Abstract
Objective
Chronic pain associated with osteoarthritis (OA) often leads to reduced function and engagement in activities of daily living. Current pharmacological treatments remain relatively ineffective. This study investigated the efficacy of photobiomodulation therapy (PBMT) on cartilage integrity and central pain biomarkers in adult male Wistar rats.
Design
We evaluated the cartilage degradation and spinal cord sensitization using the monoiodoacetate (MIA) model of OA following 2 weeks of delayed PBMT treatment (i.e., 15 days post-MIA). Multiple behavioral tests and knee joint histology were used to assess deficits related to OA. Immunohistochemistry was performed to assess chronic pain sensitization in spinal cord dorsal horn regions. Furthermore, we analyzed the principal components related to pain-like behavior and cartilage integrity.
Results
MIA induced chronic pain-like behavior with respective cartilage degradation. PBMT had no effects on overall locomotor activity, but positive effects on weight support (P = 0.001; effect size [ES] = 1.01) and mechanical allodynia (P = 0.032; ES = 0.51). Greater optical densitometry of PBMT-treated cartilage was evident in superficial layers (P = 0.020; ES = 1.34), likely reflecting the increase of proteoglycan and chondrocyte contents. In addition, PBMT effects were associated to decreased contribution of spinal glial cells to pain-like behavior (P = 0.001; ES = 0.38).
Conclusion
PBMT during the chronic phase of MIA-induced OA promoted cartilage recovery and reduced the progression or maintenance of spinal cord sensitization. Our data suggest a potential role of PBMT in reducing cartilage degradation and long-term central sensitization associated with chronic OA.
Introduction
Osteoarthritis (OA) is a prevalent chronic joint disease that affects cartilage and underlying bone integrity, leading to significant functional disability if left untreated. Current therapeutic options are mainly focused on delivering palliative treatments as with nonsteroidal anti-inflammatory drugs (NSAIDs).1,2 Nevertheless, the use of palliative pain treatments has been limited mainly due to the occurrence of side effects associated with such pharmacological interventions. 2 The characterized lack of vasculature and nerve terminals of joint cartilages hamper the delivery of potential treatments as well as contribute to the delay in diagnosing OA. 3 Therefore, non-pharmacological treatments are warranted to improve joint and pain-related impairments at late OA stages. Among many physical therapy treatments, photobiomodulation therapy (PBMT) has emerged as a promising nonpharmacological intervention. 1 PBMT is a noninvasive therapy largely employed to treat a series of neuromuscular disorders, this therapy uses lasers or light-emitting diodes (LEDs) over a target tissue. Preclinical studies have shown abundant evidence on the positive effects of PBMT in pain-like behavior, joint degeneration, and inflammation.4-19 Briefly, PBMT has been associated with regenerating cartilage, modulating inflammatory processes,4,5 and reducing pain-like behavior 6 following experimental knee OA. Table 1 summarizes the PBMT studies on knee OA in rodents to date, with a brief description of irradiation parameters and main treatment outcomes.
PBMT Effects on Pain-Like Behavior, Cartilage Integrity, and Inflammation in Experimental Models of Knee OA in Rodents. a
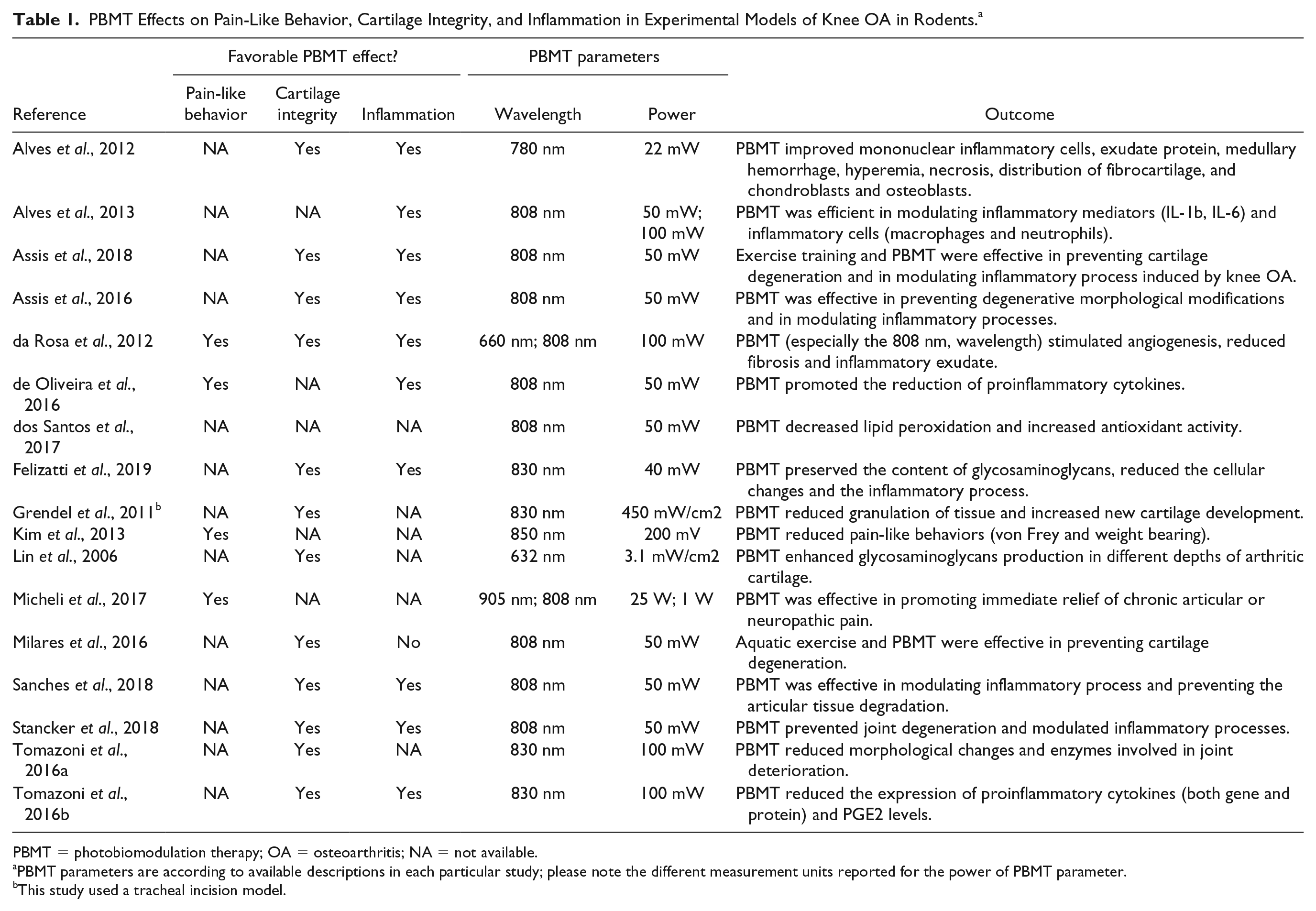
PBMT = photobiomodulation therapy; OA = osteoarthritis; NA = not available.
