Abstract
Objective
Matrix-associated autologous chondrocyte implantation (ACI) and microfracture (MF) are well-established treatments for cartilage defects of the knee. However, high-level evidence comparing microfracture and spheroid technology ACI is limited.
Design
Prospective, phase III clinical trial with patients randomized to ACI (N = 52) or MF (N = 50). Level of evidence: 1, randomized controlled trial. Both procedures followed standard protocols. For ACI 10 to 70 spheroids/cm2 were administered. Primary outcome measure was the Knee Injury and Osteoarthritis Outcome Score (KOOS). This report presents results for 36 months after treatment.
Results
Both ACI and MF showed significant improvement over the entire 3-year observation period. For the overall KOOS, noninferiority of ACI (the intended primary goal of the study) was formally confirmed; additionally, for the subscores “Activities of Daily Living” and “Sport and Recreation,” superiority of ACI over MF was shown at descriptive level. Occurrence of adverse events were not different between both treatments (ACI 77%; MF 74%). Four patients in the MF group required reoperation which was defined as treatment failure. No treatment failure was reported for the ACI group.
Conclusions
Patients treated with matrix-associated ACI with spheroid technology showed substantial improvement in various clinical outcomes after 36 months. The advantages of ACI compared with microfracture was underlined by demonstrating noninferiority, in overall KOOS and superiority in the KOOS subscores “Activities of Daily Living” and “Sport and Recreation.” In the present study, subgroups comparing different age groups and defect sizes showed comparable clinical outcomes.
Introduction
Articular cartilage defects affect around 1.5 million patients per year in Europe1,2 and are therefore a common orthopedic issue. Given the limited self-regenerative capacity of hyaline cartilage, this can be a source of pain and functional limitation of the joint. 3 Cartilage defects have been indicated as a cause of progressive degenerative changes, leading to osteoarthritis. 4 This represents a medical need for the treatment of cartilage defects in order to avoid an increasing severity, and a chronic condition, of the initial cartilage defect and to reduce the risk of the patient’s developing osteoarthritis (OA). Treatment of cartilage defects is thus important for the prevention of the need for a total knee replacement.5-7
According to recommendations by national and international medical associations, autologous chondrocyte implantation (ACI) is recommended for medium to large cartilage defects of the knee, 8 as superiority to microfracture (MF) has been shown especially in defect sizes greater than 2 to 4 cm2. 9 Long-term results of ACI are now available, including patient data obtained over up to 20 years, supporting the durability of a good clinical outcome after ACI.10-14
In contrast, more and more studies have questioned the benefits of microfracture, owing to a variety of concerns. The amount and quality of the repair tissue in MF is dependent on bone marrow stem cells, and their number and reparative capacity are decreased in older patients, therefore better results have been shown for patients younger than 40 years.15-18 Furthermore, the repair tissue has been reported to consist of fibrocartilage19-21 and these factors may account for the poorer results in larger defects as well as the deterioration observed in long-term follow-up, which has frequently been reported.5,10,12-14,22-26
However, numerous studies evaluated periosteal or collagen covered ACI and thus it is important to examine matrix-associated ACI, especially with spheroid technology, as it displays the latest advancement of ACI and to adduce scientific evidence for its effectiveness.
This study compares autologous matrix-associated ACI (Spherox; CO.DON, Teltow, Germany) with arthroscopic MF in a prospective, randomized trial.
Patients and Methods
Study Design and Surgical Treatment
Study design and surgical treatment have been described previously. 27 Briefly, the multicenter phase III clinical trial (NCT01222559; EudraCT Nr. 2009-016466-82) was designed as a randomized controlled trial comparing the efficacy and safety of treatment with (a) the matrix-associated ACI with Spherox and (b) MF.
The patients were randomized by using a fixed block size (6) stratified prospectively by age into 2 classes (18-34 and 35-50 years). Patients in each class were allocated randomly on a 1:1 basis to group A (ACI) or B (MF). The randomization was implemented by telephone by the clinical research organization during the operation. The procedures prevented blinding.
The sample-size calculation considered the overall KOOS (change from baseline to 24 months) and was focused on the test for noninferiority of ACI compared with MF (1-sided α = 0.025, power = 80%, lower equivalence bound = −8.5%, expected mean difference = 0, standard deviation = 15). The sample size was based on a repeated-measures analysis of covariance (ANCOVA) approach to estimate the overall effect. Hence, a minimum sample size of N = 90 (ie, 45 MF and 45 ACI) was calculated. As Spherox is an advanced medicinal therapeutic product, specifically tissue-engineered product, the cell culture is highly depending on the individual patient’s biopsy material. Thus, there is always the risk of insufficient cell culture, especially as the product is fully autologous. An estimated dropout rate of 7.5% for the MF group and 15.5% for the ACI group (greater for ACI than for MF because of the risk of insufficient cell culture) had to be included, which led to a final sample size of N = 102 (50 MF and 52 ACI). Within a prespecified hierarchical test design (and thus as a method of adjustment for multiple testing in terms of a closed testing procedure), noninferiority was only tested given a significant relevant change from baseline to 24 months of more than 10 percentage points in ACI. In case of noninferiority, the hierarchical structure allows for testing superiority.
Of the 163 patients screened, 102 were assessed as eligible to participate in the study and were included.
After the trial had been approved by the ethics committees responsible for the respective centers and by the local regulatory authorities, it was registered internationally. Patients who consented in writing to trial participation were included, between December 2010 and December 2014, at 8 German and 3 Polish orthopedic centers. There was no primary center within this trial. Patients were recruited within centers as eligible. The study was designed to include up to 5 years of follow-up. The results presented here are derived from the 3-year follow-up analysis, which was completed in August 2018.
Patients’ final eligibility was assessed through an arthroscopy, to ensure the inclusion of symptomatic unipolar, full-thickness focal cartilage defects of grade III or IV, with a defect size between 1 and 4 cm2 and with intact adjacent cartilage. Detailed inclusion and exclusion criteria are presented in Table 1 .
Main Inclusion and Exclusion Criteria.

ICRS = International Cartilage Repair Society; NSAIDs = nonsteroidal anti-inflammatory drugs; MRI = magnetic resonance imaging; ACI = autologous chondrocyte implantation.
