Abstract
Objective
Osteoarthritis (OA) is a chronic joint disease characterized by degeneration of articular cartilage and secondary osteogenesis. Cell-based agents, such as mesenchymal stem cells, have turned into the most extensively explored new therapeutic agents for OA. However, evidence-based research is still lacking.
Methods
We searched public databases up to February 2020 and only included randomized controlled trials. The outcomes included the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), the Knee Injury and Osteoarthritis Outcome Score (KOOS), the visual analogue scale (VAS) score, and serious adverse events (SAEs). A network meta-analysis was also performed in this work.
Results
We included 13 studies in the meta-analysis. The effect size showed that cell-based therapy did not significantly reduce the WOMAC score at the 6-month follow-up (standard mean difference [SMD] −3.6; 95% confidence interval [CI] −0.90 to 0.18; P = 0.1928). However, cell-based therapy significantly improved the KOOS at the 12-month follow-up (SMD 0.68; 95% CI 0.07-1.30; P = 0.0288) and relieved pain (SMD −1.05; 95% CI −1.46 to −0.64; P < 0.0001). The findings also indicated that high-dosage adipose-derived mesenchymal stem cells (ADMSCs) may be more advantageous in terms of long-term effects.
Conclusions
Cell-based therapy had a better effect on KOOS improvement and pain relief without safety concerns. However, cell-based therapy did not show a benefit in terms of the WOMAC. Allogeneic cells might have advantages compared to controls in the WOMAC and KOOS scores. The long-term effect of high-dose ADMSC treatment for OA is worthy of further study.
Introduction
Osteoarthritis (OA) is a common irreversible degenerative joint disease characterized by degenerative changes in articular cartilage and secondary bone hyperplasia. 1 It can cause joint pain, swelling, stiffness, and mobility disorders that greatly impact quality of life. 2 The prevalence of symptomatic OA is approximately 242 million worldwide, 3 and its prevalence is increasing because of recent increases in life expectancy. 4 The World Health Organization (WHO) indicated that OA affects 9.6% of men and 18% of women older than 60 years. 5 There is still a lack of effective ways to prevent OA progression. Microfractures can improve function and pain only in the short term but can even be harmful for elderly patients in the long term. 6 Articular cartilage is considered non-self-renewing avascular tissue, 7 and endogenous mesenchymal stem cells (MSCs) are not sufficient in quantity and quality for cartilage repair. 8
Therefore, treatment with exogenous MSCs is theoretically feasible to repair cartilage injury, and researchers are exploring this technique for OA treatment. 9 In preclinical studies, intra-articular cell injection therapy and even treatment with exosomes derived from cells have been deemed potential and safe treatments for articular cartilage repair and regeneration in OA. 10 However, the Food and Drug Administration (FDA) still has concerns about the efficacy and safety of cell therapy for OA. 11 The results of current randomized controlled trials (RCTs) on cell-based therapy are still inconclusive, especially regarding the functional results. 11
Recently, a meta-analysis of RCTs showed limited evidence that the intra-articular injection of MSCs can relieve knee cartilage pain and improve function, and cell quantity and concomitant treatment may have an impact on the results. 12 Another meta-analysis showed that expanded MSCs can relieve pain in the short term (6-12 months), but there is still insufficient evidence of functional improvement and cartilage repair. 13 Other comprehensive analyses based on RCTs and non-RCTs have shown that MSCs can improve pain and function over a relatively longer follow-up period (<28 months) and are considered ideal treatment methods.14,15 Before-after comparisons have also shown that injection with bone marrow MSCs (BMMSCs) can improve function and relieve pain but not improve range of motion.16,17 Almost all comprehensive analyses agree that there is a lack of standards for cell applications, especially regarding cell type and source, cell dosage, cell quality identification, cell vehicle, and effect evaluation criteria. The clarification of cell application standards is the basis for further RCT design.18,19 Therefore, this study was designed to analyze the effect and safety of cell therapy for OA by meta-analysis and to further analyze various cell therapy strategies in detail by network meta-analysis to provide guidance for further RCT design and even a reference for cell therapy standards in OA.
Methods
This systematic review and meta-analysis was conducted according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. 20
Search Strategy
A literature search was conducted in PubMed, Embase, and Cochrane Library by 2 authors independently. The search period was from the date of database inception to March 4, 2020. The following search terms were used: (((((((stem cell) OR (cell transplantation)) OR (mesenchymal)) OR (stromal)) OR (cytotherapy)) OR (cellular therapy)) OR (Cell therapy)) AND (((Random*) OR (Randomized)) OR (Randomized)) AND Osteoarthritis. Manual searches of the reference lists of the relevant reviews were also performed to identify additional eligible studies.
Eligibility Criteria
Inclusion and exclusion criteria were applied to the search results. A study was included if it met the following criteria: the study had an RCT design; the study included OA patients; one of the study arms adopted a cell-based treatment; the control group adopted a non-cell-based therapy; and the study reported at least one of the following outcomes: the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), the Knee Injury and Osteoarthritis Outcome Score (KOOS), or the visual analogue scale (VAS) score. The exclusion criteria were as follows: non-RCT design; examination of other non-OA patients; use of a non-cell-based intervention, such as exosome or platelet-rich plasma (PRP); use of a cell-based intervention combined with surgical treatment; lack of a non-cell-based control group; and lack of reporting of the above outcomes. In addition, both the stromal vascular fraction (SVF) and bone marrow aspirate concentrate (BMAC) contain multiple cell components, such as adipose-derived MSCs (ADMSCs), endothelial precursor cells (EPCs), endothelial cells (ECs), macrophages, and smooth muscle cells in the SVF. 21 The BMAC also contains platelets, white blood cells, red blood cells, hematopoietic precursors, and nonhematopoietic precursors. 22 Because of the presence of various cell types known to cause immunological rejection, the SVF and BMAC are suitable only for autologous treatments. Therefore, due to the difference in cell therapy with specific dosages and types, the SVF and BMAC were not analyzed in this meta-analysis. In vitro studies, animal studies, conference abstracts, and comments were also excluded. The study selection was independently undertaken by 2 authors.
