Abstract
Objective
Patients undergoing articular cartilage paste grafting have been shown in studies to have significant improvement in pain and function in long-term follow-ups. We hypothesized that ex vivo impacting of osteochondral autografts results in higher chondrocyte matrix production versus intact osteochondral autograft plugs.
Design
This institutional review board–approved study characterizes the effects of impacting osteochondral plugs harvested from the intercondylar notch of 16 patients into a paste, leaving one graft intact as a control. Cell viability/proliferation, collagen type I/II, SOX-9, and aggrecan gene expression via qRT-PCR (quantitative reverse transcription-polymerase chain reaction) were analyzed at 24 and 48 hours. Matrix production and cell morphology were evaluated using histology.
Results
Paste samples from patients (mean age 39.7) with moderate (19%) to severe (81%) cartilage lesions displayed 34% and 80% greater cell proliferation compared to plugs at 24 and 48 hours post processing, respectively (P = 0.015 and P = 0.021). qRT-PCR analysis yielded a significant (P = 0.000) increase of aggrecan, SOX-9, collagen type I and II at both 24 and 48 hours. Histological examination displayed cell division throughout paste samples, with accumulation of aggrecan around multiple chondrocyte lacunae.
Conclusions
Paste graft preparation resulted in increased mobility of chondrocytes by matrix disruption without loss of cell viability. The impaction procedure stimulated chondrocyte proliferation resulting in a cellular response to reestablish native extracellular matrix. Analysis of gene expression supports a regenerative process of cartilage tissue formation and contradicts long-held beliefs that impaction trauma leads to immediate cell death. This mechanism of action translates into clinical benefit for patients with moderate to severe cartilage damage.
Introduction
Osteoarthritis is a debilitating disease that occurs as a result of damage to the articular cartilage, resulting in pain and loss of function. Articular cartilage defects have been reported in 60% of patients undergoing knee arthroscopy. 1 Moderate to severe cartilage lesions are described as Outerbridge grade III and IV, respectively, usually at the time of arthroscopic or magnetic resonance imaging evaluation. The annual incidence for cartilage repair surgery is 90 surgeries per 10,000 patients, and increasing by 5% per year. 2 The ultimate goal of cartilage repair is to restore articular cartilage surfaces that match the biomechanical and biochemical properties of normal hyaline cartilage and to prevent the progression of focal cartilage injury to end-stage arthritis. 3 When nonoperative management fails, cartilage treatment strategies that are palliative (chondroplasty and debridement) and reparative (drilling and microfracture, autologous chondrocyte implantation, articular cartilage paste grafting, osteochondral autograft transplantation, or osteochondral allograft transplantation) can be implemented. 2
Restorative techniques focus on stimulating the growth of new cartilage or replacing damaged areas with intact plugs of autologous or allograft cartilage and bone. Articular cartilage paste grafting is a minimally invasive joint preserving cartilage resurfacing technique developed in 1991 by the senior author to repair chondral defects in knee joints. 4 The paste graft technique utilizes an osteochondral paste of autologous articular cartilage, bone marrow, and progenitor cells in combination with a morselized (completely fractured) defect bed. The combined paste of articular cartilage and bone is thought to augment the progenitor and stem cell supply from vascularized subchondral marrow access, and may present the necessary cellular signals and conductive matrix to produce an appropriate repair tissue. Current literature is inconclusive on the optimal treatment strategy for chondral lesions.2,5
Articular cartilage paste grafting is a biologic technique that uses impacted autologous chondral fragments and bone to repair the articular surface. 6 Findings reported by Xing et al. using a rabbit model support the superiority of paste implantation (articular cartilage paste grafting) 4 over the microfracture technique for cartilage regeneration through a triad of macroscopic, histological, and biochemical assessments. 7
Although paste grafting has shown long-term clinical promise in patients4,6,8 and animal models7,9-12 with isolated and severe osteochondral lesions, no in vitro biochemical or cellular characterization of this tissue has been performed. Understanding the viability and proliferative activity of the paste matrix as used in humans will provide insight into new techniques of cartilage resurfacing. The primary objective of this study was to characterize early biochemical and cellular events after impacting the osteochondral graft into a paste. We hypothesize that ex vivo impacted osteochondral autografts show higher chondrocyte and matrix production than intact osteochondral autograft plugs.
Methods
Patient Selection, Research Ethics, and Patient Consent
Consecutive patients treated with articular cartilage paste graft procedure provided written consent to participate in an institutional review board-approved prospective outcomes study, which enabled the researchers to collect and store clinical and patient-reported outcome measures in a clinical database. A subset of 20 patients treated between April 2016 and March 2017 that fit the inclusion and exclusion criteria provided additional written consent to participate in a prospective institutional review board-approved articular cartilage paste graft characterization study. Inclusion criteria were skeletally mature patients as evidenced on X-ray and over the age of 18. Patients were excluded if they had inflammatory arthritis, latent infection in the knee, or suffered from any chronic condition that may affect their participation.
