Abstract
Objective
The objective of this systematic review and meta-analysis was to report the safety of intra-articular hyaluronic acid (IAHA) in patients with symptomatic knee osteoarthritis (OA).
Methods
We identified randomized controlled trials reporting the safety of IAHA versus IA saline in adults with symptomatic knee OA. Main safety outcomes were adverse events (AEs), local AEs, serious adverse events (SAEs), study withdrawals, and AE-related study withdrawals.
Results
A total of 35 randomized controlled trials with 38 group comparisons comprising 8,078 unique patients (IAHA: 4,295, IA saline: 3,783) were included in the meta-analysis. Comparing IAHA with IA saline over a median of 6 months follow-up, there were no differences in the risk of AEs (42.4% vs. 39.7%, risk ratio [RR] = 1.01, 95% CI = 0.96-1.07, P = 0.61), SAEs (1.8% vs. 1.2%, RR = 1.44, 95% CI = 0.91-2.26, P=0.12), study withdrawals (12.3% vs. 12.7%, RR = 0.99, 95% CI = 0.87-1.12, P = 0.83), or AE-related study withdrawals (2.7% vs. 2.1%, RR = 1.37, 95% CI = 0.97-1.93, P = 0.08). Local AEs, all of which were nonserious, were more common with IAHA vs. IA saline (14.5% vs. 11.7%, RR = 1.21, 95% CI = 1.07-1.36, P = 0.003) and typically resolved within days.
Conclusion
IAHA was shown to be safe for use in patients with symptomatic knee OA. Compared with IA saline, IAHA is associated with an increased risk of nonserious, transient local reactions. There was no evidence to suggest any additional safety risks of IAHA.
Introduction
Knee osteoarthritis (OA) is the leading cause of disability in adults1-3 and its prevalence is anticipated to rise exponentially over the next several decades. The pathogenesis of knee OA is multifactorial, but largely attributable to chronic overloading of the knee joint that promotes degradation of the articular cartilage.4,5 Hyaluronic acid (HA) is an integral component of synovial fluid. As part of its intra-articular function, it acts as a joint lubricant during shear stress and a shock absorber during compressive stress. The concentration and molecular weight of endogenous HA is often reduced in knee OA, leading to reduced viscoelastic properties of synovial fluid. Altered biodynamics with loss of other protective HA properties induce proinflammatory pathways. 6 Intra-articular hyaluronic acid (IAHA) injections are intended to alleviate knee OA symptoms by reversing the OA-induced HA deficit. 7 Numerous systematic reviews have evaluated the effectiveness of IAHA for knee OA, with varying conclusions.8-16 However, relatively less attention has been paid to the safety of IAHA. In 2012, Rutjes and colleagues 15 were among the first to report potential safety concerns with IAHA for knee OA. Since then, few studies have attempted to confirm the safety of IAHA and to further investigate the potential sources of heterogeneity among studies. The purpose of this systematic review and meta-analysis of randomized controlled trials (RCTs) was to report on the safety of IAHA in patients with symptomatic knee OA.
Methods
Data Sources and Searches
We developed and followed a review protocol that adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). 17 We searched Medline, Embase, the Cochrane Database of Systematic Reviews, and the Directory of Open Access Journals, with no language restrictions, from inception to December 31, 2018 for RCTs of IAHA for symptomatic knee OA by using a combination of study design-, diagnosis-, and treatment-specific keywords (Supplemental Table 1). Additionally, reference lists of included papers and relevant meta-analyses were manually searched. We also included results from published and unpublished sources (e.g. abstracts, Food and Drug Administration website) to reduce the risk of publication bias. 18 Manuscripts published in non-English language journals were translated to English by local-language medical translators.
Study Selection
Two independent researchers (LM, DF) reviewed titles and abstracts for possible inclusion. Primary inclusion criteria were RCTs of IAHA (IAHA group) versus IA saline (Saline group) for symptomatic knee OA; sample size at least 30 patients per group; identical treatment and follow-up conditions in each group; and at least one extractable safety outcome. The 30-patient minimum sample size criterion was included to minimize bias associated with small-study effects. 19 Duplicate publications and studies that included concomitant surgical procedures were excluded.
Data Extraction
Data were independently extracted from eligible studies by 2 researchers (LM, DF). Data extraction discrepancies between the 2 researchers were resolved by discussion and consensus. The following variables were recorded in standardized data extraction forms: general manuscript information, patient characteristics (gender, age, body mass index), study characteristics (sample size, blinding assessment, industry funding, follow-up duration), HA characteristics (trade name, molecular weight, source, cross-linking, regulatory status), procedural details (number of injections), and safety outcomes. Safety outcomes included all-cause serious adverse events (SAEs), all-cause adverse events (AEs), local AEs, patient withdrawals, and AE-related patient withdrawals. Serious AE data were extracted based on investigator determination or report of any event that led to death, serious deterioration in health, life-threatening illness/injury, permanent impairment, hospitalization or prolongation of hospitalization, or medical/surgical intervention to prevent permanent impairment. Treatment relatedness data were extracted based on investigator determination. We reviewed the article text, tables, and patient flow diagrams to determine the number of patient withdrawals in total as well as those occurring due to an AE. The Cochrane Collaboration tool was used to assess risk of bias. 20
Data Analysis
Safety outcomes in each group were compared using the risk ratio (RR) where an RR >1 indicated higher risk with IAHA and an RR <1 indicated lower risk with IAHA. When one group was compared to multiple groups within the same study, the sample size of the single group was adjusted to avoid double counting of patients. 21 We used the I2 statistic to estimate heterogeneity of outcomes among studies where a value of 0% represents no heterogeneity and larger values represent increasing heterogeneity. 22 Substantial heterogeneity was defined by a Cochran Q test P < 0.1 or I2 > 50%. When substantial heterogeneity existed, a random effects model was planned; otherwise, a fixed-effect model was planned. 23 Publication bias was visually assessed with funnel plots and quantitatively assessed with Egger’s regression test. 24 Predefined subgroup analyses were performed to explore the association of study-level factors on each safety outcome. We tested the stability of our estimates with two sensitivity analyses. The first sensitivity analysis was a one-study removed analysis in which we iteratively removed one study at a time to determine whether conclusions for each outcome were influenced by any single study. The second sensitivity analysis compared results of fixed-effect versus random effects models for each safety outcome. P-values were 2-sided with a significance level <0.05. Analyses were performed using Review Manager 5.3 (The Cochrane Collaboration, Copenhagen, Denmark).
Results
Study Selection
We screened 459 studies to determine eligibility for inclusion in this review. We excluded 424 studies, mostly due to a non-IA saline control group, inadequate sample size, or nonrandomized design. Ultimately, 35 RCTs with 38 group comparisons comprising 8,078 unique patients (IAHA: 4,295, Saline: 3,783) were included in this review. A flow diagram of study identification and selection is shown in Figure 1 . A listing of excluded RCTs with reasons for exclusion is provided in Supplemental Table 2.
