Abstract
Objective
The aim of this study was to investigate early radiological and clinical outcome of autologous minced cartilage treatment as a single-step treatment option in patients with a chondral or osteochondral lesion (OCL) in the knee.
Design
Eighteen patients with an OCL in the knee were included. Cartilage from healthy-appearing loose bodies and/or the periphery of the defect were minced into small chips and sealed in the defect using fibrin glue. Preoperatively, and at 3 (n = 14) and 12 (n = 18) months follow-up, magnetic resonance imaging (MRI) was performed. The Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) 2.0 score was used to assess the cartilage repair tissue on MRI at 12 months. The International Knee Documentation Score, Knee Injury and Osteoarthritis Outcome Score, EuroQoL-5D, and Visual Analogue Scale pain were collected preoperatively and 12 months after surgery.
Results
Three months postoperative, MRI showed complete defect filling in 11 out of 14 patients. Mean MOCART 2.0 score at 12 months was 65.0 ± 18.9 with higher scores for lateral femoral chondral lesions compared to medial femoral chondral lesions (75.8 ± 14.3, 52.5 ± 15.8 respectively, P = 0.02). Clinical and statistical significant improvements were observed in the patient-reported outcome measures at 12 months postoperatively compared to preoperatively.
Conclusion
Treatment of OCLs using the autologous minced cartilage procedure resulted in good cartilage repair measured by MOCART 2.0. Clinically relevant improvements were observed in the clinical scores. This study suggests autologous minced cartilage as a promising, single-step treatment for OCLs.
Introduction
Chondral and osteochondral lesions (OCLs) are a common cause of knee pain, swelling and loss of function in young and active patients. If left untreated, these defects have a high chance to progress in size and initiate the process of osteoarthritis (OA).1 -4 Typically, patients with focal cartilage defects are young and active, and therefore joint replacement should be avoided. Hence, effective joint-preserving treatments with predictable outcome are necessary for this group of patients. 5
Treatment of OCLs is hampered by the limited capacity for self-repair of articular cartilage6-10 due to the absence of pluripotent cells, a sparse distribution of chondrocytes and the lack of vasculature. 11 Treatment of an OCL by surgical intervention should ideally generate repair tissue that resembles the structure and function of hyaline cartilage as close as possible, and prevent deterioration into OA in the long term.
Current treatment options for OCL include marrow-stimulating techniques such as microfracture and subchondral drilling, transplant procedures such as mosaicplasty and osteochondral autograft transfer system (OATS), and cell-based therapies such as autologous chondrocyte implantation (ACI) and autologous particulated cartilage chips. Microfracture is a relatively easy procedure, indicated for small lesions (<2 cm2) that provides short-term improvement. 12 However, defects treated by microfracture deteriorate between 18 and 36 months after surgery, show higher failure rates, and shorter time to failure in comparison to other techniques.13-16
Compared to microfracture, transplant procedures show better results in terms of pain and function in the short- and mid-long term,17,18 and lower reoperation rates, 19 but often result in donor site morbidity.20,21 ACI is considered to generate the best quality repair tissue and shows good long-term clinical results, 19 however is costly and is a 2-step procedure.
Each of these currently available cartilage-restoration techniques have their own limitations, such as limited defect size that can be treated or necessity of a 2-step procedure. According to the consensus statement of the Dutch Orthopaedic Society for OCL repair, ACI is advised in the larger and more challenging complex OCLs. 22 ACI is, however, been unavailable in the Netherlands in the time-period during which patients in this paper were treated due to insurance issues and until recently, ACI was not indicated in the age group younger than 18 years of age. 23 In view of these limitations, novel techniques are being developed with the goal of producing high-quality repair tissue in a single-step procedure, without high cell-producing costs and extensive national regulations/legislation.
An alternative to the aforementioned techniques is using small cartilage chips harvested from the patient’s knee in a single step procedure named the autologous minced cartilage procedure.24,25 Chondrocytes from these cartilage chips embedded in fibrin glue have been shown to migrate from their surrounding matrix and form new extracellular matrix (ECM).26,27 This procedure was first described by Albrecht et al. 28 in 1983 and was developed further ever since for the treatment of chondral lesions.17,24,26,29,30 OCLs have successfully been treated by Christensen et al. 31 using autologous minced cartilage chips along with fragmented autologous bone press-fitted into the bed of the defect. Thus, chondral lesions as well as OCLs can be treated using the autologous minced cartilage procedure. The aim of this study was to evaluate the radiological and clinical outcome 12 months after the 1-step autologous minced cartilage procedure performed in patients with an OCL of the knee. In contrast to previously published studies, no scaffold or covering membrane was used to secure the cartilage pieces besides fibrin glue. Magnetic resonance imaging (MRI) was used to assess the effectiveness of this new approach in terms of defect fill at 3 months, and to score the repair tissue at 12 months.
Materials and Methods
Study Design and Patient Selection
Patients in this study were retrospectively included from a continuous cartilage-repair registry of the knee in which data were prospectively collected. The study was conducted at a single academic hospital specialized in cartilage repair and regeneration (Joint Preserving Clinic, Maastricht University Medical Center, The Netherlands). Patients suffering from an OCL of the knee between June 2017 and January 2020 were identified through the clinical database. Patients were included if they underwent surgical treatment for an OCL of the femoral condyle, patella, or trochlea by the autologous minced cartilage procedure, and had a preoperative and 12-month follow-up MRI available. Patients were selected for the autologous minced cartilage procedure if they had loose, vital appearing chondral fragments or if they suffered from osteochondritis dissecans lesions that could not be re-fixated. Other indications were patellar lesions or defects > 4 cm2. This study was approved by the local ethical committee (METC 2021-2898).
Surgical Procedure
The autologous minced cartilage procedure was performed with or without autologous cancellous bone grafting, depending whether a bony defect was present. The surgical procedures were performed by 2 specialized knee surgeons (PJE and TAB). The surgery started with a diagnostic arthroscopy of the affected knee to set the final indication, inspect the whole joint, rule out the presence of additional intra-articular abnormalities, and remove loose cartilage fragments. After finishing the arthroscopic procedure, the portals were closed with a single transcutaneous suture.
