Abstract
Objective
The objective of this study was to compare the effects of hyperosmolar sodium (Na+), lithium (Li+) and potassium (K+) on catabolic and inflammatory osteoarthritis (OA) markers and sulfated glycosaminoglycan (sGAG) loss in TNF-α-stimulated cartilage explants.
Methods
Explants from bovine stifle joints were stimulated with TNF-α for 1 day to induce cartilage degradation followed by supplementation with 50 mM potassium chloride (KCl), 50 mM lithium chloride (LiCl), 50 mM sodium chloride (NaCl), or 100 nM dexamethasone for an additional 6 days. We assessed the effect of TNF-α stimulation and hyperosmolar ionic treatment on sGAG loss and expression of OA-associated proteins: ADAMTS-5, COX-2, MMP-1, MMP-13, and VEGF.
Results
TNF-α treatment increased sGAG loss (P < 0.001) and expression of COX-2 (P = 0.018), MMP-13 (P < 0.001), and VEGF (P = 0.017) relative to unstimulated controls. Relative to activated controls, LiCl and dexamethasone treatment attenuated sGAG loss (P = 0.008 and P = 0.042, respectively) and expression of MMP-13 (P = 0.005 and P = 0.036, respectively). In contrast, KCl treatment exacerbated sGAG loss (P = 0.032) and MMP-1 protein expression (P = 0.010). NaCl treatment, however, did not alter sGAG loss or expression of OA-related proteins. Comparing LiCl and KCl treatment shows a potent reduction (P < 0.05) in catabolic and inflammatory mediators following LiCl treatment.
Conclusion
These results suggest that these ionic species elicit varying responses in TNF-α-stimulated explants. Cumulatively, these findings support additional studies of hyperosmolar ionic solutions for potential development of novel intraarticular injections targeting OA.
Introduction
Osteoarthritis (OA) is a degenerative joint disorder characterized by degradation and loss of articular cartilage, remodeling of subchondral bone and chronic inflammation of the joint.1,2 As the most common joint disorder in the United States, OA’s prevalence has doubled since the mid-20th century, with numbers forecast to reach 25% of the U.S. adult population by 2030.1,3-6 The disease inception and progression depend on a multitude of factors such as joint injury, aging, and genetics. Although attention has been focused toward mitigating the symptoms of the disease through pain management and maintenance of function, there remains a lack of treatments available to halt or reverse the observed degeneration.7,8
Recently, synovial inflammation has been recognized to play an integral role in OA progression.8-10 Synovial inflammation is characterized by the production of proinflammatory mediators, including tumor necrosis factor-alpha (TNF-α) and interleukin-1beta (IL-1β), which alter chondrocyte differentiation and phenotype.11-15 Specifically, chondrocytes produce excess catabolic factors such as matrix metalloproteinases (e.g., MMP-1 and MMP-13), and a disintegrin and metalloprotease with thrombospondin domains (e.g., ADAMTS-5), which result in the degradation of collagen-II and aggrecan, respectively.12,13,16 Additionally, the presence of these proinflammatory mediators can induce chondrocytes to secrete inflammatory cytokines and growth factors (e.g., interleukin-6 [IL-6], vascular endothelial growth factor [VEGF]) and to release cyclooxygenase-2 (COX-2). These factors have been implicated to further contribute to the inflammatory environment and OA severity within the joint.17-20
The release of inflammatory cytokines and extracellular matrix (ECM) degradation proteins are believed to exacerbate synovial inflammation, leading to a repeated cycle of increased inflammation and cartilage degradation. 9 To target inflammation, current treatment options for OA include the administration of nonsteroidal anti-inflammatory drugs (NSAIDS), which have been shown to inhibit COX-2 activity.21-23 Long-term use of NSAIDS, however, can cause cardiovascular problems, and liver and kidney damage. 24 Alternatively, hyaluronic acid (HA) intra-articular (IA) injections have been utilized as another treatment option due to the ability of HA to inhibit MMP/ADAMTS activity and to exhibit anti-inflammatory properties in vitro through counteracting the effects of TNF-α and IL-1β.25,26 The use of HA IA injections, however, requires additional follow-up procedures and has demonstrated variable efficacy in patients.27,28 Despite the role of inflammation in OA, these therapies only provide temporary symptomatic and pain relief, and do not stop or reverse disease progression. 11 Thus, therapeutic targeting of inflammation in OA needs to be coupled with disease-modifying ability for restoration of joint structure.