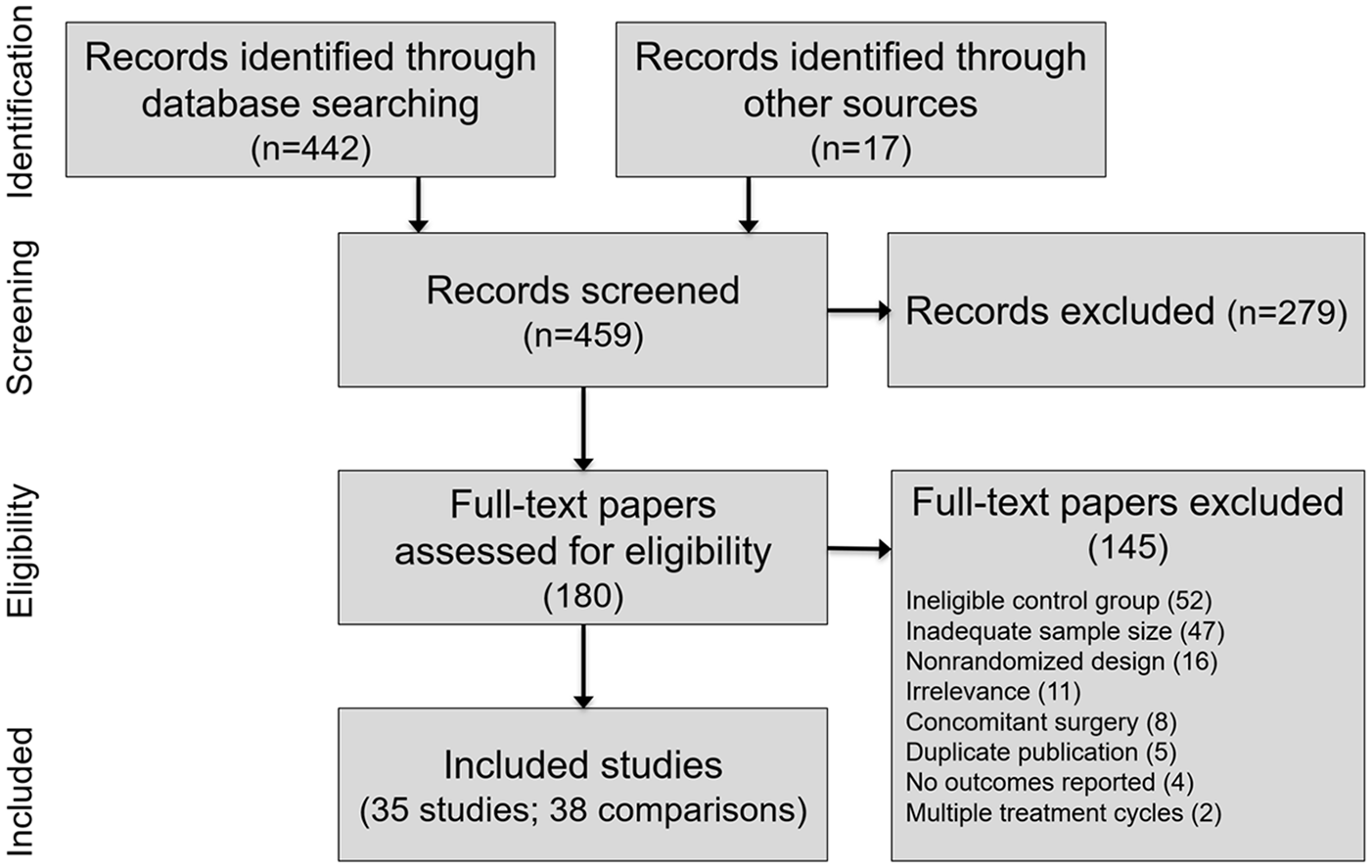
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram.
Patient and Study Characteristics
Baseline patient characteristics in the IAHA and Saline groups are reported in Table 1 . Characteristics of patients in the IAHA and Saline groups were similar, with women comprising 60% of each group, a median age of 63 years in each group, and a median body mass index of 29 kg/m2 in each group. Most studies utilized patient and outcome assessor blinding, but injector blinding was rarely specified. Most HA products were derived from avian sources. The total number of injections received in each series ranged from 1 to 5, except for one study that used 11 injections. 25 Median patient follow-up was 6 months (range: 5 weeks to 2 years) ( Table 2 ). The most common potential sources of bias were lack of investigator blinding, industry funding, and incomplete outcome data ( Figure 2 ).
Patient Characteristics in Randomized Controlled Trials Included in the Meta-Analysis.
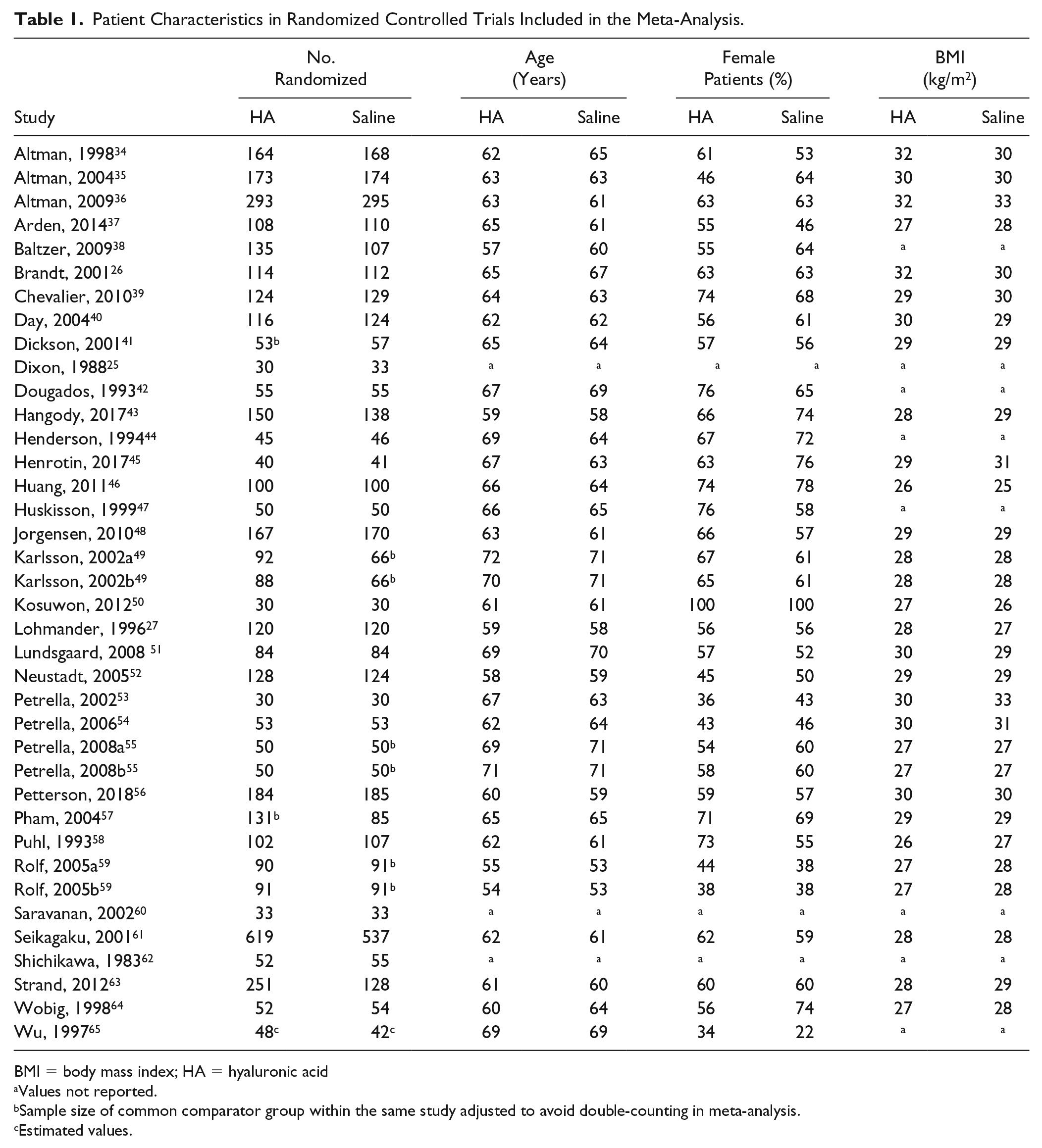
BMI = body mass index; HA = hyaluronic acid
Values not reported.
