Abstract
Study Design
Meta-analysis.
Objectives
To compare the efficacy and safety of bone marrow(BM)–derived mesenchymal stem cell(MSCs) and adipose-derived(AD) MSCs in the management of osteoarthritis of knee from randomized controlled trials(RCTs) available in the literature.
Materials and Methods
We conducted electronic database searche from PubMed, Embase, and Cochrane Library till May 2020 for RCTs analyzing the efficacy and safety of MSCs in management of osteoarthritis of knee. Visual Analog Score(VAS) for Pain, Western Ontario McMaster Universities Osteoarthritis Index(WOMAC), Lysholm Knee Scale(Lysholm), Whole-Organ Magnetic Resonance Imaging Score(WORMS), Knee Osteoarthritis Outcome Score(KOOS), and adverse events were the outcomes analyzed. Analysis was performed in R platform using OpenMeta[Analyst] software.
Results
Nineteen studies involving 811 patients were included for analysis. None of the studies compared the source of MSCs for osteoarthritis of knee and results were obtained by pooled data analysis of both sources. At 6 months, AD-MSCs showed significantly better VAS(P<0.001,P=0.069) and WOMAC(P=0.134,P=0.441) improvement than BM-MSCs, respectively, compared to controls. At 1 year, AD-MSCs outperformed BM-MSCs compared to their control in measures like WOMAC(P=0.007,P=0.150), KOOS(P<0.001;P=0.658), and WORMS(P<0.001,P=0.041), respectively. Similarly at 24 months, AD-MSCs showed significantly better Lysholm score(P=0.037) than BM-MSCs(P=0.807) although VAS improvement was better with BM-MSCs at 24 months(P<0.001). There were no significant adverse events with either of the MSCs compared to their controls.
Conclusion
Our analysis establishes the efficacy, safety, and superiority of AD-MSC transplantation, compared to BM-MSC, in the management of osteoarthritis of knee from available literature. Further RCTs are needed to evaluate them together with standardized doses.
Keywords
Introduction
Osteoarthritis (OA) of the knee is the most common degenerative joint disorder among adults that poses major morbidity affecting the functional quality of everyday life. OA knee results from an imbalance between the rate of degeneration and repair due to limited intrinsic potential for cartilage to heal. 1 It is characterized by the gradual wear of hyaline cartilage resulting in the formation of bony spurs at the margins of the joints and development of subchondral sclerosis and cysts. 2 Hence, cartilage has been targeted to regenerate and rejuvenate with the help of orthobiologics. These bioactive molecules bridge a gap between conservative and surgical management in the treatment of osteoarthritis knees.
Cellular therapy is defined as the transplantation of human cells to replace or repair damaged tissue and/or cells, including hematopoietic stem cells (HSCs), mesenchymal stem cells (MSCs), lymphocytes, dendritic cells, NK cells, and pancreatic islet cells. Among the available orthobiologics and cellular therapy, MSCs have a greater advantage in healing and regenerating the cartilage defects. 3 Intraarticular administration of MSCs may enhances cartilage regeneration and reduction of degenerative mechanisms of OA knee. MSCs were found in bone marrow, adipose tissue, amniotic fluid, placenta, dental pulp, endometrium, menstrual blood, and umbilical cord. Among these varied sources, the MSCs from bone marrow and adipose tissues are readily accessible. MSCs possess anti-inflammatory, antimicrobial, analgesic, regenerating, immunomodulatory, and immune-evasive properties. 4 The choice of MSCs in the treatment of OA knees are debatable among all orthopedic and translational medicine researchers. 5
Bone marrow–derived mesenchymal stem cells (BM-MSCs) can be derived from the iliac crest, and its isolation follows simple kinetics of centrifugation to concentrate MSCs.6,7 Bone marrow aspirate concentrate contains enormous growth factors (PDGF, EGF, TGF-β, FGF, and NGF) and bone morphogenetic proteins (BMP-2&7).8-10 Although adipose-derived mesenchymal stem cells (AD-MSCs) are found abundant in the human body, it needs complex processing to obtain adipose tissue–derived stromal vascular fraction (SVF). SVF is an aqueous fraction with a combination of adipose-derived stem cells, endothelial precursor cells, endothelial cells, macrophages, smooth muscle cells, lymphocytes, pericytes, and pre-adipocytes.11,12 SVF warrants the usage of allogenic preparation as it comprises various cells in the solution. The stem cell activity of adipose-derived SVF is 3 times higher than bone marrow–derived MSCs.13,14
Hence, with this meta-analysis, we aim to compare the efficacy and safety of BM-MSCs and AD MSCs in the management of osteoarthritis of knee from the available literature.
Materials and Methods
This meta-analysis was conducted following the guidelines of Back Review Group of Cochrane Collaboration 15 and reported based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 16
Search Strategy
Two reviewers performed an independent electronic literature search for studies evaluating the safety and efficacy of stem cell therapy for spinal cord injury. We searched the following databases: PubMed, Embase, and the Cochrane Library up to May 2020. No language or date restrictions were applied. Keywords used for the search were as follows: “Knee Osteoarthritis,” “Knee Degeneration,” “Stem Cell Therapy” and “Mesenchymal Stem Cells,” “Bone marrow,” “Adipose.” The reference list of the selected articles was also searched to identify studies not identified in the primary search. As per the inclusion and exclusion criteria, eligible studies were included for meta-analysis. The discrepancy between the authors was resolved through discussion until a consensus was obtained. A detailed study selection flow diagram is given in Figure 1 .
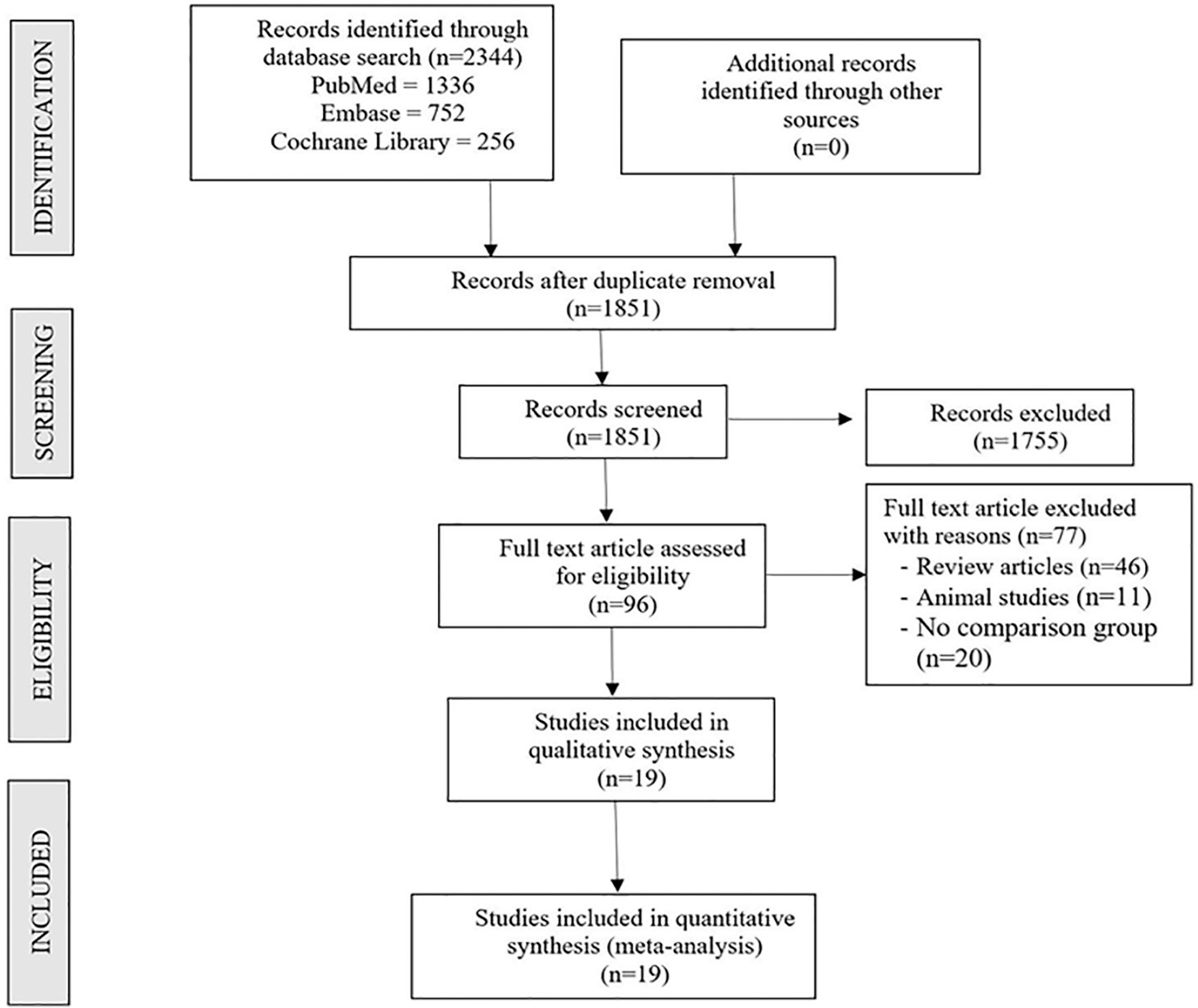
PRISMA flow diagram of the included studies.