Subsequently, a medial or lateral parapatellar (mini-)arthrotomy was performed depending on the location of the lesion. The lesion was treated using the autologous minced cartilage technique, as described by Massen et al.
25
In short, after the lesion size was measured, the defect was debrided using a scalpel or ringed curette until a stable, viable cartilage wall was encountered (
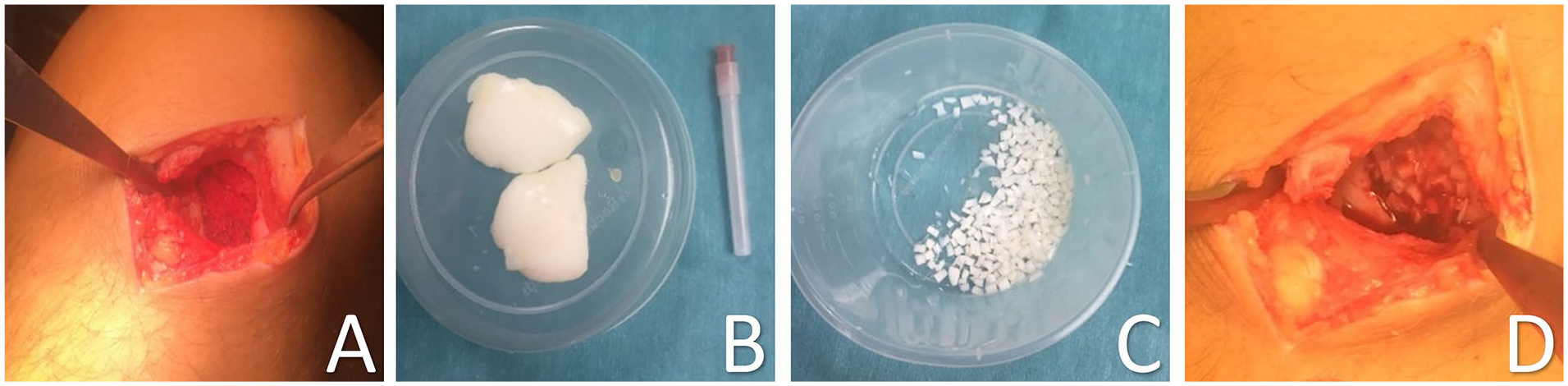
Defect of the lateral femoral condyle after debridement creating a stable viable cartilage wall (
Whenever there was sclerotic or avital bone at the bottom of the defect, this was debrided until healthy trabecular bone was encountered and if subchondral bleeding was encountered this was sealed with fibrin glue (TISSEEL, Baxter International, Deerfield, IL, USA) (Baxter Netherlands). In case of an osteochondral defect deeper than 4–5 mm, autologous cancellous bone was harvested from the proximal tibia through a cortical window. The cancellous bone fragments were impacted in the bony part of the defect to the level of the subchondral plate and sealed with fibrin glue (TISSEEL) as described by Christensen et al.
31
(
Hereafter, the minced cartilage chips were placed in the defect flush or slightly below the level of the adjacent cartilage and sealed using fibrin glue. The fibrin glue was not diluted. No collagen membrane was used. Once the fibrin glue was set, multiple flexion- and extension movements were made to verify that the cartilage chips did not dislocate from the defect or catch on opposing tissue.
Coexisting intra- and/or extra-articular abnormalities, that is, meniscus tear, patellar dislocation or patellofemoral maltracking, anterior cruciate ligament rupture, and mechanical axis deviation, were treated in the same procedure to protect the repaired cartilage. 32
Postoperative Rehabilitation
Rehabilitation was led by a physical therapist, using guidelines of previously published protocols.33-35 The focus of the rehabilitation program was to protect the newly repaired cartilage by limiting the range of motion of the knee and gradual weightbearing, depending on the location of the defect. Guidelines and timelines differed slightly depending on the location of the defect.
For condylar lesions: 2 weeks non weight-bearing, 2 weeks 25% loading, 2 weeks 50% loading, return to 100%. For patello-femoral lesions full weight bearing was allowed, range of motion was gradually expanded using a hinged knee brace: 2 weeks 0–30 degrees, 2 weeks 0–60 degrees, and 2 weeks 0–90 degrees, progression to full range of motion. Progressive muscle strengthening was pursued in a step-wise program.
Radiological Data Collection
MRI was performed pre-operatively to assess cartilage, subchondral bone, and soft tissues. Imaging was performed on whole-body 1.5 and 3 Tesla imaging systems using an extremity coil. The MRI protocol consisted of sagittal, coronal, and transversal images acquired using a turbo spin echo (TSE) sequence with and without fat suppression (FS) and a sagittal 3-dimensional proton density sequence (PD), similar to the protocol described by Schreiner et al. 36
All MRIs were evaluated by an experienced musculoskeletal radiologist (DL). Pre-operative MRIs were used to assess the size of the lesions and define the lesions as either pure chondral or osteochondral. The MRI scans at 3 months follow-up were used to assess the repair site and to quantify the fill of the defect in terms of depth. Repair sites were scored as complete fill, partial fill, or complete delamination of the implanted cartilage chips.
The 12 months follow-up MRI was graded using the Magnetic Resonance Observation of Cartilage Repair Tissue (MOCART) 2.0 score. 36 This score consists of seven parameters that assess the quality of the repair tissue that can be combined to a total score. The total score ranges between 100 (hyaline cartilage/best achievable repair tissue) and 0 (worst score).
Clinical Data Collection
Pre-operatively and 12 months after surgery, 4 different questionnaires were filled out by the patient at the outpatient clinic: the Pain Visual Analog Scale37,38 (Pain VAS; 0–100, 0 indicating no pain, 100 indicating unbearable pain), International Knee Documentation Committee Subjective Knee Evaluation Form39,40 (IKDC; 0–100, 100 being the highest score), the Knee Injury and Osteoarthritis Outcome Score40,41 (KOOS; 0–100 for five separately scored subscales; pain, other symptoms, function in daily living, function in sport, and knee-related quality of life, 100 being the highest score), and the 5-level EuroQol 42 (EQ-5D; 1-5 for five domains, 1 being the highest score).