Patient Demographics
Twenty patients were enrolled in the study with data reported on 16 subjects. The exclusion of 4 subjects was attributed to sample handling and shipment. The mean age was 39.7 ± 13.5 years (range 18-63 years), and 68.8% (11/16) were male. All patients had moderate to severe articular cartilage lesions as documented by intraoperative Outerbridge (OB) Grade III (19%) and OB grade IV (81%), respectively.
Surgical Procedures
The articular cartilage paste graft technique has been described in detail previously.4,6,8 In short, the defect is first debrided and then completely morselized, creating a fresh fracture base through the subchondral bone with extensive use of microfracture awls. Next, one to two 8 × 15-mm plugs of articular cartilage and underlying cancellous bone are harvested from the intercondylar notch using an 8-mm trephine and impacted into a paste with a mallet. The paste is impacted with an obturator through the trephine into the morselized chondral defect and held in place for up to 2 minutes or until blood clotting is observed. For the in vitro study the same harvesting protocol was used and 2 additional osteochondral plugs were harvested, left intact, transferred into sterile cell culture media, and shipped overnight at 4°C for in vitro characterization. Upon receipt at the laboratory, one osteochondral plug was impacted into a paste under normal articular cartilage paste grafting protocol 4 and another left intact as a control.
Analysis of Gene Expression
Paste graft and intact control plug samples were incubated in (modified Hamms Media) and cultured at 37°C, 95% humidity and 5% CO2. 13 The harvesting of plugs was done in a clinical setting. Analysis included cell culture 24- and 48-hour testing, for cell viability and proliferation (Promega Inc., Madison, WI, Cat # B3582). Gene expression of collagen type I (Col-I), collagen type II (Col-II), SOX-9, cartilage oligomeric matrix protein (COMP), aggrecan, and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was analyzed by qRT-PCR (quantitative reverse transcription-polymerase chain reaction) using the LightCycler 480 system (Roche, Basel, Switzerland). 14 Additionally, Safranin-O/fast green histology of cultures samples was evaluated for matrix production and cell morphology.
Analysis was conducted for cell proliferation at 24 and 48 hours postsurgery using the MTS [3-(4,5-dimethylthaizol-2-yl)-5-(3-carboxymethoxy-phenyl)-2-(4-sulfophenyl)-2H-tetrazolum] assay (Promega, Madison, WI). At each time point, samples were removed from their cell culture media, rinsed with 1× HBSS (Corning, Corning, NY), and incubated in 500-µL culture media with 125 µL MTS for 90 minutes. Three 100 µL aliquots of each sample’s incubation medium were transferred to a 96-well plate and their absorbance measured at 492 nm using an elx800 plate reader (Biotek, Winooski, VT).
Gene Expression Characterization
Gene Expression assay by qRT-PCR was performed. RNA was extracted from the samples using RNeasy Mini Kit and QIAShedder (Qiagen, Venlo, Limburg) following the manufacturer’s protocol. Both cartilage plug and paste samples were treated identically with respect to complete isolation of all RNA within the tissue.
Each cartilage sample was homogenized by Gentle MACS Dissociator with M Tubes (Miltenyi Biotec). Cell harvest and RNA extraction/purification was completed with RNeasy Mini Kit (Qiagen) and RNase-Free DNase Set (Qiagen). Complementary DNA (cDNA) was synthesized with iScript cDNA Synthesis Kit (Bio-Rad) and T100 Thermal Cycler (Bio-Rad). Gene expression analysis was performed using the Light Cycler 480 (Roche) with iQTM SYBR Green Supermix (Bio-Rad) and protocol. Fold change of gene expression (mRNA) was normalized to the housekeeping gene, glyceraldehyde 3-phosphate dehydrogenase (GAPDH), and calculated relative to the controls using the Pfaffl method (ΔΔCt method).
To confirm gene expression (mRNA) analysis was completed with the following primers: collagen type-I (COL-I), collagen type-II (COL-II), SOX-9, and aggrecan.
The primer sequences specific for the target gene and the internal control gene GADPH are listed in Table 1 .
Primer Sequences for RT-qPCR: Collagen Type I, Collagen Type II, SOX-9, Aggrecan, Cartilage Oligomeric Matrix Protein (COMP), and Glyceraldehyde 3-phosphate Dehydrogenase (GAPDH).