Inclusion Criteria
Studies were included for quantitative review if they met the following PICOS criteria:
Population: Patients with knee osteoarthritis
Intervention: MSC therapy
Comparator: Usual care
Outcomes: Visual Analog Score (VAS) for Pain, Western Ontario McMaster Universities Osteoarthritis Index (WOMAC), Lysholm Knee Scale (Lysholm), Whole-Organ Magnetic Resonance Imaging Score (WORMS), Knee Osteoarthritis Outcome Score (KOOS), and adverse events
Study Design: Randomized controlled trials
Exclusion Criteria
Trials were excluded if they had the following characteristics:
Observational studies and interventional studies without a comparator group
Animal studies involving stem cell therapy for knee osteoarthritis models
Review articles and in-vitro studies involving stem cell therapy
Data Extraction
Two reviewers retrieved independently relevant data from articles included for analysis. Following data were extracted:
Study characteristics: year of publication, authors, country, level of evidence, number of patients enrolled.
Baseline characteristics: mean age, gender proportions, Kellgren-Lawrence grade of osteoarthritis, source of MSC utilized, intervention for both the groups, follow-up duration, and assessment parameters utilized. For analytical purpose, we have included studies using BMC and isolated expanded BM-MSC therapy into one group, with SVF and isolated expanded AD-MSC therapy in another group.
Efficacy outcomes: VAS for pain, functional outcomes like WOMAC, Lysholm Score, KOOS, and radiological outcomes such as WORMS.
Safety outcomes: adverse events in the included studies.
For missing data, we tried to contact the original author first. If we failed to contact, we calculated the missed values from other available data using formulas in the Cochrane Handbook for Systematic Reviews of Interventions. Any disagreement in data collection was resolved until a consensus was attained by discussion.
Risk of Bias and Quality Assessment
The methodological quality of the included studies was assessed independently by 2 reviewers using the Cochrane Collaboration’s ROB2 tool for randomized studies, which has 5 domains of bias assessment including randomization process, deviation from intended intervention, missing outcome data, measurement of the outcome, and selection of the reported results. 17
Statistical Analysis
Meta-analysis was conducted in the R platform with OpenMeta[Analyst]. 18 For dichotomous variable outcomes, risk ratio (RR) with 95% confidence interval (CI) was used, and for continuous variable outcomes, weighted mean difference (WMD) with 95% CI was used. Heterogeneity was assessed using the I2 test. 19 If I2 < 50% and P > 0.1, we used a fixed-effects model to evaluate, otherwise, a random-effects was used. A P value <0.05 was considered significant. Sensitivity analyses were performed to explore the source of heterogeneity when it existed. Publication bias was analyzed with a funnel plot for the outcomes in the included studies.
Results
Search Results
Electronic database search resulted in 2,344 articles, which after initial screening for duplicate removal gave a total of 1,351 articles. Title and abstract screening were done in those 1,851 articles and 1,755 of them were excluded. Ninety-six articles qualified for full-text review of which 77 were excluded. Finally, 19 studies20-38 with 811 patients were included for quantitative analysis. PRISMA flow diagram of study selection is given in Figure 1 . Nine of 19 studies utilized MSCs from adipose tissue, of which 1 used allogenic source and 8 studies utilized autogenous source of AD-MSCs. Ten of 19 studies utilized MSCs from bone marrow, of which 3 used allogenic sources and 7 studies utilized autogenous source of BM-MSCs. There was no standardization noted among the included studies concerning the dose of MSCs transplanted. There was also no uniformity among the included studies for the outcome measures utilized. The general characteristics of the studies included were given in Table 1 . There was no uniformity in the dose of MSCs transplanted in the included studies. Interventions in the treatment and control groups of the individual studies are given in Table 2 .
Characteristics of Included Studies.
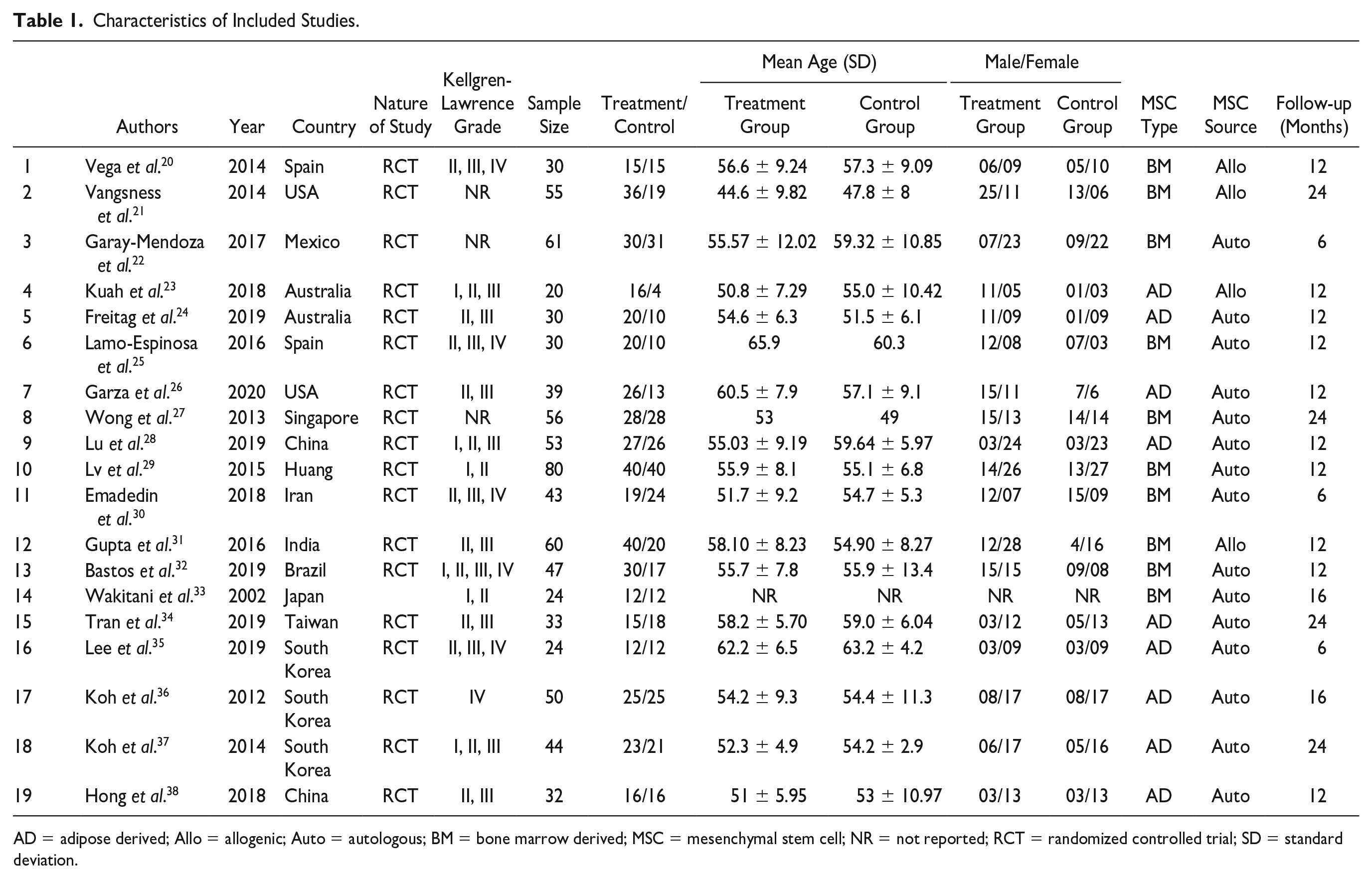
AD = adipose derived; Allo = allogenic; Auto = autologous; BM = bone marrow derived; MSC = mesenchymal stem cell; NR = not reported; RCT = randomized controlled trial; SD = standard deviation.
