Abstract
Objective
Cinnamaldehyde (CA) is an active ingredient of Wenyang Tongluo capsule. This study was performed to investigate the function of CA on human chondrocytes.
Design
Different doses of CA were used to treat C28/I2 cells, which were stimulated by interleukin-1β (IL-1β), and then the viability and apoptosis of the cells were examined by cell counting kit-8 and flow cytometry. Interleukin-6 (IL-6), interleukin-20 (IL-20), and tumor necrosis factor-α (TNF-α) were measured by enzyme-linked immunosorbent assay. Quantitative real-time reverse transcriptase polymerase chain reaction was performed to measure miR-1285-5p, miR-140-5p, IL-20, and high-mobility group box 1 (HMGB1) messenger RNA (mRNA) expression. Western blot assay was performed to detect IL-20, HMGB1, IKBα, phospho-IKBα, IKKα/β, and phospho-IKKα/β expression. Moreover, the relationships between miR-1285-5p and IL-20, as well as miR-140-5p and HMGB1, were validated by dual-luciferase reporter assay.
Results
CA promoted the viability and inhibited the apoptosis of C28/I2 cells stimulated by IL-1β and repressed IL-6, IL-20, and TNF-α levels. CA increased miR-1285-5p and miR-140-5p expression levels. MiR-1285-5p and miR-140-5p promoted the viability and inhibited the apoptosis and inflammation of C28/I2 cells. IL-20 was a target gene of miR-1285-5p, and HMGB1 was a target gene of miR-140-5p. Overexpression of IL-20 or HMGB1 could reverse the effect of CA on C28/I2 cells treated with IL-1β. In addition, HMGB1 increased phospho-IKBα and phospho-IKKα/β expression in IL-1β- and CA-treated C28/I2 cells.
Conclusions
CA protects chondrocytes via regulating miR-1285-5p/IL-20 axis and miR-140-5p/HMGB1/nuclear factor kappa B pathway.
Introduction
Osteoarthritis (OA) is a slowly progressive degenerative joint disease, with an increasing morbidity with aging and a higher morbidity in women than in men. 1 The main clinical symptoms of OA include chronic pain, joint instability, stiffness, joint deformity, and narrowing of the joint space. 2 Articular cartilage is composed of chondrocytes and extracellular matrix, which are rich in collagen and proteoglycan, and chondrocytes are the only cell type in articular cartilage, and chondrocyte apoptosis is positively correlated with the severity of cartilage destruction and matrix degradation. 3 Interleukin-1β (IL-1β) is an important proinflammatory factor that is often used to induce chondrocyte apoptosis and cartilage matrix degradation, thus mimicking OA in vivo and in vitro models. 4 Current clinical pharmacological treatments for OA are mainly analgesics such as nonsteroidal anti-inflammatory drugs, but they only relieve pain, and abuse of medication is common.4,5 Therefore, the development of novel efficient and safe drugs is important for OA treatment.
Cinnamaldehyde (CA), also known as 3-phenylprop-2-enal, is an important bioactive ingredient extracted from Ramulus Cinnamomi and has been found to play a key regulatory role in a variety of diseases. For example, in neurodegenerative diseases such as Alzheimer’s disease and Parkinson’s disease, CA can exert neuroprotective effects by modulating neuroinflammation, inhibiting oxidative stress, and improving synaptic connection. 6 In stress urinary incontinence model, CA can reduce nitric oxide level and nitric oxide synthase level in lipopolysaccharide-induced RAW 264.7 cells in a dose-dependent manner and increase urethral contractility. 7 In an animal model of ulcerative colitis, CA effectively attenuates the symptoms of colitis induced by dextran sulfate sodium salt, including weight loss, shortened colon length, and inflammatory cell infiltration, and CA inhibits the production of proinflammatory factors and NOD-like receptor (NLR) family pyrin domain containing 3 inflammasome in colonic tissues. 8 In addition, several studies have recently reported the anti-inflammatory effects of CA in OA cartilage degeneration.9,10 However, the potential mechanisms of CA in treating OA have not been fully elucidated. In this study, we used IL-1β to induce the injury of human chondrocyte C28/I2 to establish an in vitro model of OA and explored the effects of CA on chondrocyte viability and apoptosis as well as inflammatory responses, and the underlying molecular mechanisms.
Method
Cell Culture and Treatment
Human chondrocyte cell line C28/I2 was purchased from Merck Millipore (Temecula, CA). C28/I2 cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum (Gibco, Carlsbad, CA) and 100 U/ml penicillin and 100 μg/ml streptomycin (Gibco) and incubated in an incubator (5% CO2, 37 °C). When C28/I2 cell fusion reached 70%-80%, the cells were stimulated with 10 ng/ml of IL-1β for 24 hours to construct an in vitro model of OA.
The Preparation of CA
CA (C6H5CH=CHCHO) (molecular weight: 132.16, purity ≥95%) was purchased from Sigma-Aldrich (St. Louis, MO; Cat. No. W228613), and its molecular structure is shown in Figure 1A . CA was dissolved in dimethyl sulfoxide (DMSO; Sigma-Aldrich) and prepared into solutions at concentrations of 5, 10, and 20 μg/ml and stored at −20 °C. When C28/I2 cells reached 70%-80% fusion, the cells were treated with 10 ng/ml of IL-1β and different concentrations of CA (0, 5, 10 and 20 μg/ml) for 24 hours, and the control group was treated with 0.1% DMSO.