Several approaches involving the use of hyperosmolar irrigation solutions have been investigated and validated for exhibiting chondroprotective effects and limiting posttraumatic OA development in vivo and in vitro.29-32 Ionic solutions are hypothesized to influence cell behavior through changes in osmolarity, membrane potential and through the interaction of the ions with each other and other biomolecules.33-37 For instance, we have previously demonstrated the capacity of hyperosmolar potassium (K+) to attenuate catabolic and inflammatory cytokine production relative to a non-ionic hyperosmolar control in an in vitro model of OA. 38 Likewise, a number of studies have extensively characterized the anti-inflammatory capacity of lithium (Li+) and sodium (Na+) in vitro and in vivo.29,39-42 Pertaining to OA, specifically, work by Hui et al. 43 and Minashima et al. 44 demonstrated the ability of Li+ to reduce expression of cartilage catabolic markers and mitigate sulfated glycosaminoglycan (sGAG) loss in human articular chondrocytes, and bovine and murine cartilage explants, respectively. Furthermore, Eltawil et al. 29 and others demonstrated how increasing osmolarity of Na+-based irrigation solutions can reduce chondrocyte death in a cartilage injury model. 45 Thus, ionic solutions at hyperosmolar concentrations may present a means for mitigating inflammation and cartilage degradation in OA. With this in mind, we aim to expand on our previous work utilizing hyperosmolar K+ by investigating its efficacy in reducing sGAG loss and molecular OA features in an explant model of cartilage degradation.
In the present study, we sought to compare the effects of hyperosmolar ionic Na+, K+, and Li+ solutions in a TNF-α-induced cartilage degradation explant model. The synthetic glucocorticoid, dexamethasone, was utilized for comparison due to its established effects in alleviating inflammation and sGAG loss in cartilage explants.46-49 In brief, we harvested cartilage explants from bovine stifle joints and cultured them in the presence of the proinflammatory mediator TNF-α for 1 day, followed by supplementation with dexamethasone, or hyperosmolar sodium chloride (NaCl), potassium chloride (KCl) or lithium chloride (LiCl) for 6 additional days. To first evaluate our model’s capacity to recapitulate key OA phenotypic markers, we first assessed sGAG loss induced by TNF-α activation and subsequently evaluated the efficacy of NaCl, KCl, LiCl, or dexamethasone in mitigating any observed effects. We then used immunohistochemical analysis to examine changes in OA-associated catabolic and inflammatory proteins following the applied treatments.
Materials and Methods
Bovine Cartilage Explant Culture
Discs of articular cartilage were harvested from bovine stifle joints acquired from N = 4 donors younger than 30 months at New York Custom Processing, LLC. Macroscopically healthy discs were aseptically collected from the nonweightbearing regions of the cartilage at the distal femur using disposable 4-mm sterile punches (Miltex). The harvested discs were then briefly rinsed with Dulbecco’s phosphate buffered saline (DPBS; Corning) supplemented with 1% antibiotic/antimycotic solution (10,000 IU/mL penicillin, 10,000 µg/mL streptomycin, and 25 μg/mL amphotericin; Life Technologies). Explants from each joint’s condyle or a random assortment of explants from one donor joint were assigned as an individual experimental replicate (n) for a total of n = 9. These were placed one per well in a 12-well plate containing culture medium (CM) (Dulbecco’s modified essential medium (DMEM; Corning, 2 mL/well) supplemented with 50 µg/mL of
For explant collection, cartilage discs were cut in half, and weights of both pieces were recorded. One half of each disc was digested in 200 µL papain buffer (125 μg/mL papain [Sigma-Aldrich], 5 mM cysteine HCl, 5 mM EDTA [ethylenediaminetetraacetic acid], and 100 mM Na2HPO4 in distilled water) for sGAG content measurement. The other half was fixed in 10% neutral buffered formalin (Fisher Scientific) and preserved in optimum cutting temperature compound (OCT; Tissue-Tek) at −20 °C for cryosectioning and immunohistochemistry for key markers associated with OA.