The EQ-5D measures health-related quality of life in five domains (mobility, self-care, usual activities, pain/discomfort, and anxiety/depression). The EQ-5D index score was calculated based on a Dutch value set, representative of the Dutch population with regard to age and gender.43-45 Missing items in the Patient Reported Outcome Measures (PROMs) were treated as described by Irrgang et al. 39 and Roos et al. 46 For the IKDC a maximum of 2 missing values was accepted and the IKDC score was calculated based on the maximum possible score (of the filled out items). For the 5 subscales of the KOOS at least 50% of the questions needed to be filled out and the score was calculated based on the maximum possible score of the completed questions.
Statistical Analysis
Statistical analysis was performed using SPSS (IBM SPSS Statistics for Windows, Version 27.0. Armonk, NY: IBM Corp). All data were tested for normality using the Shapiro-Wilk test. To compare preoperative with follow-up variables, paired t-tests were performed on normally distributed data, while nonparametric data were analyzed using the Wilcoxon signed rank test, and Mann-Whitney-U test.
Correlations were performed using Pearson and Spearman correlation coefficient (Pearson R and Spearman’s rho respectively). Possible relationships between MOCART score and patient characteristics (age, BMI), as well as between MOCART score and defect characteristics (location, size, bone grafting), and MOCART score and PROMs were studied. Furthermore, PROMs were examined for possible relations with patient characteristics, and defect characteristics. Differences between defect locations were compared using Kruskal-Wallis analysis and post-hoc Bonferroni correction. All statistical tests were 2-sided. Descriptive results are demonstrated as mean ± standard deviation (SD) unless otherwise specified. The significance level was defined as P ≤ 0.05 for all tests.
Results
Patients and Demographics
Eighteen patients were included in this study, with a mean follow-up of 14.1 ± 2.7 months. Postoperative MRI was performed at 3.2 ± 0.8 months and 13.3 ± 2.7 months. Demographic data are displayed in Table 1 . Four patients had undergone previous surgery of the knee, of which one had an intervention for the index lesion (arthroscopic fixation in another hospital). Arthroscopic removal of loose bodies was performed in another hospital in 3 patients.
Patient Demographics (n = 18).

Data are presented as mean ± SD.
Lesions were located at the medial femoral condyle (MFC) (n = 8, 44%), the lateral femoral condyle (LFC) (n = 6, 33%), and patella (n = 4, 22%). No lesions of the trochlea were encountered. In 10 patients, autologous cancellous bone grafting was performed due to the depth of the defect. In 7 patients one or more concomitant procedures were performed. Defect characteristics and surgery details are displayed in Table 2 .
Defect Characteristics, Concomitant Procedures, Perioperative.
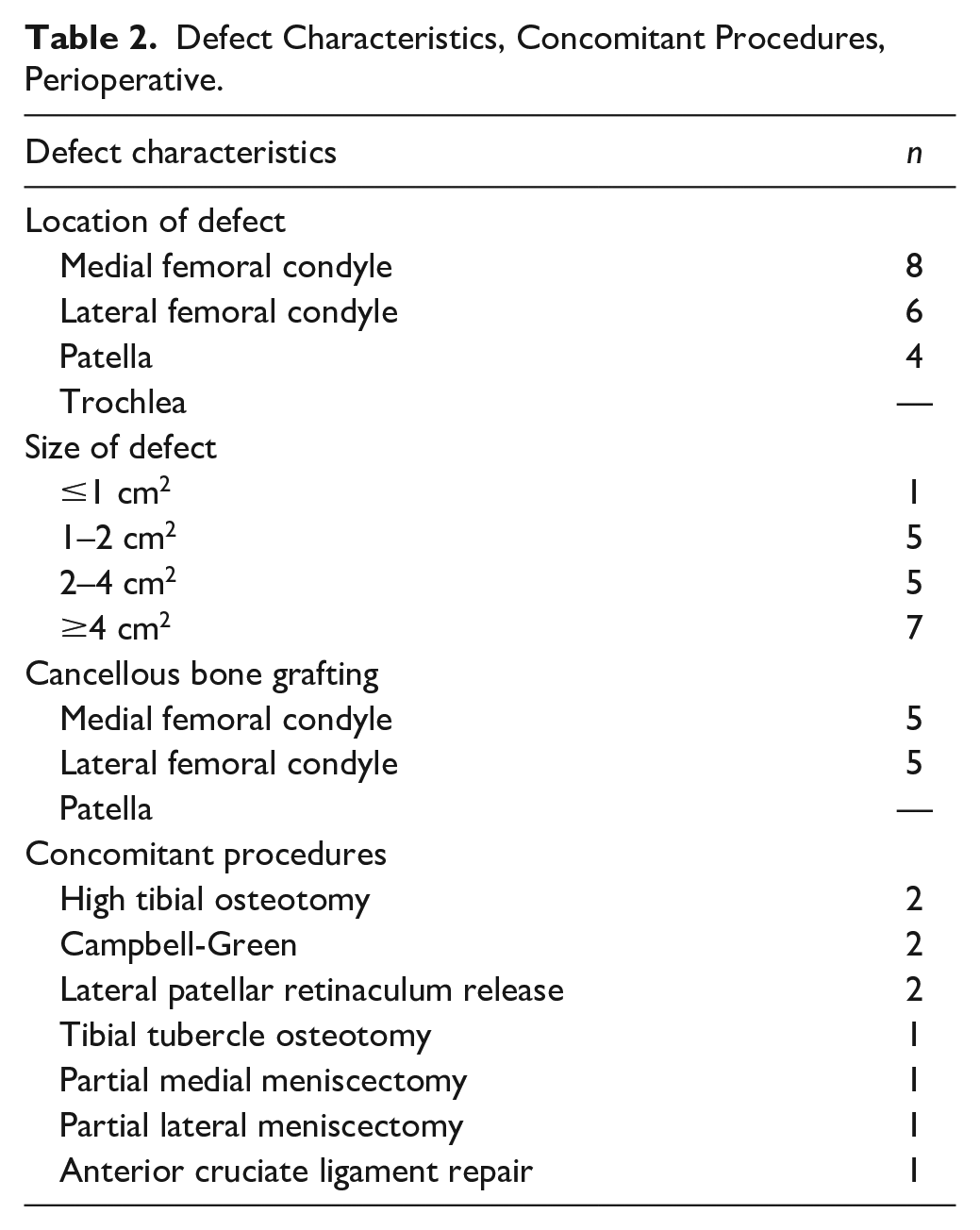
At 12 months follow-up, 1 patient had been reoperated arthroscopically for hypertrophic overgrowth of the repair tissue. One out of 2 patients who had undergone a concomitant high tibial osteotomy, had the implanted hardware removed.