PBMT parameters are according to available descriptions in each particular study; please note the different measurement units reported for the power of PBMT parameter.
This study used a tracheal incision model.
Several experimental models have been used to induce knee OA in rodents, including spontaneously-, diet-, surgically-, or chemically-induced models. 20 Chemically- induced models, such as the monoiodoacetate (MIA) model, are effective in producing acute and chronic effects that mimic chronic nociceptive pain deficits. 21 These chronic pain-related deficits often encompass peripheral and central pain modulation mechanisms.22-24 MIA-induced peripheral nociceptive and inflammatory modulation is observed by increased local concentration of tumor necrosis factor–alpha (TNFα), interleukin-6 (IL-6), and nerve growth factor (NGF), 25 axonal injury to dorsal root ganglia (DRG) cells 23 and increased expression of activating transcription factor–3 (ATF-3), growth associated protein 43 (GAP43), neuropeptide Y (NPY) and calcitonin gene-related peptide (CGRP) in DRG cells.25-27 Furthermore, it is hypothesized that the central pain sensitization changes signal transmission at the spinal cord dorsal horn through microgliosis and astrogliosis via a mechanism of chronic neuropathic pain.22,23,25,28 Administration of NSAIDs reduced pain-like behavior 24 and NSAIDs or antibiotics attenuated astrogliosis in this model. 22 To advance the scientific evidence on the PBMT anti-inflammatory effects, the evaluation of central neuroinflammation biomarkers, such as the aforementioned reactive astrogliosis, is of utmost importance.22,29 However, little is known about the effects of PBMT over such chronic pain mechanisms following MIA-induced OA. 6
Hence, more studies are necessary for the full understanding of the PBMT effects at the cellular level, and thus, to consolidate PBMT as an effective OA treatment with consistent translation to the clinical practice.1,20,30 Although PBMT parameters’ optimization should be done in clinical studies to avoid translational failure, the investigation of the PBMT effects over chronic pain modulation mechanisms is hardly accomplished in clinical trials. Here, we used the MIA-induced knee OA model to assess the mechanisms of central pain modulation following PBMT treatment at chronic OA stages. In addition to central neuroinflammation,22,29 we sought to address how PBMT affects pain-modulation by CGRP, which is involved in the transmission of nociceptive information in the spinal cord dorsal horn. 31 We hypothesized that PBMT would promote pain relief associated with cartilage recovery and desensitization of second-order neurons in the spinal cord.
Methods
Animals
Experiment was conducted on adult male Wistar rats (age 90 days; weight 355 ± 22 g, n = 20). Housing, surgery, behavioral experiments, and euthanasia were performed in compliance with the NIH Guide for the Care and Use of Laboratory Animals and AROUCA Brazilian Law (number 11.794). Our ethics committee approval number is 23354/PROPESQ-UFRGS (Porto Alegre, RS, Brazil). Animals were housed in number of 4 (Plexiglas cages; 50 × 40 × 20 cm; length × width × height) in a temperature-regulated environment under a controlled 12-hour light-dark cycle, with lights on at 6:00 a.m., with food and water freely available (experiments performed during the light cycle). One animal died during the procedures and was excluded from the analysis (n = 19).
MIA Model of OA Pain
Rats were anesthetized with an intramuscular injection of a 7% ketamine solution and 0.3% xylazine solution (Vetaset, Fort Dodge, Campinas, SP, Brazil) at a proportion of 2:1 (0.2 mL per 100 g). Joint damage was induced by a single intra-articular injection of MIA (2 mg/0.05 mL sterile isotonic saline; Sigma, St. Louis, MO) into the right knee joint cavity as described in previous studies.24,32-34 The left knee was used as a control by injecting 0.05 mL of saline solution (sterile isotonic saline). Following the injections, animals were allowed to recover from the anesthesia effects (5-10 minutes) before returning to the home cages. Analgesic treatment (metamizole sodium) was provided every 4 hours over 2 days. All animals received MIA (right knee) and saline (left knee) injections and were randomly assigned as: control group (n = 9 animals) and PBMT-treated group (n = 10 animals).
PBMT
A low-level GaAIA laser (Vectra Genisys Systems, Model 2784, Chattanooga Group, Hixson, TN, USA) was applied with an 850 nm wavelength, 100 mW of power output, 1.43 W/cm2 of power density, 4 J of energy per site, 57.14 J/cm2 of energy density per site, 40 seconds on each site using the continuous mode.8,35 The single probe consisted of 1 diode (spot treatment area of 0.07 cm2), and laser irradiation was applied in 4 sites on the following transcutaneous compartments: medial, lateral, anterior, and posterior joint (randomly; Table 2 and Fig. 1A ). 35 PBMT parameters were based on a previous study that compared different wavelengths in a similar knee OA model. 8 For safety reasons, protective eyeglasses were used by the trained researchers during PBMT treatment. In the control group, the treatment was applied in a similar manner, but with the device turned off. PBMT treatment was provided once a day for 15 days, starting at post-MIA injection day 15 (d15).
