Abstract
The aim of this study is to evaluate the mechanical and biological performance of cartilage-like constructs produced by 3D printing. During the investigation, poly(ε-caprolactone) (PCL) and polyvinylpyrrolidone (PVP) were used as a matrix polymer and low-molecular-weight chitosan (CS), hyaluronic acid (HA), and alginic acid sodium salt (SA) were integrated separately with the polymer matrix to fabricate the constructs. Thermal, mechanical, morphology, and chemical properties and swelling, degradation, and biocompatibility behaviors were evaluated in detail. With the addition of 3 fillers, the melting temperature of the matrix increased with the addition of fillers, and PCL/3wt.%PVP/1wt.%HA had the highest melting temperature value. Mechanical characterization results demonstrated that the printed PCL/3wt.%PVP/1wt.%CS displayed the highest compressive strength of around 9.51 MPa. The compressive strength difference between the PCL/3wt.%PVP and PCL/3wt.%PVP/1wt.%CS was 5.38 MPa. Biocompatibility properties of the constructs were tested by mitochondrial dehydrogenase activity, and
Introduction
Joint pain and chronic disability are caused mainly by osteoarthritis, aging, and joint damage. For self-healing of mature cartilage, blood vessels, nerves, and lymphatics must be present. Joint replacement surgery is the most common treatment for cartilage degeneration in an advanced stage, but this treatment is highly complicated and expensive.1,2 Although cell transplantation–based tissue engineering treatment for human cartilage repair has been practiced since almost 20 years, the new tissue generated is not yet been like a natural cartilage when using existing cartilage tissue engineering strategies, regional cloning, extracellular matrix composition.
1
Cartilage tissue engineering has been a developmental area for the last 20 years. However, there are still many clinical obstacles for treatment, even though there have been huge efforts on developing new biological solutions. In this period, a lot of developmental efforts ensued that have or could positively affect biomaterial technologies, cell sources, molecular mechanisms, and genetic manipulations for cartilage tissue engineering.
3
Inadequate success in tissue engineering has shown that it does not have complexity with regard to cell types and tissue patterns, so interest in the tissue engineering of the musculoskeletal system using 3-dimensional (3D) printing technology has increased.
4
Three-dimensional scaffolds, which can be produced with biodegradable, biocompatible polymers, and cells are linked in order to get
Materials and Method
Materials
Poly(ε)caprolactone (PCL; molecular weight [MW] = 80,000 Da), polyvinylpyrrolidone (PVP; MW = 40,000 Da), chitosan (CS; MW = 50,000-190,000 Da), alginic acid sodium salt (SA; MW = 120,000-190,000 Da) were obtained from Sigma Aldrich, and hyaluronic acid (HA) was obtained from Yigitoglu Chemistry (Istanbul, Turkey).
Preparation of Digital Model and G-Code Export
Preparation of the 3D cartilage constructs consisted of 2 steps: (1) PCL/3wt.%PVP matrix formation and (2) formation of PCL/3wt.%PVP/1wt.%CS, PCL/3wt.%PVP/1wt.%HA, PCL/3wt.%PVP/1wt.%SA constructs. To form both constructs, solid model of the structures were drawn in 20 × 20 × 3 mm3 dimensions, and it was transferred in Simplify program to get G-code, which gave a command to the 3D device for printing the constructs.
Preparation of PCL/3wt.%PVP Solution and Constructs
First, PCL (9.7 g) and PVP (0.3 g) were weighted by using a scientific balance (RADWAG). In the second step, PCL was put on a magnetic stirrer (Velp Scientifica) at 150°C to get the gel form, and after 20 minutes, PVP was added into this gel and mixed manually for forming a homogeneous mixture. After the mixing process was completed, the homogeneous structure was shaped to take the shape of the printer’s chamber and the mixture was put into this chamber. Constructs were built according to the G-code commands. For fabrication of PCL/3wt.%PVP constructs, 60% and 70% infill properties were adjusted to determine the ideal pore size distributions. Pore size values at these 2 infill rates were measured via SEM (scanning electron microscopy) and Optical Microscope Analysis program (Olympus Analysis, USA) by using an SEM image (50× magnification) and this image was transferred to the Analysis program to measure the pore dimensions theoretically. Almost 20 pores were measured for PCL/PVP constructs with 60% and 70% infill rates and the mean pore size values of the constructs were obtained. Another process parameter is temperature, which is a very crucial parameter for building the constructs. The device is also available for changing the print speeds and extrusion rates at the optimization stages. The fabrication process stages are given in Figure 1a-e .