Data Collection and Quality Assessment
The information from each eligible study was extracted as follows: the first author’s name, publication year, registration number, location, sample size, age, Kellgren-Lawrence grade, intervention, cell source, cell dosage, number of injections, and follow-up. The outcomes included the WOMAC, KOOS, VAS score, and serious adverse events (SAEs). If no specific raw data were provided, the data were obtained from plots given in the article. We assessed the methodological quality of the included trials using a risk of bias approach according to the methods described by the Cochrane Collaboration, which include 7 specified domains. 23
Statistical Analysis
Continuous data are reported as standard mean differences (SMDs) with 95% confidence intervals (CIs), and dichotomous data are reported as odds ratios (ORs) with 95% CIs. I 2 was used to estimate the heterogeneity among studies. For dichotomous data, the Mantel-Haenszel method was used in a fixed-effect model, and the inverse-variance method was used for continuous data. 24 The Paule-Mandel estimator was used to estimate the between-study variance. 25 Subgroup analysis was also perform according to allogeneic and autogenic cell application. Egger’s test and a funnel plot were used to check for potential publication bias.
A frequentist random-effect model was used for the network meta-analysis, and network plots were generated for each outcome according to each direct comparison. The rank of each outcome was calculated by the surface under the cumulative ranking curve (SUCRA). 26 Comparison-adjusted funnel plots were used to determine whether small-study effects were present. All P values are reported as 2-sided, and P values less than 0.05 were regarded as statistically significant for all trials. R (version 3.6.2) with packages “meta” and “netmeta,” Stata (version 14.0), and Review Manager (version 5.3) were used for the meta-analysis.
Results
A total of 1229 articles were retrieved after removing duplicates. A total of 1162 articles were excluded after screening titles and abstracts. The full texts of the remaining 67 articles were assessed. The following articles were excluded for the following reasons: non-OA patients (12); non-RCT design (9); BMAC or SVF intervention (9); non-cell-based intervention (7); cell-based intervention combined with surgical treatment (5); no desired outcomes (4); lack of a non-cell-based control group (2); target agent intervention (2); surgery applied in the control group (1); comment (1); review (1); protocol (1). Thirteen studies were included in the meta-analysis.27-39 The selection process is shown in a flow diagram ( Fig. 1 ).
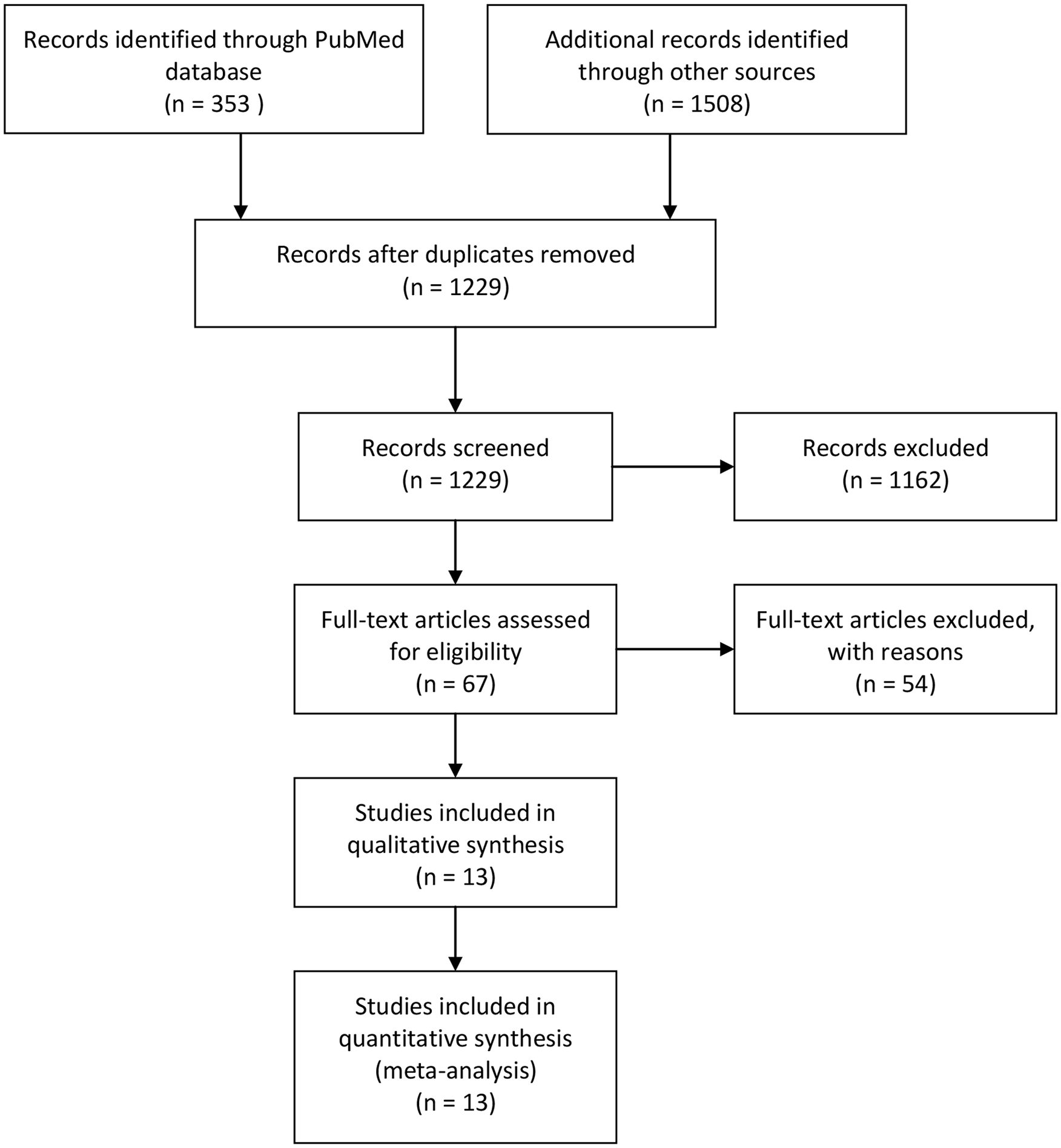
Flow diagram of the article selection process.
