Abstract
Objective
Cellular and molecular events occurring in cartilage regions close to injury are poorly investigated, but can possibly compromise the outcome of cell-based cartilage repair. In this study, key functional properties were assessed for cartilage biopsies collected from the central part of traumatic joint lesions (central) and from regions surrounding the defect (peripheral). These properties were then correlated with the quality of the initial cartilage biopsy and the inflammatory state of the joint.
Design
Cartilage samples were collected from knee joints of 42 patients with traumatic knee injuries and analyzed for cell phenotype (by reverse transcriptas-polymerase chain reaction), histological quality, cellularity, cell viability, proliferation capacity, and post-expansion chondrogenic capacity of chondrocytes (in pellet culture). Synovium was also harvested and analyzed for the expression of inflammatory cytokines.
Results
Cartilage quality and post-expansion chondrogenic capacity were higher in peripheral versus central samples. Differences between these 2 parameters were more pronounced in joints with high inflammatory features characterized by >100-fold difference in the mRNA levels of IL6 and IL8 in the corresponding synovium. Peripheral chondrocytes isolated from good- versus bad-quality biopsies expressed higher levels of collagen II/I and aggrecan/versican and lower levels of MMP13 and ADAMTS5. They also exhibited reduced proliferation and enhanced cartilage-forming capacity.
Conclusions
Chondrocytes at the periphery of traumatic lesions better maintain properties of healthy cartilage compared to those isolated from the center, even when derived from bad-quality tissues harvested from highly inflamed joints. Future studies are necessary to investigate the change of functional properties of peripheral chondrocytes over time.
Introduction
Because of the very limited capacity of articular cartilage to heal spontaneously, its damage, for example, due to trauma, requires treatment to restore the cartilaginous structure. If traumatic injuries are left untreated, the chances for the patient to develop posttraumatic osteoarthritis (OA) are highly increased, 1 leading to joint pain, loss of mobility, and decreased quality of life in the long term.2,3 Therefore, not only to restore joint function but also to decrease the probability to develop trauma-induced OA, significant cartilage lesions are commonly treated, for example, by (matrix assisted-/) autologous chondrocyte implantation (MACI or ACI). 4 Before transplanting the therapeutic cell preparation, the lesion needs to be thoroughly debrided not only of the macroscopically diseased tissue 5 but also of adjacent cartilage areas that appear intact, to create a confined lesion with a rim of healthy cartilage. 6 Despite its healthy appearance, cartilage tissue surrounding the defect might have acquired cellular and molecular changes that can compromise the clinical outcome of the cell-based cartilage repair strategies. 7 In particular, ACI failure has been linked to an improper debridement of the lesion that compromised the capacity of the implanted cells to integrate into the defect areas. 5 However, possible underlying alterations in the cartilage surrounding lesions remain largely unknown.
To date, only a few reports have described the properties of chondrocytes isolated from damaged joints.8 -12 In these studies, the characterized material, generally termed debrided cartilage, was collected from joints of patients with unidentified inflammatory statuses and consisted of mixtures of both the cartilage specimens derived from the central—more damaged—part of the lesion and the border region surrounding the lesion. Chondrocytes from these debrided tissue mixtures were mostly compared to chondrocytes collected from nonaffected cartilage tissues harvested from distant compartments of the same patients’ joints or from autopsies. Hence, even though the clinical evidence suggested that traumatic lesions affect the homeostasis of the whole joint, 13 it remains unknown to which extent the properties of cartilage and chondrocytes at the edges of lesions are affected in comparison to tissues from the same joint at a distant location.
In our study, we separately collected and analyzed cartilage tissue specimens from the central part of the lesion (central) and in closely adjacent regions surrounding the lesion (peripheral) of the same traumatic joint from a relatively large number of donors. To assess possible alterations in the biopsies from these 2 locations, we investigated the following properties: (1) histological quality of the tissue, (2) cellularity, (3) cell viability (post tissue digestion), (4) proliferation capacity, and (5) post-expansion cartilage forming capacity of the isolated chondrocytes. Additionally, we investigated the correlation of these properties with the inflammatory features of the joint and with the initial quality of the cartilage tissue.
Materials and Methods
Collection of Samples
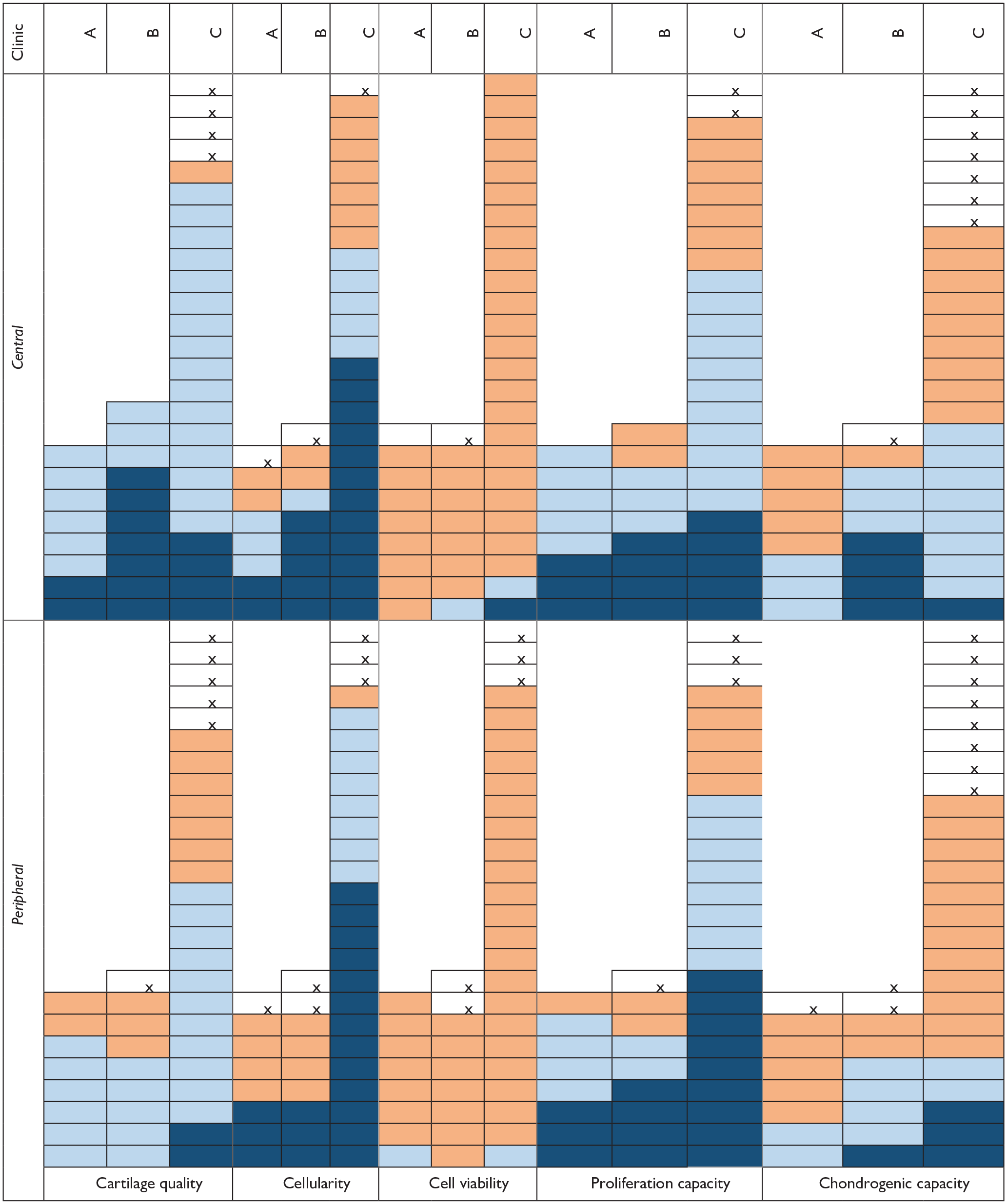
The samples described below (n = 42) were collected at the time of surgery from patients in 3 different clinics: clinic A (University Hospital Basel, Basel), clinic B (Practice LEONARDO, Hirslanden Clinic Birshof, Münchenstein), and clinic C (Schulthess Clinic, Zurich). The study was approved by the ethics committees in Basel and Zurich (EKNZ-2014-199, PB_2016-1925) and in accordance with the Declaration of Helsinki. The inclusion criteria are reported in Table S1. The recruited patients (see info in Fig. 1A ) had persistent joint pain and mechanical symptoms. We consecutively asked patients scheduled for a knee joint preserving surgery to participate and obtained written informed consent. The majority of the patients received ACI and transplantation of particulate fragments alone or in combination with cancellous bone plasty (autologous matrix induced chondroplasty).