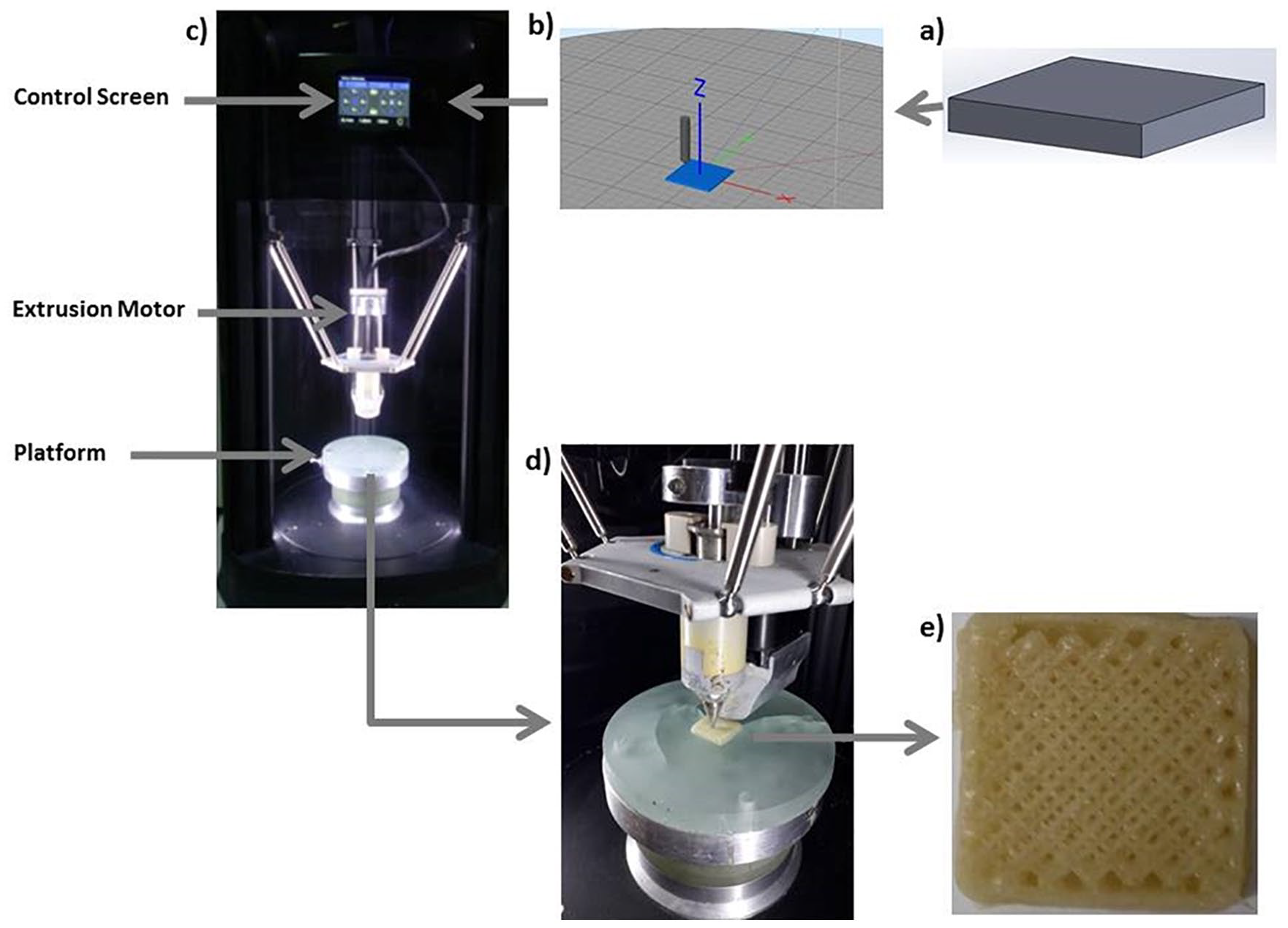
The process steps for cartilage construct fabrication: solid model (
Preparation of the PCL/3wt.%PVP/1wt.%(CS, SA, HA) Solutions and Constructs
First, PCL and PVP (3 wt.%) were mixed using the same method as described above. After that CS, SA, and HA powders were added into the matrix polymer as a filler with the same amount (1 wt.%). PCL/3wt.%PVP/1wt.%(CS, SA, HA) mixtures were printed with the same production parameters as in PCL/3wt.%PVP matrix construct to obtain the constructs.