The studies were mainly published between 2015 and 2019. Six studies included more than 2 cell-based therapy arms.27,30-33,37 One article contained the results of 2 studies. 37 Kellgren-Lawrence grades I to IV OA patients were included in this analysis. Cell types included ADMSCs, BMMSCs, umbilical cord MSCs (UCMSCs), and chondrocytes with 25% genetically modified cells with high TGF-β1 (transforming growth factor–β1) expression ( Table 1 ). The minimum cell dosage was 3.9 million, and the maximum dosage was 150 million. The cells from each included study were culture-expanded. The therapy frequency was usually a single injection, and 3 studies adopted a double injection.27,28,30 One arm of an included study used BMMSCs combined with a PRP injection. 31 The control groups included treatment with hyaluronic acid (HA), saline, cell culture media, glucocorticoids, and an untreated blank control. In terms of design quality, the included studies were all RCTs. All except 2 included allocation concealment and blinding in the design,32,36 increasing the reliability of the results. Overall, the quality of the design of the included studies was ideal ( Fig. 2 ).
Characteristics of Included Studies.
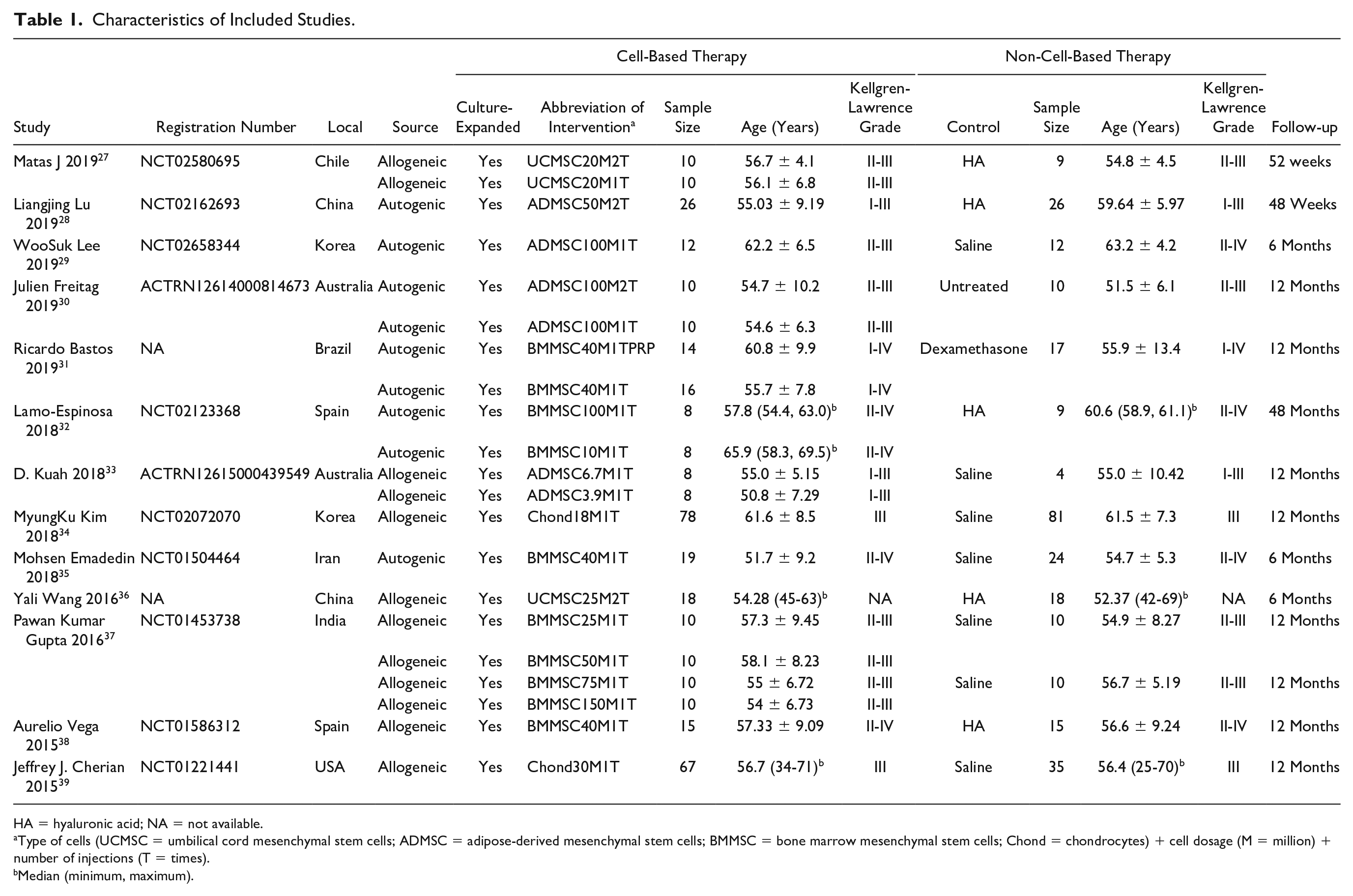
HA = hyaluronic acid; NA = not available.
Type of cells (UCMSC = umbilical cord mesenchymal stem cells; ADMSC = adipose-derived mesenchymal stem cells; BMMSC = bone marrow mesenchymal stem cells; Chond = chondrocytes) + cell dosage (M = million) + number of injections (T = times).
Median (minimum, maximum).