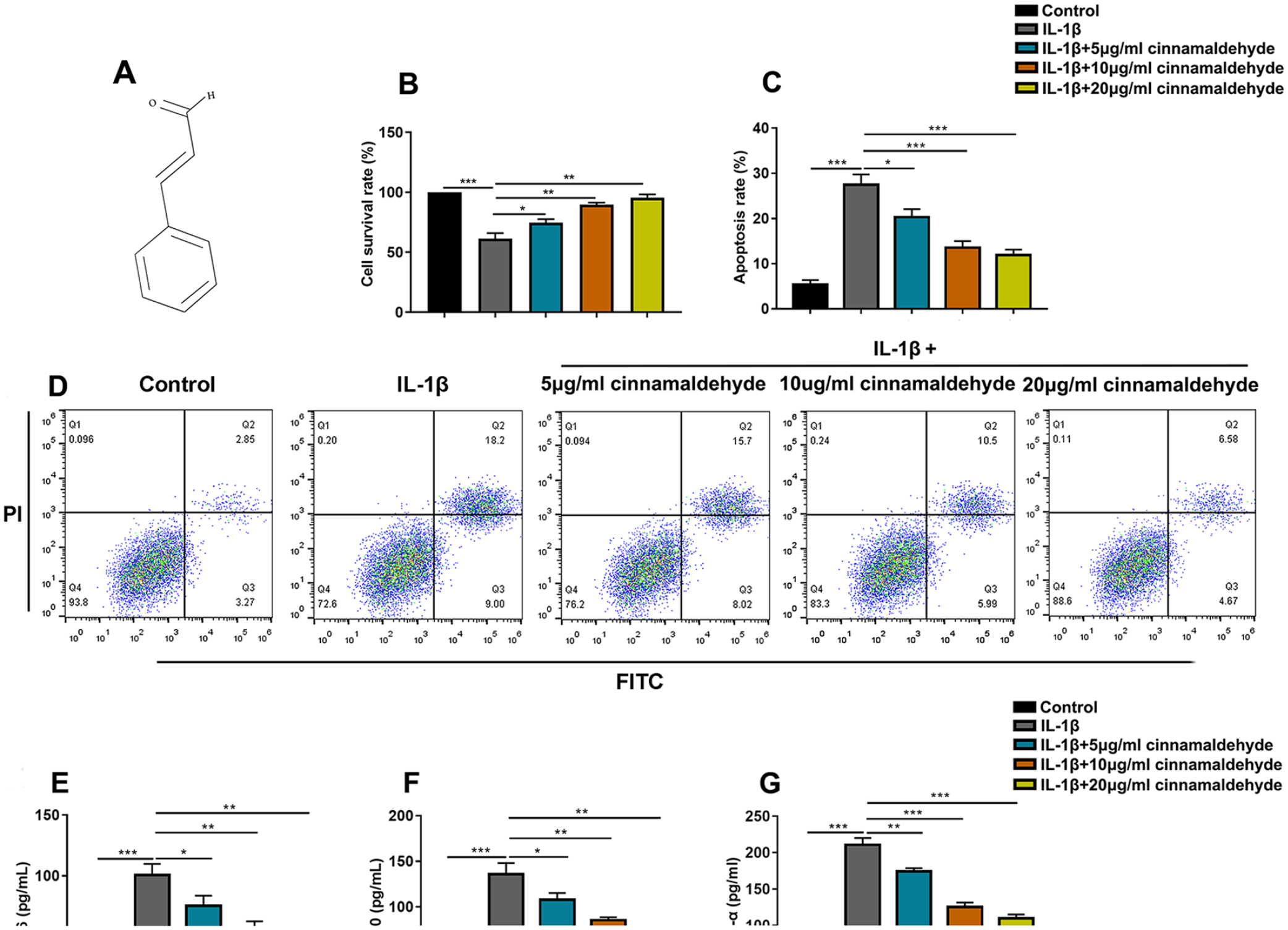
Effect of CA on the viability and apoptosis of C28/I2 cells induced by IL-1β. (
Cell Transfection
C28/I2 cells were inoculated at a density of 2 × 105 cells/ml on 6-well plates and cultured at 37 °C. Interleukin 20 (IL-20) and high-mobility group box 1 (HMGB1) overexpression plasmids, miR-1285-5p mimics, miR-140-5p mimics, and the negative control (miR-NC) were purchased from Genomeditech (Shanghai, China). When the cell fusion reached 60%, the above vectors were transfected into C28/I2 cells using Lipofectamine 2000 reagent (Invitrogen, Carlsbad, CA). Quantitative real-time polymerase chain reaction (qRT-PCR) was performed to verify the transfection efficiency.
qRT-PCR
Total RNA from C28/I2 cells was extracted using TRIzol reagent (Thermo Fisher Scientific, Waltham, MA), according to the manufacturer’s instructions, and RNA was reverse-transcribed using the ABI Reverse Transcription Kit (Applied Biosystems, Foster City, CA). qRT-PCR was then performed using SYBR Green PCR Kit (TaKaRa, Shiga, Japan) on an ABI PRISM7500 Sequence Detection System (Applied Biosystems). U6 small nuclear 1 (U6) and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) normalized gene expression levels using the 2−ΔΔCt method. The primer sequences used in this study are as follows: miR-1285-5p: Forward: 5′-CGCAGGATCTCACTTTGTTG-3′; Reverse: 5′-CAGTGCGTGTCGTGGAGT-3′; miR-140-5p: Forward: 5′-TGCGGCAGTGGTTTTACCCTATG-3′; Reverse: 5′-CCAGTGCAGGGTCCGAGGT-3′; IL-20: Forward: 5′-ATGAAAGCCTCTAGTCTTGCCT-3′; Reverse: 5′-GCCCCGTATCTCAGAAAATCC-3′; HMGB1: Forward: 5′-AAAGCGGACAAGGCCCGTTAT-3′; Reverse: 5′-AAGAGGAAGAAGGCCGAAGGAG-3′; U6: Forward: 5′-CTCGCTTCGGCAGCACA-3′;Reverse: 5′-AACGCTTCACGAATTTGCGT-3′; GAPDH: Forward: 5′-GAAGGTGAAGGTCGGAGTC-3′; Reverse: 5′-GAAGATGGTGATGGGATTTC-3′.
Cell Counting Kit-8 (CCK-8) Assay
CCK-8 kit (Dojindo, Tokyo, Japan) was used to detect the viability of C28/I2 cells. The cells were inoculated in 96-well plates with 5,000 cells per well, followed by the addition of 10 μl of CCK-8 reagent (Beyotime Biotechnology, Shanghai, China) and incubated at 37 °C with 5% CO2 for 2 hours. Optical density values at 450 nm per well were measured using a microplate reader (Molecular Devices, Sunnyvale, CA).