Photobiomodulation Therapy (PBMT) Parameters Used in the Following Equipment: Vectra Genisys Laser, Model 2784 (Chattanooga Group, Hixson, TN, USA). a
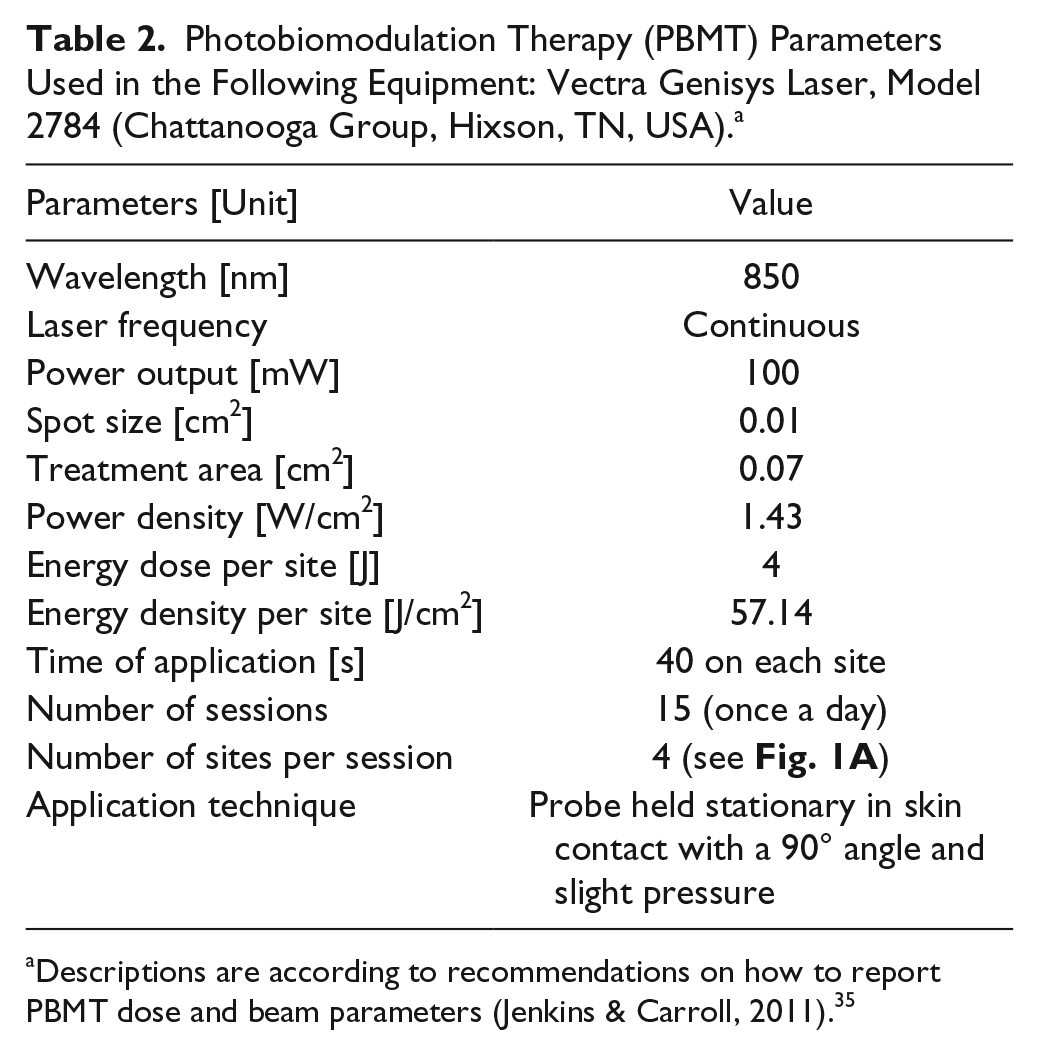
Descriptions are according to recommendations on how to report PBMT dose and beam parameters (Jenkins & Carroll, 2011). 35
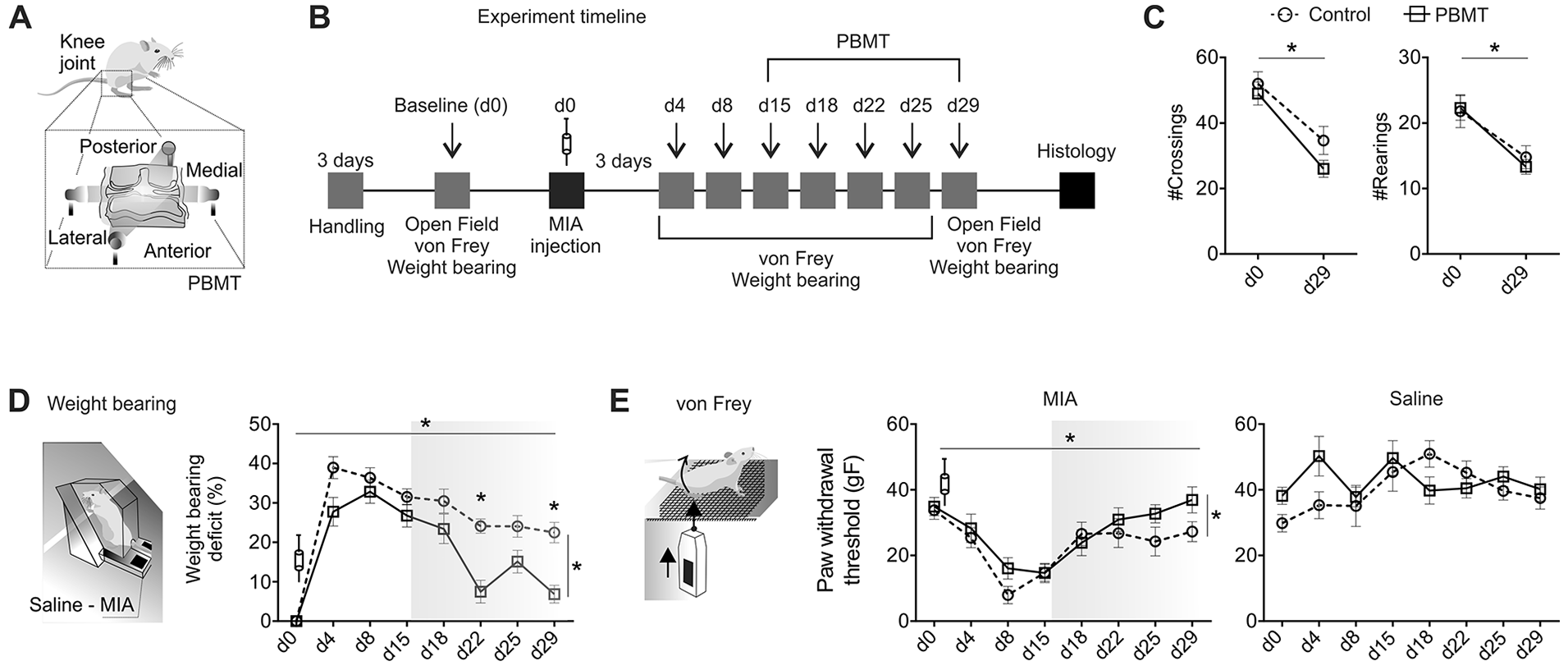
Monoiodoacetate (MIA) induced pain-like behavior, while photobiomodulation therapy (PBMT) reduced most of this behavioral deficit. (
Behavioral Testing
Behavioral measurements were conducted prior to intra-articular injection (Baseline: d0) and at post-MIA injection days 4 (d4), 8 (d8), 15 (d15), 18(d18), 22 (d22), 25 (d25), and 29 (d29) ( Fig. 1B ). Here, we used stimulus (von Frey) and nonstimulus (weight bearing and open-field) evoked methods to evaluate pain-like behaviors in rodents. 36 Specifically, we opted to use the weight bearing test as our primary outcome based on previous findings that reported high sensitivity of this test in detecting pain-like behavior following MIA and NSAIDs treatment. 24 The open-field test was used as a secondary, nonstimulus evoked behavioral testing to evaluate gross gait parameters. The von Frey test was used as a stimulus evoked method. 36 For all behavioral testing, the experimenter was blinded to experimental or control groups.
