Abstract
Objective
This study aims to (1) determine and validate living cartilage allograft transplantation as a novel source for viable osteochondral allograft (OCA) tissues and (2) perform histologic and viability comparisons of living donor cartilage tissues to currently available clinical-grade standard processed grafts.
Design
Using healthy cartilage from well-preserved contralateral compartments in 27 patients undergoing total knee arthroplasty (TKA) and 10 clinical-grade OCA specimens obtained immediately following operative implantation, standard and living donor OCA quality was evaluated at the time of harvest and following up to 3 weeks of storage on the basis of macroscopic International Cartilage Repair Society grade, histology, and viability.
Results
Osteochondral samples demonstrated a consistent decrease in viability and histologic quality over the first 3 weeks of storage at 37°C, supporting the utility of an OCA paradigm shift toward early implantation, as was the clinical standard up until recent adoption of transplantation at 14 to 35 days following donor procurement. Samples from the 10 clinical-grade OCAs, implanted at an average of 23 days following graft harvest demonstrated a mean viable cell density of 45.6% at implantation, significantly lower (P < 0.01) than the 93.6% viability observed in living donor allograft tissues.
Conclusions
Osteochondral tissue viability and histologic quality progressively decreases with ex vivo storage, even when kept at physiologic temperatures. Currently available clinical OCAs are stored for 2 to 5 weeks prior to implantation and demonstrate inferior viability to that of fresh osteochondral tissues that can be made available through the use of a living donor cartilage program.
Introduction
Osteoarthritis remains a central challenge of orthopedics, affecting more than 1 in 4 American adults and more than 300 million patients worldwide.1,2 Focal articular cartilage defects are common in young patients and play a key role in disrupting joint homeostasis and driving the inflammation- and degradation-based pathogenesis to generalized osteoarthritis. 3 In addition to significant long-term sequelae, focal cartilage defects can cause acute pain and disability similar to that of severe arthritis. 4
Articular cartilage defects have limited reparative potential due to the poor inherent regenerative capacity and the avascular nature of cartilage. Therefore, articular defects have been classically treated with surgical interventions, including microfracture, cell-based therapies such as autologous chondrocyte implantation, and tissue-based therapies such as osteochondral allograft (OCA) transplantation. 5
First described as early as 1908, OCA has become the gold standard for the treatment of cartilage defects greater than 2 cm2, with well-established safety, efficacy, and durability.6-12 The use of osteochondral tissue allows for the simultaneous transplantation of cartilage and underlying bone, enabling clinicians to address pathology extending beyond the subchondral plate if needed. Using classically obtained fresh allografts, which were often implanted within the first 24 to 48 hours of donor expiration, long-term graft survival rates of 66% to 69% have been described at 20 years of follow-up. 13 Furthermore, in addition to durability, OCA has demonstrated a broad range of clinical applications, with a proven track-record spanning the knee, ankle, hip, shoulder, elbow, and other joints.7,9,14-17
With the emergence of uniform cartilage banking and testing protocols embodied in part by the 2004 adoption of United States Pharmacopeia (USP) <71>, OCA tissues were stored at 4°C for a minimum of 14 days and up to 28 to 35 days following procurement and prior to implantation. 18 Subsequently, it has been well demonstrated that cellular demise, driven by apoptosis and cell stress at subphysiologic temperature significantly deteriorates allograft viability and quality, even within the first 14 days of storage.19-21 Alternative storage methods at room temperature (22°C to 25°C) and physiologic 37°C temperatures have subsequently been proposed and demonstrated to be superior to refrigerated storage, as is current clinical practice.22,23 Despite this, no direct comparison of alternative cartilage storage and sourcing has been made to clinical-grade tissues, which are currently poorly characterized given their high cost and relative rarity. Accordingly, many previous studies employed caprine and canine cartilage, which may possess different storage and viability profiles as compared with human tissues.
Therefore, the central limitation of increasing clinical implementation of gold standard OCA is that allografts are currently obtained from young deceased donors, leading to logistical scheduling challenges and lack of scalability of this efficacious resource. The purpose of this study was to (1) determine and validate living cartilage allograft transplantation as a novel source for viable OCA tissues and (2) perform histologic and viability comparisons of living donor cartilage tissues to currently available clinical-grade standard processed grafts.
Method
Tissue Collection and Processing
Joint resections were collected from young patients (<60 years) undergoing total knee arthroplasty (TKA) for varus or valgus pathology with well-preserved contralateral compartments demonstrating Kellgren-Lawrence grade 0 or 1 pathology. Patients with grade 2+ pathology in the preserved compartment (lateral for varus, medial for valgus) were ineligible for tissue collection and subsequent analysis. Screening was performed on the basis of preoperative TKA templating radiographs, without further advanced imaging or additional formal criteria for meniscus and ligamentous pathology. Importantly, the decision for TKA was made clinically and independently of subsequent osteochondral tissue donation, with radiographs of listed TKA patients screened to determine the presence of a minimally affected compartment appropriate for cartilage characterization. Twenty-seven young TKA donors (16 males, 11 females, age 56.2 ± 3.3 years) were screened using standard American Association of Tissue Banks (AATB) tissue donation criteria and were eligible for inclusion in this study. In addition, 10 femoral OCA specimens, obtained immediately following operative implantation of the harvested osteochondral plug in clinical practice, were collected in order to characterize currently utilized OCA tissues and serve as a point of comparison with TKA tissues (i.e., living donor cartilage). Therefore, a total of 37 osteochondral samples were analyzed. All aspects of this study were performed following institutional review board approval (IRB 13-005619).
