Abstract
Objective
The classic chondrocyte isolation protocol is a 1-step enzymatic digestion protocol in which cartilage samples are digested in collagenase solution for a single, long period. However, this method usually results in incomplete cartilage dissociation and low chondrocyte quality. In this study, we aimed to develop a rapid, high-efficiency, and flexible chondrocyte isolation protocol for cartilage tissue engineering.
Design
Cartilage tissues harvested from rabbit ear, rib, septum, and articulation were minced and subjected to enzymatic digestion using the classic protocol or the newly developed sequential protocol. In the classic protocol, cartilage fragments were subjected to one 12-hour digestion. In the sequential protocol, cartilage fragments were sequentially subjected to 2-hour first digestion, followed by two 3-hour digestions. The collected cells were then subjected to analyses of cell-yield efficiency, viability, proliferation, phenotype, and cartilage matrix synthesis capacity
Results
Overall, the sequential protocol exhibited higher cell-yield efficiency than the classic protocol for the 4 cartilage types. The cells harvested from the second and third digestions demonstrated higher cell viability, more proliferative activity, a better chondrocyte phenotype, and a higher cartilage-specific matrix synthesis ability than those harvested from the first digestion and after the classic 1-step protocol.
Conclusions
The sequential protocol is a rapid, flexible, high-efficiency chondrocyte isolation protocol for different cartilage tissues. We recommend using this protocol for chondrocyte isolation, and in particular, the cells obtained after the subsequent 3-hour sequential digestions should be used for chondrocyte-based therapy.
Keywords
Introduction
Cartilage is an avascular tissue characterized by a low density of chondrocytes surrounded by an extracellular matrix (ECM). Thus, when damaged, cartilage has a limited repair capacity due to low cellularity, low cell migration to damaged areas, and a lack of support from blood vessels. Reconstruction of cartilage defects caused by degenerative diseases, 1 trauma, tumor exsection, 2 and congenital diseases 3 is a great challenge for and places a socioeconomic burden on health care systems, especially in the context of an active aging population. Although autologous or allogenic native cartilage tissues and synthetic biomaterials have been used in clinical conditions, limitations, such as donor site morbidity, disease transmission, and immune rejection, lead to suboptimal outcomes in cartilage defect reconstruction.4,5
Various cell-based therapies, including autologous chondrocyte implantation (ACI) and cartilage tissue engineering (CTE), have been developed for cartilage defect regeneration.4,6 These techniques involve isolation, expansion, or implantation of chondrocytes on scaffolds. A prerequisite for successful clinical translation of these techniques is maximizing chondrocyte yield number and improving the cell viability, phenotype, and cartilage-specific ECM synthesis capacity from limited donor tissue. Collagenase digestion, the most frequently used method for chondrocyte isolation, gradually dissociates cartilaginous ECM from the outer layer to the inner layer and releases chondrocytes from the matrix. However, different protocols for different cartilage tissue sources vary significantly, and marked variations in user competency are also observed. In general, classic enzymatic digestion is a 1-step protocol in which cartilage samples are digested in collagenase solution for a single, long period (range of 10-22 hours, typically overnight),7-9 and the resulting cell suspension is then collected for chondrocyte isolation. However, the classic protocol usually results in incomplete digestion, which reduces the efficiency of chondrocyte yield and wastes precious tissue samples. It has been reported that the chondrocyte yield with the classic digestion protocol is typically lower than 20%10,11 and that means abundant chondrocytes are entrapped in residual cartilage tissues. Although
Here, we report a rapid, flexible, high-efficiency protocol for digesting different cartilage tissues that can overcome the abovementioned limitations of the classic protocol. We used the new sequential protocol to digest different cartilage types, including auricular, costal, septal, and articular cartilage, and compared them with the classic protocol in terms of cell-yield efficiency, cell viability, cell phenotype, and cartilage-specific ECM-forming capacity in CTE.
Materials and Methods
Cartilage Sample Preparation
Experimental procedures, animal use, and care programs adopted in this study were approved by the Animal Use and Care Committee of our institute. Auricular, costal, septal, and articular cartilage tissues were harvested from New Zealand white rabbits (0.7-1.0 kg,
Cartilage Digestion
A sequential protocol was used for digesting the minced cartilage fragments, and a classic protocol was used as a control in this study. Briefly, the collagenase solution was prepared by dissolving collagenase NB4 (Nordmark Biochemicals, Uetersen, Germany) at a concentration of 1.5 mg/mL (0.12 U/mg). Three grams of minced cartilage fragments were transferred into a 50 mL tube and 30 mL of collagenase solution was added into the tube. The samples were incubated at 37°C. For the sequential protocol, the whole digestion procedure was divided into 3 digestion rounds: the first digestion lasted for 2 hours, and the second and third digestions lasted for 3 hours. For the classic protocol, a single digestion that lasted for 12 hours was applied. For each digestion, the cell suspension was collected for further investigation, and the remaining undigested cartilage was weighed, rinsed with phosphate-buffered saline (PBS), and redigested in fresh collagenase solution (10 mL collagenase solution per milligram of cartilage) in the sequential protocol until the third digestion was completed ( fig. 1A ).
