Abstract
Objective
Articular cartilage exists in a hypoxic environment, which motivates the use of hypoxia-simulating chemical agents to improve matrix production in cartilage tissue engineering. The aim of this study was to investigate whether dimethyloxalylglycine (DMOG), a HIF-1α stabilizer, would improve matrix production in 3-dimensional (3D) porcine synovial-derived mesenchymal stem cell (SYN-MSC) co-culture with chondrocytes.
Design
Pellet cultures and scaffold-based engineered cartilage were grown
Results
The addition of DMOG resulted in HIF-1α stabilization in the exterior of the engineered constructs, which resulted in increased regional type II collagen deposition, but the stabilization did not translate to overall increased extracellular matrix deposition. There was no increase in HIF-1α stabilization in the pellet cultures. DMOG treatment also negatively affected the mechanical competency of the engineered cartilage.
Conclusions
Despite previous studies that demonstrated the efficacy of DMOG, here, short-term treatment with DMOG did not have a uniformly positive impact on the chondrogenic capacity of SYN-MSCs in either pellet culture or in scaffold-based engineered cartilage, as evidenced by reduced matrix production. Such 3D constructs generally have a naturally occurring hypoxic center, which allows for the stabilization of HIF-1α in the interior tissue. Thus, short-term addition of DMOG may not further improve this in cartilage tissue engineered constructs.
Introduction
Articular cartilage is under 2 constant stresses, mechanical strain and hypoxia. In chronically hypoxic tissue such as articular cartilage, hypoxia supports cartilage development, survival, and function. 1 Mammalian tissue responds to and regulates the cellular response to low oxygen levels via mediation with hypoxia-inducible factor 1α (HIF-1α). 2 HIF-1α is a hypoxia-dependent constituent of the protein complex HIF, a transcription factor that upregulates gene targets important for chondrogenesis in mesenchymal stem cells. In normoxic cell culture conditions, hydroxylase inhibitor enzymes degrade HIF-1α. Previous research has shown that hypoxia enhances chondrogenic differentiation of mesenchymal stem cells (MSCs) when compared with ambient oxygen tension cell culture conditions.3,4
HIF-1α levels are controlled posttranslationally and are specifically regulated by 2 types of oxygen-sensing hydroxylases, prolyl hydroxylase domain-containing proteins (PHDs) and factor inhibiting HIF (FIH).
2
Furthermore, Murphy
In an effort to generate tissue engineered cartilage (TEC), co-culture systems of chondrocytes and mesenchymal stem cells are often used.
5
Given the instability of chondrocyte phenotype and function
Methods
Chondrocyte and SYN-MSC Cell Isolation
The chondrocytes and synovial (SYN)-MSCs used in both approaches, pellet and engineered construct culture, were from the same animal and isolation for each individual experiment that comprised the biological triplicate. Porcine articular chondrocytes were obtained via an overnight collagenase digestion (Worthington Biochemical) from juvenile porcine condyles (
Pellet Study
SYN-MSC and chondrocyte co-cultures were centrifuged in a 15-mL conical tube at a concentration of 0.5 × 106 cells and cultured in chondrogenic media with or without the hypoxic agent DMOG. The ratio of cell types was 4 SYN-MSCs to 1 chondrocyte to remain consistent with the tissue engineered cartilage. Media was changed 3 times per week. Pellets were harvested at days 14, 21, and 28 for data analysis (
Tissue Engineered Cartilage (TEC)
SYN-MSC and chondrocytes (ratio of 4 MSCs to 1 chondrocyte) were photoencapsulated in 1.5% methacrylated HA (MeHA) hydrogel disks (4 mm, 2 mm thickness) at a concentration of 40 million cells/mL of MeHA and cultured in chondrogenic media (DMEM, 1% ITS+Premix, 50 µg/mL L-proline, 0.1 µM dexamethasone, 0.9 mM sodium pyruvate, and antibiotics) supplemented with ascorbate (50 µg/mL) and 10 ng/mL transforming growth factor-β3 at 37°C and 5% CO2 as previously described.
8
Culture media was replaced twice per week and harvested at weeks 4 and 6 for evaluation (
DMOG Treatment
Dimethyloxalylglycine (Sigma-Aldrich) was dissolved in dimethyl sulfoxide, filtered, aliquoted in 200 µM concentrations and stored at −20°C prior to use. Fresh aliquots were added to media and deemed “treatment media.” Treated media was changed daily at the same time from day 14 to day 21 of culture for the pellets and TECs. Nontreated pellets and TEC had media change daily from day 14 to day 21 of culture to remain consistent with media change frequency of DMOG treatment. Week 3 time point samples were harvested for HIF-1α stabilization analysis within 8 hours of treatment (
Nuclear HIF-1α Co-Localization
To understand the impact of DMOG treatment on HIF-1α, immunofluorescent staining for HIF-1α was visualized and quantified in the tissues. This approach was utilized to assess whether differences were apparent in the center versus exterior of the tissues. Pellets and tissue engineered cartilage were sectioned using a CM3050 S Research Cryostat (Leica) at 5 µm and stained for HIF-1α using anti-HIF-1α and secondary antibodies (Fisher Scientific), and with DAPI for nuclear staining. Immunofluorescent images were collected on a laser scanning confocal microscope (FV1200, Olympus), HIF-1α appearing green, and DAPI blue. Images were collected at 20× from the center of the tissue, and from the exterior edge, through 100 µm into the tissues, for comparison of HIF-1α stabilization. Co-localization of overlapping DAPI and HIF-1α signal was analyzed by Image Pro Plus 7.0 (MediaCybernetics), and an increase in HIF-1α signal considered “stabilization” of the signal.