On collection of the living donor TKA joint resections and deceased donor OCA samples, photos were taken to enable gross observation and scoring of the tissues according to the International Cartilage Repair Society (ICRS) grading system. 24 Subsequently, osteochondral samples were harvested from each sample by the creation of 4 mm cartilage discs using biopsy punches for use in histologic and cellular-level characterization. Cartilage discs were stored in 2 mL of serum-free osteochondral media at 37°C with hypoxia (2.0 + 0.5% O2) for experimental time points ranging from 1 to 4 weeks following harvest, with media changes every 7 days.
Histologic Tissue Characterization
Discs undergoing histologic analysis were transferred to 10% neutral buffered formalin for preservation. After 24 hours of fixation, samples were transferred to 70% ethanol for storage prior to embedding. Tissues were subsequently bisected and embedded in paraffin and sectioned (5 μm) along a vertical plane to get cross-sectional views simultaneously demonstrating both the superficial and deep aspects of the osteochondral samples. Slides were stained for hematoxylin-eosin (H&E) and safranin-O using standard methods.
Additional sample sections underwent immunohistochemical staining for aggrecan (mouse anti-aggrecan antibody, Novusbio NB110-6524, dilution 1:150 in phosphate buffered saline/bovine serum albumin [PBS/BSA] 5%), collagen I (rabbit monoclonal anti-Collagen 1, Abcam EPR7785, dilution 1:400 in PBS/BSA 5%) and collagen II (mouse monoclonal anti-Collagen II, DSHB, University of Iowa, dilution 1:100 in PBS/BSA 5%) with normal mouse or rabbit IgG used as a negative controls.
Cell Viability Quantification
Osteochondral discs were assessed for cell viability using a 2-color fluorescence assay based on the simultaneous determination of living (calcein acetoxymethyl [AM]: green) and dead cells (ethidium homodimer-1: red) (live-dead viability/cytotoxicity kit for mammalian cells; Molecular Probes). Fluorescent 3-dimensional confocal images (850 µm × 850 µm × ~100 µm; 7.2 µm slice thickness) were collected using an inverted LSM 780 multiphoton laser scanning confocal microscope (488 nm and 561 nm lasers) at 10× magnification. Maximum intensity projections were subsequently created in Zen (2.3 SP1, Zeiss 2015) using a threshold value of 25. The amount of red and green in each image was quantified objectively in an automated and independent process using MatLab (R2015b, 8.6.0.267246) to provide red and green pixel counts. Viable cell density (VCD) was calculated by dividing the number of green (live) pixels by the sum number of green (live) and red (dead) pixels in order to provide a quantitative and nonsubjective measure of percentage viable cells.
Microbiologic Testing
Culture-based microbiological testing was performed by a dedicated microbiologic laboratory (Mayo Clinic, Rochester, MN) by placing osteochondral samples into 35 mL of tryptic soy broth followed by vortexing and allowing the sample to mix for 10 minutes. Subsequently, 0.1 mL of culture was placed onto 2 sheep blood agar plates, 1 incubated at 37°C under aerobic conditions (21% O2, 5% CO2) and the other placed in 37°C anaerobic culture. In addition, for each sample, 8 mL of inoculated, mixed tryptic culture was placed into a sealed sterile container for anaerobic broth culture alongside the anaerobic plate specimen. Samples were monitored for 2 weeks for microbiological growth. Additionally, bacterial endotoxin (BET) quantification was performed using kinetic turbidimetric testing employing good laboratory practice (GLP) principles and test validation by an independent, third-party laboratory (Nelson Laboratories, Salt Lake City, UT) in accordance with Food and Drug Administration (FDA) standards.
Statistical Methods
Statistical analysis was performed by 2 formally trained statisticians. Histologic measurements and cell viability comparisons were made between groups using generalized linear models (GLM) and Kruskal-Wallis rank-sum testing. Multigroup P values were adjusted for the number of comparisons performed during GLM modeling using the methods described by Benjamini and Hochberg (1995), 25 P values <0.05 were considered significant. Statistics were performed using SAS 9.4 and JMP Pro 13.0 (SAS Institute, Cary, NC), and R Version 3.4.3 (R Core Team, Vienna, Austria).
