Abstract
Objective
There has been a debate as to the alignment of the collagen fibers. Using a hand lens, Sir William Hunter demonstrated that the collagen fibers ran perpendicular and later aspects were supported by Benninghoff. Despite these 2 historical studies, modern technology has conflicting data on the collagen alignment.
Design
Ten mature New Zealand rabbits were used to obtain 40 condyle specimens. The specimens were passed through ascending grades of alcohol, subjected to critical point drying (CPD), and viewed in the scanning electron microscope. Specimens revealed splits from the dehydration process. When observing the fibers exposed within the opening of the splits, parallel fibers were observed to run in a radial direction, normal to the surface of the articular cartilage, radiating from the deep zone and arcading as they approach the surface layer. After these observations, the same samples were mechanically fractured and damaged by scalpel.
Results
The splits in the articular surface created deep fissures, exposing parallel bundles of collagen fibers, radiating from the deep zone and arcading as they approach the surface layer. On higher magnification, individual fibers were observed to run parallel to one another, traversing radially toward the surface of the articular cartilage and arcading. Mechanical fracturing and scalpel damage induced on the same specimens with the splits showed randomly oriented fibers.
Conclusion
Collagen fiber orientation corroborates aspects of Hunter’s findings and compliments Benninghoff. Investigators must be aware of the limits of their processing and imaging techniques in order to interpret collagen fiber orientation in cartilage.
Introduction
Sir William Hunter published his classic text on the structure of articular cartilage using merely a hand lens and a critical eye. After identifying the smooth nature of the cartilage surface, he allowed “curiosity [to lead him] a step further” and investigated the fiber alignment along the length of the cartilage depth. 1 Following the careful boiling of the cartilage, Hunter grasped the base of the subchondral bone with a pair of tweezers and peeled the cartilage radially until the fragment reached the articulating surface. This allowed him to conclude that the collagen fibers ran perpendicular to the articular surface, with the possibility of horizontal fibers to prevent separation of the vertical fibers. Benninghoff 2 extended Hunter’s observation to indicate that the fibers run vertically near the surface and arcaded, to form the lamina splendens (surface layer), which would limit fibrillation within the cartilage construct. Despite these observations, researchers using transmission (TEM) and scanning electron microscopes (SEM), among other techniques, reported that the fibers were randomly organized.3-5 MacConaill 6 suggested that these fibers might form bundles running oblique to one another to the surface.
On reviewing the literature, it can be demonstrated that the researchers utilizing TEM and SEM technology neither reconciled their findings with those of Hunter or Benninghoff nor did they consider how Hunter came to his conclusion if the fibers were randomly oriented ( Figs. 1 and 2 ). One would suspect that, by his observations, Hunter would have been unable to pull long, uninterrupted strands of articular cartilage upward from the subchondral bone if these fibers were not radially oriented. It was not considered in the experimental design of previous TEM and SEM investigations4-6 that random fiber orientation could be inaccurate due to artifacts in tissue collection and preparation. Specifically, Hunziker et al., 5 using a scalpel to prepare tissue samples, concluded that fibers form bundles running toward the articular surface, with empty spaces in between. Hunziker et al. 5 also noted contrasting random fibers in some of the bundles and radial fibers in others. MacConaill 6 also used a scalpel to prepare tissue samples, though he came to the conclusion that collagen fibers form spirals around “cell columns,” with perpendicularly running fiber bundles connecting to these columns. Other investigations, utilizing optical polarization tractography 7 and ultrasound, 8 among other techniques, supported Hunter and Benninghoff’s findings. Table 1 provides an overview of the contributions by other researchers, in addition to their techniques used and findings. Hughes et al. 9 used polarized light microscopy and SEM to determine the parallel fiber alignment. Owen et al. 10 also utilized SEM, in addition to x-ray microscopy supporting that fibers ran perpendicular with regard to the articular surface. Yao et al. 7 used optical polarization tractography to come to the same conclusion. Inkinen et al. 8 examined tissue samples in an ultrasound, in addition to optical coherence tomography, polarized light microscopy, and Fourier transform infrared imaging, again predicting parallel fiber alignment. Other groups, such as Wilson et al. 11 and Men et al., 12 created algorithms and models to predict fiber alignment.