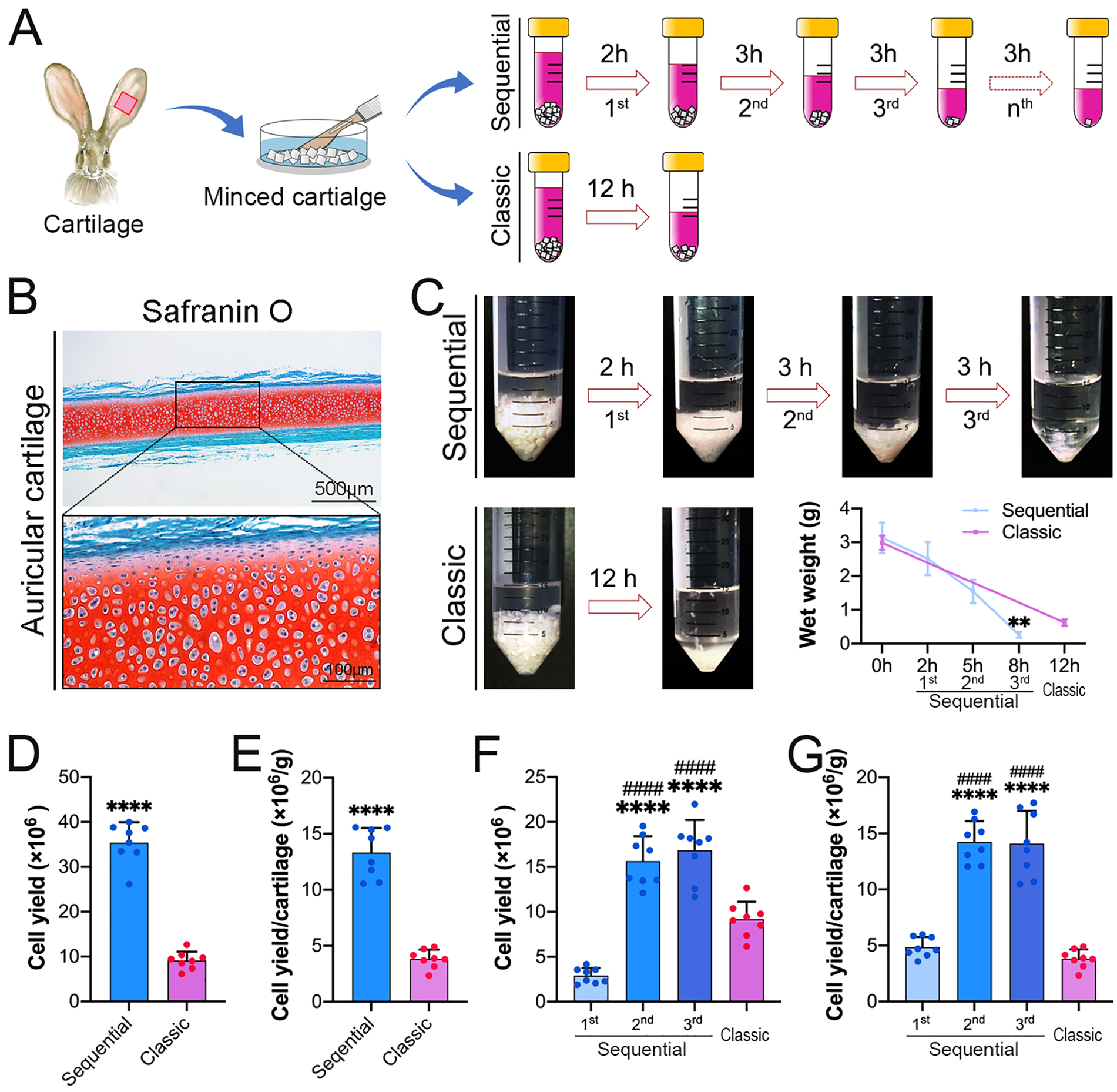
The sequential enzymatic digestion protocol improves the efficiency of cartilage digestion and cell yield.
Chondrocyte Harvest and Culture
After each digestion, the cell suspension was filtered with a 100-μm cell strainer and centrifuged at 300
For determination of cell viability, freshly harvested cells were stained with trypan blue and counted with a hemocytometer. LIVE/DEAD staining (Invitrogen, Carlsbad, USA) was also carried out immediately after digestion.
For cell expansion, the harvested cells were seeded at a density of 2.5 × 105 cells per T-25 flask in complete medium. The media were changed twice per week. Once confluent, the cells were passaged. Passage 2 cells were used in subsequent studies.
Cell Proliferation Assay
Cell Counting Kit-8 (CCK-8; Dojindo, Kumamoto, Japan) was used to measure the proliferation level of the collected cells. The cells were seeded in 96-well plates at a density of 3,000 cells/well. After 1, 2, 3, and 4 days of culture in complete medium, 10 µL of CCK-8 reagent was added for an incubation period of 4 hours. The absorption and reference wavelength were measured at 450 and 620 nm, respectively.
RNA Extraction, cDNA Synthesis, and Real-Time Polymerase Chain Reaction
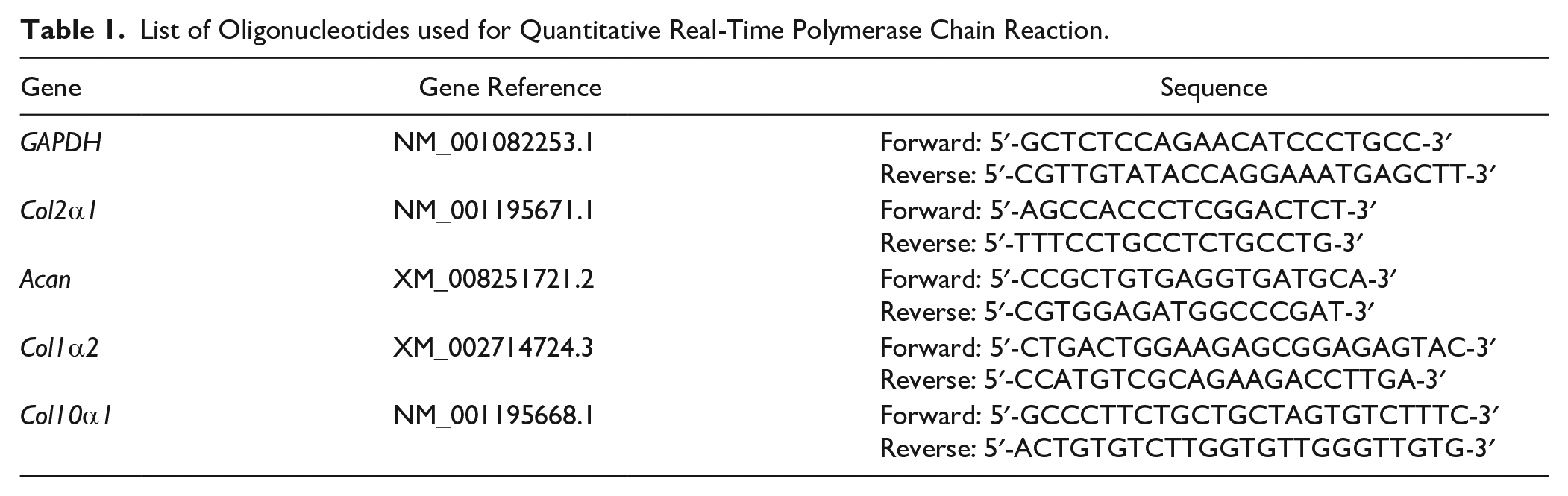
RNA from cells was extracted using a Multisource Total RNA Miniprep Kit (Axygen, New York, USA). The acquired RNA samples were reverse transcribed to cDNA by reverse transcriptase (Takara, Kusatsu, Japan). The gene expressions were quantified with iQ SYBR Green Supermix (Biorad, Hercules, USA). All primers were synthesized by Sangon Biotech (Shanghai, China), and their sequences are presented in the Table 1 .