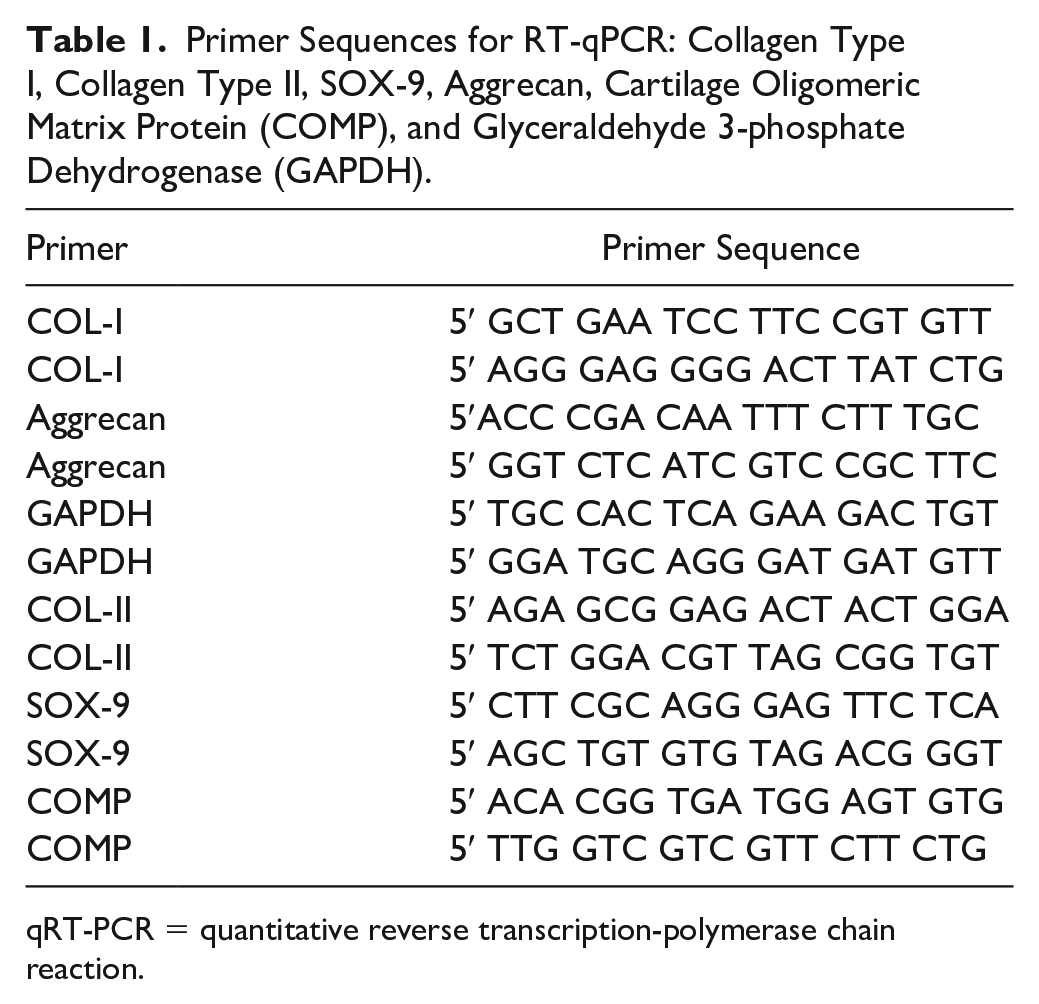
qRT-PCR = quantitative reverse transcription-polymerase chain reaction.
Histology
At 24 and 48 hours postsurgery, samples were fixed in 10% Millonig’s buffered formalin. All samples were decalcified, embedded in paraffin, and cut in 5 µm thickness in a serial fashion. Only plug specimens could be oriented in coronal plane. Sections were stained with Safranin-O/fast green and hematoxylin and eosin. Samples were examined with a bright field microscope and digital images captured. A semiquantitative analysis was performed on plug and paste sections evaluating indices of cell morphology and matrix production.
Data and Statistical Methods
Statistical analysis was performed using IBM-SPSS Statistics 24.0 software (SPSS, and IBM, Chicago, IL). To determine the difference between the viability and proliferation of the plug and paste, a paired Student’s t test (2-tailed) was used at each time point (24 and 48 hours), with background correction and data normalization conventions. One-way ANOVA was used to analyze differences between the target genes (COL-I, COL-II, aggrecan, COMP, and SOX-9). The difference was considered significant when the P value was <0.05.
Results
Reporting on 16 subjects with moderate to severe chondral defects, paste samples displayed 34% and 80% greater cell proliferation compared to plugs at 24 and 48 hours post processing, respectively. qRT-PCR analysis yielded a significant (P = 0.000) increase of aggrecan, SOX-9, collagen type I, and collagen type II at both 24 and 48 hours. Histological examination displayed cell division occurring throughout the paste samples, with deep accumulation of aggrecan content around multiple chondrocytes localized in lacunae.
Analysis of Gene Expression
Reporting on 16 subjects as an intraday group, paste samples displayed 34% (P = 0.015) and 80% (P = 0.021) greater proliferation compared to the plugs at 24 and 48 hours post processing. respectively ( Table 2 ).
Cell Proliferation in Plugs and Paste at 24 and 48 Hours.