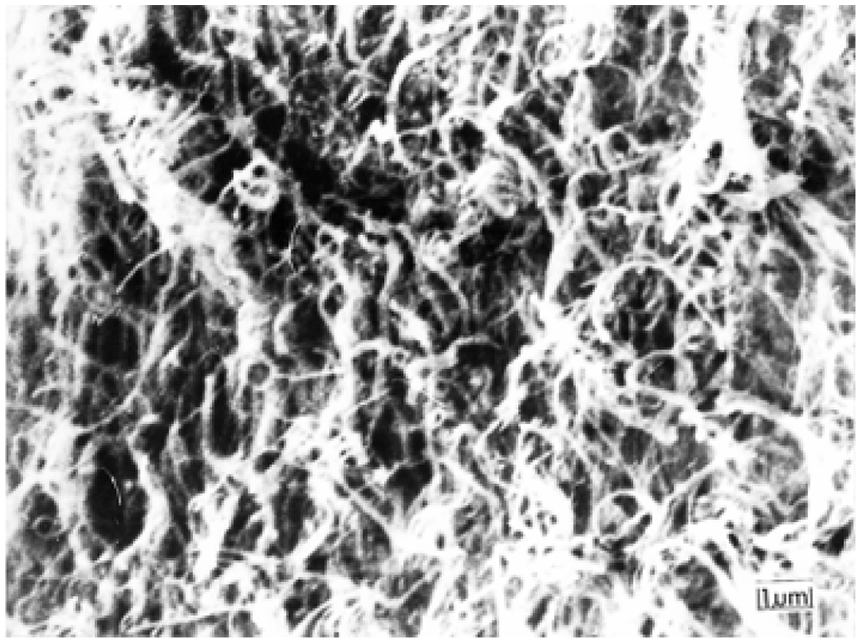
A high-powered view of the random fibers, as seen in the deep, middle/transversal, and superficial zone of the articular cartilage, created as a result of the mechanical fracture technique.

A high-powered view of the disrupted fibers traversing the 3 zones from scalpel damage. Empty lacunae can be seen.
A history of techniques, tissue preparation, and collagen fiber orientation in articular cartilage.
This investigation seeks to report an observation that allowed for the development of an experimental design that would confirm aspects of Hunter, Benninghoff, and those investigators who reported fibers running parallel to the surface, continuously traversing radially from the deep zone, to the middle zone, and to the superficial zone ( Table 1 ). An observation that allowed for serendipitous inspection of splits viewed on the surface of the articular cartilage led to the development of an experimental design that would confirm aspects of both Hunter’s and Benninghoff’s results supported under SEM imaging. During surface investigations of articular cartilage, splits were observed in specimens which clearly demonstrated that the fiber alignment was perpendicular, running normally with respect from the articular surface layer, parallel to the deep zone. This observation led to the design of an experimental protocol to test whether the preparation techniques used by various investigators may have caused artifacts, leading to conflicting results from those reported by Benninghoff and Hunter. The hypothesis tested was that the fiber alignment of the same specimens demonstrating spontaneous splits ( Figs. 3 and 4 ) would have been adversely affected by mechanical fracturing of the cartilage ( Figs. 1 and 5 ), in addition to scalpel damage during preparation ( Fig. 6 ).
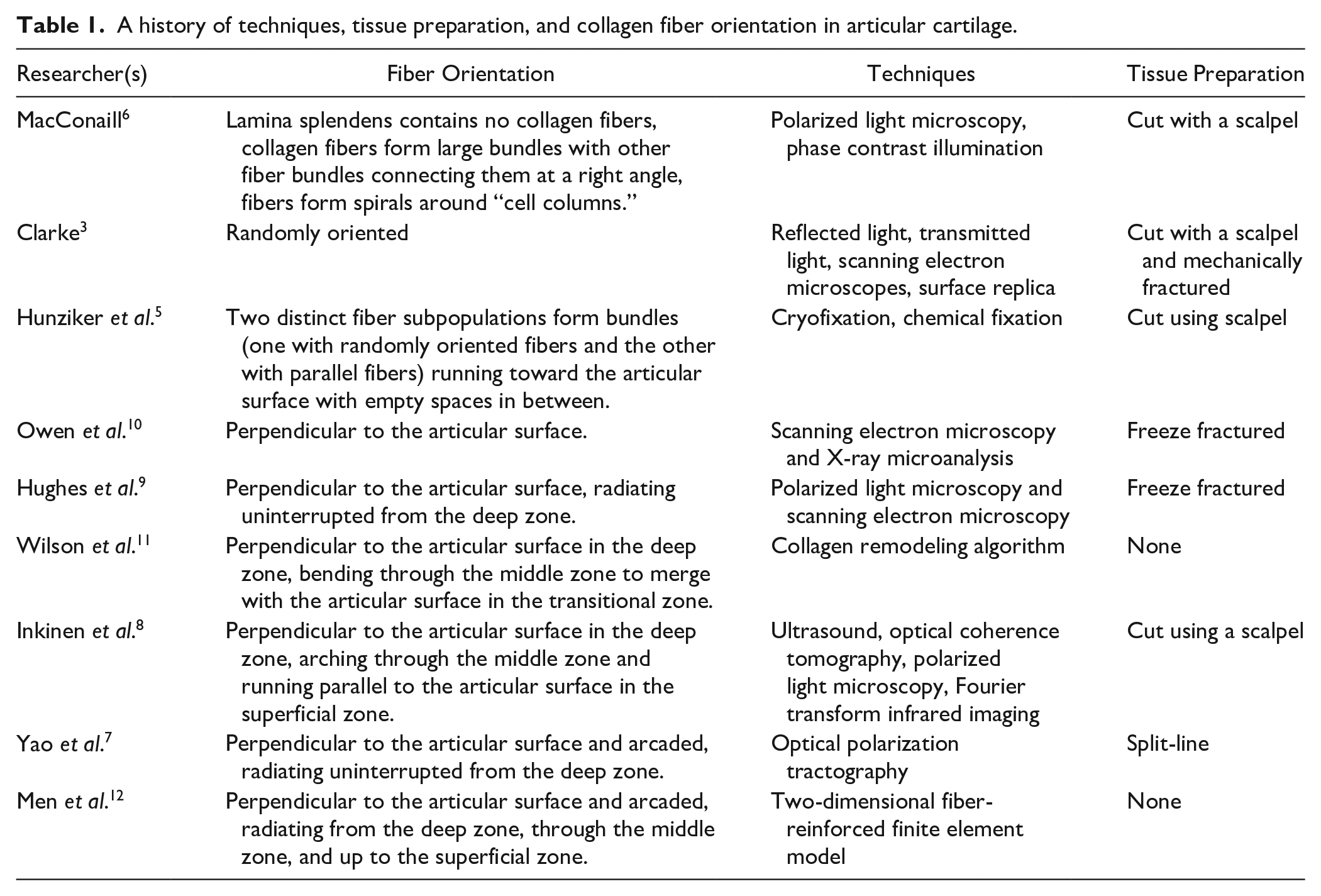
The steps taken to collect specimens with spontaneously occurring splits observed following tissue processing, due to shrinkage.
