Abstract
Objective
Though multiple high-level comparative studies have been performed for matrix-assisted autologous chondrocyte transplantation (MACT), quantitative reviews synthesizing best-available clinical evidence on the topic are lacking.
Design
A meta-analysis was performed of prospective randomized or nonrandomized comparative studies utilizing MACT. A total of 13 studies reporting 13 prospective trials (9 randomized, 5 nonrandomized) were included (658 total study participants at weighted mean 3.1 years follow-up, range 1-7.5 years).
Results
Reporting and methodological quality was moderate according to mean Coleman (59.4 SD 7.6), Delphi (3.0 SD 2.1), and MINORS (Methodological Index For Non-Randomized Studies) scores (20.2 SD 1.6). There was no evidence of small study or reporting bias. Effect sizes were not correlated with reporting quality, financial conflict of interest, sample size, year of publication, or length of follow-up (P > 0.05). Compared to microfracture, MACT had greater improvement in International Knee Documentation Committee (IKDC)-subjective and Knee Injury and Osteoarthritis Outcome Pain Subscale Score (KOOS)-pain scores in randomized studies (P < 0.05). Accelerated weight-bearing protocols (6 or 8 weeks) resulted in greater improvements in IKDC-subjective and KOOS-pain scores than standard protocols (8 or 11 weeks) for MACT in randomized studies (P < 0.05) with insufficient nonrandomized studies for pooled analysis.
Conclusions
Compared to microfracture, MACT has no increased risk of clinical failure and superior improvement in patient-reported outcome scores. Compared to MACT with standardized postoperative weight-bearing protocols, accelerated weight-bearing protocols have no increased risk of clinical failure and show superior improvement in patient-reported outcome scores. There is limited evidence regarding MACT compared to first-generation autologous chondrocyte implantation, mosaicplasty, and mesenchymal stem cell therapy without compelling differences in outcomes.
Keywords
Introduction
High-grade chondral defects of the knee are frequently debilitating 1 and, if left untreated, can lead to early osteoarthritis. 2 Brittberg et al. first described using cultured autologous chondrocytes for the treatment of high-grade articular cartilage defects. 3 This technique eventually became known as first-generation autologous chondrocyte implantation (ACI) and has been shown to be an effective means of treating focal cartilage defects. 4 Follow-up studies of first-generation ACI showed significant clinical improvements in patients. 5 However, the original procedure was technically difficult because it required the harvested autologous chondrocytes be implanted in solution form under a periosteal patch secured with sutures upon the chondral defect. 3 Furthermore, rehabilitation times for these procedures were extensive, often more than a year before return to sports activity. 6
Matrix-assisted autologous chondrocyte transplantation (MACT) techniques were developed to address some of the shortcomings of first-generation ACI. MACT involves seeding the cultured chondrocytes onto a scaffold or membrane composed of a variety of materials. 6 In 2006, membrane-associated cartilage implantation (MACI) was the first MACT therapy to receive Food and Drug Administration approval for treatment of osteochondral and chondral lesions of the knee. 7 Though multiple high-level evidence studies have been performed for MACT,3,8-10 currently there is no meta-analysis synthesizing best-available clinical evidence to assist providers with treatment decisions. Recent reviews on the topic have been limited to systematic reviews without application of meta-analytic techniques.11-13
The purpose of this study was to systematically review and synthesize high-level clinical evidence on MACT trials. The study seeks to answer several questions: (1) Are clinical outcomes equivalent for standard versus accelerated weight bearing after MACT? (2) Is MACT superior to microfracture? (3) Is MACT superior to mesenchymal stem-cell therapy? (4) Is MACT superior to first-generation ACI? (5) Is MACT superior to mosaicplasty? (6) Does chondrocyte dose affect MACT outcomes?
Methods
Search Strategy and Study Selection
We conducted a systematic review based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA)
14
guidelines. An initial PubMed search using the terms (autologous chondrocyte) AND (transplantation OR implantation) in June 2018 yielded 1,231 results for original research studies reported in English. The Embase, SCOPUS, and Google Scholar databases were cross-referenced. After screening of study titles and abstracts, 1,108 articles were excluded due to failure to meet one or more study inclusion criteria (
Systematic Review Inclusion Criteria.

