Abstract
Objective
The interaction between proteoglycan loss and collagen damage in articular cartilage and the effect of mechanical loading on this interaction remain unknown. The aim of this study was to answer the following questions: (1) Is proteoglycan loss dependent on the amount of collagen damage and does it depend on whether this collagen damage is superficial or internal? (2) Does repeated loading further increase the already enhanced proteoglycan loss in cartilage with collagen damage?
Design
Fifty-six bovine osteochondral plugs were equilibrated in phosphate-buffered saline for 24 hours, mechanically tested in compression for 8 hours, and kept in phosphate-buffered saline for another 48 hours. The mechanical tests included an overloading step to induce collagen damage, creep steps to determine tissue stiffness, and cyclic loading to induce convection. Proteoglycan release was measured before and after mechanical loading, as well as 48 hours post-loading. Collagen damage was scored histologically.
Results
Histology revealed different collagen damage grades after the application of mechanical overloading. After 48 hours in phosphate-buffered saline postloading, proteoglycan loss increased linearly with the amount of total collagen damage and was dependent on the presence but not the amount of internal collagen damage. In samples without collagen damage, repeated loading also resulted in increased proteoglycan loss. However, repeated loading did not further enhance the proteoglycan loss induced by damaged collagen.
Conclusion
Proteoglycan loss is enhanced by collagen damage and it depends on the presence of internal collagen damage. Cyclic loading stimulates proteoglycan loss in healthy cartilage but does not lead to additional loss in cartilage with damaged collagen.
Introduction
Proteoglycan (PG) loss and damage to the collagen network are among the first signs of early osteoarthritis (OA) in articular cartilage. PGs give cartilage resistance to compressive loading by generating a swelling pressure, which is maintained in the tissue by the arcade-like structure of collagen that restricts swelling and gives the tissue its resistance to tension. This mechanical coupling between PGs and collagen suggests a relationship between PG depletion, collagen damage, and mechanical loading in the onset of early stage OA. However, the exact nature of this relationship is still unclear.
1
PG loss precedes extracellular matrix (ECM) degradation and collagen loss in articular cartilage explants cultured in the presence of interleukin-1 (IL-1)2-5 or retinol.
6
Conversely, when ovalbumin is injected to induce arthritis in rabbits, collagen type II cleavage by collagenase is detected earlier in synovial fluid than PG loss.
7
Additionally, collagenase treatment decreases the PG content, especially if the treatment duration is longer.
8
Finally, collagen damage may promote damage to the ECM and
Loading regimes and biochemical treatments play an essential role in stimulating or inhibiting PG depletion from articular cartilage, and this behavior may be altered during the post-loading period. In general, submitting cartilage to cyclic loading enhances PG loss,10-15 and this effect may be altered by peak stress,12,13 load frequency10,14, and duration of unloading periods. 14
Using a methyl methacrylate indenter in confined compression, Lee and Bader
10
showed that 15% dynamic strain applied to bovine chondrocytes in agarose at 0.3, 1, or 3 Hz for 48 hours induces a statistically significant increase in glycosaminoglycan (GAG) release into the medium compared with unstrained control values, and that this release decreases with frequency. Loening
Nevertheless, other studies demonstrate that the amount of PG loss may decrease during cyclic loading but increase in the postloading period: Torzilli
Therefore, there is a discrepancy between studies with regard to the effects of collagen damage, mechanical loading, and the combination of both factors on PG loss. Part of these differences may result from an interaction between effects of the severity of collagen damage on one hand, and effects of cyclic mechanical loading on the other hand. The premise is that a disrupted collagen network would result in a more open structure, which facilitates the transport of PGs entangled within the ECM out of the tissue over time. This transport may already occur as a result of passive diffusion but may be enhanced by convection. Sah
Methods
Osteochondral Plug Preparation
A total of 56 osteochondral plugs (diameter = 7.5 mm) were harvested from 22 metacarpal proximal epiphyses of 1-year-old calves using a column drill, as described previously. 21 During drilling and cutting, osteochondral plugs were irrigated with phosphate-buffered saline (PBS) and afterwards they were stored in PBS at −20°C until mechanical testing.
