopic and Histological Evaluations of New Meniscus Tissue
Macroscopically, PCL/SF/Gel/AA scaffolds formed new meniscus tissue, and no sign of infection, edema, and inflammation was observed. Also, the level of synovial fluid was appropriate. The new meniscus in the PCL/SF/Gel/AA scaffold revealed properties similar to those of the meniscus tissue, with histological configuration and ECM deposition approximately the same as the native meniscus. Hematoxylin and eosin staining displayed the presence of elongated cells like fibroblast, round-shaped cells like chondrocyte, and structures of mature lacunae in the neo-meniscus ECM tissue ( Fig. 12D , arrows). Also, the number of cells was higher in areas of new meniscus tissue, where reconstruction had not been completed yet. Masson’s trichrome staining ( Fig. 12 ) showed proper organization of type I collagen fibers in areas of new meniscus tissue, which had undergone a relatively complete reconstruction. Since the lesion was generated in the avascular area of the meniscus tissue, the considerable repair was not observed in the negative control sample during the study period. Outcomes of histological scoring, using a modified quantitative system of meniscus scoring, 38 presented a significant difference among the groups ( Fig. 13 ).
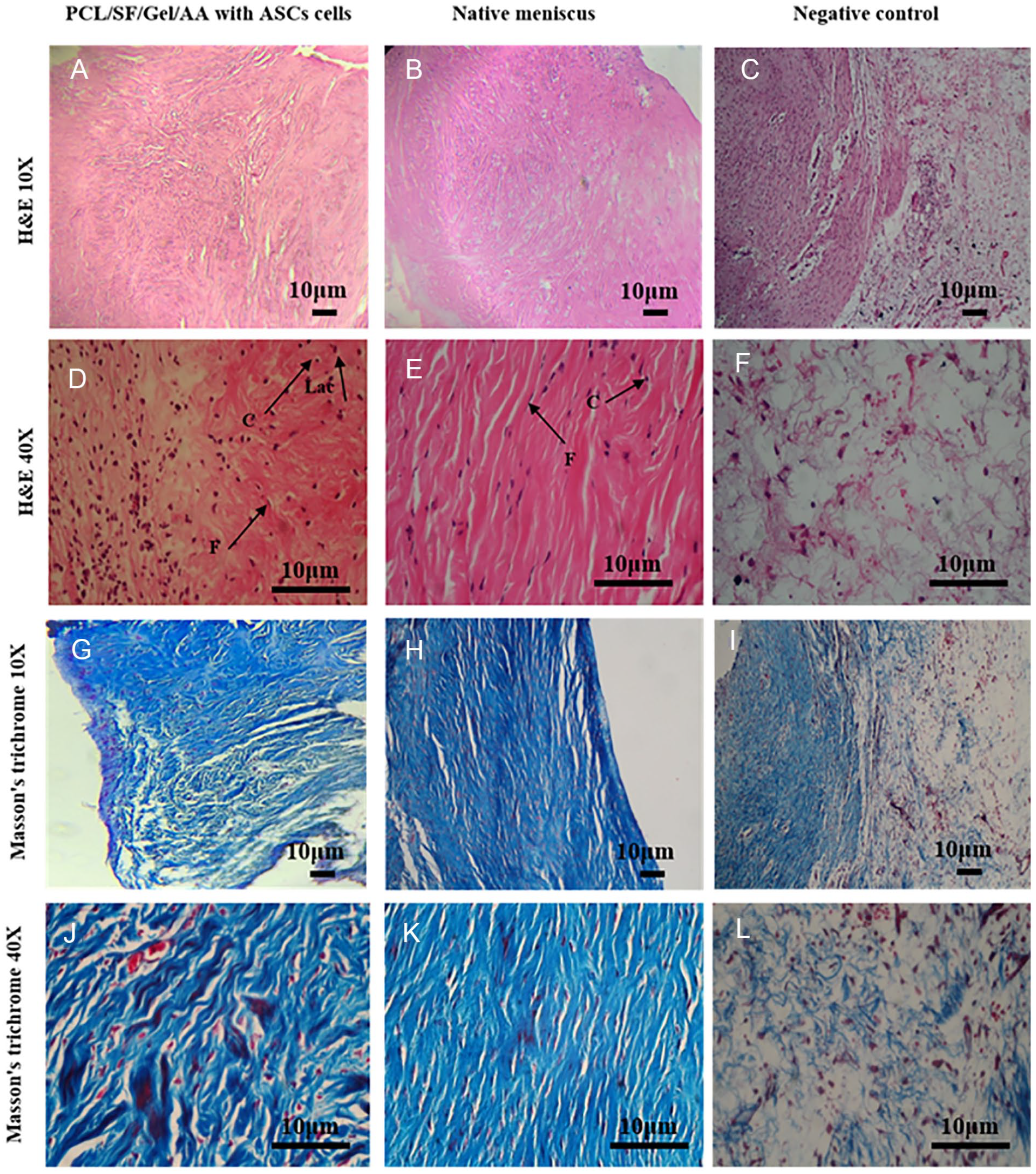
The repaired meniscus tissue after 2 months of transplantation in the negative control, native, and scaffold groups by hematoxylin and eosin (H&E) and Masson’s trichrome staining. H&E staining displayed the presence of elongated cells like fibroblast, and round-shaped cells like chondrocyte in the neo-meniscus extracellular matrix (ECM) tissue. Masson’s trichrome staining displayed type I collagen fibers. Lac, mature lacunae; C, chondrocytes-like cells; F, fibroblast-like cells.