Clinical data. (
Central cartilage tissues were harvested with a 4-mm biopsy punch with minimal 2-mm distance from the center of the defect or cut hemi-circumferentially around the defect. Full thickness cartilage tissues from the periphery of the lesion were collected from cartilage surrounding the defect (2-5 mm) with a 4-mm biopsy punch ( Fig. 1B ). Synovial tissues were also collected from the same joints with an arthroscopic grasper.
Samples (central cartilage, peripheral cartilage, and synovium) were placed in separate sterile prelabelled tubes, containing transport medium (phosphate buffered saline solution supplemented with 100 units/mL penicillin and 100 mg/mL streptomycin) and shipped to the laboratory at the University Hospital Basel at the same or following day.
At the laboratory, samples were processed for specific characterizations (see Table S2). Cartilage specimens were cut into 2 parts: half of the tissue was fixed in 4% formalin for histological and immunohistochemical analyses, and the other half was cut in small pieces and processed for cartilage digestion. Synovial tissues were also cut into 2 parts: one half was fixed in 4% formalin for histological and immunohistochemical analyses, and the other half stored at −80°C and further processed for quantitative real-time RT-PCR (reverse transcriptase polymerase chain reaction) analyses.
Chondrocyte Isolation, Expansion, and Chondrogenic Culture
Digestion of cartilage tissues from the central and peripheral regions of the lesion was performed as previously described. 14 In brief, cartilage tissues were chopped in small pieces that were treated with 0.15% type II collagenase (Worthington Biochemical Corp., Lakewood, NJ) on an orbital shaker at 37°C for 22 hours. The resulting isolated cells were counted using trypan blue to estimate the percentage of viable cells. Cells were plated in culture dishes at a density of 1000 to 2000 cells/cm2 and cultured in Expansion Medium: Dulbecco’s modified Eagle’s medium (DMEM; Gibco) containing 4.5 mg/mL D-glucose and 0.1 mM nonessential amino acids, 1 mM sodium pyruvate (Gibco), 10 mM HEPES buffer (Gibco), 100 units/mL penicillin, 100 mg/mL streptomycin, 0.29 mg/mL L-glutamine (basic medium) supplemented with 10% fetal bovine serum for 2 passages. 14 From 11 matched samples (3 from clinic A and 8 from clinic C), having a relatively large size, a portion of the post-digested cells were processed for RT-PCR analyses as described below.
As reference, macroscopically uninjured normal articular cartilage was obtained from the knee joints of 5 cadaveric patients (within 24 hours after death) with unknown clinical history of joint disorders (mean donor age: 56 years; range: 51-60 years; 1 female and 4 males) and after informed consent obtained from relatives. Additionally, macroscopically fibrillated articular cartilage was used as reference and was obtained from the knee joints of 10 patients with clinical history of OA (mean donor age: 74 years; range: 55-82 years; 6 female and 4 males) undergoing total knee replacement, after informed consent from patients. All samples were collected and processed in accordance with the local ethics committee (University Hospital Basel). Again, cartilage biopsy was minced and digested enzymatically. Freshly isolated articular chondrocytes from the different donors were pooled and frozen to further perform quantitative RT-PCR analysis.
The proliferation index was calculated as the ratio of log 2(N/N0) to T, where N0 and N are the cell numbers at the beginning and the end of the expansion phase, respectively, log 2(N/N0) is the number of cell doublings, and T is the number of days required for the expansion.
Chondrogenic differentiation was induced in 3D micromass pellet cultures using a chondrogenic medium (basic medium supplemented with 10 mg/mL insulin, 5.5 mg/mL transferrin, 5 ng/mL selenium, 0.5 mg/mL human serum albumin, 4.7 mg/mL linoleic acid, 0.1 mM ascorbic acid-2-phosphate [Sigma], 10−7 M dexamethasone [R&D Systems, Minneapolis, MN], and 10 ng/mL transforming growth factor [TGF] β3 [Novartis]). Cell suspensions were diluted at 106 cells/mL, 0.25 mL distributed per 1.5 mL polypropylene conical tubes (Sarstedt, Numbrecht, Germany) and centrifuged at 1100 rpm for 3 minutes to form spherical pellets. Pellets were cultured for 2 weeks with medium changed twice weekly and afterwards assessed histologically and biochemically.
Analytical Methods
Histology
Native tissues (central cartilage, peripheral cartilage, and synovium) and chondrogenic pellets were fixed in 4% paraformaldehyde, dehydrated, and embedded in paraffin. Synovial tissues were stained with hematoxylin and eosin according to a standard protocol. Native cartilage tissues and chondrogenic pellets were stained with Safranin O/fast green (SafO/FG). SafO/FG sections of native cartilage were used to grade cartilage quality as the following: no/very weak staining (score 0), weak or scattered staining (score 1), moderate or nonuniform staining (score 2), intense and uniform staining (score 3). SafO/FG sections of chondrogenic pellets were used to grade the extent of neo-formed cartilage by post-expanded chondrocytes using the Bern Score (BS). 15
Immunohistochemistry
Immunohistochemical staining were performed with Ventana Discovery Ultra (Roche Diagnostics [Suisse], SA) automated slide stainer. In brief, tissue sections were deparaffinized and rehydrated. Antigens were retrieved by a protease (Protease 3, 760-2020, Ventana) digestion for 20 to 44 minutes at 37°C. Primary antibody was manually applied and incubated for 1 hour at 37°C. After washing, the secondary antibody was incubated for 1 hour at 37°C. The detection step was performed with the Ventana DISCOVERY ChromoMap DAB (760-159, Ventana) detection kit. Afterwards, the slides were counterstained with hematoxylin II, followed by the bluing reagent (respectively, 790-2208 and 760-2037; Ventana). Sections were then dehydrated, cleared, and mounted with permanent mounting and coverslips.
The following primary antibodies were used for IL6 (PA1-268811, Invitrogen) 1:100, matrix metalloproteases (MMP)13 (ab39012, abcam) 1:100, type I collagen (COL1) (ab137492, abcam) 1:5000, and type II collagen (COL2) (63171, MP Biomedicals) 1:1000. Secondary antibodies used included anti-mouse polymer horseradish peroxidase (HRP), R&D Mouse IgG (VC001-025, VisUCyte), and anti-rabbit polymer HRP (414141F, Nichirei Histofine Simple Stain MAX PO).
Quantification of Glycosaminoglycans and DNA
Chondrogenic pellets were digested with 1 mg/mL protease K in 50 mM Tris with 1 mM EDTA, 1 mM iodoacetamide, and 10 mg/mL pepstatin-A for 16 hours at 56°C. For glycosaminoglycan (GAG) quantification, the method of Barbosa et al., 16 was used. Briefly, digested pellets were incubated with 1 mL of dimethylmethylene blue assay (DMMB) solution (16 mg/L dimethylmethylene blue, 6 mM sodium formate, 200 mM GuHCL, pH 3.0) on a shaker at room temperature for 30 minutes. Precipitated DMMB-GAG complexes were centrifuged and supernatants were discarded. Complexes were dissolved in decomplexion solution (4 M GuHCL, 50 mM Na-Acetate, 10% Propan-1-ol, pH 6.8) at 60°C. The absorption was measured at 656 nm and GAG concentrations were calculated using a standard curve prepared with purified bovine chondroitin sulfate. DNA content was measured by using the CyQuant Cell Proliferation Assay Kit (Molecular Probes Inc., Eugene, OR) according to the instructions of the manufacturer.