Flow Cytometry Assay
The apoptosis of C28/I2 cells was detected using the Annexin V-FITC and propidium iodide (PI) Apoptosis Detection Kit (BD Pharmingen, San Diego, CA). C28/I2 cells were trypsinized with 0.25% trypsin, centrifuged, and then the cells were collected. The cells were washed twice with phosphate-buffered saline (PBS) containing 5% bull serum albumin and then incubated for 20 minutes with 5 µl of Annexin V-FITC solution and 10 µL of PI solution in dark, and after the cells were washed by PBS, the apoptosis was detected by a flow cytometer (BD Biosciences, San Jose, CA) and the data were analyzed using FlowJo 10.0 software (Tree Star, Ashland, OR) to calculate the apoptosis rate of each group. The “Q2+Q3” cell percentage was the apoptosis rate.
Enzyme-Linked Immunosorbent Assay (ELISA)
C28/I2 cells were placed in 6-well plates, cultured for 48 hours, and then chondrocytes were lysed and the supernatant was collected. The levels of interleukin-6 (IL-6), IL-20 and tumor necrosis factor-α (TNF-α) in C28/I2 cells were measured using the corresponding ELISA kits (R&D Systems, Inc., Minneapolis, MN) according to the manufacturer’s instructions.
Luciferase Reporter Assay
Bioinformatics was used to predict the complementary binding sites between miR-1285-5p and IL-20 3′-untranslated region (3′-UTR), as well as miR-140-5p and HMGB1 3′-UTR, respectively. The sequences of IL-20 3′-UTR were subsequently amplified to construct Psi-CHECK2/IL-20 3′-UTR wild-type reporter plasmids (IL-20-WT) and Psi-CHECK2/ IL-20 3′-UTR mutant reporter plasmids (IL-20-MUT). Similarly, the Psi-CHECK2/HMGB1 3′-UTR wild-type reporter plasmids (HMGB1-WT) and Psi-CHECK2/HMGB1 3′-UTR mutation reporter plasmids (HMGB1-MUT) were constructed. Then, the above luciferase reporter plasmids were co-transfected into C28/I2 cells with miR-NC and miR-1285-5p mimics or miR-NC and miR-140-5p mimics. After 24 hours of transfection, the luciferase activity was detected using the Dual-Luciferase® Reporter Assay System (Promega, Madison, WI). Relative luciferase activity = (Firefly luciferase activity / Renilla luciferase activity) × 100%.
Western Blot Assay
Total proteins were extracted from cells using the radioimmunoprecipitation assay lysis buffer (Beyotime Biotechnology) and quantified using the bicinchoninic acid kit (Sangon Biotech, Shanghai, China). The protein samples were separated using sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to polyvinylidene difluoride membranes (Millipore, Billerica, MA). Subsequently, the membranes were blocked with 5% skimmed milk and then incubated with anti-IL-20 antibody (1:1,000, MA5-26604; Thermo Fisher), anti-HMGB1 antibody (1:1,000, ab190377; Abcam Inc., Cambridge, UK), anti-GAPDH antibody (1: 1,000, ab8245; Abcam Inc.), anti-phospho-IKBα (p-IKBα) antibody (1:1,000, ab133462; Abcam Inc.), anti-IKBα antibody (1:1,000, ab32518; Abcam Inc.), anti-IKKα/β antibody (1:1,000, ab178870; Abcam Inc.), and anti-phospho-IKKα/β antibody (p-IKKα/β; 1:1,000, ab194528; Abcam Inc.) overnight at 4 °C and the membranes were subsequently incubated with horseradish peroxidase–conjugated secondary antibody (1:2,000, ab6702/ab205719; Abcam Inc.) for 2 hours at room temperature. The protein bands were then developed using the ECL luminescence reagent (7Sea PharmTech, Shanghai, China), with GAPDH as the internal reference.
Microarray Analysis
GSE175961 dataset was downloaded from the Gene Expression Omnibus (GEO) database, and the online analysis tool GEO2R (https://www.ncbi.nlm.nih.gov/geo/geo2r) provided by the GEO database was adopted to analyze the differentially expressed microRNAs (miRNAs) in cartilage tissue of OA patients and healthy controls. Log2|fold change| <1 and P < 0.05 were used as the screening criteria.
Venn Diagram
GeneCards database (https://www.genecards.org/) was used to identify the target genes associated with OA. TargetScan database (http://www.targetscan.org/vert_72/) was applied to predict the downstream target genes of miR-1285-5p. StarBase database (https://starbase.sysu.edu.cn/) was used to predict the downstream target genes of miR-140-5p. The related genes of OA were screened from the GeneCards database. And the Venn diagram (http://bioinformatics.psb.ugent.be/webtools/Venn/) was used to plot the overlapping genes between the TargetScan database and the GeneCards database, as well as the StarBase database and the GeneCards database.
Bioinformatics Analysis Based on Annotation, Visualization, and Integrated Discovery (DAVID) Database
DAVID database (https://david.ncifcrf.gov/home.jsp), an online website for gene function annotation and analysis, was used for Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis, with “P < 0.05” as inclusion criteria.
Statistical Analysis
GraphPad Prism 7.0 was used for statistical analysis, and all experiments were conducted 3 times, and the results were expressed as “mean ± standard deviation”; Student’s t test and 1-way analysis of variance (ANOVA) were used for comparisons. P < 0.05 signified statistical significance.
