Abstract
Objective
Mesenchymal stem cells (MSCs) are a promising cell-based therapy treatment option for several orthopedic indications. Because culture expansion of MSC is time and cost intensive, a bedside concentration of bone marrow (BM) aspirate is used as an alternative. Many commercial systems are available but the available literature and knowledge regarding these systems is limited. We compared different point-of-care devices that concentrate BM (BMC) by focusing on technical features and quality parameters to help surgeons make informed decisions while selecting the appropriate device.
Methods
We compared published data on the BMC devices of Arteriocyte, Arthrex, Celling Biosciences, EmCyte, Exactech, ISTO Tech, Harvest Tech/Terumo BCT, and Zimmer/BIOMET regarding technical features (centrifugation speed/time, input/output volume, kit components, type of aspiration syringes, filter usage) and quality parameters of their final BMC product (hematocrit, concentration of platelets and total nucleated cells, concentration of MSC and connective tissue progenitor cells).
Results
The systems differ significantly in their technical features and centrifugation parameters. Only the fully automated systems use universal kits, which allow processing different volumes of BM. Only the Arthrex system allows selection of final hematocrit. There was no standardized reporting method to describe biologic potency.
Conclusions
Based on the data obtained in this review, recommending a single device is not possible because the reported data could not be compared between devices. A standardized reporting method is needed for valid comparisons. Furthermore, clinical outcomes are required to establish the true efficacy of these systems. We are conducting additional studies for more careful comparison among the devices.
Keywords
Introduction
Autologous cell-based therapies like platelet-rich plasma (PRP) and bone marrow aspirate concentrate (BMAC)/bone marrow concentrate (BMC) are promising alternative treatment options for several orthopedic indications. Both are generated by density gradient centrifugation of either whole blood or bone marrow aspirate (BMA), which leads to a concentration of platelets and their important growth factors. The major difference and possible advantage of BMC in comparison to PRP is the content of mesenchymal stem cells (MSCs) that have the ability to differentiate into different connective tissue cell types (including chondrocytes) and therefore represent promising candidates to repair damaged tissue. Apart from that, MSC also possess immunomodulatory, anti-inflammatory, antiapoptotic, proliferative and chemoattractive functions and can coordinate the differentiation process of functional tissue regeneration in host cells.1,2 Many studies show that the use of culture expanded MSC and BMC for the treatment of different orthopedic conditions, for example, tendinopathy, osteonecrosis of the femoral head and especially osteoarthritis and focal cartilage lesions is feasible, safe, and can provide some clinical efficacy in terms of pain reduction, joint function and quality of life.3-10 Furthermore, in a recent study Cassano
Although MSC can be found and extracted from different tissues, such as skeletal muscles, umbilical cord blood and Wharton’s jelly, the most common sources in orthopedic interventions are bone marrow and fat tissue. It has to be clarified that bone marrow–derived MSC represent only 0.001% to 0.01% of the mononuclear cells in the bone marrow whereas MSC occur at a 100- to 1000-fold higher concentration in adipose tissue. 1 Despite the higher concentration of stem cells in adipose tissue, the extraction of cells requires collagenase digestion, which takes 1 to 2 hours and is not approved by the Food and Drug Administration (FDA). While culture expansion of bone marrow generates more MSC than centrifugation, 12 it is a highly time- and cost-intensive procedure, requires GMP (Good Manufacturing Practice) facilities, and is not yet approved by the FDA. In contrast, a point-of-care concentration of BMA may be a reasonable alternative for the clinical practice and is permitted by the FDA due to minimal manipulation of the cells. Many commercial systems from several manufacturers are available; however, it is challenging to select the most appropriate system.
Because of the lack of clinical trials and outcome data after BMC treatments, in this review we compared published data of 8 different point-of-care devices that concentrate BM with the focus on technical features and quality of the harvested product. The primary objective was to provide surgeons with a single resource to enable selection of the appropriate device in clinical practice.