Stem Cell Transplantation Protocol of the Included Studies.
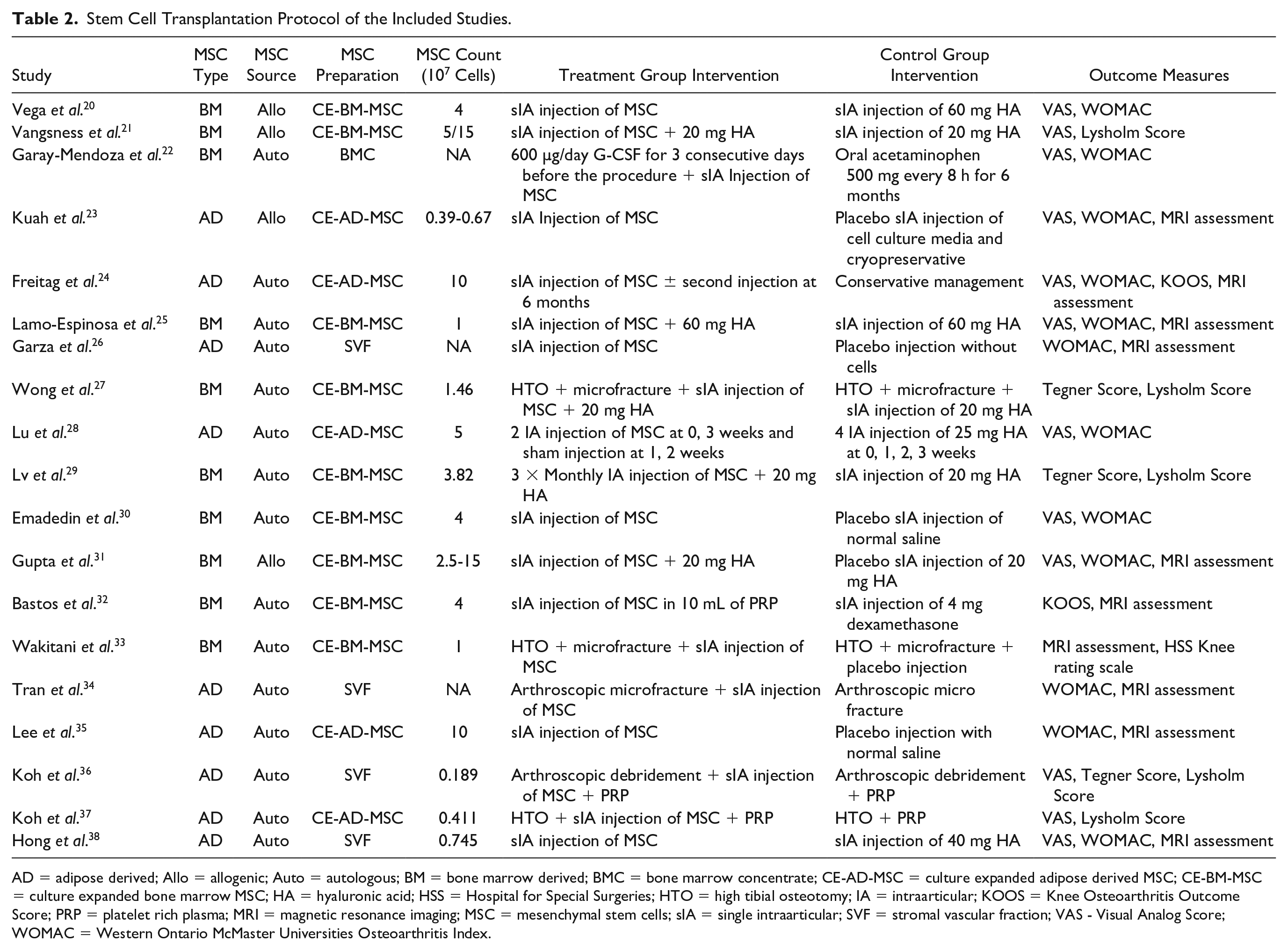
AD = adipose derived; Allo = allogenic; Auto = autologous; BM = bone marrow derived; BMC = bone marrow concentrate; CE-AD-MSC = culture expanded adipose derived MSC; CE-BM-MSC = culture expanded bone marrow MSC; HA = hyaluronic acid; HSS = Hospital for Special Surgeries; HTO = high tibial osteotomy; IA = intraarticular; KOOS = Knee Osteoarthritis Outcome Score; PRP = platelet rich plasma; MRI = magnetic resonance imaging; MSC = mesenchymal stem cells; sIA = single intraarticular; SVF = stromal vascular fraction; VAS - Visual Analog Score; WOMAC = Western Ontario McMaster Universities Osteoarthritis Index.
Quality Assessment
The methodological quality of the included studies has been mentioned in Figure 2 . None of the included studies had an overall high risk of bias to be excluded from the analysis.