Measurement of sGAG Release
The sGAG released into culture medium from each cartilage disc as well as the amount remaining within each cartilage disc at the end of the culture period were measured using the dimethylmethylene blue (DMMB, Sigma) assay. Briefly, medium aliquots and corresponding cartilage digests were diluted with blank medium at 1:2 and 1:100, respectively. Following this, 50 μL of diluted sample solution was added into a 96-well plate, mixed with 250 μL of DMMB buffer (prepared according to previously published protocol 50 ), and absorbance was measured at 525 and 540 nm. sGAG content was calculated using the difference in absorbance values relative to a standard curve prepared from chondroitin sulfate B sodium salt (Sigma) at a concentration range from 80 to 0 mg/mL. Fractional sGAG loss over the period of culture was calculated by dividing the accumulated sGAG content from the medium samples by the total sGAG per sample. Values for accumulated sGAG medium content and total sGAG content per explant weight are presented in Supplemental Table 1.
Histological Analyses
Fixed explant samples embedded in OCT compound were cut into 10 μm transverse sections using a cryomicrotome (Leica) and mounted onto glass slides. Staining protocols were carried out at room temperature, unless stated otherwise. For each antibody or histological stain, staining was conducted for all joint sections in a set simultaneously, allowing sections within each set to be semiquantitatively compared.
Immunohistochemical Staining
Standard immunostaining protocols were employed to stain sections for OA-specific markers and to visualize their relative expression and localized distribution within the cartilage tissue. In brief, following rehydration in 10% neutral-buffered formalin (Fisher Scientific), sections were incubated with a 3% hydrogen peroxide solution for 10 minutes to block endogenous peroxidases. The sections were then pretreated with a 250 U/mL hyaluronidase (Sigma) solution in PBS for 1 hour at 37 °C, unless stated otherwise (Supplemental Table 2). To prevent nonspecific interactions, the sections were then blocked using an Avidin-Biotin kit (Biocare Medical) followed by a 10-minute exposure to Terminator (Biocare Medical). Primary antibodies for OA-specific markers (Supplemental Table 2) were diluted in PBS containing 3% bovine serum albumin (BSA; Fisher Scientific) and 1% Tween 20 (Fisher BioReagents) and applied for 1 hour. Bound primary antibody was visualized after successive incubations with a biotinylated secondary antibody (Biocare Medical) for 30 minutes and a horseradish peroxidase (HRP)-conjugate (Biocare Medical) for 20 minutes, followed by the application of the chromogen 3,3-diaminobenzidine (Vector Laboratories). Sections were then dehydrated and mounted. Stained sections were imaged using an Axiovert microscope (Zeiss) at 10× magnification and assembled into mosaics for semiquantitative image analysis.
Safranin-O Staining
Sections were first rehydrated in 10% neutral-buffered formalin (Fisher Scientific), and then rinsed with a 1% acetic acid solution for 10 to 15 seconds. Subsequently, 100 μL of 0.1% safranin-O solution was added to each section and incubated at room temperature for 5 minutes. Sections were dehydrated and mounted. Stained sections were imaged using an Axiovert microscope (Zeiss) at 10× magnification.
Semiquantitative Image Analysis
Assembled mosaic images with intact layered structure and minimum background signal were chosen as representative images, and semiquantification of relative protein expression was performed by 2 blinded observers. 51 In brief, for each section, a staining intensity of the overall section was recorded by each observer on a scale of 0 to 3, with 0 representing no staining and 3 representing the highest staining intensity for that antibody within a set. The intensity values were then scaled by dividing the observed intensity of each section by 3. Following quantification of each stained set, average staining intensity for a given antibody was calculated by averaging the scaled values across sets and observers.
Statistical Analysis
All data are reported as the mean ± standard error of mean. Sample means were compared using SPSS software (version 26.0). Homogeneity of variance was confirmed using the Levene’s statistic. Experimental means were compared using either an unpaired t test or post hoc testing. For experimental means that assume homogeneity of variance, comparison was done using 1-way analysis of variance (ANOVA) followed by a Tukey honestly significant difference post hoc test. For means that violated homogeneity of variance, comparison of means was conducted using a Welch’s ANOVA followed by Games-Howell post hoc test. Statistical significance was determined at P < 0.05.