List of Oligonucleotides used for Quantitative Real-Time Polymerase Chain Reaction.
Immunofluorescence Staining
Primary cells were seeded in a 25-mm confocal dish at a density of 2.5 × 105 cells/dish and cultured in complete medium for 12 hours. The cells were then fixed with 4% paraformaldehyde and permeabilized with 0.1% Triton X-100. The cells were subsequently immunostained with type II collagen (Col II) antibody (Invitrogen) overnight and incubated with secondary antibody (Invitrogen) and phalloidin (Sigma, St.Louis, USA). The samples were covered with antifade reagent (Roche, Basel, Switzerland). The ratio of Col II-positive cells to total cells was determined with ImageJ.
In Vitro Expansion on Scaffolds
For CTE, 7 × 105 passage 2 cells were seeded onto a 4-mm diameter, 1-mm thick Col I-based cylindrical scaffold (Ultrafoam; Davol, Warwick, USA). The seeded scaffolds were then cultured in chondrogenic medium (complete medium supplemented with 10 ng/mL TGF-β3 [transforming growth factor beta 3] [R&D, Minneapolis, USA], 10 µg/mL insulin [Novo Nordisk, Copenhagen, Denmark], and 0.1 mM ascorbic acid 2-phosphate [Sigma]) for 2 weeks ( fig. 6A ). The medium was changed twice per week, and the aspirated medium was collected for subsequent biochemistry assays. After 2 weeks of culture, the constructs were collected and subsequent histology and biochemistry tests were performed.
Histological and Immunohistological Staining
The perichondrium-peeled cartilage tissues and tissue-engineered constructs were fixed in 4% paraformaldehyde and embedded in paraffin. The sections were stained with hematoxylin & eosin (H&E; Solarbio, Beijing, China) and safranin O (Solarbio). For immunohistochemistry staining, the sections were blocked with 5% goat serum and incubated with Col II antibody (Invitrogen). After sequential incubation with a biotinylated secondary antibody and an ABC-alkaline phosphatase complex (Vector, Burlingame, USA), specific staining was revealed by using Vector Red (Vector).
Glycosaminoglycan Quantification
The constructs were digested with proteinase K (Sigma) for 16 hours at 56°C. For glycosaminoglycan (GAG) quantification, the digested constructs and the collected medium supernatant were incubated in 500 µL of dimethylmethylene blue (DMMB; Sigma) solution. Precipitated DMMB-GAG complexes were centrifuged and supernatants were discarded. Complexes were dissolved in decomplexion solution at 60°C, the absorption was measured at 656 nm, and the GAG concentrations were calculated using a standard curve prepared with purified bovine chondroitin sulfate (Macklin, Shanghai, China).
Statistical Analysis
All the above data are presented as the means ± standard deviations. Student
Results
Auricular Cartilage Digestion
