Abstract
Keywords
Introduction
Various cartilage repair techniques are used to treat knee symptoms related to cartilage lesions. Autologous chondrocyte implantation (ACI) aims to heal a cartilage lesion with repair tissue that resembles normal hyaline cartilage.1,2 In this technique, the implanted chondrocytes proliferate and gradually fill the lesion with repair tissue. 3 This tissue is assumed to gradually organize and achieve structurally and biomechanically similar properties as native cartilage. According to animal studies in mongrel dogs and rabbits, repair tissue maturation seems to be a slow process, lasting at least 1 year after surgery.4-7 In humans, the maturation process after ACI has been assumed to last even longer.8,9 In clinical trials, repair tissue quality has been investigated as an outcome measure at a specific time point after surgery. However, such measurements in time series are limited and, therefore, the duration and chronological details of the maturation process are unknown.1,10-15
Indentation testing indicates the biomechanical properties of tissue and is known to reflect the histological structure of the tissue.1,12,16 Immediately after chondrocyte implantation, the stiffness of the repair area is unmeasurable, and as the new tissue is formed the biomechanical properties of the repair tissue gradually improve. In repair tissue, higher stiffness values are associated with more hyaline-like repair.1,12 The mechanical stiffness of native cartilage decreases as its degeneration advances.16-18 Therefore, the biomechanical measurement provides a nondestructive approach to assess repair tissue growth and quality and also indirectly reflects the histological structure of the tissue. However, at present only limited information is available on the biomechanical properties of repair tissue after cartilage repair with ACI.1,8,12
More knowledge regarding the repair tissue maturation process is needed to understand the biology of cartilage repair, to estimate the optimal time to evaluate the repair tissue biomechanical and structural outcome, and to design optimal postoperative rehabilitation. The current study sought to investigate the progress and duration of repair tissue maturation after ACI by using arthroscopic indentation measurements.
Materials and Methods
Patients
In this study, we included patients who underwent a biomechanical stiffness measurement of repair tissue in a second-look arthroscopy after ACI of the knee. The patients were selected from 115 patients treated consecutively between 1997 and 2008 in Jyväskylä Central Hospital, Finland. The patients were offered a second-look arthroscopy with biomechanical measurements for scientific purpose between 1- to 5-year follow-up. Of the patients, 31 were willing to participate in the follow-up arthroscopies. For these patients, 38 arthroscopies were performed due to scientific initiative. Of these arthroscopies, 23 were performed for asymptomatic patients and 15 for symptomatic patients. Additionally, the biomechanical measurements were offered for the patients whenever they returned for arthroscopic surgery due to clinical symptoms (21 arthroscopies). Thereby, 59 second-look arthroscopies with biomechanical measurements of the repair tissue were performed for 40 patients. These patients were included in this study. Thirty patients had full-thickness cartilage lesions (International Cartilage Repair Society [ICRS] grade 3 to 4) and 10 had an osteochondritis dissecans (OCD) defect (
Table 1
). Measured lesions were on the medial femoral condyle (
Patient Clinical Characteristics.
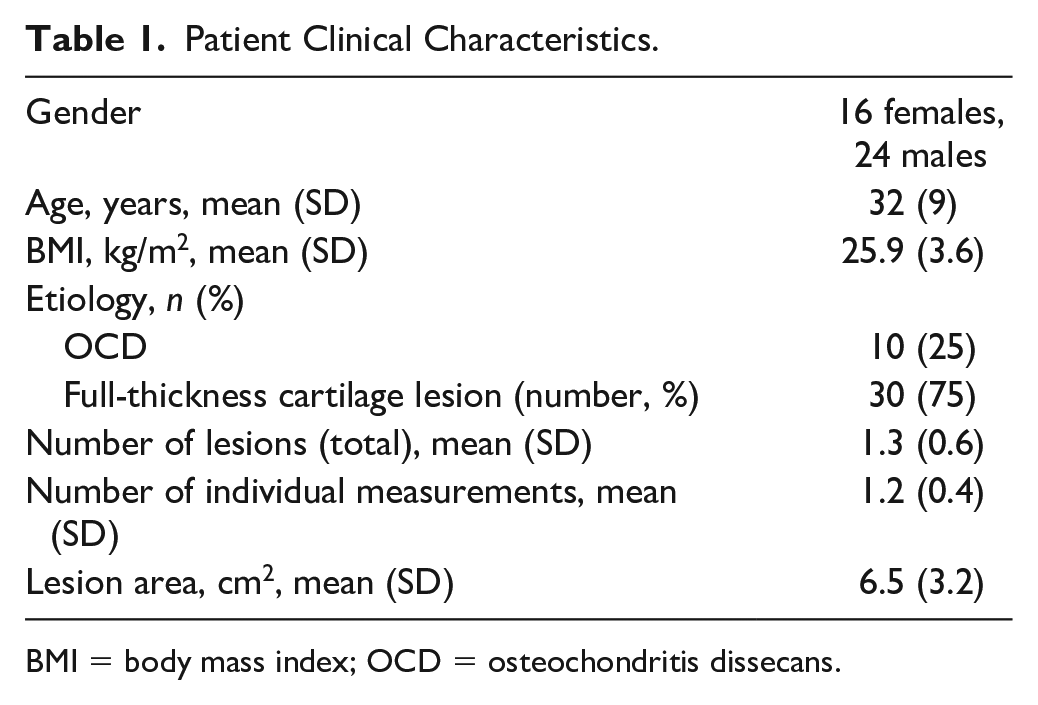
BMI = body mass index; OCD = osteochondritis dissecans.