Results
TNF-α Promotes sGAG Loss and Catabolic/Inflammatory Marker Expression in Bovine Cartilage Explants
In the present study, exposure to proinflammatory mediator TNF-α was utilized to induce cartilage degradation, a primary hallmark of OA. In accordance with previous literature,
52
treatment of bovine cartilage explants with TNF-α induced a ~1.9-fold increase in sGAG loss following 7 days of culture compared with untreated controls (
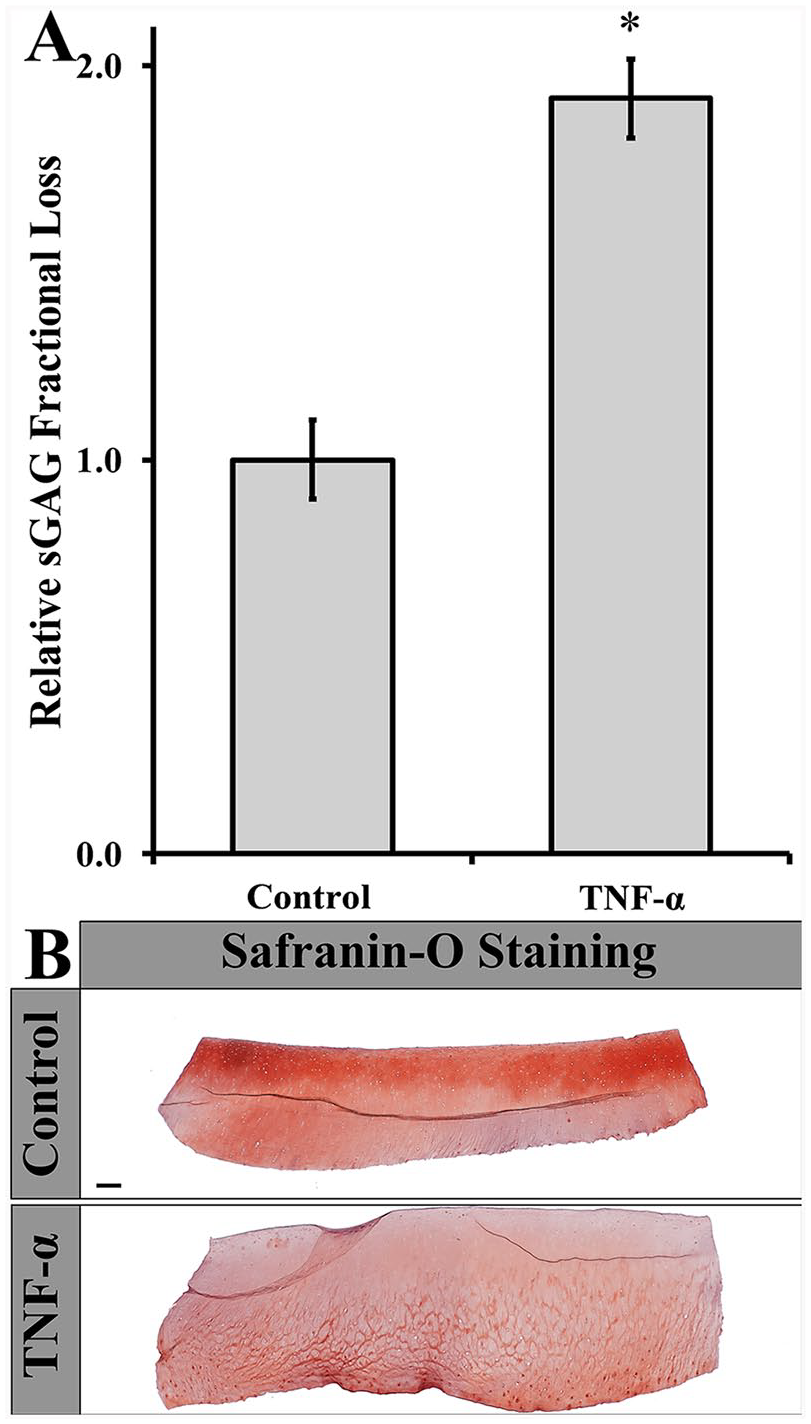
(
Furthermore, we assessed TNF-α-induced shifts in catabolic and inflammatory OA markers following 7 days of culture using immunohistochemical analysis. Representative immunostaining images are shown in Supplemental Figure 1. Stimulation of the cartilage explants with TNF-α increased the expression of COX-2 (1.8-fold, P = 0.018), MMP-13 (2.4-fold, P < 0.001), and VEGF (2.2-fold, P = 0.017), which are known to be elevated in OA (
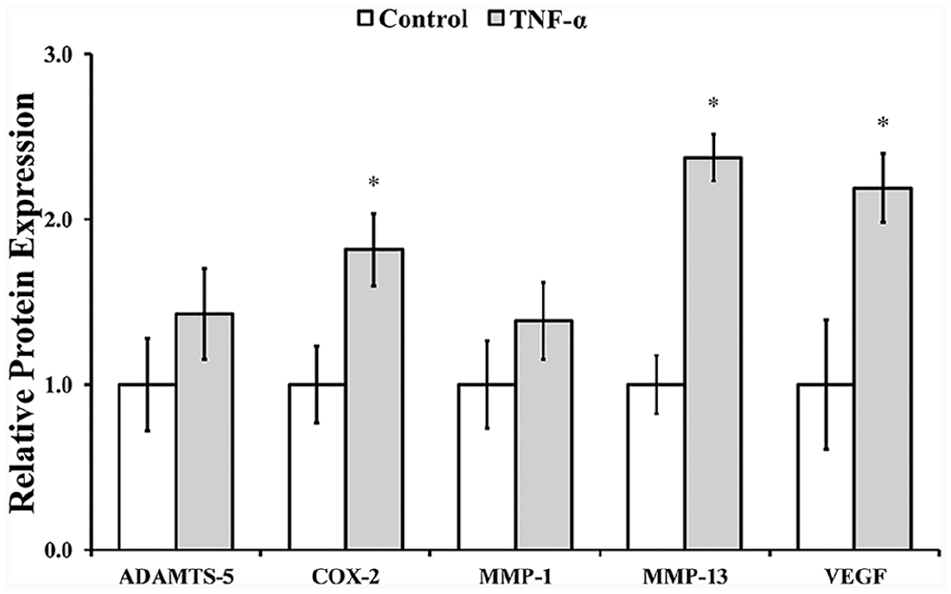
Relative protein levels of ADAMTS-5, COX-2, MMP-1, MMP-13, and VEGF expressed by cartilage explants following 25 ng/mL TNF-α stimulation for 7 days. *Denotes difference relative to control obtained by t test (P < 0.05). Error bars correspond to the standard error of the mean (n = 5-6). TNF-α, tumor necrosis factor-α; ADAMTS-5, a disintegrin and metalloprotease with thrombospondin domains; COX-2, cyclo-oxygenase-2; MMP, matrix metalloproteinase; VEGF, vascular endothelial growth factor.
Hyperosmolar Ion Solutions Modulate TNF-α-Induced sGAG Loss
