Abstract
Objective
To evaluate differences in pro-inflammatory and degradative mediator production from osteoarthritic knee articular cartilage explants treated with a hyperosmolar saline solution supplemented with anti-inflammatory components (
Design
Full-thickness 6 mm articular cartilage explants (n = 12/patient) were created from femoral condyle and tibial plateau samples collected from patients who received knee arthroplasty. One explant half was treated for 3 hours with hyperosmolar saline (600 mOsm/L) supplemented with anti-inflammatory components and the corresponding half with normal saline (308 mOsm/L). Explants were cultured for 3 days and then collected for biomarker analyses. Media biomarker concentrations were normalized to the wet weight of the tissue (mg) and were analyzed by a paired t test with significance set at P < 0.05.
Results
Cartilage was collected from 9 females and 2 males (mean age = 68 years). Concentrations of MCP-1 (P < 0.001), IL-8 (P = 0.03), GRO-α (P = 0.02), MMP-1 (P < 0.001), MMP-2 (P < 0.001), and MMP-3 (P < 0.001) were significantly lower in explant halves treated with the enhanced hyperosmolar solution. When considering only those cartilage explants in the top tercile of tissue metabolism, IL-6 (P = 0.005), IL-8 (P = 0.0001), MCP-1 (P < 0.001), GRO-α (P = 0.0003), MMP-1 (P < 0.001), MMP-2 (P < 0.001), MMP-3 (P < 0.001), and GAG expression (P = 0.0001) was significantly lower in cartilage explant halves treated with the enhanced hyperosmolar solution.
Conclusions
Treatment of cartilage explants with a hyperosmolar saline arthroscopic irrigation solution supplemented with anti-inflammatory components was associated with significant decreases in inflammatory and degradative mediator production and mitigation of proteoglycan loss.
Keywords
Introduction
During arthroscopic procedures, a constant flow of irrigation solution is utilized to distend the joint and support visualization of intraarticular structures. Hyperosmolar synovial fluid (404 mOsm/L; pH 7.7) is diluted and displaced by an isotonic irrigation fluid such as normal saline (308 mOsm/L; pH 5) or lactated Ringer’s solution (273 mOsm/L; pH 6.5).1,2 Chondrocytes are sensitive to fluctuations in osmolarity, temperature, and pH; this transition may influence the articular cartilage extracellular matrix composition by altering cytoskeletal organization, gene expression, protein synthesis, and calcium signaling.2-6 Thus, there is strong interest in developing and validating solutions that can maintain and protect the joint, rather than disrupt it.3,7-12
Following an acute injury, there is a shift in the biochemical environment within the joint.13-18 Inflammatory mediators, including prostaglandin E2 (PGE2), monocyte chemoattractant protein-1 (MCP-1), interleukin 6 (IL-6), interleukin 8 (IL-8), and growth-regulated protein alpha (GRO-α) are upregulated and promote cartilage and glycosaminoglycan (GAG) degradation through the induction of mediators such as matrix metalloproteinases (MMPs).19,20 These changes are most often seen following mechanical trauma; however, recent studies suggest that arthroscopic interventions are associated with a high rate of iatrogenic cartilage injuries.21,22 As such, strategies for alleviating arthroscopy-related contributions to inflammatory and degradative processes are critical for best practice.
Recent evidence suggests hyperosmolar saline may function to minimize chondrocyte death following mechanical trauma, contribute to diminished perioperative pain and swelling, and indirectly inhibit the production of inflammatory and degradative mediators by cartilage and meniscus explants.3,8-10,23-25 Consequently, the addition of anti-inflammatory components to a hyperosmolar solution may further augment joint health and recovery following arthroscopic surgery. Therefore, we sought to develop a hyperosmolar solution that could further mitigate inflammation and degradation following arthroscopic surgery.