Quantitative Real-Time RT-PCR
Total RNA was extracted from native synovial tissues, chondrocytes following cartilage digestion, and chondrogenic pellets. cDNA synthesis and RT-PCR (7300, Applied Biosystems) were performed as previously described 17 to quantify expression levels of type I collagen (COL1A1, Hs00164004), type II collagen (COL2A1, Hs00264051), aggrecan (ACAN, Hs00153936_m1), versican (VCAN, Hs00171642_m1), interleukin 6 (IL6, Hs00985639_m1), hyaluronan and proteoglycan binding link protein gene family-1 (HAPLN1, Hs00157103_m1), microfibrillar associated protein 5 (MFAP5, Hs00185803_m1), interleukin 8/C-X-C motif chemokine ligand 8 (IL8/CXL8, Hs00174103_m1), a disintegrin and metalloproteinase with thrombospondin motifs 5 (ADAMTS5, Hs00199841_m1), and metalloprotease 13 (MMP13, Hs00233992_m1), all from Applied Biosystems. For each sample, the Ct value of each target sequence was subtracted from the Ct value of the reference gene (glyceraldehyde 3-phosphate dehydrogenase, GAPDH, Hs02758991, Applied Biosystems) to derive the ΔCt.
Statistical Analyses
Statistical evaluation was performed using SPSS software version 22 (SPSS, Sigma Stat). Data were represented as the mean ± SE (standard error). Differences between groups were estimated by Kruskal-Wallis followed by Mann-Whitney tests for multiple comparisons and P values adjusted with Bonferroni correction. Comparisons of 2 populations were performed by Mann-Whitney test. P values <0.05 were considered statistically significant.
Results
We collected 42 samples from 3 different clinics (clinic A: n = 8; clinic B: n = 9; clinic C = 25). The clinically relevant information related to these patients are reported in Figure 1 . Briefly, patients (28% female and 72% male) with a mean age of 32 years (range 18 to 60 years) mostly (56%) had a healthy body weight range (body mass index [BMI]: 18.5-25.0 kg/m2). The patients experienced their knee injury from 3 months up to 9 years prior to the intervention. The time of symptom onset ranged between less than 6 months (14%), 6 to 12 months (8%), or more than 12 months (78%). The lesions were located either in single compartments (50% patella, 26% condyle, 15% trochlea) or multiple compartments (9%) and were generally of severe grade (International Cartilage Repair Society [ICRS] macroscopic score 3-4; Fig. 1A ).
Characterization of the Properties of Cartilage and Chondrocytes from Central and Peripheral Regions of the Cartilage Lesion
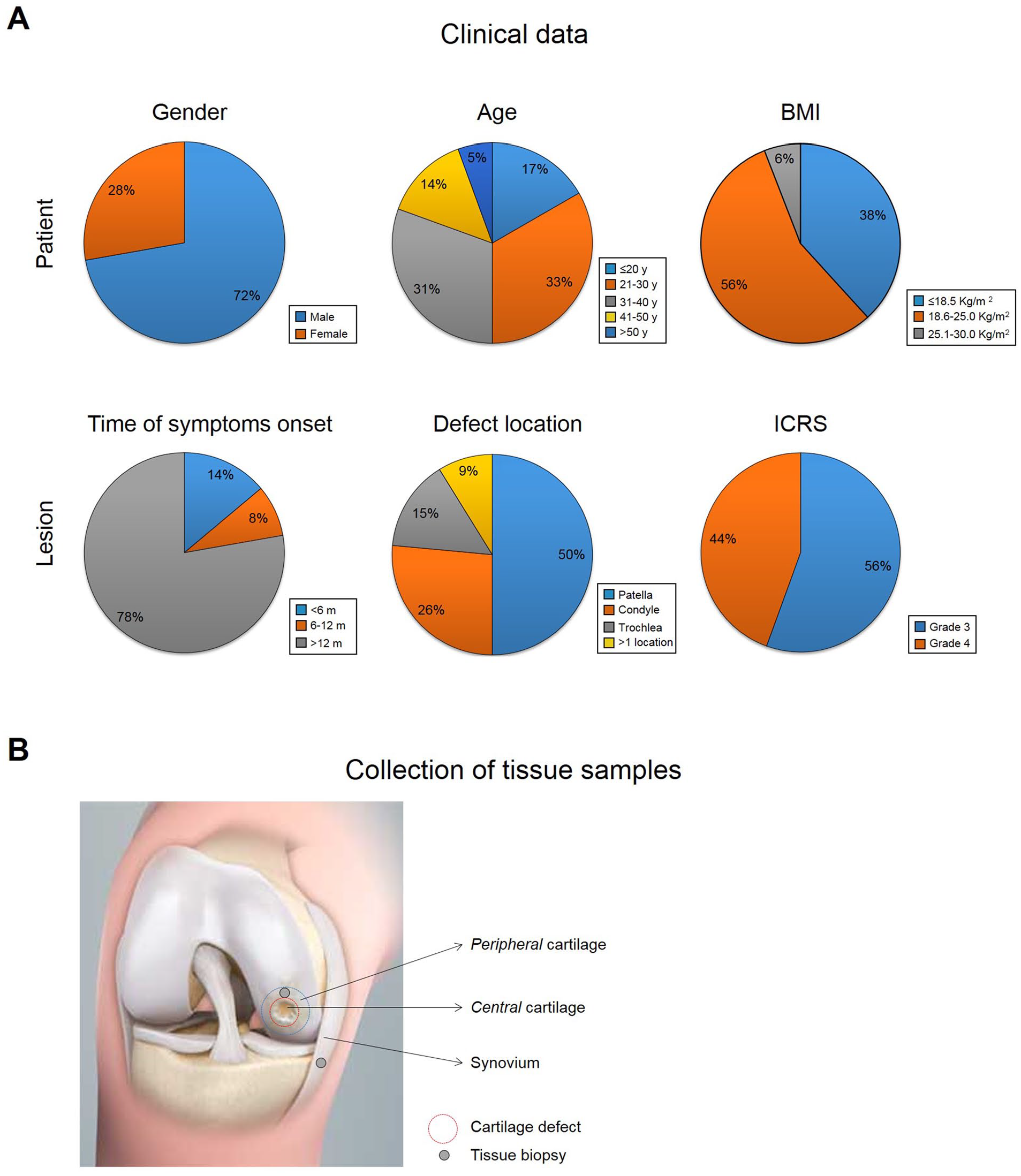
For a certain number of the collected cartilage samples, we could not perform the full set of characterizations, for example, due to their limited sizes (see Table 1 ). Overall, our results (reported in Table 1 ) showed the following trends: (1) the cartilage samples were highly viable and had heterogeneous properties, (2) cartilage biopsy quality was the parameter that mainly differed between the peripheral and central samples, while (3) the interclinical variability was relatively low ( Table 1 ). A detailed description of the results is presented in the following sections.
Overview of the Generated Results on the Cartilage Samples a .