Open Field
A square open field arena (38 × 38 × 38 cm) was divided into 9 zones. Rats were placed individually on the arena center, always facing the same direction, and were video-recorded for 5 minutes. The estimated distance traveled (#crossings) and the number of exploratory events (#rearings) were manually counted by a trained evaluator.
Mechanical Allodynia
An electronic von Frey monofilament apparatus (Insight, Ribeirão Preto, SP, Brazil) was used to gradually stimulate the plantar surface of both hind paws (for a maximum of 4 seconds). Once a withdrawal reflex was observed, the maximum force (gF) applied was computed—this procedure was repeated 3 times for each hind paw. If the animal presented instantaneous withdrawal reflex or reduced weight bearing at the affected paw (due to lesion), the withdrawal threshold was considered zero gF.
Weight bearing
Hindlimb weight bearing was measured using a custom-made apparatus (for detailed description on dimensions and calibration see Supplementary Figure 1). Briefly, this apparatus consisted of a plexiglass cage, to restrain the rat, built-in with 2 digital scales (± 0.01 g accuracy; Digital Pocket Scale, China). Animals were placed into the plexiglass test box and acclimated to the apparatus for 2 to 5 minutes. Weight (gF) distribution to each hindlimb was measured for 3 times (over a period of 5 seconds). Weight bearing deficit was calculated as follows:
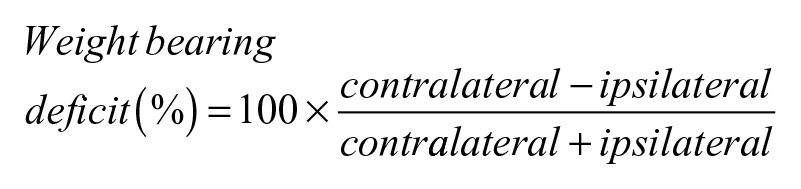
Histology
At d30, 24 hours after the last day of behavioral assessment, rats were overdosed with ketamine and xylazine (80 mg/kg and 10 mg/kg; intraperitoneally), and transcardially perfused through the left ventricle using a peristaltic pump (Control Company, Friendswood, TX, USA, 20 mL/min)—perfused by 400 mL of 0.9% saline solution followed by 400 mL of a fixative solution (4% paraformaldehyde in 0.1 M phosphate buffer, pH 7.4; PB). The lumbar spinal cords were removed, postfixed in the same solution for 4 hours, cryoprotected by immersion in 15% and 30% sucrose solutions in PB at 4°C until they sank, quickly frozen in isopentane (Merck, Darmstadt, Germany) cooled in liquid nitrogen and kept in a freezer (−70°C) for further analyses. Both hindlimbs were dissected and the soft tissue surrounding the knee joint was removed. Femorotibial joints were fixed in 10% phosphate-buffered formalin and subsequently decalcified in 5% formic acid for 72 hours. Transverse sections (24 µm) from the lumbar spinal cords (L2-L5) and coronal sections of femorotibial joints (14 µm) were obtained using a cryostat (at −20°C; CM1850, Leica, Wetzlar, Germany). Free-floating spinal cord sections were collected in PB saline (PBS; pH 7.4), pretreated with 3% hydrogen peroxide for 30 minutes, carefully washed, treated with 2% bovine serum albumin (Inlab, São Paulo, SP, Brazil) in PBS containing 0.4% Triton X-100 (PBS-Tx; Sigma Chemical Co., St Louis, MO, USA) for 30 minutes, incubated for 48 hours with polyclonal CGRP (1:2250; courtesy of Dr. Rodrigo, Cajal Institute, Spain) or glial fibrillary acidic protein (GFAP; 1:150; Sigma) antibodies, and finally were gently stirred at 4°C. The primary antibody was removed and sections washed in PBS-Tx for 30 minutes, immersed in a secondary antibody (goat anti-rabbit lgG; Sigma Chemical Co., St Louis, MO, USA) diluted 1:50 in PBS-Tx for 2 hours at room temperature with gentle stirring. After washing with PBS-Tx 3 times for 15 minutes, a soluble complex of horseradish peroxidase and rabbit anti-horseradish peroxidase (Sigma Chemical Co., St Louis, MO, USA) diluted 1:500 was applied for 1 hour 30 min at room temperature. The reaction was revealed in a medium containing 0.06% 3,3′-diaminobenzidine (DAB, Sigma Chemical Co., St Louis, MO, USA) dissolved in PBS for 10 minutes, and 1 mL of 3% H2O2/mL was added to the DAB medium for an additional 10 minutes. Finally, sections were rinsed in PBS, mounted on glass slides, dehydrated in ethanol, cleared with xylene and covered with DPX (Sigma Chemical Co., St Louis, MO, USA) and coverslips. Femorotibial joints were stained using standard blue toluidine procedures. Image J software (NIH, USA) was used to quantify spinal cord and femorotibial joint histology. All images containing the region of interest were converted to an 8-bit grayscale (0–255 gray levels). The picture elements (pixels) employed to measure the optical density were obtained from rectangles (CGRP: 200 µm × 60 µm) or squares (GFAP: 30 µm × 30 µm) overlaid on the grayscale image. Both the left and right sides of each spinal cord were used. Software was calibrated using a standard calibration image. Background quantification was performed before each optical densitometry (OD) measurement, and later subtracted from each measurement. CGRP and GFAP OD were quantified in the dorsal horn (laminas 1-2) of the lumbar spinal cord (L2-L5). CGRP analysis was conducted at 3 dorsal horn regions (lateral, central, and medial; 20 measurements/animal, 10 for each spinal cord side; 5× magnification). GFAP was analyzed in the central region of the dorsal horn in a similar manner but using 40× magnification to visualize reactive astrocytes (100 cells/animal; 50 cells/side). Femorotibial joint cartilage proteoglycan and chondrocyte content was measured in a similar manner. Briefly, images from the central region of medial tibial plateau (MTP) and medial femoral condyle (MFC; 10× magnification) underwent the same aforementioned transformation and calibration procedures, and rectangles (100 µm × 25 µm) were placed at the lateral, central, and medial aspects of the deep (closest to subcondral bone) and superficial cartilage layers (40 measurements/animal; ≈20 measurements per hind paw). Background correction was performed by subtracting basal from superficial cartilage OD. In addition, we quantified the length of tidemark violations normalized to total MTB or MFC cartilage lengths, and a qualitative subchondral bone score (data not shown).