The purpose of the present study was to evaluate differences in pro-inflammatory and degradative mediator production from osteoarthritic knee articular cartilage explants treated with a hyperosmolar saline solution supplemented with anti-inflammatory components or normal saline using an in vitro model for knee arthroscopy. It was theorized that cartilage explants treated with the supplemented anti-inflammatory solution would produce significantly lower levels of inflammatory and degradative mediators and release decreased levels of extracellular matrix components into the media compared to explants treated with normal saline.
Methods
Articular Cartilage Tissue Collection
With institutional review board approval and patient informed consent, osteoarthritic femoral condyle and tibial plateau articular cartilage was recovered from adult patients undergoing total knee arthroplasty (TKA) at our institution. Resected osteochondral tissues that would otherwise be discarded were collected aseptically in the operative suite and subsequently taken to an in-hospital laboratory for processing. Full-thickness articular cartilage was removed from the bone aseptically with the use of a scalpel blade. Six-millimeter cartilage explants (n = 132; 12 per patient) were created using a dermal punch. Explants were created from areas of the resected articular surfaces with Outerbridge grade 0-3 changes as assessed by the attending orthopedic surgeon and the orthopedic resident-researcher to ensure that areas of the joint with varying metabolic profiles were included to mimic the clinical scenario most closely.
In Vitro Split Tissue Knee Arthroscopy Model
Explants were aseptically sectioned in half and then placed into individual wells of a 24-well tissue culture plate. One half of each cartilage explant was treated for 3 hours with 2 mL of 0.9% saline (308 mOsm/L) while the corresponding half was treated with 2 mL of a proprietary hyperosmolar saline solution (600 mOsm/L) for the same duration. The hyperosmolar solution was composed of a 1.8% saline solution that was created by adding 165 mL of 7.2% saline to 1 L of 0.9% saline. This solution was then supplemented with 2 mM
Media Analyses
The concentration of PGE2 was determined using the PGE2 Express EIA Kit (PGE2, Cayman Chemical, Ann Arbor, MI). A Human Cytokine/Chemokine Magnetic Bead Panel was used to analyze concentrations of MCP-1, IL-6, IL-8, and GRO-α (Millipore, Billerica, MA). MMP-1, -2, -3, and -13 were assessed via a Luminex magnetic bead multiplex assay (R&D Systems, Inc., Minneapolis, MN). GAG content released to the media was determined using the dimethyl-methylene blue (DMMB) assay. 26
Statistical Analyses
Media concentrations from each explant half were normalized to the wet weight (mg) of each cartilage explant half. Total joint metabolism was assessed by calculating the average normalized biomarker production from all explant halves created from the articular cartilage (n = 12/patient) explant samples. These values were subsequently averaged to calculate the total global biomarker production from all articular cartilage samples. A secondary analysis was performed including only those cartilage explant pairs (n = 44 explant pairs) in the top tercile of biomarker production. Performing both analyses resulted in an evaluation of the total joint activity while also assessing those explants from areas of the joint with a higher metabolic activity. Biomarker production from the respective explant halves were analyzed via t test. Statistical analyses were performed using GraphPad Prism 8 (GraphPad Software, San Diego, CA). A P value <0.05 was deemed to be statistically significant for all analyses.
Results
Resected osteochondral tissues were recovered from femoral condyles and tibial plateaus of 11 patients who presented for TKA. These samples were collected from 9 females and 2 males with a mean age of 68.0 ± 8.0 years at the time of surgery. Patients included in this study had a mean body mass index (BMI) of 34.6 ± 7.3 kg/m2. All patients reported that they were either nonsmokers or had discontinued tobacco use more than 12 months prior to their surgical procedure. None of the patients included in this study had a medical history consistent with inflammatory arthropathy. While a portion of patients included in this study were taking prescribed medications for unrelated conditions (e.g., antihypertensives or antilipidemic agents), none of the patients had used systemic corticosteroids or immune modulators at any time.