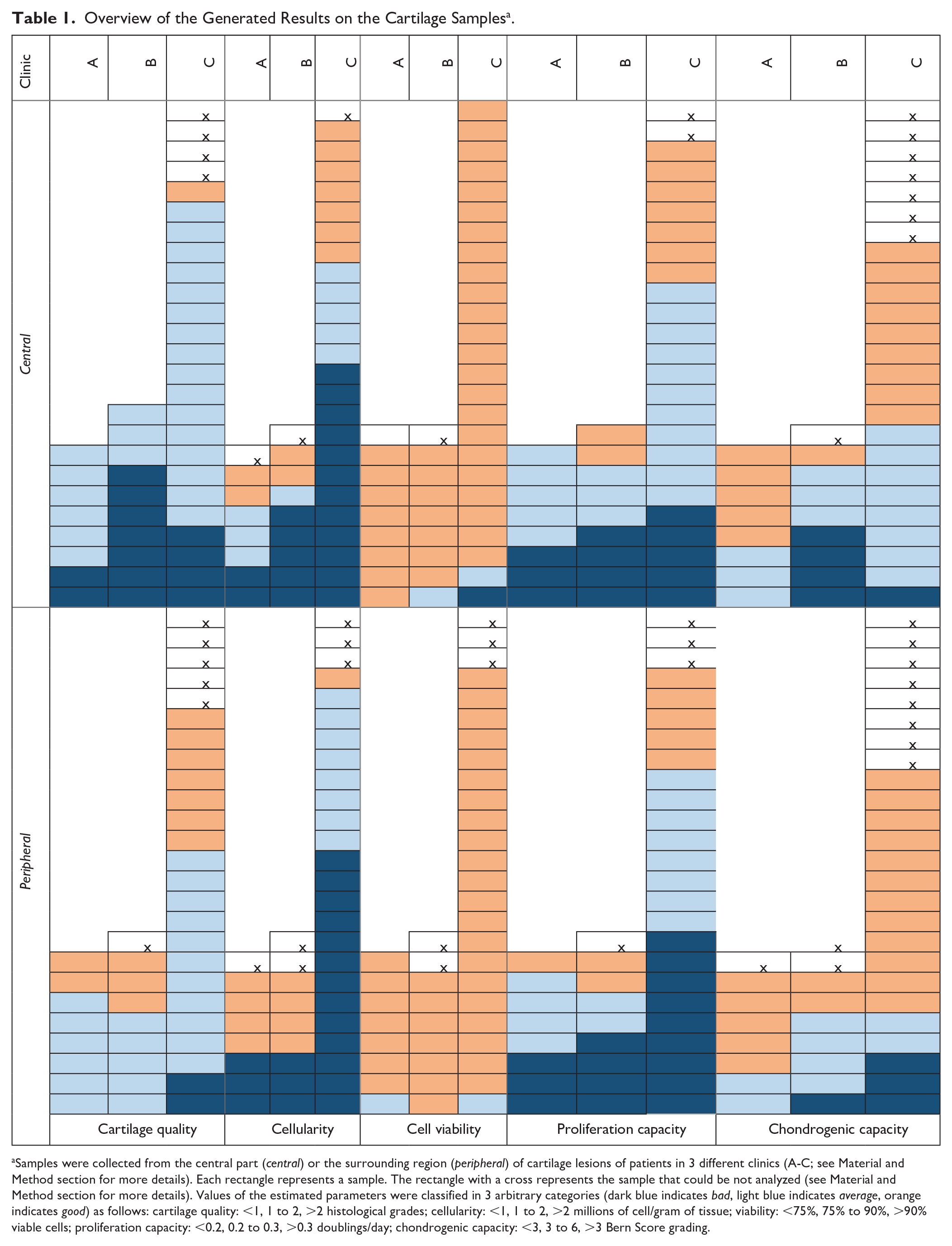
Samples were collected from the central part (central) or the surrounding region (peripheral) of cartilage lesions of patients in 3 different clinics (A-C; see Material and Method section for more details). Each rectangle represents a sample. The rectangle with a cross represents the sample that could be not analyzed (see Material and Method section for more details). Values of the estimated parameters were classified in 3 arbitrary categories (dark blue indicates bad, light blue indicates average, orange indicates good) as follows: cartilage quality: <1, 1 to 2, >2 histological grades; cellularity: <1, 1 to 2, >2 millions of cell/gram of tissue; viability: <75%, 75% to 90%, >90% viable cells; proliferation capacity: <0.2, 0.2 to 0.3, >0.3 doublings/day; chondrogenic capacity: <3, 3 to 6, >3 Bern Score grading.
Cartilage Quality
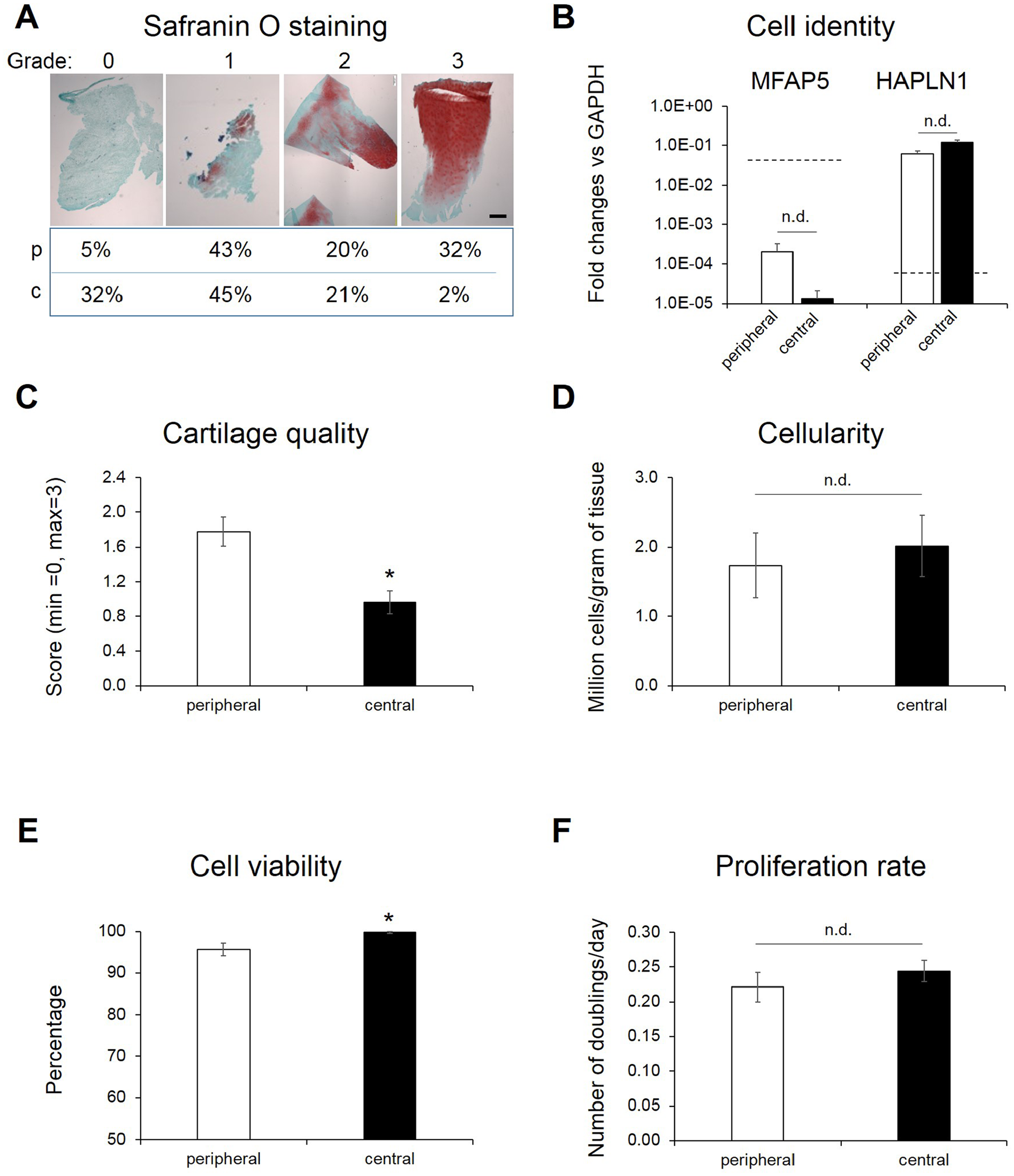
Representative Safranin O pictures of cartilage samples are displayed in Figure 2A . While similar fractions of peripheral and central cartilage samples had intermediate quality (grades 1 and 2), large differences were observed in the percentage of cartilage samples with grade 0 (5% peripheral vs. 32% central) and grade 3 (32% peripheral vs. 2% central; Fig. 2A ). To better clarify the identity of cells isolated from the central and peripheral cartilage, we assessed the expression levels of HAPLN1 (a marker highly expressed in chondrocytes from different cartilage tissues 18 ) and MFAP5 (a marker previously used as a negative marker for chondrogenic cells from articular cartilage). 19 Results reported in Figure 2B show that as compared to peripheral chondrocytes, central chondrocytes expressed similar low levels of MFAP5 and similar high level of HAPLN1, thus suggesting that the tissues collected in the lesioned cartilage areas are mainly composed by chondrocytic cells. Instead, synovial tissue (that is rich in fibroblastic cells), displays, as expected, high level of MFAP5 and low level of HAPLN1.
Cell and tissue properties of peripheral and central cartilage. (
Grading of the peripheral cartilage specimens was 1.9-fold higher (P = 0.001) than that of central samples ( Fig. 2C ). Interclinically, no significant differences in cartilage quality were observed, even if the differences in the quality between peripheral and central samples varied to some extent (histological grading peripheral vs. central were 1.9-fold [P = 0.13], 4.2-fold [P = 0.006], and 1.5-fold [P = 0.095], respectively, for clinics A, B, and C; Fig. S1A).