Therefore, a total of 13 studies reporting 13 trials were included in the final analysis (
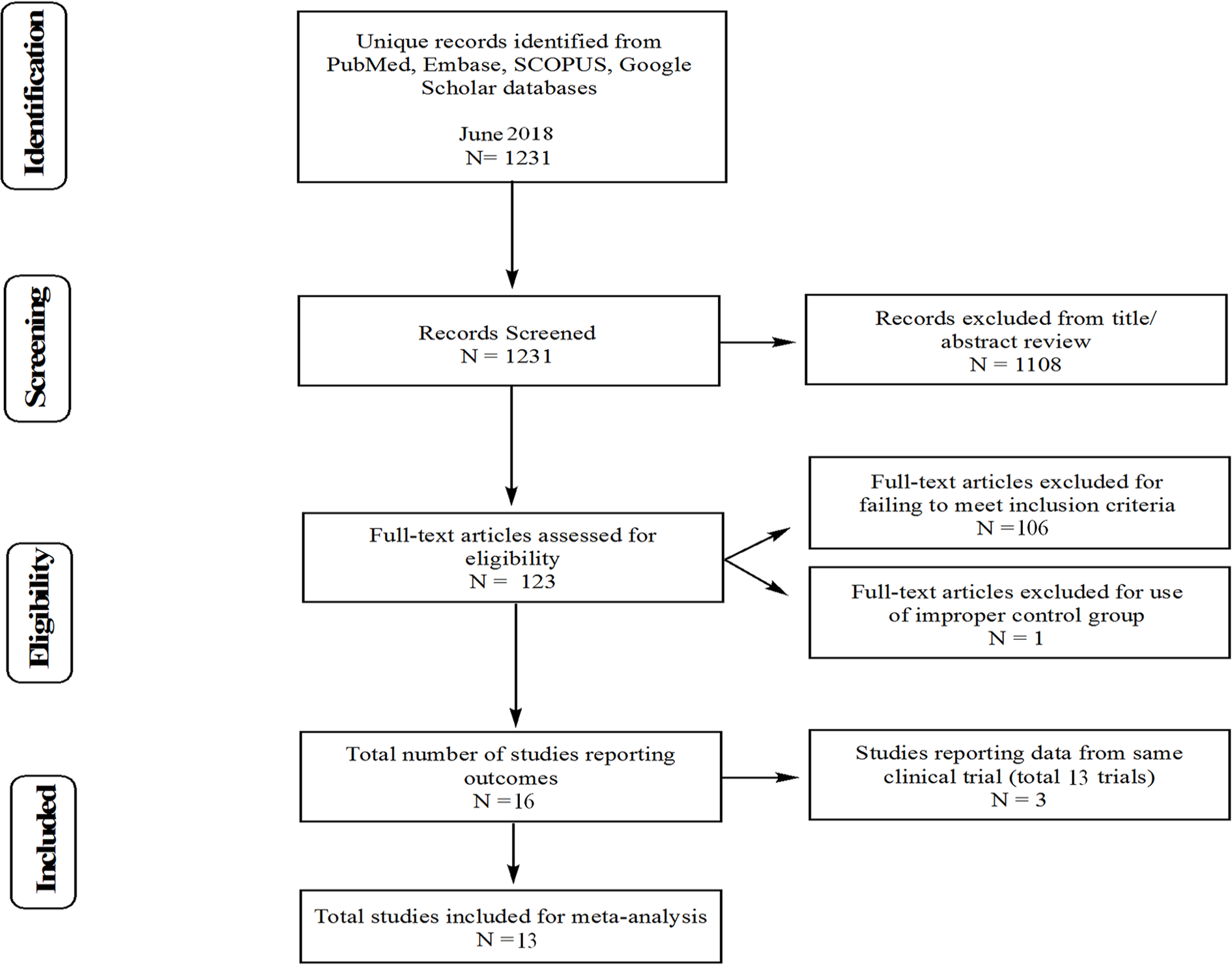
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. Fourteen studies were identified for inclusion.