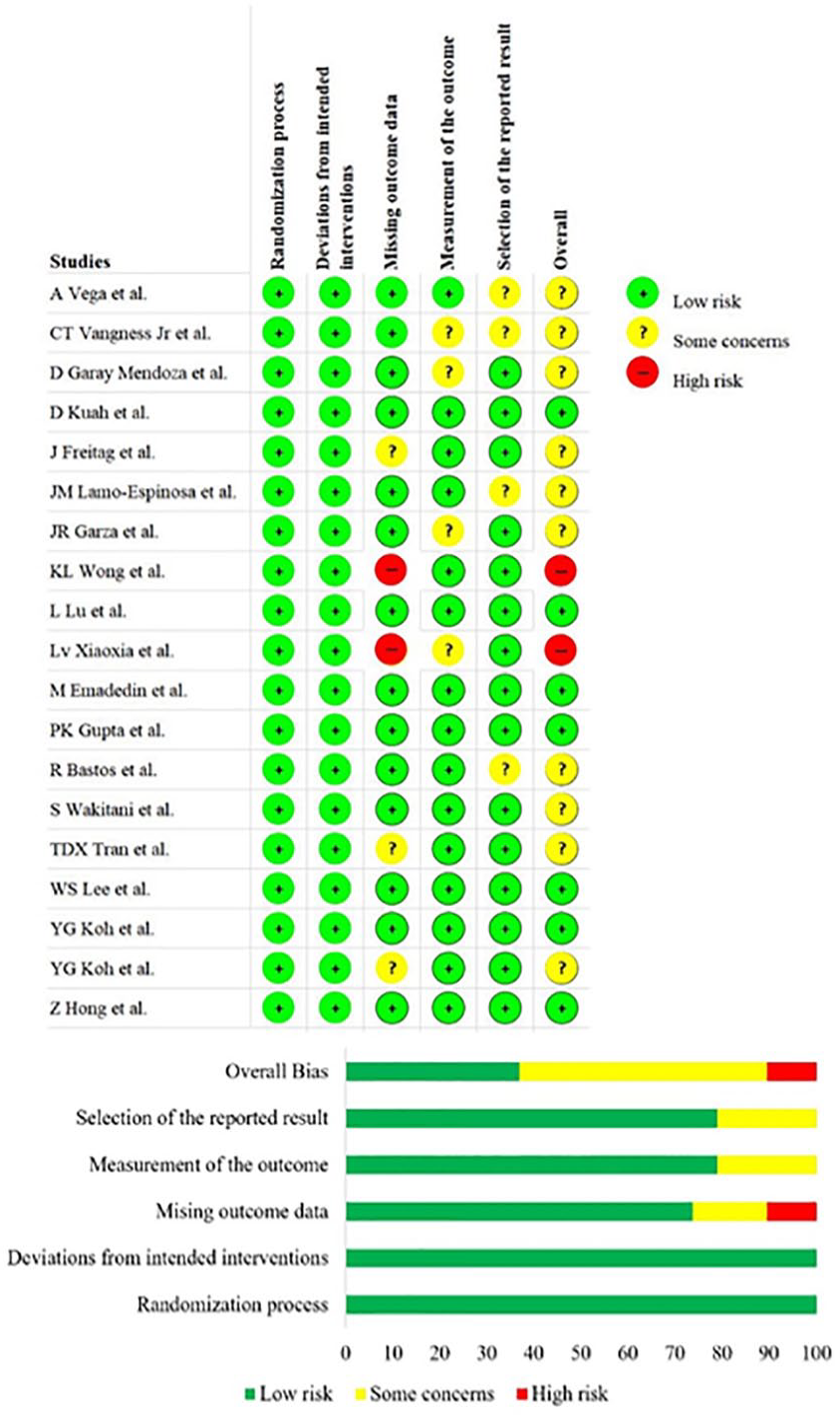
Methodological quality and risk of bias assessment of all the included studies.
Efficacy Outcomes
Visual Analog Scale for Pain at 6 Months
Five studies involving 249 patients reported VAS for pain at 6 months posttransplantation of BM-MSCs. There was a significant heterogeneity observed between the included studies (I2 = 94.17%, P < 0.001). Hence, the random-effects model was used for analysis. On analysis, no significant difference was noted compared to their controls at 6 months posttransplantation period (WMD = −9.549, 95% CI [−19.825, 0.727], P = 0.069; Fig. 3A ).
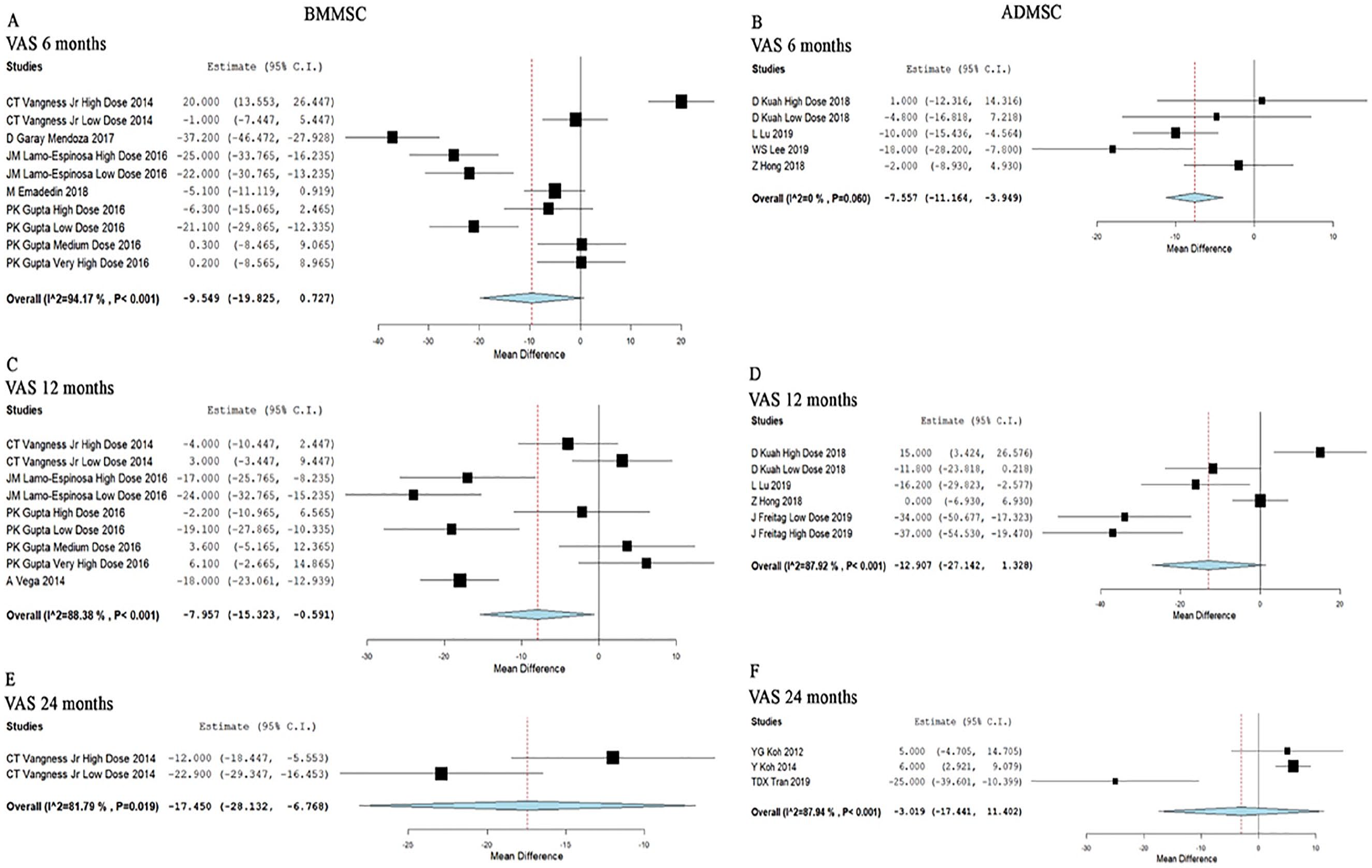
Forest plot of the included studies comparing BM-MSC and AD-MSC transplantation groups with their controls.
Four studies involving 136 patients reported VAS for pain at 6 months posttransplantation of AD-MSCs. There was a significant heterogeneity observed between the included studies (I2 = 60.63%, P = 0.026). Hence, the random-effects model was used for analysis. On analysis, a significant difference was noted compared to their controls at 6 months posttransplantation period (WMD = −13.268, 95% CI [−17.507, −9.030], P < 0.001; Fig. 3B ).
Visual Analog Scale for Pain at 12 Months
Four studies with 175 patients reported VAS for pain at 12 months posttransplantation of BM-MSCs. Since a significant heterogeneity observed between the included studies (I2 = 88.38%, P < 0.001), the random-effects model was used for analysis. On analysis, a significant difference was noted compared to their controls at 12 months posttransplantation period (WMD = −7.957, 95% CI [−15.323, −0.591], P = 0.034; Fig. 3C ).