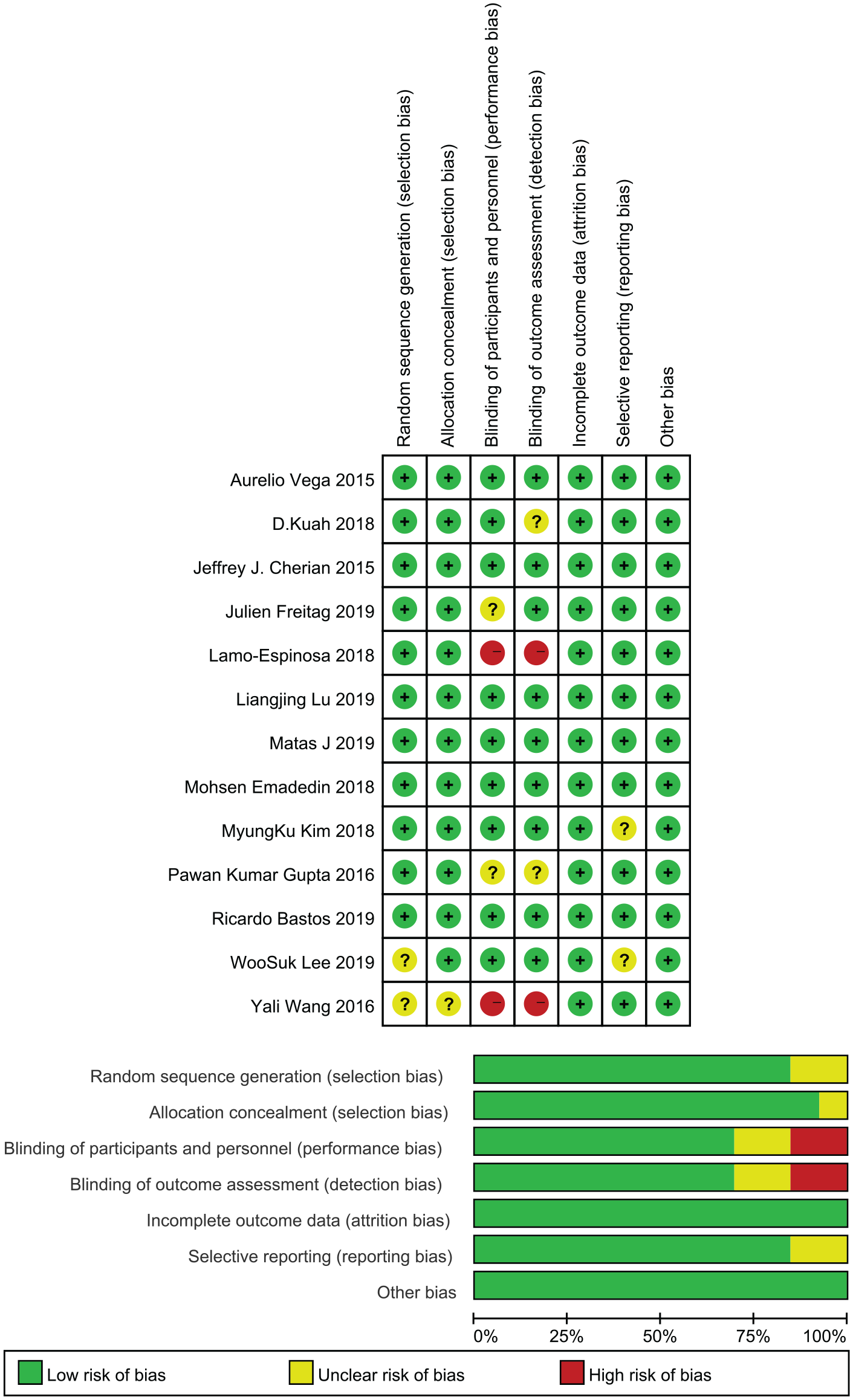
Summary of the risk of bias assessment for the included studies.
For convenience of presentation, we have abbreviated the cell-based therapy strategies according to the cell type, dosage, and number of injections. Regarding the WOMAC, we compared the cell-based therapy and control at the 6-month follow-up. Cell-based therapy did not significantly reduce the WOMAC (SMD −3.6; 95% CI −0.90 to 0.18; P = 0.1928) ( Fig. 3A ). In the subgroup analysis, allogeneic cell therapy (SMD −0.48; 95%CI −0.88 to −0.08; P = 0.018) but not autogenic cell therapy (SMD −0.15; 95%CI −1.52 to 1.23; P = 0.837) significantly reduced the WOMAC compared with the control. Further network meta-analysis was used to analyze the comparative advantages of the cell therapy strategies ( Fig. 4A ). The results showed that UCMSC25M2T (SUCRA: 82.6), ADMSC100M1T (SUCRA: 79.7), and BMMSC40M1T (SUCRA: 78.3) had relative advantages for WOMAC reduction ( Table 2 ). However, no further conclusions could be drawn from these results. Additionally, interventions with a larger number of cells were not better than those with a smaller number of cells, such as BMMSC100M1T (SUCRA: 13) and BMMSC40M1T (SUCRA: 78.3). However, the results indicated that the cell dosage may not be the main factor affecting the treatment outcome. Furthermore, we included the control type and follow-up time points to evaluate the effect size. Compared with the untreated blank control, ADMSCs could have a better effect with a large number of cells ( Fig. 5A ). The meta-regression results also showed a correlation between the ADMSC dosage and WOMAC reduction (β= -0.0057, p = 0.0075). BMMSCs did not significantly reduce the WOMAC in most cases compared to saline, culture medium, or HA. Meta-regression also showed no correlation between the cell dosage and WOMAC reduction (β = 0.0003, P = 0.9362). For UCMSCs, injection with 40 to 50 million cells may have a significant effect over the long term. However, meta-regression did not show statistically significant results (dosage: β = −0.0448, P = 0.1569; follow-up period: β = −0.1425, P = 0.1485).