A more in depth characterization was performed to assess the presence and expression of additional cartilage makers as well as cartilage-degrading markers on tissues with different histological grades. Immunohistochemical results showed that good-quality tissues (grades 2-3) contained more type II collagen and less type I collagen than bad-quality cartilage (grades 0-1). Instead, MMP13 was almost solely detected in bad quality cartilage tissues. Also, at gene expression level, a trend toward higher mRNA expression of aggrecan and type II collagen mRNA but lower versican and MMP13 expression by cells from good-quality tissues was demonstrated (see Fig. S2). These results indicate the more degenerative status of the bad- versus good-quality tissues.
Cellularity
Large interdonor variability in the cellularity was observed (from less than 0.5 to more than 10 million cells/gram of tissue; Fig. S1B) so that no statistically significant differences (overall and between clinics) in this parameter could be observed (
Cell Viability
Cell viability was generally high (ranging from 73% to 100%) in the collected samples. However, statistically significant higher percentage of viable cells were counted in peripheral as compared to central samples (98.6% vs. 96.8%, P = 0.006;
Proliferation Capacity
Proliferation rates of cells were variable (ranging from 0.06 to 0.45 number of doublings per day). Therefore, no statistically significant differences in this parameter were observed between peripheral and central chondrocytes overall and among clinics ( Fig. 2F and Fig. S1D).
Chondrogenic Capacity
Chondrogenic capacity of expanded chondrocytes was assessed by culturing the cells in micromass pellets in medium containing TGFβ. Cartilaginous extracellular matrix deposition, visualized by Safranin O staining, demonstrated that peripheral and central chondrocytes exhibit a variable capacity to form cartilaginous tissue. The quality of the tissue was scored using the grading system Bern Score (BS). We observed that both peripheral and central samples generated tissues with bad (BS <3.0), averaged (BS: 3.0–6.0), and good (BS: >6.0) cartilaginous quality ( Fig. 3A ). However, a higher percentage of peripheral (vs. central) chondrocytes produced pellets falling in the latter, good, category (60% vs. 40%, respectively). Consequently, BS was higher in tissues generated by peripheral versus central chondrocytes (6.5 ± 0.5 vs. 5.2 ± 0.4, P = 0.037; Fig. 3B ). No statistically significant interclinical difference in this parameter was observed (Fig. S1E). Biochemical analyses of the pellets demonstrated a correlation between the BS and the GAG/DNA contents ( Fig. 3C ). RT-PCR analyses of pellets revealed no statistically significant differences in the expression of COL1, COL2, and aggrecan among the groups (data not shown).
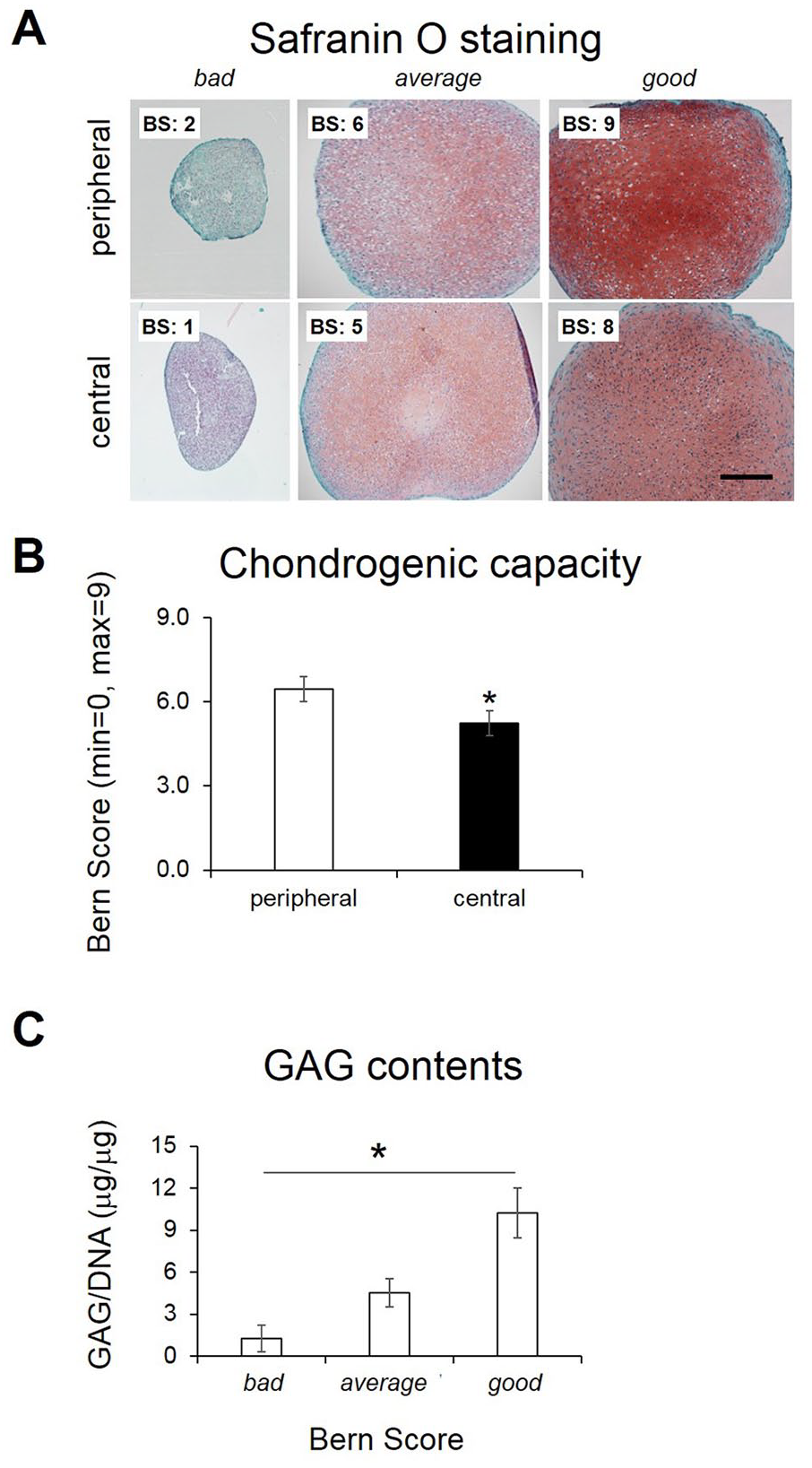
Cartilage forming capacity of chondrocytes derived from peripheral and central cartilage. (
Characterization of the Properties of Chondrocytes Isolated from Cartilage Tissues of Different Inflammatory Status
We then investigated whether the aforementioned properties of cartilage tissues/chondrocytes differed in samples derived from joints with different degrees of joint inflammation. For this purpose, synovial tissues were collected from the same joints from which cartilage samples were harvested. The majority of the collected synovium had inflammatory/degenerated appearances. Based on the expression level of IL6 and IL8, however, 2 inflammatory groups could be defined: high inflammation and low inflammation (differences in the expression levels of IL6 and IL8 in these 2 groups were 131.7-fold [P < 0.001] and 1081.3-fold [P < 0.001], respectively). Expression levels of IL6 and IL8 in the high and low inflammation groups were comparable to those measured in synoviocytes isolated from osteoarthritic patients (IL6: 0.291 ± 0.001, IL8: 0.153 ± 0.002 [N = 13; Kellgren and Lawrence grade 2-3]) or non-OA (healthy) patients (IL6: 0.057 ± 0.001, IL8: 0.027 ± 0.001 [N = 4]) during interventions performed at the Istituto Ortopedico Rizzoli, Bologna, Italy, respectively (results kindly provided by Prof. G. Lisignoli, Laboratorio di Immunoreumatologia e Rigenerazione Tissutale, Istituto Ortopedico Rizzoli; Fig. 4A ). Biopsies in the high and low inflammation categories were derived from patients with similar age (30 ± 9 vs. 33 ± 12 years), time of symptom onset (mainly >12 months), and severity of cartilage damage (mainly ICRS score 4). Hematoxylin-eosin staining of the synovial tissues demonstrated the presence of variable amounts of fibroblastic and adipocytic cells in both groups. Instead, inflammatory cells were reproducibly more present in the synovial tissues of the high inflammation group ( Fig. 4B ). Immunohistochemical analyses showed the presence of IL6 positive areas in the synovium of the high inflammation group (50% of the analyzed samples), while no detectable IL6 staining was observed in any synovium of the low inflammation group ( Fig. 4C ).