One serious adverse event occurred during high tibial osteotomy: perioperatively the popliteal artery was damaged, for which vascular repair was indicated and a transient neuropathy of the peroneal nerve was diagnosed.
Radiological Results
Defect sizes scored on pre-operative MRI ranged from 0.24 to 7.8 cm2, with a mean of 3.3 ± 2.1 cm2. At 3 months after surgery, 14 out of 18 patients underwent MRI. In 11 patients (79%), a complete fill of the defect was seen, indicating that the cartilage chips did not migrate. The remaining 3 patients had a partial fill of the defect on MRI as can been seen in Figure 2 . Table 3 presents an overview of the MOCART scoring based on MRI at 12 months postoperatively per patient, including relevant patient characteristics and concomitant procedures. The total MOCART score at 12-month follow-up ranged from 35 to 100, with a mean of 65.0 ± 18.9. The total MOCART scores are displayed by location in Figure 3 . Defects of the MFC had significant lower MOCART scores compared to defects of the LFC (mean MOCART: 52.5 ± 15.8 vs 75.8 ± 14.3 respectively, P = 0.02).
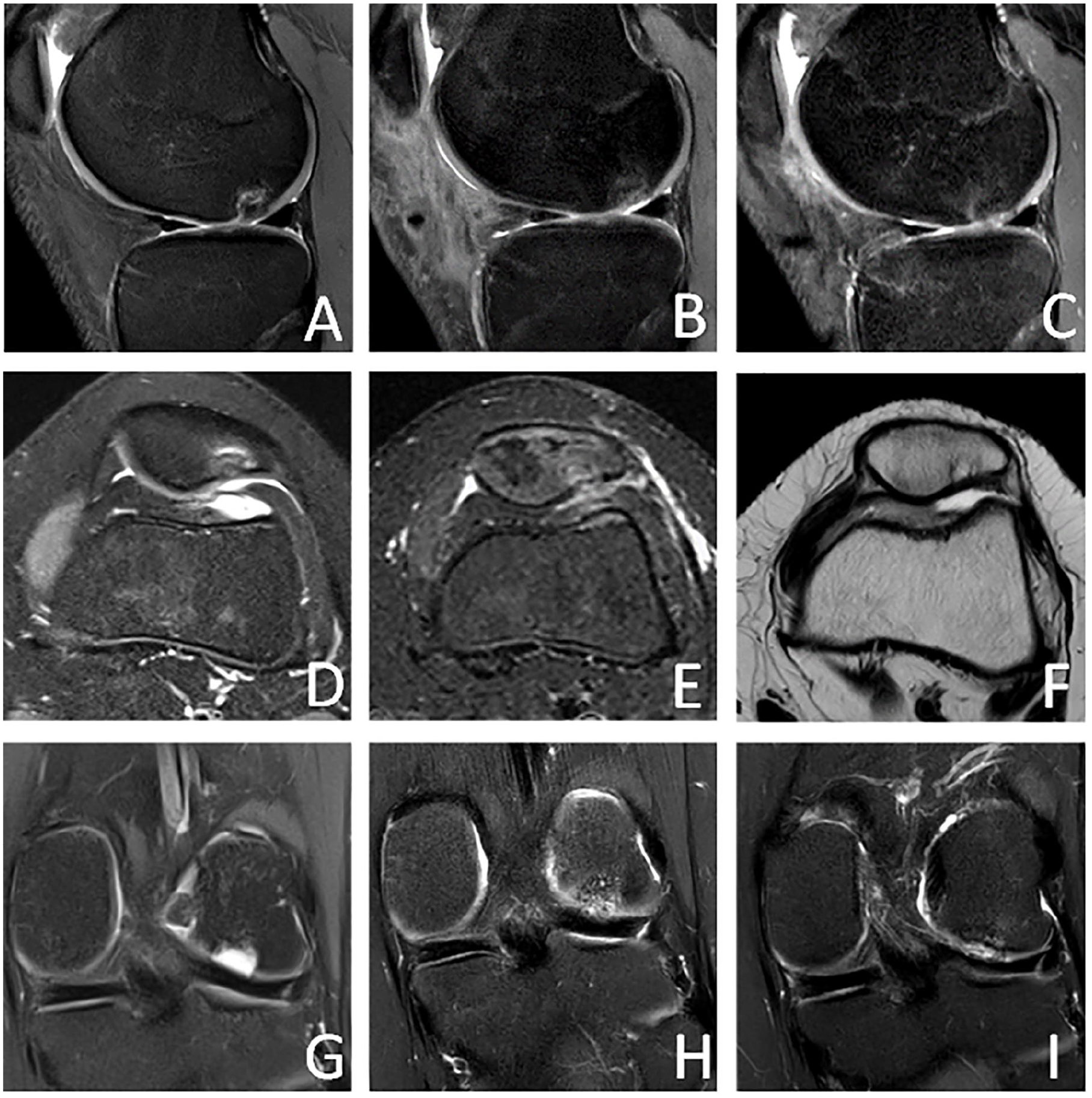
Top row: Pre-operative MR image showing the full thickness cartilage lesion with a 5mm bony defect at the lateral femoral condyle (
Overview of MOCART Scoring Per Patient, Including the Relevant Patient Characteristics and Concomitant Procedures.