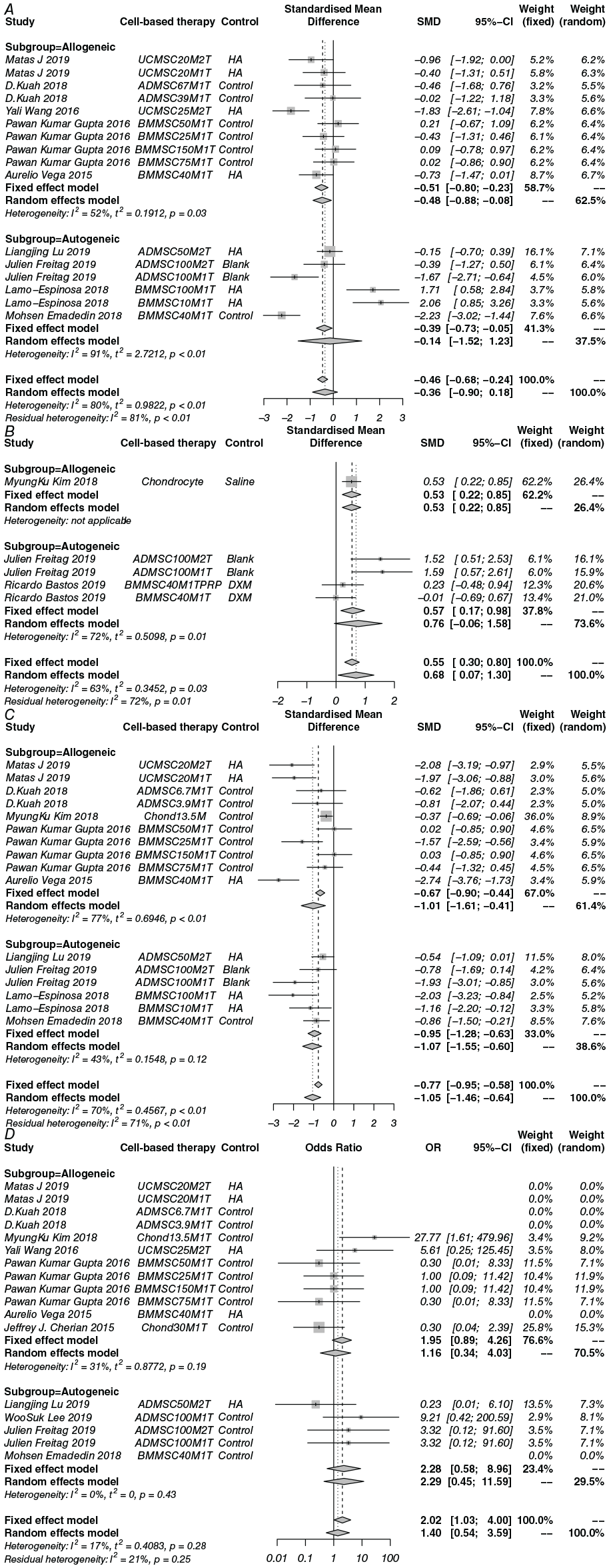
Forest plots of the effect size with the 95% CI for each outcome. (
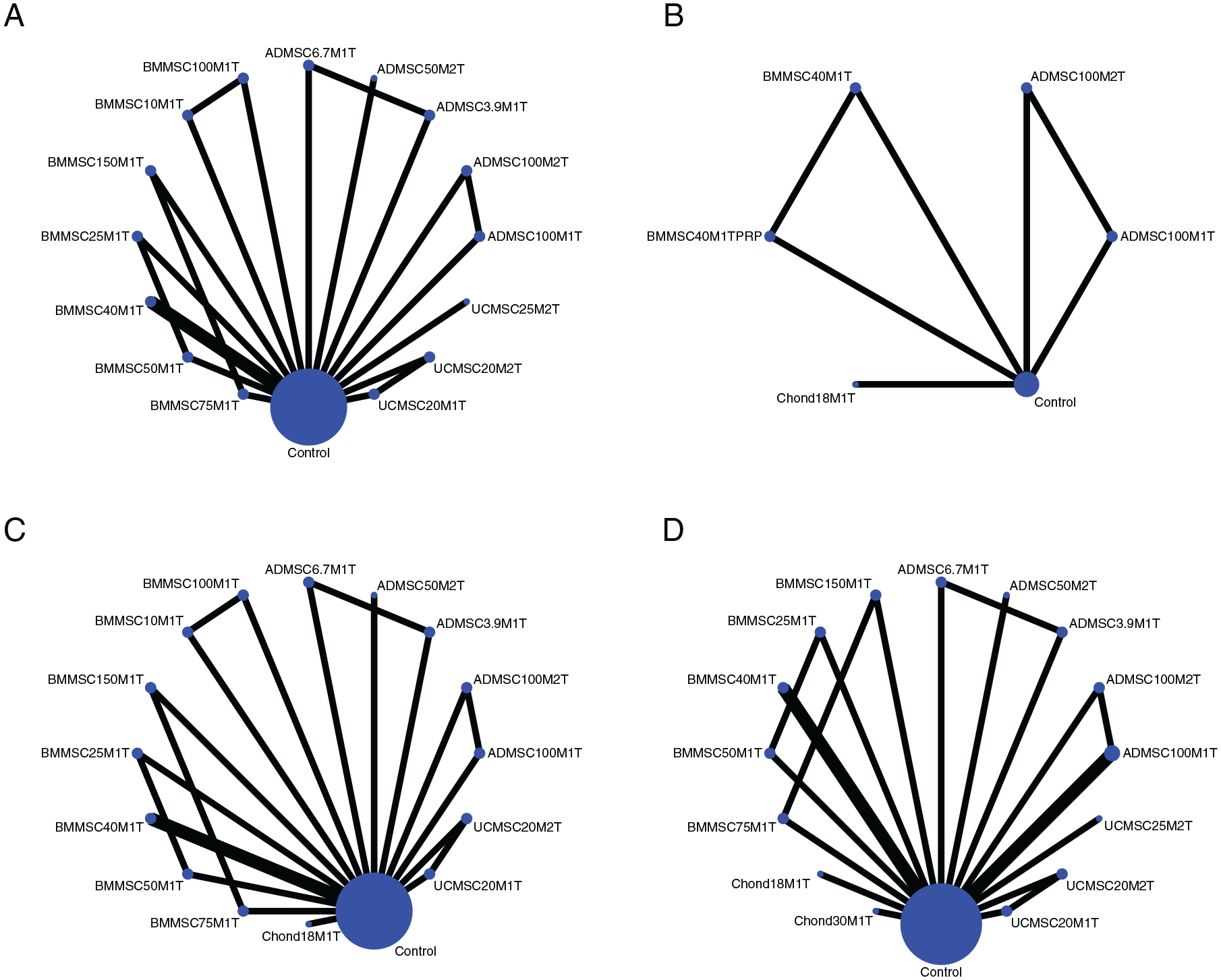
Network of comparisons for interventions included in the analyses. (
SUCRA Results of Interventions for Each Outcome.