Outcome scores of interest utilized in the analysis were the Knee Injury and Osteoarthritis Outcome Pain Subscale Score (KOOS-pain), 23 International Knee Documentation Committee (IKDC) scores, and rates of clinical treatment failure or reoperation. Both IKDC and KOOS-pain scores are acceptable measures of knee disability in cartilage repair patients. 24 Data were directly abstracted from the articles with the exception of KOOS-pain results reported by Akgun et al. 25 and Ebert et al. 10 ; in these studies, reported KOOS-pain values had an abnormally small level of dispersion for all groups at all time points; this anomaly likely represented a common reporting error in which standard error of the mean was inadvertently reported rather than standard deviation. To address this issue, the mean reported standard deviation (SD) for KOOS-pain from the remaining trials (SD = 13) was assigned to avoid excessive weighting in the meta-analysis.
Risk of Bias and Quality Assessment
Several quality assessments were used to determine risk of bias and study reporting quality. The modified Coleman Methodology score 26 (Appendix B), the Methodological Index for Non-Randomized Studies (MINORS) score 27 (Appendix C), and the Delphi criteria 28 (Appendix D) were all assessed by 2 independent reviewers (AD and SP). The modified Coleman Methodology score assessed the likelihood that biases and confounding factors were absent in the study (85-100 excellent; 70-84 good; 55-69 fair; and ≤54 poor). The MINORS scoring system was used to assess the overall quality of nonrandomized trials (scored 0 to 24). The Delphi criteria assessed the overall quality of randomized clinical trials using a 9-item checklist with scores of 1, −1, and 0 corresponding to “yes,” “no,” and “don’t know,” respectively. Agreement statistics were determined for all quality assessment tools. Discrepant scores were resolved by a third author. Reporting of a financial conflict of interest (COI), specifically funding or royalties received by one or more study authors, a manufacturer of MACT, or the control therapy, was also assessed.
Statistical Analysis
All statistical tests were performed with STATA 13.1 (StataCorp, College Station, TX). Interrater agreement for methodological quality scores was reported as the interclass correlation coefficient (ICC). Random effects meta-analyses using the Dersimian and Laird method 29 were performed for each outcome (risk of clinical failure, risk of repeat operation, improvement in IKDC scores, and improvement in KOOS-pain scores) and each treatment-control combination. Due to fundamental differences in study design between randomized and nonrandomized studies, separate meta-analyses were performed for each study design. Effect heterogeneity was assessed using the I2 measure as described by Higgins et al. 30 For studies reporting either IKDC or KOOS-pain but not both outcome scores, a formula was derived (Appendix A) from the remaining studies reporting both IKDC and KOOS-pain to allow estimation of one score from the other. A positive transformation was applied (Appendix A) to allow inclusion of studies with zero failures or reoperations in both treatment and control groups in the statistical analysis. Treatment-control combinations with fewer than 2 dedicated randomized trials or 2 dedicated nonrandomized trials were not eligible for meta-analysis and best-available evidence for these studies are discussed qualitatively. Multiple assessments of bias were performed (Appendix A) including publication bias (via Egger’s test and visual inspection of funnel plots), bias due to reporting or methodological quality, and bias due to financial conflict of interest, sample size, year of publication, or length of follow-up
Funding Source
The authors received no funding for this study.
Results
Reporting Quality and Risk of Bias
Methodological quality was moderate with a mean Coleman score of 59.4 (SD 7.6, range 50-70), Delphi score of 3.0 (SD 2.1, range −2 to 6), and MINORS of 20.2 (SD 1.6, range 18-23). Interrater agreement for the Coleman score was good (ICC 0.79), and the interrater agreement was fair for the MINORS (ICC = 0.55) and Delphi scores (ICC = 0.48). The most common methodological shortcomings as assessed by these scoring systems were lack of blinding of either the patient (11/13 studies) or care provider (13/13 studies) and lack of prospective sample size estimation (7/13 studies).
There were no significant correlations between study effect size (as defined by difference between treatment and control groups in KOOS, IKDC, reoperation rates, or failures rates) and methodological quality scores, financial conflict of interest, sample size, year of publication, or length of follow-up (P > 0.05). There was no evidence of small study bias in any of the meta-analyses in the current study per Egger’s test of bias (P > 0.05, all analyses) or visual inspection of funnel plots with IKDC or KOOS-pain as outcome measures.