Surgical Techniques and Rehabilitation
Patients were randomized (1:1) to ACI or MF. ACI treatment required 2 surgical interventions: biopsy to obtain cartilage cells that are used to grow chondrocytes in vitro and subsequent implantation of the spheroids containing these chondrocytes. Before surgery, the investigator estimated the defect size using magnetic resonance imaging (MRI) evaluation and during arthroscopy for tissue procurement this defect size was verified. Concomitant surgery (e.g. ligament repair, high tibial osteotomy, meniscal repair) was not performed in this clinical trial.
During second arthroscopy, damaged articular cartilage and/or associated flap(s) were debrided with a 4.5-mm oscillating shaver and/or a sharp curette to produce a stable tissue border to the adjacent cartilage. Debridement was performed to the subchondral bone without subchondral abrasion to leave the subchondral bone intact. The number of spheroids implanted was to be within the manufacturer’s normal recommended range of 10 to 70 per cm2 (application of the maximum producible number within this range), which was confirmed in the phase II clinical trial; details are given in an earlier publication.28,29 The spheroids are delivered on ice (4°C) and taken from the transport box directly before implantation. The ice-cold spheroids are implanted into the defect site and distributed by the surgeon over the whole defect area. This may be performed with a mini arthrotomy or arthroscopically assisted. The spheroids are delivered in an applicator, which is connected to a syringe which is filled with air. Spheroids may be applicated using the syringe and are self-adhesive to the bone. During warm-up to body temperature, the spheroids are slightly flattening and adhering to the defect surface. Twenty minutes are proven as sufficient time for initial adhesion since the joint is stabilized directly after surgery using a medical track and thus the initial adherence can expand after surgery without disturbance.
For MF, a single intervention was performed according to Steadman et al. 30
Following surgery, all patients followed a standardized rehabilitation program for the surgical treatments. Partial weightbearing was recommended for 6 weeks with 10 to 20 kg, started at the first postoperative day. Regaining full weightbearing was recommended within 7 to 8 weeks or latest after 12 weeks. Continuous passive motion was started from the day after surgery for 6 weeks, increasing from 0-0-60° in the first week to 0-0-90° until week 6. The first 6 weeks included physiotherapy aimed primarily on reduction of swelling, isometric quadriceps activity and mobilization. From week 7, increase to full range of motion was encouraged, proprioceptive and muscular training was increased and cycling or aquajogging was permitted. Physiotherapy was adjusted to individual joint status and complaints. Return to high-impact sports was recommended earliest after 12 months.
Assessment Criteria
The Knee Injury and Osteoarthritis Outcome Score (KOOS), which has been validated for focal cartilage lesions,31-33 was the primary efficacy variable. The KOOS subscores were assessed as secondary efficacy variables. Other assessments included MRI observation of cartilage repair tissue (MOCART). Adverse events, vital signs, electrocardiography, physical examination, concomitant pain medication, and laboratory values were documented. Adverse events (AEs) and serious adverse events (SAEs) were classified by the investigators according to MedDRA coding. Treatment was defined as failed if the initial defect required reoperation.
Statistical Evaluation
The main variable for this article was the overall KOOS 36 ± 2 months after treatment. The analysis was performed according to a prospectively defined hierarchical scheme (closed test procedure) by using a repeated-measures ANCOVA including baseline visit as well as visit 3 to visit 7 (at 6, 12, 18, 24, and 36 months). First, the ACI group was tested for relevant clinical improvement from baseline using within-subject effects. If the change from baseline to 36 months was significantly above 10 percentage points, the 2 treatment groups could be compared. Second, if the lower 95% confidence limit of the least square mean difference was greater than −8.5, the ACI treatment was to be regarded as significantly noninferior to microfracture. In the final step: if the lower confidence limit of the least square mean difference was not only greater than −8.5, but also greater than 0, ACI was to be regarded as superior to MF. The change from baseline to 12 and 24 months was tested simultaneously. Thus, estimates for those time points are post hoc results, which are exploratively given unadjusted for multiple testing. MF is considered an effective treatment for smaller defect sizes of 1 to 4 cm2 and it has been deemed unethical to treat patients with higher defect sizes, where microfracture already has been shown to be inferior to ACI. 8 Therefore, the present study was powered for noninferiority at visit 6 (i.e., 24 months after transplantation). All testing other than the above was at descriptive level or analyzed exploratively at full level of significance (α = 5%). Noninferiority and superiority analyses were accordingly performed for the KOOS subscores (pain, other symptoms, function in daily living, function in sport and recreation, knee-related quality of life).
Clinical outcome was also assessed for each treatment group using a threshold of 8 percentage points as the minimal clinically important difference (MCID) from baseline in the overall KOOS. 34 Subgroup analyses stratified by age group and defect size were performed.
The intention-to-treat (ITT) population was defined as comprising all patients who were (a) randomized, (b) treated with ACI or MF, and (c) completed the KOOS questionnaire at baseline. A supporting per-protocol analysis was also conducted.
The statistical analyses were performed by StatConsult GmbH (Magdeburg, Germany).
Results
Patient Population
The study population comprised 102 patients (61 male, 41 female) aged 37 ± 9 years. A detailed illustration of the grouping and flow of patients within the trial is shown in Figure 1 . In this clinical trial, for only 1 patient insufficient cell material were grown out of the biopsy material and thus no product could be produced, and no treatment occurred (the patient was included in the safety population, but not in ITT). The treatment groups were well balanced regarding demography and disease background (cf. Table 2 ). Primary defect location was the femoral condyle, with 1 patient also showing a patellar defect (and thus violating an inclusion criterion). ICRS grades were adequately balanced between the treatment groups, with more than one-half of defects in both groups being of grade IV A. Defect sizes before debridement ranged from 0.5 to 4.0 cm2 and were likewise well balanced ( Table 2 ). The compliance rate to the rehabilitation program was excellent with 98% in both groups. Concomitant surgery was not performed within the trial.