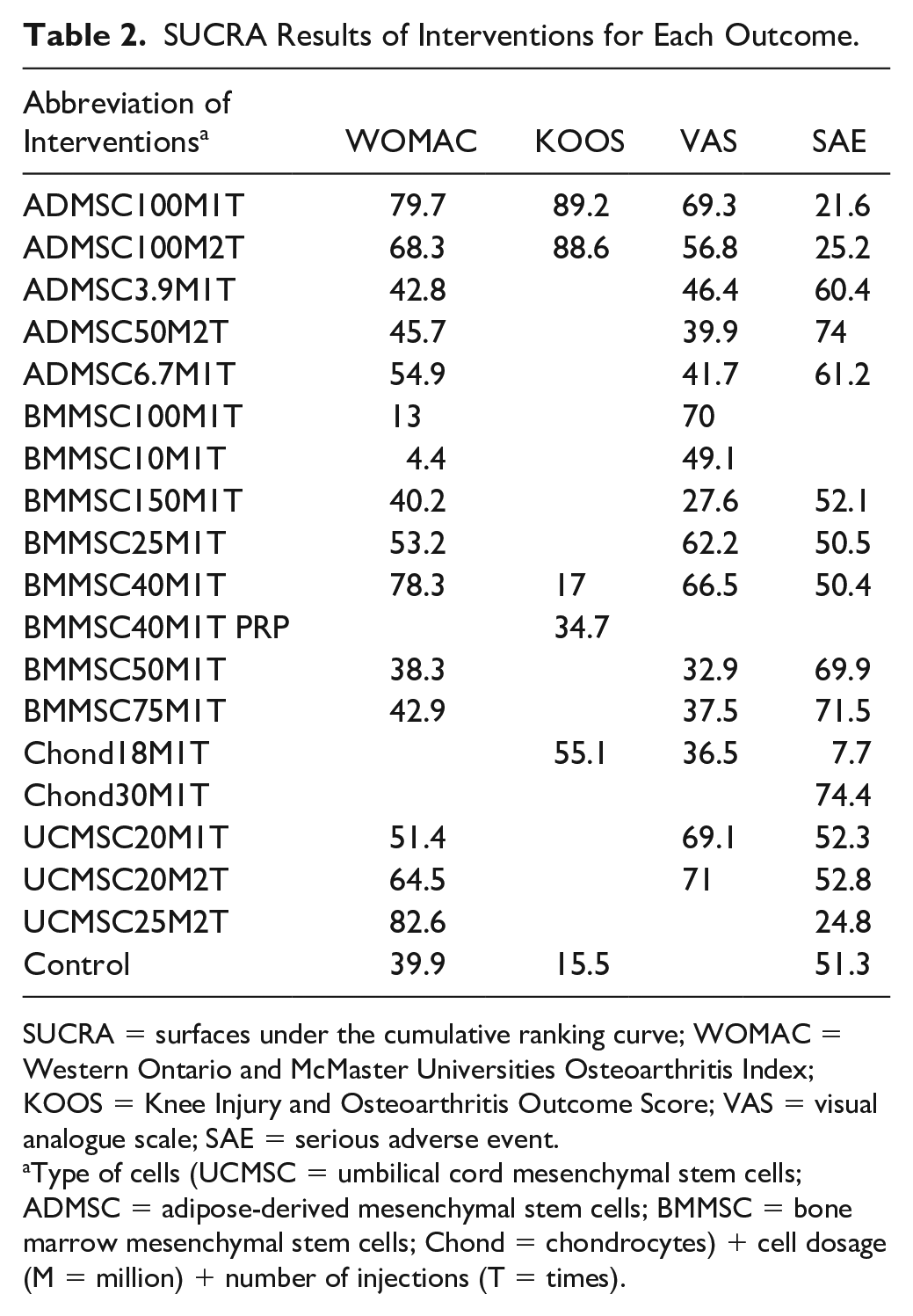
SUCRA = surfaces under the cumulative ranking curve; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index; KOOS = Knee Injury and Osteoarthritis Outcome Score; VAS = visual analogue scale; SAE = serious adverse event.
Type of cells (UCMSC = umbilical cord mesenchymal stem cells; ADMSC = adipose-derived mesenchymal stem cells; BMMSC = bone marrow mesenchymal stem cells; Chond = chondrocytes) + cell dosage (M = million) + number of injections (T = times).