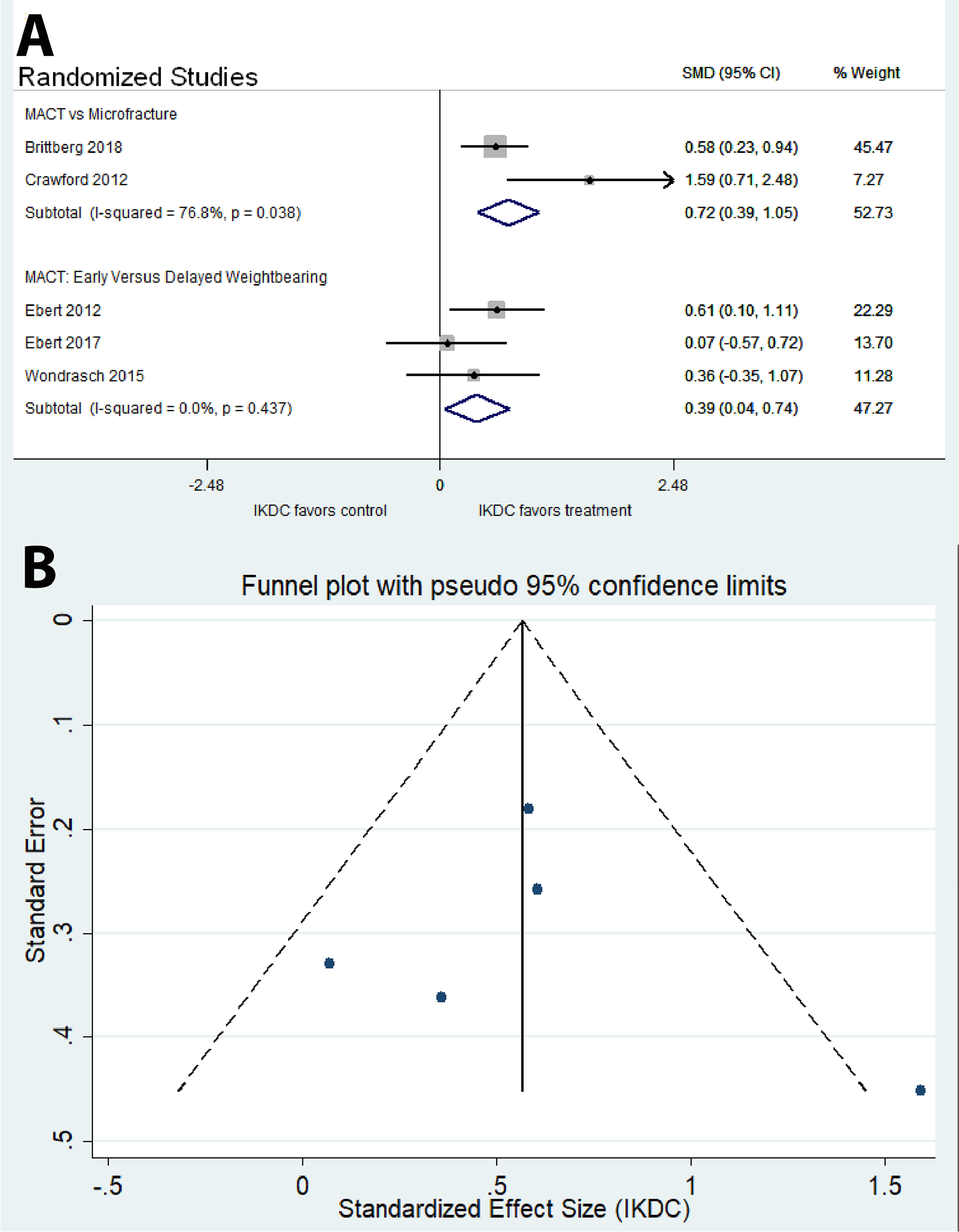
MACT versus Microfracture
Four trials compared clinical outcomes of MACT versus microfracture (
Studies of MACT versus Microfracture and MACT with Differing Weight-Bearing Protocols.