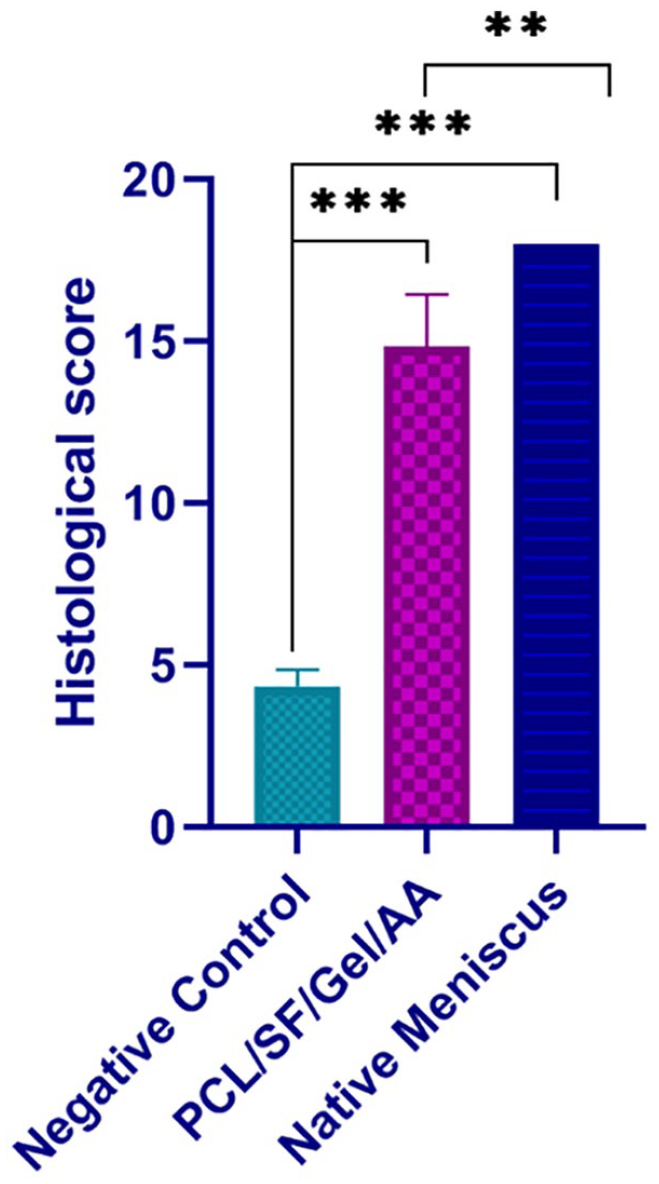
Histological scoring of the repaired meniscus after 2 months of transplantation into the punch defect. The modified meniscus scoring system was applied.
Discussion
Since the meniscus injuries in the avascular zone cannot be regenerated, it is crucial to provide constructs with appropriate function after partial and total meniscectomy. Currently, there are several options, including meniscal transplantation (for total meniscectomy), prosthesis substitutes, and recently, tissue engineering. Tissue engineering, due to its high similarity to the native tissue, avoiding injury and the donor limitation, being matched with the lesion site in size, and reduced infection risk is an optimal alternative. 8
A suitable scaffold for each tissue should provide optimal conditions for cellular behavior such as cell attachment, migration, proliferation, differentiation, organization, and also nutrient diffusion. Here, the results of SEM images showed that the scaffolds had interconnected pores, and therefore, were able to provide a suitable substrate for cells.
