Abstract
Objective
Metabolic disturbance is a known risk factor for cardiovascular disease and has been identified as a risk factor for the development of knee osteoarthritis. In this study, we sought to determine the effects of prebiotic fiber supplementation, aerobic exercise, and the combination of the 2 interventions, on the progression of knee osteoarthritis in a high-fat/high-sucrose diet-induced rat model of metabolic disturbance.
Design
Twelve-week-old male CD-Sprague-Dawley rats were either fed a standard chow diet, or a high-fat/high-sucrose diet. After 12 weeks on diets, rats consuming the high-fat/high-sucrose diet were randomized into 4 subgroups: a sedentary, an aerobic exercise, a prebiotic fiber supplementation, and an aerobic exercise combined with prebiotic fiber supplementation group. The aerobic exercise intervention consisted of a progressive treadmill training program for 12 weeks, while the prebiotic fiber was added to the high-fat/high-sucrose diet at a dose of 10% by weight for 12 weeks. Outcome measures included knee joint damage, body mass, percent body fat, bone mineral density, insulin sensitivity, and serum lipid profile.
Results
Aerobic exercise, or the combination of prebiotic fiber and aerobic exercise, improved select markers of metabolic disturbance, but not knee joint damage. However, these results need to be considered in view of the fact that the chow-fed rats had similar knee OA-like damage as the high-fat/high-sucrose–fed rats.
Conclusion
Exercise or prebiotics did not increase joint damage and might be good strategies for populations with metabolic knee osteoarthritis to alleviate other health-related problems, such as diabetes or cardiovascular disorders.
Introduction
Metabolic syndrome is a complex disorder and is defined by the World Health Organization as a pathologic condition characterized by abdominal obesity, insulin resistance, hypertension, and dyslipidemia (low high-density lipoprotein [HDL]-cholesterol and high triglyceride levels). 1 These metabolic disturbances are known risk factors for cardiovascular disease,2,3 as well as for the development of knee osteoarthritis (OA),4,5 which is now recognized as a separate “metabolic OA” phenotype. 6 Moreover, nearly 60% of the OA patient population present signs of metabolic disturbance. 7
Exposing CD-Sprague-Dawley rats to a high-fat/high-sucrose (HFS) diet leads to increased body fat, systemic inflammation, knee joint inflammation, 8 intramuscular fat deposition,9,10 and knee damage within 12 weeks 8 (for a detailed review, see Collins et al. 11 and Courties et al. 5 ). It has been shown that prebiotic fiber supplementation, aerobic exercise, and the combination of prebiotic fiber and aerobic exercise prevented the development of OA-like damage that is observed in rats on a HFS diet, when the interventions were started at the onset of the HFS diet exposure. 12 Additionally, clinical trials with elderly knee OA patients with overweight and obesity demonstrated that exercise improved self-reported measures of knee function and pain, 13 while data from prospective cohort studies showed that a diet rich in fiber lowers the risk of moderate or severe knee pain regardless of radiographic knee osteoarthritis status. 14
While there is evidence that early exercise and prebiotic fiber supplementation can prevent the development of OA in a rat model of diet-induced metabolic disturbance, and that exercise and fiber consumption improve functional outcomes in human OA studies, it remains unknown if these interventions are equally successful when started at time points after the introduction of a HFS diet when obesity is already well-established and once knee OA-like damage has begun to emerge. Specifically, it would be crucial to know the threshold in time when exercise and prebiotics are effective, when they become neutral, and possibly if they become destructive and might accelerate knee OA progression. Therefore, the purpose of this study was to determine the effects of prebiotic fiber supplementation, aerobic exercise, and the combination of prebiotic fiber and aerobic exercise on the progression of metabolic knee OA in a HFS diet-induced rat model of metabolic disturbance. We hypothesized that prebiotic fiber supplementation, an aerobic exercise regime, and the combination of prebiotic fiber and aerobic exercise, introduced 12 weeks after the onset of an obesity-inducing HFS diet, would reduce the progression of OA-like damage in the knee of rats fed a HFS diet by reducing the metabolic disturbance caused by the HFS diet.
Methods
Animals and Intervention Protocol
Seventy male, 12-week old, CD-Sprague-Dawley rats were housed individually (12:12 hour light-dark cycle, 21°C) and either fed ad libitum a standard chow diet (control group, 5% of total weight as fat, 47.5% carbohydrates [only 4% from sucrose], 25% protein, 12.5% from fiber and micronutrients, and 10% moisture; Lab Diet 5001, United States, n = 11), or a HFS diet (20% of total weight as fat, 50% sucrose, 20% protein, and 10% from fiber and micronutrients; custom Diet #102412, Dyets, United States, n = 59). After 12 weeks on diets, rats consuming the HFS diet were randomized (stratified randomization based on percent body fat) into four subgroups: a sedentary (HFS, n = 15), an aerobic exercise (HFS + E, n = 14), a prebiotic fiber supplementation (HFS + F, n = 15), and an aerobic exercise combined with prebiotic fiber supplementation (HFS + F + E, n = 15) group ( Fig. 1 ). The aerobic exercise intervention consisted of a progressive moderate treadmill training program for 12 weeks, up to 30 minutes per day, 5 days a week as described previously.15-17 The prebiotic fiber intervention consisted of supplementing the HFS diet with 10% prebiotic fiber by weight (wt/wt, Orafti P95, BENEO-Orafti, Germany).12,18,19 Calculation of sample size was performed using G*Power Software (version 3.0.10, Germany), 20 and it was based on the knee joint damage presented in a previous study with HFS and control group rats. 8 All experiments were approved by the University of Calgary Life and Environmental Sciences Animal Care Committee. Only male rats were examined thereby not allowing for identifying sex-specific differences, which is a limitation of this study.
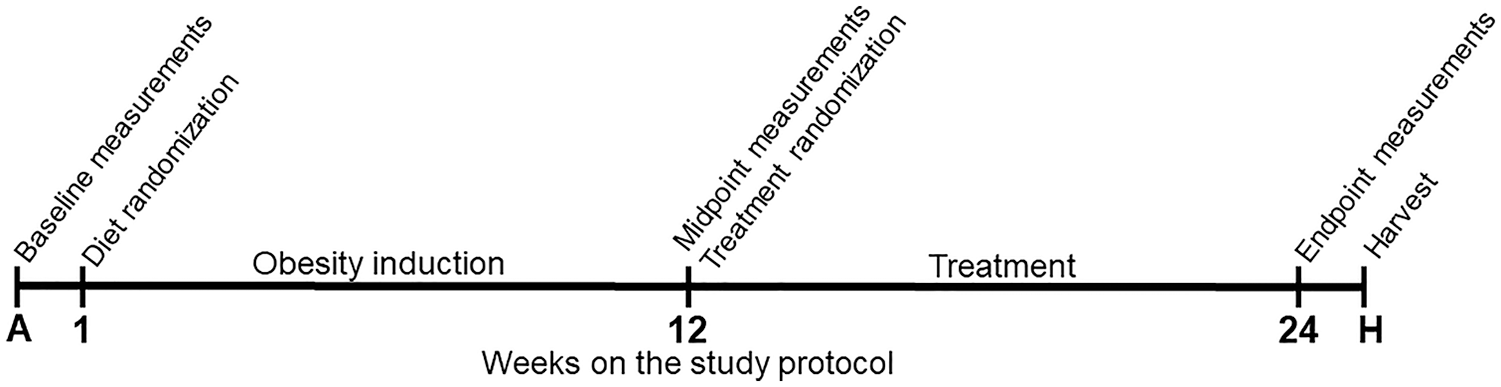
Study design. Measurements performed at baseline (acclimation week): body mass, blood glucose, blood insulin, and blood lipid profile. Measurements performed at midpoint: body mass, blood glucose, blood insulin, blood lipid profile, and body composition (DXA [dual-energy X-ray absorptiometry] scan). Measurements performed at endpoint: body mass, blood glucose, blood insulin, and body composition (DXA scan). Measurements performed at harvesting: blood lipid profile. Body mass was measured at the beginning of each week in the study protocol.