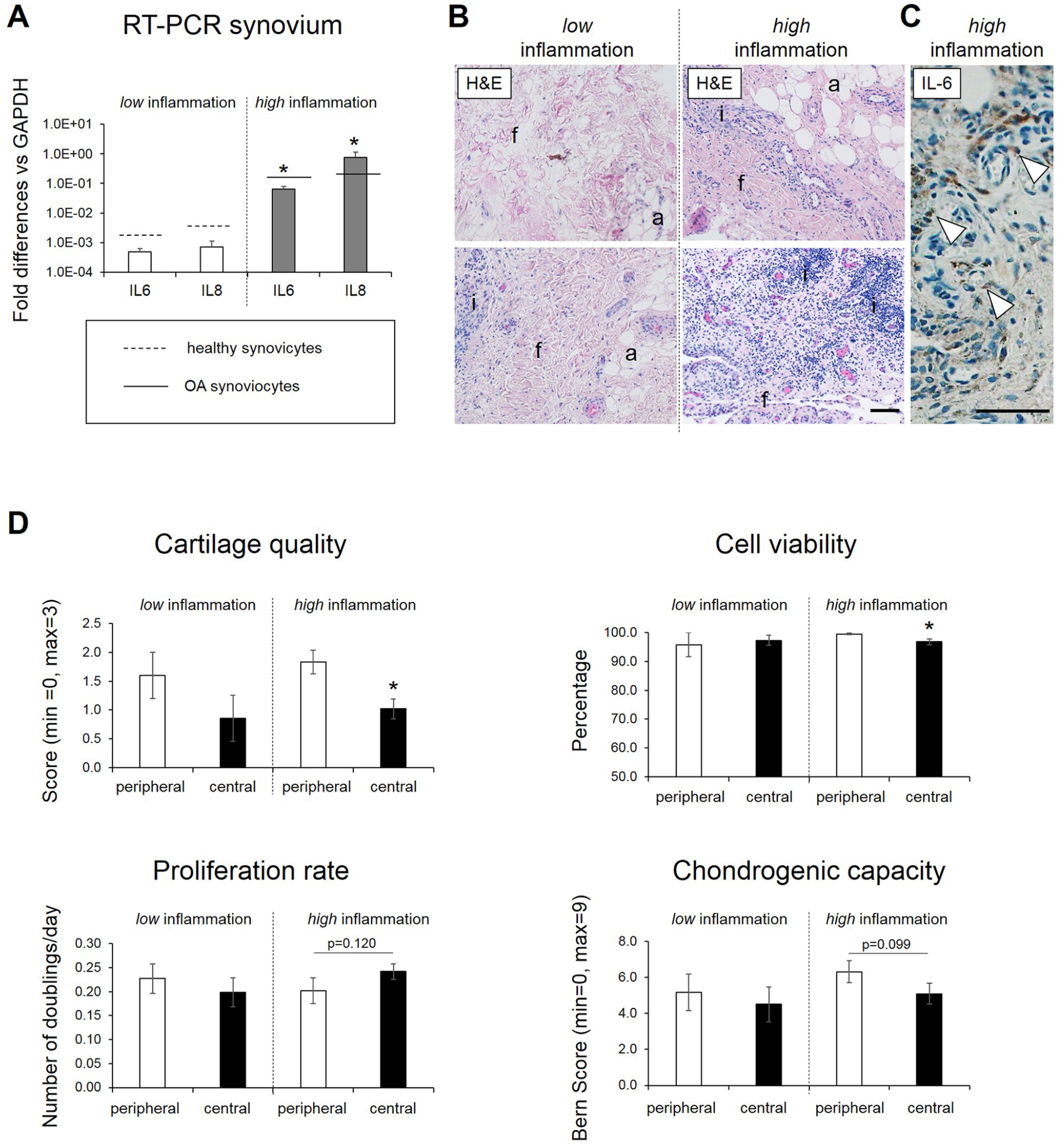
Properties of peripheral and central cartilage collected from joint having different inflammatory status. (
No statistically significant differences in any of the investigated parameters between peripheral and central samples were observed in the low inflammation group, probably due to the limited amounts of samples analyzed in this group (n = 7, corresponding to 18% of the total). Instead, in the high inflammation group (n = 27), cartilage quality (1.8 ± 0.2 vs. 1.0 ± 0.2, P = 0.009) and cell viability (99.5 ± 0.3% vs. 96.8 ± 1.0%, P = 0.003) were higher in the peripheral versus central samples ( Fig. 4D ). In addition, we observed a trend toward a higher proliferation capacity (proliferation rate: 0.24 ± 0.03 vs. 0.20 ± 0.03, P = 0.120) and lower post-expansion chondrogenic capacity (BS: 6.3 ± 0.6 vs. 5.1 ± 0.6, P = 0.099) between central versus peripheral chondrocytes ( Fig. 4D ).
Characterization of the Properties of Chondrocytes Isolated from Cartilage Tissues of Different Quality
Considering that the majority of the cartilage samples derived from high inflammatory joints, we decided to only use samples of this group for additional investigations aimed at assessing whether and to which extent chondrocyte phenotype and properties vary according to the quality of the tissue from which the cells were derived.
RT-PCR analyses of the chondrocytes after isolation showed that the phenotype of cells derived from central cartilage did not differ when derived from bad (grade 0-1) or good (grade 2-3) quality tissue as shown by similar level of the mRNA ratios for aggrecan/versican (Agg/Ver) and type II collagen/type I collagen (COL2/COL1). Contrarily, in peripheral chondrocytes significantly higher mRNA ratios of Agg/Ver (5.2-fold, P = 0.032) and COL2/COL1 (169.6-fold, P = 0.016) were observed for cells from the good- versus bad-quality cartilage tissue. Interestingly, peripheral chondrocytes from the good—but not bad—quality cartilage tissues had higher values of both ratios (22.4-fold, P = 0.016, and 121.5-fold, P = 0.032, respectively) than the corresponding central cells ( Fig. 5A and Table 2 ). For the central chondrocytes, expression levels of MMP13 and ADAMTS5 also remained unaffected irrespective of the quality of the starting tissue, while these factors were higher expressed by peripheral chondrocytes of bad- versus good-quality cartilage (31.3-fold, P = 0.008, and 2.1-fold, P = 0.056, respectively). Despite both deriving from good-quality starting tissue, the expression of MMP13 and ADMTS5 was, respectively, 19.3-fold (P = 0.056) and 3.0-fold (P = 0.111) higher in central chondrocytes as compared to peripheral chondrocytes ( Fig. 5A and Table 2 ). Noteworthy, despite these lower expression levels in peripheral chondrocytes (vs. central), they expressed the mRNA of these factors at a higher level than reference chondrocytes from uninjured healthy cartilage and at levels more similar to OA chondrocytes ( Fig. 5A ).