Four studies with 135 patients reported VAS for pain at 12 months posttransplantation of AD-MSCs. Since a significant heterogeneity observed between the included studies (I2 = 87.92%, P < 0.001), the random-effects model was used for analysis. On analysis, no significant difference was noted compared to their controls at 12 months posttransplantation period (WMD = −12.907, 95% CI [−27.142, 1.328], P = 0.076; Fig. 3D ).
Visual Analog Scale for Pain at 24 Months
Only one study involving 55 patients reported VAS for pain at 24 months posttransplantation of BM-MSCs. There was a significant improvement compared to the controls at 24 months without any heterogeneity (WMD = −17.450, 95% CI [−28.132, −6.768], P < 0.001; Fig. 3E ).
Three studies involving 127 patients reported VAS for pain at 22 months posttransplantation of AD-MSCs. There was a significant heterogeneity observed between the included studies (I2 = 87.94%, P < 0.001). Hence, the random-effects model was used for analysis. On analysis, no significant difference was noted compared to their controls at 24 months posttransplantation period (WMD = −3.019, 95% CI [−17.441, 11.402], P = 0.682; Fig. 3F ).
With regard to pain by VAS, AD-MSCs showed significant immediate pain relief posttransplantation, which did not last in the long term. Even though BM-MSCs did not provide immediate significant pain relief as that of AD-MSCs, it provided significant pain relief at the long term.
WOMAC at 6 Months
Four studies involving 214 patients reported functional outcomes with WOMAC score at 6 months posttransplantation of BM-MSCs. There was a significant heterogeneity observed between the included studies (I2 = 96.84%, P < 0.001). Hence, the random-effects model was used for analysis. On analysis, no significant difference was noted compared to their controls at 6 months posttransplantation period (WMD = −6.774, 95% CI [−24.014, 10.467], P = 0.441; Fig. 4A ).
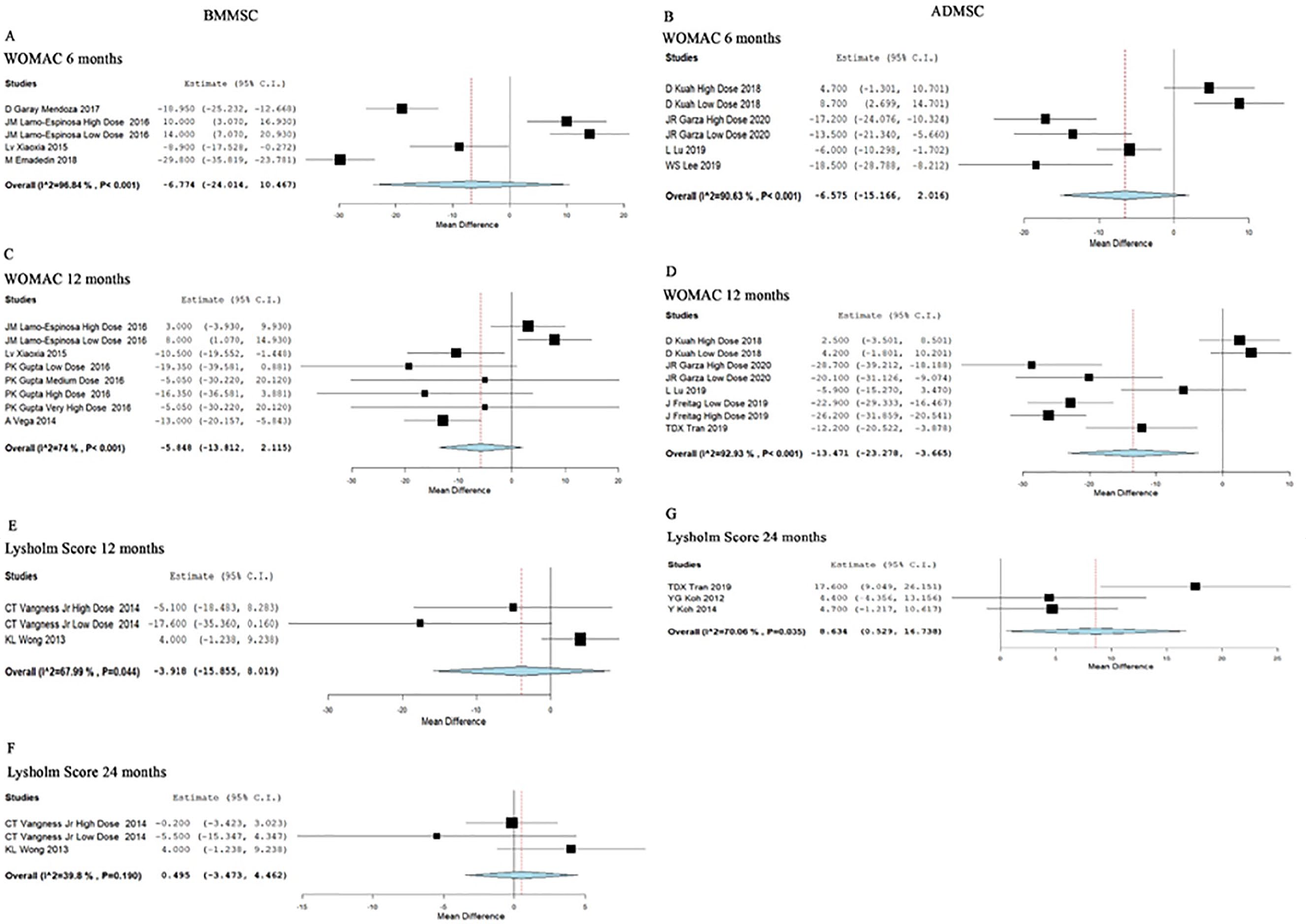
Forest plot of the included studies comparing BM-MSC and AD-MSC transplantation groups with their controls.
Four studies involving 136 patients reported functional outcomes with WOMAC score at 6 months posttransplantation of AD-MSCs. There was a significant heterogeneity observed between the included studies (I2 = 90.63%, P = <0.001). Hence, the random-effects model was used for analysis. On analysis, no significant difference was noted compared to their controls at 6 months posttransplantation period (WMD = −6.575, 95% CI [−15.166, 2.016], P = 0.134; Fig. 4B ).
WOMAC at 12 Months
Four studies involving 200 patients reported functional outcomes with WOMAC score at 12 months posttransplantation of BM-MSCs. There was a significant heterogeneity observed between the included studies (I2 = 74%, P < 0.001). Hence, the random-effects model was used for analysis. On analysis, no significant difference was noted compared to their controls at 12 months posttransplantation period (WMD = −5.848, 95% CI [−13.812, 2.115], P = 0.150; Fig. 4C ).
Four studies involving 175 patients reported functional outcomes with the WOMAC score at 12 months posttransplantation of AD-MSCs. There was a significant heterogeneity observed between the included studies (I2 = 92.93%, P = <0.001). Hence, the random-effects model was used for analysis. On analysis, a significant difference was noted compared to their controls at 12 months posttransplantation period (WMD = −13.471, 95% CI [−23.278, −3.665], P = 0.007; Fig. 4D ).
With regard to the functional outcome by the WOMAC score, AD-MSCs showed a significant improvement in functional outcome at 12 months posttransplantation period compared to their controls, while BM-MSCs failed to elicit a significant response neither at 6 nor at 12 months posttransplantation period.
Lysholm Knee Score at 12 Months
Two studies involving 111 patients reported functional outcome with Lysholm Knee Score at 12 months posttransplantation of BM-MSCs. There was a significant heterogeneity observed between the included studies (I2 = 67.99%, P = 0.044). Hence, the random-effects model was used for analysis. On analysis, no significant difference was noted compared to their controls at 12 months posttransplantation period (WMD = −3.918, 95% CI [−15.855, 8.019], P = 0.520; Fig. 4E ).
Only one study involving 33 patients reported Lysholm score at 6 months with no significant improvement posttransplantation of AD-MSCs.