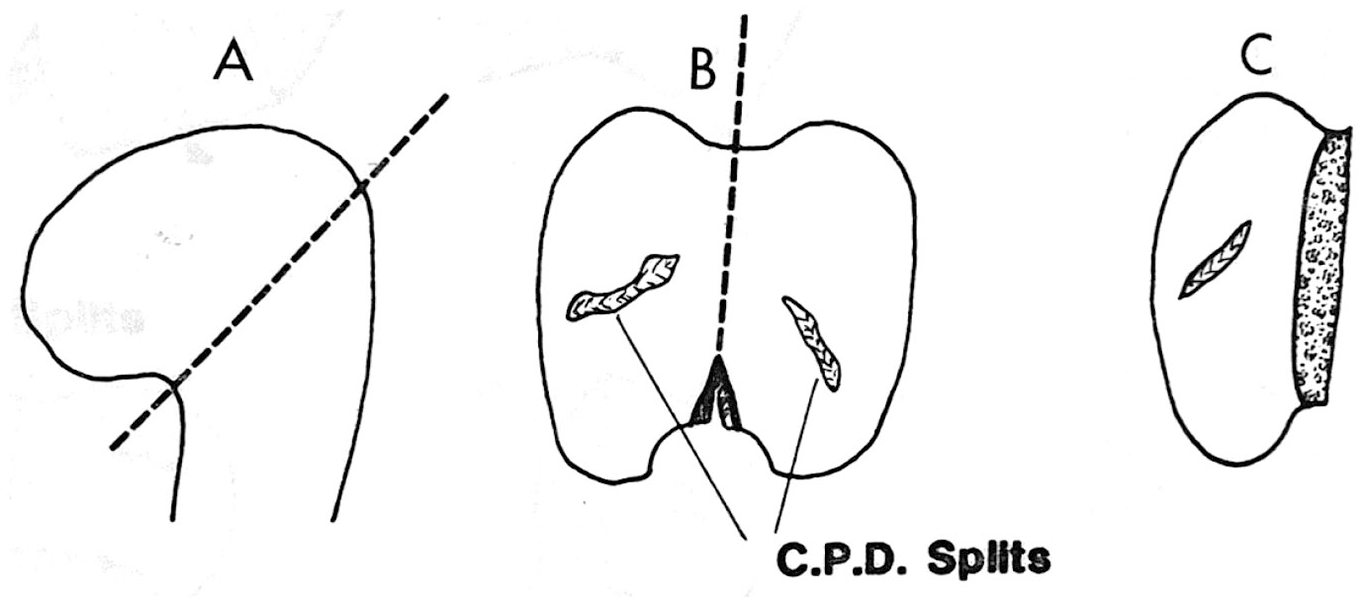
Numerous bundles with parallel fibers traversing from the deep and middle zones of the cartilage to the surface zone. This figure demonstrates the irregularly shaped splits that formed as a result of shrinkage. One could imagine that if the split region were reassembled there would be no gaps between the bundles.