Histology
Sections from pellets and tissue engineered constructs (
Biochemistry
Pellets were weighed wet, lyophilized for 48 hours, reweighed dry, and digested in 0.5 mg/mL Proteinase-K at 60°C overnight (
Tissue engineered cartilage followed the same sample preparation (
Spectral Imaging Compositional Analysis
Spectral images were obtained to assess the relative distribution of specific components of the tissue. Tissue engineered cartilage constructs were sectioned at 30 µm thickness, and pellets sectioned at 5 µm thickness onto low-e microscope slides (Kevley Technologies;
Attenuated Total Reflection (ATR) Spectroscopy
Attenuated total reflection (ATR) spectral data were collected from pellets (
Mechanical Assessment
Tissue engineered cartilage constructs were harvested for mechanical analysis at weeks 4 and 6 (
Statistical Analysis
Results of outcome parameters were reported as means and standard deviations. Differences in condition (DMOG treated or untreated) and weeks were assessed by a 2-way ANOVA and Tukey’s HSD test, with
Results
Pellets: Nuclear HIF-1α Co-Localization and Signal Quantification
HIF-1α signal can be observed co-localized with nuclei in both DMOG-treated and untreated groups at day 21 (
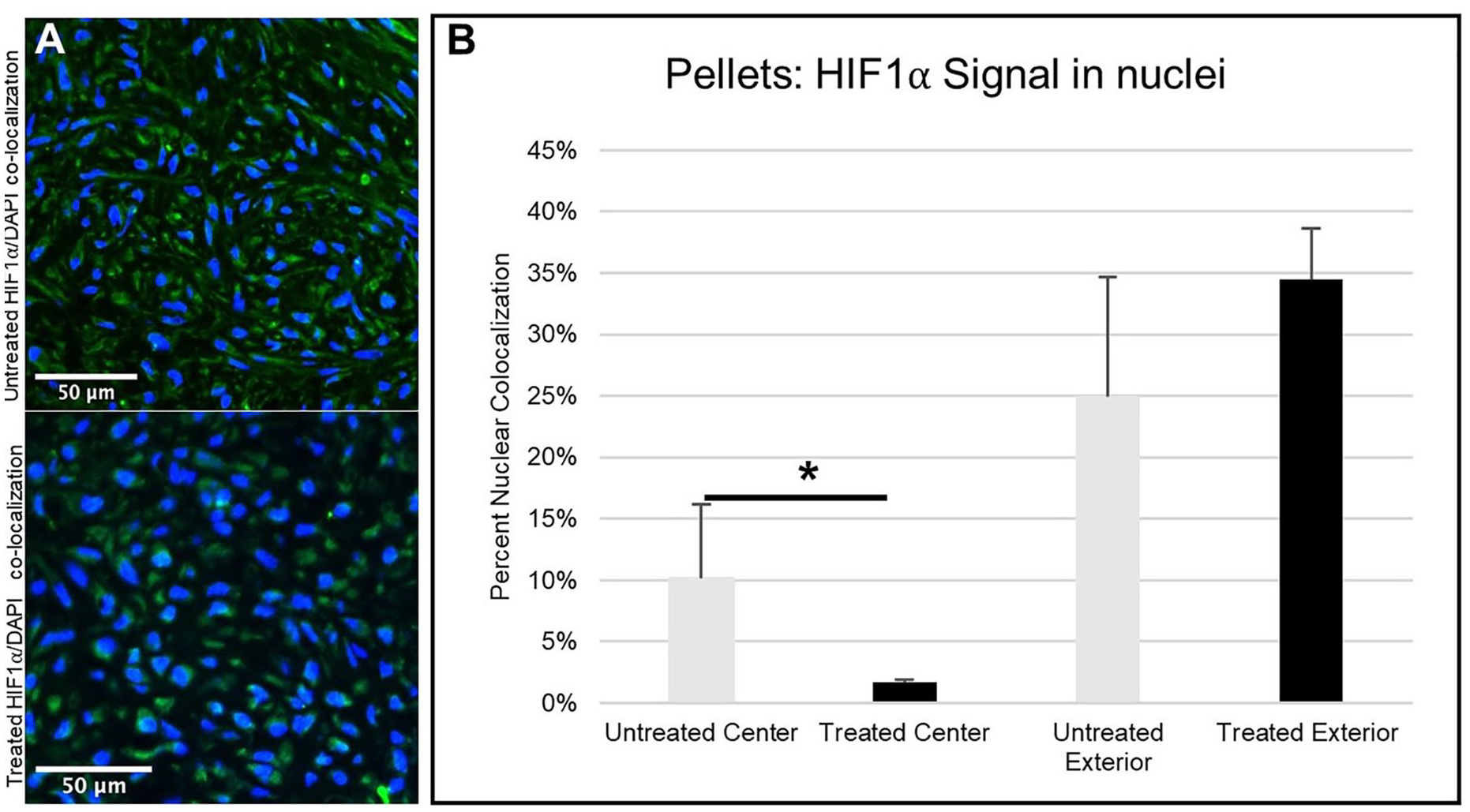
Immunofluorescence images of HIF-1α (green) co-localized with cell nuclei (blue) in (
Pellets: Qualitative Histology
The Alcian blue and picrosirius red stains demonstrated proteoglycan and collagen deposition, respectively, throughout the pellets for both the untreated and DMOG-treated pellets (
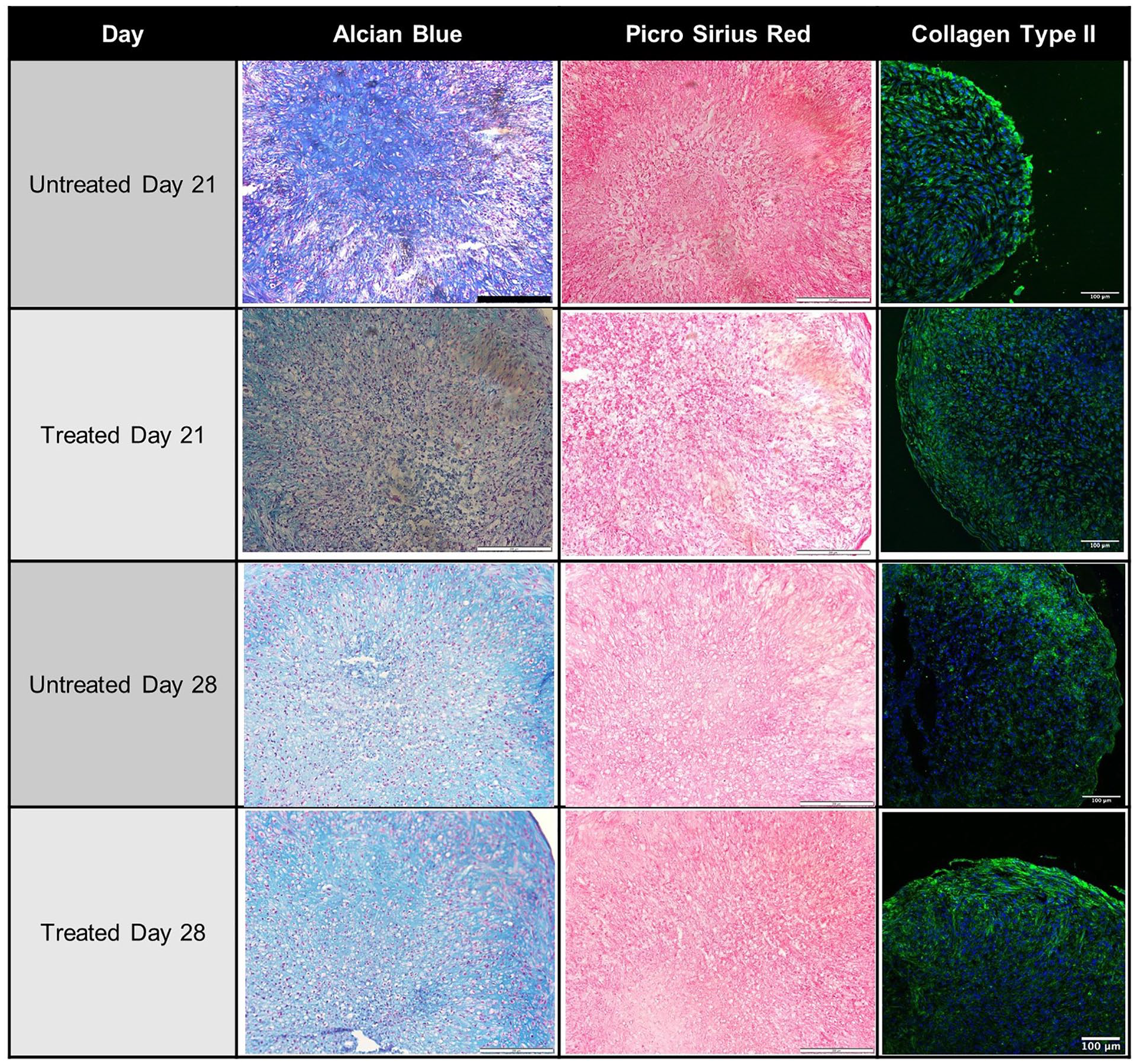
Representative histological images of each pellet culture group. Alcian blue was used to visualize deposited proteoglycans and picrosirius red to stain collagen. Scale bar for Alcian blue and picrosirius red images = 200 µm. Histological images shown are supplemented with corresponding images of immunofluorescent staining for type II collagen (green), with cell nuclei (blue) stained with DAPI. Scale bar for immunofluorescence images = 100 µm.
Pellets: Spatial Distribution and Quantification of Proteins