Results
Physiologic Storage of Osteochondral Tissues
Baseline VCD at the time of living donor TKA sample harvest was 91.8% ± 8.5%. Decreasing viability over time was observed, with average VCD of 86.1% ± 16.9% at week 1, 86.4% ± 14.9% at week 2, and 72.5% ± 15.6% at week 3 (P = 0.01 for day 0 vs. week 3,
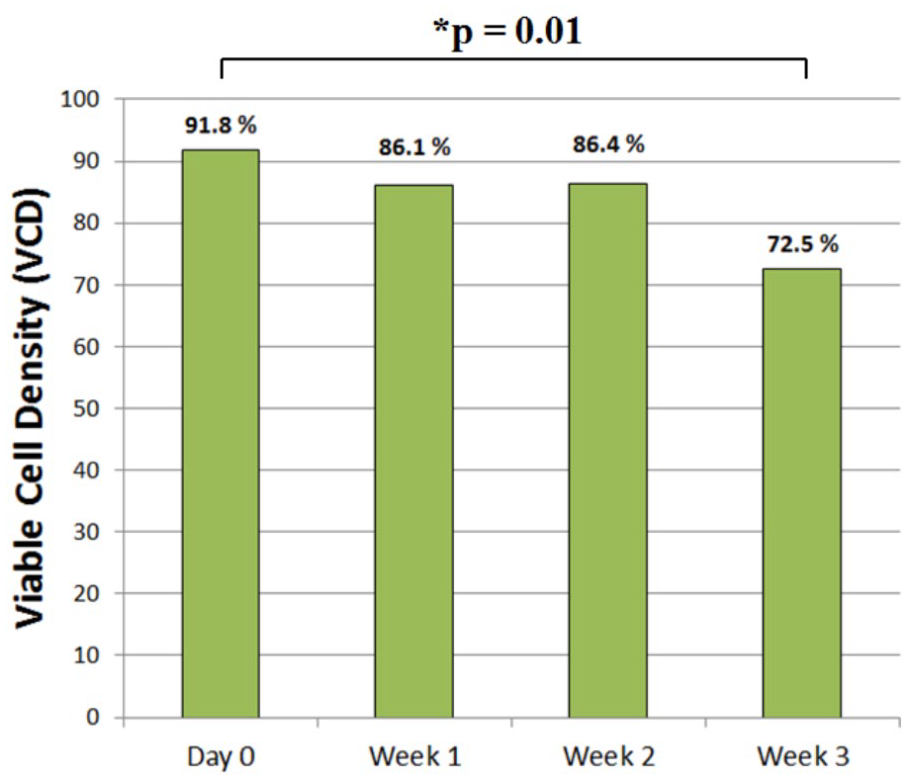
Osteochondral allograft viability over storage time. Viable cell density (VCD) of well-preserved total knee arthroplasty (TKA) compartment tissues demonstrated significant (P = 0.01) decreases in VCD over the course of 3 weeks of physiologic (37°C + hypoxia) storage.
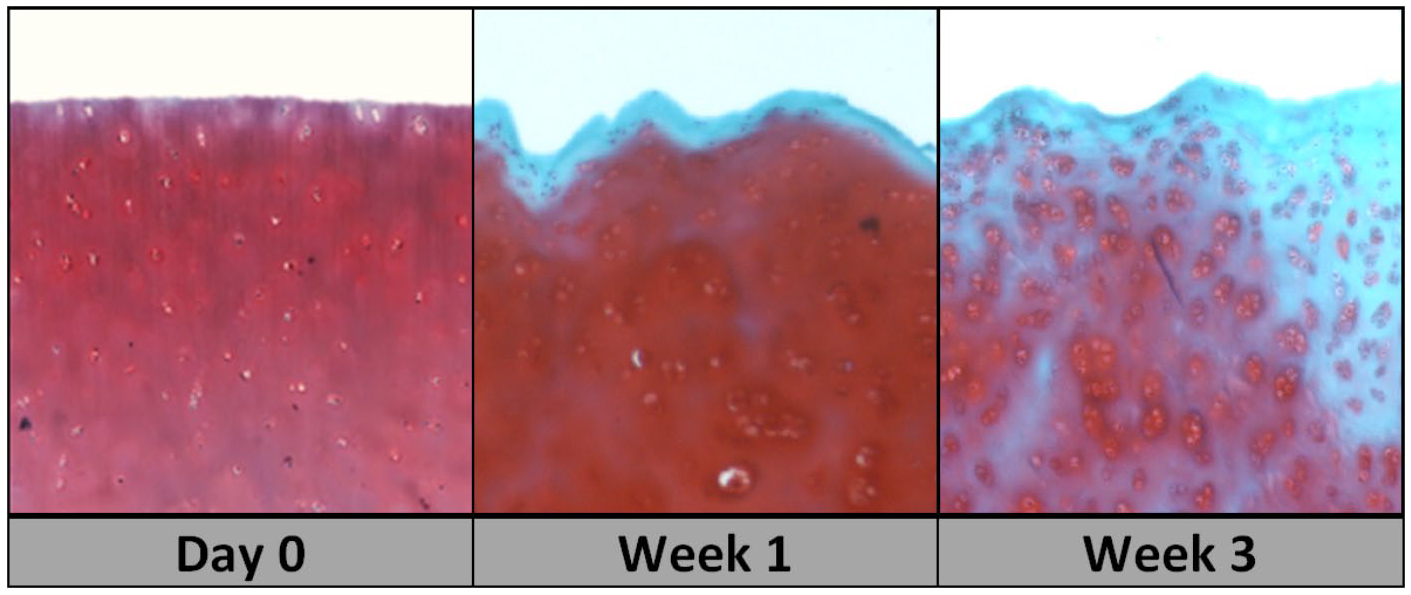
Osteochondral allograft histology and glycosamine staining over storage time. Representative images of decreasing safranin-O staining (red), indicative of decreasing glycosaminoglycan content at articular surface of stored cartilage samples over 3 weeks with substituting fibrosis (light blue) and associated atypical chondrocyte hypertrophy.