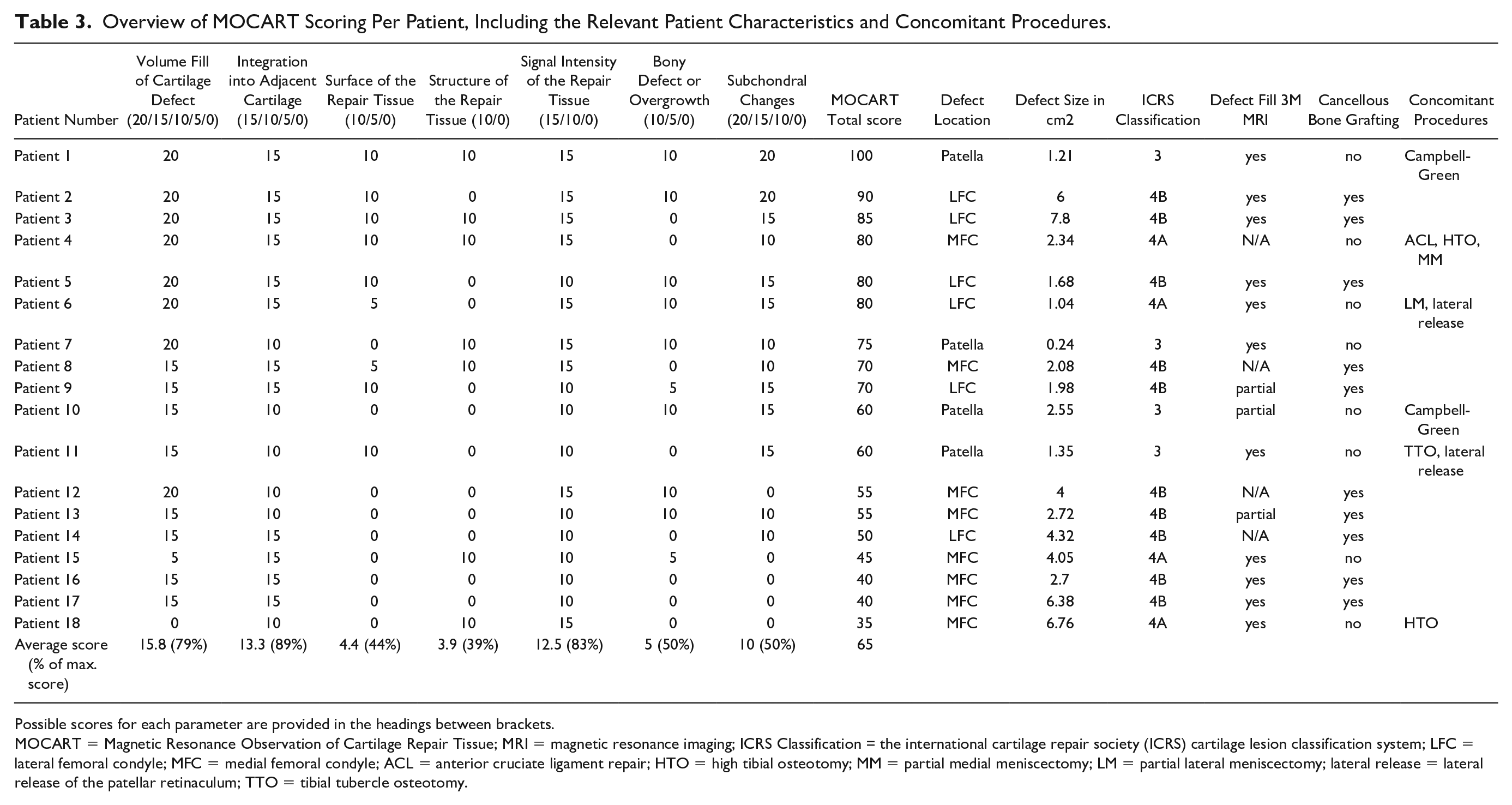
Possible scores for each parameter are provided in the headings between brackets.
MOCART = Magnetic Resonance Observation of Cartilage Repair Tissue; MRI = magnetic resonance imaging; ICRS Classification = the international cartilage repair society (ICRS) cartilage lesion classification system; LFC = lateral femoral condyle; MFC = medial femoral condyle; ACL = anterior cruciate ligament repair; HTO = high tibial osteotomy; MM = partial medial meniscectomy; LM = partial lateral meniscectomy; lateral release = lateral release of the patellar retinaculum; TTO = tibial tubercle osteotomy.
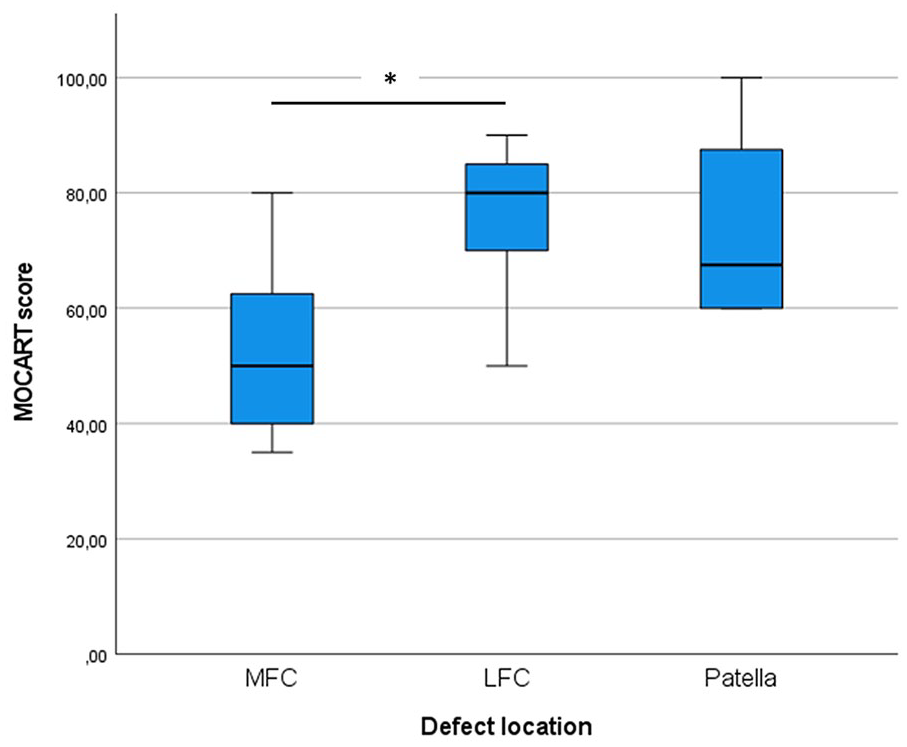
MOCART score per defect location. The asterisk depicts a statistically significant difference between the MOCART score of LFC and MFC lesions (p = 0.02). MOCART = Magnetic Resonance Observation of Cartilage Repair Tissue; LFC = lateral femoral condyle; MFC = medial femoral condyle.