Body Composition
Twelve weeks after the diets started (midpoint) and at the end of the intervention protocol, rats were lightly anesthetized with isoflurane and body composition (for percent body fat and bone mineral density calculations) was measured using Dual X-ray absorptiometry (DXA) with software for small animals (Hologic ODR 4500; Hologic, Bedford, MA, USA). The mean of 2 repeat scans for each animal was used for analysis. Body mass was measured at the beginning of each week.
Blood Glucose and Insulin
During the baseline acclimation week (week A), after 12 weeks on their respective diet (midpoint), and at the end of the intervention period (endpoint), rats were given an oral gavage of 2 g/kg glucose following a 16-hour food deprivation period. Blood was collected at 0, 15, 30, 60, and 120 minutes postgavage via tail nick into a chilled tube for insulin analysis (Multiplex Discovery Assay; Eve Technologies, Alberta, Canada). Blood glucose was measured immediately with a blood glucose meter (OneTouch Verio and Blood Glucose Monitoring System, Lifescan, Switzerland). Whole body insulin sensitivity was determined using proxy measures from the glucose tolerance tests (composite insulin sensitivity index [CISI]).21,22
Blood Lipid Profile
During the baseline acclimation week (week A – via tail nick: 800 µL), after 12 weeks on their respective diet (midpoint – via tail nick: 800 µL), and at the end of the experimental protocol (via cardiac puncture: 7-10 mL), blood was collected following a 16-hour food deprivation period. Serum was analyzed for lipid profiles (total cholesterol, low-density lipoprotein [LDL]-cholesterol, high-density lipoprotein [HDL]-cholesterol, and triglycerides) using colorimetric assays (Calgary Lab Service, Calgary, Alberta, Canada).
Knee Joint Histology
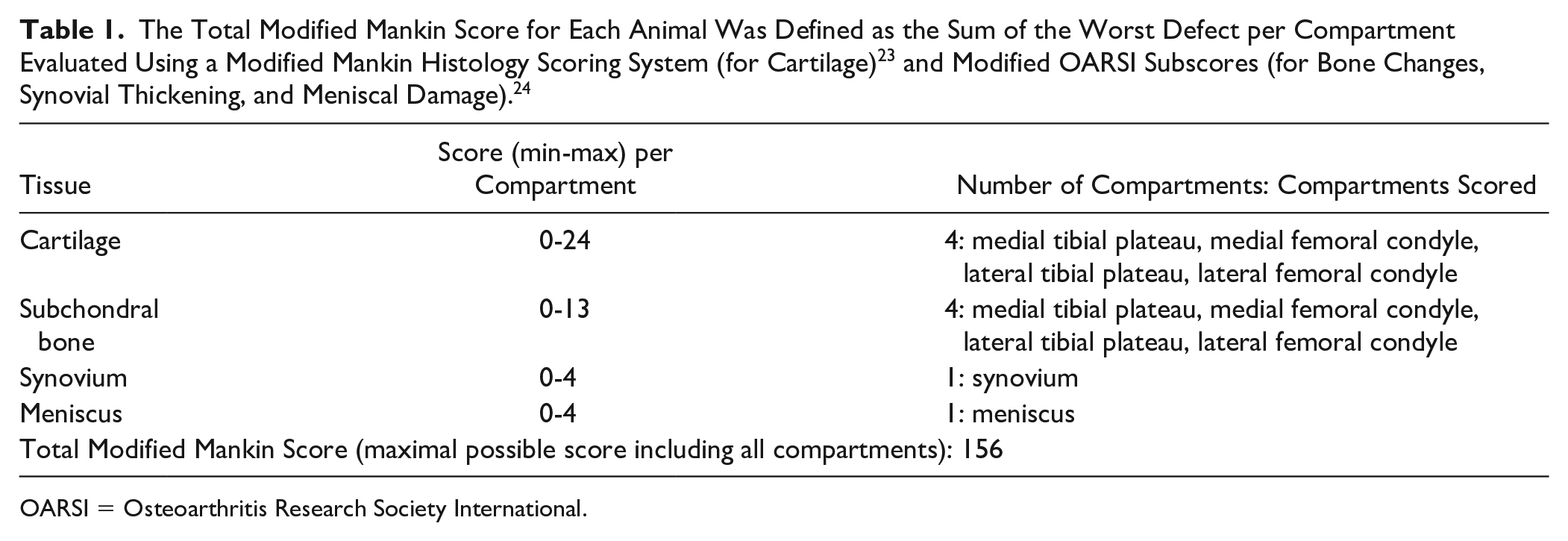
Following euthanasia, the left knee was harvested by cutting the femur and tibia/fibula 2 cm above and below the joint line, respectively. Muscles were removed, and joints were fixed in a 10% neutral buffered formalin solution for 14 days at room temperature. Knees were then decalcified, dehydrated in a graded series of alcohols, embedded in paraffin wax, and stored at room temperature until sectioning. 15 Serial sagittal plane sections of 10 µm thickness were obtained and mounted. Subsequently, every second section was stained sequentially with hematoxylin, fast green, and safranin-O. 15 Histological sections were quantified using a Modified Mankin Histology Scoring System, 23 and a Modified Osteoarthritis Research Society International (OARSI) histologic 24 subscore for bone changes, synovial thickening and meniscal damage for each joint. The worst defect for tibia, femur, synovium, and meniscus for each compartment (medial and lateral) were quantified per joint. The sum of all the scores per compartment was defined as the Total Modified Mankin Score for each animal ( Table 1 ) and were used for the statistical analyses. 12
OARSI = Osteoarthritis Research Society International.
Statistical Analysis
All data are presented as means ± 1 SEM (standard error of the mean). Nonparametric Kruskal-Wallis testing and Dunn’s post hoc testing with Bonferroni adjustment was used to determine differences in outcome variables between groups. We further investigated differences between time-points within groups using Wilcoxon testing (percent body fat and bone mineral density), and Friedman’s test with Dunn’s post hoc testing and Bonferroni adjustment (body mass, CISI, blood lipid profile). To assess the association between knee joint damage, body mass, percent body fat, and metabolic profile at week 24 in the experimental protocol, the Spearman Correlation Coefficient was calculated. Analyses were done using SPSS V25.0 software (IBM Corp, Armonk, NY, USA). Data were considered statistically significant at P < 0.05, 2-sided test.