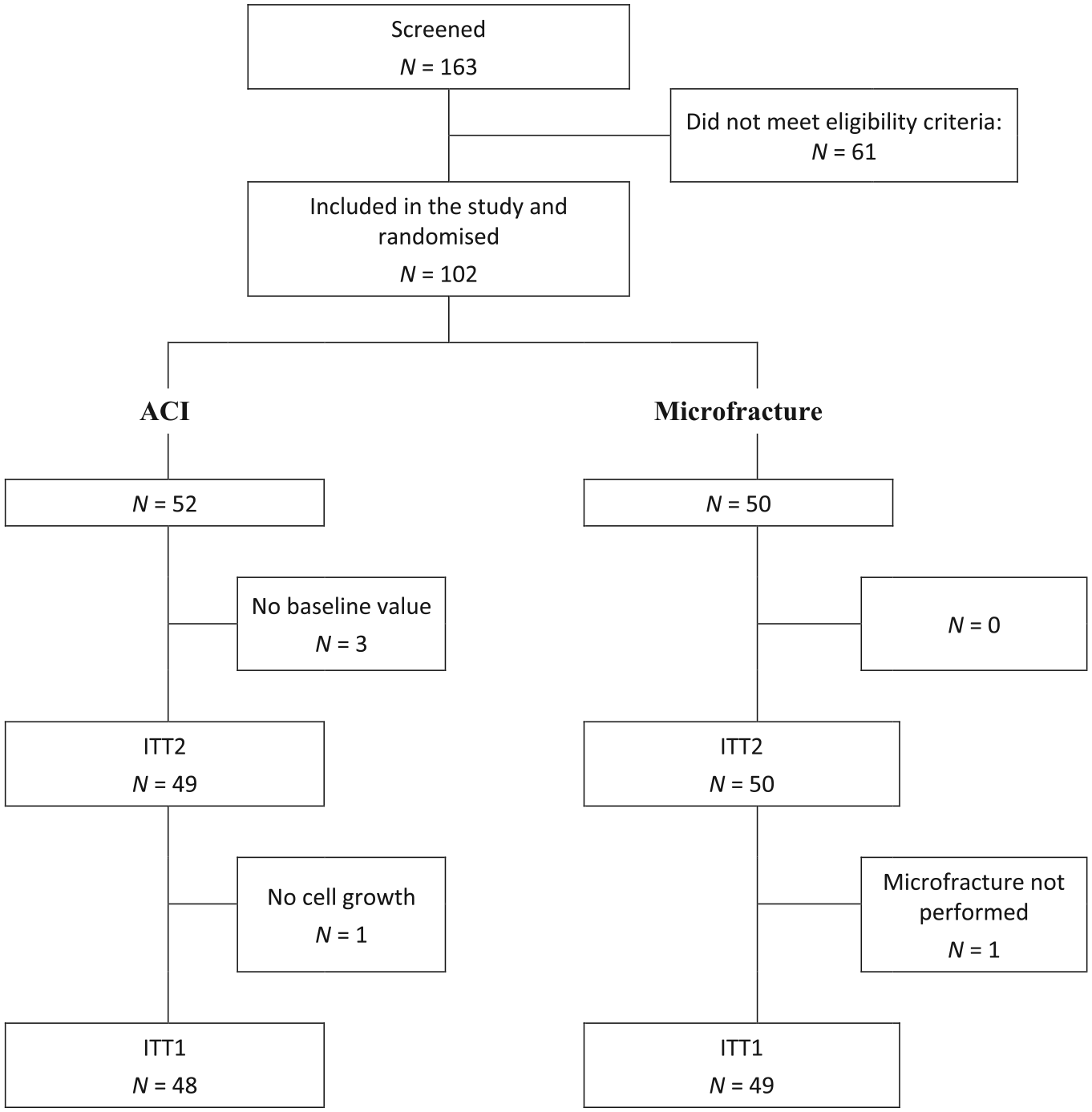
Patient disposition. ACI, autologous chondrocyte implantation; ITT, intention to treat; PV, protocol violation.
Patients’ Demographic and Baseline Data.
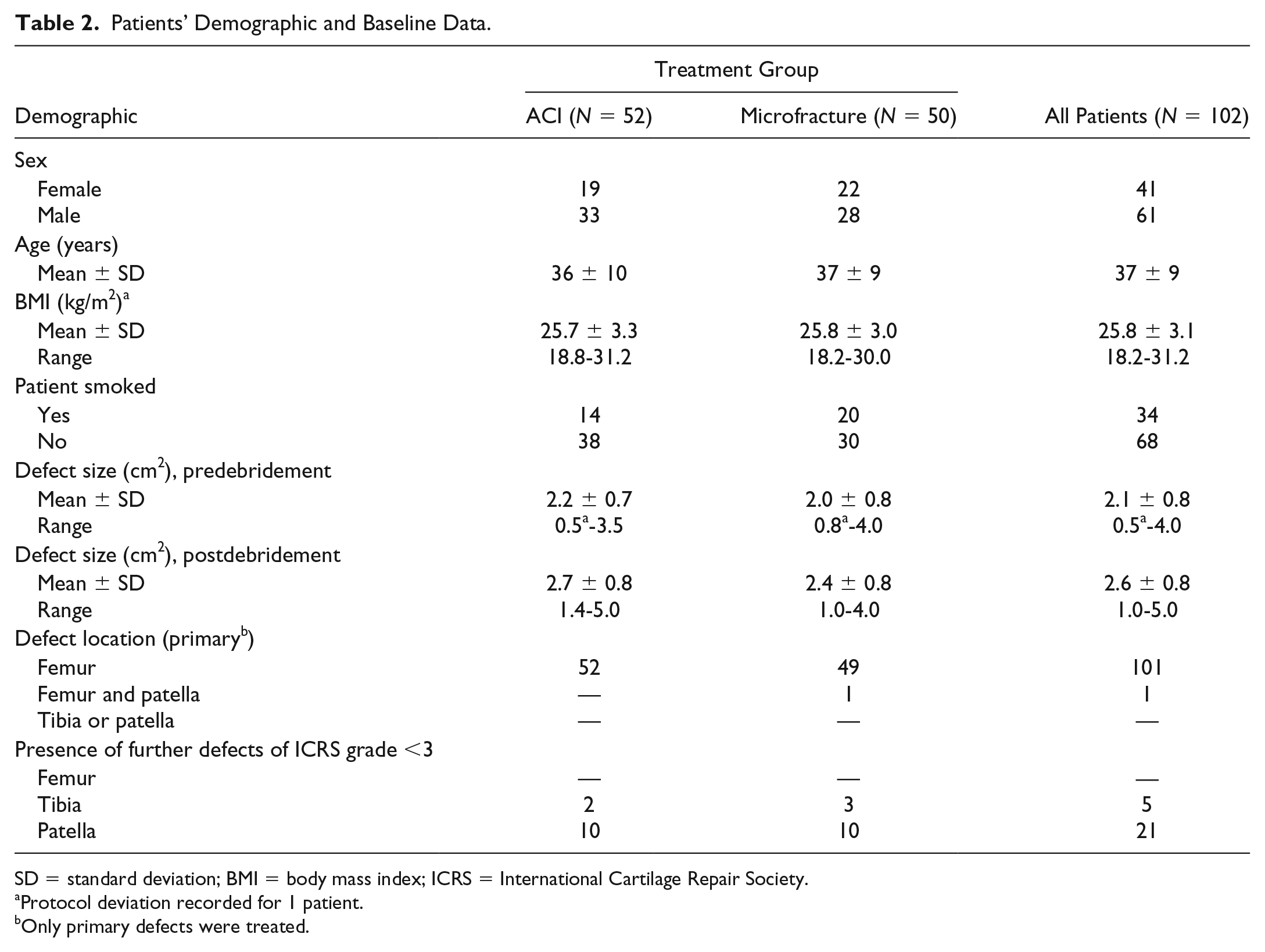
SD = standard deviation; BMI = body mass index; ICRS = International Cartilage Repair Society.