Methods
An initial search was conducted from February until July 2016 using existing brochures and a Google search to find companies that manufactured point-of-care devices to concentrate bone marrow. All companies were contacted by email or phone to get detailed information about their devices. Devices and companies were excluded if they were not FDA approved, not approved for intra-articular injection, not considered point-of-care devices, did not provide information, or did not provide information in English. The following devices where included in our comparison: the MAGELLAN MAR0Max device from Arteriocyte Medical Systems, the Arthrex Angel System Bone Marrow Concentration, the ART BMC device from Celling Biosciences, the PureBMC device from EmCyte Corporation, the Accelerate: Autologous Platelet Concentrating System from Exactech, the CellPoint Concentrated Bone Marrow Aspirate System from ISTO Technologies, the BMAC 2 device from Harvest Technologies/Terumo BCT, and the BioCUE Blood and Bone Marrow Aspirate Concentration System from Zimmer/BIOMET. We then performed a literature search in PubMed to find publications in which the devices were used or in which different devices were compared. The search strategy included (BMAC or bone marrow aspirate concentrate), (BMC or bone marrow concentrate) as well as only the name and the company of the different devices. Finally, based on the information obtained from our searches, all devices were compared for technical features as follows: centrifugation speed and time, input and output volumes, kit components, type of syringe used to aspirate, and usage of a filter before processing the bone marrow. We also assessed each device on the quality of their BMC product as follows: hematocrit (HCT), concentration of platelets and total nucleated cells (TNC), concentration of MSC, and colony forming unit fibroblasts/connective tissue progenitor cells (CFU-F/CTP). We excluded manufacturer specifications and publications that were not peer-reviewed data. Because of our focus on the treatment of orthopedic conditions (especially