Characterization of the Printed Constructs
To observe the characteristic properties of the printed constructs, characterization tests were performed. Fourier-transform infrared spectroscopy (FTIR, JASCO-4000) was used to detect the presence of typical functional groups of pure polymers. Scans were recorded in transmittance mode, between 4,000 and 400 cm−1 with a resolution of 4 cm−1. Printed constructs were shaped as plate and the test was performed for all constructs and compared with pure components. Morphological properties were obtained by using SEM (EVO MA10). The constructs were coated with gold for 90 seconds, 18 mA prior to imaging, to enable conductivity in the constructs. Differential scanning calorimetry (DSC, Shimadzu) was used to determine the thermal transitions of the constructs in the closed pan, and heating temperature ranges were adjusted from room temperature to 200°C and the heating rate was selected as 10°C/min. Mechanical properties of the constructs were determined with a compressive testing device (Shimadzu) to examine the mechanical behavior of the constructs. Test parameters, temperature, force, and speed, were adjusted to 20°C, 0.1 N, and 1 mm/min, respectively. Two experiments were performed for each constructs allowing for the mean values to be determined. The swelling test is similar to the method used in Correia
Results and Discussion
Morphology and Pore Size Analysis of Printed Cartilage Constructs
Scaffold pore size is an important parameter in tissue regeneration, and pores enable cells to an unstable environment to control the formation of new tissues.
21
According to the SEM results in
Figure 2a-d
, it was observed that pore sizes were getting smaller when the infill rates were increased. Sixty percent infill rate gave ~528 µm and 70% infill rate gave ~400 µm mean pore size values. Additionally, it was determined that 60% infill rate gave a better distribution than 70% infill rate. Therefore, other constructs were produced with this infill rate value. As a result of the pore distribution for constructs fabricated with 60% infill rate, different pore sizes and structures were observed. The average pore sizes for PCL/PVP/CS (1 wt.%), PCL/PVP/HA (1 wt.%), and PCL/PVP/SA (1 wt.%) were 512 µm, 448 µm, and 436 µm, respectively. Pore size range in tissue engineering usually ranges from ~150 µm to ~500 µm.22-24 According to the Zhang

SEM images of the cartilage construct with 60% (
FTIR Analysis
In Figure 3A , pure components of the composites were subjected to FTIR to compare the chemical structures of the composites with their components. In Figure 3(A, a) , 3 main absorption peaks were observed at ~2940 cm−1 (asymmetric CH2 stretching), ~2865 cm−1 (symmetric CH2 stretching), ~1720 cm−1 (carbonyl stretching) for PCL. 26 In Figure 3(A, b) , CH2 asymmetric stretching vibration occurred at ~2943 cm−1. The peaks at ~1016.3 cm−1 and ~1282 cm−1 refer to the CH2 rocking mode and twisting of CH2, respectively. The peak at ~1491 cm−1 refers to the CH2 scissor mode. 27 CS had main absorption peaks at ~3283 cm−1 (N-H stretching), ~2869 cm−1 (CH stretching), ~1589 cm−1 (C=O stretching; Fig. 3(A, c) ). 26 In Figure 3(A, d) , SA had absorption peaks at ~3227 cm−1 due to the O-H stretching, asymmetric stretching of COO observed at ~1592 cm−1, and symmetric stretching of COO observed at ~1404 cm−1. HA showed 3 main absorption peaks at ~2888 cm−1 (C-H stretching), ~1605 cm−1 (amid II group), ~1403 cm−1 (C-O; Figure 3(A, e) ). 28 The FTIR spectra of PCL/3wt.%PVP, PCL/3wt.%PVP/1wt.%CS, PCL/3wt.%PVP/SA, and PCL/3wt.%PVP/1wt.%HA are shown in Figure 3B(a, b, c, d) , respectively. In the PCL/3wt.%PVP spectrum, ~1720 cm−1 and ~2864 cm−1 belong to the PCL. The peak at ~2941 cm−1 was due to the interaction between the PCL (~2940.9 cm−1) and PVP (~2943.8 cm−1). Absorption peaks at ~1365 cm−1 and ~1160 cm−1 were observed after interaction between the polymers. The FTIR spectrum of the PCL/3wt.%PVP/1wt.%CS is shown in Figure 3B(b) . Small differences were observed between the PCL/3wt.%PVP and PCL/3wt.%PVP/1wt.%CS absorption graphics. The absorption peak for PCL/3wt.%PVP was shifted from ~1720 cm−1 to ~1721.2 cm−1 after CS addition. Another interaction peak changed from ~450 cm−1 to ~453 cm−1 with the addition of CS. In Figure 3B(c) , a similar spectrum was observed with the other composites except for 2 absorption peaks, which were ~1721 cm−1 and ~452 cm−1 after SA addition. In Figure 3B(d) , a similar tendency was observed for HA, and the peaks at ~2942 cm−1 and ~452 cm−1 were different from the PCL/3wt.%PVP absorption spectrum.