Sample size of common comparator group within the same study adjusted to avoid double-counting in meta-analysis.
Estimated values.
Treatment Regimens of Randomized Controlled Trials Included in the Meta-Analysis.
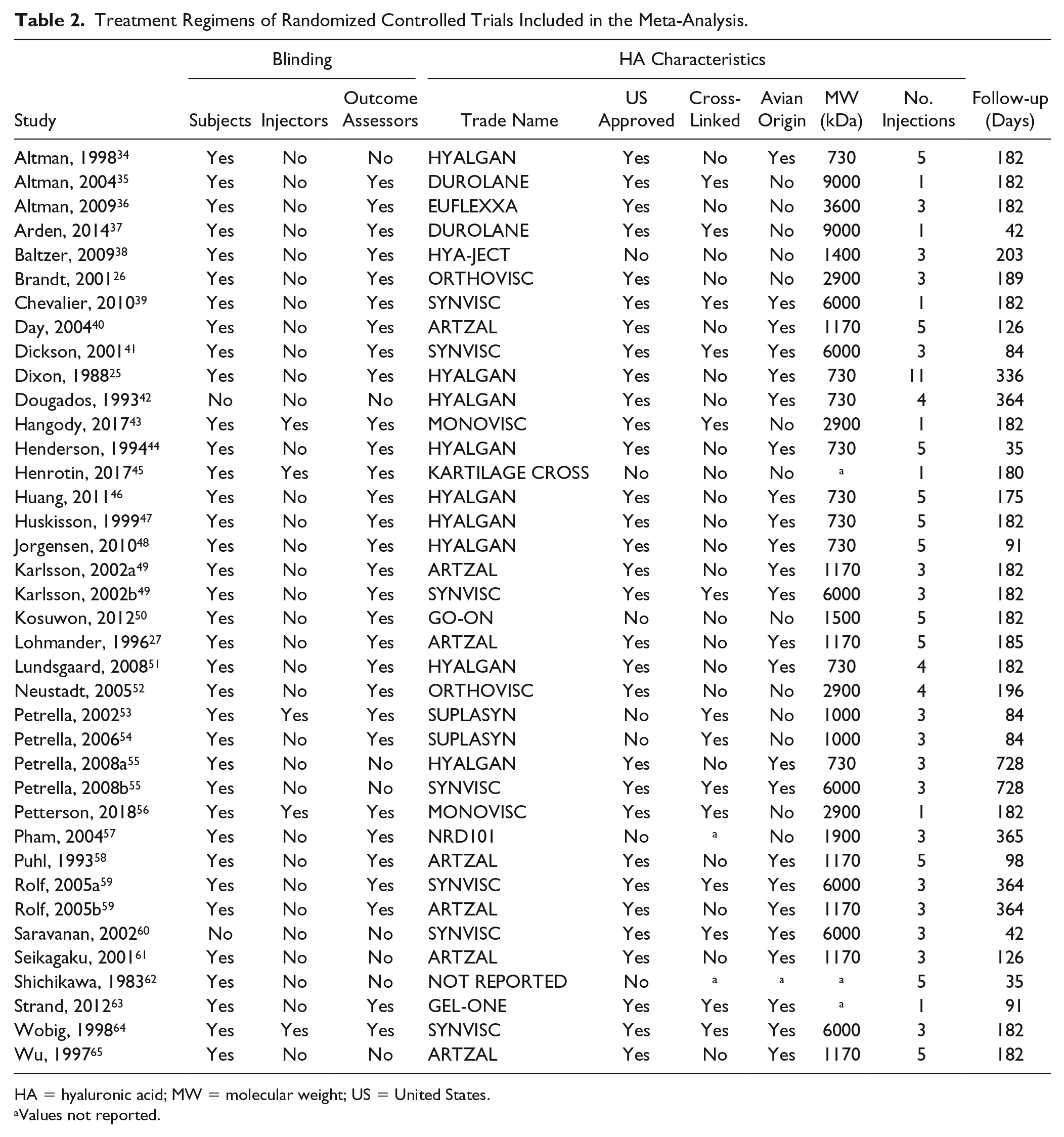
HA = hyaluronic acid; MW = molecular weight; US = United States.
Values not reported.