osteoarthritis and focal cartilage lesions), we did not compare the presence of hematopoietic stem cells (HSCs). “Enrichment” is the relative increase of cell concentration in BMC relative to BMA using commercially available cell counters. “Percentage of recovery” was calculated by dividing the total number of cells recovered in the BMC by the total number present in the volume of BMA processed. CFU assays are commonly used to quantify the number of progenitor or MSC, in which bone marrow cells are seeded and cultured for a certain period of time, and the resulting cell colonies are counted. Information regarding CFU could be found for almost all systems, although the results were reported in different units, which precluded direct comparisons between devices.
Finally, all information was summarized. Tables 1 and 2 list the complete detailed comparison of all devices, including the sources from where this information was gathered. Table 1 compares the technical features of the devices and Table 2 lists the independent peer-reviewed data regarding the quality criteria of the BMC. The asterisk (*) symbol identifies the sources from which the information was gathered. The absence of any symbol indicates that the source of the information was from manufacturers’ specifications. This review was independent and was not funded by industry.
Detailed Comparison of Different Point-of-Care Devices to Concentrate Bone Marrow (Technical Aspects).
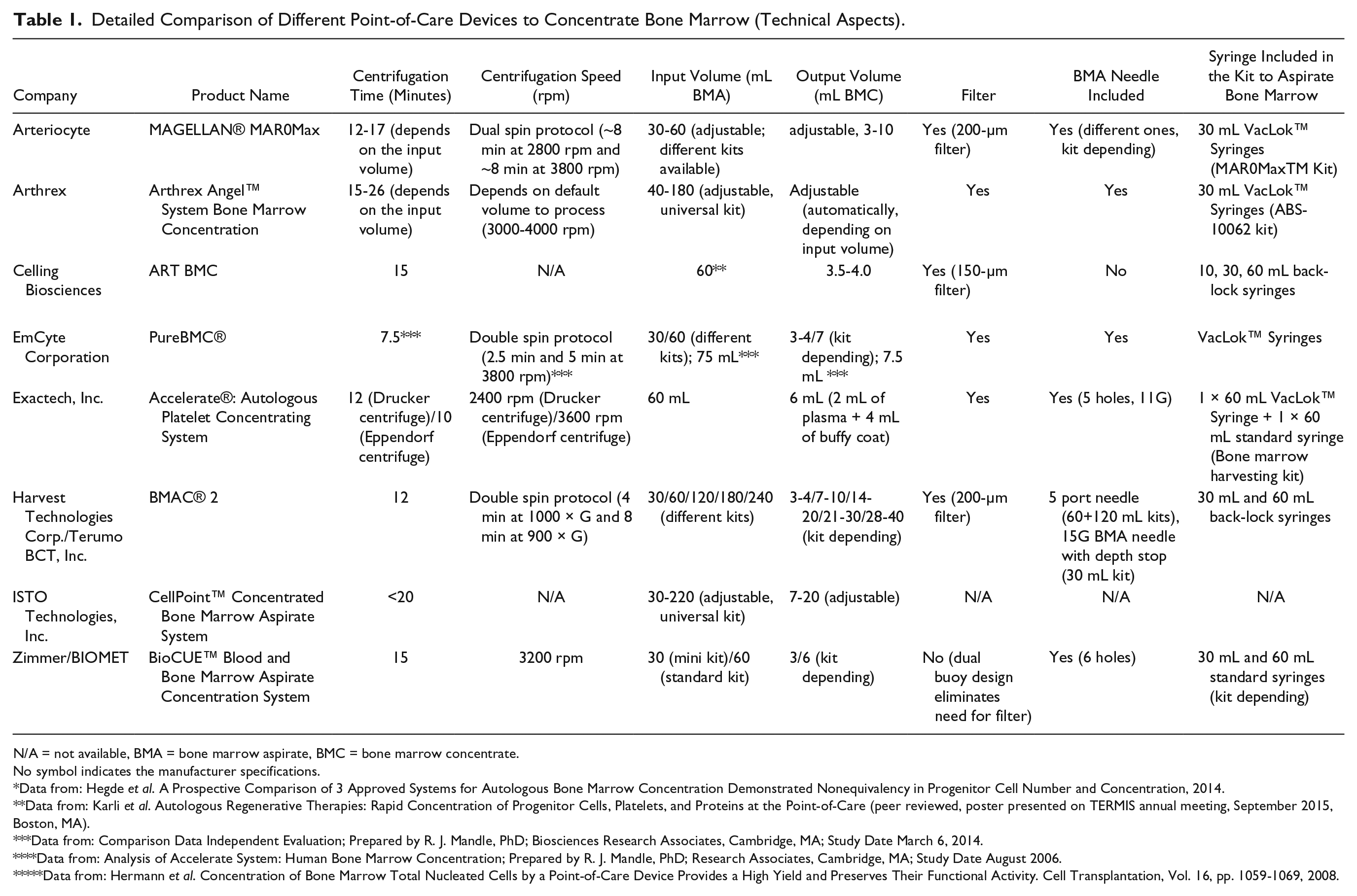
N/A = not available, BMA = bone marrow aspirate, BMC = bone marrow concentrate.
No symbol indicates the manufacturer specifications.
Data from: Hegde
Data from: Karli
Data from: Comparison Data Independent Evaluation; Prepared by R. J. Mandle, PhD; Biosciences Research Associates, Cambridge, MA; Study Date March 6, 2014.
Data from: Analysis of Accelerate System: Human Bone Marrow Concentration; Prepared by R. J. Mandle, PhD; Research Associates, Cambridge, MA; Study Date August 2006.
Data from: Hermann
Detailed Comparison of Different Point-of-Care Devices to Concentrate Bone Marrow (BMC Quality Criteria).