Living Donor Cartilage Compared with Clinical Grade Fresh-Stored OCA
The average age of clinical OCAs (
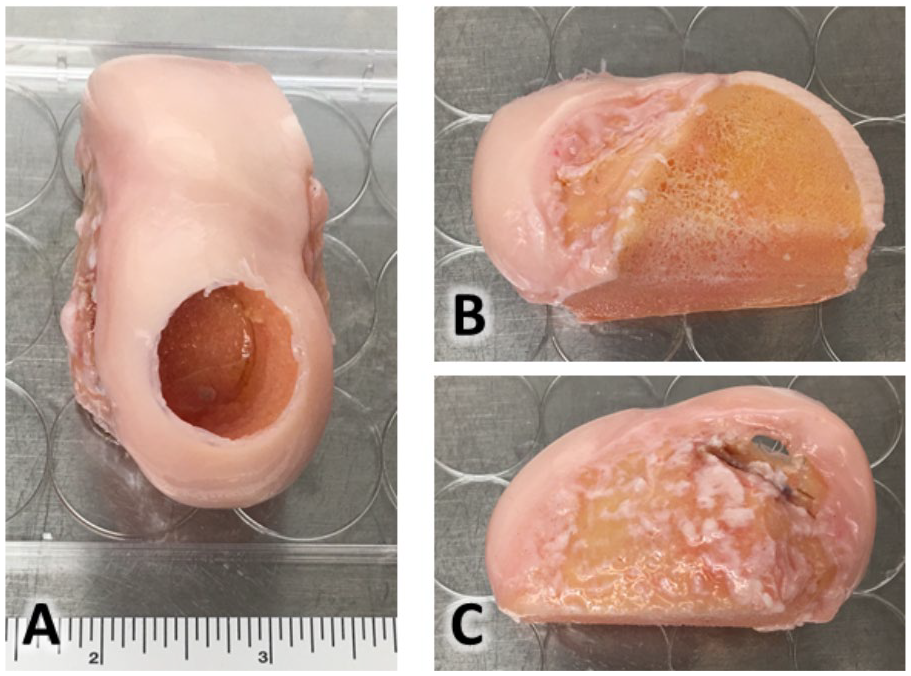
Clinical grade osteochondral allograft sample morphology. Medial femoral condyle osteochondral allograft (OCA) obtained immediately after clinical graft preparation and implantation (visualized as area of previously excised OCA plug) with (
Macroscopic Comparisons
Living donor cartilage from unaffected TKA compartments exhibited healthy macroscopic architecture at the time of harvest, with median ICRS grade 0 pathology (range 0-2). Patients without uniform grade 0 pathology demonstrated focal ICRS grade 1-2 defects, which were readily avoidable in both 4 mm disc and future allograft plug preparation. In contrast, the contralateral articular compartment demonstrated severe degenerative changes, with median ICRS grade 4 pathology (range 3-4), as would be expected in the case of arthroplasty patients (
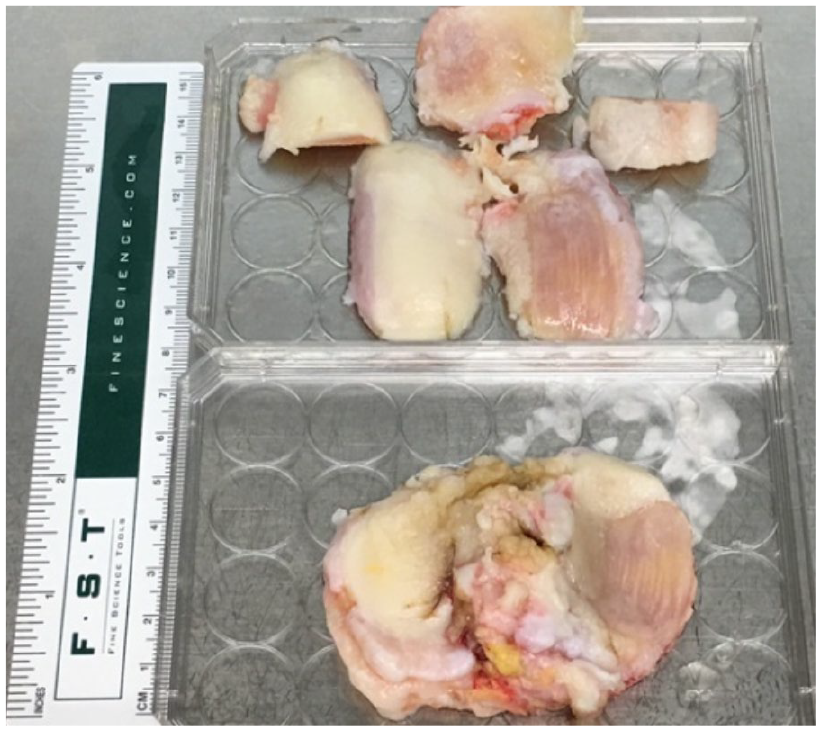
Living donor cartilage allograft sample morphology. Representative living donor osteochondral sample demonstrating full thickness medial compartment cartilage loss (eburnated bony tissues on right side) with a well-preserved lateral compartment (white articular surface on left). The resected articular surfaces are displayed in anatomic positioning as femoral and patellar cuts on the superior aspect of the image and the tibial cut at the bottom of the image.