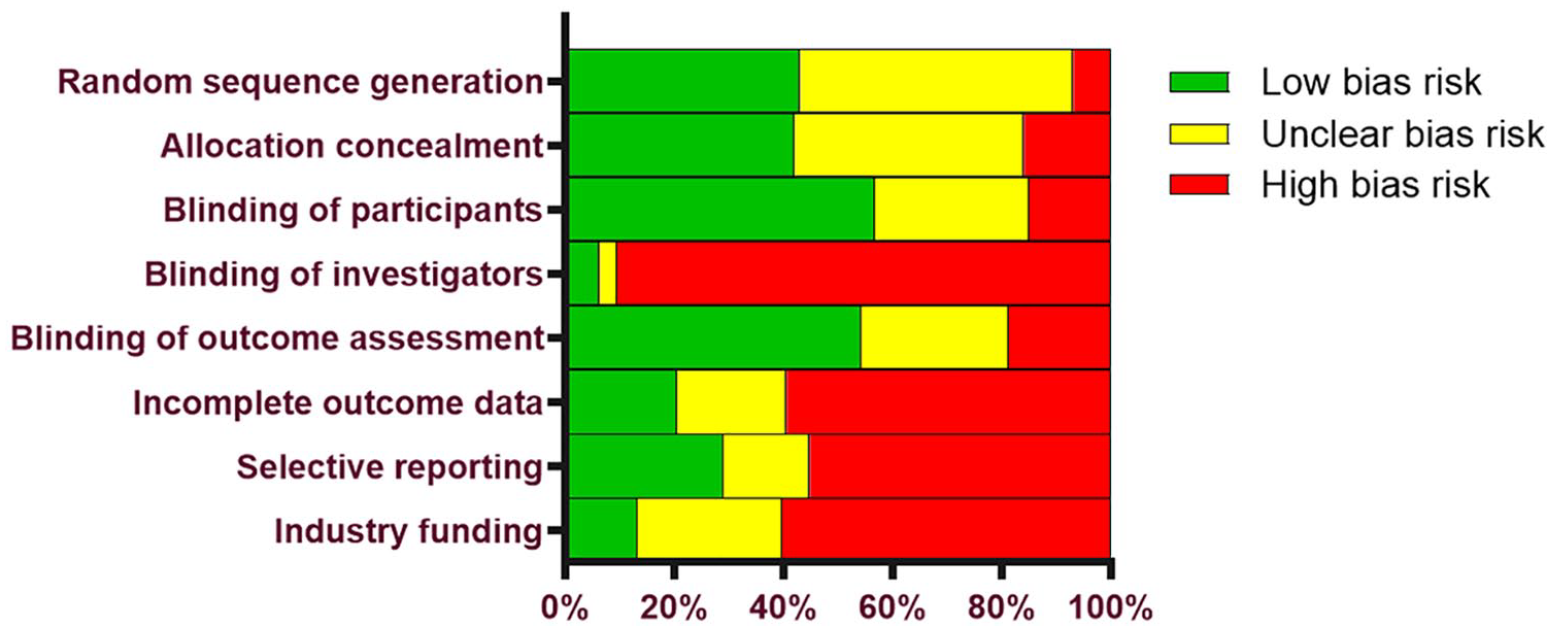
Risk of bias assessment with the Cochrane Collaboration Tool.
Main Outcomes
Comparing IAHA with Saline groups, there were no differences in the risk of AEs (42.4% vs. 39.7%, RR = 1.01, 95% CI = 0.96-1.07, P = 0.61;
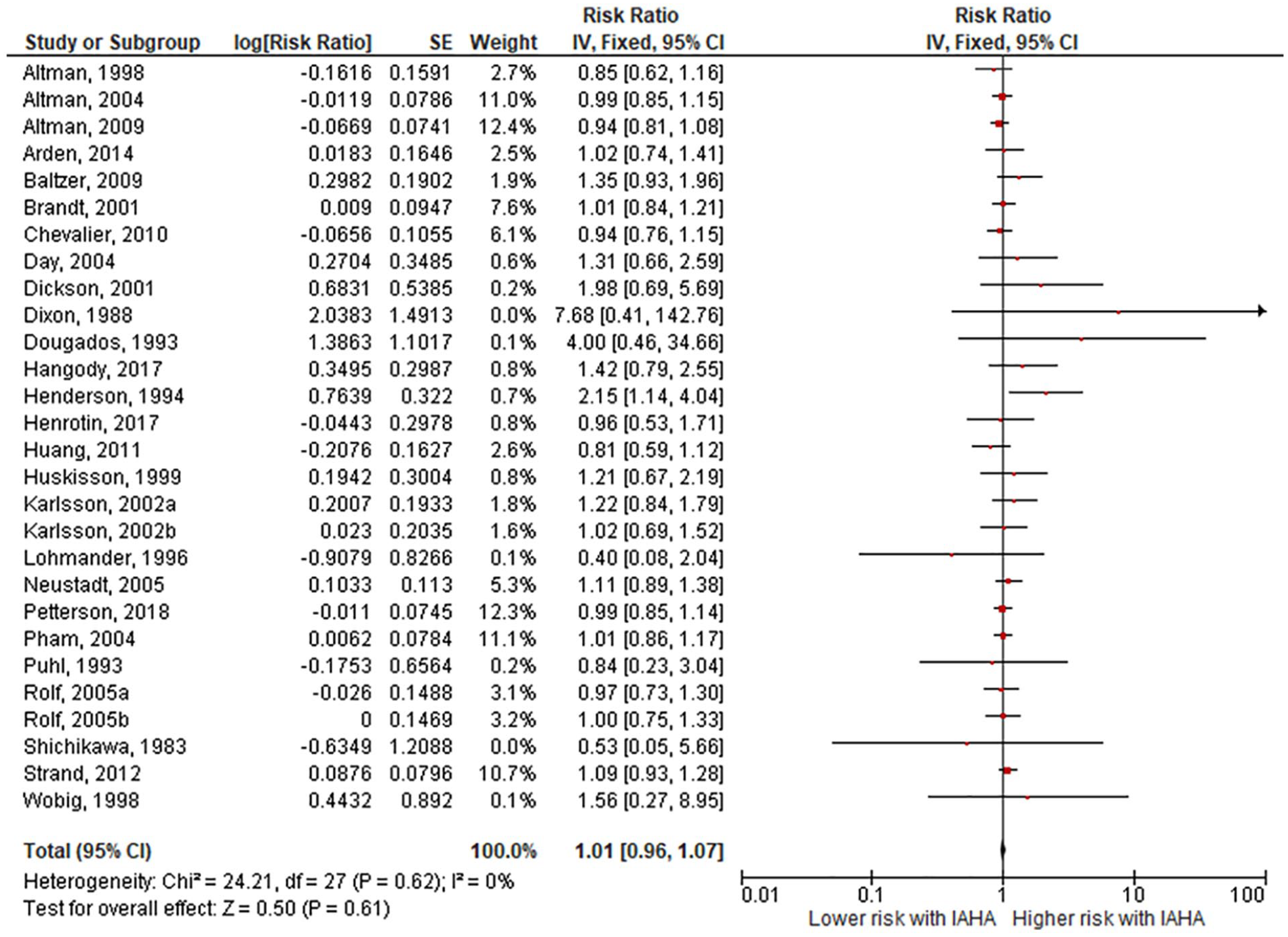
Forest plot of pooled risk ratio for adverse events with hyaluronic acid (HA) or saline injections. The risk ratio and 95% confidence interval are plotted for each study. The size of the square is proportional to the sample size of the study. The pooled risk ratio is denoted by the diamond apex and 95% confidence interval denoted by the diamond width. A pooled risk ratio of more than 1 indicates higher risk with HA. A pooled risk ratio of less than 1 indicates lower risk with HA. Heterogeneity: I2 = 0%, P = 0.62. Publication bias: Egger’s P = 0.04.