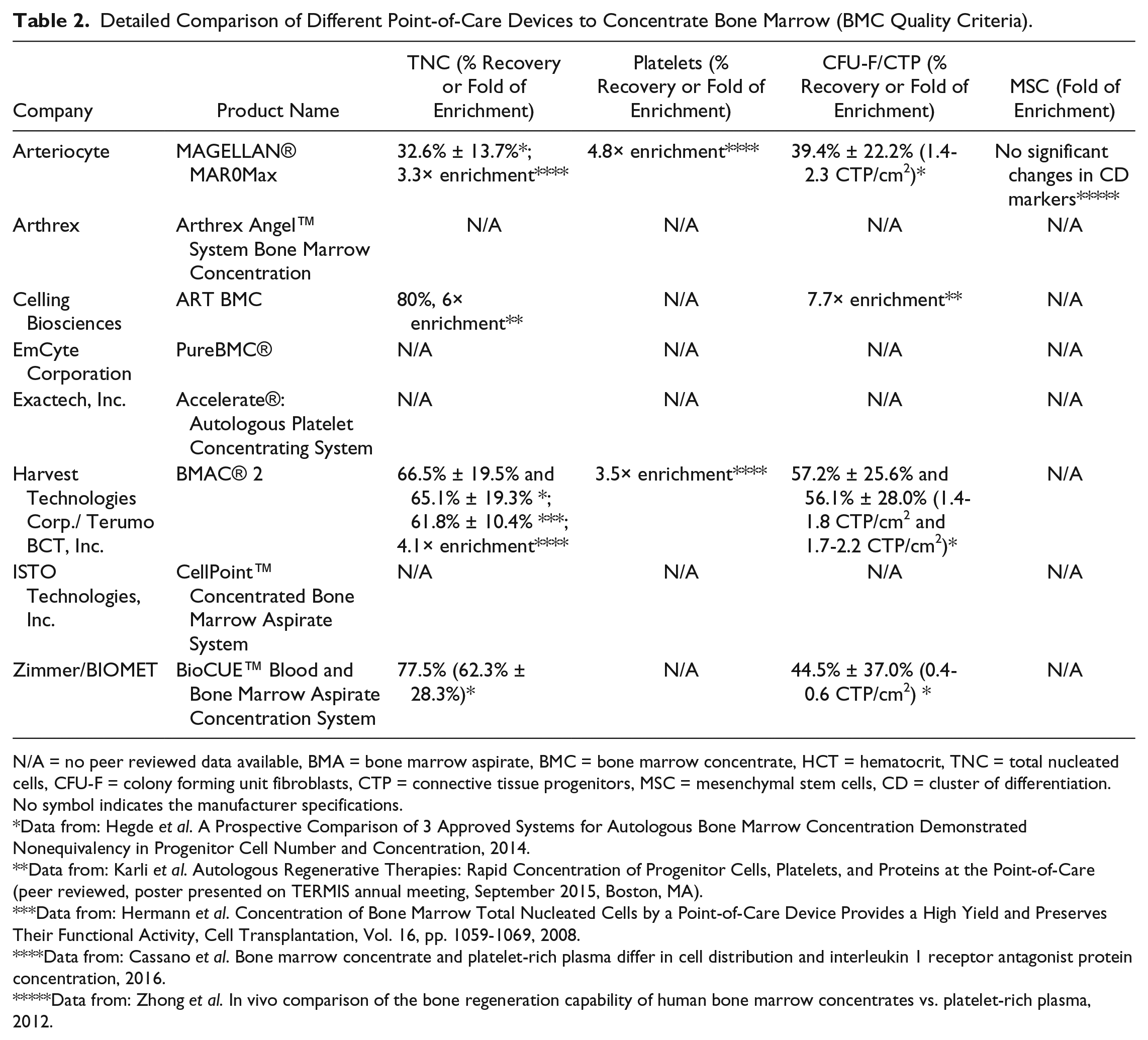
N/A = no peer reviewed data available, BMA = bone marrow aspirate, BMC = bone marrow concentrate, HCT = hematocrit, TNC = total nucleated cells, CFU-F = colony forming unit fibroblasts, CTP = connective tissue progenitors, MSC = mesenchymal stem cells, CD = cluster of differentiation.
No symbol indicates the manufacturer specifications.
Data from: Hegde
Data from: Karli
Data from: Hermann
Data from: Cassano
Data from: Zhong
Clinical Use of BMC
There have been several encouraging reports of the use of autologous BMC as intra-articular injections for the treatment of OA and as implants for the treatment of full-thickness chondral defects.3,13-20 Even though long-term results are pending, a recent prospective randomized study showed no significant differences in short-term pain relief between injection of BMC and saline in knees of patients with OA. 21 Nevertheless, BMC is extensively used for orthopedic applications by surgeons worldwide and there are diverse commercial systems from several manufacturers that incorporate a vast number of technical features. Given the lack of high-quality studies and definitive evidence regarding clinical efficacy, the important parameters that are likely to be relevant to the clinician are safety, convenience, time involved, and reproducibility. It is not yet known if differences in the technical features of the different devices have an impact on the quality of patient care and clinical outcomes. We conducted this review to assist the surgeon in making informed comparisons when choosing among the different systems.