Given that Na+, K+, and Li+ have shown chondroprotective effects in vitro, we investigated the effects of hyperosmolar Na+, K+, and Li+ solutions on TNF-α mediated sGAG loss using our model system.29,31,43,48 All measures were compared to the clinical control dexamethasone. Treatment of TNF-α-stimulated cartilage explants with a 50 mM KCl supplemented medium resulted in a ~1.4-fold increase in sGAG loss (
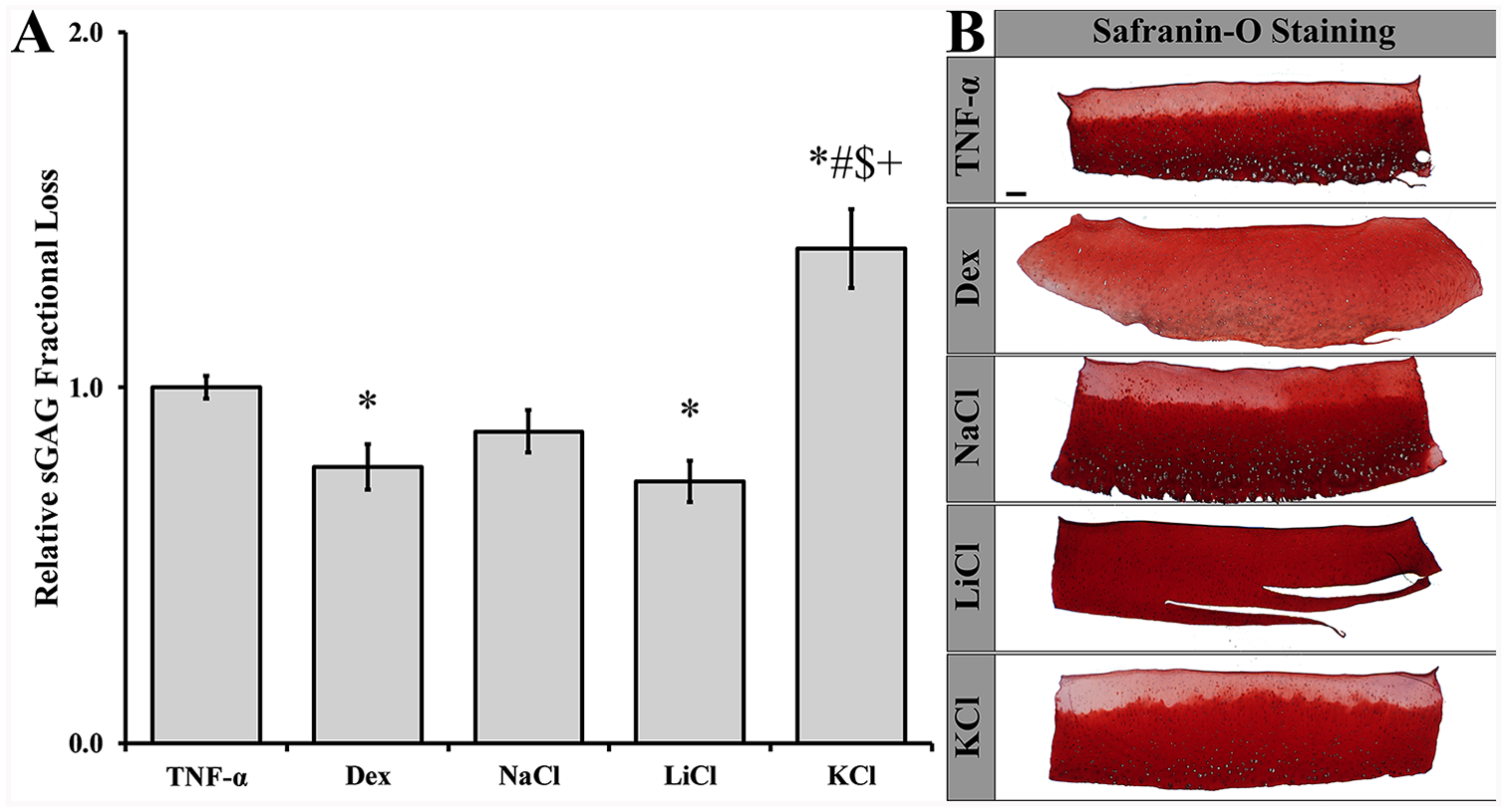
(
Hyperosmolar Ion Solutions Effect on TNF-α Stimulated OA Features
The capacity of NaCl, LiCl, or KCl to modulate changes in OA-associated catabolic and inflammatory markers was assessed—using immunohistochemical analysis—following 6 days of treatment. Representative immunostaining images are shown in Supplemental Figure 2. Relative to the TNF-α-stimulated explants, MMP-1 levels were elevated following KCl treatment (~1.6-fold; P = 0.010;
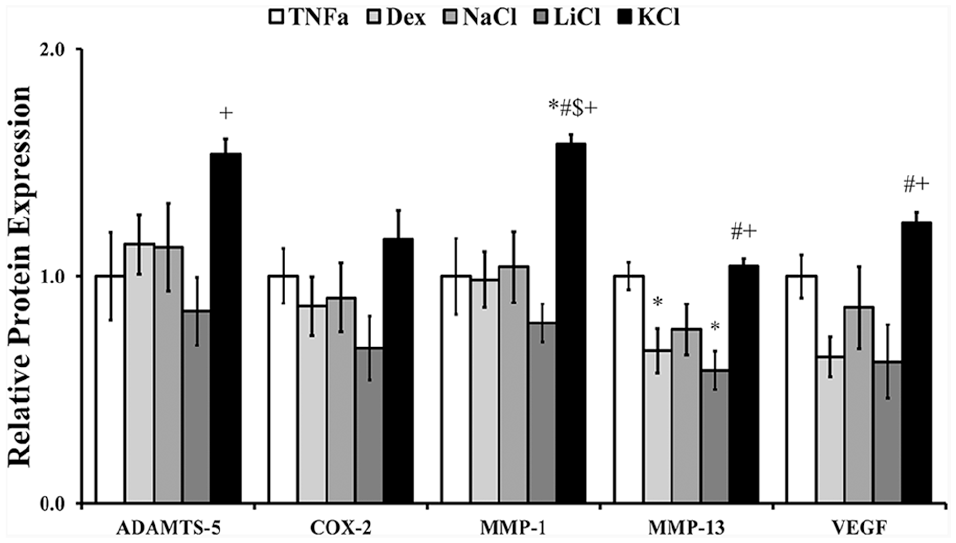
Relative protein levels of ADAMTS-5, COX-2, MMP-1, MMP-13, and VEGF expressed by TNF-α-stimulated cartilage explants following 6-day treatment with activation medium supplemented with dexamethasone (Dex; 100 nM), NaCl (50 mM), LiCl (50 mM) or KCl (50 mM). *, #, $, + denote statistical difference relative to TNF-α, Dex, NaCl, and LiCl, respectively, obtained by Tukey post hoc test (P < 0.05). Error bars correspond to the standard error of the mean (n = 5-6). TNF-α, tumor necrosis factor-α; ADAMTS-5, a disintegrin and metalloprotease with thrombospondin domains; COX-2, cyclo-oxygenase-2; MMP, matrix metalloproteinase; VEGF, vascular endothelial growth factor.