Spectral imaging was used to assess the spatial distribution of proteoglycan and collagen in pellets treated with or without DMOG. The spectral imaging method allows not only for the visual spatial distribution of proteins, but also the relative quantification of each protein. Proteoglycan was found distributed throughout the tissues (
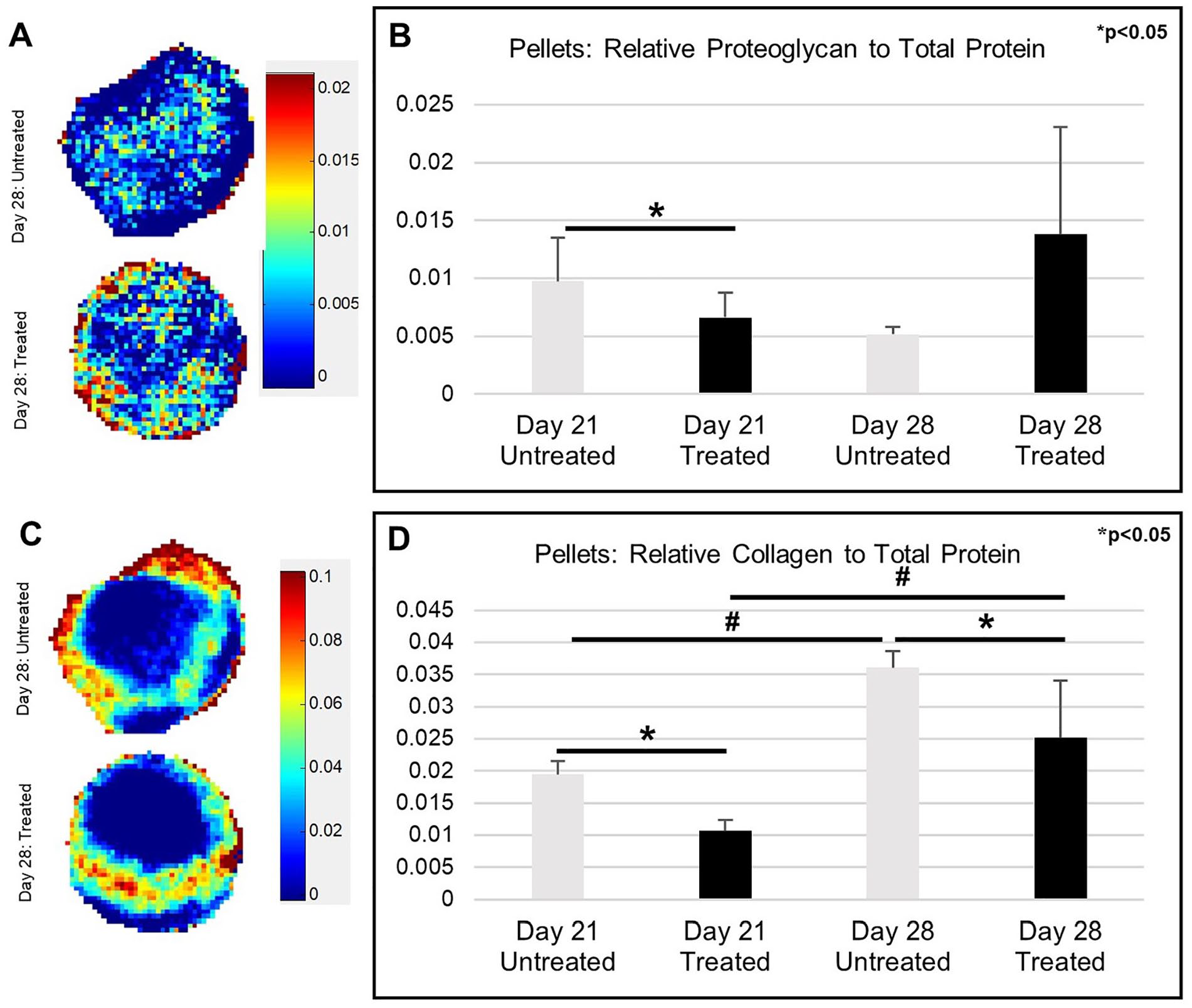
(
Pellets: Bulk Quantification of Collagen by ATR
The ATR technique was used to quantify differences in collagen content in the pellets, in lieu of biochemistry, which was not sensitive enough for the small amounts of tissue analyzed. The collagen peak in the untreated day 28 sample is both visibly clear (
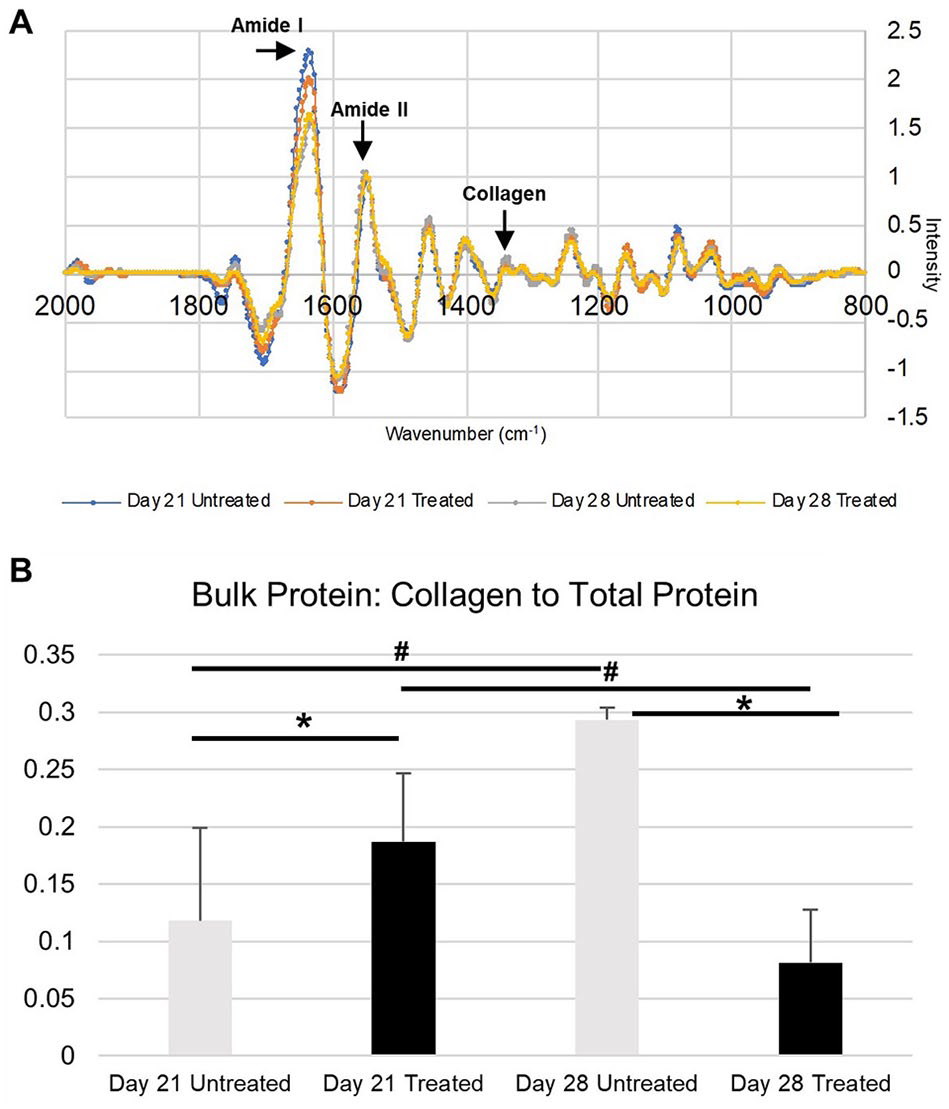
(
Pellets: Biochemical Assessment
Pellets were assessed for differences in proteoglycan content by the sGAG DMMB assay. DMOG did not impact bulk proteoglycan deposition at day 21 or day 28 when compared to the untreated groups (
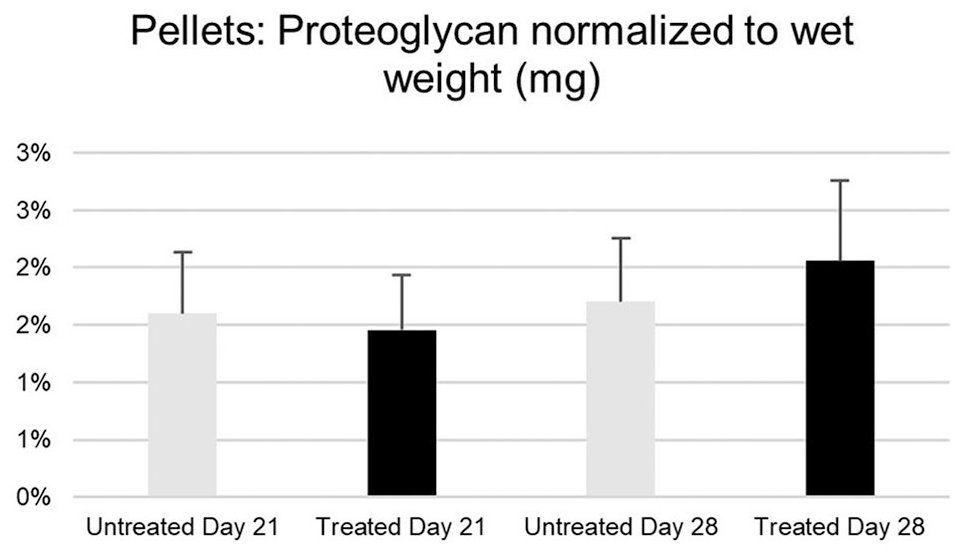
Quantification of bulk proteoglycan in cartilage pellets for DMOG-treated and untreated groups at days 21 and 28 (using the DMMB assay). No statistical differences among groups.