Comparison of Technical Features
The harvesting systems can be broadly classified into 2 groups. The first includes the computer-controlled, fully automated, and closed-loop systems with an integrated centrifuge, which are used by the devices from Arteriocyte, Arthrex, and ISTO Technologies. An important feature of these devices is the optical sensor to detect the different wavelengths corresponding to the various blood fractions to separate the buffy coat after centrifuging. In a fully automated system, after connecting the BMA syringe to the device and after selecting appropriate parameters in the computer software, the system will deliver the BMC ready to use in a syringe without any syringe switching or further operations required by the user. In contrast, the second group of systems uses a manual extraction of the buffy coat. Therefore, the systems of Celling Biosciences, EmCyte, Exactech, Harvest Technologies/Terumo BMC, and Zimmer/BIOMET provide different closed sterile processing disposables in which the BMA is injected. This disposable has to be placed in an external centrifuge to separate the various blood fractions. Next, the device has to be taken out of the centrifuge, a syringe has to be connected, and the buffy coat has to be extracted manually according to respective manufacturer’s specifications.
The advantages of a closed-loop system are greater convenience and possibly less risk of infection or contamination, although no publications could be found to prove scientific evidence of likelihood of these risks. The advantage of an integrated centrifuge is convenience and reduced equipment in the procedure room. Manual extraction gives the surgeon greater control over the harvested material, but with the disadvantage of variation due to differences in extracted material.
Input/Output Volumes
One important criterion for selection is the volume of bone marrow the surgeon is able to harvest and the quantity of BMC needed for the treatment. As depicted in Table 1 , the input volumes of BMA for the different devices range between 30 and 240 mL and the output volumes of BMC between 3 and 40 mL. For clinical applications requiring variable volumes of BMC, the fully automated devices possess certain advantages. The systems of Arteriocyte and ISTO Technologies allow selection of the final BMC volume, whereas the Arthrex device automatically adjusts the output volume depending on the volume of BMA input. Furthermore, the fully automated systems provide universal disposable kits, which allow processing different volumes (30-60 mL with the Arteriocyte device, 40-180 mL with the Arthrex device, and 30-220 mL with the ISTO Technologies device). All other companies provide individual kits, which allow only processing and delivery of predefined volumes of BMA and BMC, respectively.
Centrifugation Parameters
In Table 1 , the centrifugation parameters of the different devices are shown. The centrifugation times range between 7.5 and 20 minutes. The centrifugation speed ranges between 2400 and 4000 rpm (Harvest Technologies: 900 and 1000 × G). Three systems (Arthrex, EmCyte, and Harvest Technologies) use a double spin protocol in which a first centrifugation cycle separates the red blood cell fraction followed by a second cycle to separate the buffy coat. Except Celling Biosciences and ISTO Technologies, which did not provide information regarding the centrifugation speed, all other devices employ a single centrifugation cycle. We only found one study that compared different centrifugation protocols to separate BMC. 22 In that study, single and double centrifugation of BMA for sinus floor elevation were compared but no statistically significant differences were found. Therefore, no evidence-based superiority of a specific device using a specific protocol to concentrate bone marrow can be determined at this time.
Kit Content
The quality of the final BMC likely depends on the quality of the bone marrow and the aspiration technique. Fast and continuous aspiration of bone marrow with a high differential pressure, using smaller volume syringes, results in a better quality specimen and in more stem cells.23,24 Furthermore, multiple advancements of the bone marrow needle and multiple site aspiration increases initial nucleated cell counts, CFU-Fs, and generates more MSC at the first passage. 25 Therefore, so-called back-lock syringes, which can be locked at a certain volume, are able to hold a continuous high differential pressure while aspirating and could be beneficial to improve BMA quality. The devices of Celling Biosciences, Arteriocyte, Arthrex, Harvest Technologies, Exactech, and Emcyte have these syringes included in their kits, which could be an advantage compared with systems that provide standard syringes.