On the other hand, the expression of the ECM degradation protein, MMP-13, was significantly decreased following treatment with dexamethasone (~0.7-fold, P = 0.036) and LiCl (~0.6-fold, P = 0.005) relative to stimulated controls, while other markers (ADAMTS-5, COX-2, IL-6, MMP-1, and VEGF) remained unchanged by the 6 day treatment. Although LiCl treatment appeared to support decreased expression of COX-2 (~0.7-fold) this apparent difference fell below statistical significance. Interestingly, NaCl treatment did not seem to alter sGAG fractional loss or catabolic and inflammatory protein expression relative to stimulated explants. Cumulatively, however, these data suggest the following: (1) the ionic content of hyperosmolar solutions have varying effects on the OA phenotype, (2) hyperosmolar KCl—contrary to previous in vitro results 38 —increases TNF-α-mediated sGAG loss and expression of catabolic and inflammatory markers, and (3) hyperosmolar LiCl mitigates cartilage degradation and the expression of important OA markers, similar to previously published results.39,43,44
Discussion
The objective of this study was to compare the effects of Na+, K+, and Li+ hyperosmolar solutions on cartilage degradation and catabolic protein expression in a TNF-α-mediated cartilage degradation explant model. The effects of these solutions were compared to dexamethasone (100 nM) for its previously established role in attenuating inflammation and mitigating sGAG loss in cartilage.46-49 In our assessment, we measured sGAG loss in cartilage due to TNF-α treatment, followed by