H&E staining revealed that 2 layers of overlaying tissue were still observed over the central cartilaginous region ( fig. 1B ). As shown in figure 1C , in the sequential protocol, after the 3 digestions, the wet weight of the residual cartilage tissue was decreased to 0.26 ± 0.09 g on average. While, in the classic protocol, the wet weight of the residual cartilage tissue was decreased to 0.62 ± 0.10 g on average. Although a long time was needed for the classic protocol, the residual cartilage was 2.4 times heavier than that in the sequential protocol ( fig. 1C ). In total, (13.32 ± 2.20) × 106 cells were collected from the sequential protocol, but only (3.84 ± 0.82) × 106 cells with the classic protocol ( fig. 1D ). The cell-yield efficiency of the sequential protocol was approximately 3.5-fold higher than that of the classic protocol ( fig. 1E ). These data suggest that the sequential protocol is a timesaving but high-efficiency protocol for auricular chondrocyte isolation that achieves a high cell yield.
With the sequential protocol, the number of cells yielded from each digestion was (2.91 ± 0.85) × 106, (15.65 ± 2.77) × 106, and (16.87 ± 3.35) × 106 on average. With the classic protocol, the number of cells yielded from each digestion was (9.17 ± 1.96) × 106 on average ( fig. 1F ). After each digestion, the cell-yield efficiency of the second and third digestions was significantly higher than that of the first digestion and the classic protocol ( fig. 1G ). These results indicate that the second and third digestions exhibit higher cell-yield efficiency than the first digestion and the classic protocol.
Cell Viability
Flow cytometry analysis showed that live cells collected from the second and third digestions were (91.81 ± 4.18)% and (93.20 ± 3.79)% on average, respectively, which were significantly higher than that obtained with the first digestion ([82.2 ± 8.41]%) and the classic protocol ([79.84 ± 9.73]%), respectively (
fig. 2A
and
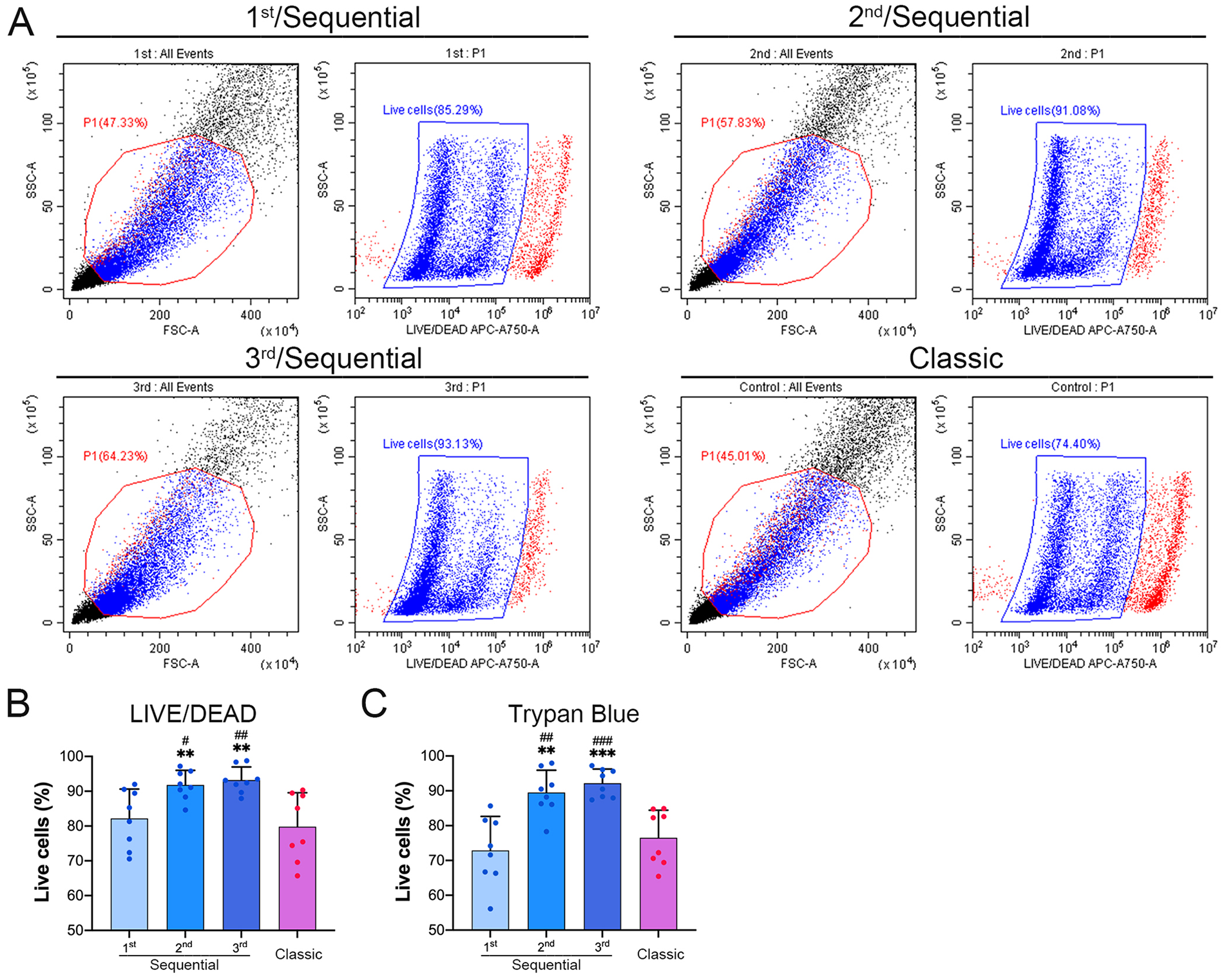
The sequential protocol improves the viability of the collected cells.
Cell Morphology and Col II Protein Expression
As shown in figure 3A , after a 12-hour culture, most of the cells harvested from the first digestion were attached to the dish but still resided in cell aggregates. Few Col II-positive cells were scattered within the cell aggregates. Most of the cells from other digestions were single cells. Among them, Col II-positive cells showed a round or polygonal shape, which is a typical morphology of chondrocytes. Some spindle-shaped Col II-negative cells were also observed. More interestingly, the cells from the third digestion showed a higher percentage of round/polygonal shape and larger spreading area than those from the others. The percentages of Col II-positive cells in the first, second, and third digestions were (6.90 ± 0.80)%, (19.10 ± 1.11)%, and (29.50 ± 3.75)%, respectively, and the percentage in the classic protocol was (11.47 ± 1.88)% ( fig. 3B ). The percentages of Col II-positive cells from the second and third digestions were significantly higher than those from the first digestion and the classic protocol.
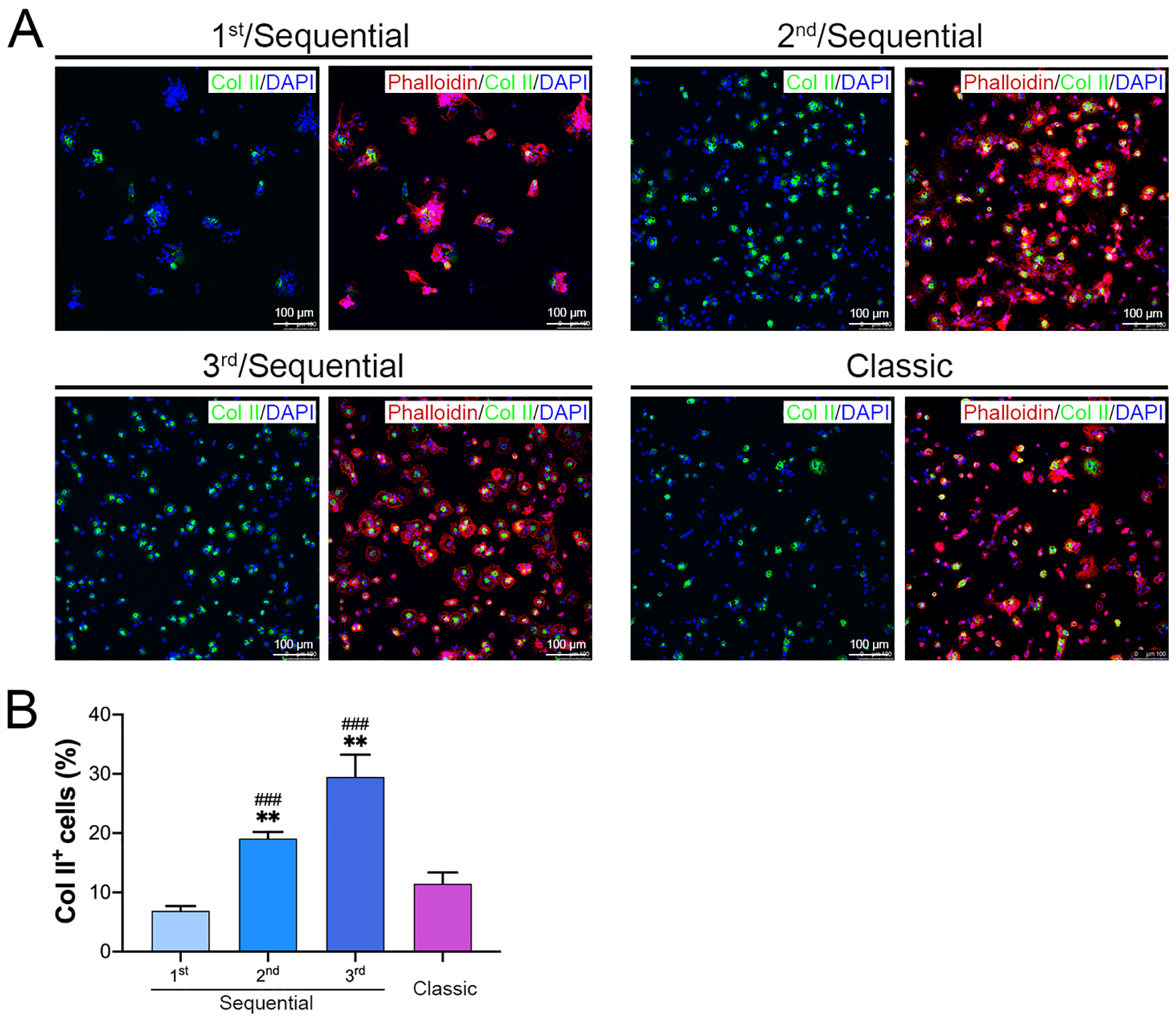
The second and third digestions of the sequential protocol harvested a high percentage of Col II-positive cells.