Mechanical Loading
After thawing at room temperature for 1 hour, the osteochondral plugs were allowed to swell freely in 20 mL PBS for 24 hours (
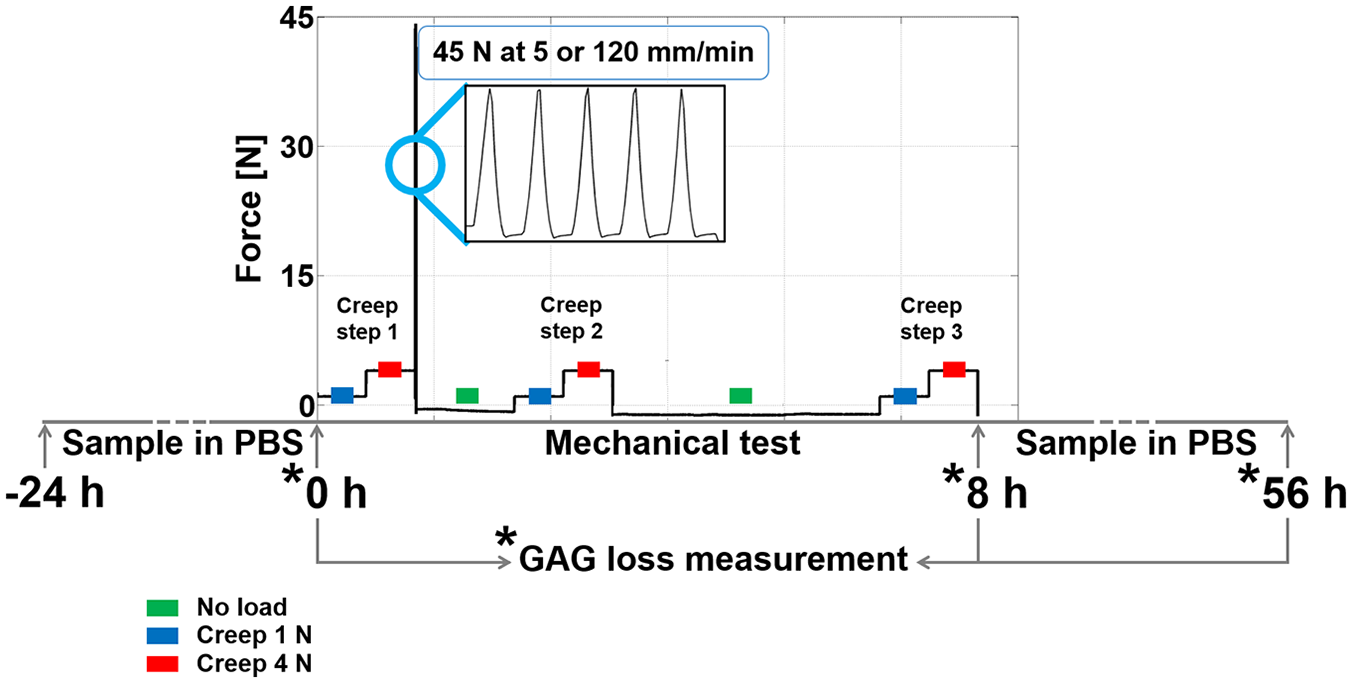
Mechanical testing scheme for the first research question. The 5 ramps up to 45 N were applied to induce collagen damage in articular cartilage. The creep steps at 1 N and 4 N were applied to measure changes in stiffness during the mechanical testing regime. The periods in which cartilage was unloaded were long enough to allow full recovery. The asterisks indicate the time points at which PBS was refreshed and collected to measure the amount of PG loss in media. The total test duration was 8 hours.
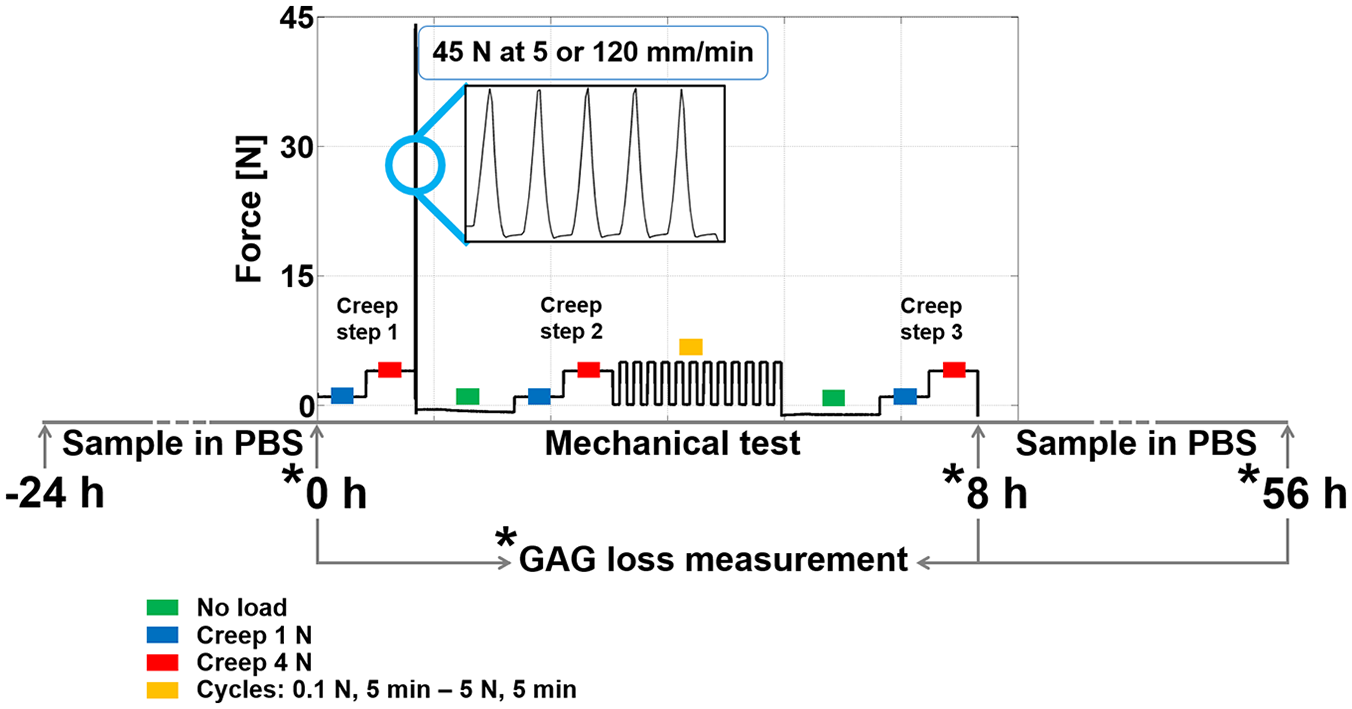
Mechanical testing scheme for the second research question. The 5 ramps up to 45 N were applied to induce collagen damage in articular cartilage. The cyclic loading consisted of 12 low-frequency cycles applied for approximately 2 hours. The creep steps at 1 N and 4 N were applied to measure changes in stiffness during the mechanical test regime. The periods in which cartilage was unloaded were long enough to allow full recovery. The asterisks indicate the time points at which phosphate-buffered saline (PBS) was refreshed and collected to measure the amount of proteoglycan (PG) loss in media. The total test duration was 8 hours.
Mechanical indentation experiments were performed with a tensile testing machine (Universal Testing Machine BT1-FB010TND30, nominal force 2.5 kN, Zwick/Roell, Ulm, Germany). A 2-mm-diameter hemispherical impermeable indenter was used for all loading protocols to ensure the effects of loading pertain to the center and not to the cut edges of the samples. All indentations were applied in displacement-control at the displacement rates defined below, until the required force was reached. For the creep steps, the prescribed force was kept constant during the desired loading period.