The level of collagen type I, collagen type II, aggrecan, COMP, and SOX-9 genes was significantly higher (P ≥ 0.0001) in the paste compared to plug at both 24 and 48 hours; there was no significant difference between these genes when comparing the 24 and 48 hour paste samples ( Fig. 1 ).
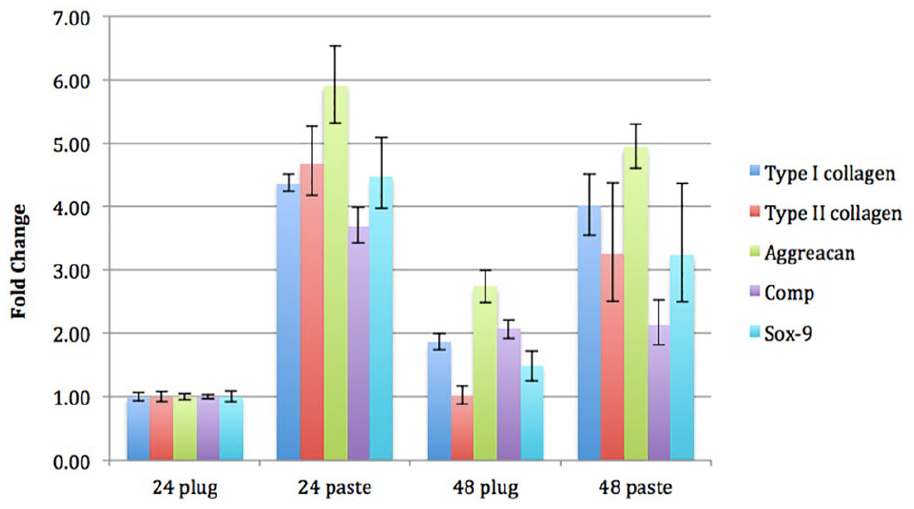
Results of gene expression obtained by qRT-PCR.
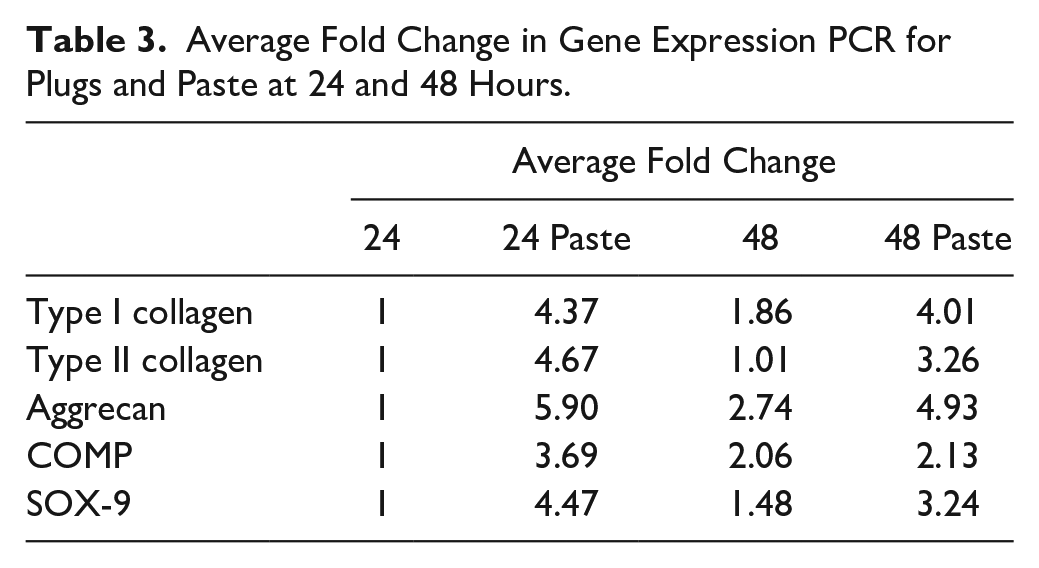
When compared to the 24-hour plug, the expression of aggrecan, COMP, and SOX-9 was significantly higher in the 48-hour plug whereas the expression of Col-I, Col-II, and aggrecan was significantly higher in the 48 hour paste ( Table 3 ).
Average Fold Change in Gene Expression PCR for Plugs and Paste at 24 and 48 Hours.