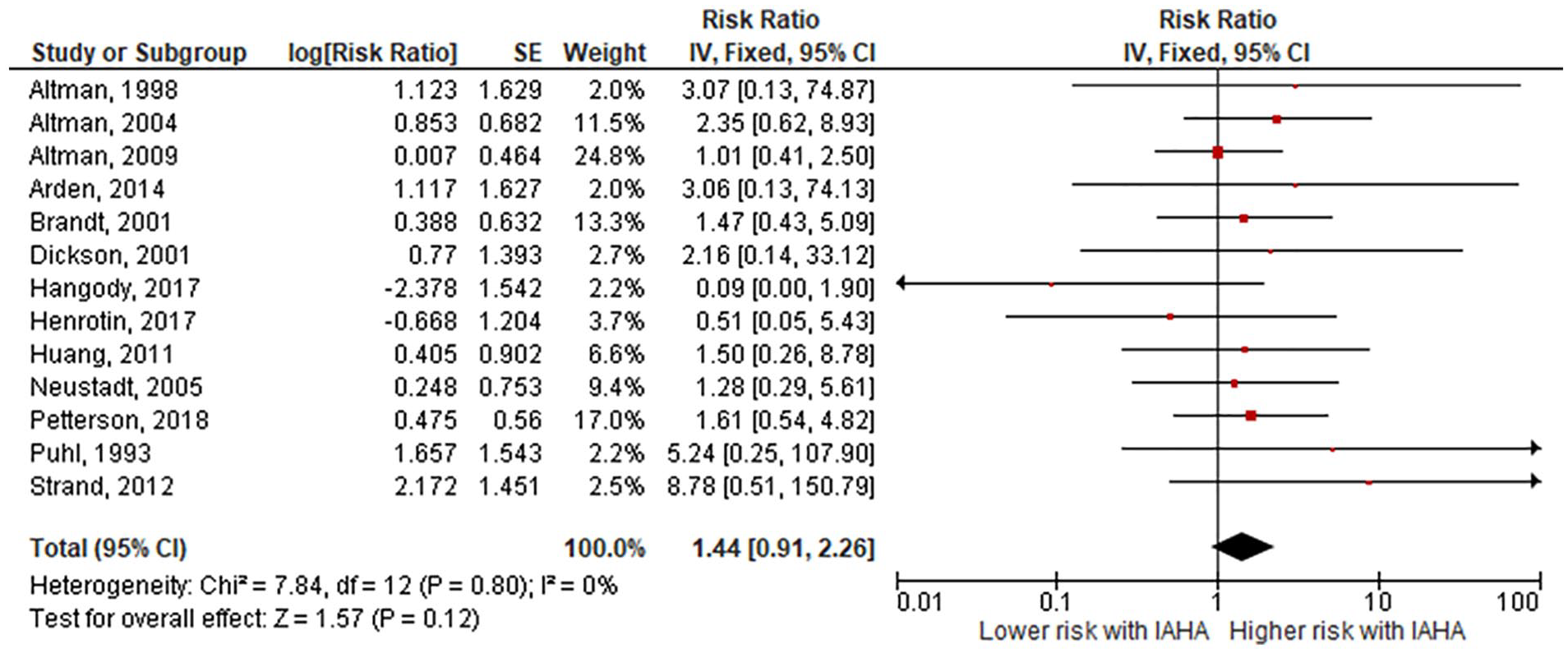
Forest plot of pooled risk ratio for serious adverse events with hyaluronic acid (HA) or saline injections. The risk ratio and 95% confidence interval are plotted for each study. The size of the square is proportional to the sample size of the study. The pooled risk ratio is denoted by the diamond apex and 95% confidence interval denoted by the diamond width. A pooled risk ratio of more than 1 indicates higher risk with HA. A pooled risk ratio of less than 1 indicates lower risk with HA. Heterogeneity: I2 = 0%, P = 0.80. Publication bias: Egger’s P = 0.55.
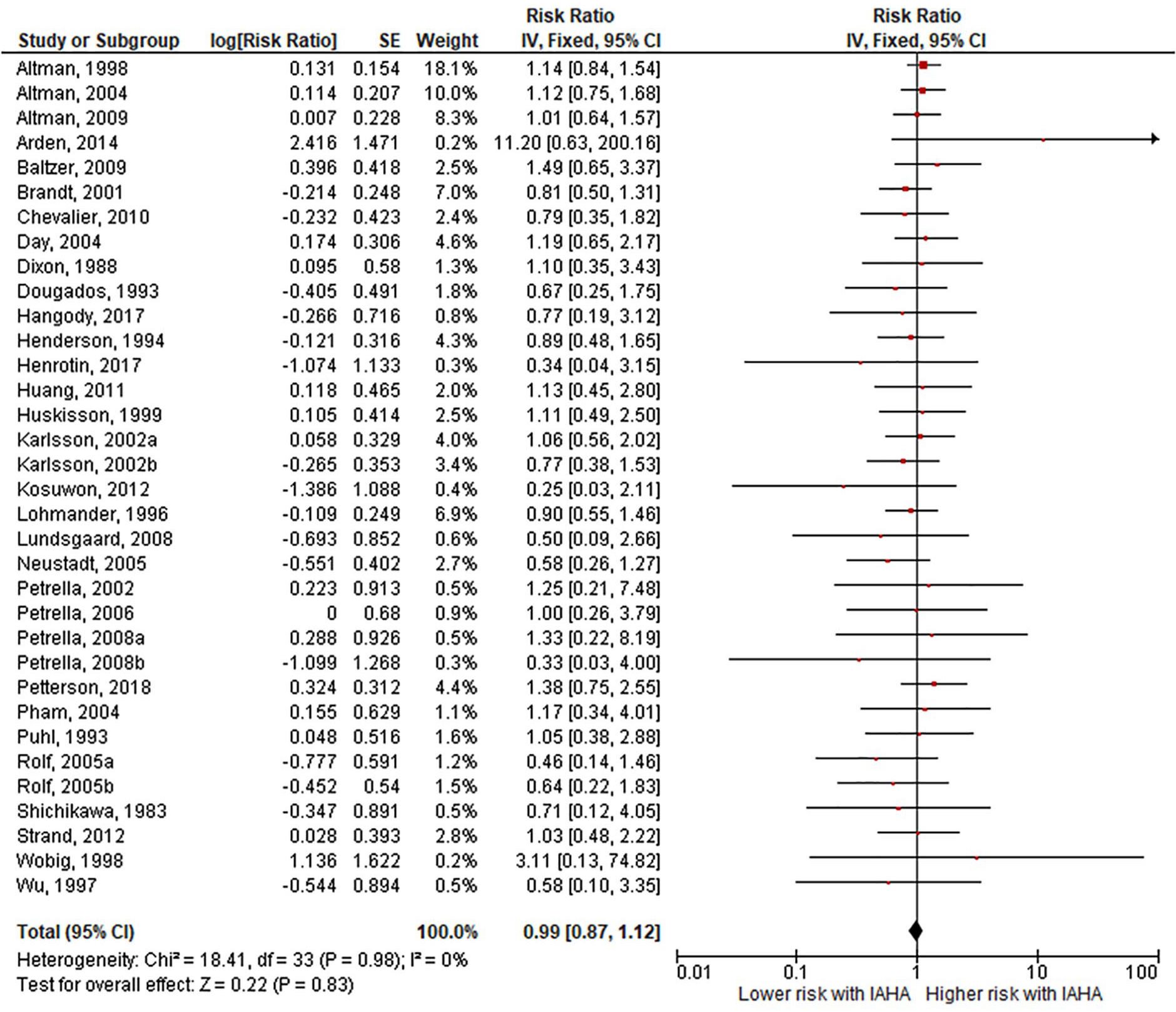
Forest plot of pooled risk ratio for study withdrawal with hyaluronic acid (HA) or saline injections. The risk ratio and 95% confidence interval are plotted for each study. The size of the square is proportional to the sample size of the study. The pooled risk ratio is denoted by the diamond apex and 95% confidence interval denoted by the diamond width. A pooled risk ratio of more than 1 indicates higher risk with HA. A pooled risk ratio of less than 1 indicates lower risk with HA. Heterogeneity: I2 = 0%, P = 0.98. Publication bias: Egger’s P = 0.13.