Results
Knee Joint Histology
Prebiotic fiber supplementation, moderate aerobic exercise, and the combination of prebiotic fiber supplementation and moderate aerobic exercise did not prevent the progression of knee joint damage observed in rats fed a HFS diet. These interventions also did not accelerate the progression of knee joint damage (Total Modified Mankin Score: HFS = 50 ± 7.6, HFS + F = 47 ± 6.3, HFS + E = 50 ± 7.2, HFS + F + E = 53 ± 6.7; Fig. 2 ). Interestingly, the age-matched (36 weeks) chow-fed rats had similar joint damage scores as the HFS-fed rats (chow 51 ± 7.2). The tibia and femur had similar damage profiles, while the medial knee compartment was more affected than the lateral knee compartment ( Fig. 3 , and Table 2 ). We observed OA-like damage in both the cartilage and the subchondral bone ( Table 2 ). Damage to the subchondral bone was characterized by the replacement of the healthy bone marrow with fibrous and granulation tissue. We defined these damages as “bone marrow lesions” and identified them with white thick arrows in Figure 2 .
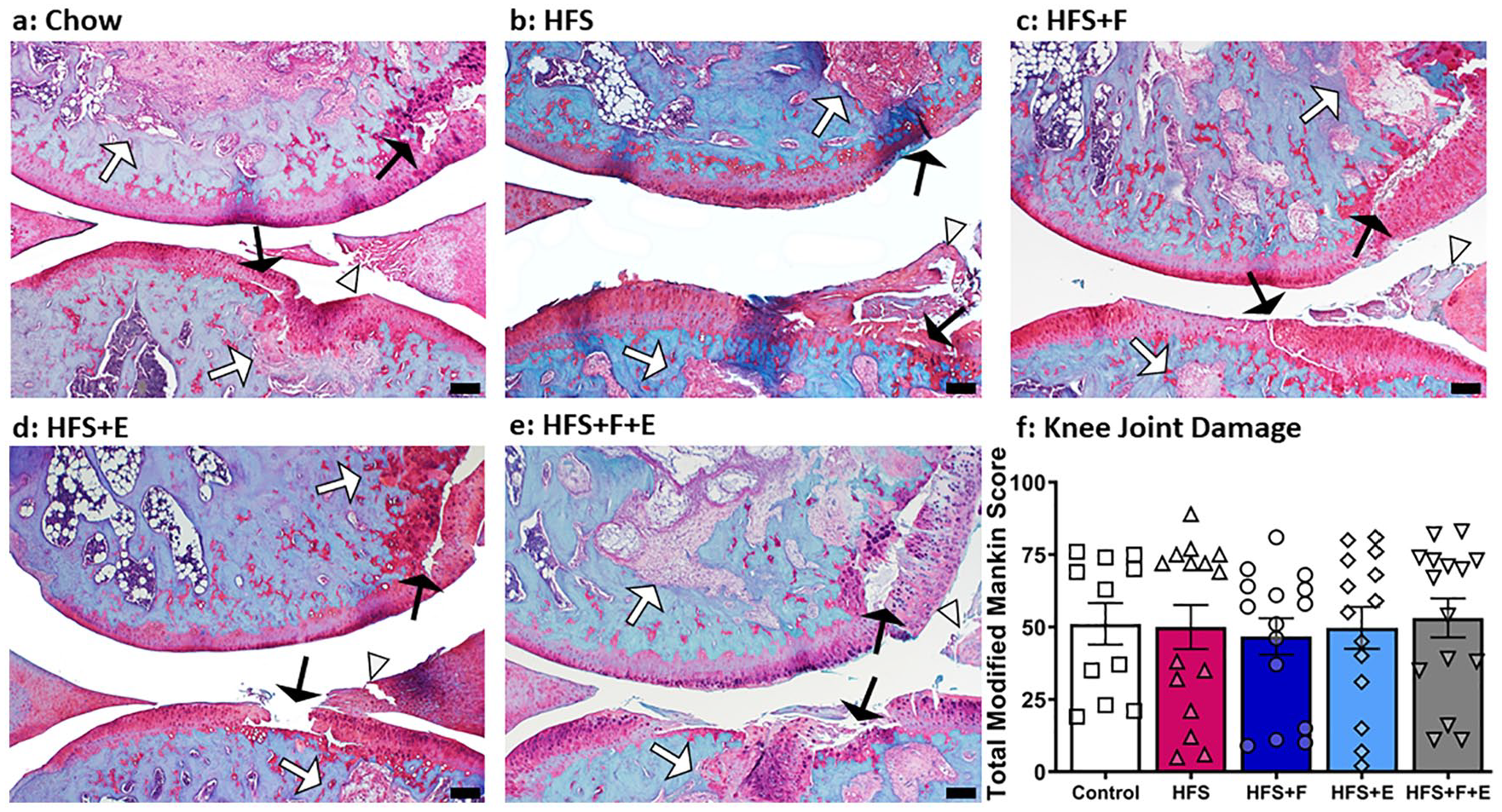
Knee joint damage. Images represent the mean rat joints for each experimental group. White thick arrows indicate subchondral bone damage (bone marrow lesion). Black thin arrows indicate cartilage defect. White arrow heads indicate meniscal damage. Chow, chow control diet; HFS, high-fat/high-sucrose diet; F, prebiotic fiber; E, aerobic exercise. Black bar = 200 μm. Values are means ± 1 SEM.
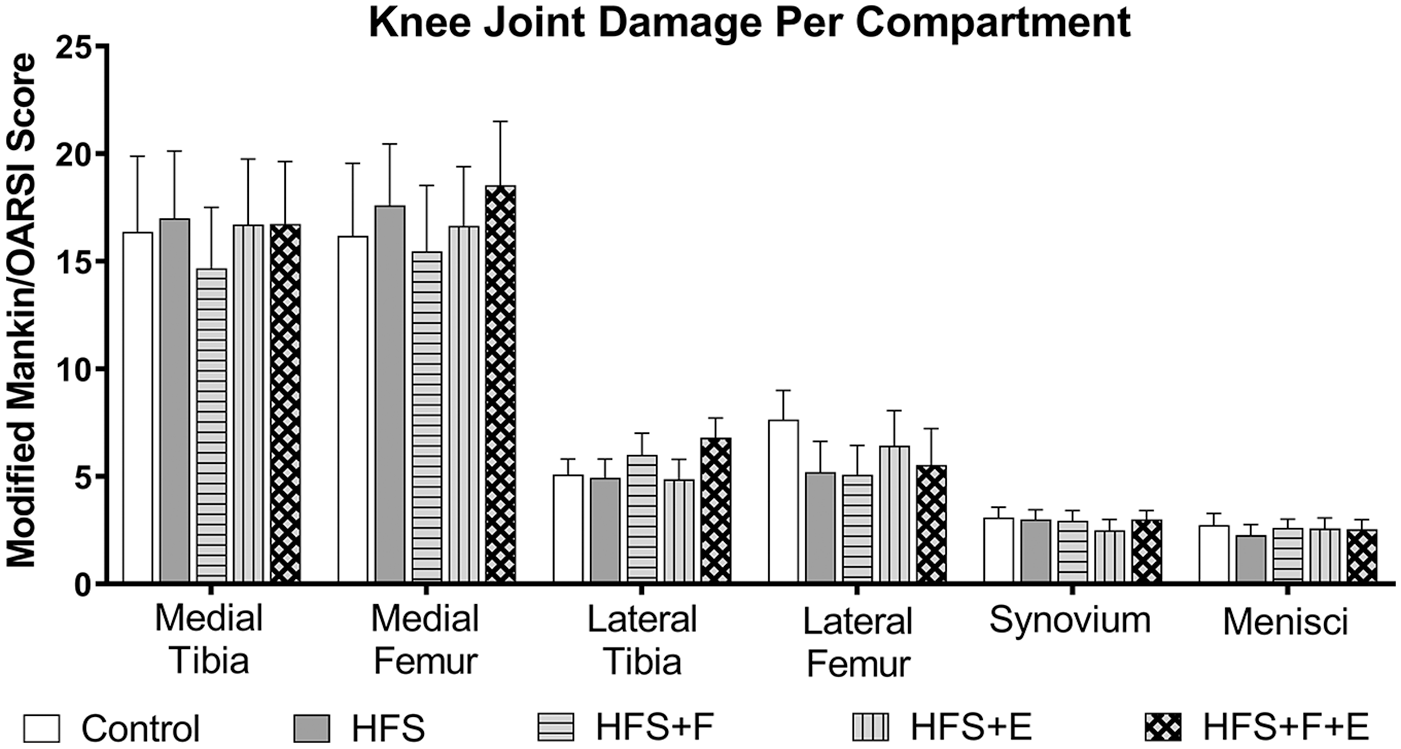
Knee joint damage per compartment. Medial knee compartment is more affected than the lateral compartment. Chow, chow control diet; HFS, high-fat/high-sucrose diet; F, prebiotic fiber; E, aerobic exercise. Values are means + 1 SEM.