Results
Effect of CA on Proliferation and Apoptosis of C28/I2 Cells Which Were Treated With IL-1β
C28/I2 cells were treated with IL-1β and different doses of CA (0, 5, 10, and 20 μg/ml) for 24 hours, and the cell viability was detected by CCK-8 assay. The results showed a significant decrease in C28/I2 cell survival rate after stimulation with IL-1β compared with the control group, whereas cell viability was significantly promoted after treatment with 5, 10, and 20 μg/ml of CA compared with the IL-1β group in a time- and concentration-dependent manner (
CA Promotes MiR-1285-5p and MiR-140-5p Expression Levels
To investigate the mechanism of CA with the in vitro model of OA, miRNAs differentially expressed in cartilage tissues of OA patients and healthy controls were analyzed with a dataset GSE175961, and miR-1285-5p expression was found to be downregulated (log2fold change = −5.05, P < 0.001) (
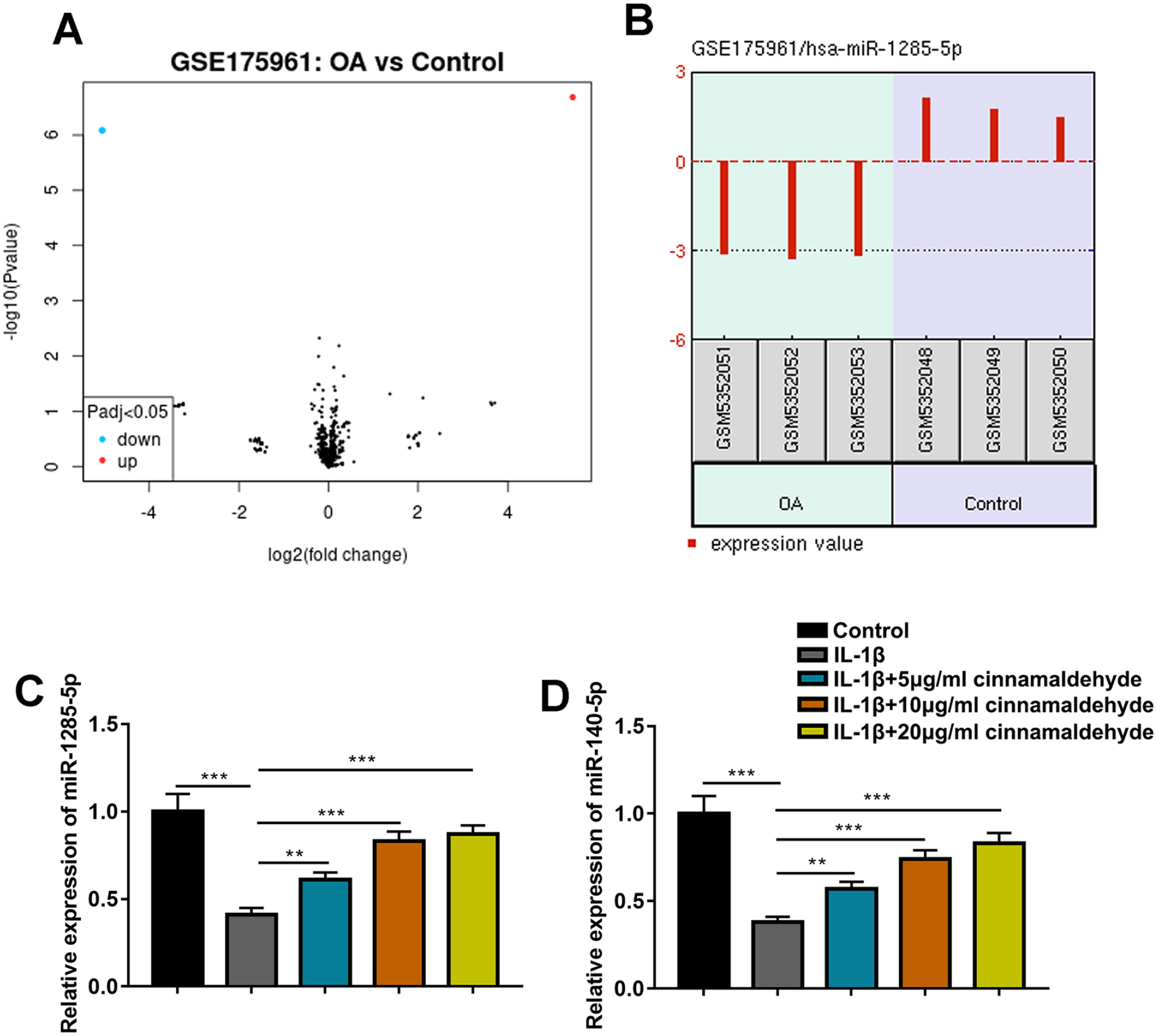
CA promotes miR-1285-5p and miR-140-5p expressions. (
MiR-1285-5p and MiR-140-5p Attenuate IL-1β-Induced Injury of C28/I2 Cells
To explore the function of miR-1285-5p and miR-140-5p in the in vitro model of OA, miR-1285-5p mimics and miR-140-5p mimics and negative control miR-NC were transfected into C28/I2 cells, respectively, to construct miR-1285-5p and miR-140-5p overexpression models, and qRT-PCR validated the successful transfection (
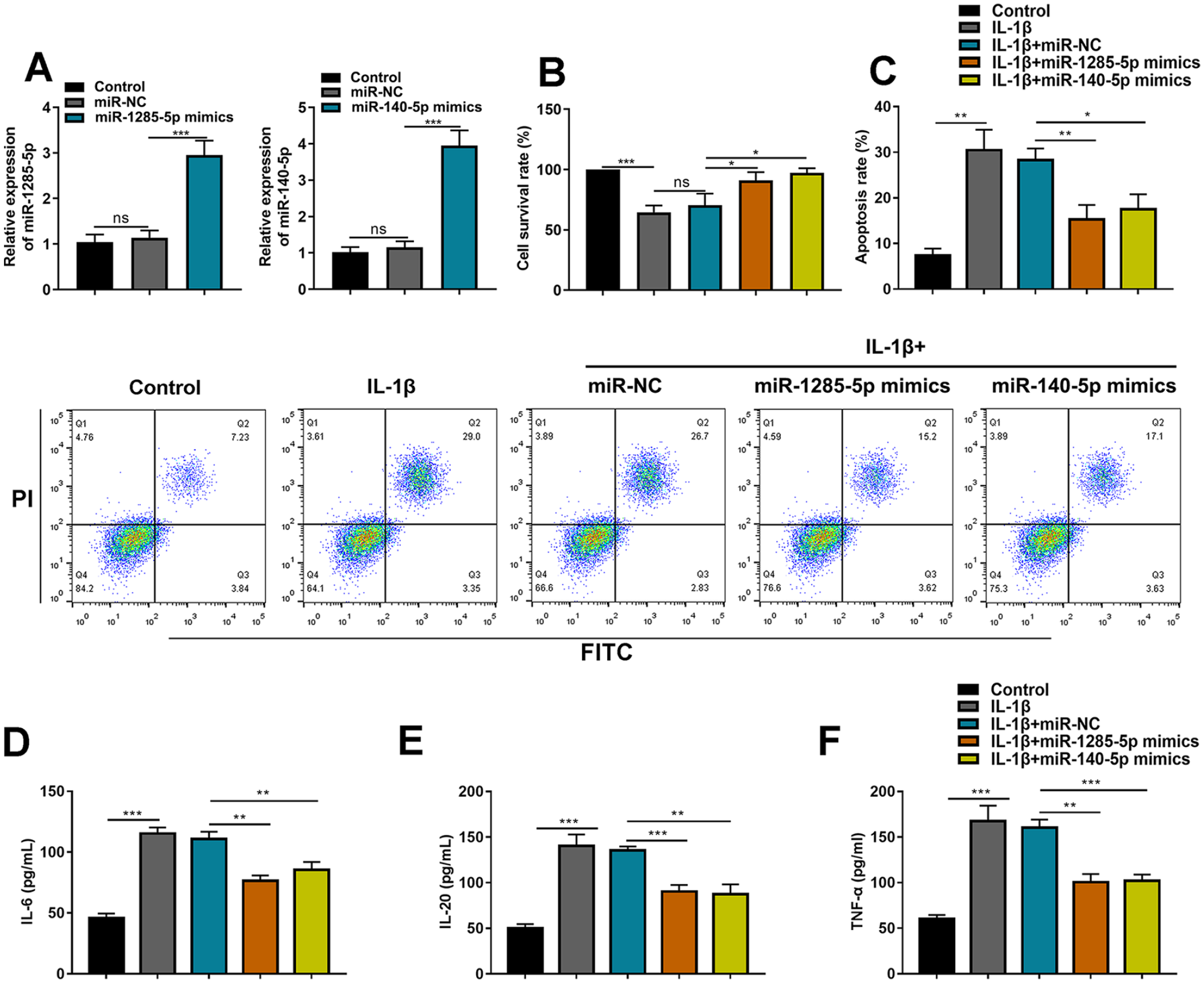
MiR-1285-5p and miR-140-5p attenuate IL-1β-induced injury in C28/I2 cells. (
IL-20 Is a Downstream Target of MiR-1285-5p
To investigate the downstream regulatory mechanism of miR-1285-5p in OA, the GeneCards database was used to analyze genes associated with OA, and the TargetScan database was used to predict the messenger RNAs (mRNAs) containing complementary binding sites to miR-1285-5p, and 452 overlapping genes were screened by plotting a Venn diagram (