Lysholm Knee Score at 24 Months
Two studies involving 111 patients reported functional outcome with Lysholm Knee Score at 24 months posttransplantation of BM-MSCs. There was no heterogeneity among the included studies (I2 = 39.8%, P = 0.190). Hence, the fixed-effects model was used for analysis. On analysis, no significant difference was noted compared to their controls at 24 months posttransplantation period (WMD = 0.495, 95% CI [−3.473, 4.462], P = 0.807; Fig. 4F ).
Three studies involving 127 patients reported functional outcome with Lysholm Knee Score at 24 months posttransplantation of AD-MSCs. There was significant heterogeneity among the included studies (I2 = 70.06%, P = 0.035). Hence, the random-effects model was used for analysis. On analysis, a significant difference was noted compared to their controls at 24 months posttransplantation period (WMD = 8.634, 95% CI [0.529, 16.738], P = 0.037; Fig. 4G ).
With regard to the functional outcome by Lysholm score, AD-MSCs showed a significant improvement in functional outcome at 24 months posttransplantation period compared to their controls, while BM-MSCs failed to elicit a significant response neither at 12 nor at 24 months posttransplantation period.
KOOS at 12 Months
Only one study involving 47 patients reported functional outcomes with KOOS at 12 months posttransplantation of BM-MSCs. There was no significant improvement compared to the controls at 12 months without any heterogeneity (WMD = 2.643, 95% CI [−9.047, 14.333], P = 0.658; Fig. 5C ).
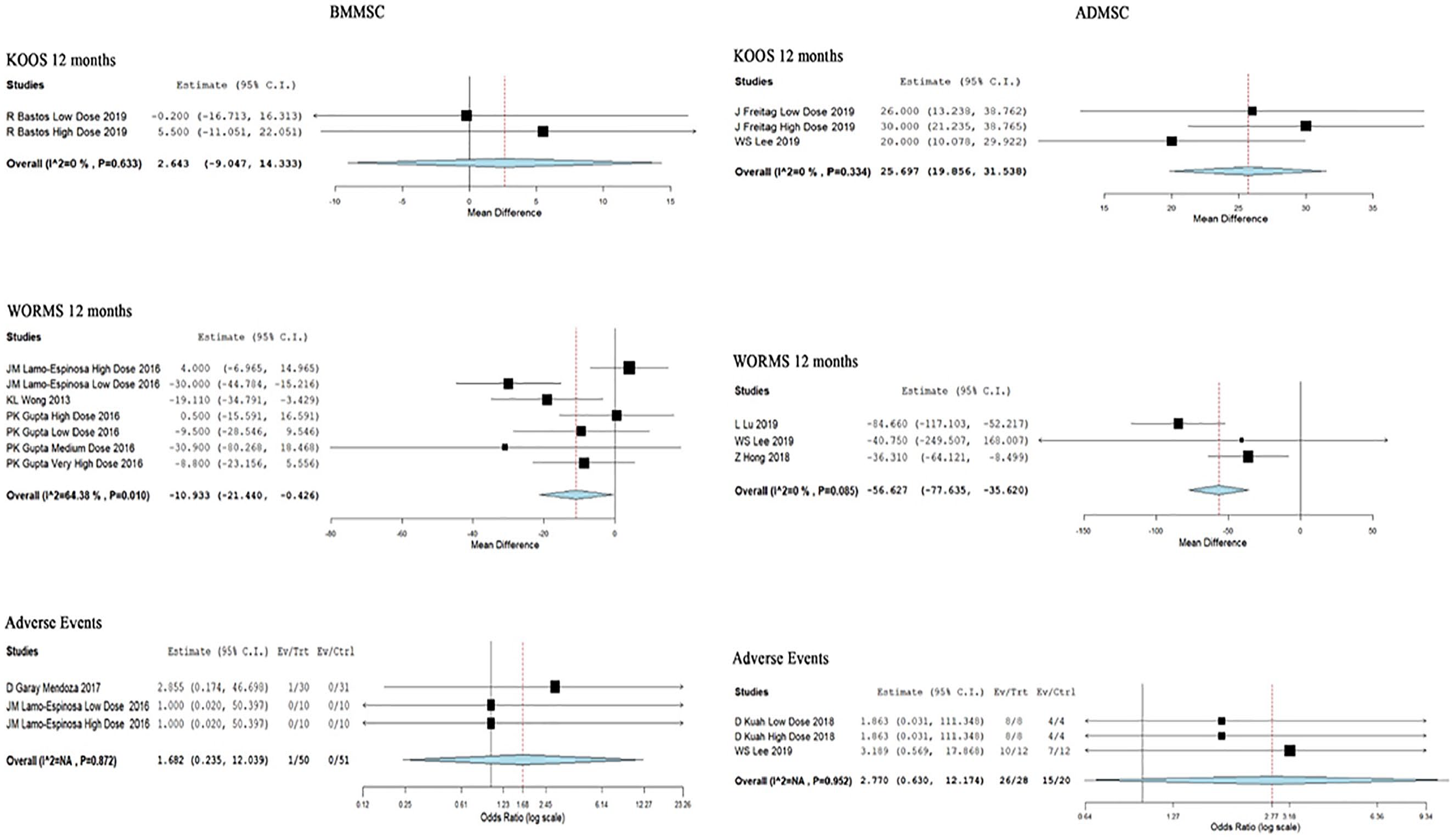
Forest plot of the included studies comparing BM-MSCs and AD-MSCs transplantation group with their controls.
Two studies involving 54 patients reported functional outcomes with KOOS at 12 months posttransplantation of AD-MSCs. There was no heterogeneity among the included studies (I2 = 0%, P = 0.334). Hence, the fixed-effects model was used for analysis. On analysis, a significant difference was noted compared to their controls at 12 months posttransplantation period (WMD = 25.697, 95% CI [19.856, 31.538], P < 0.001; Fig. 5D ).
With regard to the functional outcome by KOOS, AD-MSCs showed a significant improvement in functional outcome at 12 months posttransplantation period compared to their controls while BM-MSCs failed to elicit a significant response.
WORMS at 12 Months
Three studies involving 146 patients reported radiological outcomes with MRI based on WORMS at 12 months posttransplantation of BM-MSCs. There was significant heterogeneity among the included studies (I2 = 64.38%, P = 0.010). Hence, the random-effects model was used for analysis. On analysis, a significant difference was noted compared to their controls at 12 months posttransplantation period by regeneration in the cartilage noted on MRI (WMD = −10.933, 95% CI [−21.440, −0.426], P = 0.041; Fig. 5A ).
Three studies involving 109 patients reported radiological outcomes with MRI based on WORMS at 12 months posttransplantation of AD-MSCs. There was no heterogeneity among the included studies (I2 = 0%, P = 0.085). Hence, the fixed-effects model was used for analysis. On analysis, a significant difference was noted compared to their controls at 12 months posttransplantation period by regeneration in the cartilage noted on MRI (WMD = −56.627, 95% CI [−77.635, −35.620], P < 0.001; Fig. 5B ).
Although both AD-MSCs and BM-MSCs showed a significant regeneration of the targeted cartilage based on MRI evaluation, AD-MSCs (P < 0.001) outperformed BM-MSCs (P = 0.041) at 12 months posttransplantation period compared to their controls.
Safety
Two studies involving 91 patients reported adverse effects with low heterogeneity among the included studies with BM-MSCs (I2 = 0.0%, P = 0.872). Hence, a fixed-effects model was used for analysis. There was no significant increase in the adverse events compared to the controls (RR = 1.682, 95% CI [0.235, 12.039], P = 0.604; Fig. 5E ).
Two studies involving 44 patients reported adverse effects with low heterogeneity among the included studies with AD-MSCs (I2 = 0.0%, P = 0.952). Hence, a fixed-effects model was used for analysis. There was no significant increase in the adverse events compared to the controls (RR = 2.770, 95% CI [0.630, 12.174], P = 0.177; Fig. 5F ).