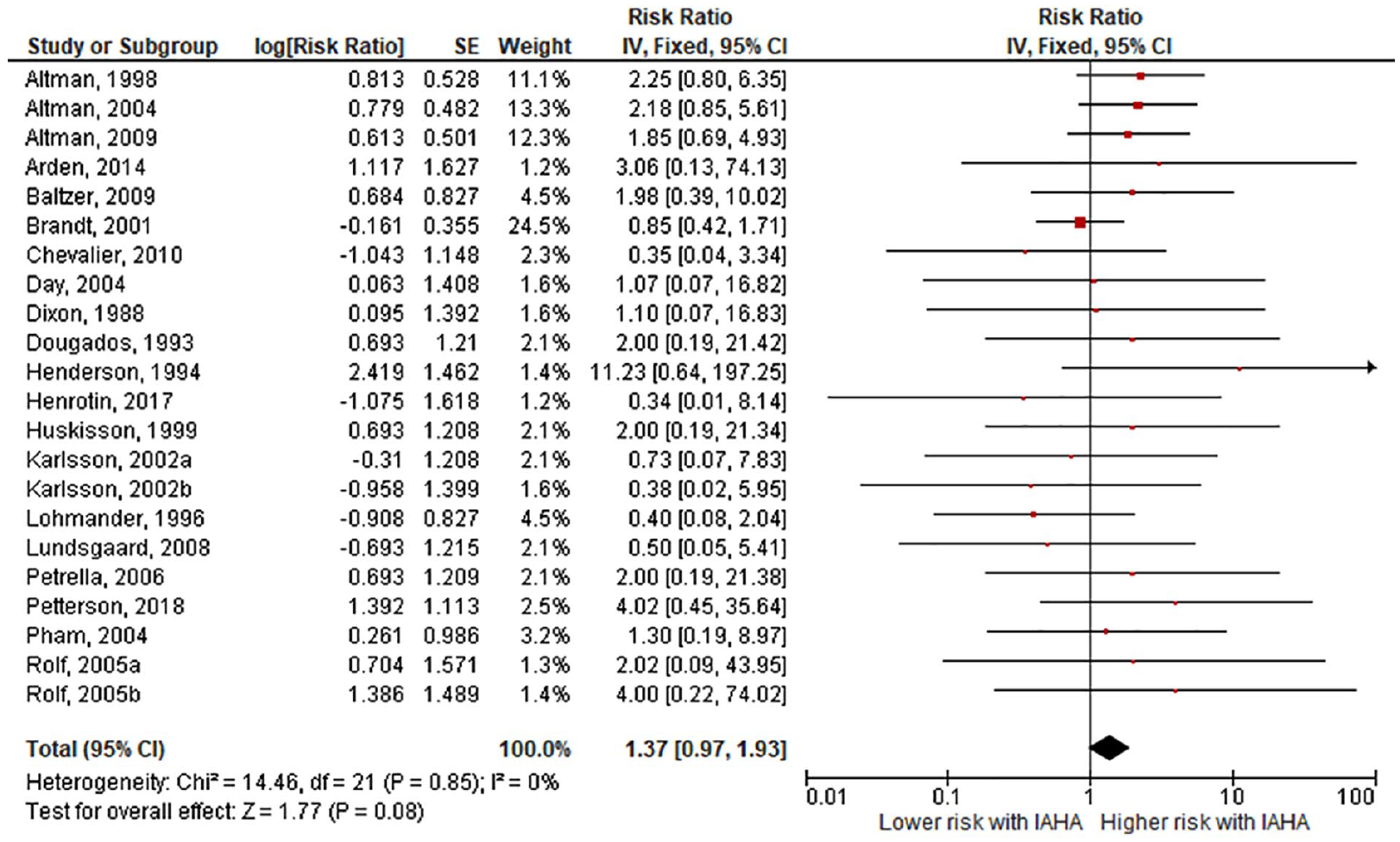
Forest plot of pooled risk ratio for adverse event-related study withdrawal with hyaluronic acid (HA) or saline injections. The risk ratio and 95% confidence interval are plotted for each study. The size of the square is proportional to the sample size of the study. The pooled risk ratio is denoted by the diamond apex and 95% confidence interval denoted by the diamond width. A pooled risk ratio of more than 1 indicates higher risk with HA. A pooled risk ratio of less than 1 indicates lower risk with HA. Heterogeneity: I2 = 0%, P = 0.85. Publication bias: Egger’s P = 0.87.