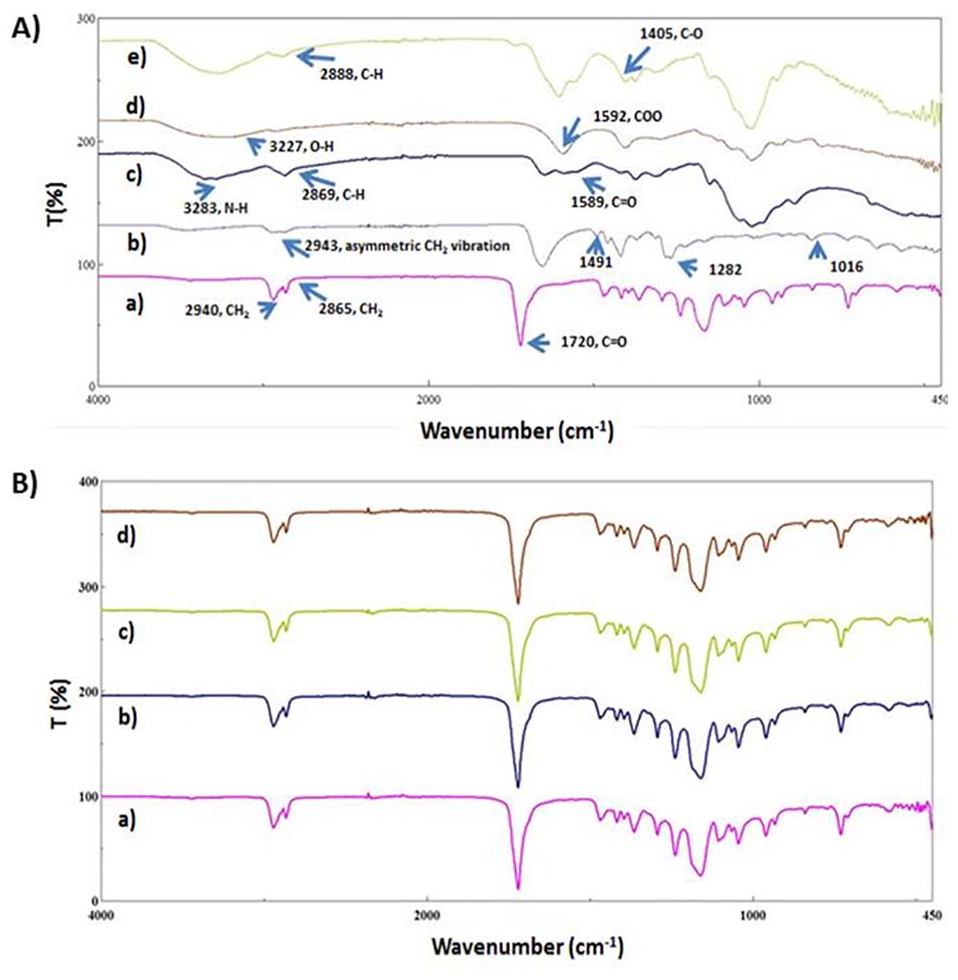
FTIR spectrum of pure components: PCL (
Thermal Analysis of Constructs
The resulting DSC thermograms are shown in Figure 4 . Sharp single endothermic peaks were observed at around 62°C to 65°C for all groups. This temperature range correlates to the melting points of the PCL. 29 The melting temperature of the PCL/3wt.%PVP matrix was 62.66°C ( Fig. 4a ). This value was increased with the addition of the CS, HA, and SA to 65.71°C, 67.33°C, and 64.98°C, respectively ( Fig. 4a, b, c, d ). The melting temperature of PCL increased with the addition of the PVP, CS, HA, and SA. These changes might be due to the interactions between the components. According to the DSC results, it was observed that the 3D-printing method and high processing temperature did not cause a significant change in the chemical structure of PCL. 29 The melting point of the PVP (at ~179°C) was not seen in the curve, and this might be due to its dominant amorphous structure and its low amount in the mixture. 30
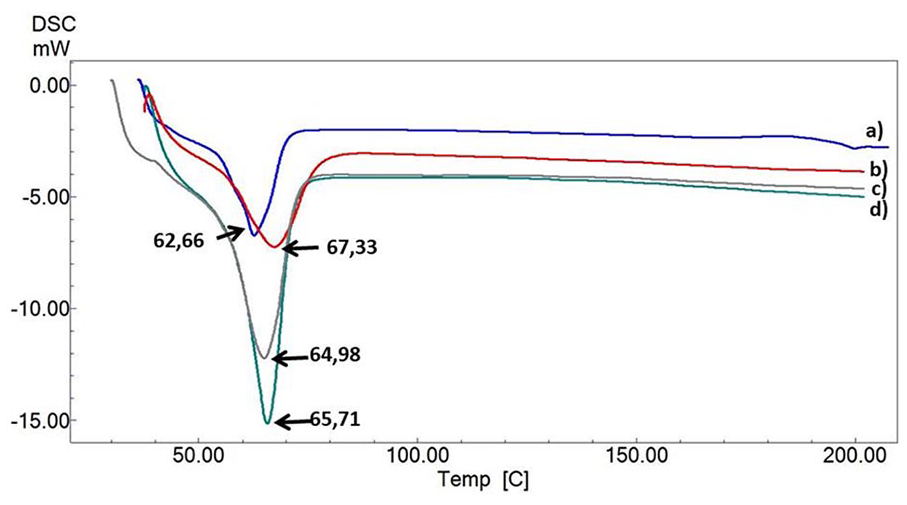
DSC thermograph for all constructs with their melting points values and labels: PCL/3wt.%PVP (
Compressive Strength of the Constructs
Mechanical properties of articular cartilage are vital due to its structure, which enables the body to smooth surfaces for movement of joints. 31 According to the compressive testing results given in Table 1 and Figure 5 , it can be said that with the addition of polymer, compressive strength values were increased and PCL/3wt.%PVP/1wt.%CS had the highest compressive strength value (9.51 MPa) under the same test conditions. PCL/3wt.%PVP had the lowest compressive strength value (4.13 MPa) compared with the other compressive strength values of the constructs. This result indicated that additive polymers increased the mechanical properties of PCL/3wt.%PVP. Although higher strength values were obtained with addition of the polymer into the matrix, elongation (%) values were nearly the same for all constructs. It means that they increased the resistance to impacts with the same elongation properties. Native cartilage had ~1.5 MPa compressive modulus value, 12 and the compressive strength values found in this study showed that printed cartilage constructs had high enough strength in terms of mechanical properties for cartilage tissue applications.