To address the first research question, on the dependence of PG loss on the amount and type of collagen damage in articular cartilage, overloading-induced collagen damage was achieved by ramping 5 times from 0.05 to 45 N at either 5 mm/min (OL5,
Overview of the Conditions Applied per Group. a
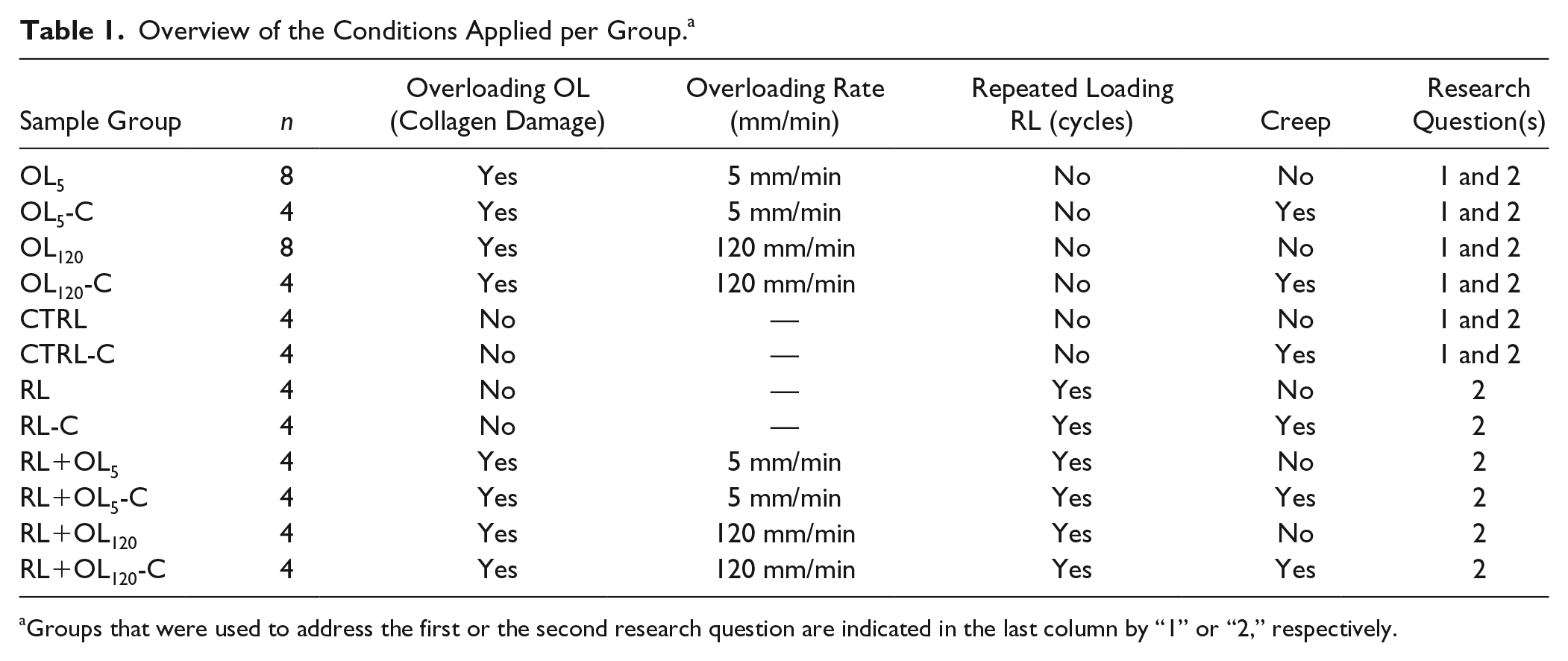
Groups that were used to address the first or the second research question are indicated in the last column by “1” or “2,” respectively.
To address the second research question, on whether repeated loading further increases the already enhanced PG loss in cartilage with collagen damage, the same protocol was used, but with additional repeated loading (RL). A total of 12 RL cycles were applied, each consisting of holding the load at 0.1 N for 5 minutes, ramping up to 5 N at 5 mm/min, and holding at 5 N for 5 minutes (
Fig. 2
). The repeated 5-minute loading periods are long enough to induce convection.
15
Therefore, differences in PG loss between samples that received and did not receive the cyclic compression may be attributed to convection-induced transport of PGs. The RL cycles were applied either in absence of the overloading step (RL,
After 8 hours of mechanical testing, samples were kept in PBS for 48 hours. Subsequently, cartilage was separated from the bone with a scalpel. The cartilage was then cut longitudinally through the middle of the indented area. One half was embedded in Tissue-Tek compound (Sakura Finetek, Torrance, CA, USA, Inc.), rapidly frozen in liquid nitrogen and stored at −30°C until histological processing. The other half was frozen without prior embedding and was not used in the current study. PBS was collected at
Histology
To detect denatured collagen, 7 µm longitudinal cryosections were stained using col2-3/4m antibody, using a previously described protocol. 21 Briefly, sections were dried for 90 minutes at 37°C, fixed for 5 minutes in 3.7% 0.1 M phosphate-buffered (pH 7.4) formaldehyde and then rinsed extensively in a large volume of PBS. Sections were dipped in 0.1% tween PBS incubated with 1% hyaluronidase (testicular, Type I-s, EC 3.2.1.35, Sigma-Aldrich, St. Louis, MO, USA) for 30 minutes at 37°C to enhance the permeability of the extracellular matrix by removing PGs. Afterwards, they were incubated in 10% normal horse serum for 30 minutes to block nonspecific staining and incubated overnight at 4°C with 1/20 col2-3/4m antibody. The next day, sections were incubated in biotin-labeled horse anti-mouse antibody (1/400, IgG (H + L), produced in horse, Vector Laboratories, Inc., Burlingame, CA, USA) for 1 hour at room temperature. Finally, they were incubated with streptavidin 555 reagent (streptavidin, Alexa Fluor 555 conjugate, Invitrogen, Waltham, MA, USA) for 30 minutes. To detect nuclei, they were counterstained with 1:1000 DAPI (Thermo Fisher, Waltham, MA, USA). After each preparation step, sections were rinsed with PBS. After mounting with mowiol, stained sections were digitized at 10× (Zeiss Axiovert 200M, Carl Zeiss, Oberkochen, Germany).