Inflammatory Mediators
There were no statistically significant differences between treatment cohorts for PGE2 production from cartilage explants with regard to total biomarker production (P = 0.34) or when considering only those cartilage explants in the top tercile of tissue metabolism (P = 0.14) ( Fig. 1 ).
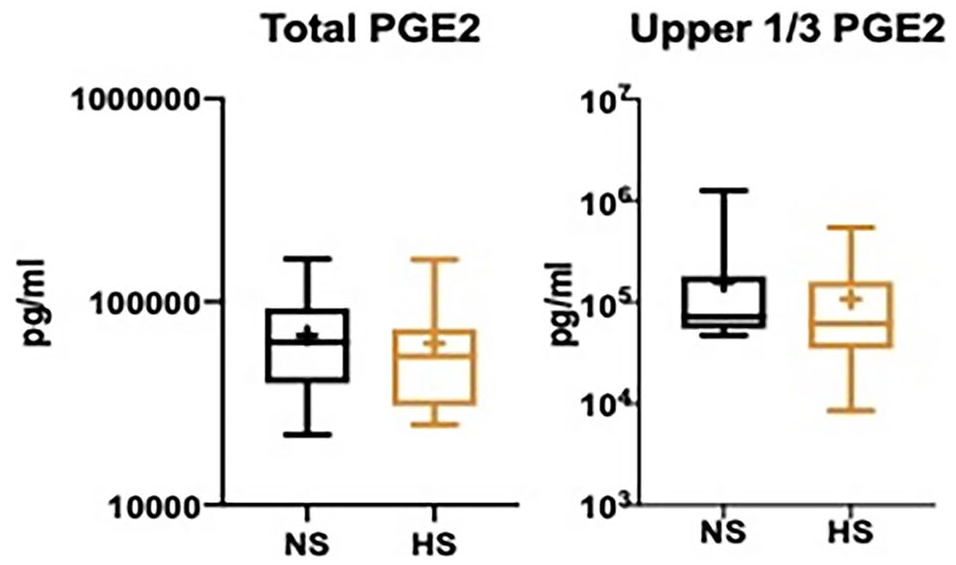
Box-and-whisker plots of articular cartilage normal saline (NS) and enhanced hyperosmolar saline (HS) total and top tercile media concentrations of prostaglandin E2 (PGE2). + Indicates mean value.
Cytokine/Chemokine Production
Total media concentrations of MCP-1 (P < 0.001), IL-8 (P = 0.03), and GRO-α (P = 0.02) were significantly lower in explant halves treated with the enhanced hyperosmolar solution as compared with normal saline controls. In the explant pairs that were more metabolically active, there was significantly lower IL-6 (P = 0.005), IL-8 (P = 0.0001), MCP-1 (P < 0.001), and GRO-α (P = 0.0003) production in cartilage explant halves treated with the enhanced hyperosmolar solution as compared with the normal saline controls ( Fig. 2 ).