Histology
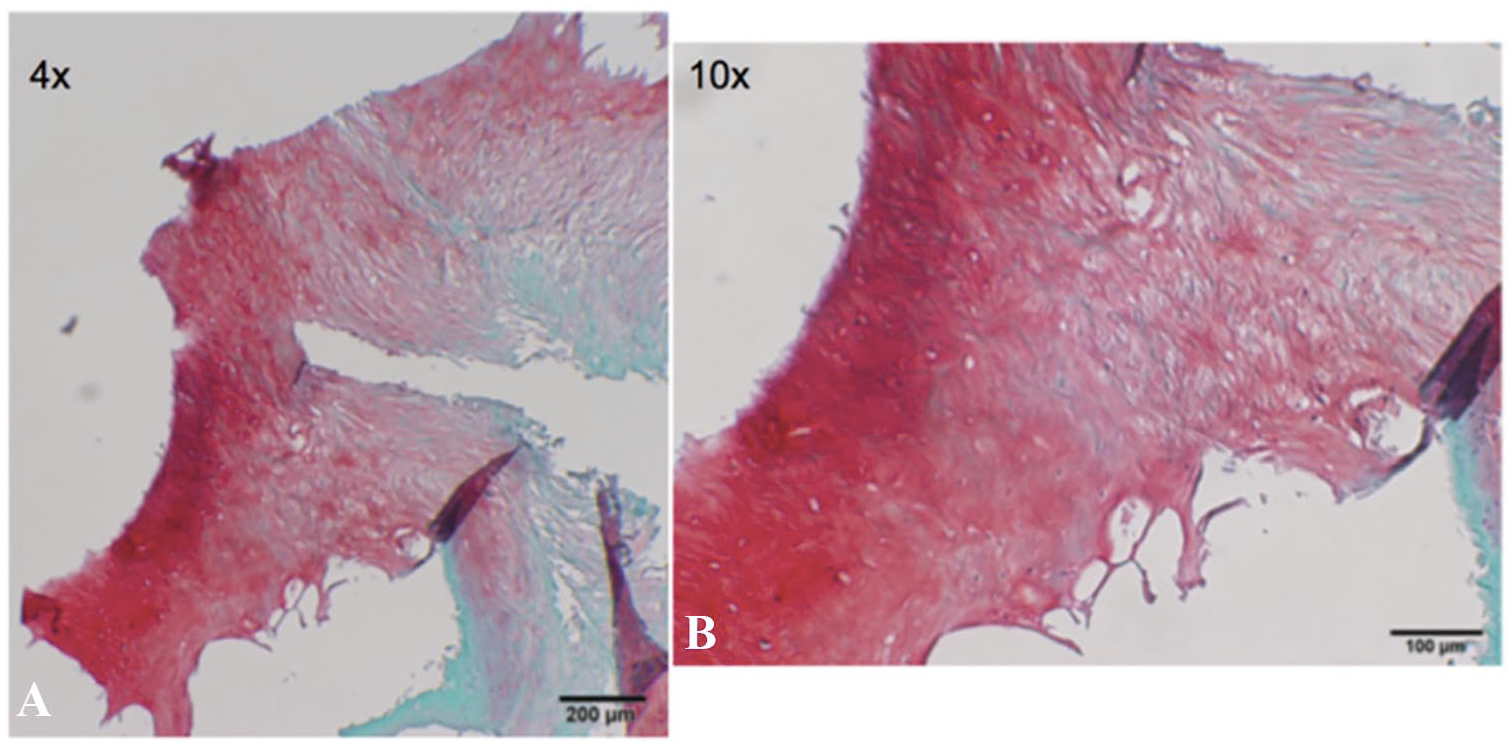
Histological examination shows cell division occurring throughout the paste samples with limited amounts of fibrous tissue and fibrocartilage. A mixture of hyaline-like cartilage tissue is present with deep accumulation of aggrecan content around the chondrocytes localized in lacunae as illustrated in Figure 2A and B , Figure 3A to D , and Figure 4A to C .
(
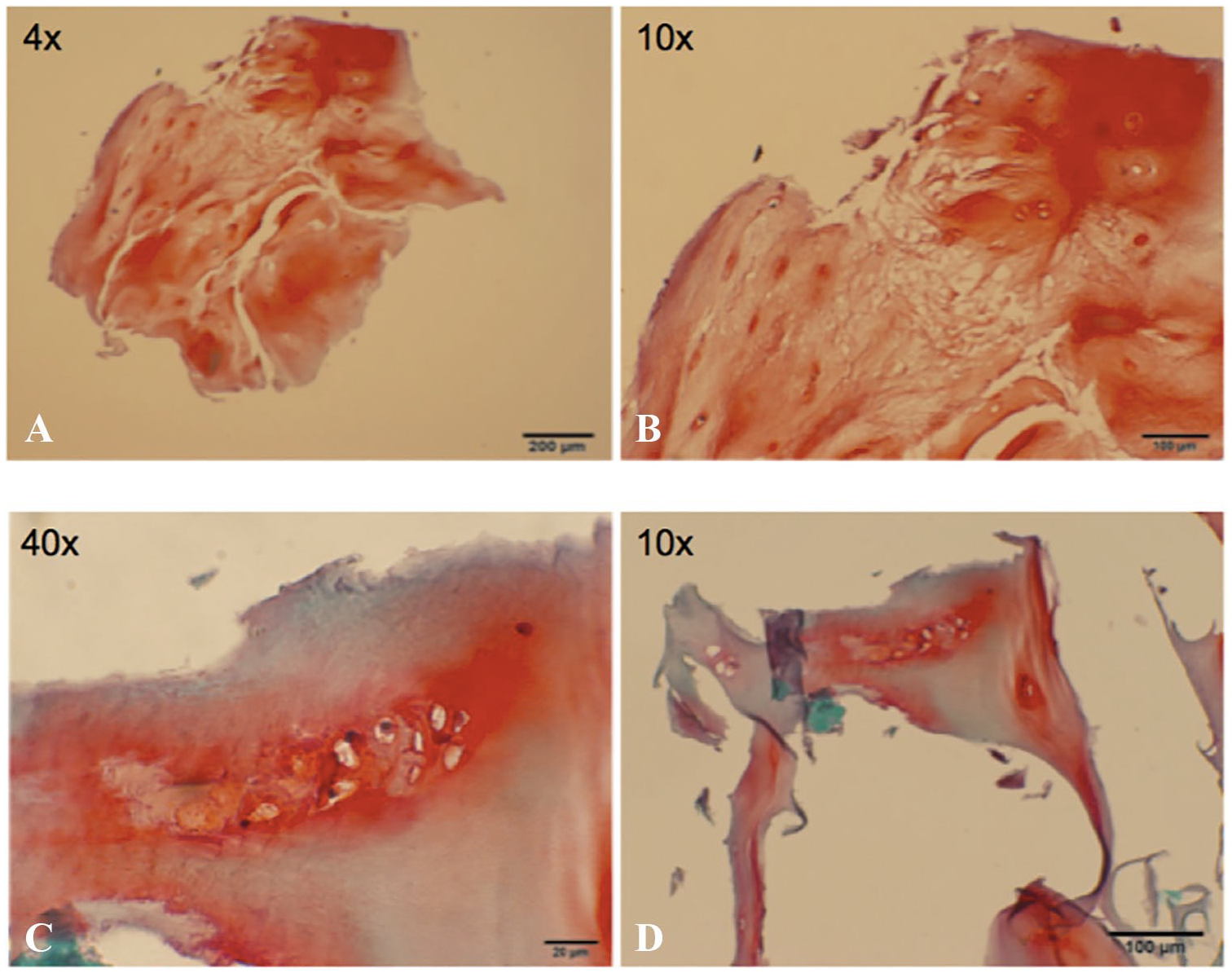
(
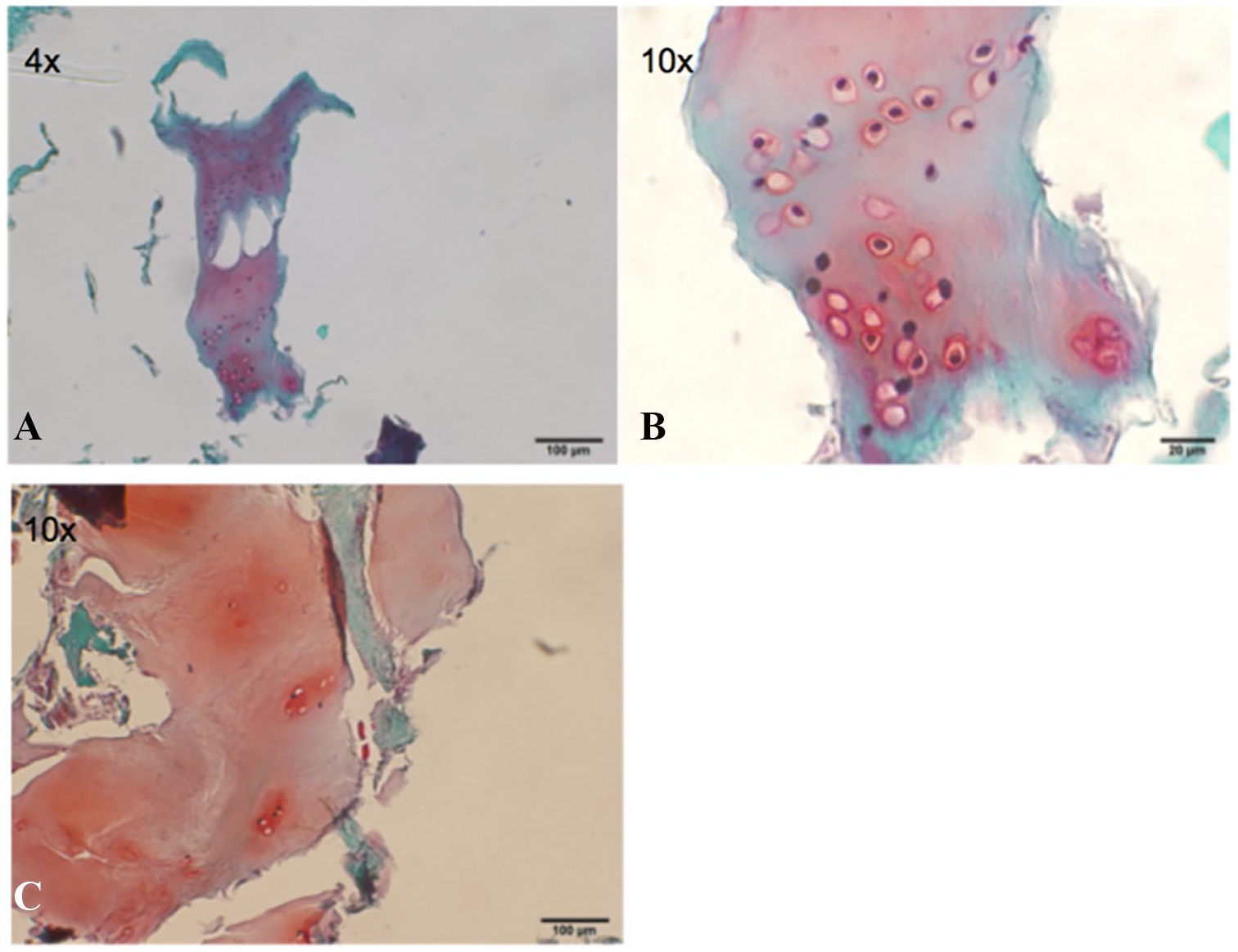
(
Discussion
This study demonstrates that the technique of paste graft preparation results in increased mobility of chondrocytes by virtue of matrix disruption, stimulating the cells to divide, and attempt a cellular response to reestablish native extracellular matrix. In culture and compared to intact cartilage cores, paste graft preparation resulted in increased mobility of chondrocytes by virtue of matrix disruption without loss of cell viability.
The concept of paste grafting was based on 4 principles; first, chondral lesions and arthritic areas of joints are mostly nonviable. Second, the larger the fracture bed the more robust the healing response. Third, bone marrow pluripotent cells will form cartilage if exposed to the signaling factors in articular cartilage matrix and live active chondrocytes, and finally, if motion is applied with non–weight bearing then articular cartilage repair tissue will form. 23
This contrasts with the logic behind particulated cartilage methods called CAIS and DeNovo NT, where autograft cartilage, when mechanically minced into cubes of 1 to 2 mm, was proposed to affect cartilage repair. Chondrocytes in the cartilage pieces were hypothesized to “escape” from the extracellular matrix, migrate, multiply, and form a new hyaline-like cartilage tissue matrix that would integrate with the surrounding host tissue. In addition, unlike cultured chondrocytes that take on a spindle-shaped morphology during culture, the chondrocytes from the impacted cartilage retained the standard chondrocyte spheroid shape.