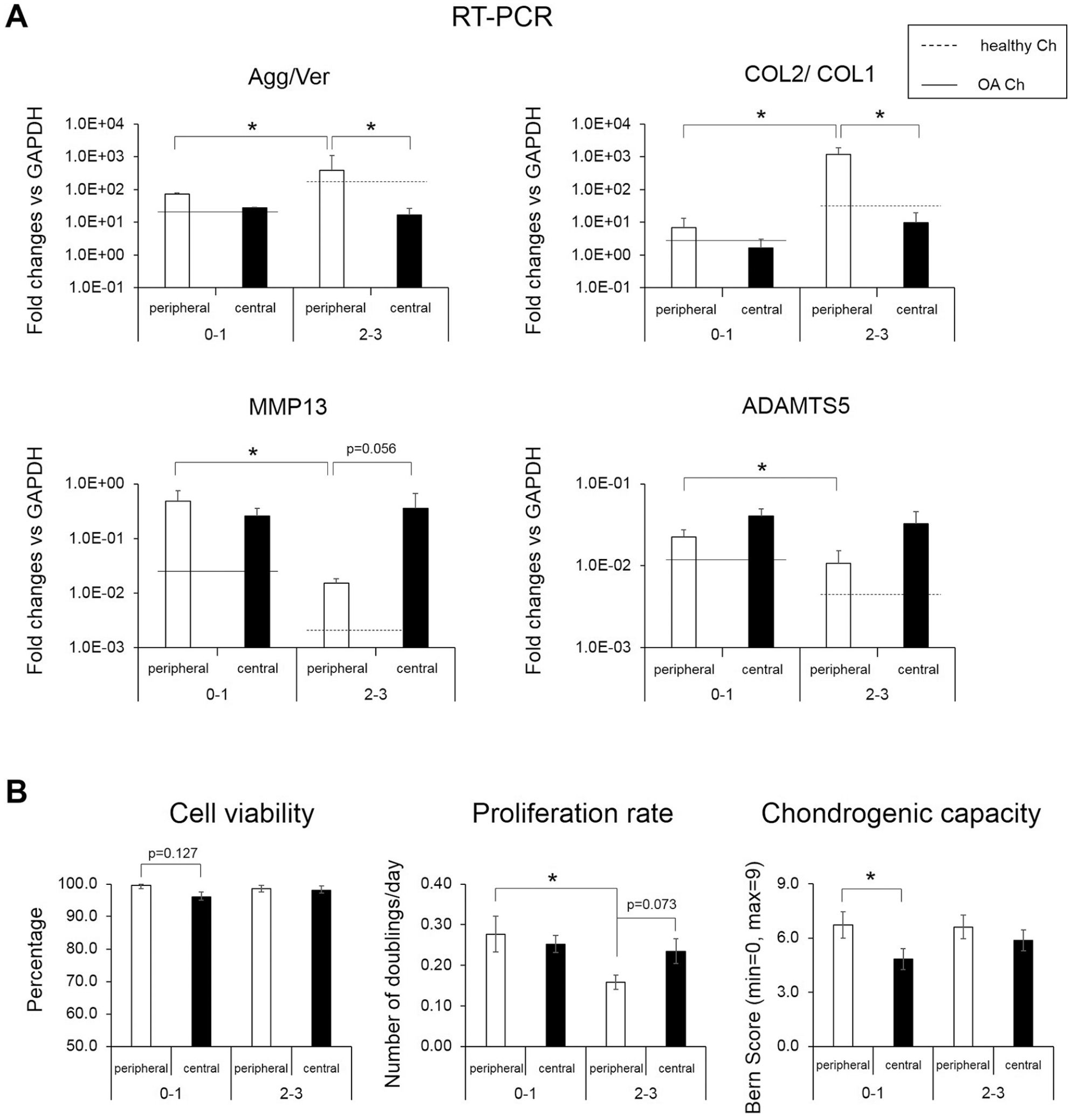
Properties of peripheral and central chondrocytes collected from cartilage tissues with different histological quality. (
Comparison of Chondrocytes from the Periphery and Center of Cartilage Lesions. Summary of the Differences between Peripheral (p) and Central (c) Chondrocytes Derived from Cartilage Tissues of Different Qualities.
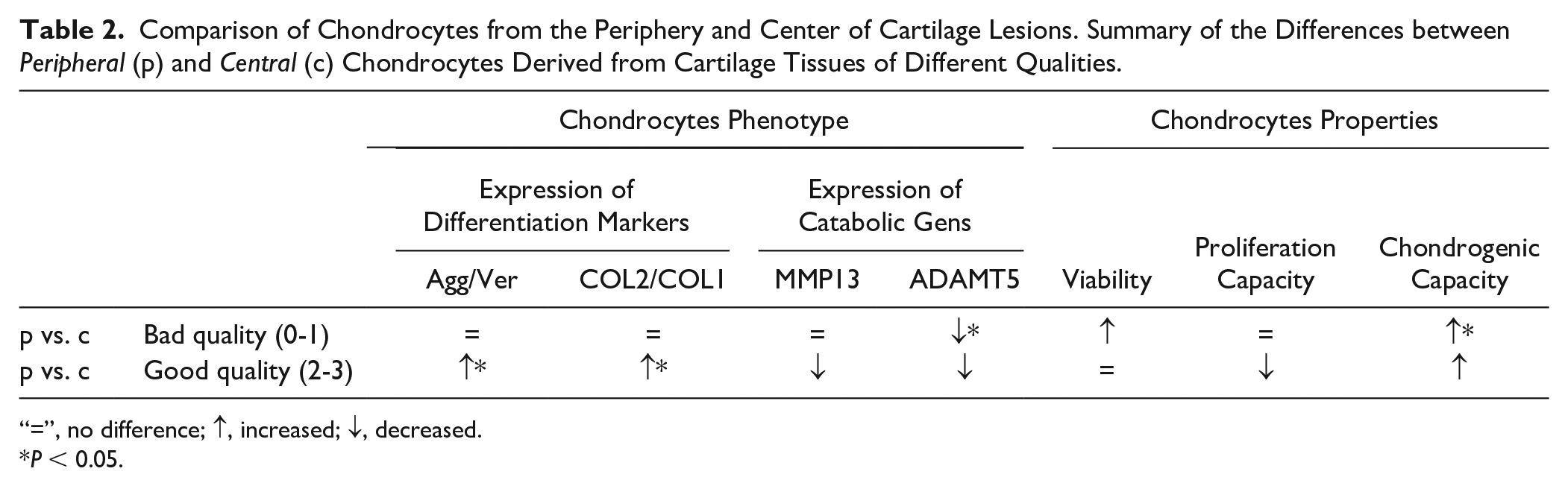
“=”, no difference; ↑, increased; ↓, decreased.
P < 0.05.
Among the different properties investigated, cell viability was observed to not significantly differ between chondrocytes (both peripheral and central) in bad- versus good-quality cartilage tissues, even if trends toward a lower percent of viable cells were seen between central versus peripheral chondrocytes in the bad-quality cartilage tissues ( Fig. 5B and Table 2 ). Proliferation rates of central chondrocytes did not differ in cartilage tissues of different quality. Instead, peripheral chondrocytes exhibited a reduced proliferation capacity (1.7-fold, P = 0.041) in tissue with good (vs. bad) quality, so that in these good-quality tissues this parameter was lower as compared to that of the central chondrocytes (1.5-fold, P = 0.073; Fig. 5B and Table 2 ). The post-expansion chondrogenic capacity of central and peripheral chondrocytes was observed not to significantly differ among tissues with different qualities. However, in bad-quality cartilage tissue peripheral chondrocytes were observed to have a superior post-expansion differentiation capacity as compared to central chondrocytes (P = 0.047; Fig. 5B and Table 2 ).
Discussion
We demonstrated that samples collected from the center of the cartilage lesion (central) exhibit lower qualities and contained chondrocytes less viable and with inferior cartilage-forming capacity as compared to closely adjacent (peripheral) areas. Even in highly inflamed joints, peripheral cartilage exhibited superior properties as compared to the central cartilage. Finally, we showed that even if peripheral chondrocytes from tissues with degenerated properties acquire phenotypic alterations, they maintained superior functional properties as compared to the central chondrocytes.
The overall relatively low interclinical variability in the measured parameters observed in our study highlights the importance of setting standard operating procedures for the collection human specimens for laboratory investigations. We collected samples from patients without radiological evidences of OA. However, since the localized cartilage degeneration was quite severe (ICRS score: 3-4), it cannot be excluded that also the macroscopically heathy appearing cartilage tissues may have acquired molecular changes, as characteristically described in (pre)osteoarthritic cartilage. 18 However, we observed that tissues collected in adjacent areas of a damaged cartilage displayed superior histological and molecular properties as compared to those collected in the lesions. In particular, peripheral cartilage samples contain more GAG and express higher levels of type II collagen and aggrecan mRNA as compared to the corresponding central cartilage samples. Our results are in agreement with those of Squires et al., 20 indicating inferior histological quality and amounts of type II collagen and aggrecan proteins in cadaveric cartilage collected from lesions as compared to those harvested from adjacent regions. Despite these differences, similar amounts of viable cells having good proliferation capacities were isolated in both peripheral and central cartilage samples. These results correlate with previous reports showing that chondrocytes isolated from damaged cartilage exhibit similar propensity to proliferate as compared to those isolated from normal cartilage tissues.8,21 Nevertheless, we here demonstrated that central chondrocytes exhibited an inferior post-expansion differentiation capacity as compared to peripheral chondrocytes. More in depth, molecular characterization are necessary to identify differences in the expression profiles between peripheral and central chondrocytes that can account for their distinct biosynthetic activities. In particular, it would be important to understand whether genes dysregulated in (pre-)osteoarthritic chondrocytes, including TGFβs, 22 BMPs, 23 and Wnts 24 are also differentially expressed in chondrocytes isolated in focal cartilage lesions and in adjacent areas to the lesion.