Protocol deviation recorded for 1 patient.
Only primary defects were treated.
KOOS Overall and KOOS Subscores “ADL” and “Sport/Rec”
Table 3 shows mean scores ± standard deviation (SD) and median for the overall KOOS and the KOOS subscores Activities of Daily Living (ADL) and Sports and Recreation (Sport/Rec) at baseline, and after 12, 24, and 36 months (see also Fig. 2 ). Both treatment groups showed the greatest improvement in the overall KOOS as well as in the subscores within the first year and continued to improve slightly until the third year as shown in Table 3 .
Overall KOOS and the KOOS Subscores “ADL” and “Sport/Rec” at Baseline and after 12, 24, and 36 Months. a
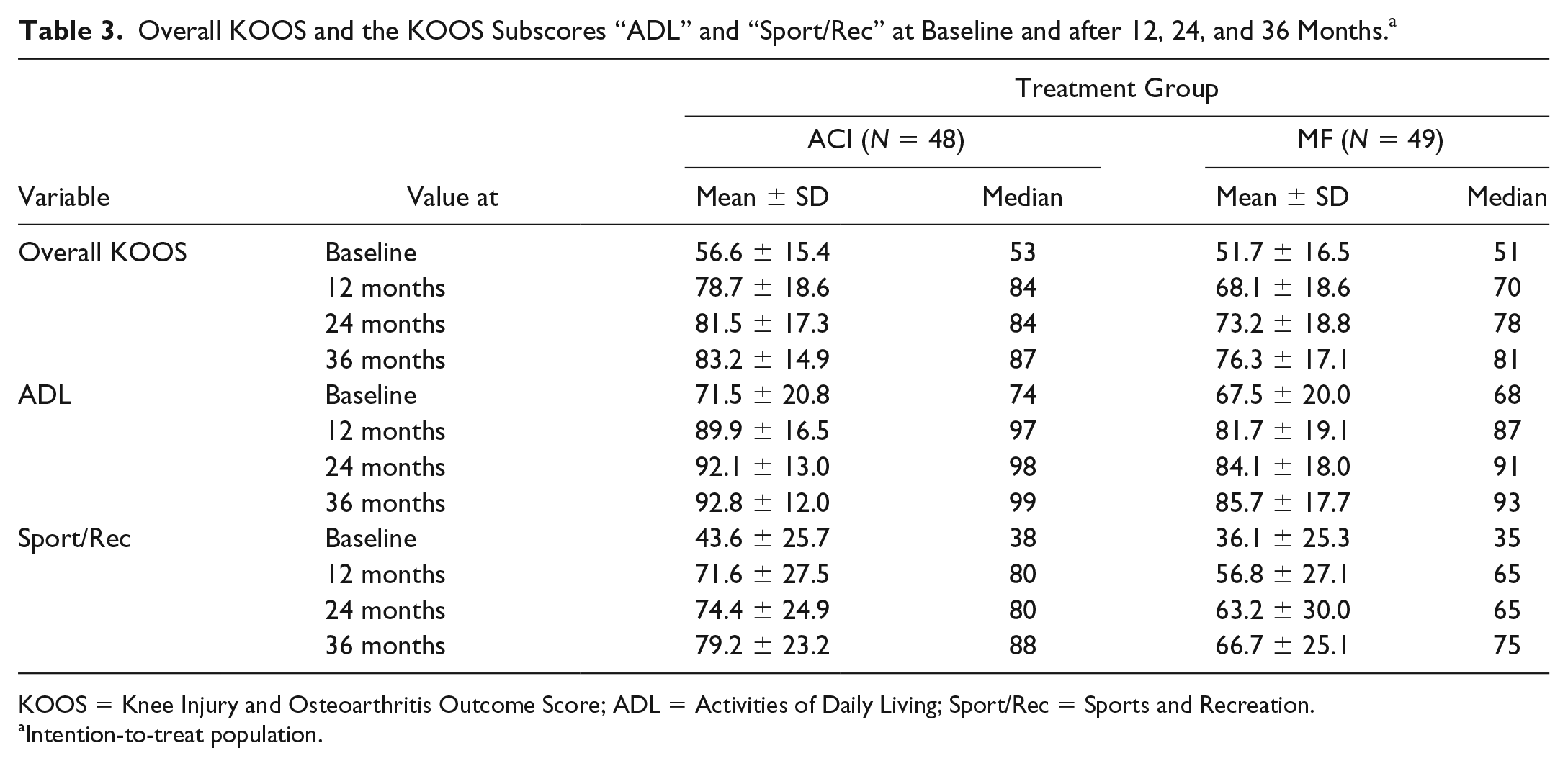
KOOS = Knee Injury and Osteoarthritis Outcome Score; ADL = Activities of Daily Living; Sport/Rec = Sports and Recreation.
Intention-to-treat population.