Glycosaminoglycan Assay
Collected PBS was thawed at 37°C, real volumes were measured and then each tube was centrifuged for 5 minutes at 1000 rpm to separate small pieces of cartilage and bone from GAGs. Then, to concentrate the samples, 4 mL of PBS supernatant was poured into centrifugal filters (Amicon Ultra-4 centrifugal filter units, Merck, Darmstadt, Germany) and centrifuged at 2500 RCF (relative centrifugal force) for 20 minutes. Papain digestion buffer and diluted GAG standard were used to prepare the GAG standards in duplicate. The same amount of papain digestion buffer was added to the filtered PBS. Both standards and samples were digested overnight at 60°C, after which they were centrifuged at 12000 rpm for 5 minutes. 40 μL of the samples and standards were then added in duplicate to a 96-well plate and stained with dimethylmethylene blue (DMMB), pH 3.0, to quantify GAGs in media using photospectroscopy according to the protocol of Farndale, described in more detail in Farndale
Data Processing and Statistics
The severity of the superficial and internal collagen damage each received a score between 0 and 5, such that the total collagen damage ranged from 0 to 10. This scoring system was developed and validated previously for use with col2-3/4m-stained cartilage. 21 In short, in the digitized col2-3/4m-stained sections, superficial damage was scored based on visible macroscopic irregularities at the cartilage surface, while microscopic internal damage was scored based on the area covered by the staining. Superficial damage was scored 0 in case of a perfectly smooth surface, 1 in case of macroscopic mild roughening of the surface, and 5 in case clefts penetrated through the cartilage and reached the bone. Internal damage was scored between 0 and 4 depending on the size of the area stained with the col2-3/4m probe, and 1 point was added to the score if the staining advanced into the cartilage surface.
During the creep steps, force and displacement values were recorded. The deformed height Δ
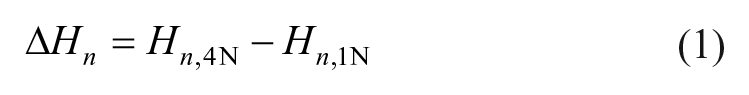
Using this equation, the stiffness

with Δ
Spearman’s rank-order correlation was used to assess the relationship between the amount of PG loss and the grade of collagen damage. A visual inspection of the scatterplot was performed to evaluate whether the relationship was monotonic. An independent-samples
A 2-way analysis of variance (ANOVA) was used to examine the effects of loading condition (i.e., “cycles” vs. “no cycles”) and collagen damage (i.e., “damage” vs. “no damage”). Residual analysis was performed to test for the assumptions of the 2-way ANOVA. Outliers were assessed by boxplot inspection, normality was assessed using Shapiro-Wilk’s normality test, and homogeneity of variances was assessed by Levene’s test.
A 2-way mixed ANOVA was used to determine whether there was an influence of the interaction between loading condition (i.e., “cycles” vs. “no cycles”) and the 3 creep steps on changes in stiffness. Outliers were assessed by boxplot inspection, normality was assessed using Shapiro-Wilk’s normality test, homogeneity of variances, and covariances were assessed by Levene’s test and Box’s
Results
Examples of different categories of collagen damage are shown in Figure 3 .
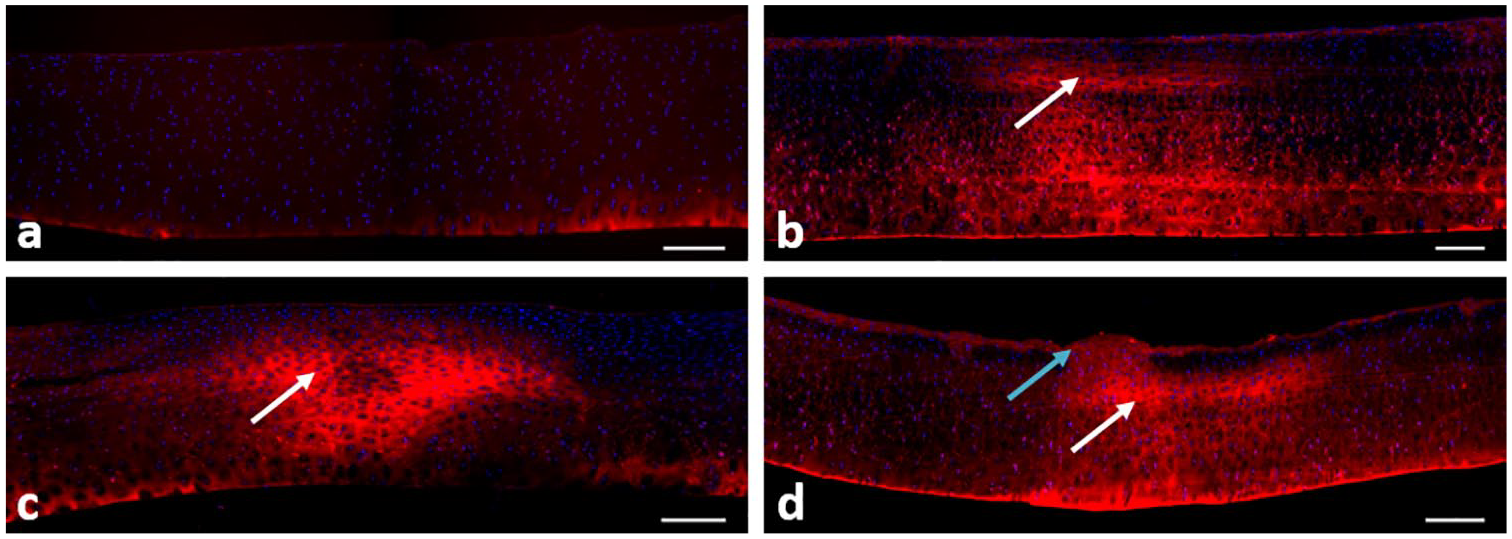
Typical examples of samples stained with col2-3/4m for collagen damage (stained areas) and DAPI for cell nuclei (background dots): (
The total damage score in loaded samples ranged from 0 (no damage) to 5.5, with damage occurring both at the surface and internally (as shown by col2-3/4m staining). Superficial damage was mild and ranged between 1 and 2. The maximum internal damage score was 3.5, meaning that more than 50% of the cartilage between articular surface and bone stained positive for collagen damage. Control samples never stained for internal damage, but mild irregularities were visible at the surface of most samples ( Fig. 4 ).