Microfracture versus Morselization
The rationale for microfracture technique is to establish a clot to provide a suitable environment for a viable population of mesenchymal stem cells (MSCs) from the marrow to differentiate into stable tissues within the lesion. 7 However, microfracture holes leave bone bridges and only slightly if any perforation of the subchondral plate occurs. The regenerated cartilage following microfracture is mostly fibrocartilage, 15 which degenerated quickly resulting in return of symptoms. 16 Complete fracture or morselization of the base of the lesions is believed to stimulate a more robust healing response. The quantity of MSCs, cellular differentiation, and proliferation appears crucial for cartilage repair. 7 The larger the stimulation the more powerful the healing response. Therefore, in the articular cartilage paste graft technique the exposed bone was morselized, not just microfractured, resulting in a complete fracturing of the surface into and through the subchondral plate. 4
Migration of Pluripotent Cells
It was postulated that pluripotent cells migrating from the bone marrow would be more likely to form cartilage if they encountered a paste of articular cartilage extracellular matrix, articular cartilage cells, and bone marrow. Xing et al. compared the paste graft technique to microfracture in a rabbit model and reported that the osteochondral paste contained chondrocytes and MSCs in the cancellous bone at 4 weeks, which had a positive effect on cell proliferation and the development of a cellular matrix and was superior to microfracture alone. 7