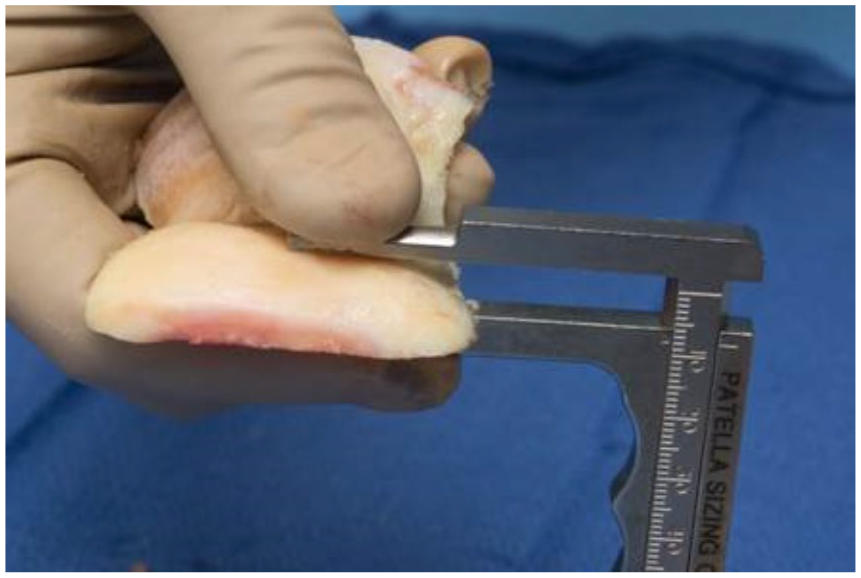
Intraoperative measurement of living donor allograft sample thickness. Graft thickness is measured using sterile calipers (measured here as 8 mm) for living donor cartilage allograft osteochondral sections obtained at the time of total knee arthroplasty from a well-preserved lateral femoral condyle.
Living Donor Cartilage Histology
Living donor cartilage allograft samples exhibited substantial and uniform appearing collagen II staining throughout the superficial and deep cartilage layers, with no staining present on IgG controls (
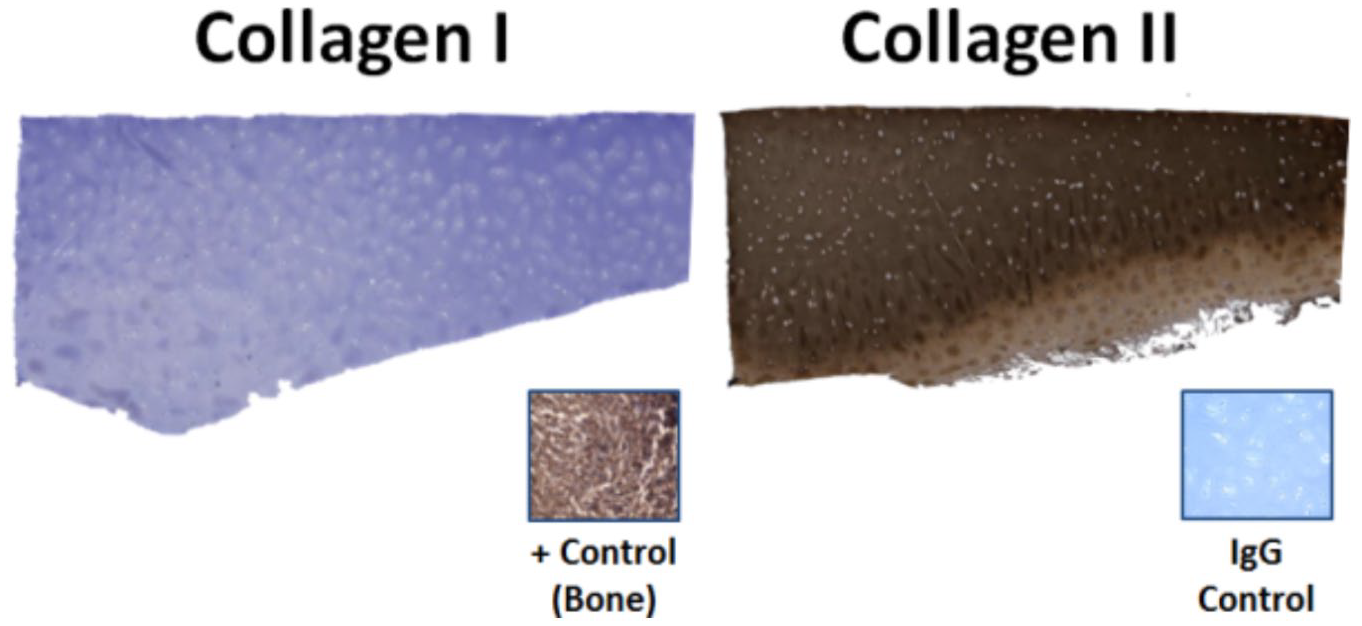
Living donor cartilage histology. The articular surface is oriented upward and displays no visible collagen I staining in contrast to the diffuse and dark staining present for collagen II.