evaluation of the impact of NaCl, KCl, LiCl or dexamethasone in altering the observed effects. We then used immunohistochemistry to evaluate changes in cartilage catabolic protein and inflammatory marker expression arising from TNF-α activation and subsequent treatment with KCl, NaCl, LiCl, or dexamethasone.
The progression of OA is manifested by an increase in sGAG loss, collagen degradation and expression of many catabolic and inflammatory proteins.2,12 These attributes, which are associated with cartilage degradation, can be simulated in vitro through treatment with inflammatory cytokines, such as TNF-α.52-55 Stimulating bovine cartilage explants with TNF-α displayed features found in OA such as increased sGAG loss (
Treatments utilizing dexamethasone and Li+ have been widely studied in multiple in vitro and in vivo models that recapitulate OA symptoms.39,43,44,47,61 Aligning with our current results, Lu et al. 48 demonstrated the capacity of dexamethasone to mitigate sGAG loss in TNF-α stimulated bovine cartilage explants. Furthermore, Richardson and Dodge, 62 and Huebner et al. 46 have shown reduced MMP-13 gene expression following dexamethasone treatment in TNF-α-stimulated equine chondrocytes and rabbits in a posttraumatic OA model, respectively. Similarly, Hui et al. 43 demonstrated LiCl (10 mM) treatment for 14 days mitigated bovine cartilage degradation and MMP production in both IL-1β- and TNF-α-mediated cartilage loss. Our results offer support to the potential of Li+ in reducing cartilage damage and catabolic protein expression.