Knee Joint Damage Subscores for Cartilage and Bone per Compartment. a
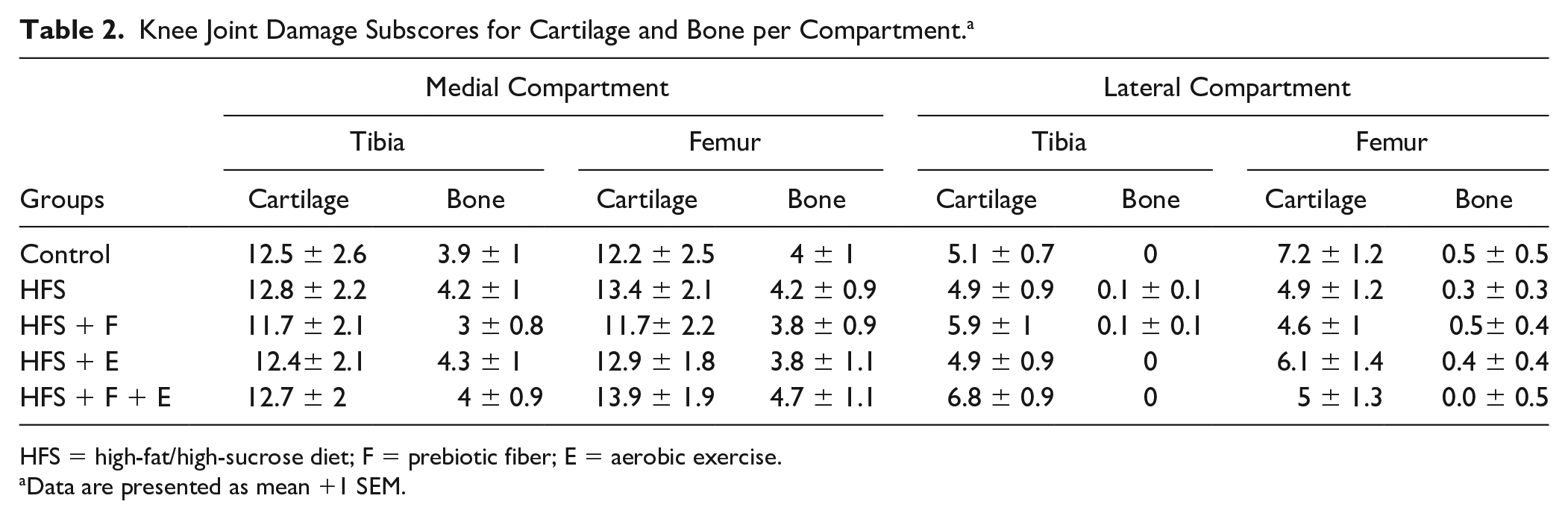
HFS = high-fat/high-sucrose diet; F = prebiotic fiber; E = aerobic exercise.
Data are presented as mean +1 SEM.
Body Mass and Body Composition
The mean body mass at the beginning of the experimental protocol was 489 g (SEM ±4.7). Body mass increased for all groups from week 1 to week 12 (before the intervention started). At week 12 of the experiment protocol, the HFS-fed rats were heavier (795 ± 11.0 g) than rats fed the control-chow diet (652 ± 15.6 g; P < 0.05). From week 12 to 13, there was a slight decrease in body mass, likely due the midpoint measurements performed at the end of week 12. In week 13, the exercise and fiber interventions started, and rats in the control-chow, HFS, and HFS + F group continued to gain weight, while rats in the HFS + E and HFS + F + E groups did not ( Fig. 4a ). At week 24, rats in the HFS + E and HFS + F + E had similar body mass as rats fed a control-chow diet (P > 0.05; 796 ± 25 g, 756 ± 25 g, and 731 ± 19 g, respectively), while rats in the HFS (943 ± 24 g) and HFS + F (893 ± 32 g) groups were heavier than control-chow fed rats (P < 0.05).