Cellular-Level Comparisons (Live/Dead Analysis)
The mean VCD for all living donor cartilage allograft samples undergoing viability testing at the time of harvest was 93.6% (range 88.4% to 97.8 %). In comparison, the mean VCD for operatively implanted OCA samples was 45.6% (range 2.2% to 90.9%, P < 0.01,
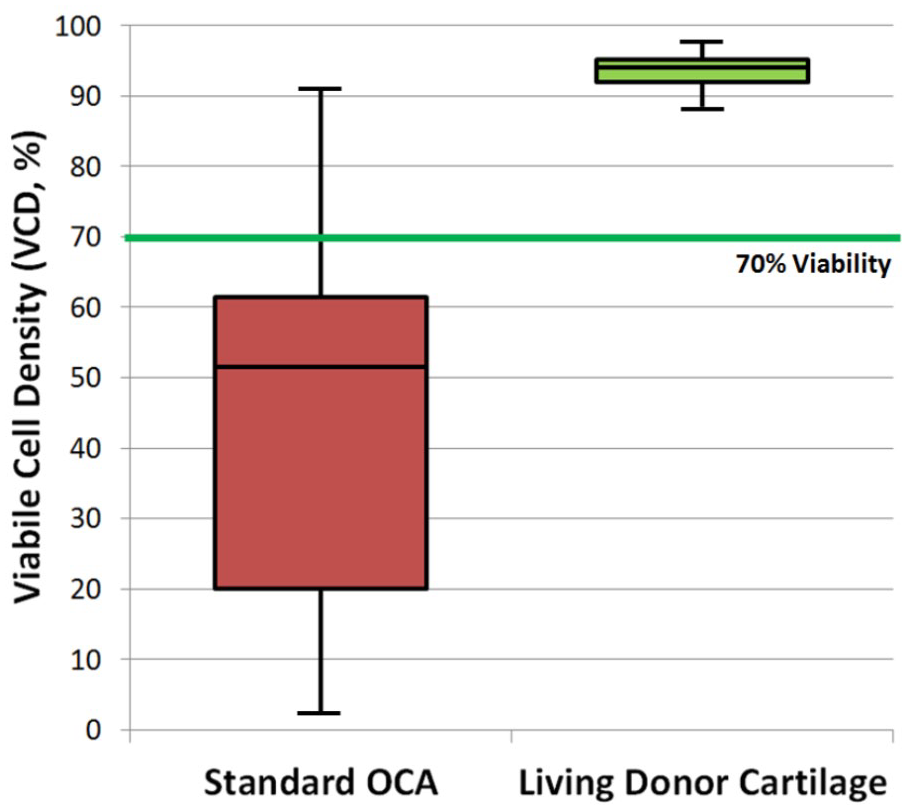
Comparisons of living donor and standard fresh-stored clinical osteochondral allograft viability. The box and whisker plot of OCA and living donor cartilage allograft VCD demonstrates significantly higher and more homogenous VCD values in living donor tissues when compared with current clinically available OCA. Whiskers represent the minimum and maximum values while boxes show the first quartile, median, and third quartile. OCA = osteochondral allograft; VCD = viable cell density.
Living Donor Cartilage Microbiological Proof-of-Concept
Samples for 9 tested living donor cartilage patients were culture negative (0% contamination) on aerobic and anaerobic plates, as well as anaerobic tryptic broth culture at all time points, 0 to 4 weeks following harvest. In addition, in accordance with FDA tissue standards, BET was performed following 2 and 3 weeks of storage for 4 samples as part of a validated, third-party analysis. All BET values in the tests that were performed were under the detection limit of 0.250 endotoxin units (EU) per milliliter, and this stringent threshold is well below the 0.5 EU/mL limit set by the FDA.
Discussion
The main findings of this study are 3-fold: (1) fresh osteochondral samples obtained at the time of TKA demonstrate a consistent decrease in viability and histologic quality during the first three weeks of storage, (2) decreased viability observed in laboratory-stored samples are also observed in clinical grade osteochondral allograft at the time of surgical implantation, and (3) living donor osteochondral allograft from relatively well-preserved compartments at the time of TKA demonstrates satisfactory graft viability and histology when compared with OCA samples from current clinical practice. These findings are substantial given the recent shift in delayed OCA implantation at 14 to 35 days following donor procurement to permit further sterility testing which has yet to be accompanied by assurance or disclosure of graft viability. 18 Furthermore, employing living donor transplantation from the time of TKA has the potential to simultaneously increase viability, ease logistic scheduling, and expand the availability of OCA, supporting the implementation of what is considered the gold standard treatment for large cartilage and osteochondral defects.