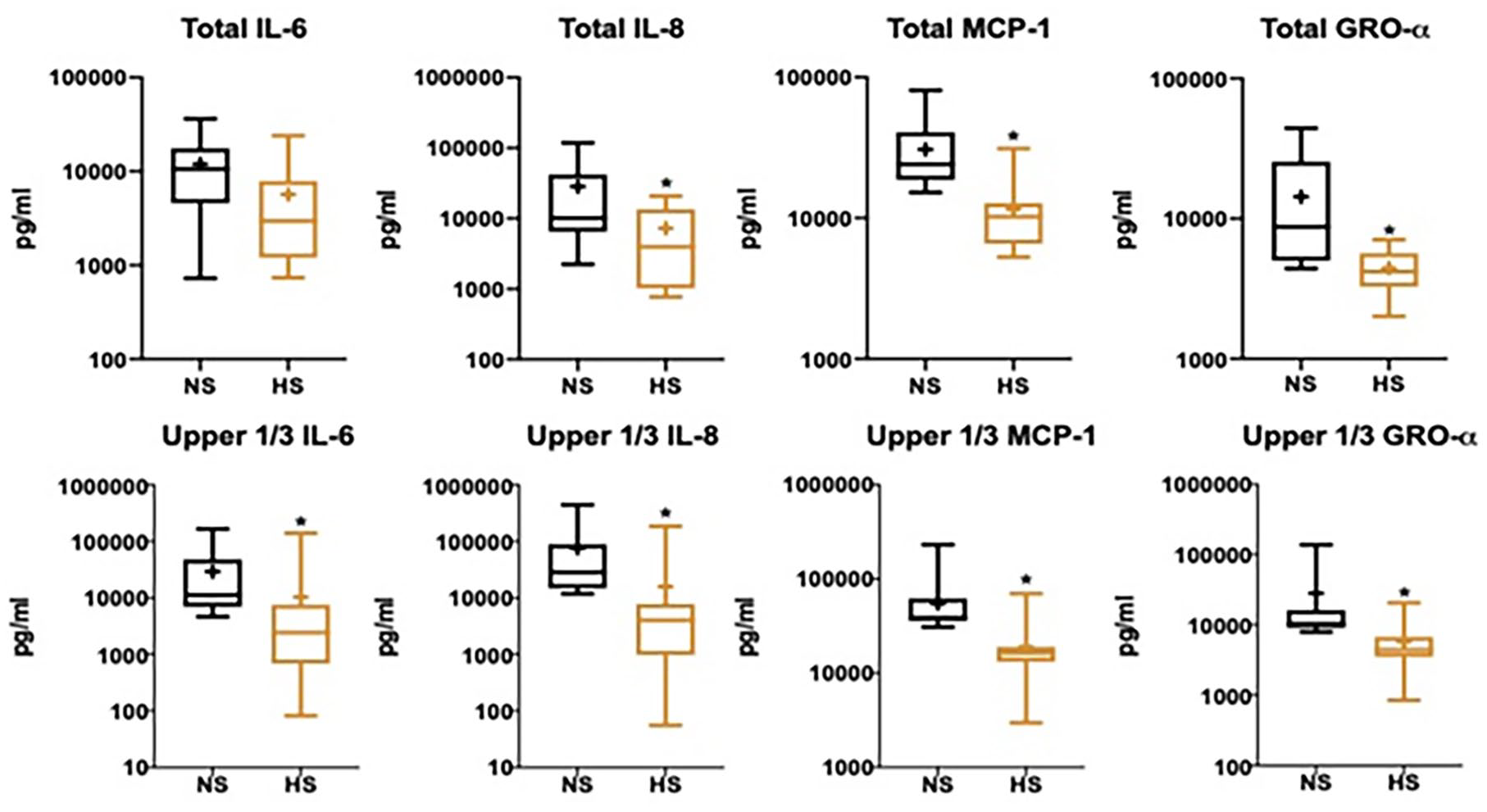
Box-and-whisker plots of articular cartilage normal saline (NS) and enhanced hyperosmolar saline (HS) total and top tercile media concentrations of monocyte chemoattractant protein-1 (MCP-1), interleukin 6 (IL-6), interleukin 8 (IL-8), and growth-regulated protein alpha (GRO-α). + Indicates mean value. * Indicates significant (P < 0.05) difference in biomarker expression.
Degradative Mediators
With regard to degradative mediators, there were significantly lower levels of MMP-1 (P = 0.001), MMP-2 (P = 0.002), and MMP-3 (P < 0.001) production from cartilage explant halves treated with the enhanced hyperosmolar saline solution. The subgroup analysis also revealed significant lower production of MMP-1 (P < 0.001), MMP-2 (P < 0.001), and MMP-3 (P < 0.001) from cartilage explant halves in the enhanced hyperosmolar saline treatment group ( Fig. 3 ).