TECs: Nuclear HIF-1α Co-Localization and Signal Quantification
The co-localization of HIF-1α in the nuclei in Week 3 tissue engineered cartilage (
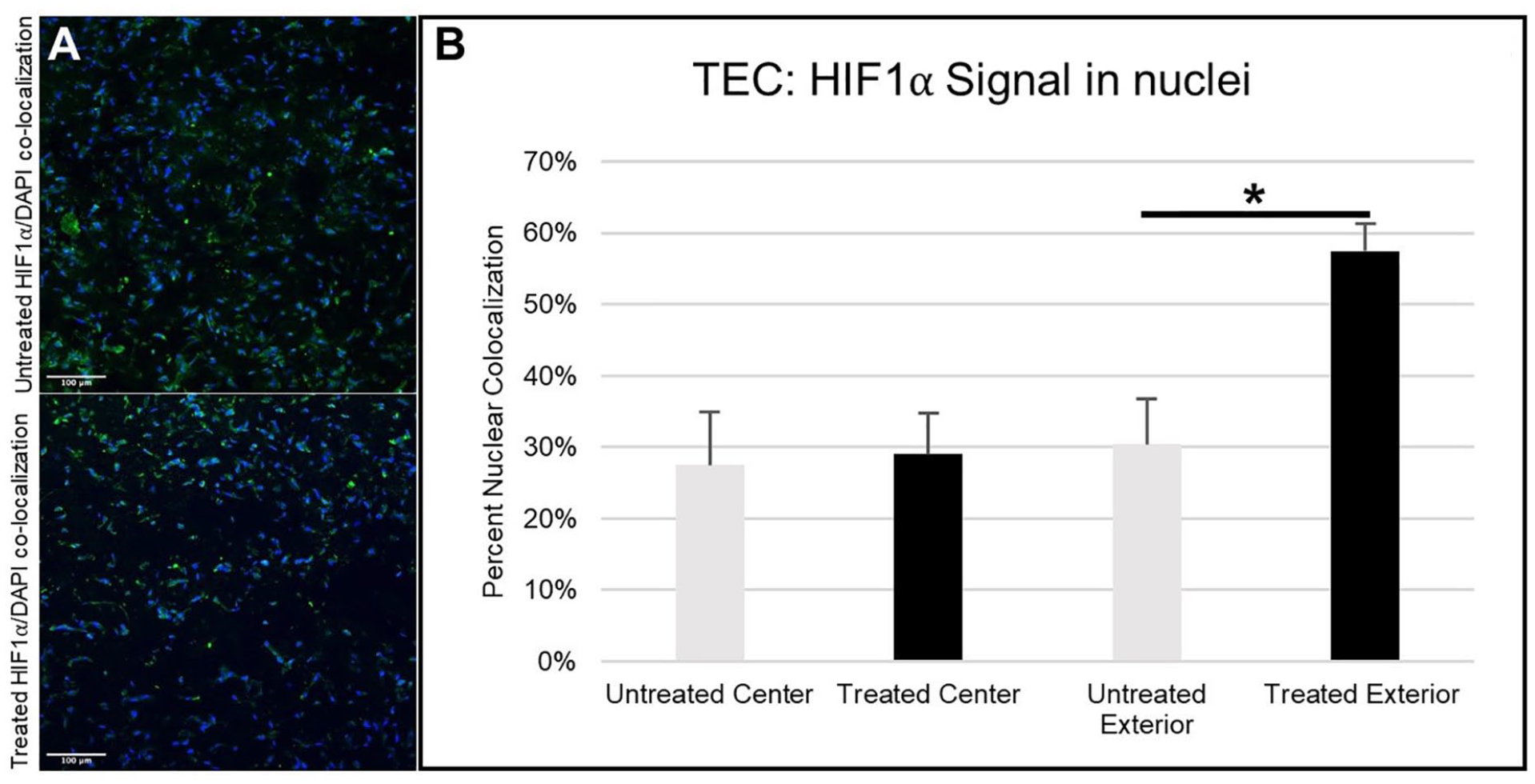
Immunofluorescence images of HIF-1α (green) co-localized with cell nuclei (blue) in (
TECs: Qualitative Histology
Tissue sections stained with Alcian blue (
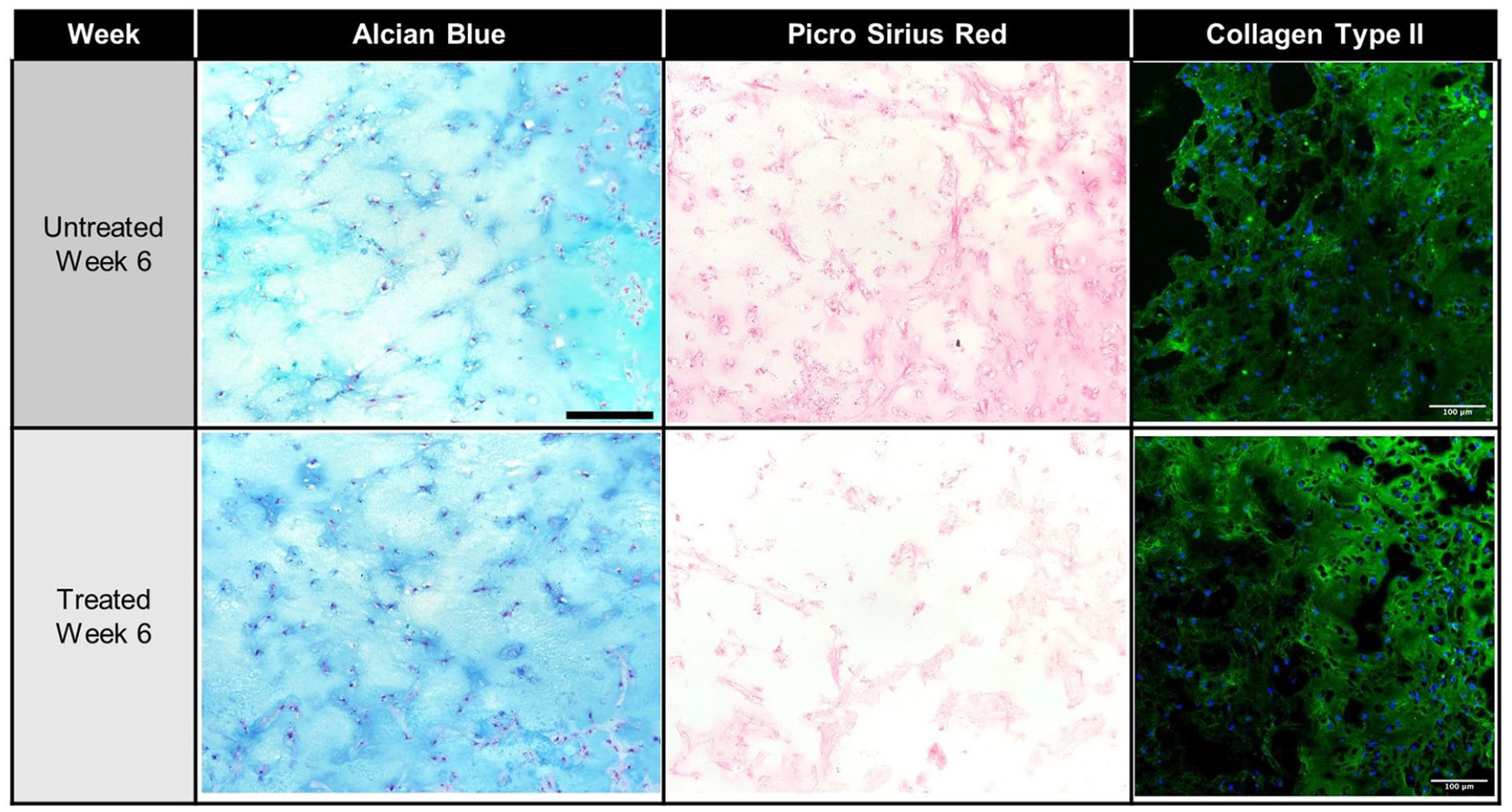
Representative histological images of the engineered cartilage construct groups at week 6. Alcian blue was used to visualize deposited proteoglycans and picrosirius red to stain collagen. Scale bar for Alcian blue and picrosirius red images = 200 µm. Histological images shown are supplemented with corresponding images of immunofluorescent staining for type II collagen (green), with cell nuclei (blue) stained with DAPI. Arrows indicate edge of construct. Scale bar for immunofluorescence images = 100 µm.