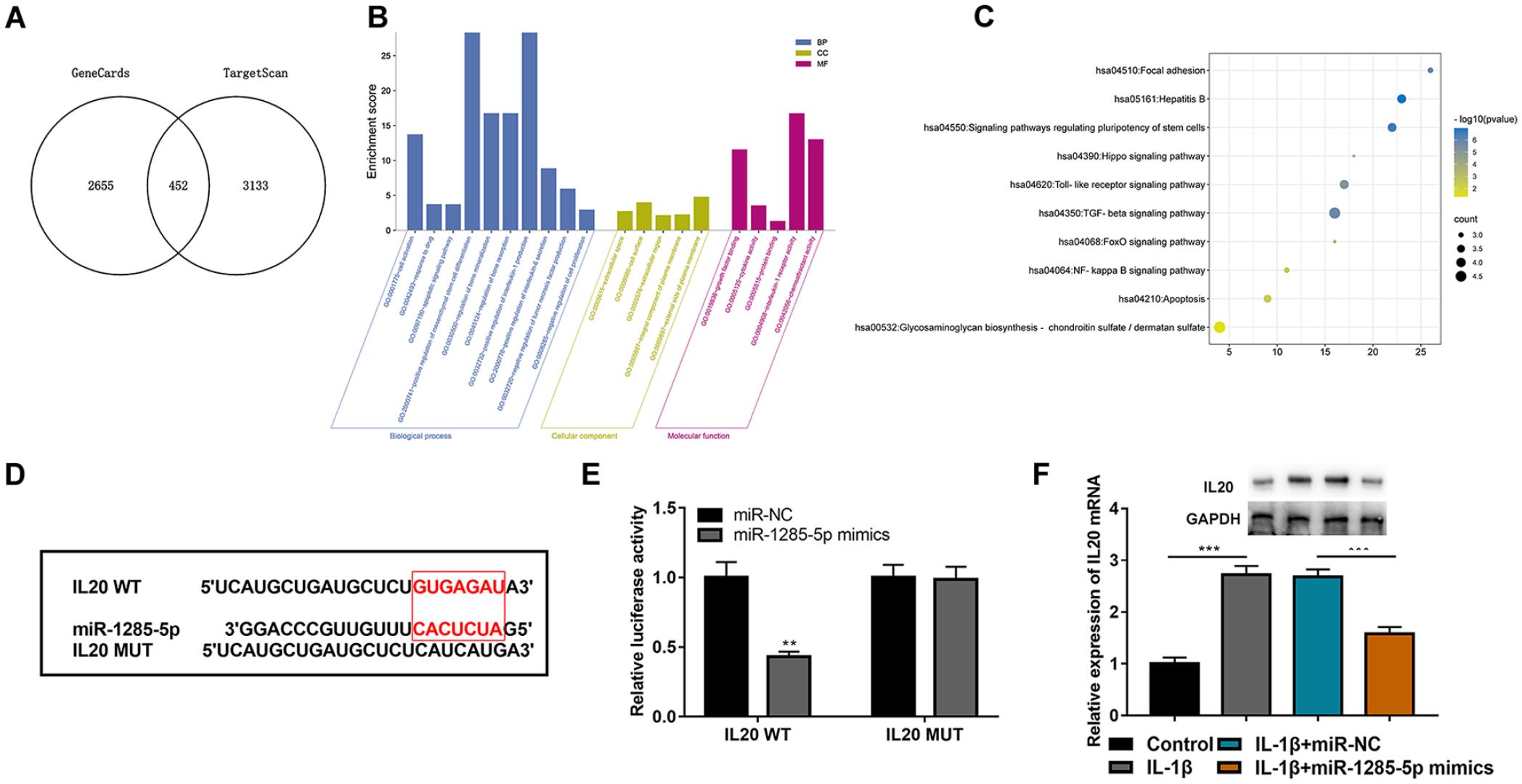
IL-20 is a downstream target of miR-1285-5p. (
HMGB1 Is a Downstream Target of MiR-140-5p
The downstream mechanism of miR-140-5p in OA pathogenesis was also investigated. The OA-related genes from the GeneCards database and the mRNAs containing complementary binding sites with miR-140-5p were plotted by a Venn diagram, and 336 genes were screened out (
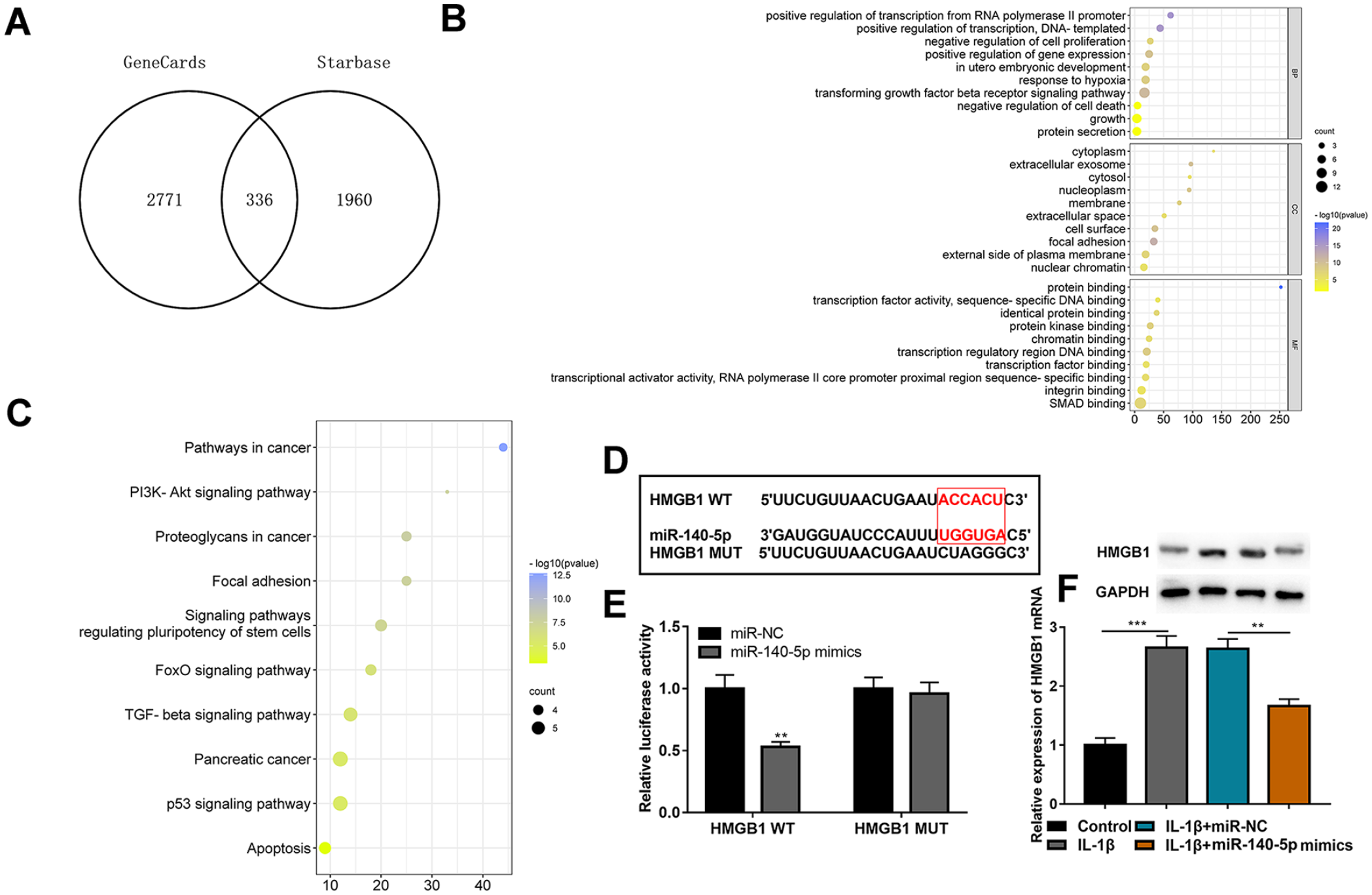
HMGB1 is a downstream target of miR-140-5p. (
CA Attenuates IL-1β-Induced Injury of C28/I2 Cells by Modulating IL-20 and HMGB1
Next, IL-20 and HMGB1 overexpression plasmids were transfected into C28/I2 cells, respectively. Western blot assay indicated that CA could inhibit the expression of IL-20 and HMGB1, and the transfection was successful (