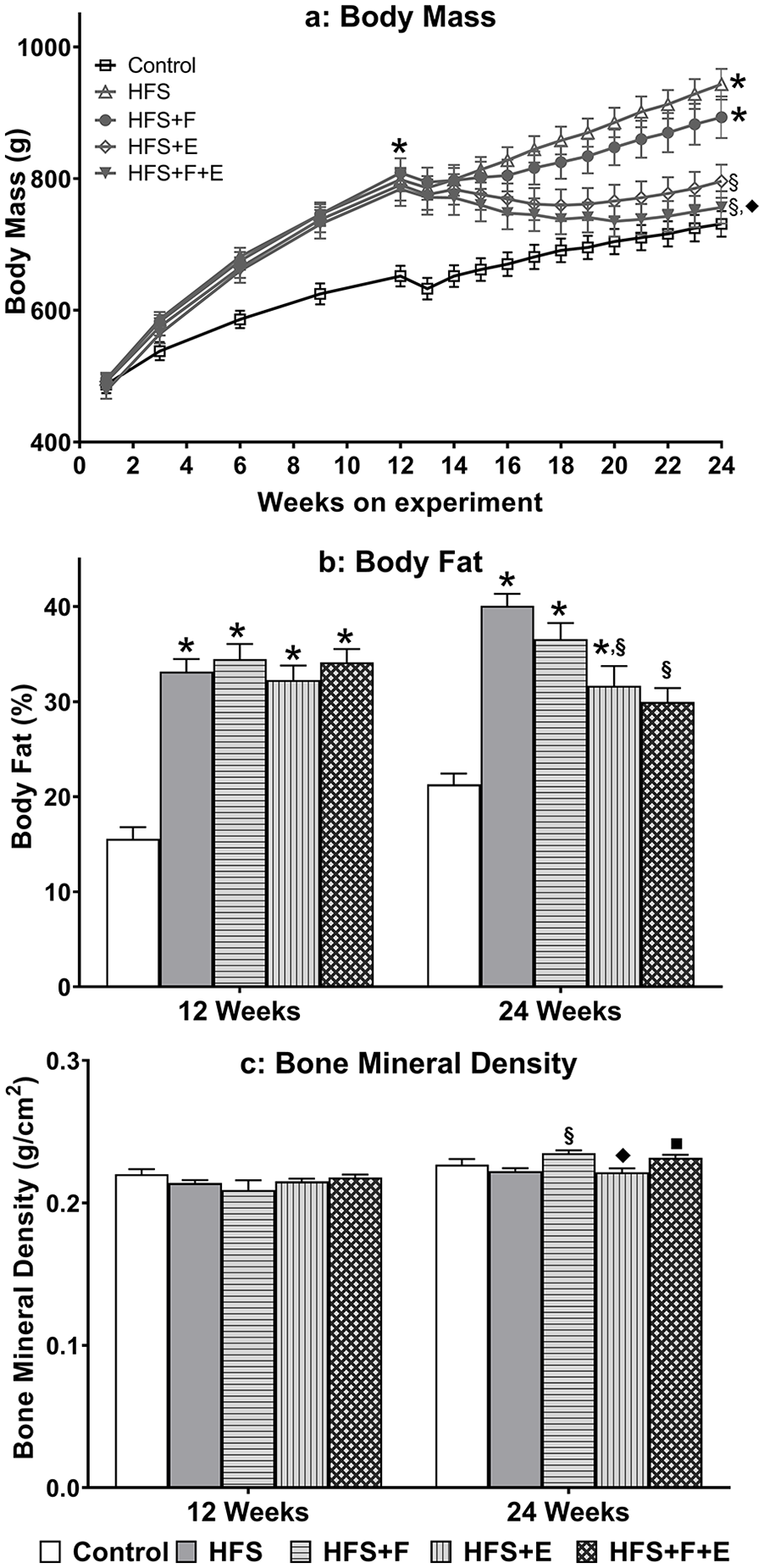
Body composition. (
At week 12 (prior to the interventions), the percent body fat for the rats fed a HFS diet was higher than that measured for the chow fed rats (P < 0.05, 33.5% and 15.5%, respectively, Fig. 4b ). At week 24, rats in the HFS + E and HFS + F + E groups had a lower percent body fat when compared with HFS group animals (P < 0.05). Rats on the control-chow diet, HFS diet, and in the HFS + F group had a significant increase in percent body fat from week 12 to week 24 (P < 0.05), while rats in the HFS + E did not have a change in percent body fat (P = 0.572), and rats in the HFS + F + E had a decrease in percent body fat (P = 0.003) from week 12 to week 24.
There were no differences in bone mineral density at week 12 ( Fig. 4c ). At week 24, there were no differences in bone mineral density between control-chow and HFS group rats. However, HFS + F group rats had a higher bone mineral density than HFS and HFS + E group rats (P < 0.05, Fig. 4c ). Rats in all groups had an increase in bone mineral density from week 12 to week 24 (P < 0.05).
There was no association between knee joint damage, percent body fat, and body weight at week 24 ( Table 3 and Supplementary Table S1 for group specific associations between knee joint damage, percent body fat, and body weight).
Relationship Between Knee Joint Damage (Total Modified Mankin Score), Body Mass, Percent Body Fat and Rat Metabolic Profile at Week 24 in the Experimental Protocol.
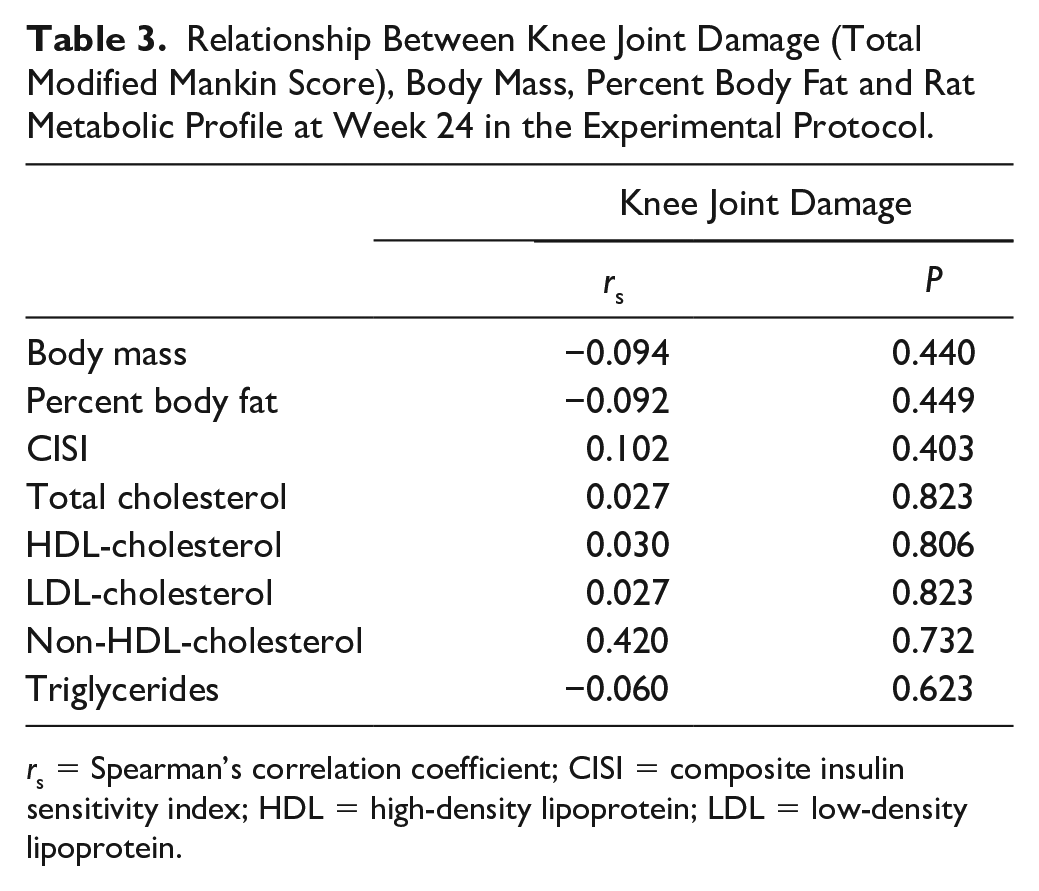
rs = Spearman’s correlation coefficient; CISI = composite insulin sensitivity index; HDL = high-density lipoprotein; LDL = low-density lipoprotein.
Systemic Metabolic Alterations
Blood Glucose and Serum Insulin
At baseline, insulin sensitivity was similar across the 5 experimental groups, ranging from 1 to 1.5 (P > 0.05, Fig. 5a ). At week 12, there was a significant decrease in insulin sensitivity for all groups fed a HFS diet when compared to rats fed a chow-control diet (P < 0.05). At week 24, HFS group rats had lower insulin sensitivity than rats in the chow-control group (P = 0.004). No differences were detected between HFS + F, HFS + E, HFS + F + E group animals when compared with chow-fed rats (P > 0.05); and when compared with the HFS group rats (P > 0.05).