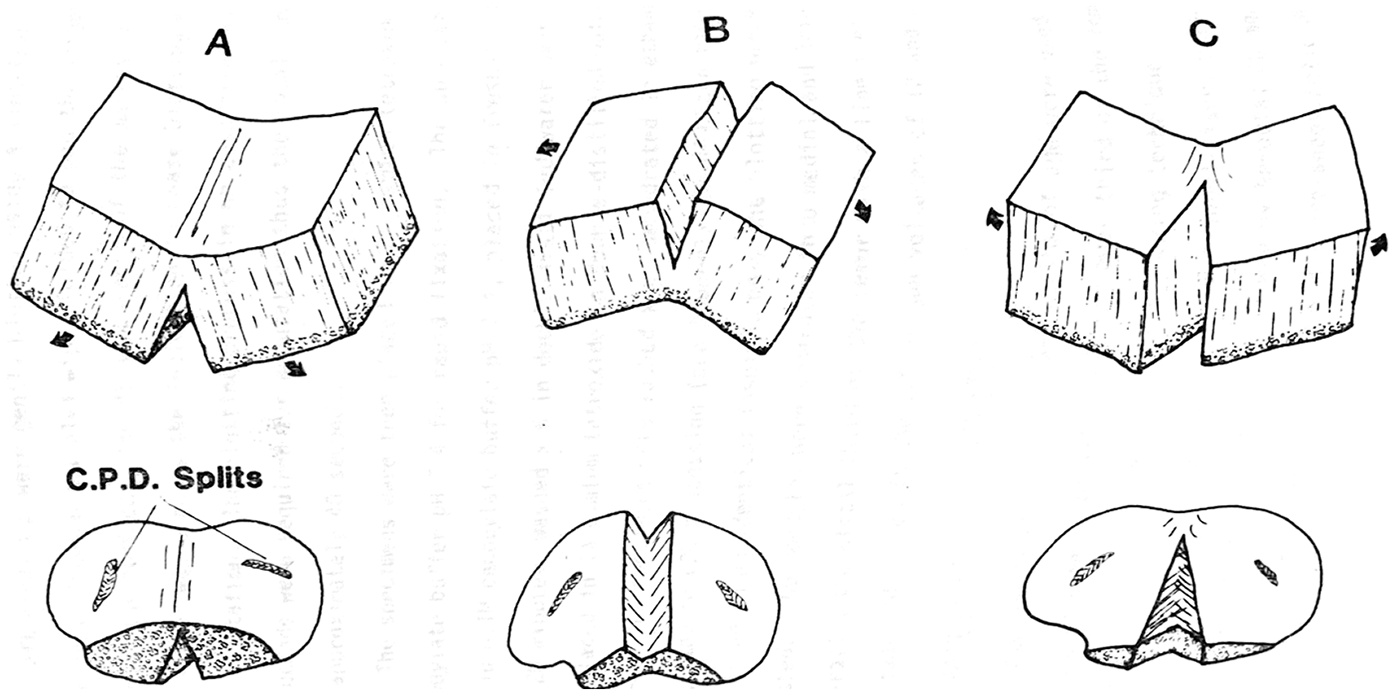
A diagram of the fracture technique, following Clarke’s 1971 protocol. 3 The specimens were prepared to retain the calcified zone and remnants of the subchondral bone to assure that the 3 zones (deep, middle, and superficial) were preserved.

A low-powered view of the scalpel damage. This damage was incomplete in this specimen, allowing the remainder of the tissue to split open, exposing the deep zone parallel fiber bundles, undamaged by the blade. The articular surface (AS), the scalpel region of damage (EX) shows the pattern of the edge of the scalpel blade, and the split edge (CP).
Methods
While performing experiments on the articular surface of rabbit articular cartilage, it was observed that random regions in the distal condyles of the adult rabbit cartilage had surface splits revealing parallel aligned fibers traversing from the calcified zone to near the surface where the fibers arched to entangle into the surface lamina ( Fig. 4 ). This observation allowed the development of the hypothesis and the experimental design below.
After receiving animal utilization permission, 10 mature adult New Zealand rabbits, 5 to 6 months of age were used to obtain 20 specimens each consisting of the lower third of the femur along with the attached condyles by the following technique ( Fig. 3 ).
A preoperative dose of Promex 2 tranquilizer, 0.25 mL/4.5 kbw was administered intravenously followed by Nembutal 60 mg/mL using 40 mg/kg within 15 to 20 minutes after the preanesthetic drug. This proved to be the best technique for deep anesthesia, avoiding pain and overdose. The condyles and distal third of the shaft were removed ( Fig. 3 ) and immediately immersed in 2.5% glutaraldehyde in 0.1 M cacodylate buffer adjusted to pH 7.4 for 7 days. The specimens were then washed in 0.1 M cacodylate buffer with 3 changes over a period of 30 minutes, and then processed in the OTOTO (osmium tetroxide and thiocarbohydrazide) method described by Bloebaum and Wilson 13 and Bloebaum and Radley. 14
The specimens were passed through ascending grades of alcohol 70% to 100% over a period of 1 to 2 weeks and subjected to critical point drying (CPD). On removing the specimen from the CPD apparatus, the femoral shaft was separated from the condyle close to the articular margin with a fine fret saw, assuring that the calcified zone and a remnant of the subchondral bone remained intact ( Fig. 3 ). This resulted in 40 specimens for inspection under the dissecting microscope, to assess for the presence of surface splits.
Twenty-five specimens of the 40 condyles revealed surface splits, a 62% yield. The condyles with the splits were then mounted on studs with silver conducting paint and viewed in the Phillips 501 SEM at 12 and 25 kV. Fibers were directed predominantly in a radial direction, normal to the surface ( Fig. 4 ) in all 25 cartilage specimens with the surface splits before performing the mechanical and scalpel experimentation portion on the specimens with the surface splits.