Statistical Analysis
Data normality was accessed using the Shapiro-Wilk test. Two-way analyses of variance (α = 0.05) were used to investigate general main effects in all planned comparisons (factors: lesion and treatment). To confirm the main effects, Sidak post hoc correction was applied (using GraphPad Prism software). LabVIEW 8.5 was used for computational statistics—used to explore the complex dataset, calculate Pearson correlation maps and dimensions reductions using principal component (PC) analysis. For (1) spinal cord histology the unit of analysis was considered the cell (astrocyte) or region (CGRP); for (2) behavior and cartilage the unit of analysis was considered the animal and the knee, respectively. All PC analysis was conducted in z-score transformed data to avoid magnitude artifacts. Statistical significance was set for P < 0.05.
Results
Experiment timeline is described in Figure 1B .
MIA Induced Acute and Chronic Behavioral Deficits in Rats
The MIA model of knee OA induced deficits in locomotion. In such gait impairment, animals crossed less quadrants (#crossings: Ftime(1, 35) = 45.49, P < 0.001) and explored less (#rearings: Ftime(1, 35) = 33.57, P < 0.001) the open field arena. PBMT treatment was not effective in reverting these deficits (#crossings: Ftreatment(1, 35) = 3.81, P = 0.107; #rearings: Ftreatment(1, 35) = 0.12, P = 0.797) ( Fig. 1C ). MIA injection also produced robust weight bearing deficit immediately following the intra-articular injection (d4)—sustained throughout the experiment (Ftime(7, 137) = 11.61, P < 0.001). PBMT reduced this deficit at d22 and d29 (Ftreatment(1, 137) = 11.61, P = 0.001; Fig. 1D ). Stimulus evoked mechanical allodynia was also evident (Ftime(7, 137) = 8.84, P < 0.001), and PBMT reduced this deficit without post hoc effects (Ftreatment(1, 137) = 4.61, P = 0.032; Fig. 1E ).
The Cartilage Optical Density Changed Following PBMT Treatment
Reduction of cartilaginous matrix and cellular content was evident 30 days post-MIA injection, and proteoglycan and chondrocyte loss at the superficial cartilage was evident 37 ( Fig. 2A ). In less affected areas, there was noticeable partial damage that extended approximately one-third to one-half of cartilage thickness. At the transition zone between damaged and viable cartilage, chondrocytes were isolated, variable in size and tightly aggregated in clusters or chondrones. Tidemark violations were also evident alongside with hypertrophic chondrocytes inside the newly forming calcified cartilage layer.