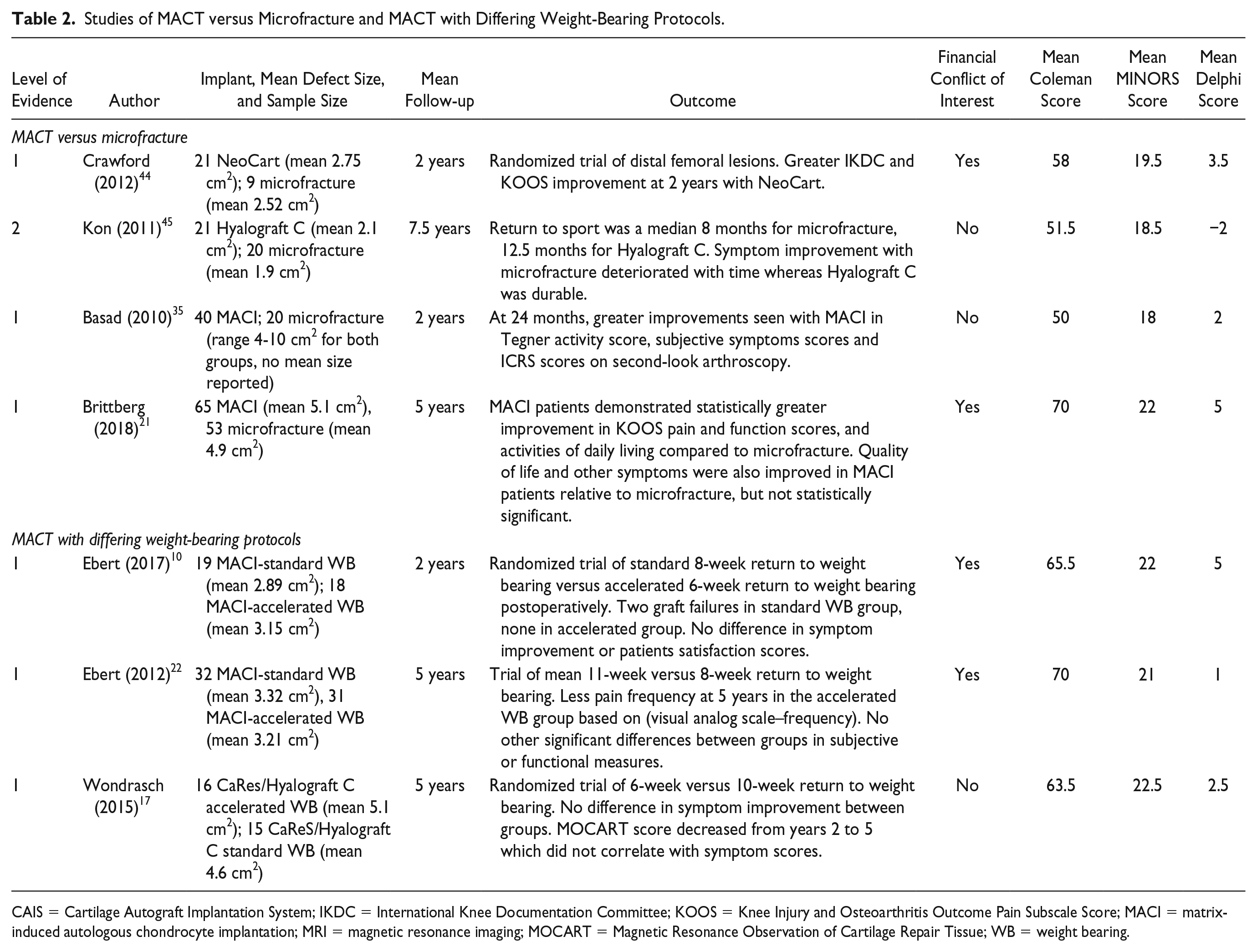
CAIS = Cartilage Autograft Implantation System; IKDC = International Knee Documentation Committee; KOOS = Knee Injury and Osteoarthritis Outcome Pain Subscale Score; MACI = matrix-induced autologous chondrocyte implantation; MRI = magnetic resonance imaging; MOCART = Magnetic Resonance Observation of Cartilage Repair Tissue; WB = weight bearing.