Compressive Testing Results of All Constructs.
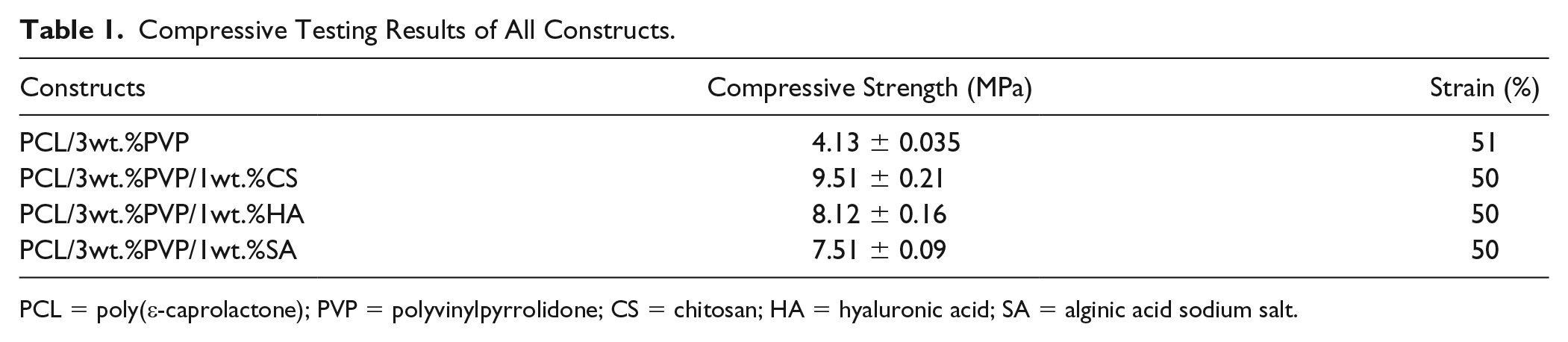
PCL = poly(ε-caprolactone); PVP = polyvinylpyrrolidone; CS = chitosan; HA = hyaluronic acid; SA = alginic acid sodium salt.
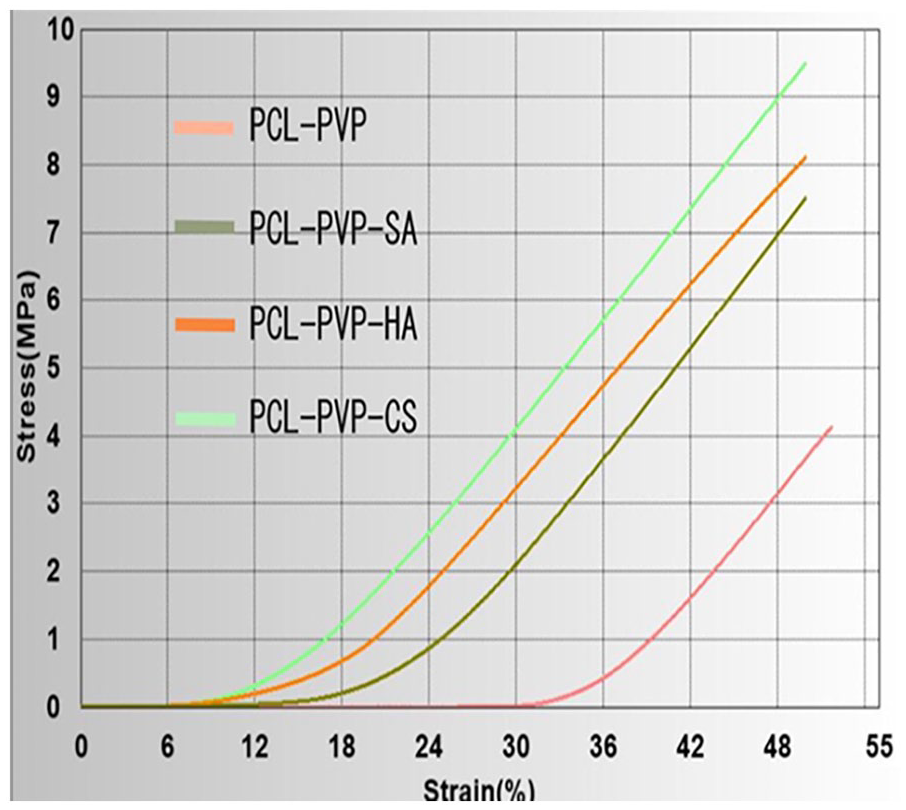
The stress-strain curve obtained from the compressive test.