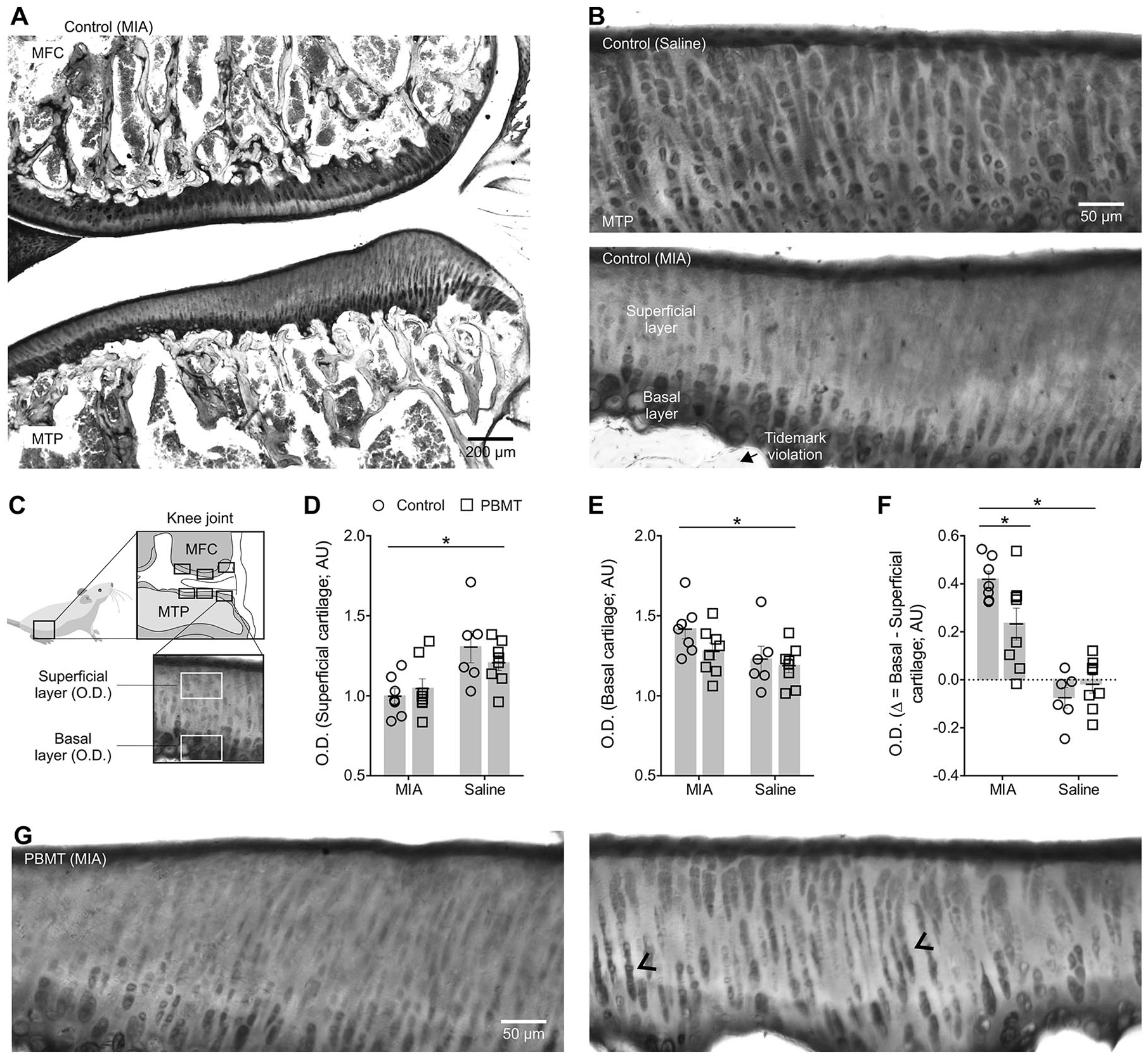
Osteoarthritis (OA) induced by monoiodoacetate (MIA) was characterized by cartilage degradation that was partially rescued following 14 days of photobiomodulation therapy (PBMT). (
Control knees injected with saline displayed abundant toluidine blue staining at d30, evenly distributed throughout the superficial and deep cartilage layers (
We next used dimension reductions to understand the relationship between the changes in cartilage density induced by PBMT and pain-like behavior. Here we showed that cartilage optical density alterations were consistently correlated with weight bearing deficits at d4 (superficial: r = −0.64, P = 0.010; basal-superficial: r = 0.52, P = 0.044), d15 (basal-superficial: r = 0.67, P = 0.006), d18 (superficial: r = −0.67, P = 0.006), and d22 (basal-superficial: r = 0.56, P = 0.028; Fig. 3A ). Furthermore, PBMT promoted superficial cartilage change that was correlated with early weight bearing recovery following treatment onset: at d18 (superficial: r = −0.74, P = 0.034) and d22 (superficial: r = −0.72, P = 0.042; Fig. 3B ). Supplementary Table 1 shows variables that significantly correlated with at least one cartilage optical density measurement (also shown in the correlational mapping: white hashed rectangles in Figs. 3A and B ). PC analysis revealed a principal component related to superficial and ∆ cartilage changes, which explained most of the variance in the dataset of PBMT animals ( Fig. 3C ).
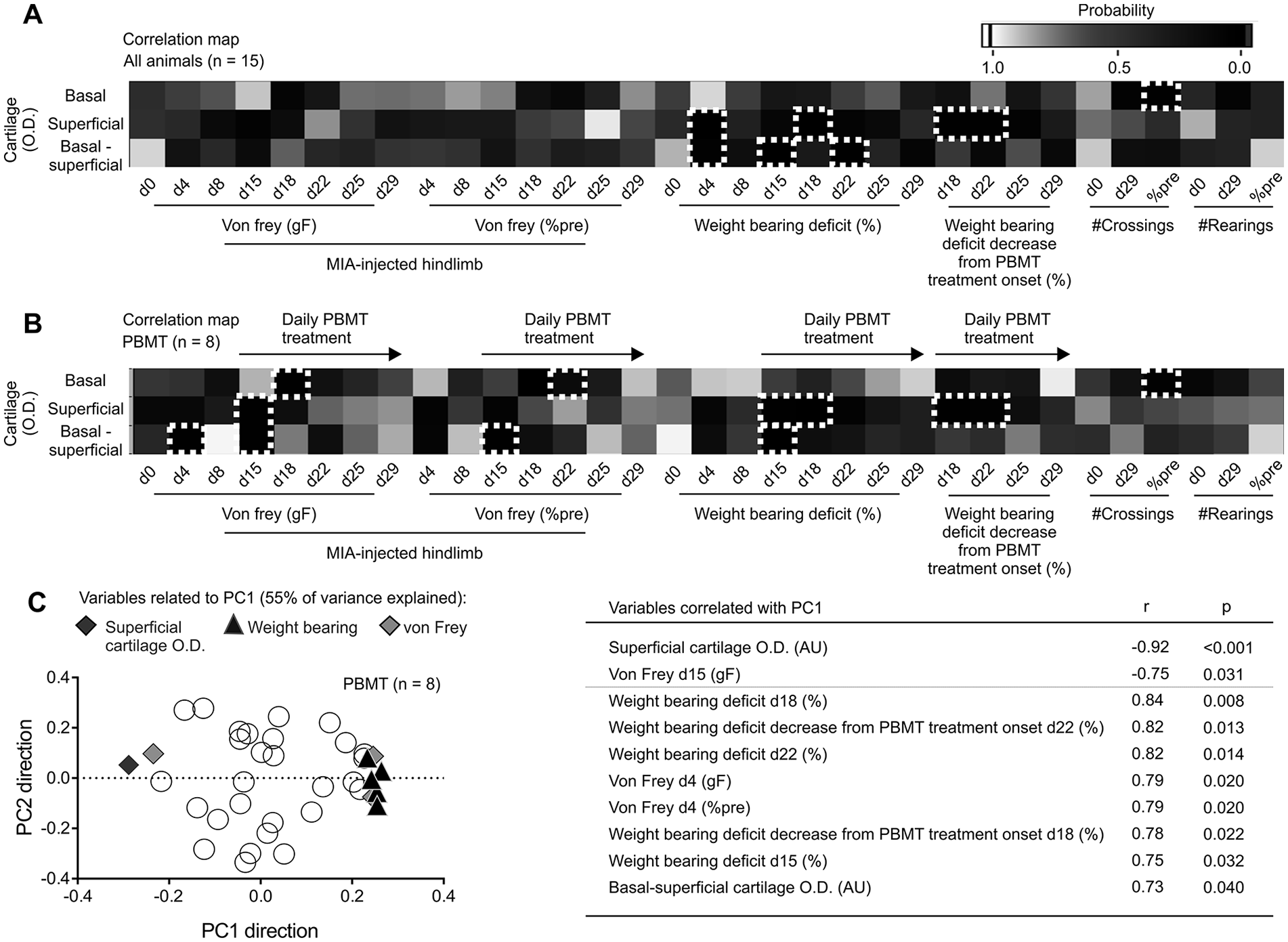
Cartilage optical densitometry (OD) was correlated with pain-like behavior, and photobiomodulation therapy (PBMT)–promoted pain relief also correlated with cartilage OD (
MIA Increased Pain Transmission in the Spinal Cord Dorsal Horn, while PBMT Reduced the Spinal Astrocytes Contribution to Chronic Pain Behavior
Here we explored the contribution of central pain modulation mechanisms in recovering pain-like behavior following PBMT. While MIA injections increased CGRP signaling in the ipsilateral spinal cord dorsal horn (Flesion(1, 218) = 11.59, P < 0.001; Fig. 4A and B ), PBMT had no effect over such CGRP-mediated pain modulation. Conversely, we found that PBMT reduced the reactive astrogliosis widespread in both ipsilateral and contralateral spinal cord segments, with significant post hoc for the contralateral spinal cord (Ftreatment(1, 688) = 10.39, P = 0.001; Fig. 4C and D ).