Articular cartilage defects and subsequent osteoarthritis remain central challenges of orthopedic surgery, causing significant disability and loss of productivity in a large portion of the global population. 2 Osteochondral allograft has become the gold standard treatment for large focal cartilage and osteochondral defects, and has a long-standing track record in successful joint preservation surgery. However, this resource remains rare due to the limited scalability of young deceased donor based solutions and the importance of viable tissues for clinical success.13,22,26 The purposes of this study were to determine and validate living donor cartilage allograft transplantation as a novel source for viable OCA tissues, and to compare living donor cartilage transplantation to currently available clinical-grade tissues. These aims were achieved by showing that living donor cartilage TKA tissues have the appropriate clinical properties (i.e., macroscopic cartilage grade, cell viability, and microbiological sterility) for use as a novel and potentially improved OCA source as part of a living donor cartilage program (LDCP).
Previous studies have shown that storage of cartilage at room temperature and 37°C is superior over classical refrigerated methods, especially in animal tissues.19-23 However, these storage temperatures have not been evaluated in human tissues, especially over multiple weeks of storage. Our work focused on determining tissue quality under ideal, physiologic temperatures for OCA storage during a clinically relevant time frame by monitoring the biological properties of cartilage. While studies employing intra-articular thermometers at the time of knee exercise have demonstrated that physiologic knee temperature varies on the range of approximately 33°C to 39°C, other investigations into the efficacy of ice- and cryotherapy-based cooling systems have demonstrated that physiologic and even therapeutic temperatures rarely, if ever, approach values near 25°C (i.e., room temperature).27,28 Therefore, it is intuitive that long-term maintenance of osteochondral tissues may be preferable within physiologic and biologic parameters near 37°C, as performed in this study.
Given that physiologic storage offers viability benefits for osteochondral tissues, it is also important to establish the ideal timeline for cartilage implantation following storage. While classic OCA surgeries were performed on the order of hours to days following procurement, sterility testing paradigms have shifted practice toward implantation at 14 to 35 days following graft harvest, without simultaneous testing or validation of tissue viability.6,18,29 These changes raise the concern of decreasing clinical benefit given the established link between tissue viability and graft success. 26 Accordingly, we tested the viability and histology of osteochondral samples under optimal physiologic conditions during storage for up to 3 weeks, which is near the average time of 23 days to OCA implantation at our institution. The observed decreases in viability, as well as significant accompanying loss of glycosaminoglycan content in safranin-O staining, certainly raises the concern of storing tissues on the order of weeks, even in the most optimal and physiologic of conditions.
Previous research by Schmidt et al. 30 has investigated whether storage time influences outcomes of osteochondral allograft transplantation performed after a mean of 6.3 days (early release) versus 20.0 days (late release) of storage at 4°C. While the authors did not find a significant effect of storage time on graft survival, grafts were considered early release for up to 14 days following procurement, which is noteworthy considering that this methodology masks the effects of early graft degradation or loss of viability occurring within the first 2 weeks of storage. Furthermore, as noted by the authors, there is mounting evidence which includes animal data from their institution 31 as well as others 23 demonstrating that refrigerated storage at 4°C performs inferior to more physiologic temperatures such as 37°C in terms of viability. Finally, although their study was well powered with 150 patients, no direct measurements of graft viability were performed. Therefore, outcomes must be interpreted in light of the limitations and potentially dominant effects of nonphysiologic refrigeration and indirect, correlation-based causality between viability and outcomes. 30
In addition to the inherent limitations of scaling allografts obtained from deceased donors, the unexpected passing of donors adds an additional layer of logistical complexity for scheduling OCA transplantation. At the time of this study, our institution, which is a high-volume cartilage center, has 3 patients that have been on OCA waiting lists for greater than 6 months. Such limitations and volumes could be improved by the implementation of LDCPs, considering the high volume of varus and valgus total knee replacements performed in the United States and worldwide and the fact that TKAs are performed on an elective and prescheduled basis.32,33 We have demonstrated that candidate living donors can be successfully screened to provide optimal osteochondral grafts, and that safety testing can be performed immediately prior to tissue donation. Therefore, patients in need of living donor cartilage allograft could also be scheduled in advance to follow operative dates for living donors undergoing TKA. Given that distal femoral OCA costs are on the order of $8,500 to $15,000 per graft, significant cost savings may also result and fund the establishment of the necessary tissue banks, infectious disease screening for donors, and more advanced preoperative testing such as magnetic resonance imaging (MRI) for donors.
To further assess the sterility and safety of tissues harvested in the operating room for use as allograft, the sterility record of our facility Bone Bank, which employs identical personnel and sterile containers as those used in this study to intraoperatively harvest femoral head cancellous allograft was queried. Over the course of the 2017 calendar year, of the 377 femoral head samples obtained, 1 sample (0.27%) was determined to be contaminated at the time of storage. These values, in addition to our LDCP culture and BET testing data, indicate that concerns about the potential for microbial contamination are minimal. These encouraging findings support the clinical implementation of physiologic storage of osteochondral allograft as well as the expansion of currently limited OCA supply with living donor cartilage allograft. Given the evolution of new, rapid microbiological testing machinery such as that embodied by 3-dimensional colorimetry or DNA based methods, expedited sterility clearance and OCA implantation within hours to days of allograft procurement comprise goals that are well supported by the literature and may serve to optimize patient outcomes.