The commonly reported adverse events of the intervention include minor discomfort and bruising which resolved spontaneously or with treatment. However, no major serious adverse events with permanent effects such as death, tumor, or immune reaction to the intervention were noted during follow-up.
Subgroup Analysis
We performed subgroup analysis among the studies utilizing AD-MSCs based on the dose of the MSCs transplanted and adjuvant surgeries associated with the transplantation procedure.
For analysis of the dosage of the AD-MSCs transplanted we categorized the available studies using AD-MSCs into 2 subgroups. Studies using dosage <5 × 107 cells were compared with studies using dosage ≥5 × 107 cells across various outcome measures. On analysis, it was established that studies involving MSC dosage ≥5 × 107 cells showed statistically significant outcomes than the studies with MSC dosage <5 × 107 cells as shown in Table 3 .
Subgroup Analysis of the Included Studies.
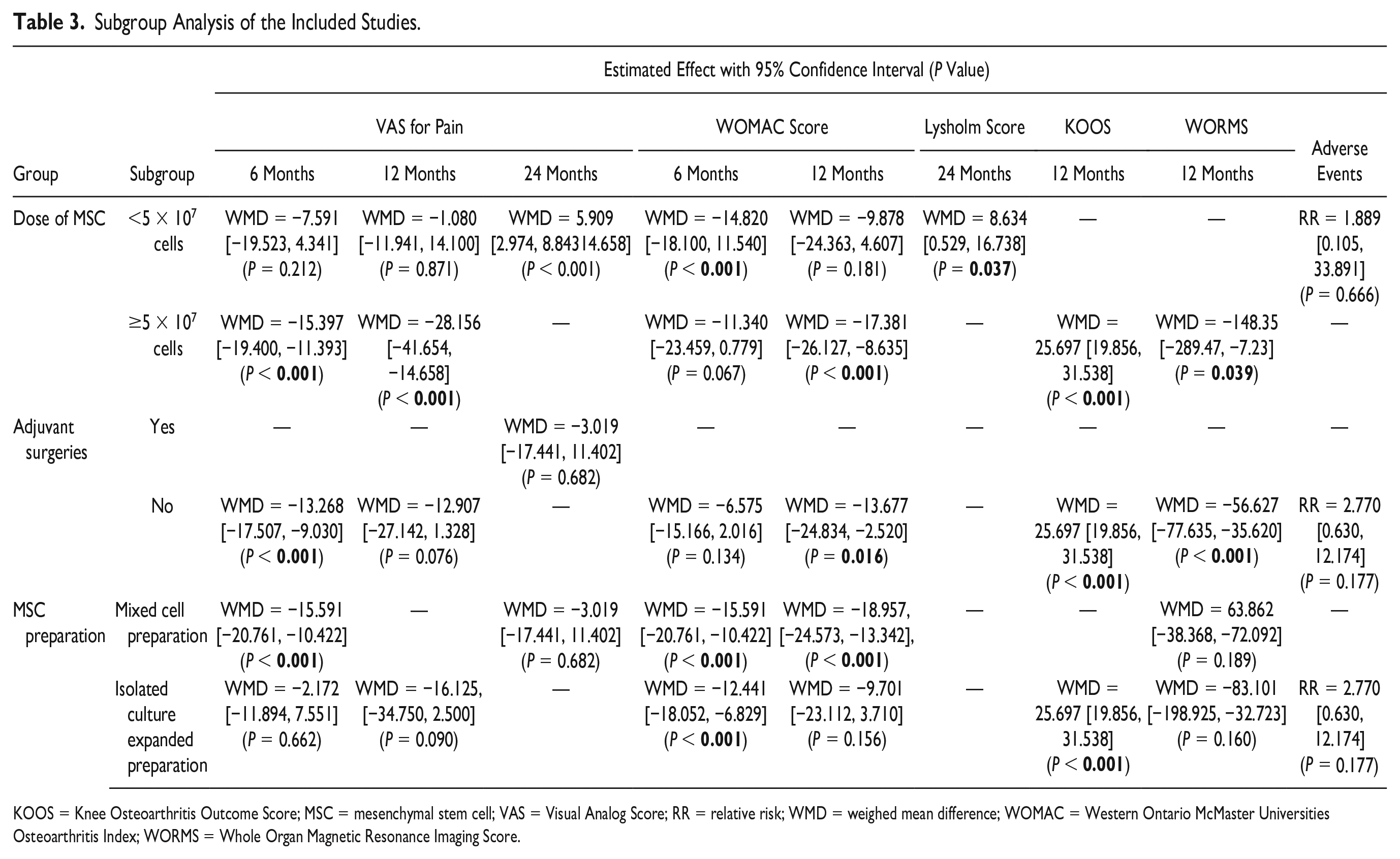
KOOS = Knee Osteoarthritis Outcome Score; MSC = mesenchymal stem cell; VAS = Visual Analog Score; RR = relative risk; WMD = weighed mean difference; WOMAC = Western Ontario McMaster Universities Osteoarthritis Index; WORMS = Whole Organ Magnetic Resonance Imaging Score.
We also compared the outcome across studies that used adjuvant surgeries like high tibial osteotomy, arthroscopic debridement or arthroscopic microfracture to facilitate cartilage regeneration with other studies that used only AD-MSC transplantation. On analysis, it was noted that statistically significant outcomes were achieved in studies without the use of any adjuvant surgical procedures as shown in Table 3 .
On analyzing the effect of MSC preparation methods, we noted that mixed cell preparation was not inferior to isolated culture expanded preparation of MSC among the outcome measures available for comparison between them.
Sensitivity Analysis
A sensitivity analysis was performed in each analysis. All the results (VAS for Pain, WOMAC, Lysholm, WORMS, KOOS, and adverse events) were not significantly altered by sequentially omitting each study in the meta-analysis. On the other hand, consistency of the results was maintained after reanalysis by changing to the random-effects model.
Publications Bias
Publication bias was analyzed utilizing the Funnel plot and Egger regression test and for the meta-analysis of efficacy and safety of BM-MSCs and AD-MSCs in the management of osteoarthritis of the knee, there was no evidence of publication bias by Egger regression test (P = 0.564) and funnel plot as shown in Figure 6 . All the studies lied within the 95% CI and were distributed evenly about the axes, implying minimal publication bias.
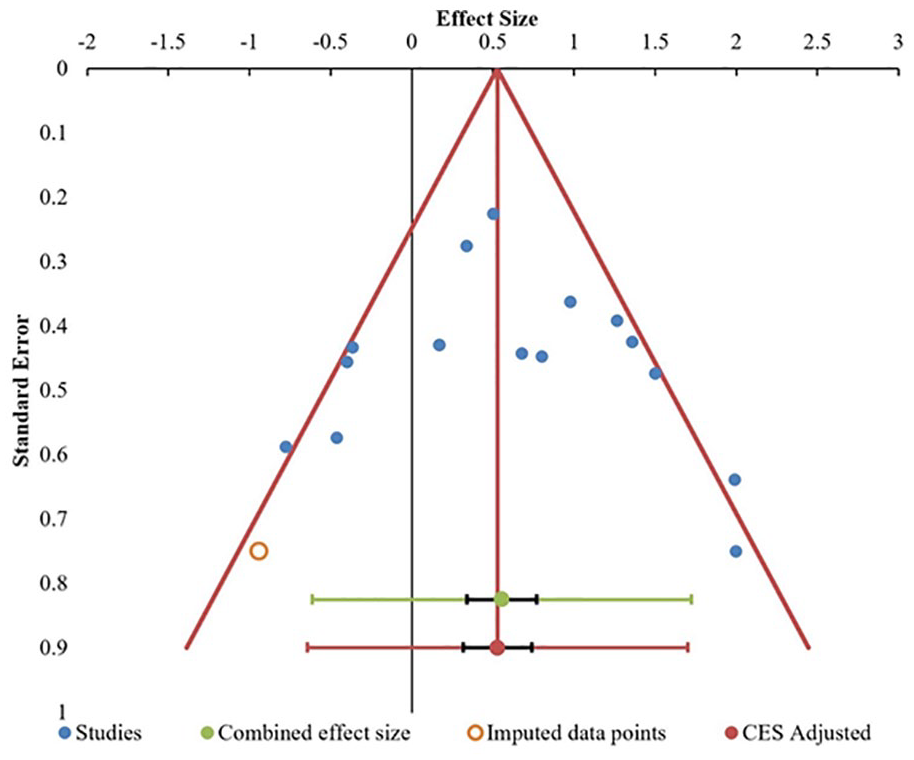
Publication bias assessment with funnel plot for Visual Analog Score at 12 months in the included studies.