We were then interested to know whether the properties of cartilage and chondrocytes in peripheral versus central samples differed in joints of different inflammatory status. We thus assessed the inflammatory status of the patients’ joints by quantifying the mRNA expression level of 2 key inflammatory cytokines IL6 and IL8 in the harvested synovial tissues. We are aware that several cytokines might show dissociated patterns of mRNA and protein expression and, thus, posttranscriptional and posttranslational regulation of cytokine production must be taken into consideration. 25 However, Seitz et al. 26 showed that, among several inflammatory cytokines, IL6 and IL8 increased both at mRNA and protein levels in synovial fibroblasts from rheumatic patients in response to IL1β. Hence, we assume that the here measured high versus low mRNA expression levels of IL6 and IL8 by synovial tissue samples would reflect the high or low presence of these inflammatory mediators in the joints. The most important finding is that even in the high inflammation group, biopsies collected at the edges of the lesion better maintained features of healthy cartilage as compared to those from central regions (as demonstrated by the higher histological score) and contained more viable and competent chondrocytes to newly form cartilage after expansion).
We showed that the majority of central cartilage samples, but also a certain fraction of peripheral cartilage samples, display features of degenerated tissues. This is in agreement with the accepted theory that damage in posttraumatic joint “radiates out from the foci over time, progressively involving once-healthy cartilage.” 18 We thus addressed the so far not investigated issue whether phenotype and functional properties of chondrocytes differed if cells derived from cartilage areas within a joint with traumatic cartilage damage, having different degenerative appearances. Our results demonstrate that peripheral chondrocytes isolated from more degenerated cartilage tissues acquire a more de-differentiated phenotype and an enhanced expression of genes coding for cartilage-degrading enzymes, characteristics of osteoarthritic cells, 27 while those isolated from less degenerated cartilage tissue display a healthier phenotype. Instead, central chondrocytes display a similar aberrant phenotype if derived from cartilage tissue with good or bad quality. Consequently, they display a more pronounced tendency to proliferate and a reduced capacity to newly form cartilage tissue as compared to the peripheral counterparts. Interestingly, even if MMP13 and ADAMTS5 mRNA were expressed at lower levels in peripheral chondrocytes from good- versus bad-quality cartilage, their expression levels were higher than those measured in unaffected healthy cartilage and more similar to the ones in OA chondrocytes. These results indicate that although not macroscopically evident, a molecular shift toward an “early/pre-OA” phenotype has already occurred. In the long term, if left untreated, this could evolve to a fully defined OA pathology.1,28 These results are also in line with those reported in a previous study in which high expression levels of several degradative mediators (including MMP13 and ADAMTS5) were quantified in adjacent areas of cartilage lesions of 2 cadaveric joints. 29 Future studies in large animal models should be performed to elucidate whether and to which extent the removal of such healthy-like cartilage tissues from the periphery of the defect during refreshment of the cartilage lesion is required to ensure successful clinical outcomes of (cell based) therapies.
The current study has additional limitations to those previously discussed. First, the majority of the patients (~80%) had >12 months’ time since symptom onset. Therefore, we could not perform subgroup analysis for this key variable, 7 to assess whether the investigated properties differed with the degree of “chronicity” of the lesion. This important investigation would require the collection of a much larger number of samples as compared to the one available in the current study. Second, none of our analyses was performed at single cell level. Hence, future studies will be required to investigate whether the differences reported here between peripheral and central chondrocytes are due to alterations of specific cell subpopulations or by overall properties of the residents’ cells. Third, we did not perform any mechanical characterizations of the cartilage tissues (due to the limited size of the collected samples). However, considering previous published results showing inferior mechanical properties of fibrous cartilage versus hyaline cartilage tissues, 5 it is reasonable to state that the here classified as bad-quality cartilage has acquired altered mechanical properties. In addition, optical investigations on cartilage tissues using relatively new techniques, that is, electromechanical assessment 30 or Raman spectroscopy 31 could be considered to acquire more information on the quality/composition of peripheral versus central cartilage samples.
Conclusions
Our results indicate that—although cellular and molecular changes can be observed in healthy-like cartilage following trauma—chondrocytes at the edges of the lesion maintain healthier properties as compared to those isolated from the central parts. The superiority of these cells was maintained when joints were characterized as highly inflamed. Future studies will be necessary to investigate how functional properties of peripheral chondrocytes change over time and, consequently, to identify a “point of no return” (since first knee surgery or onset of symptoms) to define time windows to increase the likelihood of successful cartilage repair strategies. In addition, future studies using new technologies, such as laser-assisted methods to reproducibly debride cartilage lesions/areas, will be required to assess whether and to which extent removal of peripheral margins of the lesion is required to enable cartilage repair/regeneration.
Supplemental Material
FigS1 – Supplemental material for Comparison of Human Articular Cartilage Tissue and Chondrocytes Isolated from Peripheral versus Central Regions of Traumatic Lesions
Supplemental material, FigS1 for Comparison of Human Articular Cartilage Tissue and Chondrocytes Isolated from Peripheral versus Central Regions of Traumatic Lesions by Lina Acevedo, Lukas Iselin, Majoska H. M. Berkelaar, Gian M. Salzmann, Francine Wolf, Sandra Feliciano, Nicole Vogel, Geert Pagenstert, Ivan Martin, Karoliina Pelttari, Andrea Barbero and Markus P. Arnold in CARTILAGE
Supplemental Material
FigS2 – Supplemental material for Comparison of Human Articular Cartilage Tissue and Chondrocytes Isolated from Peripheral versus Central Regions of Traumatic Lesions
Supplemental material, FigS2 for Comparison of Human Articular Cartilage Tissue and Chondrocytes Isolated from Peripheral versus Central Regions of Traumatic Lesions by Lina Acevedo, Lukas Iselin, Majoska H. M. Berkelaar, Gian M. Salzmann, Francine Wolf, Sandra Feliciano, Nicole Vogel, Geert Pagenstert, Ivan Martin, Karoliina Pelttari, Andrea Barbero and Markus P. Arnold in CARTILAGE
Supplemental Material
Supplementary_File_Table_S1-2 – Supplemental material for Comparison of Human Articular Cartilage Tissue and Chondrocytes Isolated from Peripheral versus Central Regions of Traumatic Lesions
Supplemental material, Supplementary_File_Table_S1-2 for Comparison of Human Articular Cartilage Tissue and Chondrocytes Isolated from Peripheral versus Central Regions of Traumatic Lesions by Lina Acevedo, Lukas Iselin, Majoska H. M. Berkelaar, Gian M. Salzmann, Francine Wolf, Sandra Feliciano, Nicole Vogel, Geert Pagenstert, Ivan Martin, Karoliina Pelttari, Andrea Barbero and Markus P. Arnold in CARTILAGE
Footnotes
Acknowledgments and Funding
We thank Dr. Amir Steinitz (University Hospital Basel) and Dr. Raphael Kaelin (Practice LEONARDO) for their help in the collection of several tissues and Dr. Gyözö Lehoczky (University-Hospital Basel) for his help in the cell culture work. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Stiftung Lindenhof Bern, Switzerland (Grant Number: 14-05-F), and the Swiss National Science Foundation (SNF Project Numbers: 310030_175660/1 to AB and PMPDP3_151396/1 to KP).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of Basel (No. EKNZ 2014-199/PB_2016-09125).
Informed Consent
All patients gave their informed consent for inclusion before they participated in the study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.