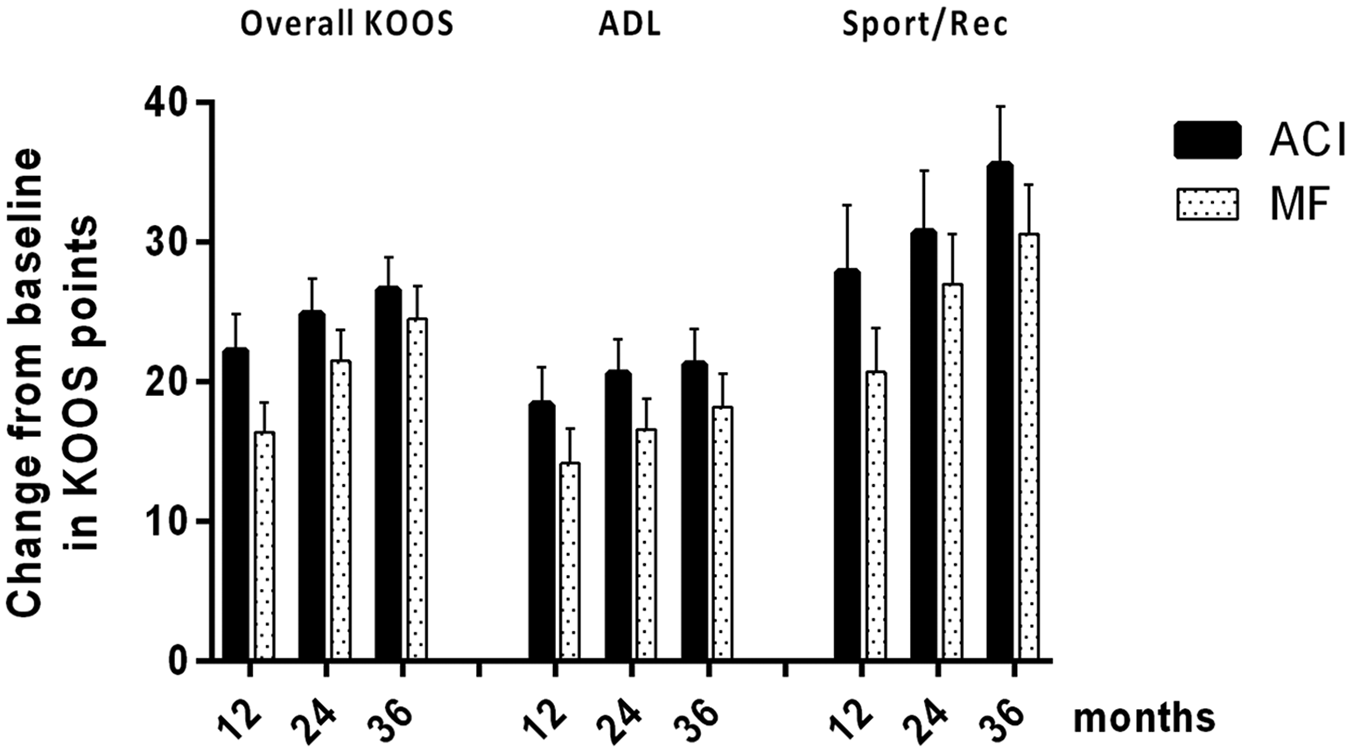
Change from baseline in overall Knee Injury and Osteoarthritis Outcome Score (KOOS) and the KOOS subscores “function in activities of daily living” (ADL) and “function in sport and recreation” (Sport/Rec) at baseline and after 12, 24, and 36 months for ACI and MF group. Means and standard error of the means are displayed. ACI, autologous chondrocyte implantation; MF, microfracture.
The results of hierarchical statistical testing are summarized in Table 4 (step 1, follow-up vs. baseline, and step 2, ANCOVA). All the within-group comparisons (follow-up vs. baseline) had a lower confidence interval (CI) bound above 10 percentage points (relevant clinical improvement) for ACI.
Test for Relevant Clinical Improvement from Baseline to Follow-up. a
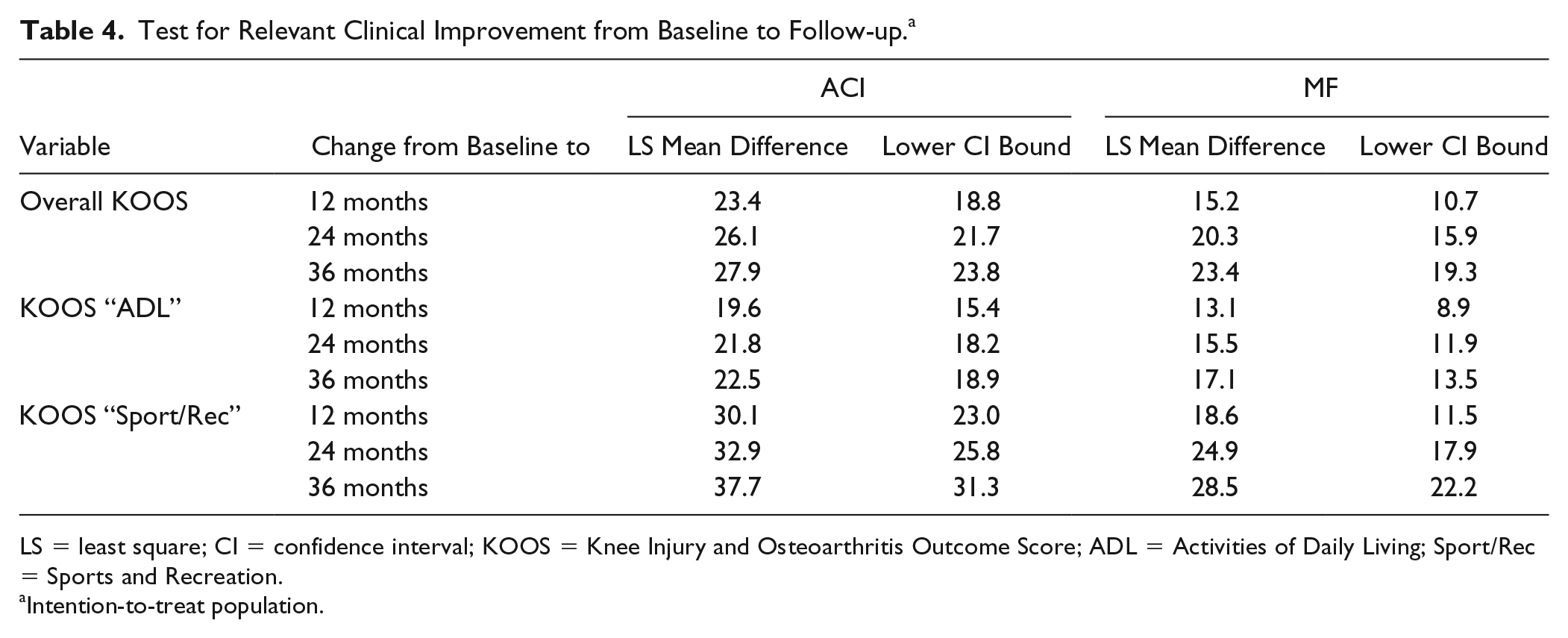
LS = least square; CI = confidence interval; KOOS = Knee Injury and Osteoarthritis Outcome Score; ADL = Activities of Daily Living; Sport/Rec = Sports and Recreation.