The specimens were then removed from their studs following SEM observation and processed in the following manner. The subchondral bone was partially undercut to reduce the mechanical rupturing force required for the fracturing by hand. Force was applied in 1 of the 3 directions shown ( Fig. 5 ) in accordance with Clarke. 15 Nine of the 25 specimens were fractured by the method shown in ( Fig. 5a ), and 8 each by the methods shown in Fig. 5b and c . This resulted in 50 mechanically fractured surfaces for cross-sectional SEM examination of the collagen fiber perpendicular to the complete cross-section of the cartilage fiber complex. All observances were then made in the SEM at 55 to 14,000 times magnification.
To test the effects of scalpel damage replicating TEM sampling methods during specimen collection and preparation, scalpel cuts were made with new No. 15 scalpel blades through the depth of the cartilage deep into the subchondral bone thereby penetrating the complete thickness of articular cartilage in specimens with previously observed splits with known parallel fibers radiating from the deep zone, uninterrupted to the superficial zone ( Fig. 6 ). For purposes of comparison with scalpel cuts before and after tissue processing, scalpel cuts were also made into condyles from five rabbits, attempting to penetrate all three zones (deep, middle, and superficial), prior to fixation at the time of surgery and into 5 condyles following OTOTO tissue processing as mentioned above. Specimens were remounted on studs and viewed in the SEM.
Results
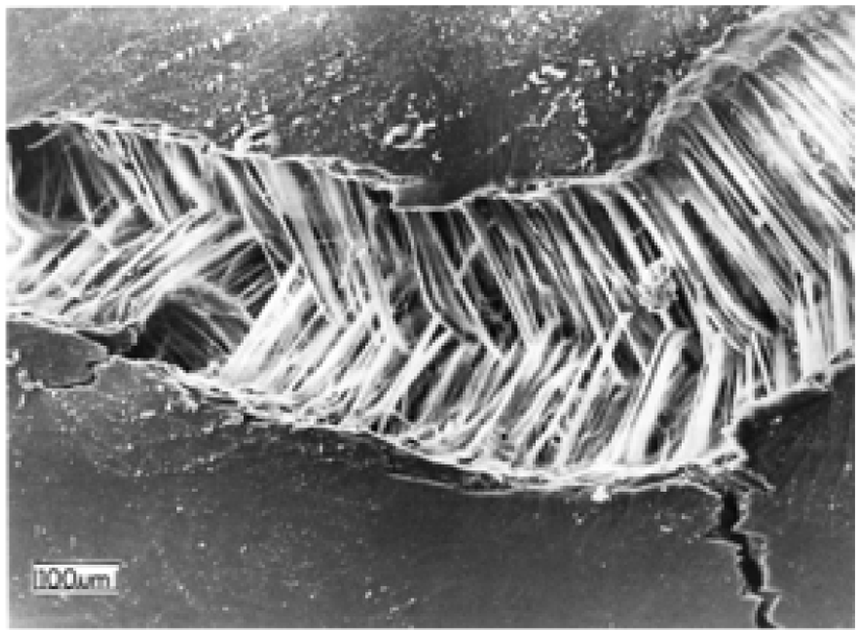
The splits produced random lesions within the 25 specimens creating deep fissures exposing parallel bundles of fibers ( Fig. 4 ). The lesions measured 0.479 to 1.53 mm2. On higher magnification, the individual fibers (0.1-0.5 µm) were observed to run approximately parallel to each other traversing radially from the deep and middle zones toward the surface layer, arcading near the surface ( Figs. 4 , 7 , and 8 ).

A high-powered view of the continuous parallel fibers communicating to the surface.
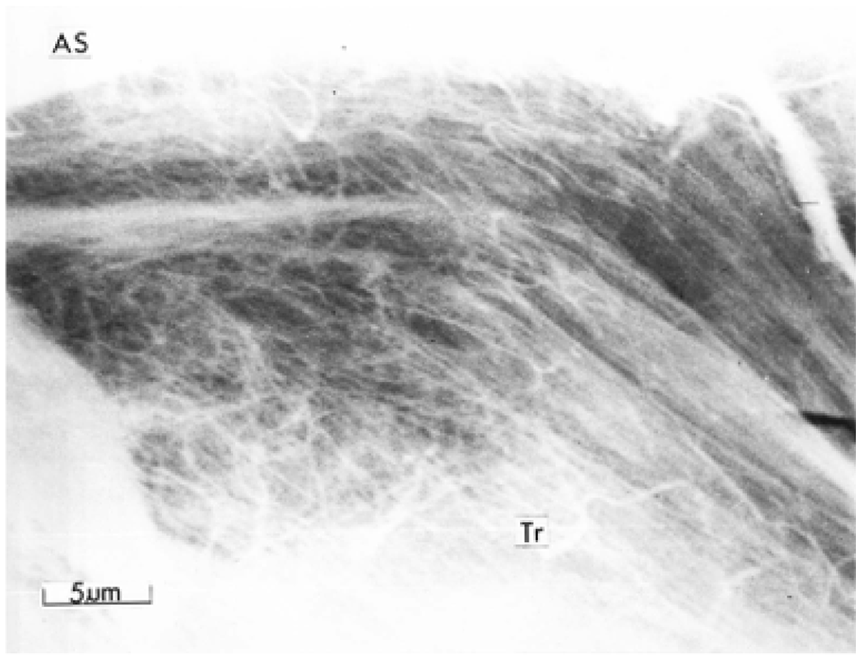
Arcading parallel fibers (Tr) bending toward the articular surface (AS).