Cell Proliferation
As shown in figure 4A , most of the cells from each digestion, especially those from the second and third digestions, displayed a uniform morphology with a typical round or polygonal shape at day 3. More spindle-shaped cells were observed in the first digestion. Furthermore, the cells from the first digestion and the classic protocol had a lower cell density ( fig. 4A ), indicating that the cells from the second and third digestions may have higher cellular proliferative activity. The CCK-8 proliferation assay confirmed that the cells from the 3 digestion rounds in the sequential protocol, especially those from the second and third digestions, had comparable but higher proliferative capacities than those obtained with the classic protocol at days 3 and 4 ( fig. 4B ). These data imply that the sequential protocol does not affect the proliferation ability of the cells.
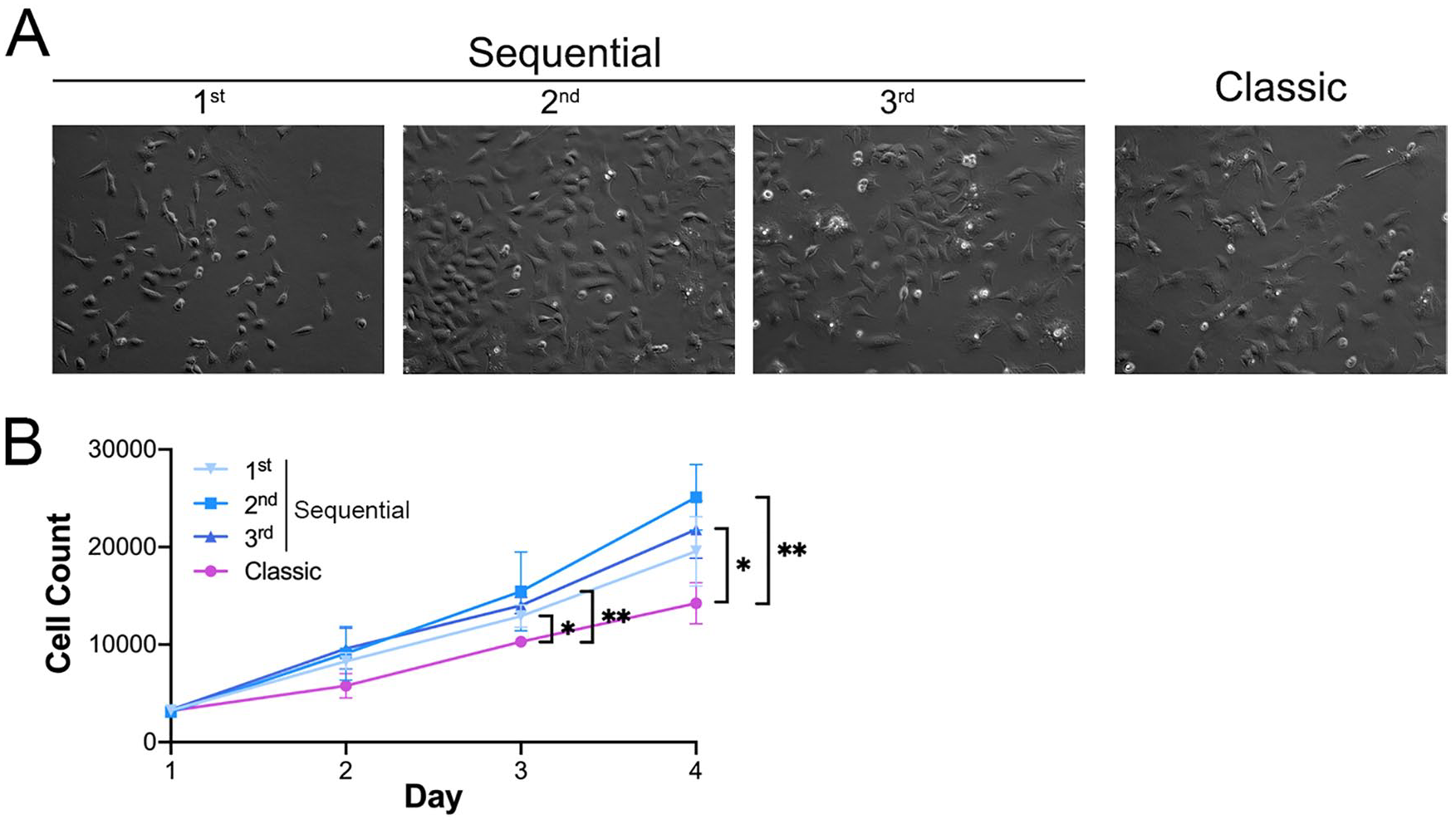
The sequential digestion protocol did not impair cell proliferative activity.