Intention-to-treat population.
In the repeated-measures ANCOVA ( Table 5 ) for the overall KOOS noninferiority was demonstrated, statistically significant superiority was narrowly missed at all assessed time points, except at the 12-month assessment. As the study was designed and powered for non-inferiority, achievement of noninferiority is defined as study success. Nevertheless, the subscore “Sport/Rec” even showed, at a descriptive level, superiority of ACI over MF at 12 and 36 months and “ADL” showed this at all 3 time points (12, 24, and 36 months). For the other subscores at each visit, greater values were found in the ACI group than in the MF group, again demonstrating noninferiority, and superiority could be shown for the KOOS subscore “Quality of Life” at 12 months (data not shown). The results of the per-protocol analysis were in line with the findings from the ITT population (data not shown).
Repeated-Measures ANCOVA to Test Superiority of ACI over MF. a

ANCOVA = analysis of covariance; ACI = autologous chondrocyte implantation; MF, microfracture; LS = least square; CI = confidence interval; KOOS = Knee Injury and Osteoarthritis Outcome Score; ADL = Activities of Daily Living; Sport/Rec = Sports and Recreation.
Intention-to-treat population.
Primary variable, shown here for comparison.
Minimal Clinically Important Difference in Overall KOOS ACI Versus MF
Roos and Lohmander 34 reported a limit of at least 8 percentage points for the MCID, which is defined as the smallest change in treatment outcome that an individual patient would identify as important and which would require a change in the patient’s management. In both groups, the great majority of patients showed an increase in overall KOOS of 8 or more points 36 months after treatment, with a greater rate in the ACI group than in the MF group (90% and 84%, respectively).
Safety of ACI Versus Microfracture
During the 3-year follow-up, 28 out of 52 (54%) patients in the ACI group experienced a total of 67 adverse events that were assessed as treatment-related by the investigators (adverse reactions). In the MF group, 28 of 50 (56%) patients experienced a total of 68 adverse reactions. While in the ACI group no SAE was assessed as treatment-related, in the MF group there were 5 possibly treatment-related SAEs were reported, all of moderate severity (cf. Table 6 ), affecting 4 patients. Four out of these 5 treatment-related SAEs in the MF group are considered as treatment failure (resurgery necessary), while no such failure occurred in the ACI group. The affected patients were treated as follows: The “joint adhesion” was salvaged by arthroscopic arthrolysis, the patient suffering from “cartilage injury” and “arthralgia” was treated with mosaicplasty. The event of “osteochondrosis” required treatment by open osteochondral transplantation, implantation of alloplastic bone surrogate material.
Treatment-Related Serious Adverse Events (Safety Population).

In the same patient.
The majority of the adverse reactions for both groups occurred within the first 12 months. In the ACI and MF groups, respectively, 78% and 79% of the adverse reactions or treatment-related SAE events were in the MedDRA system organ class (SOC) “musculoskeletal and connective-tissue disorders.” Table 7 shows adverse reactions related to ACI and MF. The most frequently reported terms were “joint effusion” (n = 23 for ACI, n = 18 for MF), “arthralgia” (n = 13 for ACI, n = 21 for MF), and “joint swelling” (n = 11 for ACI, n = 10 for MF). Evaluation of the individual events in other SOCs, most of which occurred once or twice only, did not reveal any likely association with either treatment. The only conspicuous difference was the reporting of 4 cases of bone marrow edema in the MF group, while no such event was reported for the ACI group.
Adverse Events Classified as Probably or Possibly Related to the Study Treatment, by MedDRA SOC and Preferred Term. a
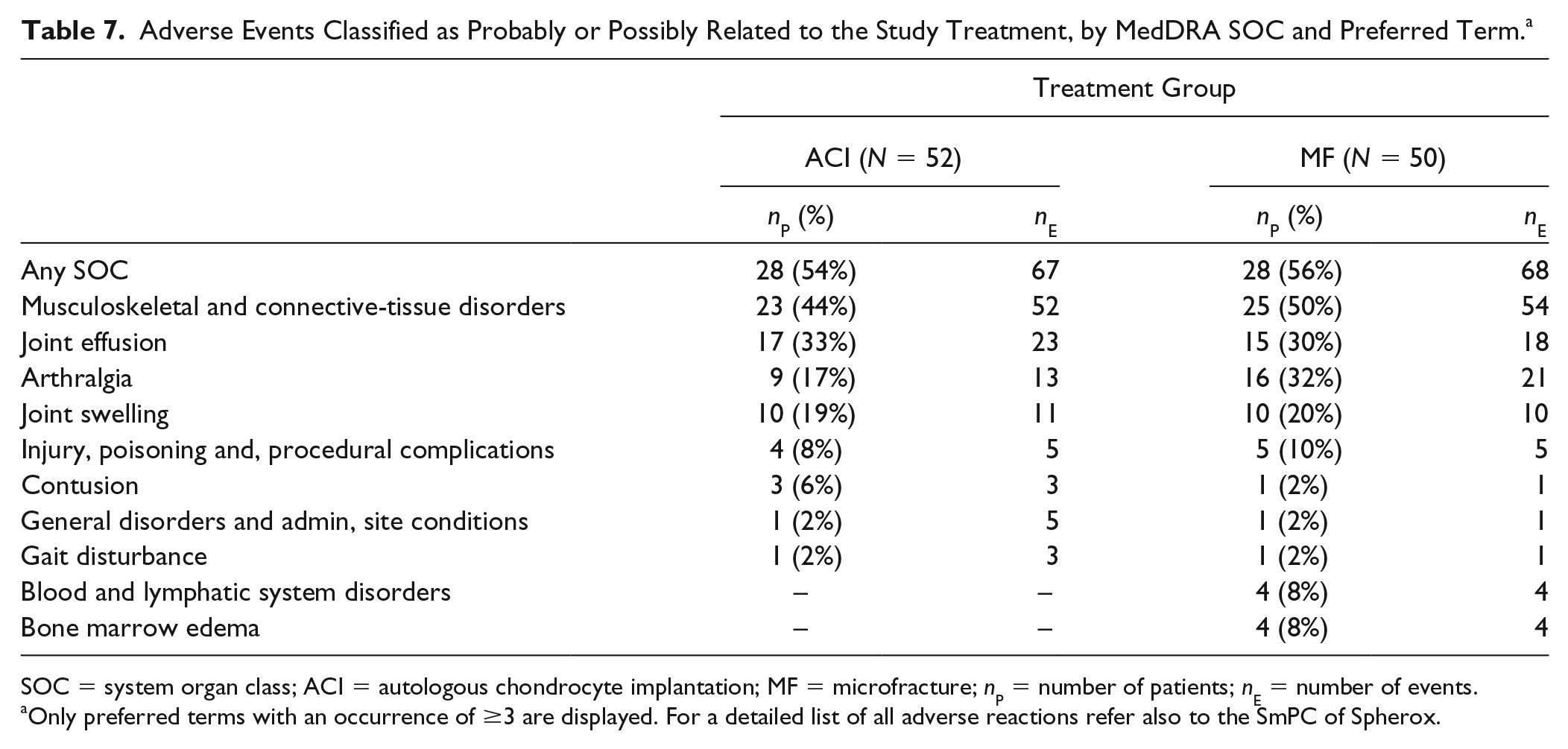
SOC = system organ class; ACI = autologous chondrocyte implantation; MF = microfracture; nP = number of patients; nE = number of events.