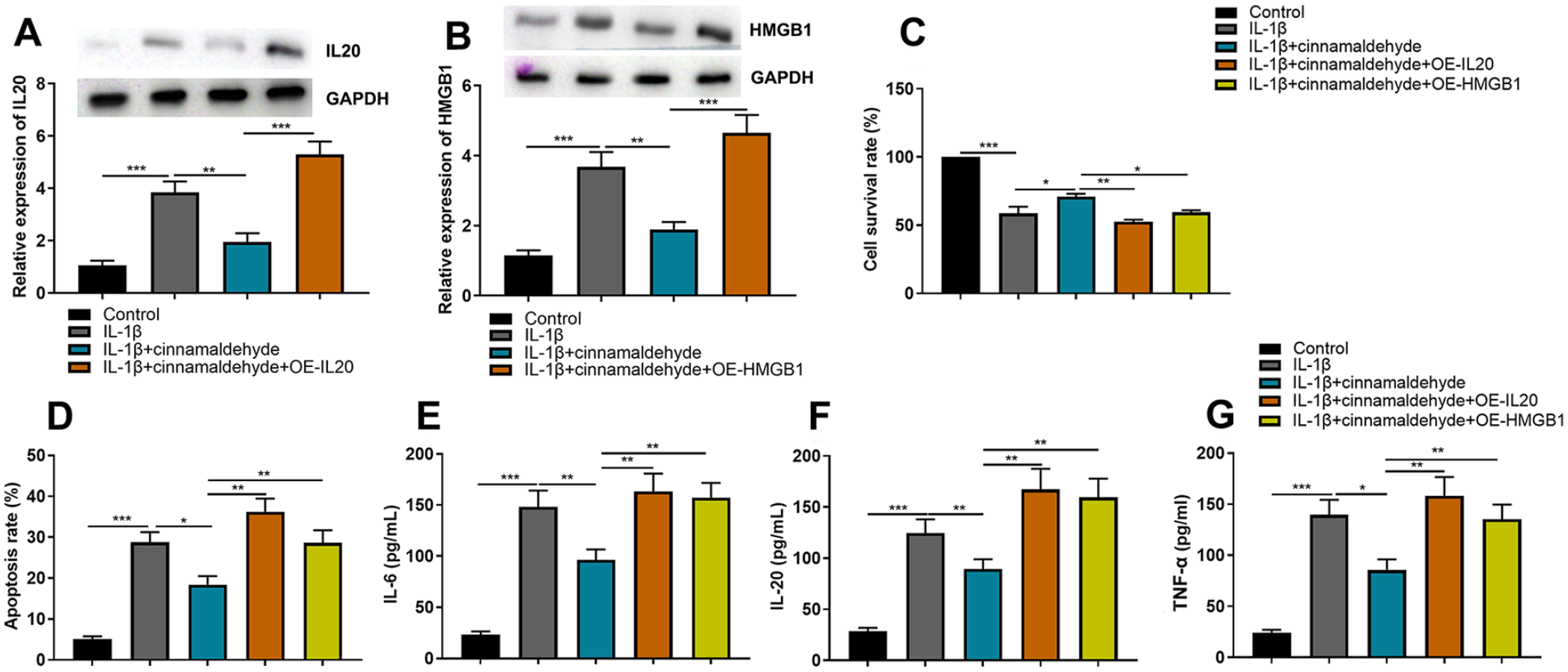
CA attenuates IL-1β-induced injury of C28/I2 cells by regulating IL-20 and HMGB1. (
CA Could Regulate Nuclear Factor-Kappa B (NF-κB) Pathway via HMGB1
In addition, to identify the signaling pathway which was activated by IL-20 and HMGB1 in OA, we detected the expression levels of NF-κB pathway–related proteins. The results showed that HMGB1 overexpression could elevate the expression of p-IKBα and p-IKKα/β which were inhibited by CA; however, IL-20 had no effect on p-IKBα and p-IKKα/β expression (
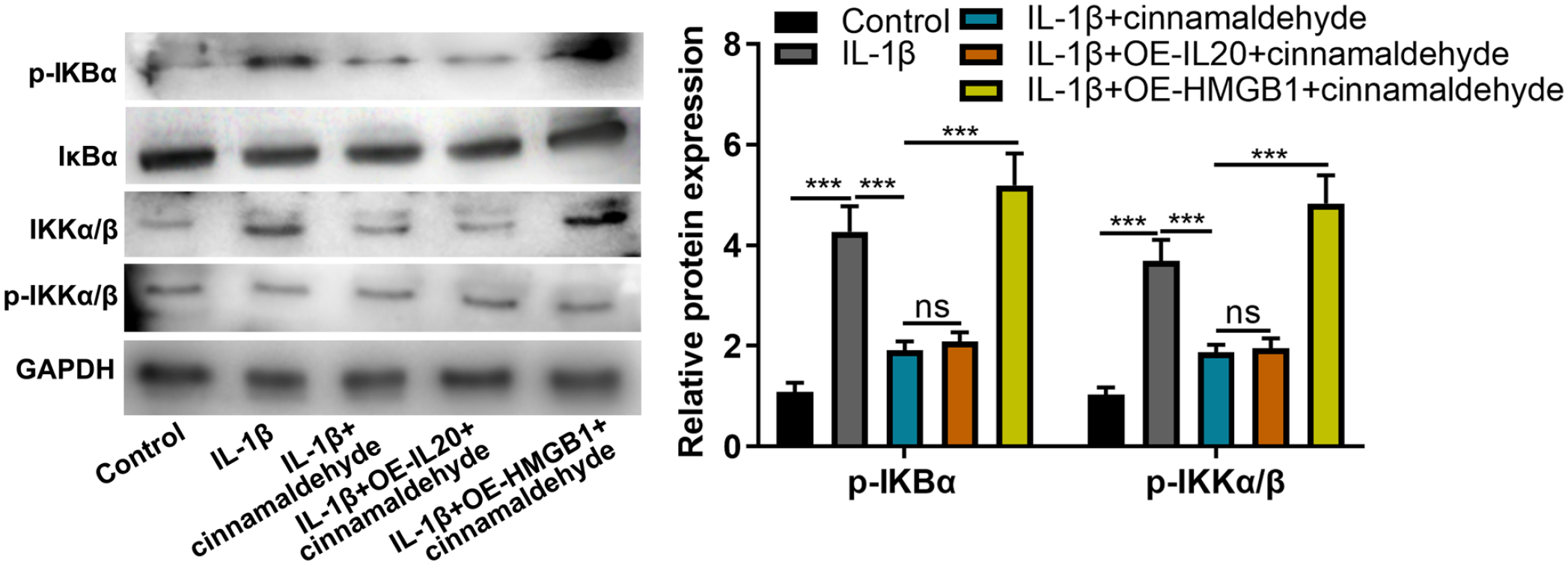
CA could regulate NF-κB pathway by HMGB1. Western blot assay was used to detect IKBα, p-IKBα, p-IKKα/β, and IKKα/β expression. CA = cinnamaldehyde; NF-κB = nuclear factor-kappa B; HMGB1 = high-mobility group box 1; GAPDH = glyceraldehyde-3-phosphate dehydrogenase; OE = overexpression. NS P > 0.05, ***P < 0.001.