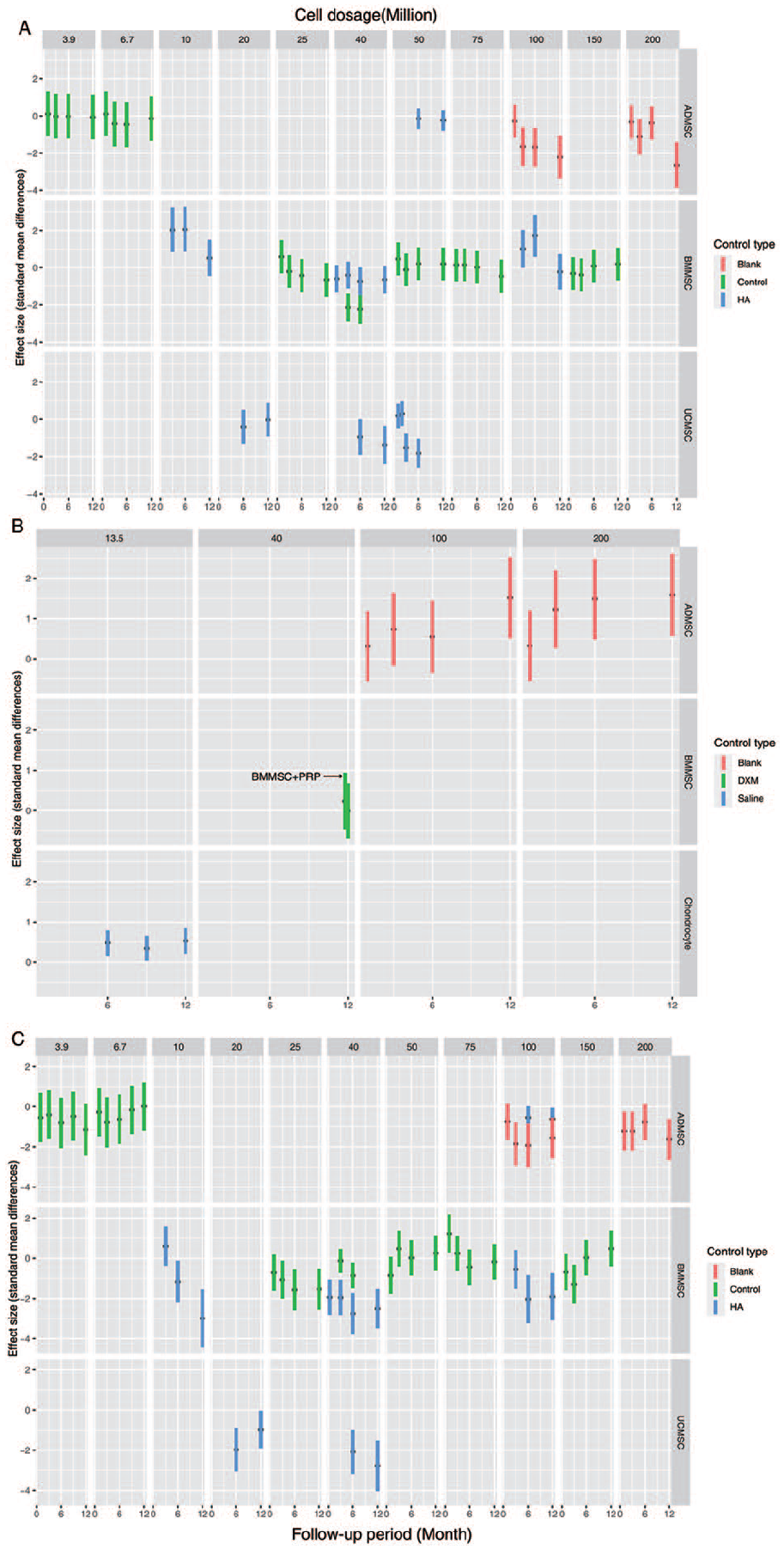
Effect sizes with 95% CIs of cell-based therapies compared with controls for each cell dosage and follow-up time points. (
Regarding the KOOS, at the 12-month follow-up time point, when results were reported more frequently, cell-based therapy significantly improved the KOOS (SMD 0.68; 95% CI 0.07-1.30; P = 0.0288). In the subgroup analysis, allogeneic cell therapy (SMD 0.53; 95%CI 0.22-0.85; P < 0.001) but not autogenic cell therapy (SMD 0.76; 95%CI −0.06 to 1.58; P = 0.069) significantly improved the KOOS. Network meta-analysis was adopted to show comparatively superior cell application strategies and showed that ADMSC100M1T (SUCRA: 89.2) and ADMSC100M2T (SUCRA: 88.6) yielded relatively good results. After adding the cell dosage and follow-up factors, the results showed that a large dosage of ADMSCs had a more significant effect on the KOOS over the long term than the untreated blank control. Because the cell dosage has only two variables of 100 million and 200 million and the duration of the follow-up was a confounding factor, the meta-regression results showed no statistical correlation between the cell dosage and KOOS increases (β = 0.0037, P = 0.2775), but a correlation was identified between the follow-up period and KOOS increases (β = 0.0998, P = 0.0174). In addition, chondrocytes also showed a significant effect on the KOOS during the 6- to 12-month follow-up period.
Considering the VAS scores, cell therapy could significantly reduce the VAS score at the 6-month time point by traditional meta-analysis (SMD −1.05; 95% CI −1.46 to −0.64; P < 0.0001). Network meta-analysis showed that UCMSC20M2T (SUCRA: 71), ADMSC100M1T (SUCRA: 69.3), and UCMSC20M1T (SUCRA: 69.1) were superior for pain relief. Specifically, ADMSCs were more effective at high dosages, and meta-regression also showed a correlation between the dosage and VAS score (β = −0.0035, P = 0.0282). Compared with HA, BMMSCs had an obvious effect over the long-term regardless of the cell dosage. However, compared with saline or cell culture medium, BMMSCs were advantageous at low dosages. Nevertheless, overall, the effect was not related to the cell dosage (β = 0.0061, P = 0.1706). In addition, the results suggest that there is publication bias (Egger’s test: P = 0.01267; Begg’s: P = 0.01922).
Because the sample size in each included study was small, there were multiple double-zero SAE outcome reports. In general, cell therapy did not result in more SAEs than non-cell-based interventions (fixed-effect model: OR 2.02, 95% CI 1.03-4.00, P = 0.422; random-effect model: OR 1.40, 95% CI 0.54-3.59, P = 0.4895). Although not significant, we found that Chond30M1T (SUCRA: 74.4), ADMSC50M2T (SUCRA: 74.0), and BMMSC75M1T (SUCRA: 71.5) resulted in relatively fewer SAEs. Because SAEs are represented by the cumulative incidence at the last follow-up, the effect size could not be comprehensively determined according to the follow-up period and cell dosage.