No significant correlation was found between MOCART score and defect size (Pearson R = -0.305, P = 0.219). However, defects <2.5 cm2 had significant higher MOCART scores (P = 0.02) than defects >2.5cm2.
Neither cancellous bone grafting (mean MOCART: 63.5 ± 18.1 vs 66.9 ± 21.0 for bone grafting and no bone grafting respectively, P = 0.73), BMI (Pearson R = -0.021, P = 0.935), or age at time of surgery (Spearman’s rho = -0.222, P = 0.376) were significantly related to MOCART score at 12 months follow-up.
Clinical Results
A statistically significant improvement from preoperative to 12 months postoperative was found in the VAS pain, IKDC score, all 5 subscales of the KOOS, and the EQ-5D-5L ( Table 4 ). No correlation was found between the clinical outcome scores and the size of the defect. Also, the use of a cancellous bone graft was not related to the clinical scores, neither was the ICRS score of the defect related to clinical scores or the MOCART score.
Patient Reported Outcome Measures.
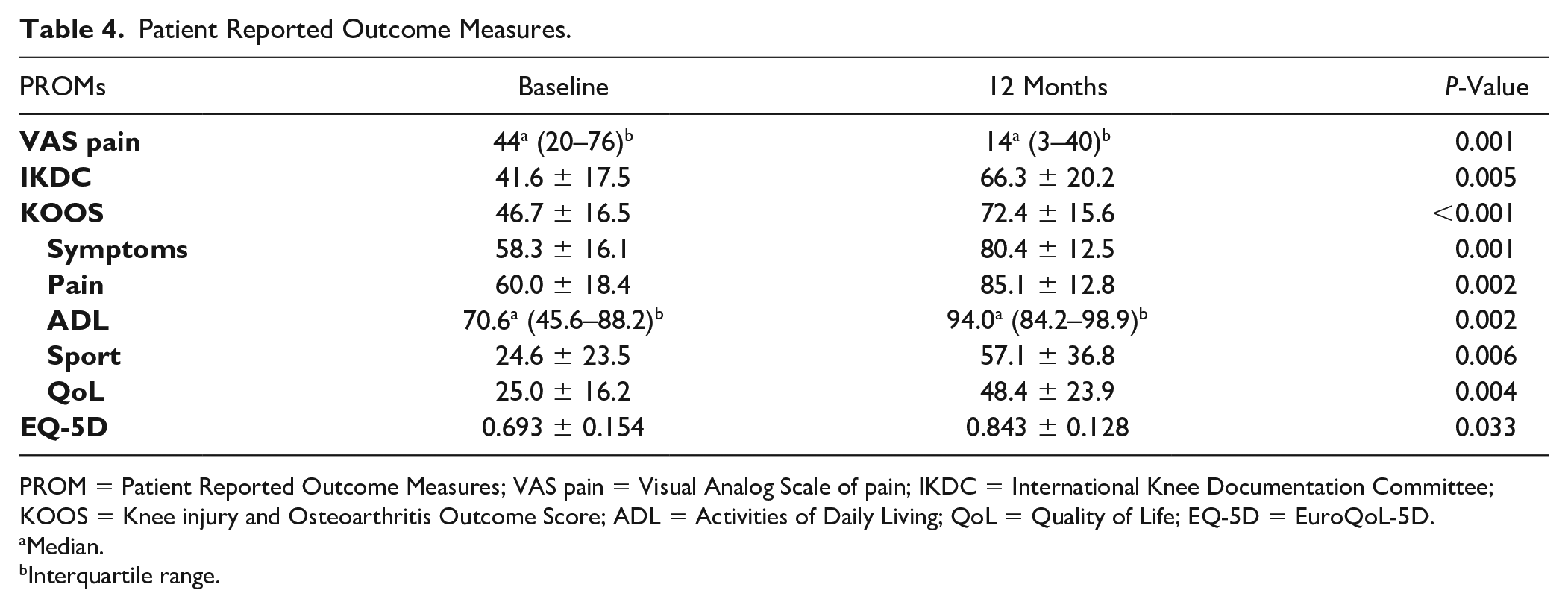
PROM = Patient Reported Outcome Measures; VAS pain = Visual Analog Scale of pain; IKDC = International Knee Documentation Committee; KOOS = Knee injury and Osteoarthritis Outcome Score; ADL = Activities of Daily Living; QoL = Quality of Life; EQ-5D = EuroQoL-5D.
Median.
Interquartile range.
IKDC and KOOS QoL subscale were moderately and statistically significantly correlated to the MOCART score (Pearson R = 0.529, P = 0.05, and Pearson R = 0.541, P = 0.05 respectively).
BMI of the patient was negatively correlated with the KOOS pain subscale (Pearson coefficient R = -0.597, P = 0.02), so the higher the BMI of a patient, the more pain he or she experiences. Furthermore, age was negatively correlated with the KOOS ADL subscale (Spearman’s rho = -0.612, P = 0.02), so the older the patient is, the more limitations in daily life he or she experiences (Supplementary material for this article is available online).