Cartilage Repair
Cartilage repair surgery was performed according to the originally published ACI technique. 2 For eligible patients, a cartilage biopsy was first acquired during a knee arthroscopy. The biopsy was shipped to the Cell Matrix Laboratory at the Cell Matrix Laboratory at the Sahlgrenska University Hospital in Gothenburg, Sweden for further processing. At the laboratory, the chondrocytes were separated and cultured. An autologous chondrocyte suspension was produced and shipped to the Jyväskylä Central Hospital on the day of the definitive cartilage repair. In this procedure, the cartilage lesion was exposed through an arthrotomy of the knee. The damaged cartilage was removed. A periosteum patch removed from the proximal tibia was sutured with absorbable sutures to cover the lesion. The seam was secured watertight with fibrin glue (Tisseel, Baxter, Switzerland). The autologous chondrocyte suspension was injected under the periosteum to fill the lesion. Finally, the seam was closed with sutures and fibrin glue. The patients followed a standardized rehabilitation protocol. The protocol included restricted weightbearing for 8 to 12 weeks. Immediate training of knee movement was encouraged and, if deemed necessary, supported with a continuous passive motion machine during the hospital stay. Patients systematically visited a physiotherapist during the first 12 weeks; thereafter the visits to the physiotherapist were arranged according to the individual needs of the patient. Return to high-impact activities was recommended 1 year after surgery.
Biomechanical Evaluation
During a second-look arthroscopy, the orthopedic surgeon measured the stiffness of repair tissue and adjacent intact cartilage with an arthroscopic indentation probe (Artscan Inc, Helsinki, Finland). A detailed description of the arthroscopic instrument and the
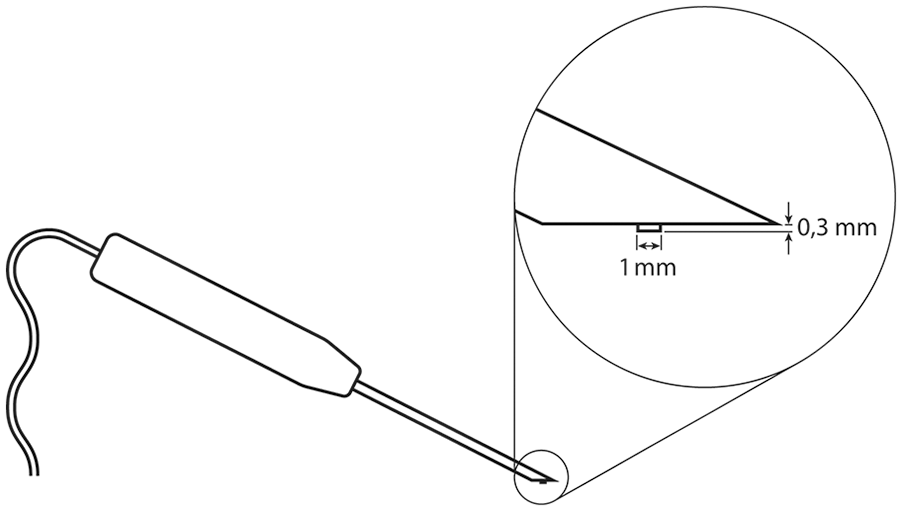
Schematic drawing of the indenter device.
The biomechanical properties of native cartilage are unique and variable in different areas of the knee. 20 They also vary between individuals. 20 Cartilage repair aims to restore native tissue-like biomechanical properties in the knee. Therefore, in addition to the lesion, an individual-specific reference value was acquired from the intact cartilage surrounding the measured lesion. The orthopedic surgeon measured the repair tissue and the surrounding intact cartilage with the indentation probe. The average of three repeated measurements was calculated as the representative stiffness of that site. 8 Each repair site was distinguished by a demarcating line that could be faintly but reliably visualized at the margins of all lesions. The measurements were obtained from the center of the repair tissue, and from the macroscopically intact adjacent cartilage. To enable comparisons between individuals and lesions from different joint surfaces, a lesion-specific relative indentation value was calculated as a ratio between the repair tissue and surrounding cartilage stiffness.1,8 All independent relative indentation values were pooled to form a single time series of measurements illustrating the biomechanical progress of tissue maturation (Supplemental table).