TECs: Spatial Distribution and Quantification of Proteins
Spectral imaging allowed for the evaluation of relative amounts and spatial distribution of protein within the DMOG-treated and untreated constructs (
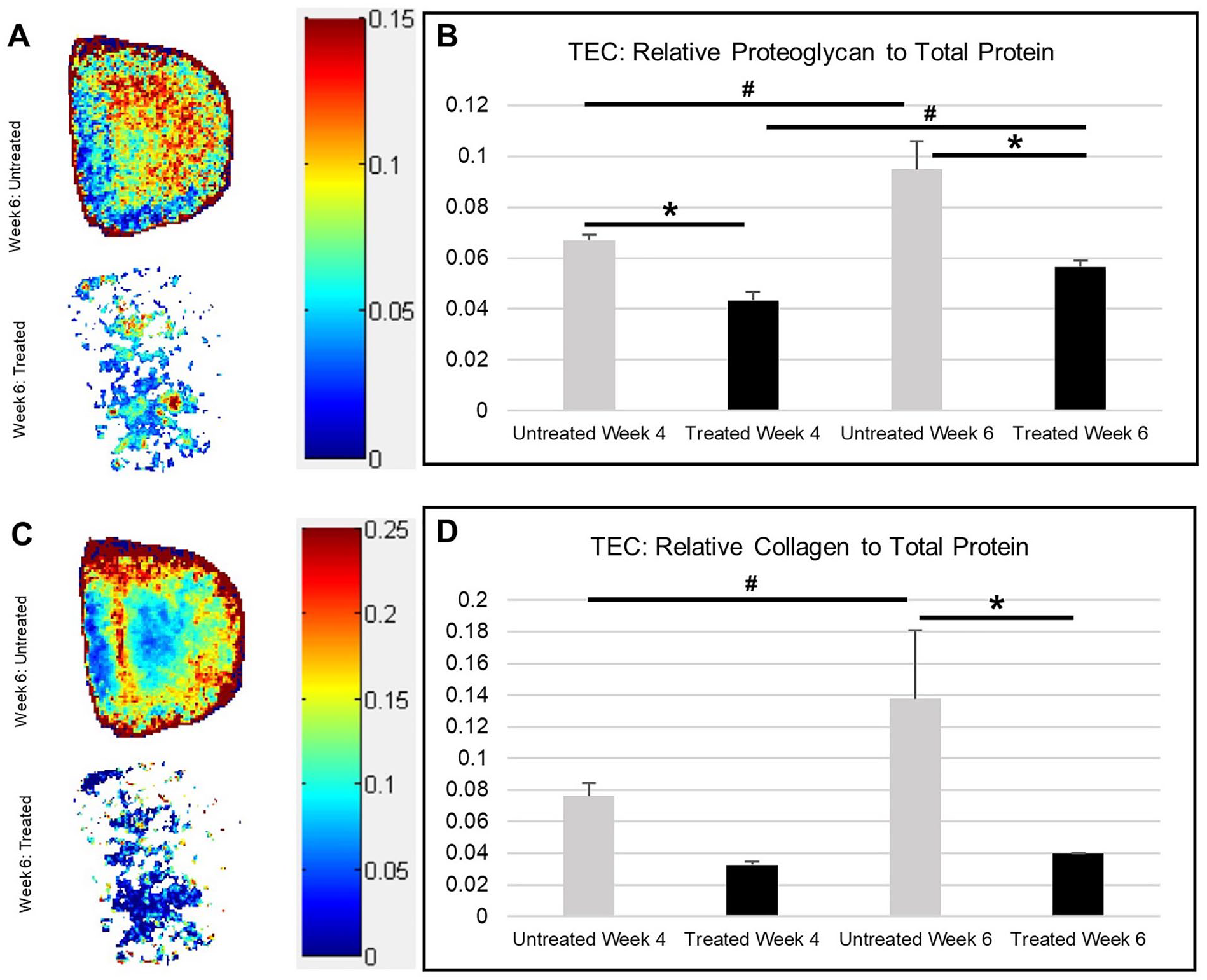
(
TECs: Biochemical Assessment
Consistent with the spatial distribution analysis and quantification of proteins by spectral imaging, the biochemical results of proteoglycan (
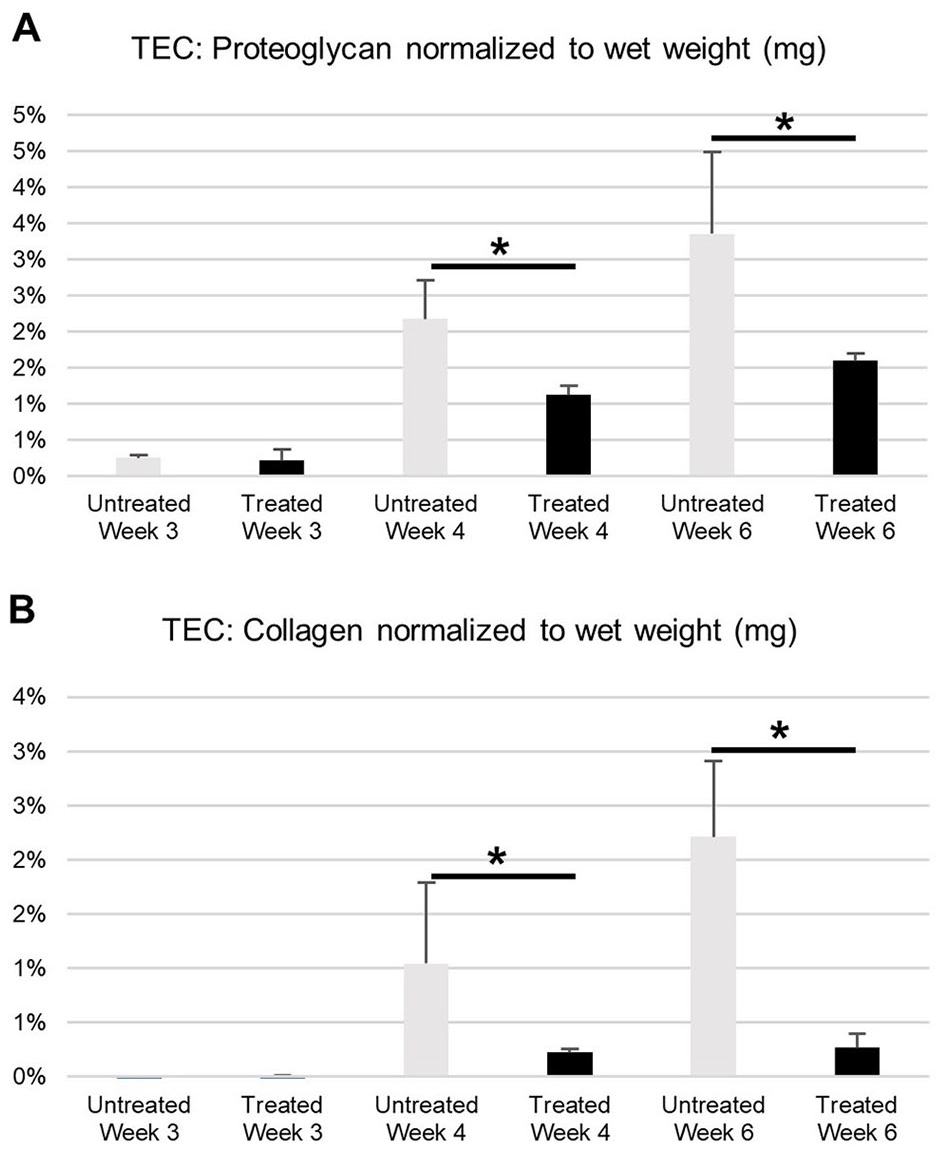
(
TECs: Mechanical Assessment
Deposited proteoglycan and collagen play a critical role in the compressive properties of cartilage. Thus, the statistical decrease in both equilibrium modulus (