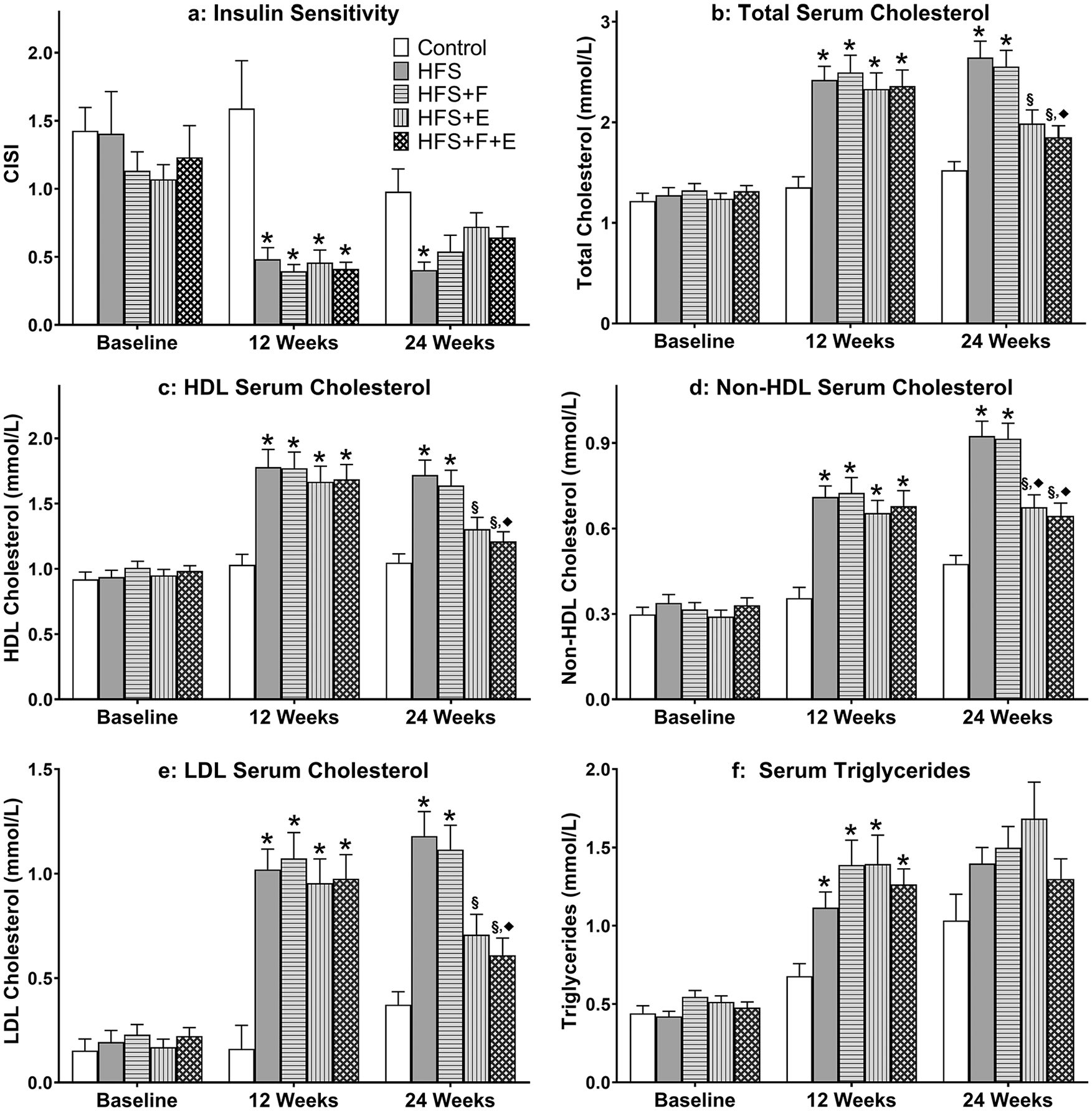
Metabolic profile. Composite insulin sensitivity index, and serum lipid profile for rats in the control, HFS, HFS + F, HFS + E, HFS + F + E groups at baseline, week 12, and week 24. *Indicates difference from chow group, while § indicates difference from HFS group, and ♦ indicates difference from HFS + F. HFS, high-fat/high-sucrose diet; F, prebiotic fiber; E, aerobic exercise. Values are means + 1 SEM.
Further analyses revealed that the rats fed the chow diet had the same insulin sensitivity at baseline and week 12 (P = 0.99), but their insulin sensitivity decreased from week 12 to week 24 (P = 0.005). Rats fed a HFS diet had a significant decrease in insulin sensitivity from baseline to week 12 (P < 0.05). Rats in the HFS, and HFS + F, HFS + E, and HFS + F + E groups had similar insulin sensitivity at weeks 12 and 24 (P > 0.05).
Baseline values for blood insulin and glucose were similar for all experimental group animals ( Fig. 6 ). At week 12, the blood insulin and glucose values were different for animals in the HFS group. Specifically, the glucose and insulin values for the HFS group animals did not recover to fasted values (time 0 minutes) at the end of the 120 minutes, while control group rats showed a recovery within 60 and 90 minutes. At week 24, blood glucose levels were similar for all groups. Insulin levels at the week 24 remained increased compared to baseline (week A) measurements. Additionally, at week 24, insulin levels for chow-fed rats did not recover within the 120 minutes testing period to pre-glucose challenge levels ( Fig. 6c and f ). There was no association between knee joint damage and CISI at week 24 ( Table 3 and Supplementary Table S1).