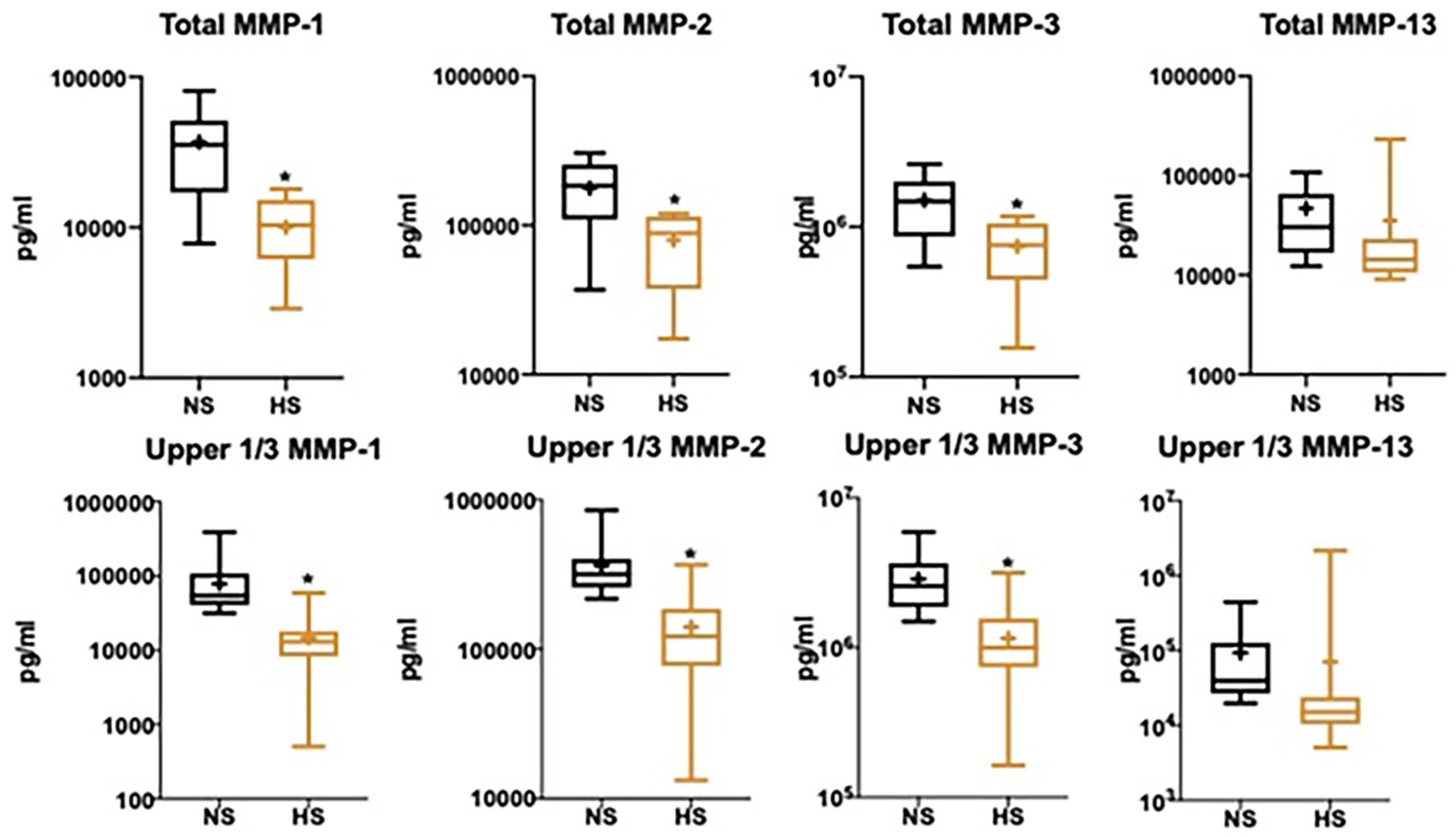
Box-and-whisker plots of articular cartilage normal saline (NS) and enhanced hyperosmolar saline (HS) total and top tercile media concentrations of matrix metalloproteinase-1 (MMP-1), MMP-2, and MMP-3. + Indicates mean value. * Indicates significant (P < 0.05) difference in biomarker expression.