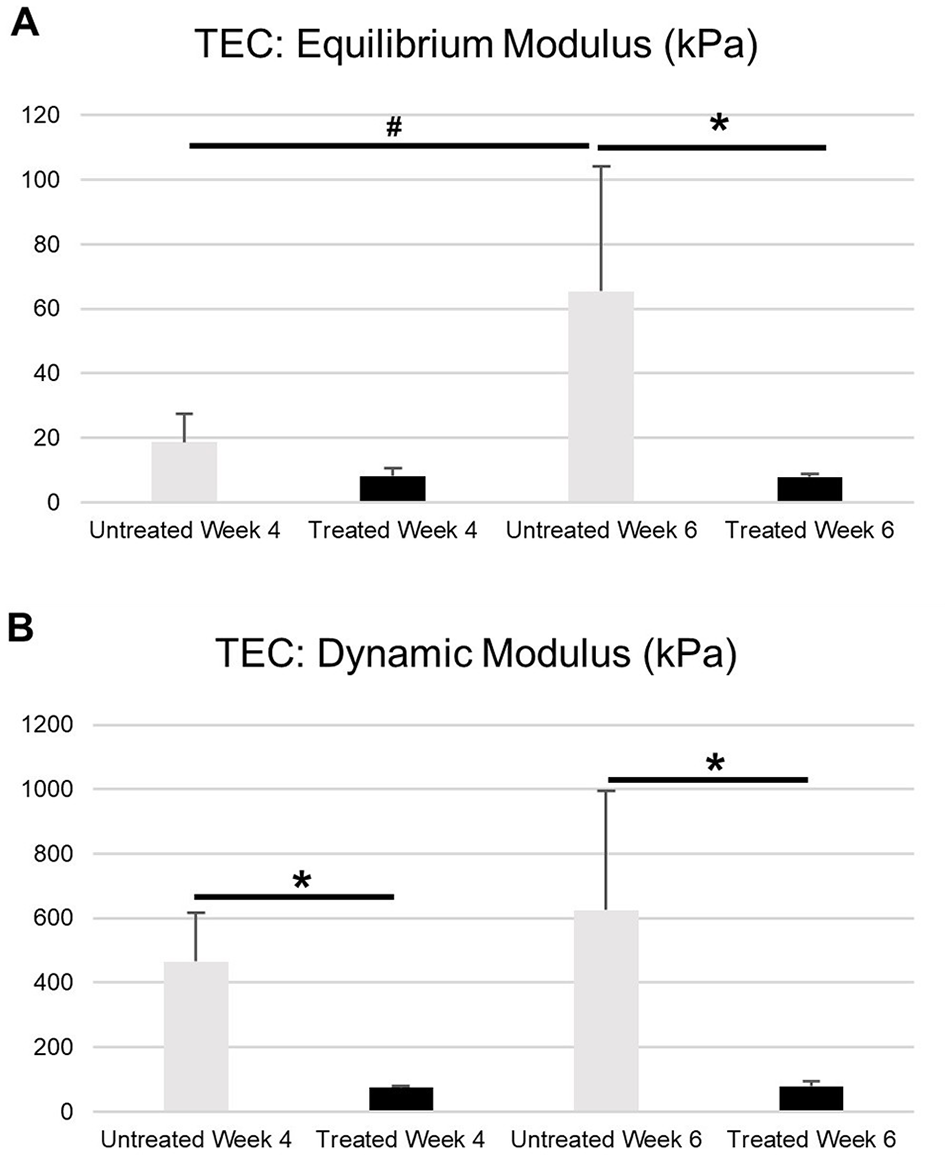
(
Discussion
The motivation behind this study was to investigate the impact of a hypoxia simulating agent, DMOG, on the differentiation on SYN-MSCs in scaffold-free and scaffold-based tissue engineered cartilage. This study, which represents the first DMOG treatment for cartilage tissue engineering, found that DMOG had a negative effect on the cartilaginous extracellular matrix deposition despite an increase in HIF-1α stabilization in regions of the tissues.