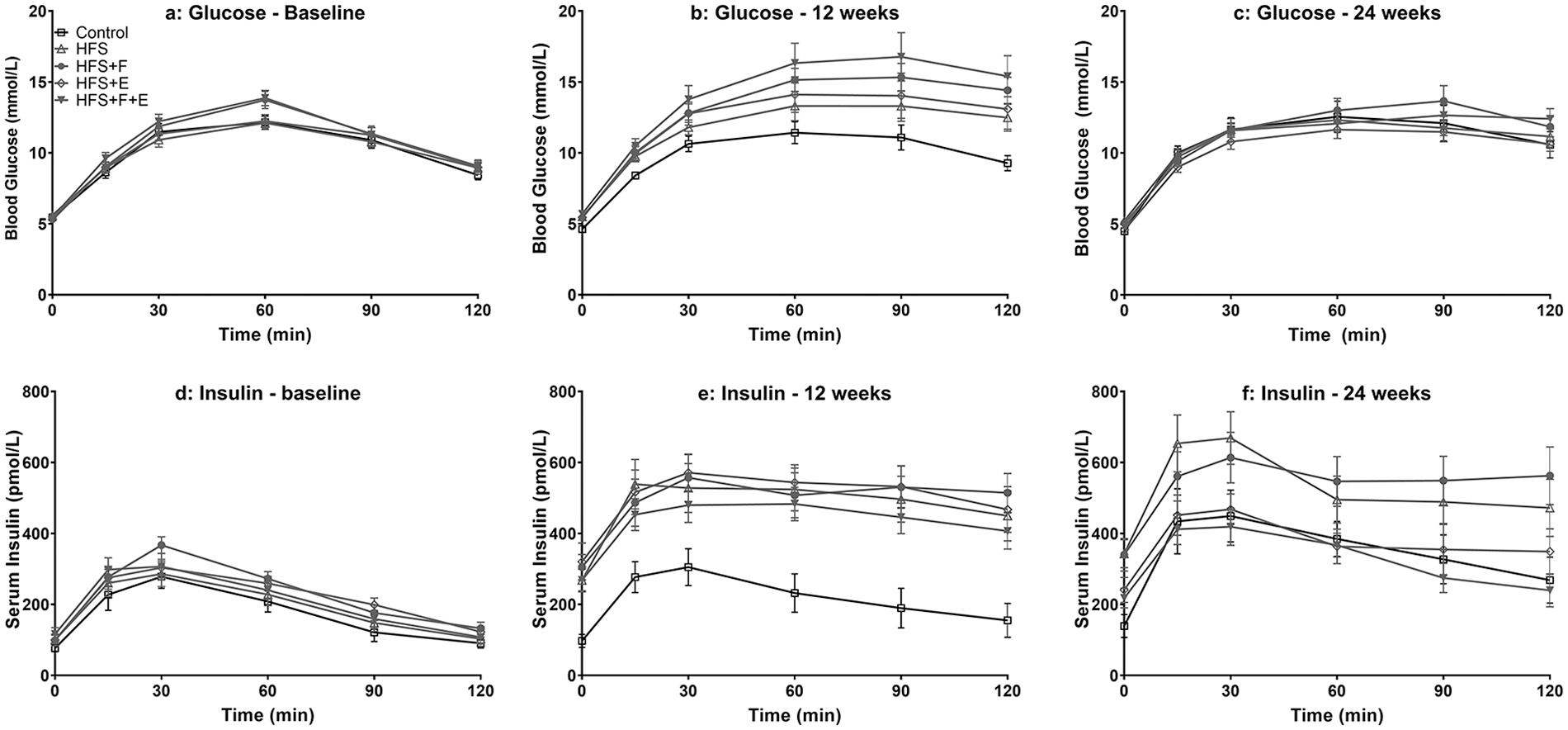
Glucose and insulin curves during glucose challenge at baseline, week 12, and week 24. HFS, high-fat/high-sucrose diet; F, prebiotic fiber; E, aerobic exercise. Values are means ± 1 SEM.
Serum Lipid Profile
At baseline, there were no differences in serum lipid profiles between groups. At week 12, the HFS-fed rats had an increase in serum total cholesterol, HDL cholesterol, non-HDL cholesterol, LDL cholesterol, and triglyceride levels compared with the control-chow fed rats (P < 0.05, Fig. 5 ). At week 24, with the exception of triglycerides, the HFS and HFS + F group rats had higher serum lipid levels than the control-chow-fed rats; while rats in the HFS + E and HFS + F + E groups had similar serum lipid levels as the chow-control rats (P > 0.05, Fig. 5 ). There were no differences in triglyceride levels between groups at week 24.
Serum total cholesterol, HDL-cholesterol, LDL-cholesterol, and triglyceride levels increased from baseline to week 12 for the HFS-fed groups (P > 0.05); while only HDL-cholesterol levels were increased from baseline to week 12 for control-chow-fed rats. From week 12 to week 24, serum total cholesterol did not change for rats in the control-chow fed, HFS, HFS + F, and HFS + E group animals, and decreased for the HFS + F + E group animals (P = 0.019). However, total cholesterol was increased for all experimental groups from baseline to week 24 (P < 0.05).
From week 12 to week 24, serum HDL-cholesterol levels did not change for rats in the control-chow fed, HFS, and HFS + F groups, and decreased for rats in the HFS + E (P = 0.024) and HFS + F + E (P = 0.006) groups. From week 12 to week 24, serum triglyceride levels did not change for rats in any of the 5 experimental groups (P > 0.05), but from baseline to week 24, triglyceride levels increased in all groups (P < 0.05).
There were no changes from baseline to week 12 for non-HDL cholesterol and LDL-cholesterol for the chow-fed rats. The non-HDL cholesterol and LDL-cholesterol were increased for the 4 HFS group rats from baseline to week 12. From week 12 to week 24, the non-HDL cholesterol did not change for rats in the HFS + E and HFS + F + E groups, while it increased for control, HFS, and HFS + F groups. From week 12 to week 24, serum LDL-cholesterol did not change for rats in the control-chow fed, HFS, HFS + F, and HFS + E groups, and decreased for HFS + F + E group rats. LDL cholesterol increased from baseline to week 24 for control-chow-fed rats. There was no association between knee joint damage and lipid profile at week 24 ( Table 3 and Supplementary Table S1).
Discussion
While there is evidence that early intervention with exercise and/or prebiotic fiber supplementation prevents the development of OA in a rat-model of diet-induced metabolic disturbance, 12 it remains unknown if these interventions remain successful in preventing or slowing the progression of OA when initiated at time points after the introduction of the HFS diet and once knee OA-like damage has been established to a certain degree. Therefore, the purpose of this study was to determine the effects of prebiotic fiber supplementation, aerobic exercise, and the combination of prebiotic fiber and aerobic exercise on the progression of knee OA-like damage in a HFS diet-induced rat-model of metabolic disturbance. We used a CD-Sprague-Dawley-model of diet-induced metabolic syndrome, that has been shown to produce reliable and consistent knee OA-like damage as early as four weeks following HFS diet exposure, 25 with consistent increase in OA severity at 12 weeks8,12 following HFS diet exposure. We subjected this model to an aerobic exercise intervention and a prebiotic fiber supplementation at 12 weeks following the HFS diet exposure to evaluate the effects of these interventions on the progression of knee OA-like damage. We showed that neither prebiotic fiber supplementation nor aerobic exercise affected the existing knee OA-like damage in this preclinical model, despite modestly improving selected markers of metabolic disturbance for some of the interventions. However, these results need to be considered in view of the fact that the chow fed control rats had similar knee OA-like damage as all the experimental HFS fed rats, suggesting that factors other than diet played a role in the progression of the knee OA-like damage observed in this study. Furthermore, all experimental groups showed a dichotomy in the knee OA outcomes: some animals showed severe cartilage and subchondral bone lesions and had high damage scores, while other animals did not show such lesions, and had low damage scores. However, none of the metabolic markers measured in this study showed an association with the observed knee joint damage, thus diminishing the possibility that differences in metabolic factors can explain the findings of this study. Additional research will be needed to address the dichotomous results found in this study.
Despite not being able to predict our dichotomous results, we speculate that the HFS diet might accelerate senescence in the CD-Sprague-Dawley rat, and that an accelerated senescence might be one reason why CD-Sprague-Dawley rats present with OA-like damage after a 12-week exposure to a HFS diet (starting at 12 weeks of age). Unpublished data from our lab revealed that nine out of ten CD-Sprague-Dawley rats fed a standard chow diet for 50 weeks had spontaneous knee OA-like damage based on gross anatomical inspection. Moreover, longevity in Sprague-Dawley rats is reduced compared to other rat strains, mainly because of degeneration processes associated with their rapid growth and high body weight.26,27 Preliminary data also indicates that CD-Sprague-Dawley rats have a more advanced biological age when compared to Wistar Han rats, and that after being fed a HFS diet over 12 weeks, these biological age differences are even more pronounced. 28 We speculate that the rapid growth in Sprague-Dawley rats is associated with an early senescence, thereby potentially explaining why a subset of our chow-fed control rats had spontaneous knee OA-like damage at 36 weeks of age. Further studies are required to test this hypothesis.