Impaction of the Paste
Osteochondral autograft transfer procedures using an osteochondral plug have been demonstrated to relieve pain and provide long-lasting improvement in pain and function; however, harvest of the plugs often results in significant pain and morbidity. 17 Additionally, matching recipient site to graft surface contours can be problematic, leaving gaps in the repaired surface. When an osteochondral autograft transplantation procedure fails, defect base voids or large holes in the surface lead to critical graft failure.18,24 For these reasons the paste graft procedure was devised that relies on the creation of a paste of viable articular cartilage, articular cartilage cells, and bone marrow by impacting harvested osteochondral plugs. Clinical observation of notchplasty harvest sites and subsequent natural restoration of the notch led to the use of intercondylar notch cores as the source of osteochondral autograft. The concern is that impaction of the cartilage may lead to cell death 19 ; however, it is reported that the rate of loading significantly affects the degree of matrix damage, the distribution of dead cells, and the amount of cell death. 20 A study by Lewis et al. reported that when mature bovine articular cartilage on the intact patella was impacted with a flat impactor to 53 MPa in 250 ms, cell viability in the impacted regions distant from visible cracks was no different than the nonimpacted control. 21 Furthermore, the results of this study demonstrate that the level of collagen type I, collagen type II, aggrecan, COMP, and SOX-9 genes was significantly higher in the impacted paste compared to intact plug at both 24 and 48 hours. In addition, the paste samples displayed 34% and 80% greater proliferation compared to the plugs at 24 and 48 hours post processing, respectfully.