Cartilage tissue was engineered using a co-culture system comprised of chondrocytes and synovial-derived mesenchymal stem cells. SYN-MSCs were chosen because of their
Dimethyloxalylglycine, cobalt chloride (CoCl2), and deferoxamine (DFO) have been shown to augment chondrogenic differentiation by mimicking hypoxic conditions required for chondrocyte growth. Cobalt chloride does so by replacing iron in prolyl hydroxylase 2 (PHD2), acting as a chelating agent to prevent the degradation of HIF-1α, a necessary transcription activator that is upregulated in hypoxia to promote ECM growth. Cobalt chloride has been shown to increase chondrogenic and osteogenic potential, as well as inhibit adipogenesis. 17 When 0.1 mM CoCl2, a concentration empirically determined to limit toxicity to cells, was added to Murine MSCs (C3H/10T1/2) for 24 or 48 hours before growth in chondrogenic medium, HIF-1α mRNA was significantly increased, and accompanied by a significant increase in aggrecan and type II collagen (Col2a1) at 48 hours of exposure, and a slight increase in Sox9 at 24 hours of exposure.
Deferoxamine (DFO or desferrioxamine) is a chelating agent initially designed to remove excess iron in the bloodstream and has been shown to have additional biological applications by synergizing with the action of TGF-β to induce chondrogenesis. 18 The mechanism of action of DFO is related to the sequestration of iron, which is necessary for the action factor inhibiting HIF (FIH) and PHD2, thereby amplifying the concentration of HIF-1α within the cell and simulating the effects of hypoxia. 7 With the addition of DFO and TGF-β to chondrocytes, Col2a1, Sox9, and aggrecan expression, as well as cell proliferation, were significantly increased, compared to the null control, DFO alone, and TGF-β alone. 18
A previous study using PC12 cells (rat adrenal phaeochromocytoma) grown in monolayer treated with DMOG at a concentration of 1 mM for 2 hours showed HIF stabilization by Western blot analysis. 19 The current study is the first to assess the effect of DMOG by consideration of spatial localization, and investigation of HIF-1α stabilization co-localized with nuclei. Co-localization with nuclei was an important step in this evaluation, as HIF-1α needs to be translocated into the nucleus to initiate transcription of target genes. Furthermore, co-localization within specific regions was representative of a spatially stabilized molecule, thereby confirming that hypoxia was achieved throughout the tissue and not only at the naturally hypoxic core. The exterior of the tissue engineered construct is not typically hypoxic, as is the construct center, and therefore the presence of HIF-1α in the exterior was enhanced by DMOG treatment. The results, a demonstration of increased HIF-1α stabilization in the exterior of tissue engineered cartilage constructs treated with DMOG, unfortunately did not translate to positive changes in the cartilaginous matrix. Accordingly, at the time points used in this study, DMOG likely did not improve chondrogenic differentiation in SYN-MSCs co-cultured with chondrocytes in pellets or in tissue engineered cartilage and therefore was not a suitable alternative to traditional hypoxia chambers.
The reduced cartilaginous extracellular matrix found in DMOG-treated pellets and treated TEC are indicative of a negative impact of DMOG. These results were not consistent with previous studies where low oxygen settings, such as chambers, were used, to enhance chondrogenesis.15,20,21 The extracellular matrix attenuation in the DMOG treated tissue engineered cartilage were subsequently reflected in the mechanical properties of the tissue, which is the ultimate determination of tissue function.
Previous research indicated that DMOG is effective at inducing HIF-1α nuclear localization and regulation of gene targets in 2D monolayers of human bone marrow mesenchymal stem cells. However, this research also showed that DMOG inhibits extracellular matrix (ECM) protein incorporation, ultimately yielding significantly reduced cartilage-like ECM with less collagen type II and glycosaminoglycans (GAGs) unless exposed only during later stages of chondrogenesis. 7 Here, samples were treated with DMOG from day 7 to day 14 of culture as previously described for the upregulation of chondrogenic genes. 7 Further exploration into dosing options may be a potential strategy for the use of DMOG for chondrogenesis.
Although here ECM deposition was impaired with DMOG treatment, Taheem
One explanation for the results of the current study is that DMOG, which is a 2-oxoglutarate analog, can inhibit prolyl 4-hydroxylase, an enzyme involved in the synthesis of 4-hydroxyproline residues, which are crucial for stabilizing collagen triple helices in physiological conditions.
23
Although sustained exposure to DMOG may in fact inhibit incorporation of ECM proteins into hydrogel matrix, some hydrogel-embedded cultures have shown success using DMOG with short-term exposure. Sathy
Another potential path that may have affected DMOG efficacy was a possible competition with HIF-1α stabilization through natural hypoxia. It is well established that 3-dimensional cell culture generates a naturally occurring hypoxic core due to the lack of oxygen diffusion. 25 The HIF-1α co-localization study showed that HIF-1α was stabilized in both the treated and untreated groups, indicating that oxygen tension remained the same on the interior center and exterior of the pellets. In the tissue engineered constructs, which were larger than pellets at 2 mm thick and a circumference of 4 mm, DMOG did elevate HIF-1α stabilization in the exterior region. However, the interior center results indicated that the lower oxygen tension had already generated a hypoxic core regardless of DMOG supplementation. Untreated tissue engineered cartilage deposited greater proteoglycan and collagen than the DMOG-treated group, suggesting that the naturally occurring hypoxic environment was sufficient for robust cartilaginous output.
Furthermore, clinical use of tissue engineered cartilage embedded with DMOG must also take into consideration the potential side effect of DMOG diffusion to adjacent tissues. It is known, for example, that DMOG can act independently of the PHD/HIF pathway. Zhdanov
In conclusion, this study investigated the potential use of a hypoxia-simulating agent to improve the chondrogenic capacity of SYN-MSCs in engineered cartilage tissue by pellet formation and hydrogel-based scaffolds. The work herein demonstrated that sustained DMOG treatment is not a suitable alternative for traditional methods to control oxygen tension
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded by NIH R01 AR056145.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not sought for the present study because ethical approval was not required for this study.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because no live animals were used to conduct the research described herein.