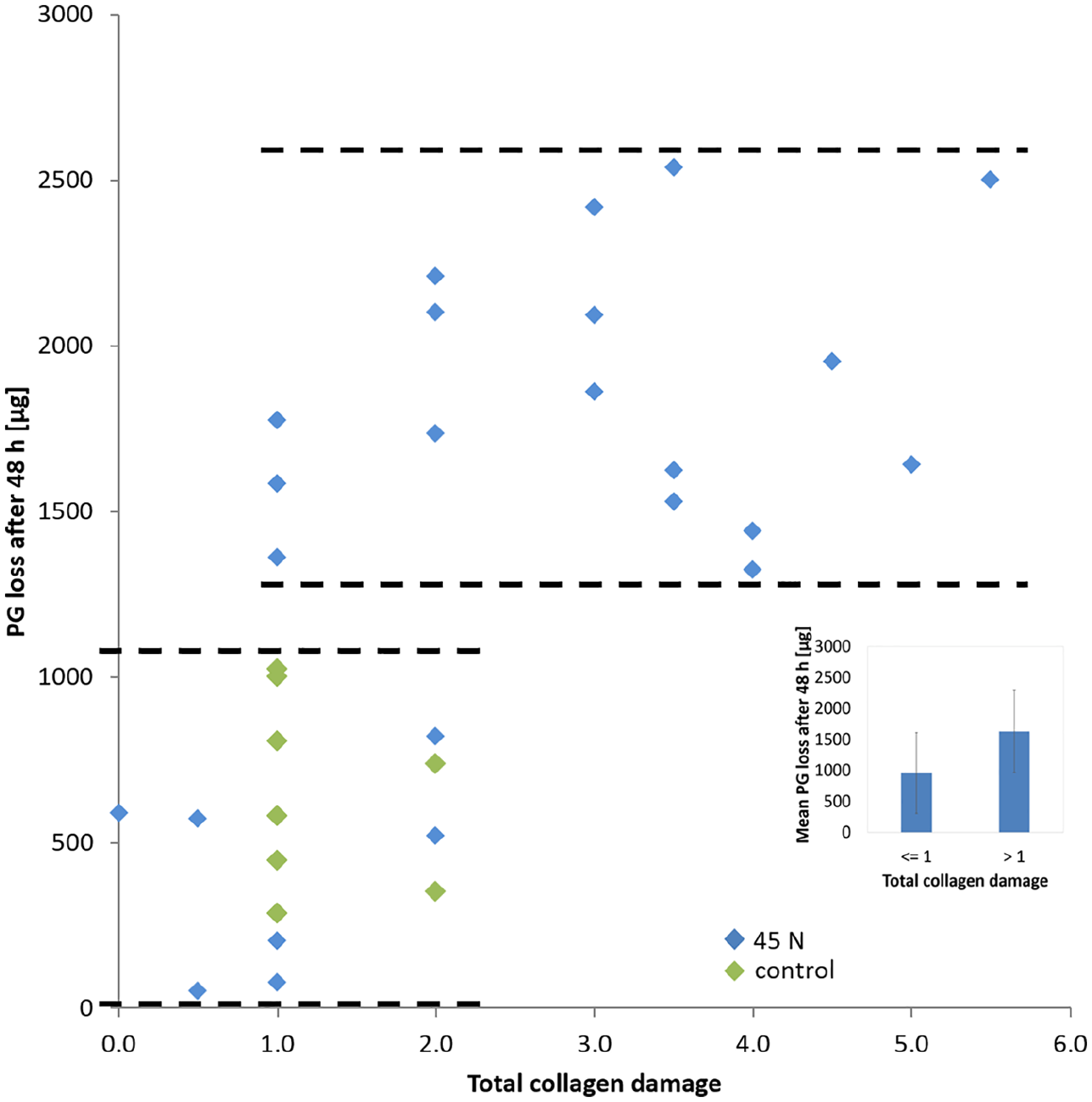
Scatter plot of total collagen damage and PG loss after 56 hours. Markers corresponding to overloaded samples at 45 N applied at either 5 or 120 mm/min and markers corresponding to control samples that did not receive the 45 N load are indicated in the legend.