While our previous in vitro work 38 indicated hyperosmolar K+ was effective in reducing expression of catabolic proteins, herein KCl treatment exacerbated sGAG loss and expression of ADAMTS-5 and MMP-1 in TNF-α-stimulated cartilage explants. We hypothesize that this discrepancy is the result of multiple variables including the type of model used. Specifically, our previous model utilized osteoarthritic chondrocytes encapsulated within poly(ethylene glycol) hydrogels, which do not fully recapitulate the natural microenvironment of cartilage tissue. Using cartilage explants, however, supports chondrocytes in their natural microenvironment with a preestablished ECM. 63
Ionic solutions are hypothesized to influence cell behavior through changes in osmolarity, membrane potential and through the interaction of the ions with each other and other biomolecules.33-37 The present data obtained for NaCl, LiCl and KCl suggest that more than hyperosmolarity is at play and that these ionic species elicit different responses in cartilage explants. As it relates to NaCl, we observed no beneficial effects relating to mitigating sGAG loss and expression of catabolic and inflammatory proteins. This is in partial support of Eltawil et al.’s investigation where no differences were found in inflammatory cytokine expression following treatment with 300 or 600 mOsm saline in a cartilage injury model. 29 Our observations, however, diverge from their report which suggested improved cartilage repair following hyperosmolar saline treatment. This discrepancy may be due to the in vivo nature of Eltawil et al.’s work as well as their use of sucrose to increase osmolarity of saline without alteration of its electrolyte concentrations. 29 On the other hand, prior investigations by Amin et al. and others have alluded to the detrimental effects of calcium and sodium ions on chondrocyte health and inflammation, respectively.30,64-66 To elucidate the effects of these ionic species further, future work will aim to test multiple ionic concentrations/doses as well as whether different inflammatory environments (e.g., TNF-α, IL-1β, or a combination of the two) alter the response to hyperosmolar ionic solutions. Additionally, important future work will aim to unravel the mechanisms involved in the observed effects as they relate to ionic identity and osmolarity of solution.
Conclusions
In summary, our results compared the effect of hyperosmolar Na+, Li+, and K+ solutions on sGAG loss and catabolic/inflammatory protein expression in TNF-α stimulated bovine cartilage explants. Li+ attenuated sGAG loss and catabolic protein expression relative to stimulated controls and K+. In contrast, Na+ exhibited no changes in sGAG loss and catabolic protein expression in stimulated explants. Additionally, relative to the clinically used synthetic corticosteroid dexamethasone, LiCl performed equally in attenuating the observed OA features in our explant model. Therefore, future studies will be needed to expand on the utility of LiCl for use as an IA injection for OA treatment.
Supplemental Material
Cartilage_Explant_Supporting_Information_Revision2_Final_Blinded_for_Review – Supplemental material for Hyperosmolar Ionic Solutions Modulate Inflammatory Phenotype and sGAG Loss in a Cartilage Explant Model
Supplemental material, Cartilage_Explant_Supporting_Information_Revision2_Final_Blinded_for_Review for Hyperosmolar Ionic Solutions Modulate Inflammatory Phenotype and sGAG Loss in a Cartilage Explant Model by Ahmad S. Arabiyat, Hongyu Chen, Josh Erndt-Marino, Katie Burkhard, Lisa Scola, Allison Fleck, Leo Q. Wan and Mariah S. Hahn in CARTILAGE
Footnotes
Supplemental Material
Acknowledgments and Funding
The authors would like to acknowledge funding sources from the National Institutes of Health NIA R03AG064550 and NIA R03AG056168 to M.S.H. The authors would also like to acknowledge Timothy Kamaldinov, Jhair Avila-Quiroga and Nicole Ragone for their contribution toward immunohistochemical analysis of cartilage and imaging.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JEM and MSH have a patent related to this work (US Patent App. 16/285,348, 2019).
Ethical Approval
Ethical approval was not sought for the present study because the bovine stifle joints were obtained from a third party vendor (New York Custom Processing, LLC).
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because it did not directly involve the use of live animals. Live animal handling was conducted by the third party vendor from which the bovine stifle joints were obtained (New York Custom Processing, LLC).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