Mechanical Fracture Results
The mechanical fractured en face surfaces previously observed showed randomly oriented fibers (0.1-0.5 μm) in the same condyle with the pre-established surface splits ( Figs. 4 and 9 ). However, larger fiber bundles consisting of random fibers could still be observed traversing radially to the surface ( Figs. 8 and 9 ). Careful observations suggested that fibers running parallel to one another were present in the background of the superficial randomized fibers at the forefront of the images observed in the mechanical fracture specimens. En face regions near the surface layer demonstrated mechanically fractured surfaces with fiber bundles arcading near the surface. ( Fig. 10 ).
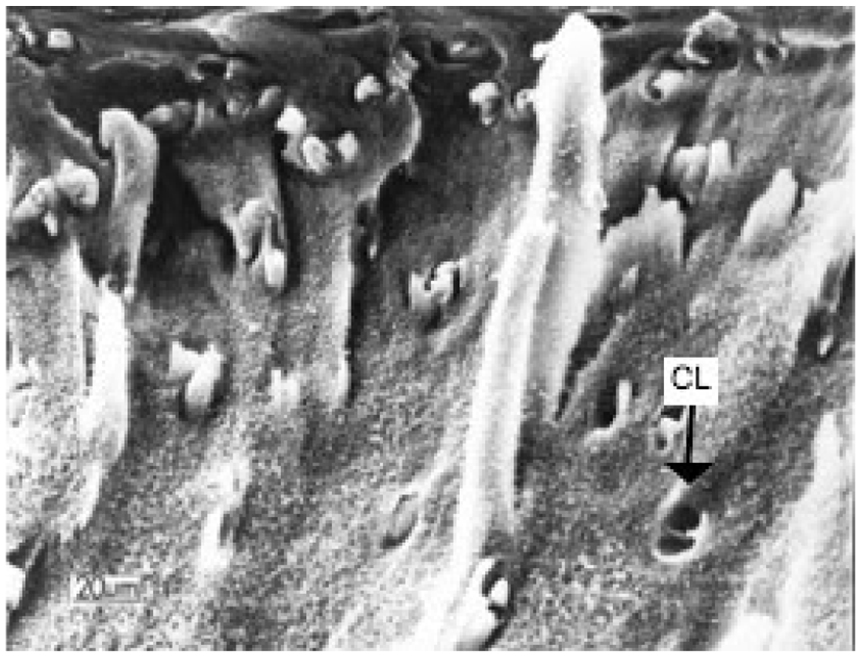
A mechanically fractured surface, revealing random fibers and bundles along the depth of the articular cartilage structure from deep zone, middle zone, and superficial zone to the surface. Chondrocyte lacunae (CL) can be observed.
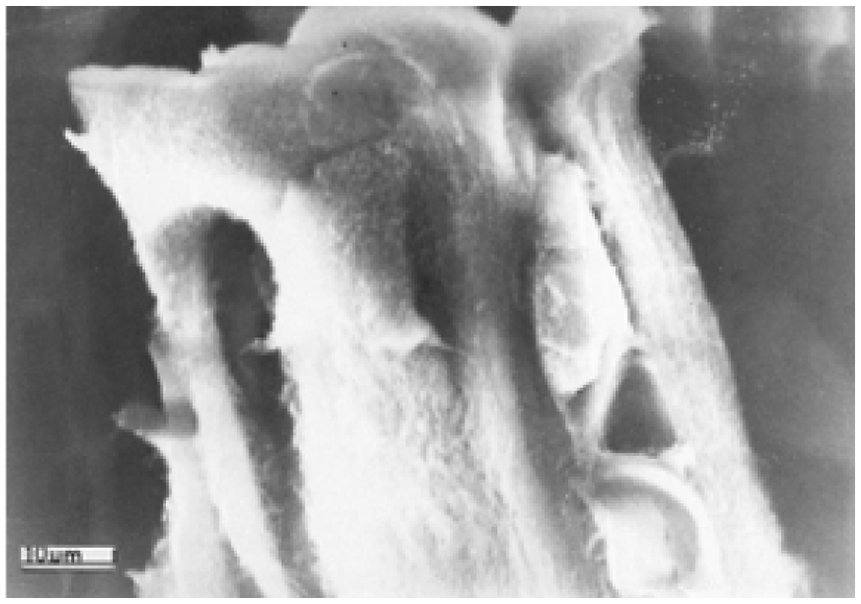
Arcading fiber bundles observed in the mechanically fractured specimen, near the surface layer.
Scalpel Technique
The scalpel damage created the most artifacts compared to mechanical fracturing and split features ( Fig. 11 ). The striation in the damaged cartilage from the scalpel edge was clearly present ( Figs. 6 and 11 ). The fiber bundle effect was completely obscured and only randomized fibers (0.1-0.5 μm in diameter) were observed throughout the 3 zones. In 4 of the specimens where the scalpel damage was not completely through the depth of the cartilage, a combination of the exposed deeper fiber bundles with parallel fibers could be seen, in contrast to bundles with random fibers from the scalpel damage ( Fig. 6 ). These observations were consistent whether the scalpel damage was performed on the specimens with scalpel damage at the time of operation or after tissue processing.