Swelling Behaviors of the Constructs
Swelling properties are related to the diffusion of nutrients and waste throughout the whole structures 20 and providing liquid absorption of secretions. 32 The swelling capacity of the constructs was examined for 7 days. The mean weights were taken between the 3 values for each construct and error bars were added as standard deviation. In Figure 6a , it was observed that PCL/3wt.%PVP/1wt.%HA had higher swelling rate than the other constructs. In the first 72 hours, all constructs increased their weight rapidly and this situation continued until 144 hours. In Figure 6a , the composites that consisted of CS and SA had a lower swelling rate than the control group. The swelling results showed that HA could improve the hydrophilicity and wetting properties of the PCL/3wt.%PVP construct. This aspect may indicate that the diffusion and absorption of solutes throughout the pores in PCL/3wt.%PVP are improved by the addition of HA. 21
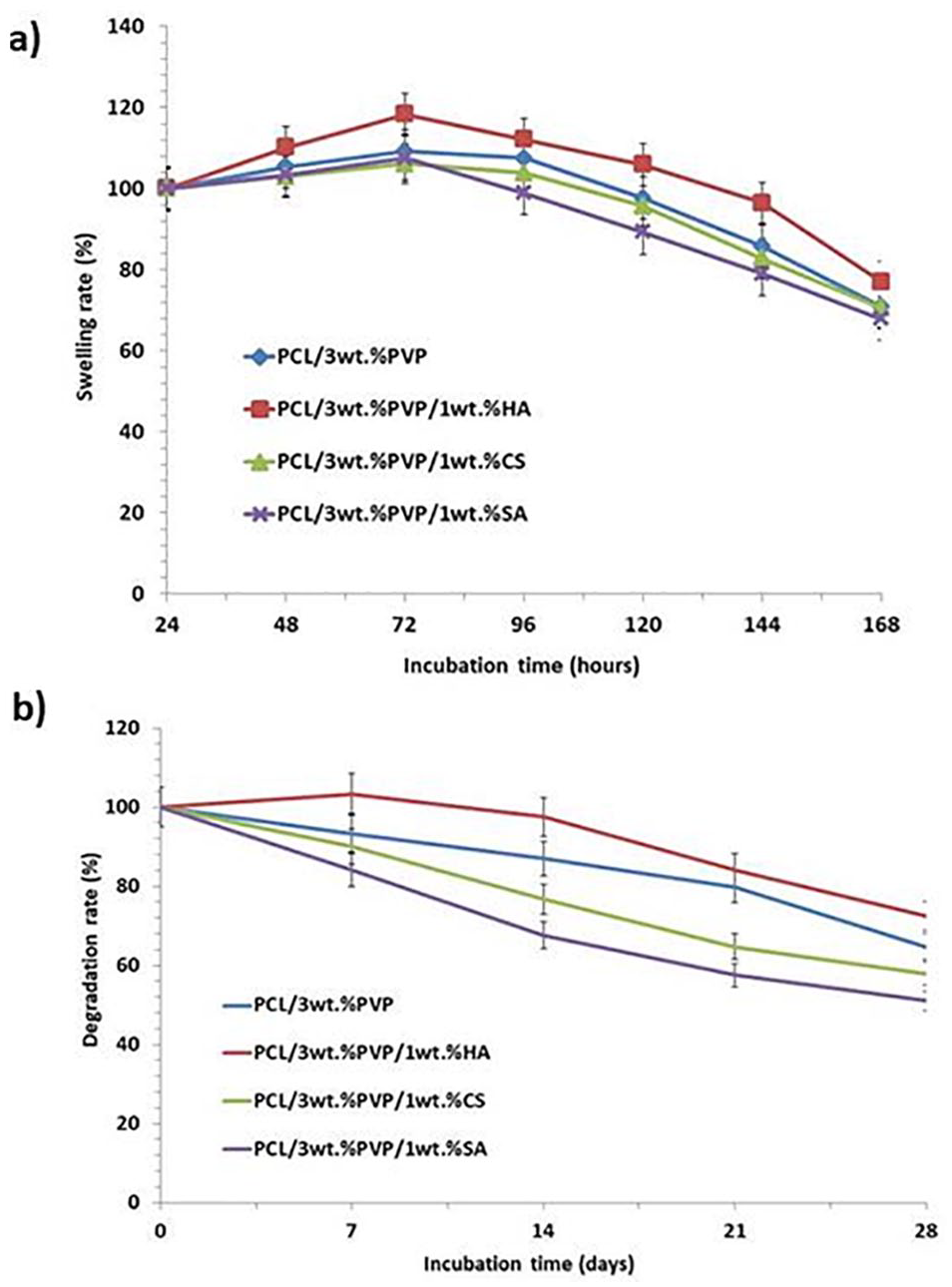
Swelling (
In Vitro Degradation of Constructs
The
Cytocompatibility of Cartilage Constructs
The cell adhesion, proliferation, and cytotoxicity properties were evaluated with MTT assay. Figure 7 shows the MTT absorption (590 nm) values for MSCs seeded in the microplate during the cell culture period (3 days). According to the results after 72 hours of incubation, it can be said that constructs had non-cytotoxicity and each construct has an impact on the growth of cells distinctively. Although proliferation rates were different for all constructs, no statistically significant effect was observed. However, the highest viability (%) was observed for PCL/3wt.