Discussion
The aim of this study was to report early radiological and clinical outcome of a single-step autologous minced cartilage procedure for OCLs of the knee. The primary finding of this study was a good healing response of the repair tissue as measured by MOCART score. Moreover, this study demonstrates improvements in pain, knee function, and quality of life 12 months after surgery.
The mean MOCART score (65.0 ± 18.9), observed 12 months after surgery, is comparable to the results found previously with the minced cartilage technique. Christensen et al. 31 observed a MOCART score of 52.5 ± 14.4 1 year after autologous dual tissue transplantation and Massen et al. 25 found a MOCART score of 40.6 ± 21.1 at 6 months after surgery. The MOCART score shows continuous improvement over time47,48 and therefore, no direct comparison can be made between the MOCART scores presented by Massen et al. 25 and the scores presented in the current study and in the study by Christensen et al. 31 ACI is still considered to result in the best quality repair tissue. 19 This is supported by McCarthy and Roberts. 49 who found a median MOCART score of 70 in 163 patients 1 year after ACI using either a periosteal or collagen membrane (first generation ACI). Recently published studies looking into the results of matrix-assisted chondrocyte implantation (MACI), and ACI with scaffold (second/third generation ACI) found MOCART scores of 76 ± 16, 47 and 78 ± 13 50 2 years after surgery.
The VAS pain showed a median score of 13.5 (2.8–39.8) at 12 months follow-up compared to 44.0 (20.0–76.0) before surgery. This improvement in VAS (30.5 points on a 0–100 scale) is comparable to the average outcomes found in studies of first-, second-, and third generation ACI looking into the VAS pain on a 0–10 scale: Nawaz et al. 51 found a decrease in VAS pain of 2.39 in 827 patients at a mean follow-up of 6.2 years, a systematic review by DiBartola et al. 52 found a mean decrease in VAS of 36.4% (3.64 points on a 10 point scale) at a follow-up of 4 to 6 years.
Knee function score at 12 months postoperatively, as reflected by the IKDC and KOOS, showed significant improvement compared to pre-operative scores. These improvements of 25 and 26 points, respectively, are larger than the minimal clinically important changes (MIC) of these scores (MIC IKDC 11 points, 53 MIC KOOS 8–10 points) 46 . Moreover, these scores are regarded as substantial clinical benefit, 54 and reached the Patient Acceptable Symptomatic State threshold. 55
The IKDC and KOOS scores are comparable to studies looking into the results of ACI. Schlumberger et al. 56 found IKDC and total KOOS scores of 66 ± 10 and 73 ± 19 points, respectively, at 31 months follow-up. Niemeyer et al. 47 found improvements in IKDC and total KOOS scores of 24.2 ± 16.9 and 24.9 ± 17.4, respectively at 24 months follow-up.
The IKDC score might be an important predictor for long-term outcomes according to a systematic review by Howard et al. 57 comparing the various patient-reported outcome measures used in assessing the effects of ACI.
The use of small autologous cartilage pieces to treat OCLs was first demonstrated by Albrecht et al. 28 in 1983 in an experimental rabbit model. More recently, chondrocytes have been observed to migrate from these cartilage chips and form new ECM.26,27 Moreover, chondrocytes are viable when cartilage is cut using sharp instruments58,59 as opposed to blunt trauma as can be seen at the edges of osteochondral autografts in mosaicplasty. 60 Increased fragmentation of the cartilage pieces positively affects the production of ECM in vitro, 61 indicating the importance of extensive fragmentation until a paste-like consistency has been reached.
Preparation of the cartilage defect consists of creating a stable, viable cartilage wall and removal of the calcified layer at the bottom of the defect. The harvested cartilage from the defect edge is vital25,62 and shows superior redifferentiation of chondrocytes compared to non-weight bearing cartilage.63,64
Also, the shear-off lesions remain viable, as we also have seen with the “modified hedgehog technique,” where we press-fitted back the flake, and ingrowth of vital cartilage was seen after 1 year. 65 While we are aware that this is still a topic for discussion, we chose to harvest from the defect edge in this technique and avoid possible damage of healthy zones of the joint. A healthy joint status leads to higher chances of success for cell-based techniques. 66 Moreover, future studies may consider techniques for per-operative testing of graft quality.
Other sources for cartilage harvest are macroscopically healthy appearing loose bodies67,68 or non-weight bearing regions such as the intercondylar notch or lateral rim of the trochlea. Cartilage harvested from the edge of the defect and any existing loose bodies provided enough cartilage pieces to cover the defect in all patients.
A number of different techniques have been described to secure the cartilage pieces. Cole et al. 24 used fibrin glue in conjunction with a synthetic scaffold (CAIS, discontinued product) showing good results in 24 patients at 2 year follow up. Qualitative MRI did not favor MFX or CAIS in terms of graft fill, integration into adjacent cartilage, or presence of subchondral cysts. However, IKDC and KOOS were statistically and clinically significant better in the CAIS group at 2-year follow-up. Christensen et al. 31 used fibrin glue only to fixate osteochondritis dissecans lesions by combined autologous bone and cartilage chips transplantation showing good MOCART scores of 52.5 ± 14.4 at 1 year follow-up and significant improvements in IKDC, 4 KOOS subscales and Tegner score.
Massen et al. 25 combined the use of fibrin glue with a Chondro-Gide membrane to fixate the cartilage fragments. Nineteen out of 27 patients in Massen’s study had lesions of the patello-femoral joint. Such coverage as the Chondro-Gide membrane might be important in a weakly constrained joint compartment with high shearing forces.29,69Average MOCART score at 6 months was 40.6 ± 21.1, knee function and pain significantly improved as measured by numeric analog scale.
Despite different follow-up moments for MRI and different PROMs being used, these few published studies show good radiological and clinical outcomes in the short term. However, studies with long-term follow-up, larger cohorts, and standardized methods of measuring outcomes are needed to establish the place for the autologous minced cartilage technique in the treatment algorithm for OCLs.