In this experiment, the metabolic response of articular chondrocytes to the paste graft preparation technique was compared to chondrocytes maintained in their 3-dimensional milieu within cartilage plugs. Cell proliferation was found to be significantly enhanced in paste graft chondrocytes. It is proposed that cell mechanical disassociation from lacunae stimulates cell division. This stimulation is similar to in vitro enzymatic digestion of the matrix for use in cartilage tissue culture or in vivo cells nests observed in osteoarthritic cartilage. The initial increase observed in the intact plug is most likely a result of the trauma in the tissue from the tissue located adjacent to the periphery of the cylindrical walls of the plug.
QRT-PCR analysis yielded an increased fold change of aggrecan, SOX-9, and collagen type I and type II at both 24 and 48 hours. The significant increase in gene expression for the important phenotypic markers of chondrocytes represents a metabolic attempt to replicate the cells and territorial matrix as a result of the paste graft manipulation. The same profile of gene upregulation can be seen in traditional cartilage cell culture. 22
Histology
The histological results of this ex vivo study demonstrate cell division occurring throughout the paste samples with limited amounts of fibrous tissue and fibrocartilage with a mixture of hyaline-like cartilage tissue present and deep accumulation of aggrecan content around the chondrocytes localized in lacunae. Cell division occurred throughout the paste samples within the osteochondral matrix, without fibrous morphology. A mixture of hyaline-like cartilage tissue is present with deep accumulation of aggrecan content around multiple chondrocytes localized in lacunae. Parallel to the upregulation of both collagen I and II, increase matrix staining was observed when comparing paste to plug samples. In the same manner, hyaline-like cartilage tissue was present with deep accumulation of aggrecan content around the chondrocytes localized in lacunae, indicating a regenerative process of cartilage tissue formation.
Previous studies in a rabbit cartilage defect model utilizing the paste graft technique demonstrated significantly superior defect fill, aggrecan staining, and collagen type II presence in the paste when compared to microfracture. 7 The authors further concluded that the articular cartilage paste graft technique 4 can result in improved quality of reparative tissue with a positive effect on the integration to surrounding cartilage, when compared to microfracture alone. 7 Histological analysis of human clinical cartilage biopsy after previous articular cartilage paste graft demonstrated that 65% of patients presented with strong and consistent evidence of articular cartilage surface regeneration. Of the 65 patients biopsied, 28% of the biopsies had areas of hyaline cartilage that were indistinguishable from normal cartilage. 8
Limitations
This study has a several limitations. First, the sample size may be considered small with data for 16 participants. Second, culture condition time points for analysis were limited to 24 and 48 hours, with no time zero baseline or characterization beyond 48 hours. Third, we did not record the force used to smash the cartilage into a paste; therefore, this might lead to different outcomes if other surgeons replicate the technique. Fourth, the patients in this study all had grade III and IV OB scores indicating the presence of moderate to severe chondral defects; it would be prudent to compare the results in patients with less joint damage to determine if the biochemical environment of more or less severely damage joints affects the chondrocyte behavior. Finally, histology assessment was limited as comparison of particulate to intact cartilage samples cannot be performed quantitatively.
Conclusions
This study identifies the early biochemical and cellular events that occur after impacting osteochondral graft into a paste. Previous studies have identified the scientific evidence for the use of chondral fragments in a one-stage cartilage repair procedure. 25 The technique of paste graft preparation results in increased mobility of chondrocytes by virtue of matrix disruption, stimulating the cells to divide and attempt a cellular response to reestablish their native extracellular matrix. This mechanism of action explains the previously documented articular cartilage regeneration documented in patients with moderate to severe articular cartilage damage treated with the paste graft technique and may form the basis for the clinical benefits documented in these difficult to treat cartilage injuries.
Footnotes
Authors’ Note
This investigation was performed at The Stone Research Foundation, San Francisco, and Feinstein Institute for Medical Research, Manhasset, New York, NY, USA.
Acknowledgment and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by a restricted grant from N. D’Arcy Roche.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
A subset of 20 patients treated between April 2016 and March 2017 that fit the inclusion and exclusion criteria provided additional written consent to participate in a prospective institutional review board-approved articular cartilage paste graft characterization study.
Informed Consent
Patients treated with articular cartilage paste graft procedure provided written consent to participate.