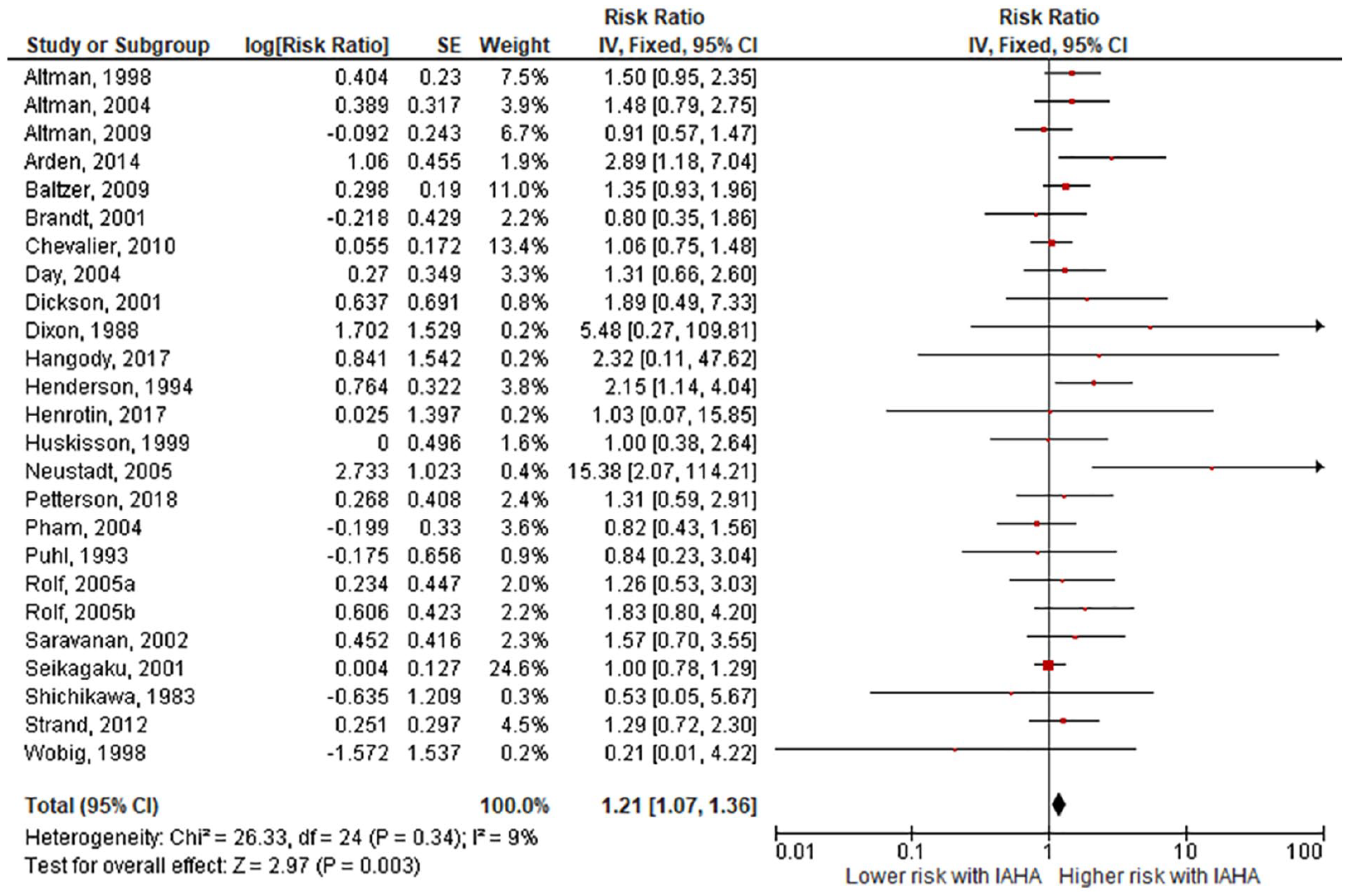
Forest plot of pooled risk ratio for local adverse events with hyaluronic acid (HA) or saline injections. The risk ratio and 95% confidence interval are plotted for each study. The size of the square is proportional to the sample size of the study. The pooled risk ratio is denoted by the diamond apex and 95% confidence interval denoted by the diamond width. A pooled risk ratio of more than 1 indicates higher risk with HA. A pooled risk ratio of less than 1 indicates lower risk with HA. Heterogeneity: I2 = 9%, P = 0.34. Publication bias: Egger’s P = 0.12.
Subgroup and Sensitivity Analyses
In subgroup analyses, no study-level factor influenced the risk of any safety outcome. The associations of these factors on the risk of local AEs are provided in Table 3 . Subgroup analysis results of other safety outcomes were not reported since no heterogeneity was observed for the other safety outcomes (all I2 = 0%). In the first sensitivity analysis, study conclusions were upheld in most scenarios when the meta-analysis was recalculated after removing one study at a time. The only safety outcome in which conclusions were influenced by individual studies was AE-related withdrawals in which one-at-a-time removal of 20 (91%) studies did not alter conclusions, but removal of Brandt et al. 26 or Lohmander et al. 27 led to the conclusion of increased risk of AE-related withdrawal with IAHA injections (Supplemental Table 3). In the second sensitivity analysis, results of each safety outcome were largely identical when comparing fixed-effect and random effects meta-analysis models owing to low heterogeneity among studies (Supplemental Table 4).
Subgroup Analysis of Study-Level Factors on the Risk of Local Adverse Events.
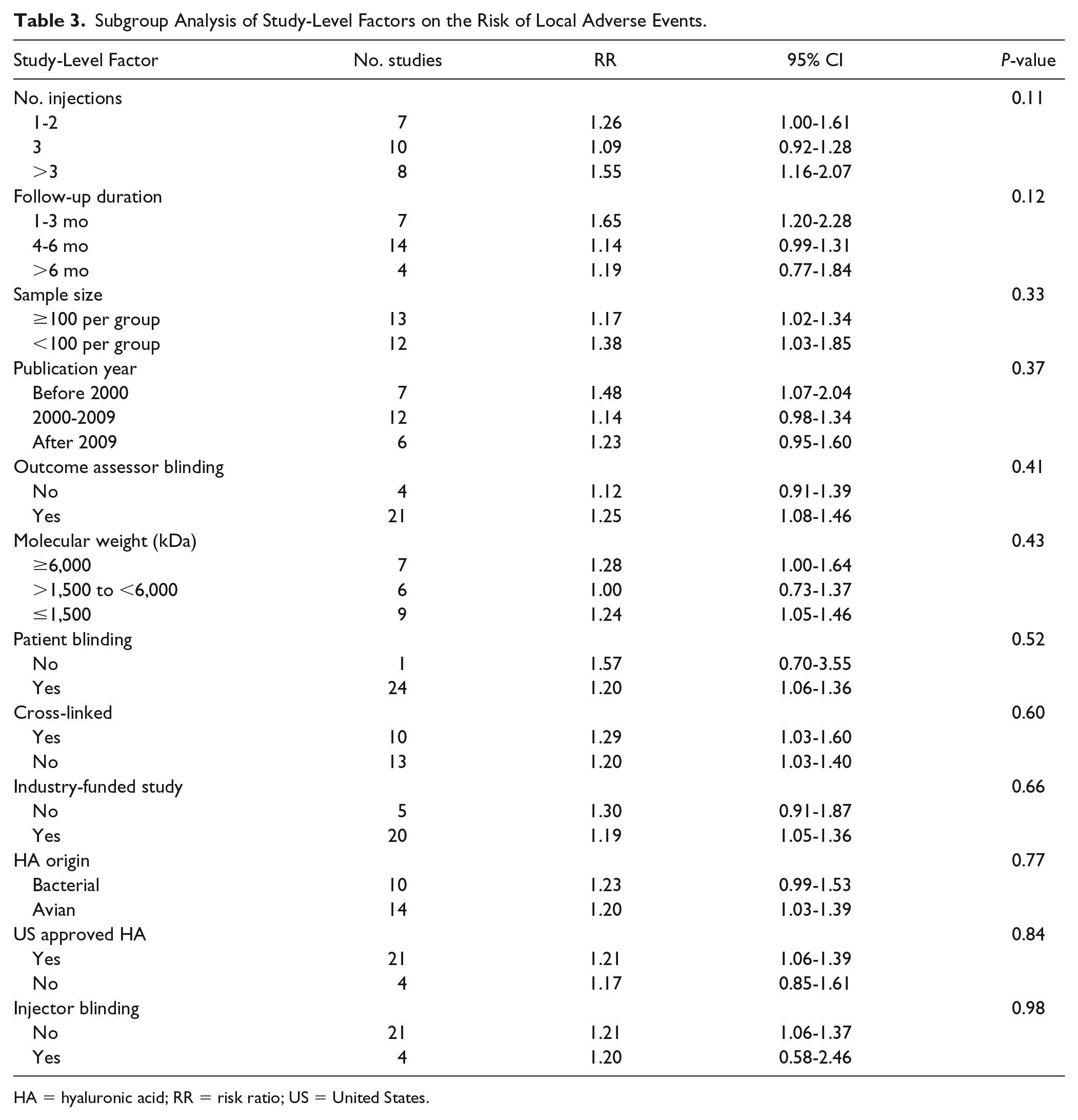
HA = hyaluronic acid; RR = risk ratio; US = United States.