Six devices (Arteriocyte, Arthrex, Celling Biosciences, Emcyte, Exactech, and Harvest Technologies) require the use of special filters after harvesting and before processing the bone marrow. This filter serves to remove bone marrow fat cells, clots, and small bone fragments from the bone marrow before centrifugation.
26
Hegde
Heparin Coating
Another aspect that could affect BMC quality is the heparin coating of the syringe, which is recommended in protocols of some devices (Arteriocyte, Arthrex, Exactech, and Harvest Technologies). Heparin could reduce the binding of CTPs to the surface of the syringes or transfer bags used with the different systems, which could lead in a smaller loss of CTPs during processing.27-29
Comparison of Different Blood Components and Quality Criteria
For orthopedic treatments, especially for the treatment of OA and focal cartilage defects, the primary possible advantage of BMC, besides the presence of platelets and their released growth factors, is the content of tissue progenitor cells, which could have the ability to form new cartilage. We selected the following surrogate criteria to compare the quality of the BMC: (1) the HCT; (2) the platelet count, because they release the important growth factors; (3) the TNC, which include MSC but, as mentioned above, only in a very small number (most of these cells are leukocytes); (4) the number of CFU-F or CTP, which represent progenitor cells; and (5) the concentration of MSC.
Hematocrit
The amount of residual red blood cells in the final BMC is a measure of the quality of cell separation. A recent
Enrichment and Recovery of Platelets and TNC
Table 2 displays the information regarding the recovery and enrichment of platelets and TNC in the BMC delivered by these systems. Direct comparisons were not possible because analyses were done on different volumes of BMA and BMC, using different cell counting machines, and using different counting protocols.
Only one study could be found that compared different devices using the same cell counter.
11
In that study, Cassano
Amount of Colony Forming Unit Fibroblasts/Connective Tissue Progenitor Cells
As a measure of the concentration of MSC in the final BMC, CFU-F/CTPs assay results were analyzed. In a CFU-assay, bone marrow is seeded into culture and the plastic-adherent colonies are counted after incubation for several days or weeks. Table 2 shows the information that could be gathered regarding the concentration of MSC and number of CFU-F/CTP in the BMC created by the different devices. Unfortunately, significant differences in the protocols such as the definition of a colony, incubation time, and reported units (CFU/mL, CTP/cm2, CTP/cm3, percentage of enrichment), precluded direct comparisons.
Only 2 independent research studies could be found that compared different devices using the same CFU-assay protocol.11,27 Hegde
Amount of Mesenchymal Stem Cells
The CFU assay is a commonly used method, which is reproducible and relatively inexpensive. However, CFU assays may not be the most quantitative measure of the presence of bone marrow MSC. The International Society for Cellular Therapy proposed minimal criteria for identification of MSC: MSC must be plastic-adherent under culture conditions, must express CD105, CD73, and CD90 while lacking expression of CD45, CD34, CD14 or CD11b, CD79α, or CD19 and HLA-DR surface molecules, and must differentiate into different cell types such as osteoblasts, adipocytes, and chondroblasts
Limitation of the Review
One weakness of this study was the limited availability of independent peer-reviewed publications. This is even more important because all devices reviewed are FDA approved and commercially available for clinical use. Nevertheless, this comparison summarizes the most relevant information in a single source to assist the surgeon in making more informed decisions when choosing among different devices. Cost is also important because intra-articular injections of BMC are not covered by medical insurance. However, each manufacturer negotiates a different price with the health care provider, which is why we could not provide any prices or cost of the compared devices or their kits. We are planning a more controlled comparison of the performance of different BMC devices followed by assessment of clinical efficacy.
Conclusion
Based on the data obtained in this review, recommending a single harvesting system is not possible. In our opinion, the fully automated systems have some technical advantages (among others, universal kits including small volume back-lock syringes and the use of a filter) and might be more convenient. In terms of “chondrogenic or anti-arthritic potency” of the BMC, we cannot recommend a single system because the reported data could not be compared between devices. A standardized reporting method is needed for valid comparison among devices. Furthermore, clinical outcomes are required to establish the true efficacy of these systems. We are conducting additional studies for more careful comparison among the devices.
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: WDB consulted for Arthrex for unrelated product development.