Only preferred terms with an occurrence of ≥3 are displayed. For a detailed list of all adverse reactions refer also to the SmPC of Spherox.
Vital signs, electrocardiography, laboratory values, and recorded concomitant pain medication were monitored: There were only 5 cases of slightly abnormal laboratory values in the ACI group (2× mild hypertriglycemia, 1× mild γ-glutamyl transferase increase) and 1 in the MF group (mild bilirubin increase). None of the results from vital sign measurement gave rise to any clinical concern about the safety of the study treatment. Almost all findings in the ECG were normal; the few abnormal findings were considered to be clinically insignificant by the investigators. Thus, they do not yield any sign of a safety concern associated with either treatment.
Influence of Age on the Overall KOOS
In both treatment groups, the age group “’35 to 50 years” started with lower baseline values than the group “’18 to 34 years,” though the difference was more pronounced in the ACI group (for these 2 age groups, respectively, 51.4 ± 14.2 and 65.2 ± 13.8 in the ACI and 49.7 ± 18.5 55.2 ± 12.2 and in the MF group). The change from baseline at 36 months after implantation was 28.9 ± 17.1 in the age group “’35 to 50 years” and 22.9 ± 14.2 in the group “’18 to 34 years” for patients treated with ACI; in the MF group, the change from baseline was 26.3 ± 18.0 and 21.6 ± 14.1, respectively. Thus, the treatment results in both, ACI and MF, seemed comparable between the age groups even though the study was underpowered for statistical comparison.
Influence of Defect Size on the Overall KOOS
In the size range included in this study, that is, 1 to 4 cm2, the increase in overall KOOS and in the subscores were found independent of defect size for both, ACI and MF group. The change from baseline in overall KOOS until 36 months after implantation was compared between groups of 1 to 2 cm2 and 2.1 to 4 cm2 defect size revealing comparable mean values of 29.3 ± 14.2 and 24.2 ± 17.7 for the ACI group and 26.4 ± 15.7 and 20.6 ± 18.4 for the MF group.
Structural Comparison between ACI and MF
As further secondary outcome, both treatment groups were assessed radiologically by the MRI-based MOCART score. 27 For the overall MOCART score, “0” represents the worst possible diagnosis and “100” represents a normal joint. Both ACI and MF lead to an improvement from the 3-month visit up to a nearly normal joint status, but no significant differences between treatment groups could be shown. There was a limitation due to the fact that—as in all cartilage-regeneration studies—MOCART assessment before treatment was technically impossible since the score is designed to assess the quality of repair tissue, which does not yet exist before treatment. Thus, a strict “before-after” comparison was impossible.
Discussion
The 3-year results of a prospectively designed, randomized pivotal phase III study comparing the efficacy and safety of 2 treatments for cartilage defects of the knee: (a) matrix-associated autologous chondrocyte implantation with spheroid technology and (b) arthroscopic MF are described.
The objective of the study, the formal demonstration of noninferiority of the ACI treatment compared with MF, was achieved. Conspicuously, even though the study was not powered to demonstrate superiority, the latter was also narrowly missed (overall KOOS values, Table 5 ). Moreover, the 2 KOOS subscores “ADL” and “Sport/Rec” did even show superiority of ACI over MF at the descriptive level ( Table 5 ) and the results were independent of age and defect size. This was possible due to the fact that the study was powered for noninferiority assuming a mean difference of zero (cf. sample size calculation). But since results for ACI showed better values than MF, the effect size and consequently the power were much higher than expected and even superiority could be shown in parts.
Both ACI and MF are treatments widely used for the repair of cartilage defects. It is generally accepted that ACI offers advantages in the treatment of large defects compared with MF, and it is therefore especially recommended by expert associations for such indications.8,22
Against this background, the study included only patients with smaller cartilage defects up to 4 cm2, and therefore, considering the existing knowledge of the efficacy of MF in this defect-size range, it was conducted as a noninferiority study. 8 This might also account for the results of the present study showing partial superiority of ACI over MF and is in accordance with the results of similar trials.5,19,35 Saris et al. 35 evaluated ACI using characterized chondrocytes at a mean defect size (2.6 cm2) comparable to that of the present study (2.1 cm2); they reported superiority of ACI over MF in the KOOS subscores “pain” and “ADL.” However, overall superiority was missed. For the study of Basad et al. 9 no average defect size was reported, but the inclusion criteria allowed the inclusion of defects of 4 to 10 cm2. Consequently, while the present study and the study of Saris et al. 35 only showed partial superiority due to the smaller defects included, Basad et al. 9 showed an overall superiority of ACI.