It has been reported previously that approximately 30% of the chow fed control group rats (36-40 weeks old) have similar Modified Mankin Scores for the knee joint as the HFS-fed rats. 29 The authors of that study attributed the similarities between control and HFS group rats to meniscal damage of the chow fed rats via unknown trauma. In our study, control chow-fed group rats (36 weeks old at sacrifice) had similar mean Total Modified Mankin Scores and similar knee joint damage characteristics as rats in the HFS fed group. Therefore, it appears that the HFS diet accelerates knee joint damage in CD-Sprague-Dawley rats through pathways identified earlier (24-week-old rats)8,12 in the short term, but does not seem to affect in the same way all the knee joints in older (36 weeks) rats.
Our 36-week-old rats fed a chow diet displayed signs of metabolic disturbance, exhibiting decreased insulin sensitivity and increased non-HDL cholesterol levels compared to when they were 24 weeks old. Serum triglyceride levels in the 36-week-old chow-fed rats were similar to those of the 24-week-old HFS-fed rats. It is known that aging it is a contributor to metabolic dysregulation, 30 therefore, one might hypothesize that CD-Sprague-Dawley rats have age-related signs of metabolic disease that may lead to OA-like damage. However, markers of metabolic disturbance quantified in this study did not correlate with the damage observed in the knee joint. The metabolic disturbance in the chow fed rats may be related to the sedentary behavior imposed by the laboratory environment. Laboratory rats presented with a running wheel are highly active, and they will exercise voluntarily,31,32 running at good speeds (40-50 m/min) for long distances ranging from 1.5 to 2.5 km per day.
Rats fed a HFS diet had higher HDL cholesterol levels compared to baseline values or to the chow fed rats. The same feature has been observed in Wistar rats fed a HFS diet when compared with Wistar rats fed a standard laboratory diet over 24 weeks. 33 HDL replacement therapy has been suggested to be a good strategy to lower cardiac risks associated with metabolic dysregulation, 34 and it has been suggested that high serum HDL levels may play a protective role in the development of osteoarthritis and osteoporosis, 35 thus we speculate that increases in HDL levels in our model might be an adaptive mechanism to the metabolic dysregulation.
Rats in the HFS + E and HFS + F + E groups had a better metabolic profile at week 24 compared with HFS group rats. However, metabolic markers did not return to baseline values, suggesting that these rats still had some metabolic disturbance. It has been shown that elevated plasma cholesterol levels, as seen in our study, trigger oxidative stress in mitochondria and degradative changes in chondrocytes that, in combination, are thought to lead to OA development and progression. 36 Additionally, drugs that lower lipid levels inhibit OA development in mouse models of atherosclerosis. 37 Perhaps, combining the exercise intervention, or the exercise plus fiber intervention, with a change in diet (e.g., switch to a chow diet), may have allowed for a return to the basal metabolic profile levels, and may have prevented the progression of knee joint damage in this rat model of metabolic OA.
In summary, moderate aerobic exercise, and the combination of prebiotic fiber supplementation and moderate aerobic exercise improved markers of metabolic disturbance. However, in view of the present results (no differences between the control and any of the HFS groups—HFS, HFS + F, HFS + E, and HFS + F + E) when animals were sacrificed at 36 weeks, and those obtained in our earlier study (significant differences between the control animals and intervention [HFS + F, HFS + E, HFS + F + E] group animals compared to the HFS-fed animals) when animals were sacrificed at 24 weeks, 12 we conclude that the HFS diet in this animal model accelerates the progression of spontaneous knee OA. However, with aging, the effects of the HFS diet on the knee are eclipsed and all knees show damage, independent of diet (chow vs. HFS) or intervention (prebiotic fiber supplementation, exercise, prebiotic fiber supplementation combined with exercise). Prebiotic fiber supplementation, moderate aerobic exercise, and the combination of prebiotic fiber supplementation and moderate aerobic exercise did not seem to slow/stop; nor did it seem to accelerate, the progression of knee OA-like damage. Therefore, exercise and prebiotic fiber supplementation may be good strategies for populations with metabolic knee OA who chose exercise and/or prebiotic to alleviate metabolic disturbances. However, these conclusions need to be considered in view of the fact that the chow-fed rats had similar knee OA-like damage as the HFS-fed rats, and that there was a distinct dichotomy in knee joint damage scores across all experimental groups without a satisfactory explanation why some animals were well protected from damage while others were not. Additional research is needed to clarify why a subset of rats in all experimental groups was protected from developing knee OA-like changes, while another subset was not.
Study Limitations
The experiments described in this manuscript have limitations. The primary knee joint outcome in this manuscript was cross-sectional rather than longitudinal (e.g., using micro-computed tomgoraphy); the latter would likely have been more revealing in describing the progression of the knee damage. Additionally, it would be important to describe and characterize the effect of moderate exercise and prebiotic fiber supplementation on other joints (such as shoulders and hips) and tissues not evaluated here, such as muscles and tendons. Another limitation of the studies presented in this manuscript is that only male CD-Sprague-Dawley rats were evaluated. Due to the higher incidence of OA in women, future studies should consider incorporating females. Based on previous findings,8,12,25 it appears that the HFS diet accelerates the natural occurrence of knee OA-like damage in young CD-Sprague-Dawley rats, but is not its only cause. Therefore, caution should be taken when designing future studies aimed at investigating the metabolic OA phenotype in this particular animal model. Additionally, we did not investigate gut microbiota dysbiosis, systemic and local inflammatory markers, biomechanical factors, knee function, or pain in this cohort; factors that should be included in future studies to help elucidate potential mechanisms underlying knee OA development in CD-Sprague-Dawley rats.
Supplemental Material
CART-20-0148_Supplementary_Material_Rescue_Study_R_02 – Supplemental material for Prebiotic and Exercise Do Not Alter Knee Osteoarthritis in a Rat Model of Established Obesity
Supplemental material, CART-20-0148_Supplementary_Material_Rescue_Study_R_02 for Prebiotic and Exercise Do Not Alter Knee Osteoarthritis in a Rat Model of Established Obesity by Jaqueline Lourdes Rios, David A. Hart, Raylene A. Reimer and Walter Herzog in CARTILAGE
Footnotes
Acknowledgments and Funding
The authors thank Baaba Sekyiwaa Otoo, Graham Zeno MacDonald, Ian Curtis Smith, Kevin Rudi Boldt, Ruth-Anne Seerattan, Stela Maris Mattiello, and Venus Joumaa for technical contributions to this study. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Institutes of Health Research # RT736475 (WH) and MOP 115076 (RAR), the Canada Research Chair Programme (WH), the Alberta Innovates Health Solutions Osteoarthritis Team Grant (DAH, WH), Alberta Innovates Health Solutions, Killam Foundation (WH), Alberta Innovates (JLR), and the Ministry of Education, Brazil (CAPES Foundation Grant 13157-13-2, JLR). The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RAR has received honoraria from Beneo GmbH for presentations distinct from the current work. JLR, DAH, and WH declare that they have no conflicts of interest to disclose.
Ethical Approval
All experiments were approved by the University of Calgary Life and Environmental Sciences Animal Care Committee (AC16-0130).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Trial Registration
Not applicable.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.