Tissue Extracellular Matrix
Total media GAG concentrations were similar between the 2 treatment cohorts (P = 0.44). However, in the more metabolically active pairs, GAG elution into the media was significantly lower in explant halves treated with the enhanced hyperosmolar solution (P = 0.0001) ( Fig. 4 ).
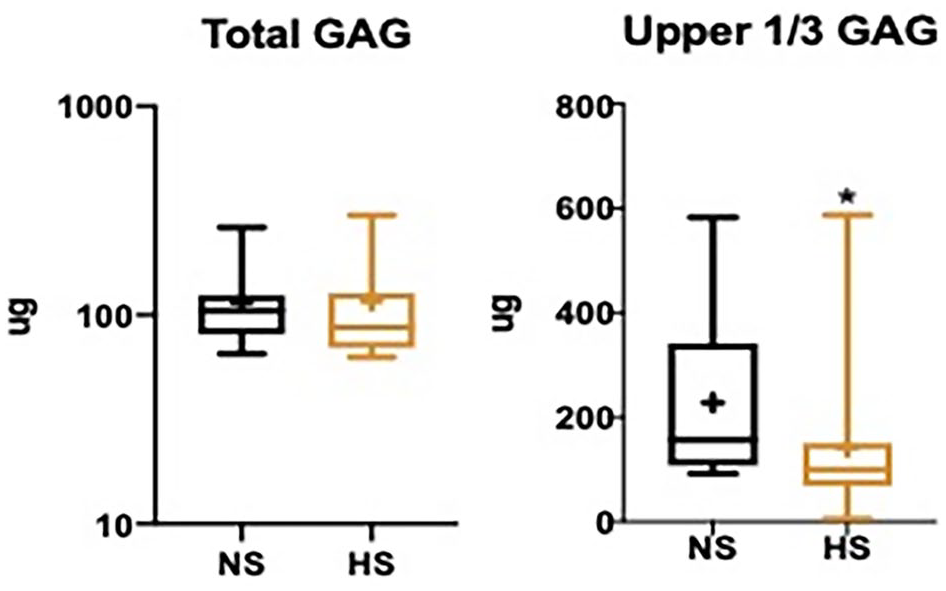
Box-and-whisker plots of articular cartilage normal saline (NS) and enhanced hyperosmolar saline (HS) total and top tercile media concentrations of glycosaminoglycan (GAG). + Indicates mean value. * Indicates significant (P < 0.05) difference in biomarker expression.
Discussion
The principal finding of the present study was that articular cartilage explants treated with a hyperosmolar saline solution supplemented with anti-inflammatory components produced significantly lower levels of pro-inflammatory and degradative mediators without detrimentally impacting extracellular matrix composition compared with paired explants treated with normal saline. Arthroscopic surgery creates an intraarticular environment that significantly alters joint homeostasis. Previous work has demonstrated that targeted modifications to the composition of standard arthroscopic irrigation fluid can positively affect joint health and patient recovery. 24 While further investigation is warranted, the results of the present study suggest that the enhanced hyperosmolar saline solution may potentiate a chondroprotective environment by minimizing articular cartilage inflammatory and degradative mediator production.
Arthroscopic surgery offers a variety of benefits for surgeons and patients; however, it is associated with short- and long-term sequelae that could potentially be minimized by modifying the composition of irrigation fluid. Perioperatively, patients treated with an isotonic irrigation fluid experience a greater degree of fluid extravasation that can lead to increased swelling, pain, functional limitations, and even airway complications following arthroscopic surgery.24,25,37 There are also long-term complications that could potentially be minimized via targeted modifications to arthroscopic irrigation solutions. Patients treated with arthroscopic surgery to address acute or chronic knee pathology often experience cartilage degradation and early-onset OA, which are associated with inflammatory and degradative mediators within the joint.14,38-43 The enhanced hyperosmolar saline solution evaluated in the present study was formulated to address many of the challenges associated with standard isotonic irrigation fluids and further augment the beneficial effects associated with hyperosmolar irrigation fluids. The inclusion of a 1.8% hyperosmolar saline solution, EGCG, and dexamethasone was based on numerous studies that have highlighted the promise of these components.3,8-10,24-33 Therefore, this solution is functionally a hyperosmolar irrigation fluid with additives that impart further anti-inflammatory properties.