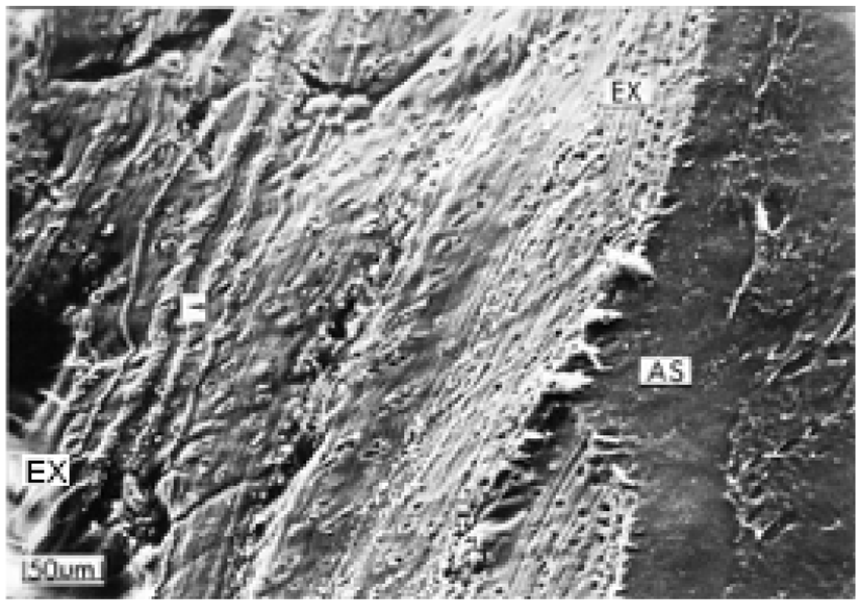
A low-powered view of scalpel damage showing striation pattern from the scalpel blade edge (EX) traversing the 3 zones. The articular surface is represented by AS.
Arcading Observations
Observations were made on both of the split and mechanically fractured specimens revealing the arcading of the fiber bundles forming the surface lamina ( Figs. 8 and 10 ).
Discussion
There is no doubt that, regardless of the technique utilized, artifacts will be produced during tissue removal and processing. As such, it remains the role of the investigator’s interpretation to be critical in assessing what may actually be the in vivo structure of a tissue, based on potential artifacts induced during specimen collection and preparation.
If investigators are to suggest that either the collagen network in articular cartilage were either random or a combination of random and perpendicularly aligned or oblique, then a critical argument must be made to explain how Hunter was able to pull continuous strands of articular cartilage from the subchondral bone surface without mechanical interruption. Conversely, one can ask how Hunter was able to track the parallel pattern at all, if the fibers were anything but parallel in alignment. The results from this study, caused by the surface splits, support Hunter’s dissection observations 1 and Benninghoff’s polarized light investigation. 2
This experimental design demonstrated how mechanical fracture and scalpel damage could have created the artifact of random fiber organization. This random pattern was due to damage from these sampling methods. It may be that the nature of the comparatively intact larger tissue samples of the rabbit condyles in this study, allowed the surface splitting to occur during processing, compared to other samples (i.e., from mice, rat, or human samples) with an interrupted surface lamina compared with the mechanical or scalpel damage. The split artifact observed in this experiment was likely the result of pure tensile forces on the intact surface of the cartilage, causing the mechanical breakdown of the surface lamina, and thereby exposing the radial fibers aligned in bundles. It may be that even the bundles observed are an artifact of dehydration during processing and mechanical forces.
Numerical forces of tensile stress on tissue evaporation have been estimated anywhere from 5.84 MPa to 4.68 GPa. 16 This would help explain the split artifact created during processing. It would be difficult to imagine how mechanically fracturing the articular cartilage specimen by hand could create pure tensile stresses initially on the surface lamina and underlying fibers, allowing the radial pattern to be exposed in all three zones. 15 The experimental results in this study support this hypothesis.
The surface lamina and the arcading network of fibers would also be of significant mechanical importance in keeping the radial fibers under tension during loading. One could imagine when a load was placed on the articular surface, that the articular surface would become displaced, causing the surface and the underlying parallel fiber structure to be under tensile forces. This could act in concert within the swelling pressure in articular cartilage and high water content, to resist loads and mechanical failure in healthy articular cartilage.
Recent studies, utilizing modern technology, have supported Benninghoff’s observations and aspects of Sir William Hunter’s results. Hughes et al. 9 used polarized light microscopy and SEM to determine the fiber orientation. In their study, specimens were subjected to critical point drying and freeze fracturing before being observed. 9 Similarly, Owen et al. 10 also used critical point drying and freeze fracturing to prepare specimens for SEM and X-ray microanalysis observations. Freeze fractures allowed the internal layout of specimens to be preserved before analysis, though the act of freezing the specimen might create unintended artifacts such as fibers that were pulled from the tissue or broken. Another study, utilizing optical polarization tractography to determine fiber orientation, used split-line techniques to prepare samples for observation. 7 Split-line techniques allow researchers to understand the orientation of the collagen fibers in the surface layer without having to mechanically fracture or use scalpels to disturb the underlying fiber orientation in specimens, thus limiting potential artifacts. On the other hand, the insertion of ink into samples using a needle could, in of itself, create a different artifact by altering the structure of the cartilage.