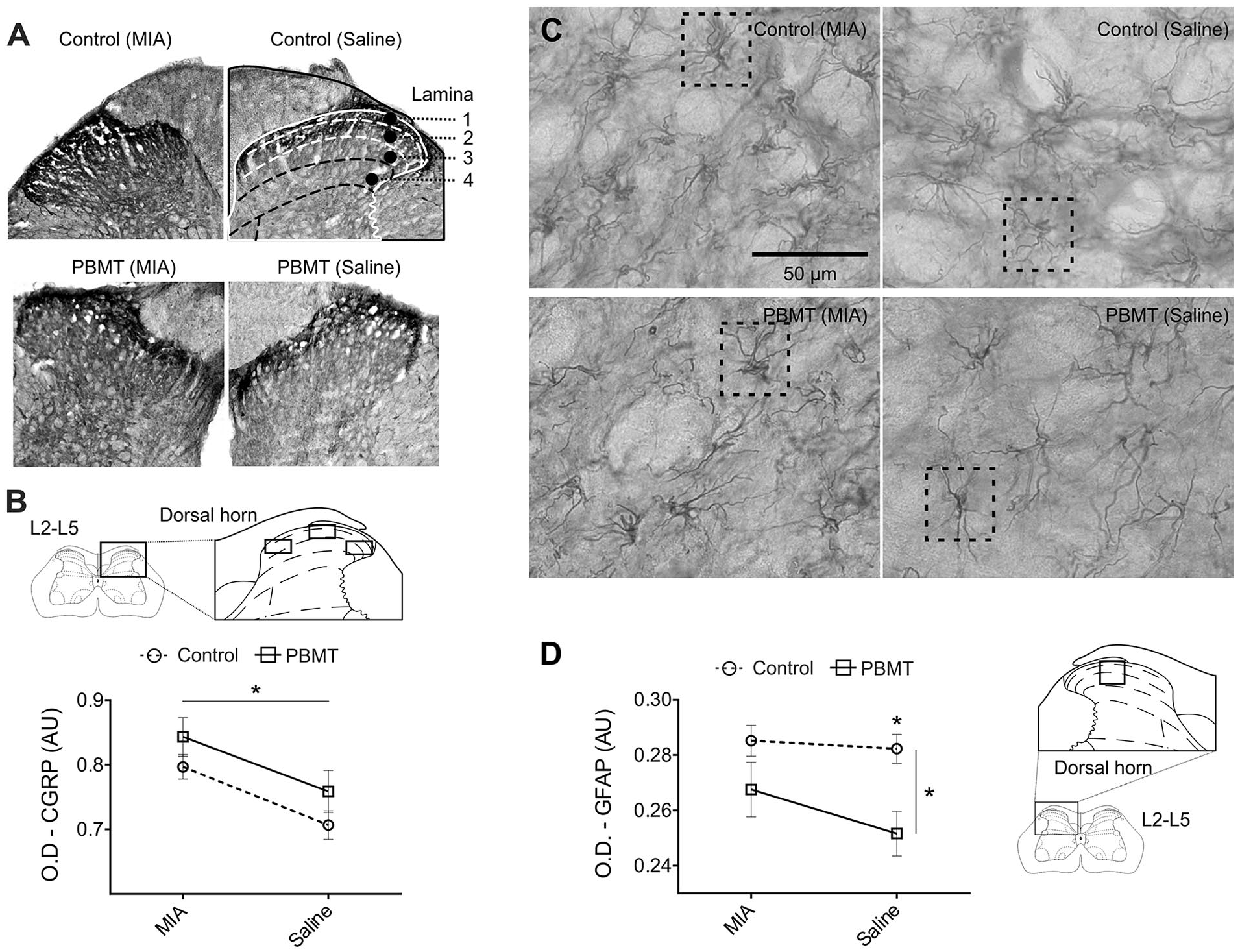
Photobiomodulation therapy (PBMT) was effective in reducing reactive astrogliosis in the spinal cord dorsal horn. (
Discussion
MIA injection induced persistent pain-like deficits that consistently and negatively correlated with cartilage optical density measures. PBMT reduced pain-like behavioral deficits of weight support and mechanical allodynia but did not affected general locomotor activity. The behavioral improvement seen with PBMT was accompanied by a reduction of the widespread reactive astrogliosis in the spinal cord dorsal horn. Overall, our results support a potential use of PBMT to promote cartilage recovery and to control chronic pain in rats.
PBMT has shown the ability to regenerate cartilaginous4,5,8,9,15,17 and nervous38,39 tissues. In the present study, PBMT had positive effects in treating MIA-induced knee OA. However, the OA observed in humans has other origins, as such, the results of this small animal model should be transferred with caution to human clinical OA. The PBMT effects for OA treatment remain to be fully determined; however, PBMT potentially acts through mechanisms of photostimulation at the cellular level. 40 A leading hypothesis on the PBMT’s mechanism of action involves mitochondrial mechanisms related to cytochrome activity, such as cytochrome-c-oxidase, which absorbs light into the near-infrared region.41,42 Several signaling pathways are activated, leading to increased activity of transcription factors related to genes expression, which, in turn, are associated to protein synthesis, cell migration and proliferation, anti-inflammatory signaling, 42 and chondrogenesis. 43 This proposed mechanism is especially beneficial to chondrocytes, which maintain cartilage homeostasis by synthesizing cartilage-specific extracellular matrix. 43 Here we showed that PBMT promoted cartilage recovery potentially associated with a greater proteoglycan and chondrocyte content in superficial layers along with reduced calcification level of deeper cartilage zone. We suggest that PBMT was effective to stimulate cartilage proteoglycan and collagen synthesis and chondrocytes replacement.44-47 Similar studies reported the increase of cartilaginous biosynthesis and of stress proteins in articular chondrocytes following PBMT intervention.17,48 PBMT may also induce differentiation of mesenchymal cells or activation of surviving chondrocytes, leading to cell differentiation, proliferation, and extracellular matrix synthesis—such as collagen and proteoglycans.17,43,49 We suggest that the complex cellular and extracellular changes promoted by PBMT reflected in the stronger cartilage observed post treatment. Another possibility is that PBMT may act at the matrix level changing the microenvironment, for exampe, reducing matrix stiffness or fibrosis, which would also reduce the mechanical stress on chondrocytes, and thereby increase cellular metabolism (reviewed elsewhere 49 ).