Chondrocyte-Specific Gene Expression
Compared with the primary cells obtained with the classic protocol, the primary cells from the second and third digestions expressed higher levels of
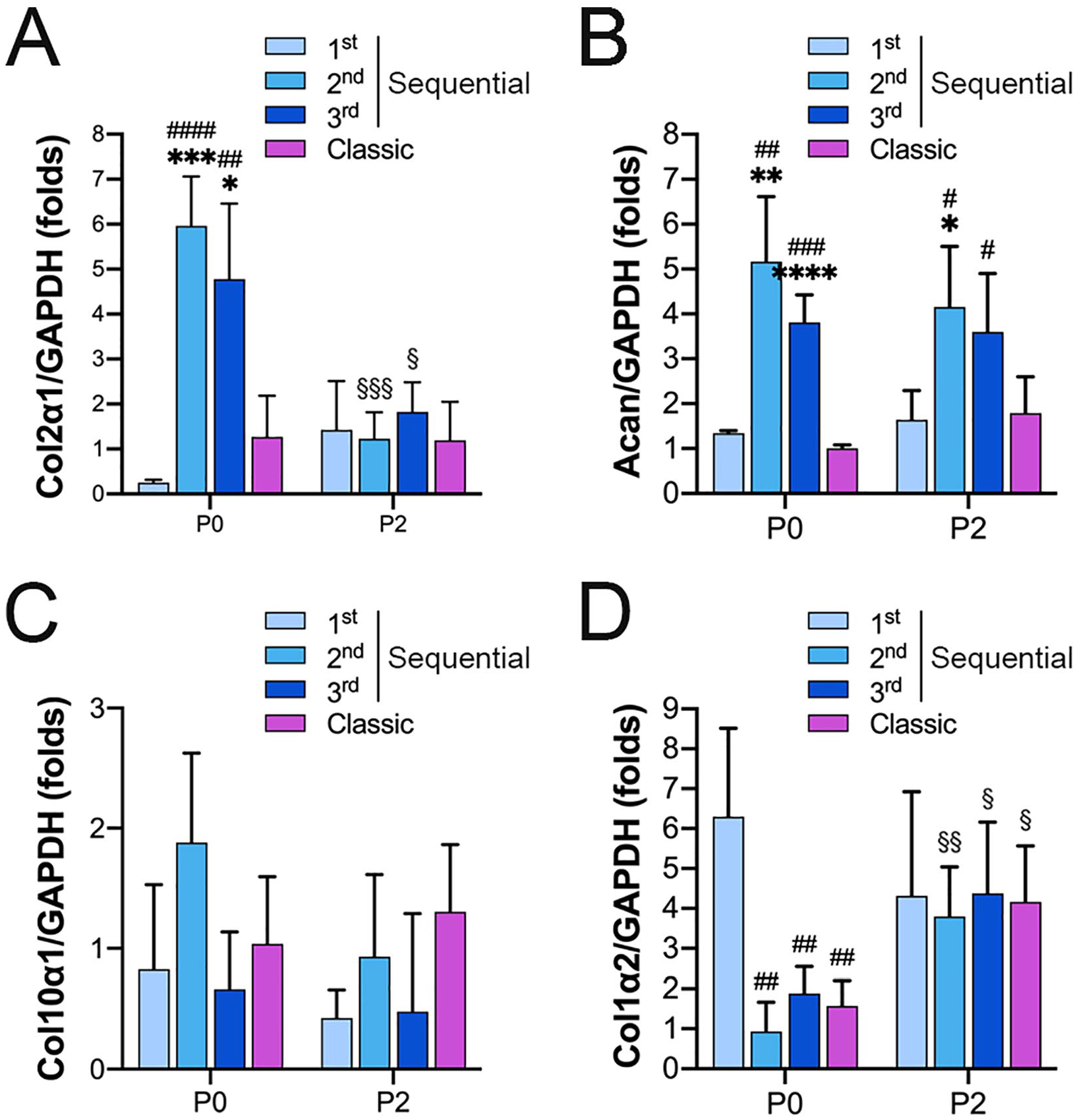
Gene expression analysis of
The expressions of
Application in CTE
After 2 weeks of
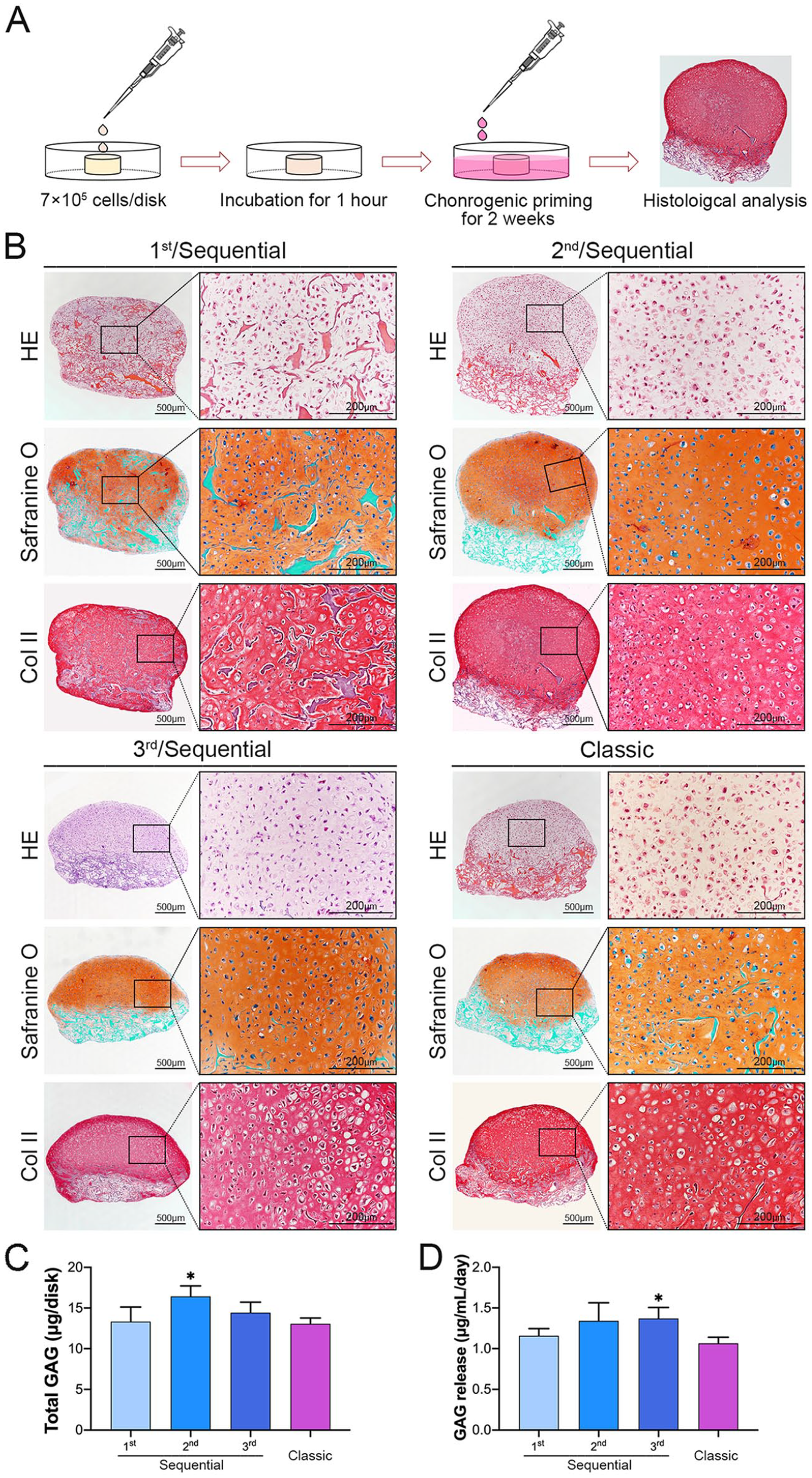
Cartilaginous matrix synthesis capacity of the harvested cells collected in CTE.