The current study is not without limitations. First, the presented living donor allografts comprise a highly selected subset of TKA patients with varus or valgus pathology with well-preserved contralateral compartments. While approximately 15 to 20 patients per month at our institution meet local and American Association of Tissue Banks (AATB) criteria for osteochondral donation, less than a third demonstrate single compartment predominant varus or valgus pathology amenable for osteochondral donation. It is important to note that all patients are independently evaluated and consented for TKA based on clinical exams and imaging, prior to being contacted by this study or future Living Donor Cartilage Allograft Project efforts. Second, further research on the biomechanical properties of living donor cartilage would better characterize this novel tissue source and assist with determining the utility and durability of such tissues obtained at the time of well-selected TKA. Third, while our outcomes are supported by previous studies demonstrating significantly decreased graft viability with storage over time and decreasing glycosaminoglycan content,34,35 the absolute magnitude of the observed decrease in viability has varied from article to article. This is likely partially related to variations between species given that animal data may not be directly applicable to clinical human experience and also related to variations between research institutions and private entities and their individual and sometimes proprietary storage solutions. Finally, both obtaining and maintaining physiologic storage of osteochondral tissues requires high-volume arthroplasty institutions with dedicated staff for patient consenting, tissue transportation, and storage.
In conclusion, osteochondral tissue viability and histologic quality progressively decreases with ex vivo storage, even when kept at physiologic temperatures. Currently available clinical OCAs are stored for 2 to 5 weeks prior to implantation and demonstrate inferior viability to that of fresh osteochondral tissues that can be made available through the use of an LDCP.
Footnotes
Acknowledgments and Funding
We thank the members of our laboratories and the laboratories of our colleagues, Elizabeth Bradley and Jennifer Westendorf for assistance, encouragement and stimulating discussions. We are also indebted to the kind support of the Mayo Clinic Bone Bank and the significant efforts of Susan Puffer and Renae Boyum. We also thank William and Karen Eby for their generous philanthropic support. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by National Institutes of Health Grant R01-AR069049 (to AJvW). This project was also supported by a grant provided by the Eby Family Foundation and funding provided by the Mayo Clinic Department of Orthopedic Surgery Orthopedic Research and Review Committee (ORRC).
Author Contributions
Mario Hevesi, MD: Conception and design, analysis and interpretation of the data, drafting of the article, critical revision of the article for important intellectual content, final approval of the article, statistical expertise, administrative, technical, or logistic support, collection and assembly of data. Janet M. Denbeigh, PhD: Conception and design, analysis and interpretation of the data, drafting of the article, critical revision of the article for important intellectual content, final approval of the article, statistical expertise, administrative, technical, or logistic support, collection and assembly of data. Carlo A. Paggi, MS: Collection and assembly of data, critical revision of the article for important intellectual content, final approval of the article. Catalina Galeano-Garces, PhD: Collection and assembly of data, critical revision of the article for important intellectual content, final approval of the article. Leila Bagheri, PhD: Collection and assembly of data, critical revision of the article for important intellectual content, final approval of the article. A. Noelle Larson, MD: Conception and design, critical revision of the article for important intellectual content, final approval of the article. Michael J. Stuart, MD: Conception and design, critical revision of the article for important intellectual content, final approval of the article. Daniel B. F. Saris MD, PhD: Conception and design, critical revision of the article for important intellectual content, final approval of the article Andre J. van Wijnen, PhD: Conception and design, analysis and interpretation of the data, critical revision of the article for important intellectual content, final approval of the article, provision of study materials or patients, obtaining of funding, administrative, technical, or logistic support. Aaron J. Krych, MD: Conception and design, analysis and interpretation of the data, critical revision of the article for important intellectual content, final approval of the article, provision of study materials or patients, obtaining of funding.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M. Hevesi is a paid consultant for Moximed. M. J. Stuart is on the editorial or governing board of the American Journal of Sports Medicine; received IP royalties from an is a paid consultant for Arthrex, Inc.; and receives research support from Stryker. D. B. F. Saris is a paid consultant for Cartiheal, Ivy Sports, and Smith & Nephew; is on the editorial or governing board, of Cartilage; and receives research support from Ivy Sports and Smith & Nephew. A. J. Krych receives research support from Aesculap/B. Braun, Arthrex, Inc., Arthritis Foundation, Ceterix, and Histogenics; receives IP royalties from Arthrex, Inc.; is a paid consultant from Arthrex, Inc., JRF Ortho, and Vericel; is on the editorial or governing board of the American Journal of Sports Medicine; and is a board or committee member of the International Cartilage Repair Society, International Society of Arthroscopy, Knee Surgery, and Orthopaedic Sports Medicine, Minnesota Orthopedic Society, and Musculoskeletal Transplantation Foundation. The other authors have no disclosures to make.
Ethical Approval
All aspects of this study were performed following institutional review board approval (IRB 13-005619).
Informed Consent
Written informed consent was obtained from all subjects before the study.
Trial Registration
Not applicable.