Discussion
IAHA injections are generally perceived as safe for use in patients with symptomatic knee OA. This conclusion was corroborated in the current meta-analysis where we confirmed that IAHA was associated with an increase in local nonserious AEs compared with saline injections (14.5% vs. 11.7%), but with no differences between treatment groups in the risk of AEs, SAEs, study withdrawals, and AE-related study withdrawals. These conclusions were robust in subgroup analyses and were not influenced by heterogeneity or publication bias.
Results of this meta-analysis confirmed that the risk of SAEs was comparable in patients receiving IAHA or saline. This finding agrees with a previous meta-analysis in which the risk of SAEs was no different with IAHA versus saline controls (RR = 1.24, P = 0.32). 16 However, this is in contrast to the results of Rutjes et al. 15 who concluded that IAHA increased the risk of SAEs (RR = 1.41, P = 0.04). In the current study, the risk of experiencing an SAE following IAHA was low, occurring in 1.8% of patients. Furthermore, as was shown in the meta-analysis of Rutjes et al., 15 none of the SAEs were related to IAHA but instead to unrelated conditions. Overall, it appears that SAEs following IAHA are rare, occur with a similar frequency as with IA saline, and are unrelated to the treatment itself.
We determined that IAHA increased the risk of nonserious, transient local reactions compared with IA saline. The most common local reactions were injection site pain, arthralgia, joint swelling, and joint effusion, which subsided within 2 to 3 days in most instances. Despite this increased risk in local reactions, IAHA was well tolerated with the percentage of patient withdrawals overall (12.3% vs. 12.7%) and withdrawals due to an AE (2.7% vs. 2.1%) comparably low in both IAHA and IA Saline groups. It was interesting that no factors related to study design or HA product characteristics influenced safety results, but this was not particularly surprising since there was little heterogeneity among studies.
Given this evidence supporting the safety of IAHA in relation to IA saline, it would be interesting for future studies to compare the safety of IAHA to active therapies recommended for treatment of symptomatic knee OA. In a clinical practice guideline for treatment of knee OA, nonsteroidal anti-inflammatory drugs (NSAIDS) were the only nonsurgical knee OA treatment recommended by the American Academy of Orthopaedic Surgeons (AAOS). 28 Yet, knee OA pain is chronic in nature and NSAIDS are not indicated for chronic use because of safety concerns. Furthermore, selective and nonselective oral NSAIDs increase the risk of serious cardiovascular events and should be used with caution in patients with high cardiovascular risk. 29 Finally, drug fact labels specify that “NSAIDs can increase the chance of a heart attack or stroke, either of which can lead to death.” 30 Given that cardiovascular disease is highly prevalent in patients with OA, 31 there appears to be a disconnect in societal guidelines regarding the safety profile of recommended therapies for knee OA. Rigorous safety evaluations of IAHA versus oral NSAIDs would help clarify the comparative risk profile of these therapies in patients with knee OA.
Strengths of this meta-analysis were inclusion of only RCTs utilizing saline controls and robust study conclusions that were not influenced by heterogeneity among studies, publication bias, or analysis specifications. There were also several limitations of this meta-analysis that warrant additional discussion. First, patients treated with oral therapies or injection of active products were excluded from the control group. While safety comparisons to these other therapies are certainly warranted, inclusion of all potential therapies in a common control group would introduce significant biases that would complicate data interpretation. Second, in accordance with best evidence practices and following the methodology used by the AAOS in their systematic review of IAHA, 32 we excluded studies with small sample sizes. While it is possible that important safety events may be missed using this methodology, exclusion of small studies reduces bias risk since they tend to report greater treatment benefits than large trials and often suffer from lower methodological quality. 19 Finally, the effect of repeat IAHA cycles on safety outcomes could not be evaluated in this review, but has previously been reported to be safe with no reported SAEs in a previous review. 33
Conclusions
IAHA was shown to be safe for use in patients with symptomatic knee OA. Compared with IA saline, IAHA was associated with an increased risk of nonserious and transient local reactions. There was no evidence to suggest any additional safety risks of IAHA.
Supplemental Material
Supplement – Supplemental material for Safety of Intra-Articular Hyaluronic Acid for Knee Osteoarthritis: Systematic Review and Meta-Analysis of Randomized Trials Involving More than 8,000 Patients
Supplemental material, Supplement for Safety of Intra-Articular Hyaluronic Acid for Knee Osteoarthritis: Systematic Review and Meta-Analysis of Randomized Trials Involving More than 8,000 Patients by Larry E. Miller, Samir Bhattacharyya, William R. Parrish, Michael Fredericson, Brad Bisson and Roy D. Altman in Cartilage
Footnotes
Acknowledgments and Funding
We thank David Fay, PhD, for assistance with literature review and data extraction. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: DePuy Synthes provided funding for this study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: L. Miller has an affiliation with DePuy Synthes that is related to this study and with OsteoArthritis Centers of America that is not related to this study. S. Bhattacharyya is an employee of DePuy Synthes. W. Parrish is an employee of DePuy Synthes. M. Fredericson has an affiliation with Ipsen that is not related to this study. B. Bisson is an employee of DePuy Synthes. R. Altman has affiliations with GlaxoSmithKline, Novartis, Pfizer, Sanofi, Sorrento, and Theralogix that are not related to this study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