GAG quantification results demonstrated that the constructs engineered by the cells from the second digestion contained more GAG than that engineered by the cells obtained with the classic protocol ( fig. 6C ) and the constructs engineered by the cells from the third digestion released more GAG than that engineered by the cells obtained with the classic protocol ( fig. 6D ).
Digestion of Cartilage Tissues from the Rib, Septum, and Articulation
To test the efficiency of the sequential protocol in other cartilage tissue types, costal, septal, and articular cartilage tissue samples were harvested from rabbits for chondrocyte isolation ( fig. 7A ). Safranin O staining revealed that all cartilage tissue samples had a red cartilaginous region and 1 or 2 green overlying tissue regions ( fig. 7B ). The chondrocyte density differed among different cartilage types. The average number of cells in a 1 mm2 area was 800 ± 161 in costal cartilage, 1,025 ± 219 in septal cartilage, and 965 ± 204 in articular cartilage ( fig. 7D ). All cartilage tissues underwent enzymatic digestion via the classic protocol and the sequential protocol. After long-term digestion via the classic protocol, the wet weight of residual cartilage tissues was 1.00 ± 0.07 g in costal cartilage, 0.23 ± 0.06 g in septal cartilage, and 0.58 ± 0.09 g in articular cartilage. After 3 digestions with the sequential protocol, the wet weight of residual cartilage tissues was 0.72 ± 0.08 g in costal cartilage, 0.07 ± 0.03 g in septal cartilage, and 0.42 ± 0.04 g in articular cartilage. Compared with the results obtained with the classic protocol, significantly lighter residual wet weights were found for the 3 different cartilage tissues with the sequential protocol ( fig. 7E ).
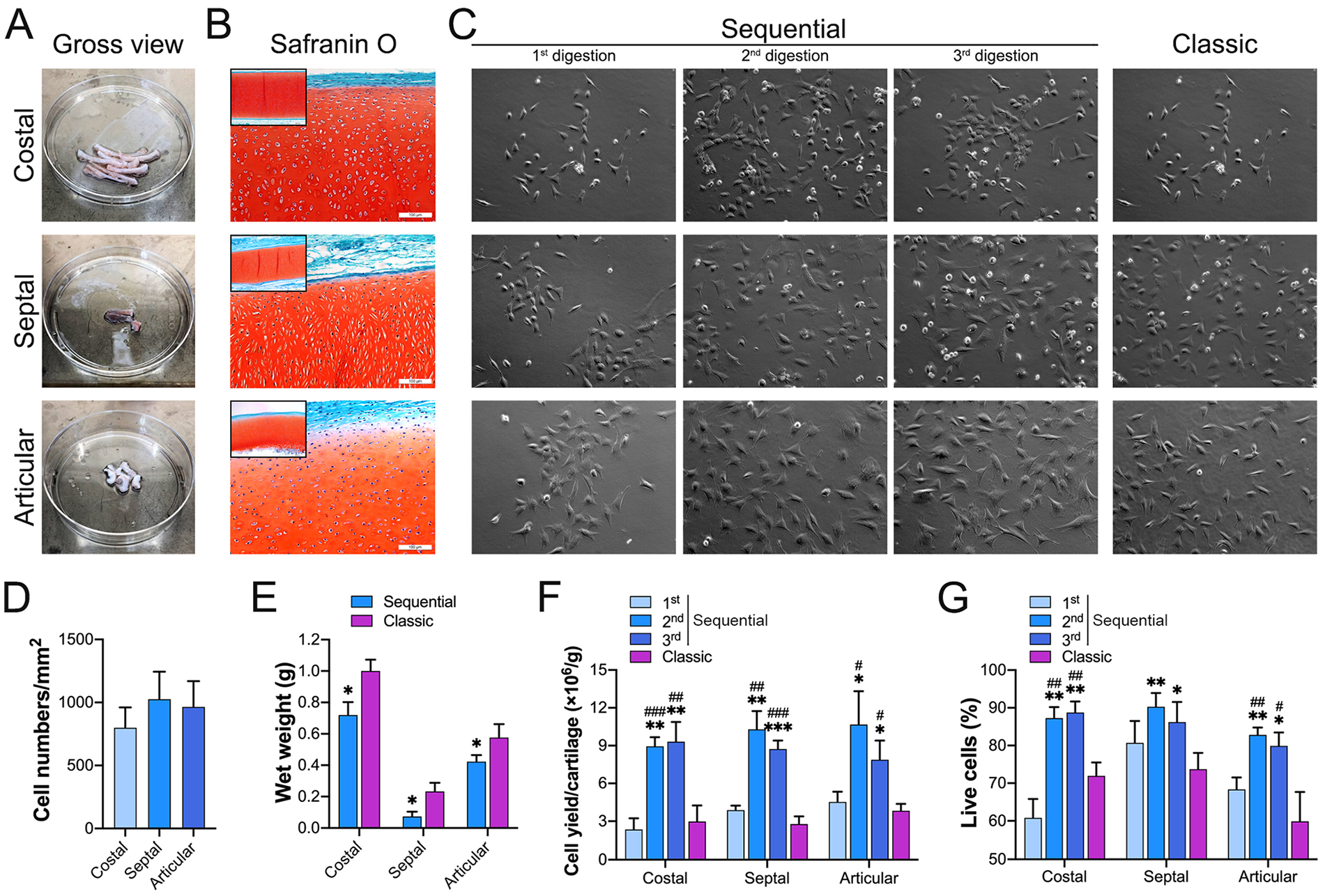
The sequential enzymatic digestion protocol yields isolated high-quality chondrocytes with high efficiency from costal, septal, and articular cartilage.
After a 3-day culture in complete medium, most of the cells from costal, septal, and articular cartilage displayed a round or polygonal shape, and some of them displayed a fibroblastic-like morphology ( fig. 7C ). Similar to the results in auricular cartilage, the second and third digestions of the sequential protocol showed significantly higher cell-yield efficiencies ( fig. 7F ) and significantly increased viable cell ratios ( fig. 7G ) in costal, septal, and articular cartilage tissue.