Discussion
In an era of modern orthopedics, regenerative and translational medicine has revolutionized the newer modalities of management to bridge the gap between medical and surgical management for osteoarthritis of knees. MSCs have the ability of differentiation, plasticity, immunomodulatory, immune evasive, antimicrobial, and anti-inflammatory properties. 39 MSCs work on the principles of neo-angiogenesis and anti-apoptosis with the help of growth factors, cytokines, chemokines, and bioactive micromolecules released by MSCs. 40 The selection of MSC in treatment of osteoarthritis knees are imperative in achieving functional results. The isolation, harvest, preparation, and characterization of MSCs from bone marrow aspirate concentrate (BMAC) and stromal vascular fraction (SVF), respectively, follow different kinetics. 41 The mechanism of action of AD-MSCs and BM-MSCs leading to functional and structural benefits is dependent upon the number and quality of the delivered MSCs.
Main Finding
We comprehensively and systematically reviewed all the available literature on MSC transplantation for OA knee and found the following:
MSC transplantation showed a statistically significant improvement in all functional outcome measures like VAS for Pain, WOMAC, Lysholm, KOOS, and radiological outcome parameters like WORMS at varied time intervals with their corresponding controls. It is promising to see the regenerative potential of the MSCs to repair the degenerated cartilage at the articular surface.
At 6 months, AD-MSCs showed significantly better VAS (P < 0.001, P = 0.069) and WOMAC (P = 0.134, P = 0.441) improvement than BM-MSCs, respectively, compared to controls. At 1 year, AD-MSCs outperformed BM-MSCs compared to their control in measures like WOMAC (P = 0.007, P = 0.150), KOOS (P < 0.001, P = 0.658), and WORMS (P < 0.001, P = 0.041), respectively. Similarly at 24 months, AD-MSCs showed significantly better Lysholm Score (P = 0.037) than BM-MSCs (P = 0.807) although VAS improvement was better with BM-MSCs at 24 months (P < 0.001). There were no significant adverse events with either of the MSCs compared to their controls.
Comparison with Other Studies
Although several meta-analyses were analyzing the efficacy and safety of MSC therapy for osteoarthritis of the knee,42-45 only meta-analysis by Han et al. 46 with 9 studies including 377 patients answered our research question. The major limitation of their study was less number of studies included and the lack of subgroup analysis. We not only had more studies included in our analysis(n = 19; 811 patients) but also analyzed them in terms of dosage of MSC transplanted to throw more light on the available evidence and identify the potential lacunae in literature which will indirectly widen the scope for future research. Moreover, apart from subjective functional outcome measures, we also included an objective MRI based outcome measure to further strengthen the results of our analysis.
Our subgroup analysis of the dosage of MSC transplantation was concurrent with the meta-analysis by Doyle et al. 47 who concluded that moderate-high cell numbers (4 × 107) were most likely to achieve optimal response in osteoarthritis of the knee. Our study identified a cutoff limit of 5 × 107 cells, which was sufficient for significant functional benefits of MSC therapy compared to the higher dosage which comes at a cost with a higher risk of adverse events. Our analysis also established that adjuvant surgical procedures are not necessary to achieve optimal results from MSC transplantation for cartilage regeneration.
Despite following the recommendation for minimal manipulation of cellular products by federal regulations, 48 expanded stem cells have to be checked for tumorigenic and immune-evasive potentials before administering for treatment. In the available literature, there are no studies that compares between the effect of BMAC and culture expanded BM-MSCs or SVF and culture expanded AD-MSCs for treating osteoarthritis of knee.
Researchers consider culture expanded stem cells to be a superior source for cartilage regeneration than mixed cell preparation. Hence, we tested the hypothesis by subgroup analysis among AD-MSCs source from the studies included. From our analysis, noninferiority was established for the mixed cell culture preparation (SVF) when its outcomes were compared to that of the culture expanded AD-MSCs. Although culture expanded AD-MSCs has increased number of stem cells than the uncultured isolated stem cells in SVF, the quality remains the same as established by their equivalence in functional outcome from the included studies. Hence it could be derived from our results that the functional outcome of cellular therapy depends more on the quality of cytokines, chemokines, and growth factors released from the stem cells than quantity of stem cells.
The selection of sources of MSCs in the treatment of osteoarthritis knees is imperative in achieving optimal functional results. Wolfstadt et al. 49 stated that MSCs obtained from bone marrow are relatively less in volume especially among the elderly population. Hass et al. 50 concluded AD-MSCs give an MSC yield of 500-fold more than BM-MSCs. Kim and Im 51 concluded that BM-MSCs are more prone toward cartilage regeneration than AD-MSC, but addition of growth factors and cytokines to SVF derived from AD-MSC, cartilage growth potential can be enhanced. Despite the difficulty in the preparation of SVF compared to BMAC, the quantity of MSCs derived from adipose tissue is relatively higher than bone marrow. A few studies proved that the MSCs from synovium have greatest chondrogenic potential.52-54 A large number of patients have to be recruited for testing the efficacy and functional outcome of synovial MSCs for osteoarthritis knees.
Direction for Future Research
Although MSCs have a potential role in the management of osteoarthritis of the knee, the scope of regenerative and translational medicine in this field needs to be evaluated by large randomized controlled interventional trials for the optimization of therapeutic protocols in terms of the type of MSC, preparation methods, quality, and quantity of MSCs to be transplanted. Ethical issues involved in minimal manipulation of tissue and cellular products and its functional outcome have to be addressed. Challenges and logistics involved to channelize stem cell basics into optimal clinical practice need an interdisciplinary approach to make this opportunity a reality in the management of osteoarthritis of the knee.
Limitations
Our analysis has some limitations. Although we used MRI-based outcome measure (WORMS) to objectively analyze the effect of MSC transplantation, most of the studies included in the analysis used functional outcome measures that were all subjective, which bears an inherent risk of bias. Blinding was not established in most of the studies which might invite room for treatment bias from patient or observer. Heterogeneity was observed in most of the outcomes reported across the studies which might be due to the variability in the treatment protocols followed in the individual studies as shown in Table 2 . Moreover, patients in various stages of the disease process were included in the studies which might also contribute to the heterogeneity of their results. Hence, a large multicentric trial with standardized dosage and frequency protocol with established outcome assessment measures, without any adjuvant procedures is needed to further confirm the results of our analysis.
Conclusion
To overcome the morbidity and to improve the functional quality of patients with osteoarthritis of the knee, the transplantation of mesenchymal stem cells plays a major role to bridge the gap between conservative and surgical care. Our analysis establishes the efficacy, safety, and superiority of AD-MSC transplantation, compared to BM-MSC, in the management of osteoarthritis of the knee. However, research and developmental work need to be further directed to standardize the dose and validate our results on the effect of the source of MSCs used for transplantation to achieve optimal results.
Footnotes
Author’s Note
Sathish Muthu is also affiliated with Orthopaedic Research Group, Coimbatore, Tamil Nadu, India.
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