In the present analysis, superiority of ACI over MF was achieved for the “Sport/Rec” and “ADL.” For the other KOOS subscores at each visit, greater values were found in the ACI group than in the MF group, again demonstrating noninferiority and superiority was shown for the KOOS subscore “Quality of Life” at 12 months (data not shown). Hambly and Griva 36 assessed the clinical impact of different subscores of the KOOS and IKDC (International Knee Documentation Committee) questionnaires: aspects assigned to the KOOS subscores “Sport/Rec” as well as “knee-related quality of life” (QoL) showed the highest impact on patient outcome. Thus, the superiority found in “Sport/Rec” may be of high clinical importance. The lack of superiority in the subscore “QoL” after 12 months can be explained by the ceiling effect for this subscore, as shown by Bekkers et al., 31 which probably makes it difficult or even impossible to distinguish the beneficial effect of MF from the putatively better outcome after ACI.
Even though Hambly and Griva 36 rated the subscore “ADL” as being of minor clinical impact, Marx et al. 37 found it to be highly representative of knee joint function. Thus, the superiority found in this subscore by the present analysis represents a superior knee joint function in patients after ACI compared with MF.
In a study by Saris et al., 35 using a matrix-associated ACI with a collagen type I/III membrane (MACI; Genzyme, Cambridge, MA, USA), ACI showed advantages in functional outcome as measured by 2 KOOS subscores (“pain” and “ADL”) after 24 months. This advantage was confirmed by the present analysis for the KOOS subscore “ADL” at 24 and 36 months. Furthermore, an advantage in the KOOS subscore “function sport and recreation” was shown, which confirms the results of an earlier study by Basad et al., 9 in which clear superiority of ACI vis-à-vis MF according to the Tegner Activity Score was found after 24 months. Also, our analyses of the MCID rates showed the advantage of ACI over MF, though without statistical significance.
The lower treatment failure rate for ACI compared with MF is in accordance with a recently published population representative matched-pair claims data analysis showing a 20.3% rate of reoperation after MF versus 12.5% after ACI (P = 0.0199). 38
In the ACI as well as in the MF group, the increase in overall KOOS and its subscores seems similar in small (1-2 cm2) compared with larger (2-4 cm2) defect sizes, even though the present study was not powered for statistical comparison. The current guideline issued by the German Association of Orthopaedics (DGOU) recommends the treatment of defects of ≥2 cm2 with ACI. 8 However, further studies are needed to substantiate the observations obtained in this study and to possibly justify the application of ACI even in smaller defects. Since no patients with larger defects were included, no conclusions can be drawn about the limitation of MF described elsewhere for defects above 4 cm2.7,8,17,18,23,39,40 The effective treatment of cartilage defects up to 10 cm2 was shown for matrix-associated ACI with spheroid technology during a phase II clinical trial.28,29
Since the reparative capacity of MF is based on stem cells, the numbers of which are decreased in older patients, studies have shown that mainly younger patients can benefit from MF.15-18 Interestingly, in the present analysis descriptively change from baseline in the overall KOOS seemed comparable between patients aged 18 to 34 and 35 to 50 years, both in the ACI and the MF group.
Thus, our results might suggest that even elderly patients can benefit from cartilage repair to an extent similar to that experienced by younger patients. This of course requires confirmation by other clinical trials specifically powered for statistical comparison between age groups.
Despite the high evidence level of the current study, some limitations have to be mentioned. One major limitation of the study was that blinding the subjects was impossible, because of the different numbers and types of procedures involved, and because of ethical restrictions on performing sham operations; thus, the risk of bias due to a placebo effect cannot be completely ruled out.
Furthermore, concomitant surgery was not performed in this clinical study and previous surgical procedures in the affected knee prior to the first arthroscopy (e.g., previous ACI, MF less than 1 year before baseline, meniscal implants, meniscal suture less than 3 months before baseline, mosaicplasty) were exclusion criteria. Furthermore, patients included in this study did not experience previous MF or meniscal suture. Thus, no conclusion can be drawn regarding the performance of either MF or ACI as second-line treatment or in patients requiring concomitant surgery. This, however, should be analyzed in future studies.
A second important limitation was the fact that the study was—within a hierarchical design—powered for noninferiority in the primary variable (overall KOOS). Even though its formal aim was attained, powering for superiority would have been preferred from a clinical perspective, to broaden the use of ACI in a clinical setting. The fact that overall superiority was closely approached, and achieved for 2 subscores, despite that the study was not powered for this, underlines the importance of the results. This is further supported by the fact that large cartilage defects were not included in the study.
In conclusion, in this prospective, randomized, open-label, multicenter phase III clinical trial, patients treated with matrix-associated ACI with spheroid technology had at least noninferior and in some points greater improvement in clinical outcomes (KOOS as well as in all other scores like IKDC, modified Lysholm, etc.) up to 36 months after treatment when compared to patients treated with MF. This advantage of ACI was backed up by a demonstration of non-inferiority in the primary outcome variable (overall KOOS) and was even more pronounced for the high-clinical-impact KOOS subscore “function in sport and recreation” as well as the subscore “activities of daily living,” which is highly representative of current knee joint function. This advantage is reflected by the increased rate of patients whose overall KOOS increased by 8 points (or more) in the ACI group compared with MF. Identifying patients at risk of progression of cartilage degeneration and better prediction of those unlikely to respond to MF treatment remain important challenges to ensure the greatest possible benefit from the not only more sophisticated but also more expensive, regenerative joint-resurfacing approach of ACI.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:The study was funded by CO.DON AG.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.H. received royalties for medical consultancy for CO.DON AG. W.Z. has received consultant fees from CO.DON AG for supporting a Scientific Advice of the EMA on a different project in 2018. S.F. has received consultant fees from Arthrex and Bauerfeind. Furthermore, some of the authors are participating as investigators in other clinical trials and receive fees for that. Each author has been rewarded with an investigator fee as outlined in the initial clinical trial authorization documents and accepted by the corresponding ethic committees. No further sponsorship was granted.
Ethical Approval
The trial was approved by the ethics committees responsible for the respective centers and by the local regulatory authorities. The main ethics commitee was Mannheim, Germany (2009-070F-MA).
Informed Consent
Written informed consent was obtained from all participants before the study.
Trial Registration
NCT01222559; EudraCT Nr. 2009-016466-82.