We also showed that superficial cartilage optical density strongly and negatively correlated with PC 1 (r = −0.92, P < 0.001), which could explain most of the variance in the dataset. Previous studies have suggested that a correlation between pain-like behavior and tissue architecture repair is required to accurately indicate disease progression and repair therapies efficacy.50,51 Interestingly, we further reported that PC 1 correlated with pain-like behaviors in an inverse manner, if compared to cartilage optical density. This result indicates that a greater superficial cartilage optical density is strongly associated to a greater behavioral performance—such as weight bearing and von Frey. Taken together, the results from this PC analysis, alongside with the correlational mapping, support the use of optical density measurements as a quantitative surrogate of cartilage integrity following MIA-induced knee OA.
The peripheral inflammatory response to OA may cause central inflammation with contribution of microglia and astrocytes,22,23,25 also observed in other animal models of peripheral inflammation. 29 PBMT was previously associated with promoting analgesic52-54 and anti-inflammatory effects,55,56 and involved in peripheral reduction of TNFα, 57 cyclooxygenase-2 (COX-2), 58 prostaglandin E2 (PGE2), 59 fibrinogen, 60 edema, 61 inflammatory cells, 56 and mediators (IL-1β, IL-6). 62 Here, we showed that PBMT was also able to indirectly control central inflammation by reducing astrogliosis in the spinal cord. We suggest that PBMT may reduce nociceptive signaling to the spinal cord dorsal horn by decreasing peripheral inflammation. This effect may result in decreased astrocytes activation and consequent desensitization of first- and second-order neurons, and subsequent reduction of the MIA-induced hyperalgesic state.22,63,64 Future studies should unveil the link between peripheral and central inflammation processes and the role of microglia in this knee OA experimental model.
PBMT may also act on nerves by reducing pain transmission. 65 As such, the effects of PBMT in pain relief occur following several models of peripheral nerve lesion through direct inhibition of nociceptors. For example, at the spinal cord level, long-term PBMT treatment can result in pain transmission depression—with respective reduction of neuronal Aδ and C transmission. 66 Here, we reported that PBMT promoted recovery of weight bearing deficits correlated with superficial cartilage density increase at early treatment (i.e., following 3 [d18] or 7 days [d22] of PBMT), but lacked correlation at later time points. This finding suggests that chronic pain modulation may involve other structures, hence, not fully captured by exclusive changes to the cartilaginous tissue. The MIA knee OA model used in the present study (at dose of 2 mg) has shown to induce not only joint degeneration but also axonal injury to DRG cells. 23 We suggest that PBMT may have affected neural structures, such as nerve terminals surrounding the cartilage and nearby structures.
Nevertheless, Huang et al. 1 reported that the current evidence does not support the PBMT efficacy as a therapy for patients with OA. However, our research team and others have reported positive PBMT effects in increasing health status, reducing pain, and increasing physical functionality in women with knee OA.67,68 Here, the findings of this small animal study are interesting, both the cartilage seemingly restorative effect and the effect on spinal glial cells that might influence pain reactions. However, clinical translation of these findings is not straightforward but should stimulate to further animal studies and future pilot human trials.
In summary, here we report that PBMT during the chronic phase of MIA-induced OA promoted cartilage and weight support recovery, and also reduced the progression or maintenance of central sensitization in the spinal cord of rats. Interestingly, our results showing decreased reactive astrogliosis following PBMT corroborate late effects of NSAIDs on MIA-induced pain-like behavior, with similar reduction of astrocyte activation at d28. We suggest that PBMT has an important role in promoting cartilage recovery and controlling long-term central sensitization associated with chronic knee OA.
Supplemental Material
4.Supplementary_Table_1_17.06.19 – Supplemental material for Photobiomodulation Therapy Partially Restores Cartilage Integrity and Reduces Chronic Pain Behavior in a Rat Model of Osteoarthritis: Involvement of Spinal Glial Modulation
Supplemental material, 4.Supplementary_Table_1_17.06.19 for Photobiomodulation Therapy Partially Restores Cartilage Integrity and Reduces Chronic Pain Behavior in a Rat Model of Osteoarthritis: Involvement of Spinal Glial Modulation by Gustavo Balbinot, Clarissa Pedrini Schuch, Patricia Severo do Nascimento, Fabio Juner Lanferdini, Mayra Casanova, Bruno Manfredini Baroni and Marco Aurélio Vaz in CARTILAGE
Supplemental Material
Supplementary_Figure_1_08.01.19 – Supplemental material for Photobiomodulation Therapy Partially Restores Cartilage Integrity and Reduces Chronic Pain Behavior in a Rat Model of Osteoarthritis: Involvement of Spinal Glial Modulation
Supplemental material, Supplementary_Figure_1_08.01.19 for Photobiomodulation Therapy Partially Restores Cartilage Integrity and Reduces Chronic Pain Behavior in a Rat Model of Osteoarthritis: Involvement of Spinal Glial Modulation by Gustavo Balbinot, Clarissa Pedrini Schuch, Patricia Severo do Nascimento, Fabio Juner Lanferdini, Mayra Casanova, Bruno Manfredini Baroni and Marco Aurélio Vaz in CARTILAGE
Footnotes
Author Contributions
GB, PSN, BMB and MAV designed the experiments. GB, CPS, FJL, MC and PSN performed the experiments. GB analyzed the data and wrote the first draft of the manuscript. GB, CPS, BMB and MAV revised the manuscript. All authors approved the final version of the manuscript.
Acknowledgments and Funding
We thank PhD Milton Antônio Zaro and PhD Matilde Achaval for supervision and support. We also thank PhD Lidiane Isabel Filippin for insightful comments and directions at the initial stages of study design. MAV and BMB are productivity fellows from CNPq, Brazil. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES/Brazil).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by our ethics committee (approval number: 23354/PROPESQ-UFRGS; Porto Alegre, RS, Brazil).
Animal Welfare
Housing, surgery, behavioral experiments, and euthanasia were performed in compliance with the NIH Guide for the Care and Use of Laboratory Animals and AROUCA Brazilian Law (number 11.794).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