Discussion
This study comprehensively analyzed the WOMAC, KOOS, VAS score, and SAEs of various cell-based therapy strategies for OA. To clarify the different cell strategies in detail, we first analyzed the cell-based therapy strategies by network meta-analysis. Then, the effect size was comprehensively determined for different cell dosages and follow-up time points. The results indicate that cell-based therapy cannot significantly reduce the WOMAC, which is one of the main indices of OA. While there were relatively few studies reporting KOOS results, cell-based therapy showed an effect on increasing the KOOS at the 12-month follow-up time point. The results of the subgroup analysis showed that allogeneic cells had advantages over autogenic cells in the WOMAC and KOOS scores. The analysis also indicates that high-dosage ADMSCs may be more advantageous for long-term effects. Cell-based therapy could significantly reduce the VAS score, but with publication bias. Regarding safety, cell-based therapy did not cause more SAEs than non-cell-based therapy.
High dosages of ADMSCs seem to have a better therapeutic effect. In one included study, after ADMSC injection, the volume of femur articular cartilage was significantly increased compared with after HA injection. 28 This is an exciting result that may determine the effect of ADMSCs in reversing the pathological progression of OA. Unexpectedly, although many studies used BMMSC injection as a treatment, the effect was not as good as expected. Although they are sourced from different tissues, it is generally believed that ADMSCs and BMMSCs will show no differences in clinical applications. They have similar characteristics, such as a pericyte origin and similar cell markers, differentiation potential, gene expression, and paracrine characteristics. 40 However, when emphasizing the difference between ADMSCs and BMMSCs, a study showed that ADMSCs have a larger cell count per unit volume of tissue, proliferate faster, and survive better than BMMSCs. 5 Considering that OA is more common in elderly patients, when the amount of BMMSCs extracted from the unit volume of bone marrow was much smaller than the amount of ADMSCs extracted from adipose tissue, the advantage of ADMSCs in terms of available quantity was highlighted. 41 A recent study on the single-cell profiles of ADMSCs and BMMSCs has shown that ADMSC populations have lower transcriptome heterogeneity and that ADMSCs are less dependent on mitochondrial respiration for energy production. 42
In this research, different results were obtained for the WOMAC and KOOS. The KOOS results favored cell-based therapy for OA. The KOOS was developed based on the WOMAC. The WOMAC evaluates the function of the knee in terms of pain, stiffness, and physical function. The KOOS has patient-relevant dimensions: pain, other disease-specific symptoms, activities of daily living function, sport and recreation function, and knee-related quality of life. More questions are included in the pain and other disease-specific symptoms subscales of the KOOS compared to the WOMAC pain and stiffness subscales. The activities of daily living function subscale of the KOOS was equivalent to the physical function subscale of the WOMAC. The sport and recreation function and knee-related quality of life subscales of the KOOS are not evaluated in the WOMAC. With the improvement of the treatment requirements for young and early-stage OA patients, the requirements for the assessment of daily activities have also increased. On the basis of the WOMAC, the KOOS focuses more on the judgment of status changes for younger and more active patients. 43 Although the number of included studies reporting results for the KOOS was relatively small, the positive results also increase the confidence of further research on stem cell treatments for OA, especially in young and early-stage OA patients.
Among the cells used in strategies included in this study, allogeneic chondrocytes do not have multiple differentiation potential, and 25% of these cells were genetically modified and irradiated to show increased TGF-β1 expression and not proliferate in the cavity.34,39 The expression of TGF-β1 could promote extracellular matrix deposition and improve local cartilage regeneration. 44 Although this study did not find an advantage of chondrocytes compared to MSCs for OA treatment, the findings expand the cell-based therapies for OA. The safety of genetically modified cells that particularly concerning in the treatment of other diseases, such as stroke, 45 was demonstrated in the closed joint cavity microenvironment.
Due to the many regions of interest measured by radiography and the various evaluation measures, including the Whole-Organ Magnetic Resonance Imaging Score (WORMS), Magnetic Resonance Imaging Osteoarthritis Knee Score (MOAKS), cartilage volume, defect area, and cartilage thickness, a quantitative meta-analysis was difficult to perform; therefore, we listed the qualitative radiographic results in Supplementary Table 1, which shows that cell-based therapy has the potential to increase the cartilage volume and alleviate cartilage defects.
In conclusion, cell-based therapy has a better effect on improving the KOOS and relieving pain without safety concerns. However, cell-based therapy showed no benefit in improving the WOMAC. Allogeneic cells might have advantages compared to controls in the WOMAC and KOOS scores. Further research could examine the long-term effects (more than 12 months) of high-dosage ADMSC (more than 100 million) treatments on OA and include evaluations of changes in cartilage volume.
Limitations
There are several limitations to this research. First, the study-level and not individual-level analysis prevented demonstration of the effects of cell-based therapy in OA patients of different ages or Kellgren-Lawrence grades. Second, the network meta-analysis contained a large number of arms according to the cell-based therapy strategies, resulting in a small sample size in each arm. Third, because of the various cell-based arms and small samples, this study is not suitable for making conclusions about which strategy is more advanced in the clinic but is more suitable for making recommendations for the design of further RCTs. Fourth, there was publication bias in the VAS score results; since the VAS score was not the primary result, this study did not use the trim-and-fill method to correct this bias. Fifth, this study did not consider the policies, regulations and related specifications for cell isolation and expansion in vitro around the world, so this study may not apply in some countries or regions based on the regulations for cell-based applications.
Supplemental Material
Supplement_table_1 – Supplemental material for Efficacy and Safety of Intra-Articular Cell-Based Therapy for Osteoarthritis: Systematic Review and Network Meta-Analysis
Supplemental material, Supplement_table_1 for Efficacy and Safety of Intra-Articular Cell-Based Therapy for Osteoarthritis: Systematic Review and Network Meta-Analysis by Wei Ding, Yong-qing Xu, Ying Zhang, An-xu Li, Xiong Qiu, Hong-jie Wen and Hong-bo Tan in CARTILAGE
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is funded by The National Key Research and Development Program of China (2017YFC1103904; 2018HB001).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