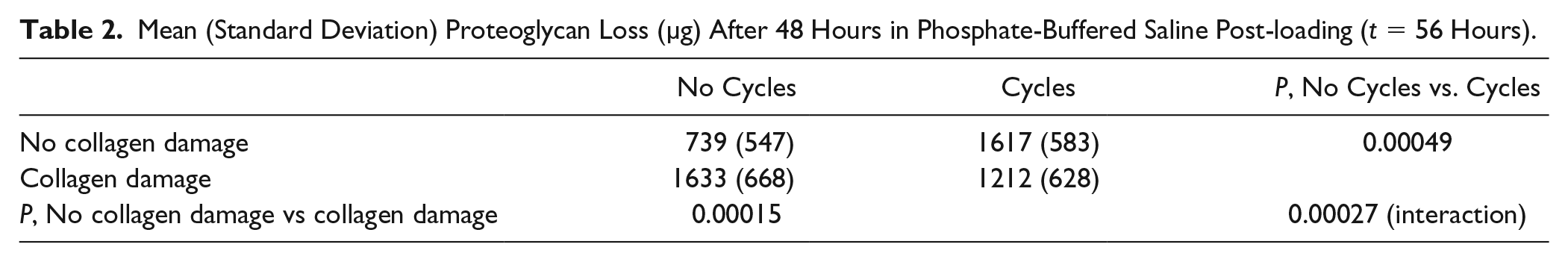
With regard to the first research question of the study, on the relationship between PG loss and collagen damage, Spearman’s rank-order correlation revealed a statistically significant correlation between PG loss after 8 hours of mechanical testing (
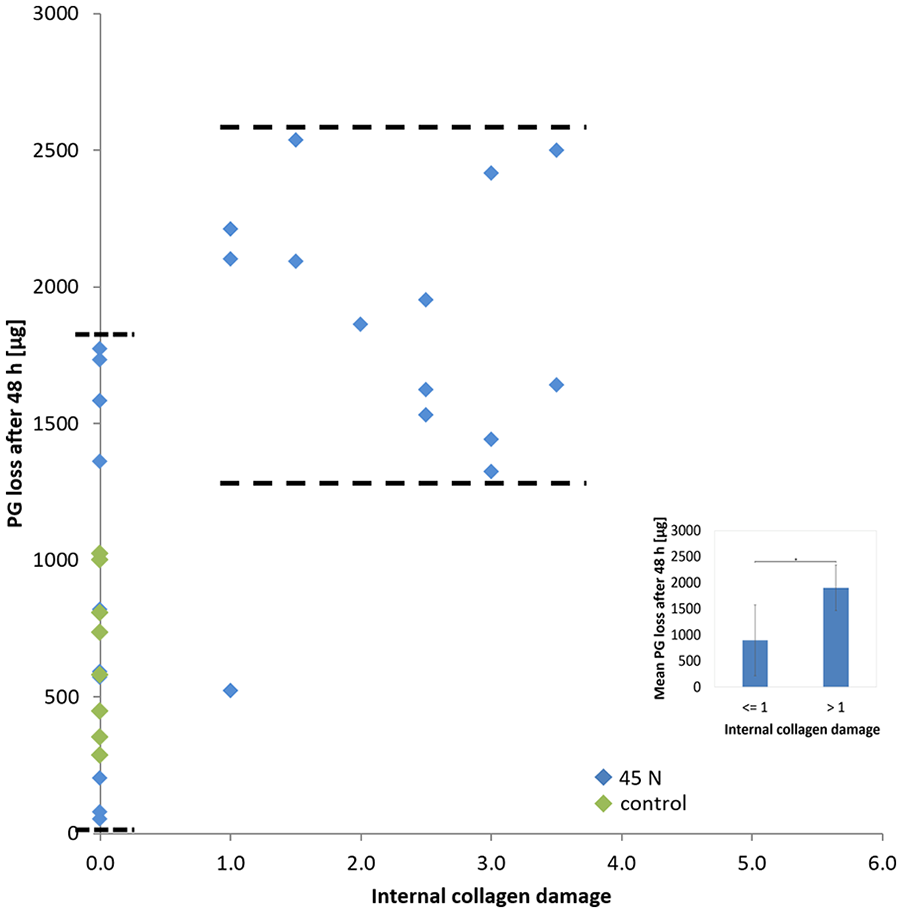
Scatter plot of internal collagen damage and PG loss after 56 hours. Markers corresponding to overloaded samples at 45 N applied at either 5 or 120 mm/min and markers corresponding to control samples that did not receive the 45 N load are indicated in the legend.
As concerns to the second research question of the study, on the effect of repeated loading (cycles) on the already enhanced PG loss in cartilage with collagen damage, 2-way ANOVA showed an interaction between loading condition (i.e., cycles/no cycles) and collagen damage (i.e., damage/no damage;
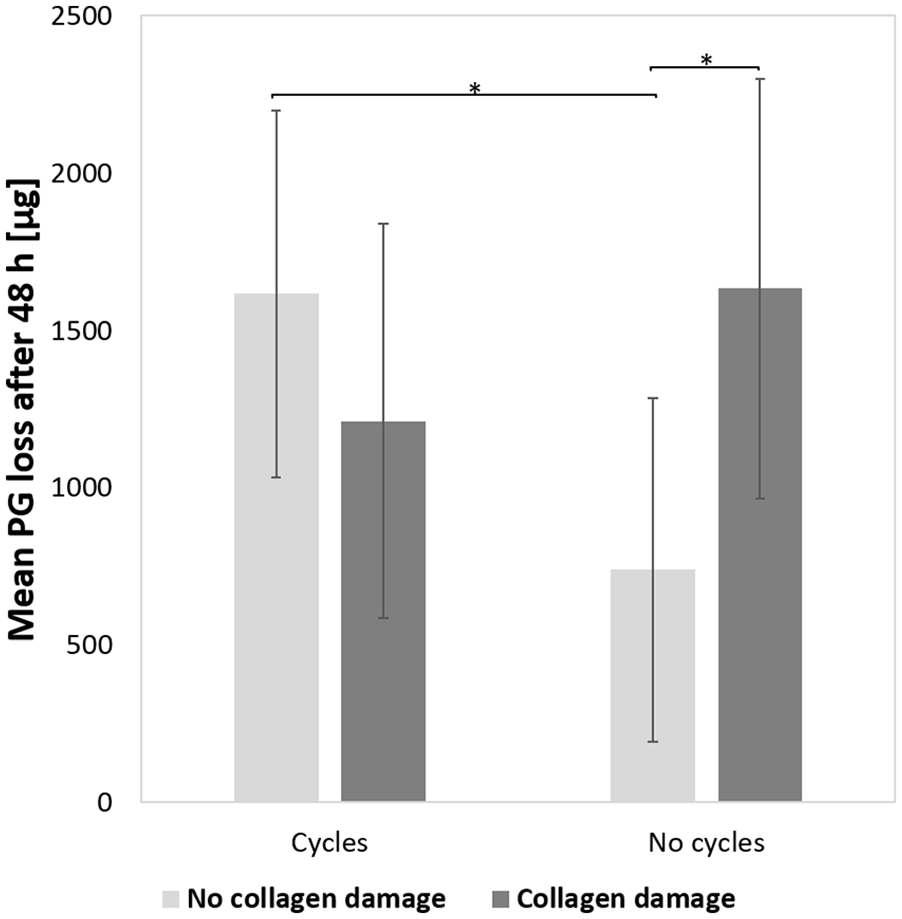
Effects of loading condition (cycles vs. no cycles) and collagen damage (damage vs. no damage) on proteoglycan (PG) loss after 48 hours in phosphate-buffered saline (PBS) post-loading (
Mean (Standard Deviation) Proteoglycan Loss (µg) After 48 Hours in Phosphate-Buffered Saline Post-loading (
Finally, the change in mechanical properties of the samples during the experiment was monitored using the three 1 N/4 N creep steps. Initial cartilage layer thickness at
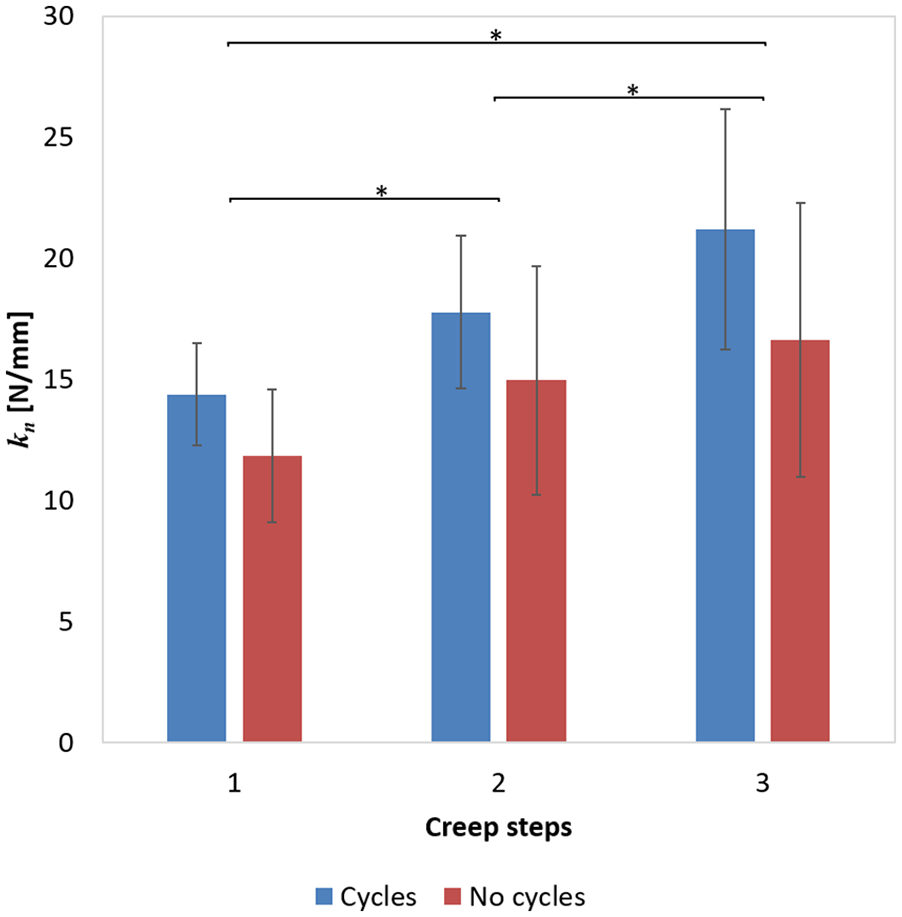
Stiffness as calculated between the low (1 N) and high (4 N) displacement equilibration portion during the first, second, and third creep steps, according to Equation (2).
Discussion
The aims of the present study were (1) to assess whether there is a relationship between total PG loss and the amount and type of collagen damage in articular cartilage and (2) to determine whether cyclic loading further increases the already enhanced loss of PGs in cartilage with collagen damage. The premise was that repeated loading would enhance the transport of PGs out of the tissue through convection, in addition to the diffusive transport due to a damaged collagen network. In one subset of cartilage samples PG loss was measured at different time points and correlated with mechanically induced collagen damage, while in another subset loading cycles were additionally applied to investigate the effect of damage and repeated loading on this loss. Changes in mechanical properties of the tissue were monitored in terms of cartilage stiffness, measured by creep indentation.
The results showed that, 48 hours postloading, PG loss was increased in cartilage samples that had more total collagen damage. Interestingly, PG loss was dependent on the presence, but not the amount of internal damage. Furthermore, the study revealed that repeated compressive loading alone significantly enhanced PG loss in cartilage with intact collagen, supporting the hypothesis that PG transport and loss depend on convection. However, it was shown that once PG loss is increased because the collagen network is damaged, it is not further enhanced by cyclic compression. This suggests that the enhanced diffusivity of PGs in cartilage with mild collagen damage may dominate over the potential effect of increased convection induced by the repeated loading protocol applied in this study.