Hyperosmolar saline solutions are an alluring alternative to the standard isotonic arthroscopic irrigation fluids. In vitro studies demonstrate that exposure to a hyperosmolar solution minimizes chondrocyte death and proteoglycan elution when compared with isotonic solutions in the setting of mechanical injury.3,8-10 Furthermore, hyperosmolar saline can mitigate select pro-inflammatory and degradative mediator production from knee articular cartilage and meniscus explants in an in vitro model of knee arthroscopy. In vivo studies have also demonstrated positive results. 23 Capito et al. 25 arthroscopically irrigated the shoulders of dogs with either a hyperosmolar saline solution (600 mOsm/L) or normal saline (300 mOsm/L) and demonstrated that the hyperosmolar solution led to decreased shoulder girth from fluid extravasation without affecting glenoid and humeral head chondrocyte viability. In a follow-up study, this research group randomized 50 patients undergoing arthroscopic shoulder surgery to treatment with either a hyperosmolar (593 mOsm/L) or isotonic (273 mOsm/L) arthroscopic irrigation solution. Their findings indicated that patients treated with the hyperosmolar solution had significantly less perioperative weight gain, smaller shoulder girth, and lower immediate visual analog pain scores. 24 Taken together, basic science, preclinical, and clinic studies support hyperosmolar solutions for use as arthroscopic irrigation fluids, such that solution modifications designed to further enhance their effectiveness for mitigating the inflammatory and degradative sequelae of arthroscopic surgery are warranted.
EGCG is a polyphenol present in green tea that exhibits anti-inflammatory activity 25 to 100 times more potent than vitamins C and E. 34 Previous research indicates that EGCG may play roles in modulating inflammatory and degradative mediators associated with cartilage degradation, including nitric oxide, PGE2, IL-6, IL-8, and MMPs.29-33 Leong et al. 30 evaluated the degree to which EGCG could alter the progression of OA and affect OA-associated pain in a mouse model of post traumatic OA and reported that mice treated with EGCG demonstrated less cartilage loss and lower OARSI (Osteoarthritis Research Society International) scores in comparison to those treated with vehicle. Furthermore, articular cartilage from the EGCG mouse cohort exhibited reduced levels of mRNA for pro-inflammatory and degradative mediators including IL-1β, TNF-α, MMP-1, MMP-3, MMP-8, and MMP-13, as well as a reduction in OA-related pain.
Dexamethasone has high affinity for intracellular glucocorticoid receptors and functions to repress the transcription of proinflammatory cytokines, enhance chondrocyte differentiation and proteoglycan synthesis, and reduce glycosaminoglycan loss in injured cartilage.26-28 Previous research suggests that dexamethasone may assist in reducing the acute inflammatory surge that is seen immediately following injury or surgery. Heard et al. 35 evaluated the efficacy of a single intraarticular injection of dexamethasone in mitigating acute postsurgical inflammation and posttraumatic OA in rabbits and reported that the dexamethasone injection was associated with significantly lower mRNA expression of IL-1β, IL-6, and IL-8 and a significantly lower histopathologic severity grades for synovium and lateral compartment articular cartilage. Based on these findings, the authors concluded that dexamethasone may effectively inhibit inflammation and the associated drivers of posttraumatic OA that ensue following joint injury and/or surgery.