%PVP/1wt.%HA, and can be due to HA, which is the major component of the extracellular matrix, 34 and thus facilitates increase in cell aggregation, proliferation, and migration. 35 For further studies, we need to undertake molecular or protein preliminary analysis to determine the potential of printed constructs as a cartilage structured template.
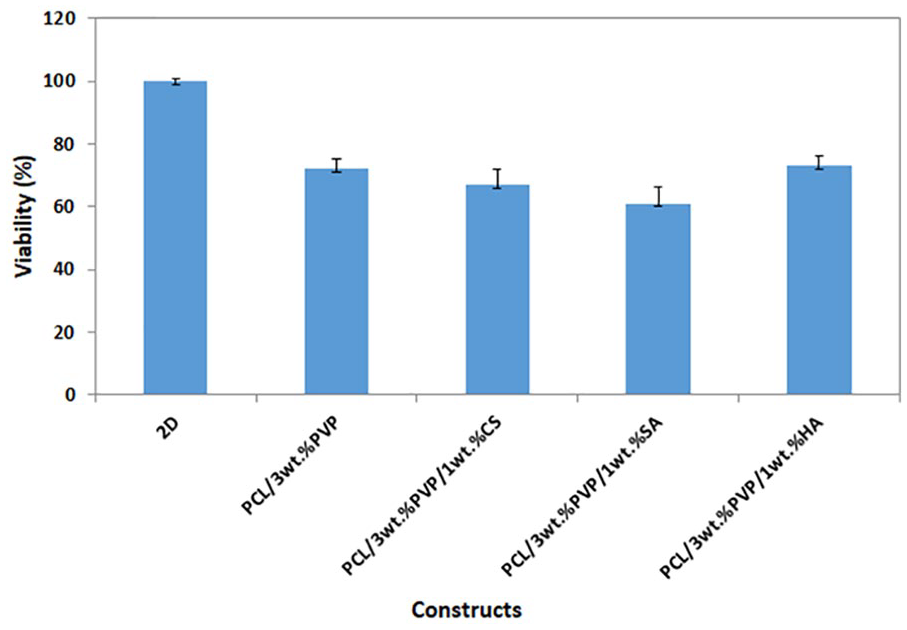
Cell viability graph of all constructs with the control group (2D cells).
Cell Attachment
SEM analysis was performed to investigate the MSCs’ attachment and morphology on PCL/3wt.%PVP, PCL/3wt.%PVP/1wt.%CS, PCL/3wt.%PVP/1wt.%SA, and PCL/3wt.%PVP/1wt.%HA compared with the acellular structure ( Fig. 8a ). According to the results obtained by SEM, it was possible to observe the cells attached to the surface of scaffolds. Cell-seeded scaffolds demonstrated the presence of cellular networks for all scaffolds. This network can be associated with the extracellular matrix attached to the scaffolds. Figure 8b-h demonstrates the formation of some colonies of cells attached between the scaffolds. With the results obtained by SEM, it can be easily said that MSCs can attach and spread on all the scaffolds.36,37 Biocompatibility of the structures with MSCs can be considered as candidate and reference combinations in the differentiation of the stem cells into chondrocytes for further studies.