Discussion
CA is a natural compound with various pharmacological effects such as antibacterial, anti-inflammatory, and antioxidant effects.11-13 In OA, it was found that trans-cinnamaldehyde (TCA) decreases the protein expression levels of interleukin-8, TNF-α, and prostaglandin E2 in chondrocytes by regulating the PI3K/Akt pathway. 9 It is also found that TCA at a concentration of 10 μg/ml significantly reduces matrix metalloproteinase (MMP)-1, MMP-3, MMP-13, ADAM metallopeptidase with thrombospondin type 1 motif (ADAMTS)-4, and ADAMTS-5 expression levels in the in vitro model of OA induced by IL-1β. Furthermore, TCA significantly reduces cartilage degradation in a rat model of OA and exerts a protective effect on chondrocytes by inhibiting NF-κB and p38-mitogen-activated protein kinase 8 pathways, suggesting that TCA may act as an anti-inflammatory agent for OA. 10 In this work, we found that 5, 10, and 20 μg/ml of CA significantly promoted the viability and inhibited apoptosis and inflammatory responses of C28/I2 cells, which were treated with IL-1β, suggesting a protective effect of CA on IL-1β-induced C28/I2 cells.
MiRNA is a highly conserved endogenous noncoding RNA of about 25 nt in length, which can modulate the expression of target genes by binding to the 3′-UTR of target mRNA, thus affecting cell proliferation, differentiation, apoptosis, senescence, and other biological processes and participating in the pathogenesis and progression of various diseases. 12 A growing number of studies have found that miRNAs are involved in the progression of OA, such as miR-29b-3p, 14 miR-146a, 15 and miR-27a. 16 However, the role of miR-1285-5p in OA is unknown. Reportedly, miR-1285-5p is involved in the pathological processes of a variety of diseases. For example, in non–small cell lung cancer, miR-1285-5p promotes the proliferation, migration, and invasion of non–small cell lung cancer cells by targeting cadherin 1 and Smad4. 17 In breast cancer, miR-1285-5p significantly inhibits the proliferation of breast cancer cells by targeting transmembrane protein 194A. 18 In prostate cancer, miR-1285-5p is a downstream target of hsa_circ_0001206, and transfection of miR-1285-5p mimics partially reverses the inhibitory effects of hsa_circ_0001206 overexpression on the proliferation, migration, and invasion of prostate cancer cells in vitro and tumor growth in vivo. 19 In addition, previous studies have shown that miR-140-5p is involved in the pathological progression of OA. For example, miR-140-5p expression is downregulated in OA cartilage tissue, and downregulation of miR-140-5p significantly reverses the inhibitory effect of knockdown of H19 on chondrocyte matrix degradation and calcification markers. 20 It is also found that miR-140-5p expression is downregulated in cartilage tissues of OA patients, and miR-140-5p overexpression inhibits apoptosis and promotes proliferation and autophagy of human primary chondrocytes by downregulating fucosyltransferase 1. 21 In this work, we found that miR-1285-5p and miR-140-5p were downregulated in IL-1β-induced C28/I2 cells, and overexpression of miR-1285-5p and miR-140-5p promoted the viability of C28/I2 cells and inhibited apoptosis and inflammatory responses. These data support that miR-1285-5p and miR-140-5p are 2 protective factors for chondrocytes in the pathogenesis of OA.
IL-20, a member of the IL-10 family, is a proinflammatory factor that regulates inflammation, angiogenesis, chemotaxis, and apoptosis, and it is involved in the pathogenesis of OA. 22 For example, IL-20 is found to induce the expression of proinflammatory factors and OA-related genes in OA synovial fibroblasts and chondrocytes by activating mitogen-activated protein kinase 1 and mitogen-activated protein kinase 8 signaling, thus promoting OA progression. 23 It is also reported that IL-20 level is elevated in the serum of OA patients and may be involved in the systemic inflammatory response. 24 In this work, we found that IL-20 was a downstream target of miR-1285-5p, and IL-20 expression was upregulated in IL-1β-induced C28/I2 cells, and miR-1285-5p overexpression inhibited IL-20 expression. In addition, HMGB1 is a highly conserved nonhistone DNA-binding protein expressed in the nucleus of eukaryotic cells. 25 In OA, HMGB1 expression is found to be upregulated in mouse chondrocytes treated with lipopolysaccharide, and HMGB1 is a downstream target of miR-410-3p, which targets HMGB1 and regulates chondrocyte apoptosis and inflammation through the NF-κB signaling pathway. 26 It is also found that HMGB1 expression is upregulated in IL-1β-induced C28/I2 and that HMGB1 is a downstream target of miR-140-5p, which inhibits IL-1β-induced inflammation, MMP expression, and apoptosis in chondrocytes through regulation of HMGB1. 27 In this work, we found that HMGB1 was a downstream target of miR-140-5p and CA could inhibit p-IKBα and p-IKKα/β expression levels by regulating HMGB1, indicating that in OA, CA could regulate miR-1285-5p/IL-20 axis and miR-140-5p/HMGB1/NF-κB pathway.
In conclusion, this study confirms that CA can promote the viability of C28/I2 chondrocytes and inhibit apoptosis and inflammatory response by regulating miR-1285-5p/IL-20 axis and miR-140-5p/HMGB1/NF-κB pathway, thereby inhibiting OA progression. Our study suggests that CA is a promising natural drug for OA treatment.
Footnotes
Acknowledgments and Funding
(1) Project of Guangdong Provincial Administration of Traditional Chinese Medicine; (2) Project of Guangdong Provincial Department of Education.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