In the present study, treatment of articular cartilage explants with a hyperosmolar saline solution enhanced by supplementation with L-glutamine, ascorbic acid, sodium pyruvate, 1 mM EGCG, and dexamethasone was associated with decreased production of IL-6, IL-8, MCP-1, GRO-α, MMP-1, MMP-2, and MMP-3. Previously, cartilage and meniscus explants recovered from patients undergoing knee surgery and treated with a 1.8% hyperosmolar saline arthroscopic irrigation solution demonstrated decreased production of PGE-2, MCP-1, IL-6, IL-8, and MMP-2. 23 Preclinical and clinical studies have validated the safety and efficacy of a hyperosmolar saline arthroscopic irrigation solution.24,25 Taken together, these data suggest that a hyperosmolar saline solution formulated to include anti-inflammatory components may potentiate a chondroprotective environment and enhance postoperative outcomes following arthroscopic surgery, warranting clinical validation studies.
Limitations
There are limitations that should be considered with regard to the present study. The experimental design focused on assessment of production of 10 key mediators and proteoglycan release from articular cartilage explants. While assessment of additional mediators, proteoglycan, collagen, and water content, and material properties of cartilage and other key intra-articular tissues would be informative, the present study was designed to focus on key mechanistic pathways that have been previously reported to be associated with clinical benefits for use of hyperosmolar arthroscopic irrigation solutions.
The solution assessed in the present study contained various compounds that were added to a hyperosmolar saline base. To date, there is no evidence that the components included in this study are detrimental to articular cartilage, however further study is warranted to examine their long-term impact on cartilage health. It is also necessary to directly compare this novel hyperosmolar solution to a standard hyperosmolar saline solution to verify the potential benefits reported in the present study.
Cartilage explants included in the present study were sampled from areas of cartilage with Outerbridge grade 0-3 changes based on gross assessments of cartilage obtained from patients undergoing TKA. This sampling method was designed to allow for inclusion of explants covering the spectrum of pathology typically noted in patients undergoing knee arthroscopy. Articular cartilage was from osteoarthritic knees with many explants classified as Outerbridge grade 2 or 3. In addition, a 3-hour duration of treatment was chosen for the present study. As such, the data reported may be more representative of longer, more complex knee arthroscopies (e.g., anterior cruciate ligament reconstruction with meniscal repair, meniscal transplants, multiligament reconstruction procedures). Therefore, while a full spectrum of cartilage pathology was assessed such that the resultant data have translational relevance, additional studies are necessary to determine the clinical applicability of these data with regard to nonarthritic joints, shorter duration arthroscopic procedures, and long-term effects of the novel arthroscopic irrigation solution. The average BMI of patients included in this study was 34.6 ± 7.3 kg/m2. This is reflective of the average BMI for many patients in this age group across much of the United States. However, it may not reflect the mean BMI of patients in this age group internationally. Future studies may be necessary to validate the findings of this study in patients with a lower mean BMI.
Finally, although statistical significance or desired power was not reached for all analyses, statistical significance was achieved for multiple comparisons between treatment groups for inflammatory and degradative mediators and extracellular matrix components indicating that sample size was effective for hypothesis testing. Type-2 statistical errors may have occurred for some comparisons, but the experimental design and data produced addressed the stated objective.
Conclusions
Treatment of cartilage explants with a hyperosmolar saline arthroscopic irrigation solution supplemented with anti-inflammatory components was associated with significant decreases in inflammatory and degradative mediator production and mitigation of proteoglycan loss. These results suggest that the addition of anti-inflammatory components to a hyperosmolar irrigation solution may promote a chondroprotective environment by minimizing post-surgical inflammation and degradation thereby potentiating joint health and recovery following arthroscopic knee surgery when compared to normal saline at an exposure time of 3 hours in patients with Outerbridge grade 0-3 articular cartilage pathology.
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved and executed under #1208392 from the University of Missouri Institutional Review Board.
Informed Consent
All patients provided informed consent.
Trial Registration
Not applicable.