Inkinen et al. 8 utilized scalpel techniques to prepare tissue samples before examining with ultrasound. Their group examined samples using optical coherence tomography, polarized light microscopy, and Fourier transform infrared imaging. 8 Using a scalpel to prepare tissue samples likely created artifacts vis-a-vis disrupting fiber orientation. This was likely countered through the operation of an ultrasound, which grants a view of the fibers underlying the scalpel damaged fibers, allowing the group to avoid the artifact created by the scalpel. This method is not without concerns, though, as ultrasound could potentially disrupt the morphology of the articular cartilage, depending on the frequency and power used.
Wilson et al. 11 avoided the possibility of artifacts via the creation of a collagen remodeling algorithm. This algorithm analyzed fiber strains to predict fiber orientation. 11 Although this group did not experiment on actual cartilage samples, the predictions of the algorithm corroborate other researchers’ findings, including those of this study. A similar study conducted by Men et al. 12 explains why cracks in cartilage spread preferentially along the tangent direction of the fibers, using a 2-dimensional fiber-reinforced finite element model, which explains the splits observed in this experiment.
In the past, studies have proposed other potential fiber orientations. For example, Hunziker et al. 5 proposed in their 1997 study reported that two distinct subpopulations of fibers form bundles running toward the articular surface, with empty spaces in between. In one of these subpopulations, fibers were randomly oriented, whereas the other subpopulation consisted of parallel fibers. In Hunziker et al.’s study, 5 researchers utilized cryofixation and chemical fixation before cutting the specimens with a scalpel for observation. The scalpel damage incurred by this investigation explains differences in observations made by Hunziker et al. Hunziker et al.’s research also failed to explain how fiber bundles integrated with the articular surface. The act of cutting the samples with a scalpel would have caused grooves and disrupted the orientation of the collagen fibers, thus allowing for the suggestion of gaps between bundles. Additionally, empty spaces between fiber bundles would have caused subsurface fissures in the articular surface, compromising the ability to resist stress. Hunziker et al.’s observations are understandable, as Figure 4 displays empty spaces between fibers caused by scalpel damage. If one imagines that the “split puzzle” observed were reassembled, there would be no gaps or empty spaces between fibers. As the split forms, fibers are pulled apart from one another, leaving empty spaces where these fibers previously were attached. This would lead to the appearance of gaps between fiber bundles.
In 1971, Clarke proposed that the collagen fibers arcaded, supporting Benninghoff as well as the findings of this study, among others.2,7-10 Researchers utilized reflected light, transmission light, and scanning electron microscopes to observe the fiber structure. 3 Similar to Hunziker et al.’s investigation, the use of a scalpel in preparing specimens created artifacts, which give the façade of random fiber orientation. Additionally, the act of mechanically fracturing specimens would have created additional artifacts by further disrupting fiber orientation. Clarke was correct in his observation that fibers arcaded toward the surface layer. This finding is consistent with the observations of this study, Benninghoff, and recent literature.7-10
Finally, MacConaill 6 proposed that the surface layer contained no collagen fiber. MacConaill also proposed that collagen fibers form spirals around “cell columns,” with perpendicularly running fiber bundles connecting to these columns. These observations were made through the utilization of polarized light microscopy and phase contrast illumination. 6 Similar to previous studies, the usage of a scalpel would have resulted in artifacts, leading to inaccurate findings. The fact that specimens had to be viewed from various angles left room for error, as it would be impossible to account for fibers that were already seen. Furthermore, MacConaill’s study, in its assumption that the surface layer contains no collagen fibers, fails to acknowledge the method in which the fibers would connect and interact with the articular surface.
Investigators must be aware of the artifact that arises in tissue preparation and collection when investigating collagen fiber orientation in articular cartilage. In conclusion, the results of this study generally corroborate those of Sir William Hunter 1 and specifically support Benninghoff’s findings, 2 as well as those of others.7-10 In general, the experimental data from this SEM study supports their observations on collagen fiber orientation of radial fibers running normal to the surface from the deep zone and middle zone, to the surface layer, arcading to form the “lamina splendens.”
Footnotes
Authors’ Note
This research was conducted at the Department of Anatomy, School of Human Sciences, University of Western Australia.
Author Contributions
RDB developed the experimental design and performed all the imaging. ASW helped with data interpretation and manuscript interpretation. WNM was essential for manuscript interpretation and data analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Department of Anatomy, School of Human Sciences, The University of Western Australia.
We would like to thank Dr. Joseph Martin for his insight regarding the usage of ultrasound.
Dr. Bloebaum would like to thank Lois and his children for their love during his absence while conducting this research.
Ethical Approval
Ethical approval for this study was obtained from the Animal Utilization Committee at the Department of Anatomy at the University of Western Australia.
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