Statistical Analysis
The descriptive statistics were presented as means with standard deviations (SDs) or as counts with percentages. We used random-effects regression model for longitudinal data to analyze relative indentation stiffness values over time. Curvilinear relationships between indentation and follow-up time were derived from random-effects regression model including quadratic term and follow-up time. Correlation coefficients were calculated by the Pearson method. The normality of variables was evaluated with the Shapiro-Wilk
Results
The curvilinear correlation between relative stiffness values and the follow-up time was 0.31 (95% CI 0.07-0.52),
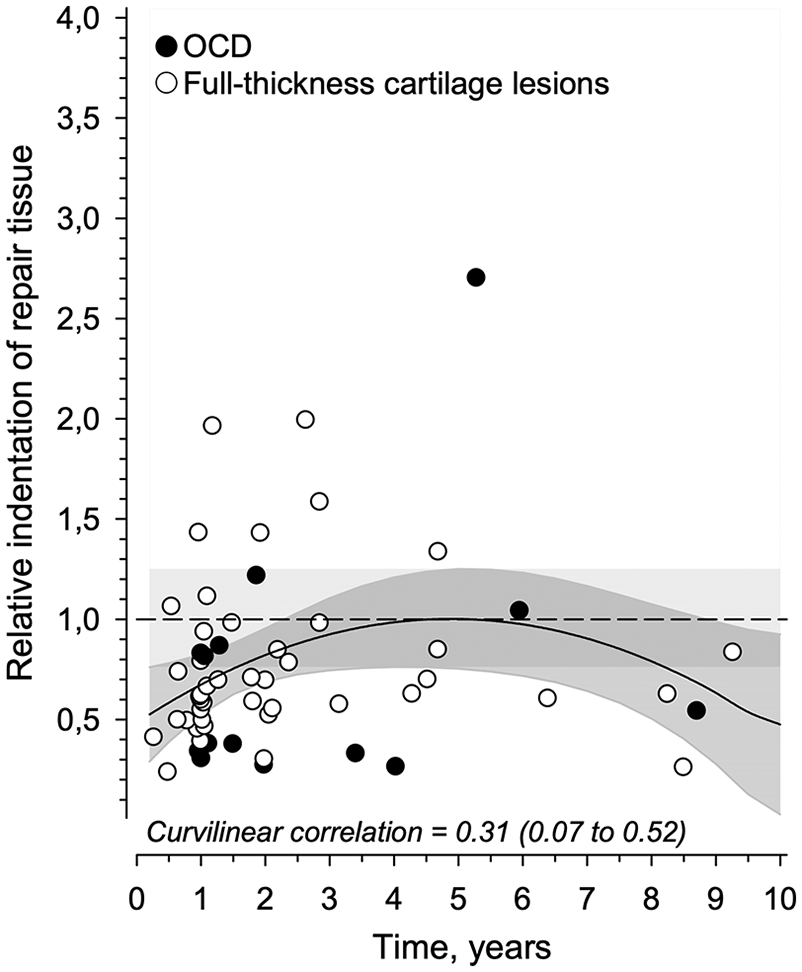
Relative indentation stiffness values of individual biomechanical measurements (
Discussion
Our study demonstrated that the biomechanical stiffness of the repair tissue gradually increases for several years, approaching the properties of native cartilage at approximately 5 years after ACI. However, after that the estimate of biomechanical progression becomes uncertain due to low number of measurements. Additionally, our study showed high variation in the biomechanical quality of the repair tissue between individual lesions. These findings describe a timeline of the maturation of repair tissue after ACI. Importantly, the repair tissue seems to mature for a longer duration than previously described.
The first 2 years of our results are consistent with the histological repair process observed in animal studies after chondrocyte implantation-based techniques.5-7 In humans, histological samples after ACI show a chronological increase of type II collagen content in repair tissue, indicating gradual repair tissue maturation toward hyaline-like cartilage. 15 These observations, together with mathematical models of cartilage regeneration, propose that repair tissue matures up to 3 years postoperatively.8,9 However, our results suggest that the repair tissue maturation process may be even longer. Since animal studies show continuous maturation for at least 2 years after cartilage repair also with techniques other than ACI,4-7,21 it is possible that prolonged remodeling follows not only ACI but also various cell therapies and bone marrow stimulation–based techniques. This implies that the repair tissue is immature during the first postoperative years. Therefore, the most optimal time to assess the quality of the repair tissue would be at least 2 to 3 years after surgery.