Results of the swelling test showed that supplementation of SF to the scaffolds could reduce the water absorption in the scaffold. Although SF is an amphiphilic polymer, its major component is composed of hydrophobic domains with a high molecular weight.42,43 Consequently, in this study, the presence of large hydrophobic domains associated with SF in the PCL/SF composite scaffolds decreased the swelling rate compared to the PCL scaffolds. However, the swelling rate of PCL/SF/Gel/AA hybrid constructs increased compared with the PCL/SF composite scaffolds, which was due to the presence of gelatin biomacromolecule. gelatin is a natural polymer with a high swelling rate. 39 Except for the peripheral area of the meniscus, other parts of it are avascular and feed through diffusion from synovial fluid and behave like a sponge by absorbing synovial fluid.21,22 Since gelatin biomacromolecules exhibit high water absorption, their presence in the composition of the PCL/SF/Gel/AA scaffolds was beneficial and made spongy constructs with higher similarity to the natural meniscus tissue.
Due to the presence of vitamin C in our proposed structure of the PCL/SF/Gel/AA scaffold, the highest antioxidant property was observed in this scaffold. Antioxidants such as vitamin C can prevent cell and tissue damages through their free radical scavenging property. These cellular and tissue damages may occur directly through oxidative degradation or via protease/antiprotease imbalances. Studies have shown that a variety of tissue inflammations are caused by reactive oxygen metabolites. These reactive oxygen metabolites may initiate and/or amplify inflammation through the upregulation of several genes involved in the inflammatory reaction, while antioxidants such as vitamins C and E may protect the cell against free radical-mediated inflammation and tissue damage. 44 Therefore, maintaining adequate antioxidant conditions can alleviate cellular damage and dysfunction, which is noticed in some inflammatory disorders as well. Moreover, meniscus damages in the inner avascular zone are in general not repaired spontaneously, which can lead to osteoarthritis (OA). 22 Although OA is known as a noninflammatory disease, studies have contributed inflammation to the symptoms and progression of OA, and proinflammatory cytokines are essential mediators of this disease.45,46 In addition, surgery and meniscus transplantation are associated with inflammation, and hence, the application of antioxidant compounds such as vitamin C in the scaffolds can be beneficial. The most abundant component of the meniscus extracellular matrix is collagen, and since vitamin C is essential for collagen synthesis, its application in the scaffold structure seems favorable. In the current research, the PCL/SF/Gel/AA scaffolds indicated an appropriate concentration of antioxidants that can provide a desirable condition for implantation in damaged meniscus areas.
Since the meniscus is a load-bearing structure, the mechanical aspects are crucial in meniscus tissue engineering. In this regard, tensile and compression tests were accomplished on the scaffolds in dry and wet conditions at room temperature. Results of the tensile test in dry conditions revealed that the tensile modulus in the PCL/SF/Gel/AA scaffold was significantly higher than PCL and PCL/SF scaffolds. This might be due to robust covalent bonds between polymer chains under mixing, which restrict the movement of the chains and consequently, lead to the rise of the tensile modulus in the PCL/SF/Gel/AA scaffold. 12 In the wet condition, there was no significant difference in Young’s modulus among the groups. However, the presence of gelatin in the PCL/SF/Gel/AA scaffold resulted in higher water absorption that, compared with dry condition, caused a significant decrease in Young’s modulus, which was almost similar to the PCL/SF scaffold.
In meniscus tissue engineering, the scaffold should possess suitable mechanical features to endure forces applied to the knee joint. In 2 separate studies, the values of 52.5 ± 44.5 and 119.73 ± 38.91 MPa were reported for the tensile modulus of the human native meniscus.47,48 Our obtained results showed that the average tensile modulus of the PCL/SF/Gel/AA composite construct in wet and dry conditions were 12.59 ± 2.12 and 218.30 ± 21.90 MPa, respectively.
Results of the compression test in the dry conditions indicated that the compressive modulus of the scaffolds was not considerable. However, the compressive modulus in the PCL/SF and PCL/SF/Gel/AA scaffolds was significantly decreased compared with the PCL scaffold. The water absorption by SF and gelatin biopolymers in wet conditions led to this reduction.