Taken together, the results may be interpreted as follows. If the collagen network in cartilage is damaged, PGs may leave the tissue. Under unloaded conditions this effect may be attributed to enhanced diffusivity through a more open collagen network. The presence of damage to the internal collagen network, as demonstrated herein with col2-3/4m staining, more than doubles the amount of PGs that is released in the culture medium. Because this internal collagen damage starts in the intermediate or high deep zone, it may be speculated that PGs from these zones may find their way to the surface and leave the cartilage. However, this effect was not significant enough to become visible in histological slides stained with Alcian blue (data not shown). Indeed, assessing the PG content in tissue has been reported to be less sensitive than quantifying PG loss in media. 17 Furthermore, total PG loss increased with total collagen damage, suggesting that although there is no significant correlation between surface damage and PG loss, damage to the superficial zone may allow more PGs to leave the cartilage.
If PGs from deeper zones are to be transported to the surface, it may be expected that convection would stimulate such transport, in particular because PGs are relatively large molecules.
25
Interestingly, however, this is not the case, as PG loss from damaged cartilage is not different between repeatedly loaded and free swelling samples. One explanation may be that the damage already increased the diffusivity of the PGs to such extent that either the diffusion overrules any additional effects of convection, or the increase is negligible and falls within the variability of the measurements. Another explanation may be found in the compaction of the cartilage after loss of PGs in combination with the cyclic compressive loading. Indeed, indentation was significantly deeper after the 4 N holding period in the first creep step than in the second and third, but the difference in indentation depths were only 4.2% between the first and second step (
These findings are largely in line with previous literature. PG loss was often reported in combination with collagen network damage, reflected as changes in cartilage shape,
27
fissures or cracks,28,29 internal cleavage,
8
or denaturation.11,30 Enzymatic degradation of cartilage discs with collagenase for different periods of time resulted in loss of PG content from the cartilage zones where collagen cleavage occurred.
8
In agreement with our results, osteochondral plugs subjected to unconfined compression showed more macroscopic damage (i.e., cracks) and an increase in PG loss with higher stresses.
29
PG loss as a result of cyclic loading was reported by Loening
With regard to the effects of cyclic loading, the enhanced PG loss in healthy cartilage observed here has also been reported by various studies.5,11,12,14,20,30 Using even lower frequency cycles (2 hours on–2 hours off) than those applied in the present study, Sah
Measured tissue stiffness significantly increased from the first to the second and third creep steps (
A high variability in PG loss between samples was observed in the current study. Some samples that were not graded as being damaged lost over 1000 µg of PGs, while some damaged samples lost as little as 521 µg of PGs. This may be a sample-dependent effect, but it is also possible that a type of micro-damage occurred in the collagen fibrils that is not detectable with col2-3/4 antibody, yet still stimulates PG release. 18
Another important consideration in the interpretation of the results presented herein is the age of the animal from which the tissue was harvested. Compared to immature bovine cartilage (e.g., 4- to 8-week-old calves), in adult bovine cartilage (e.g., 3- to 5-year-old steers) both PGs and chondroitin sulfate–rich regions become smaller,
33
which might directly affect the fixed charge density and GAG content of the tissue and, consequently, its compressive properties.
34
In human articular cartilage, a rapid decline of GAG content with age has been observed: When body growth is complete, the content of GAG (dry weight) in the superficial zone of cartilage is about 15%, amount which decreases during aging and reaches about 7% by the age of 70 years.
35
Immature cartilage from 8-week-old rabbits was shown to contain more GAGs and to have a higher wet weight and hydrodynamically larger chondroitin sulfate–rich PG than mature cartilage, therefore acting as an elastic cushion of water which protects the bone surface.
36
As concerns the collagen network, its structure and content differ depending on the specimen age. Changes of the collagen network during growth and maturation in pigs include a decrease in collagen content and gradual changes in collagen fibril orientation from parallel to the cartilage surface throughout the uncalcified tissue in 4-month-old animals to a classical Benninghoff architecture structure in 21-month-old animals.
37
Importantly, the development of articular cartilage is different at different joint surfaces, as was observed in the findings of Julkunen
The samples used for the present study were harvested from 1-year-old cows. Histology reveals an arcade-like collagen orientation, indicating that cartilage stiffness and resistance to deformation may be close to those of adult bovine tissue. However, cows at this age have probably not developed fully mature cartilage yet. For instance, Otsuki
Finally, it is possible that the overloading used in the present study damaged not only the collagen network but also the PG molecules. This would have resulted in smaller PG fragments, which could then have diffused faster through the tissue even in an intact collagen network. Theoretically, such effect may have produced a similar type of results. Therefore, determination of the distribution of PG fragment size in the collected PBS is advised in future studies.
To conclude, PG loss increases with the amount of total collagen damage, and in the presence of internal collagen damage PG loss is considerably enhanced. This suggests that collagen damage increases the diffusivity of PGs in cartilage and that not only superficial PGs but also those residing in deeper zones may leave the cartilage through enhanced diffusion. The repeated loading applied in the present study enhanced PG loss in cartilage without collagen damage, suggesting a significant impact of convection in the transport of PGs. However, it did not further stimulate PG loss in damaged cartilage, in which PG loss is already enhanced without cyclic loading. This suggests that either enhanced diffusion in damaged cartilage overrules the effects of convection, or that tissue compaction due to mechanical loading counteracts the increased permeability of the open collagen network.
Footnotes
Acknowledgments and Funding
The authors would like to thank Marina van Doeselaar for her valuable help with the laboratory procedures of collagen staining and quantification of PG loss. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the grant program “Programa de Formación Doctoral Francisco José de Caldas Generación del Bicentenario” awarded by the Francisco José de Caldas Institute for the Development of Science and Technology (COLCIENCIAS), LASPAU ID 20110352.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not sought for the present study because experiments were performed on bovine explants obtained from the slaughterhouse after animal sacrifice.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because only slaughterhouse waste products were used.