Our study shows how repair tissue quality varies substantially throughout the follow-up, suggesting an inconsistent outcome of ACI. Consistent with previous reports, the repair tissue biomechanics were equal to intact cartilage in some lesions, while others were filled with significantly softer or harder repair tissue.1,12 The equal biomechanics of repair tissue and intact cartilage suggest that some lesions repaired with hyaline-like tissue, as has been observed in histological analyses following ACI. 15 In humans, neither ACI nor bone marrow stimulation techniques seem to lead consistently to hyaline-like repair. Biopsies 1 to 2 years after cartilage repair show in only some patients hyaline cartilage (in 10%-48%) and in most patients fibrocartilage (20%-64%).1,3,10,11,22-24 Concordantly, our results suggest that ACI is capable of producing hyaline-like repair in some individuals, but on average repair tissue remains softer than native cartilage and with substantial interindividual variation. From a clinical perspective, the highly variable and slowly progressing biomechanical outcome might produce varying clinical outcomes between individuals. A clinician encountering an unfavorable repair response should carefully bear in mind the possible ongoing maturation of the repair tissue before performing a reoperation.
Methodologically, biomechanical testing has advantages compared to the more commonly used histological assessment.15,25-29 As a nondestructive method, biomechanical testing can be repeated without damaging tissues. The reproducibility of measurements has been shown to be 95% and the precision of the device 11%.
19
The arthroscopic biomechanical probe has been shown to be accurate for scientific use.16,30 Biomechanical testing with repeated measurements from different locations of the potentially heterogenous repair tissue provides, in theory, a more representative overview of the tissue than a single histological biopsy. Our analysis acknowledged the interindividual and knee compartment-dependent variation in stiffness values of the normal cartilage.
20
Therefore, the use of relative stiffness enables a comparison between independent lesions as has been introduced previously.1,8 The design of the arthroscopic indentation instrument minimizes uncertainty related to indentation: measurements are made instantaneously, the indenter tip elevates only 0.3 mm from the reference plate, and the indenter tip is small in diameter (
In addition to arthroscopic indentation, several other methods are available for monitoring repair tissue quality over time. Arthroscopy enables visual evaluation of repair tissue. Thereby, repair quality may be assessed with semi-quantitative scores according to tissue fill, structure, integration to surrounding cartilage, and feel on probing.39-41 Furthermore, arthroscopy provides possibility to measure repair tissue with techniques that require direct contact, such as biopsies for histological analysis, biomechanical measurements, and intra-articular ultrasound imaging. Histology provides an accurate description of repair tissue structure that may be assessed with validated scores.
26
However, only a limited proportion of repair tissue may be analyzed with biopsies. Typically, studies use a single biopsy obtained once at a certain follow-up time, limiting evaluation of repair progression over time.11,42 Arthroscopic biomechanical measurements were designed for less invasive assessment of cartilage and repair tissue. Since 1999 indentation measurements have been used for arthroscopic
The major limitation of our study is the lack of a histological control. Furthermore, the clinical importance of repair tissue biomechanics remains open due to mismatching clinical follow-up data. The repair tissue biomechanics and clinical outcome were measured at different follow-up times. Additionally, unsuccessful repairs might be overrepresented in our study, especially later during the follow-up. The preoperative knee symptoms at the time of second-look arthroscopy were more frequent by time. After 5 years, only a relatively small number of measurements were performed, and all in symptomatic knees.
In conclusion, our study describes a biomechanical maturation process of cartilage repair that may continue even longer than expected. A substantial increase in repair tissue stiffness proceeds for the first 2 years postoperatively, and minor progression proceeds for even longer thus indicating that repair tissue may remodel for several years after ACI. In some lesions, the biomechanical result was equivalent to native cartilage, suggesting hyaline-type repair. The marked variation in biomechanical results indicates substantial inconsistency in the structural outcome following ACI.
Supplemental Material
Revised_Supplemental_table – Supplemental material for Biomechanical Changes of Repair Tissue after Autologous Chondrocyte Implantation at Long-Term Follow-Up
Supplemental material, Revised_Supplemental_table for Biomechanical Changes of Repair Tissue after Autologous Chondrocyte Implantation at Long-Term Follow-Up by Teemu Paatela, Anna Vasara, Heikki Nurmi, Hannu Kautiainen, Jukka S. Jurvelin and Ilkka Kiviranta in CARTILAGE
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was financially supported by the Department of Orthopaedics and Traumatology of the Helsinki University Hospital.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study protocol was approved by the ethical review board of Jyväskylä Central Hospital, Finland (ID 6/2002).
Informed Consent
All participating patients in the study provided signed informed consent.
Trial Registration
Not applicable.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