No comparative studies regarding the use of a covering membrane for different anatomical regions have been conducted. Using fibrin glue only in this series of patients, a complete fill of the defect was observed 3 months after surgery in 11 out of 14 patients who underwent MRI. The remaining 3 patients had a partial fill of the defect. The advantages of not using a membrane are that this allows to perform the procedure arthroscopically, and eliminates the need for possibly damaging sutures to connect the membrane with the native healthy cartilage. Moreover, several studies have shown fibrin sealant is a medium that promotes growth and migration of chondrocytes.70,71
In contrast to a retrospective study by Schreiner et al. 72 of 114 OCL repairs by means of microfracture, OATS, and ACI, defects smaller than 2.5 cm2 had significant higher MOCART scores than larger defects. Moreover, defects of the LFC had significantly higher MOCART scores compared to MFC defects. A possible explanation may be found in that the medial knee compartment bears more load than the lateral compartment73-75 and therefore leads to higher pressure on the newly forming regenerative tissue. This finding is in line with Emre et al. 76 finding lower Lysholm knee score in lesions located on the MFC in a retrospective series of 152 patients undergoing mosaicplasty.
Numerous studies use MR imaging to evaluate cartilage repair strategies. While McCarthy et al. 77 found a significant correlation between the MOCART score and clinical outcome in the long term, others only found correlations between clinical outcome and certain individual parameters of the MOCART as defect fill, subchondral bone, structure, and signal intensity of the repair tissue.78-81
This study suffered from a number of limitations. First, with the short clinical follow-up of 14.1 ± 2.7 months and radiological follow-up of 13.3 ± 2.7 months, the results of this study cannot be compared to previous studies on long-term outcomes of ACI,19,82 OAT,83,84 and microfracture. 85 Long-term follow-up is necessary to distinguish between continuous clinical improvement as seen in ACI and deterioration of clinical results as seen in microfracture after 18 or 24 months.14,15,86
Second, the heterogeneity of the patients included in this study should be considered. Of the 18 patients, 7 (39%) suffered co-abnormalities and needed one or more additional procedures beside the repair of the OCL, and 4 patients (22%) had already been operated on the index knee. These concomitant procedures may have affected the results of this study, but we considered treatment of these co-existing pathologies essential to protect the repair site and the developing cartilage.32,87,88 Third, 3-month follow-up MRI was performed in only 14 out of 18 patients. Four patients did not show up for the MRI exam for unknown reasons. While their MOCART scores were average to good ( Table 3 ), it is not possible to retrospectively evaluate whether they had a complete fill of the defect at 3 months. Without this necessary information, no confident assertion can be made whether fibrin glue, without the use of a covering membrane, is enough to secure the cartilage pieces in the defect.
Fourth, MRI exams were performed at both 1.5 T and 3.0 T. In a comparative study performed by Kijowski et al., 89 a 3.0 T MR protocol showed improved diagnostic performance for evaluation of the cartilage compared to a 1.5 T protocol. This lower resolution of some MRI exams in our study made it harder to accurately fill out the MOCART 2.0 score.
Fifth, as only young patients were included in this study, we can only speculate about the appropriateness of this technique in patients aged 30–40 or even 50 years. For example, we may question if the results of this study might be similar to those of microfracture (bone-based) or cell-based techniques 90 in these age groups. However, further research with longer follow-up is necessary to elucidate the appropriateness of minced cartilage technique for middle-aged patients.
Lastly, due to the heterogeneity of the observed lesions regarding lesion size and nature of the lesions (traumatic versus chronic, chondral versus osteochondral) no direct comparison to other existing cartilage repair techniques is possible. Further standardized datacapture a and registry formation are needed to establish the optimal indication and personalized treatment plan identification.
To conclude, promising results of 18 patients treated by the autologous minced cartilage procedure were presented, showing significant improvements in knee function, quality of life and pain at 12 months follow-up. Additionally, radiological evaluation showed good cartilage quality at 12 months follow-up. A significantly higher MOCART score was found in defects of the LFC compared to MFC and in defects smaller than 2.5 cm2 compared to larger defects.
Future studies are required to proof durability and the position of this technique in comparison to existing techniques in cartilage treatment algorithms which include patient age, size and features of the defect, and other variables.
Supplemental Material
sj-docx-1-car-10.1177_19476035221126343 – Supplemental material for Minced Autologous Chondral Fragments with Fibrin Glue as a Simple Promising One-Step Cartilage Repair Procedure: A Clinical and MRI Study at 12-Month Follow-Up
Supplemental material, sj-docx-1-car-10.1177_19476035221126343 for Minced Autologous Chondral Fragments with Fibrin Glue as a Simple Promising One-Step Cartilage Repair Procedure: A Clinical and MRI Study at 12-Month Follow-Up by M.H.H. Wodzig, M.J.M. Peters, K.S. Emanuel, P.P.W. Van Hugten, W. Wijnen, L.M. Jutten, T.A. Boymans, D.V. Loeffen and P.J. Emans in CARTILAGE
Supplemental Material
sj-docx-2-sav-10.1177_19476035221126343 – Supplemental material for Minced Autologous Chondral Fragments with Fibrin Glue as a Simple Promising One-Step Cartilage Repair Procedure: A Clinical and MRI Study at 12-Month Follow-Up
Supplemental material, sj-docx-2-sav-10.1177_19476035221126343 for Minced Autologous Chondral Fragments with Fibrin Glue as a Simple Promising One-Step Cartilage Repair Procedure: A Clinical and MRI Study at 12-Month Follow-Up by M.H.H. Wodzig, M.J.M. Peters, K.S. Emanuel, P.P.W. Van Hugten, W. Wijnen, L.M. Jutten, T.A. Boymans, D.V. Loeffen and P.J. Emans in CARTILAGE
Footnotes
Author’s Note
Investigation performed at Maastricht University Medical Centre, Maastricht, The Netherlands.
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the local ethical committee (METC 2021-2898).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