In the pictures obtained from DAPI staining, cell proliferation in the PCL/SF/Gel/AA scaffolds appeared to be higher compared with other groups. This is probably due to the effect of silk and gelatin. Protein and polysaccharide materials have desirable properties such as biocompatibility, biodegradability, and nontoxicity. 49 Integrin-binding sites exist in the configuration of gelatin protein (like RGD), thus increasing proliferation, adhesion, and cell migration. 20 Therefore, the further proliferation of cells in the PCL/SF/Gel/AA scaffold can be attributed to this issue.
Although since 1982 meniscectomy has been proposed as an alternative method to total meniscectomy, few studies have been conducted in this area, and it needs to be further investigated. Therefore, we evaluated the meniscus repair by tissue-engineered constructs after punch defect. The punch model lacks the intense mechanical challenges that occur in severe meniscus tears and injuries, however, it has been used as a method of creating a lesion in some studies.38,50,51 The punch model disrupts the integrity of the meniscus tissue, and thus, it increases the risk of meniscal damage from mechanical stresses applied to the knee joint. Moreover, it increases the risk of articular cartilage abrasion and OA.
Stem cells can differentiate into other cells in various tissues and play a role in tissue repair.52,53 In 2017, Toratani et al. 51 studied the effects of a 3D scaffold-free construct composed of allogeneic adipose-derived stem cells to regenerate a damaged meniscus after a punch defect in a rabbit model. The results showed the positive effects of this construct on tissue repair. 51 In 2018, Koch et al. 7 evaluated the impact of a polyurethane scaffold either loaded or unloaded with mesenchymal stromal cells to reconstruct meniscal lesions followed by a partial meniscectomy in a rabbit model. They showed that the repair process was desirable in both approaches, but the speed of repair was higher in the scaffold loaded with cells. 7 The results of another study by Zellner et al. 54 showed the positive effects of hyaluronan/collagen scaffold seeded with autologous mesenchymal stem cells, compared with a cell-free scaffold or platelet-rich plasma-loaded scaffold, in the repair of meniscal tear.
The application of composite scaffolds is an approach to overcome the limitations of a single polymer, which brings together desired properties of multiple polymers. Therefore, the most important finding of this research is to improve the physicochemical, mechanical, and biological properties in the PCL/SF/Gel/AA composite scaffold, which will lead to better healing of damaged tissue. Also, since collagen is the main component of the meniscus ECM, and AA is one of the essential factors for collagen synthesis, loading of AA in the scaffold structure can increase tissue repair. Collectively, these composites showed better in vitro and in vivo performance.
The limitations of this study are the lack of larger animal models (than rabbits), longer follow-up times, and broader histological studies such as immunohistochemistry and other histological staining methods such as Alcian blue and SO/FG to further investigate the restored ECM. In addition, due to limited funding, we could not perform biochemical and biomechanical stimuli using bioreactors to increase the rate of restoration.
Altogether, the current study showed that the fabricated PCL/SF/Gel/AA scaffolds seeded with ASCs were effective in successful meniscus healing in a rabbit that underwent unilateral punch defect and could reduce inflammation in the implantation site.
Conclusion
In this study, the scaffolds based on SF and gelatin biomacromolecules blended with PCL and AA were prepared and evaluated for meniscal repair in a rabbit model. The composite scaffolds exhibited improved mechanical and physicochemical properties and revealed appropriate chondrocyte scaffold interactions. This scaffold with allogeneic ASCs could properly improve meniscus healing in a rabbit model. Due to the high antioxidant concentration in the composite scaffolds, the inflammation was also reduced after implantation. In vivo analyses with longer tracking and time and bigger animal models are needed to verify the clinical setting of the 3D PCL/SF/Gel/AA composite constructs seeded with mesenchymal stem cells for meniscus tissue engineering.
Footnotes
Acknowledgments and Funding
The authors would like to express their appreciation to Dr. Abbas Shafiee (Diamantina Institute, Translational Research Institute of the University of Queensland) for his technical assistance and critical comments. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from Shahrekord University of Medical Sciences (Grant No.: 3151).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from the Ethics Committee of the Shahrekord University of Medical Sciences (IR.SKUMS.REC.1398.119).