Discussion
The isolation of abundant viable chondrocytes from cartilage tissue is a necessary and critical step for ACI techniques and CTE. However, the classic digestion protocol, which digests cartilage biopsies via a single long period, usually results in incomplete digestion and impaired cell quality. To improve the quantity and quality of yielded cells, we developed a sequential enzymatic digestion protocol that digests cartilage tissue with sequential short-term digestions. In this study, the results showed that the sequential protocol significantly improved the cell-yield efficiency and cell quality in isolating chondrocytes from auricular, costal, septal, and articular cartilage tissues. Furthermore, the cells harvested from the second and third digestions showed superiority over those harvested with the classic protocol in terms of cell viability, cell proliferation, cell phenotype, and cartilage-specific ECM-forming capacity.
The enzymatic digestion protocol is based on degrading the cartilaginous collagen network, cleaving the core protein of proteoglycans or their connecting element of hyaluronidase, and releasing chondrocytes from the matrix. In this study, collagenase NB4, a widely used collagenase with a balanced mix of collagenolytic and additional enzymatic activities,20,21 was used for chondrocyte dissociation. Incomplete digestion and low cell-yield efficiency are frequently observed in chondrocyte isolation, 9 although the digestion periods are usually prolonged to 10 to 22 hours. The incomplete digestion may be caused by the gradual decrease in collagenolytic activity after a long period of digestion and could be solved by increasing the collagenase concentration or prolonging the digestion period. However, these solutions also increase cytotoxic activity to the cells. Therefore, we developed a new protocol with sequential short digestions to improve the quantity and quality of harvested chondrocytes.
We first used auricular cartilage to evaluate the efficiency of the newly developed protocol. Auricular cartilage is a subtype of elastic cartilage that is easy to harvest and has been widely used in CTE.3,22 After 3 digestions, the wet weight of residual auricular cartilage tissue was significantly decreased to approximately 42% of that with the classic protocol. Although the digestion time was shorter (8 hours in total), the average cell yield with the sequential protocol (13.32 × 106 cells/g) was significantly higher than that with the classic protocol (3.84 × 106 cells/g) in this study and that with other commonly used protocols (1 ~ 5 × 106 cells/g) in published studies.10,11,14 More importantly, the cells isolated using the sequential protocol showed higher cell viability and better proliferative activity. A long period of collagenase digestion has been reported to decrease both the quantity and quality of yielded cells during cell isolation from different tissue types.17,23 In the sequential protocol, the short digestion period can result in retaining a higher collagenolytic activity that benefits enzymatically gradating cartilage matrix, and the shorter exposure time in the collagenase solution can reduce cytotoxicity that results in the maintenance of high cell viability and high proliferative activity.
In this study, the perichondrium was peeled before mincing. However, H&E staining results revealed that 2 layers of overlaying tissue were still attached to the 4 types of cartilage tissues. Therefore, a 2-hour digestion was set as the first round, and 2 sequential 3-hour digestions were set as the following 2 rounds to improve cell-yield efficiency. As expected, the cells isolated from the first digestion had a lower cell-yield efficiency, viability, proliferative activity, and chondrocyte phenotype than those isolated from the following 2 digestions. During the first digestion, collagenase may contribute to digesting the outer overlaying tissues and exposing the core cartilage matrix, which creates conditions for subsequent digestions to achieve high cell-yield efficiency and quality. Although several similar 3-step digestions protocols have been reported in chondrocyte isolation, there was no convincing data that elaborated their superiority. Lau
In general, the chondrocyte quality is determined by phenotype and phenotype stability, cellular proliferative activity, and cartilage-specific matrix synthesis capacity. Although different chondrocyte isolation protocols have been reported, such as predigestion with trypsin, dispase, or hyaluronidase before collagenase digestion,10,26 the isolated chondrocytes are unavoidably contaminated by other cell types, such as fibroblasts and perichondral cells, which cannot deposit cartilaginous matrix
11
during CTE. In this study, the cells from the second and third digestions showed a comparably higher percentage of Col II-positive cells, higher cellular proliferative captivity, and higher cartilage-specific gene expression than those from the first digestion and with the classic protocol. CTE requires abundant chondrocytes, which can usually be achieved by cell expansion in a monolayer culture condition. However, cell passage may result in a lost chondrocyte-specific phenotype and dedifferentiation toward a terminal chondrocyte phenotype.27,28 Our results demonstrated that the expression levels of
Costal,29,30 septal,1,31 and articular chondrocytes32,33 have also been reported in CTE for different study purposes. Due to discrepancies among histological structures, individuals, and species, a fixed 1-step protocol is unsuitable for isolating chondrocytes with high quantity and quality. The sequential protocol provides a flexible and controllable solution for achieving this goal in different cartilage types. We therefore further assessed the efficiency of the sequential protocol in dissociating chondrocytes from costal, septal, and articular cartilage. After 3 digestions in the sequential protocol, the residual septal cartilage fragments almost disappeared. However, approximately 0.7 g of costal and 0.4 g of articular cartilage tissues still resided, which suggests that additional digestions are required for costal and articular cartilage tissues to achieve complete digestion. Despite the similarity with the results in auricular cartilage, in costal, septal, or auricular cartilage, the cells yielded from the second and third digestions showed higher cell-yield efficiency and higher viability than those from the first digestion and the 1-step protocol.
Conclusions
In this study, our results demonstrated that the cells isolated from the sequential 3-hour digestions exhibited higher cell-yield efficiency, higher cell viability, higher cell proliferative capacity, better chondrocyte phenotype, and higher cartilage-specific matrix synthesis ability than those from the first digestion and with the classic protocol. Therefore, overall, we recommend using the sequential protocol, particularly the cells obtained from the subsequent 3-hour sequential rounds of digestions, for chondrocyte-based therapy and CTE.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by grants from the National Natural Science Foundation of China (81871571 to R.-L.H.), the Shanghai Pujiang Program (2019PJD023 to R.-L.H.), and the Shanghai Municipal Key Clinical Specialty (shslczdzk00901 to R.-L.H.).
Author Contributions
Y.Y. and R.F. contributed to the design, data acquisition, and interpretation; performed all statistical analyses; and wrote the first draft of the manuscript. C.L. and J.Y. contributed to the data acquisition and interpretation. Q.L. contributed to the conception, design, and data interpretation. R.-L.H. contributed to the conception, design, data interpretation, manuscript writing, and revising. All authors revised and approved the submitted manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
All data supporting the findings of this study are available from the corresponding author upon reasonable request.