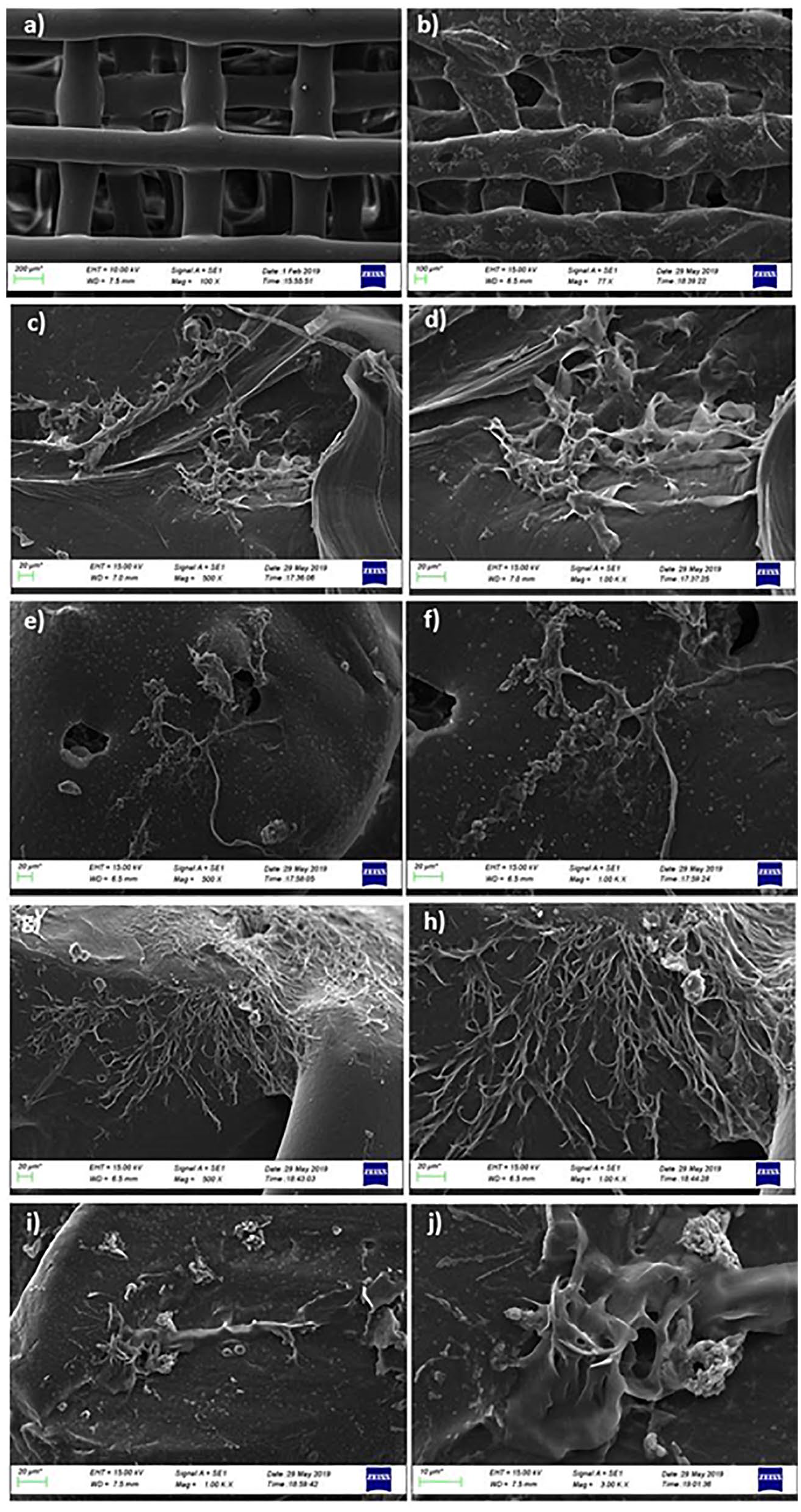
SEM images of PCL/3wt.%PVP (
Conclusions
In this study, PCL and PVP were combined to form the main component of the constructs. Chitosan, hyaluronic acid, and sodium alginate were integrated with this base construct separately in order to find the ideal correlations between them for advancing the biological alternatives that can restore the damaged tissue. 3D printing is the rapid and versatile technique that can provide the required geometry of the diseased tissues to mimic the exact structure of the tissues with adjustable porosity. Replacing diseased tissue is possible by mimicking the extracellular matrix structure. According to optimization results, it was found that the structure of the constructs with a 60% infill rate had a suitable and uniform pore distribution. With this infill rate, other composites were printed keeping this value fixed. The compressive strength values of the constructs increased with different additive polymers. The swelling ratios of the PCL/3wt.%PVP have been enhanced with the addition of the HA and also HA added composites had the highest viability value compared with the others. As a result,
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not required for this article.
Informed Consent
Informed consent was not required for this article.