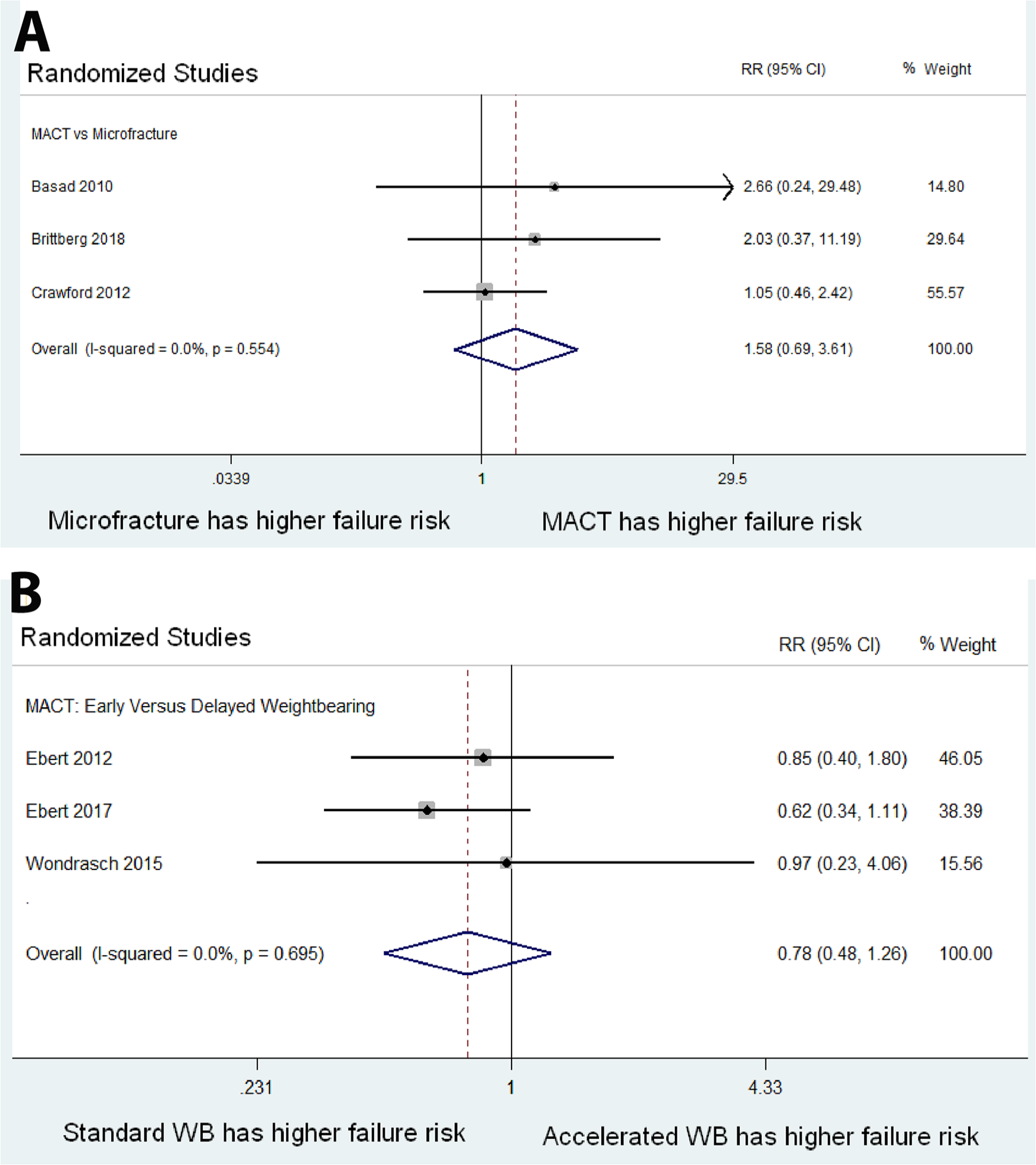
Meta-analysis of risk of treatment failure among randomized studies. Too few nonrandomized studies reported treatment failure to perform a meta-analysis of nonrandomized studies (not shown). (
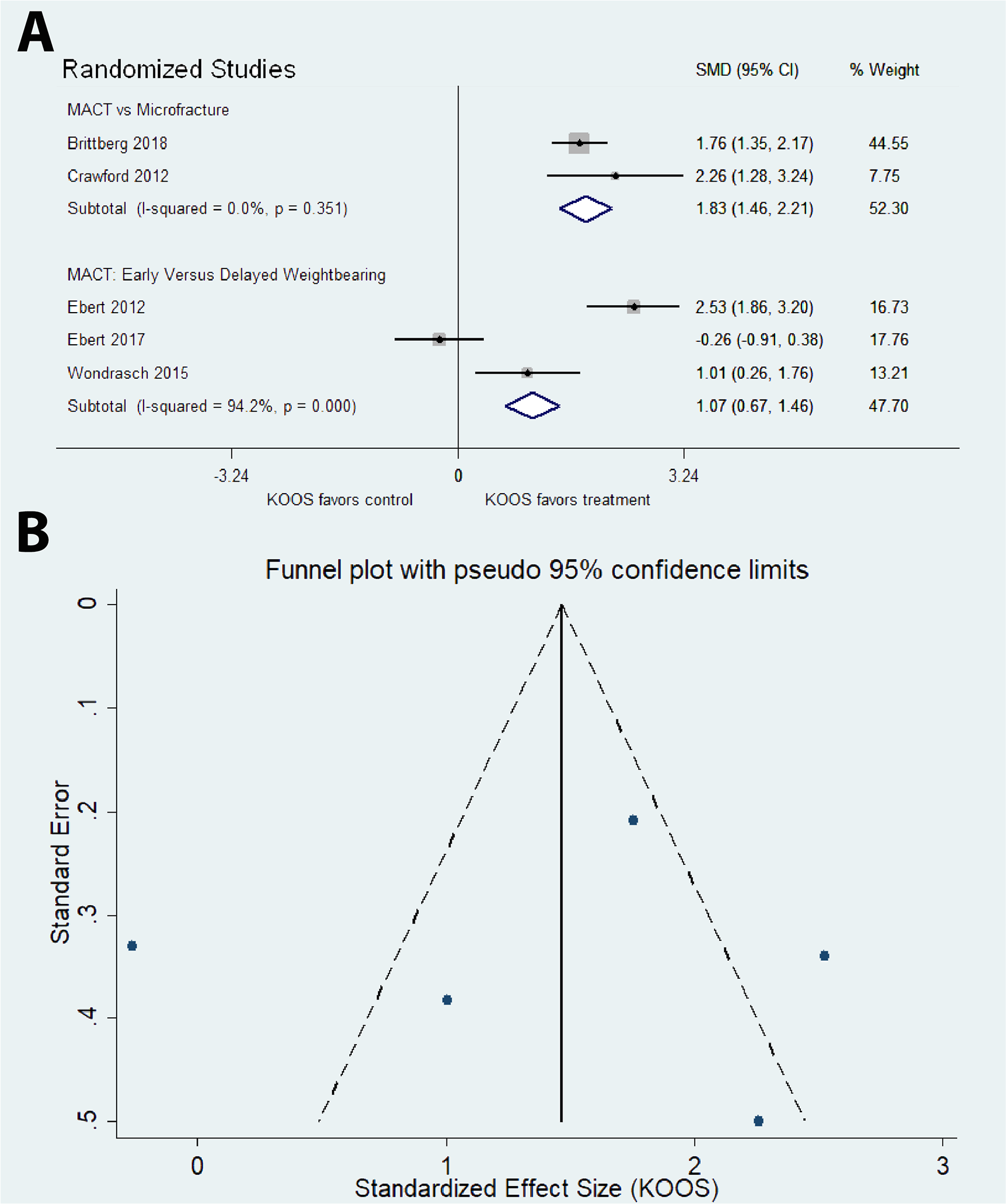
(
(
MACT with Early versus Delayed Weight Bearing
Three trials compared early versus delayed weight bearing (WB) protocols following MACT (
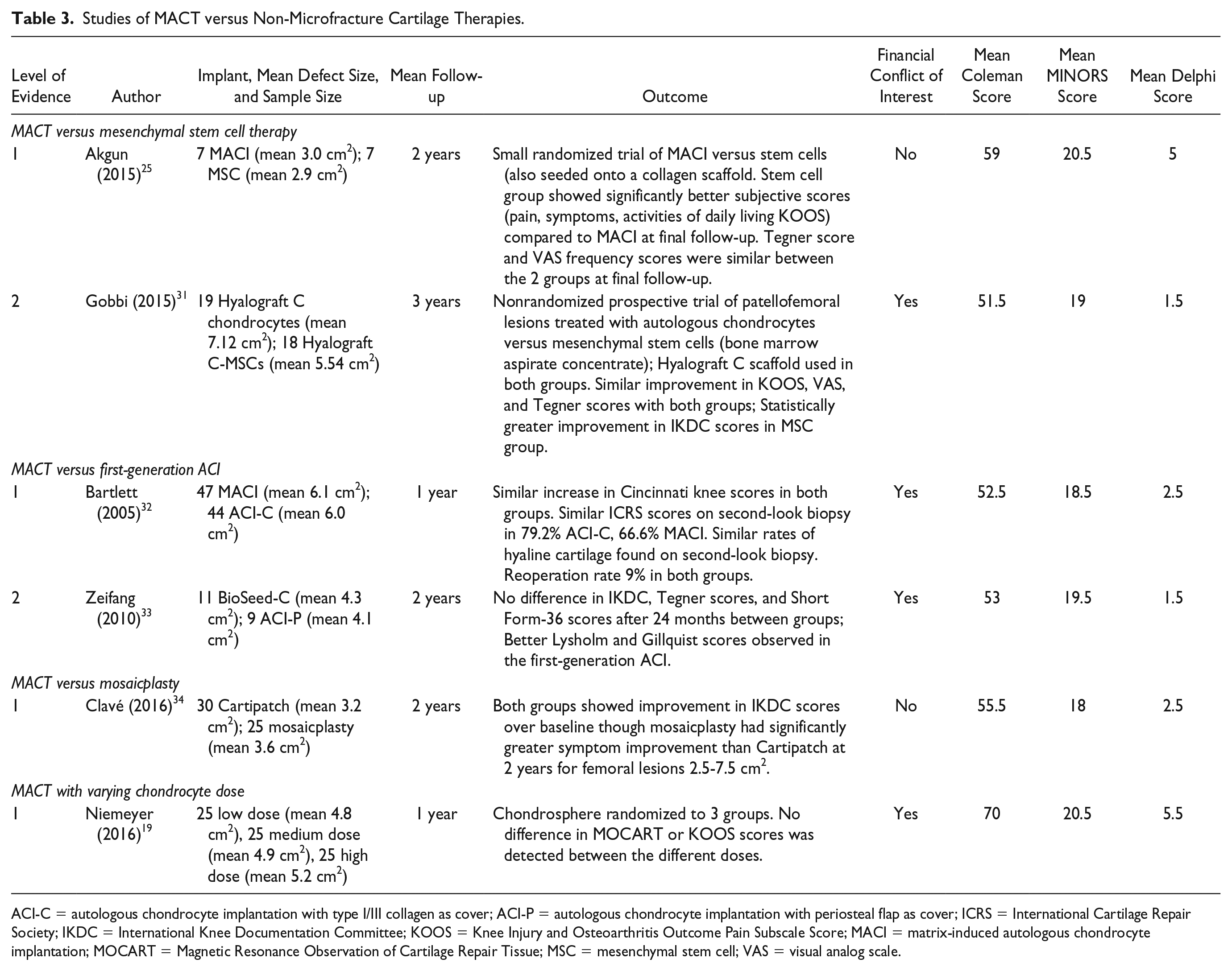
MACT versus Mesenchymal Stem Cell Therapy
Two trials compared outcomes of MACT against mesenchymal stem cell therapy (MSC) (
Studies of MACT versus Non-Microfracture Cartilage Therapies.
ACI-C = autologous chondrocyte implantation with type I/III collagen as cover; ACI-P = autologous chondrocyte implantation with periosteal flap as cover; ICRS = International Cartilage Repair Society; IKDC = International Knee Documentation Committee; KOOS = Knee Injury and Osteoarthritis Outcome Pain Subscale Score; MACI = matrix-induced autologous chondrocyte implantation; MOCART = Magnetic Resonance Observation of Cartilage Repair Tissue; MSC = mesenchymal stem cell; VAS = visual analog scale.
MACT versus First-Generation ACI
Two trials (
MACT versus Mosaicplasty
One study by Clavé et al.
34
evaluated MACT against mosaicplasty (
MACT at Varying Chondrocyte Doses
One trial by Niemeyer et al.
19
evaluated different chondrocyte dosing when performing MACT (
Discussion
The purpose of this meta-analysis was to assess the available literature and determine (1) whether MACT is superior to other procedures, (2) whether clinical outcomes are altered with accelerated WB postoperatively after MACT, and (3) whether outcomes differ with different MACT chondrocyte doses. Our analyses reveal that accelerated WB (as early as 6 weeks) is safe for MACT, and that MACT is superior to microfracture for defects up to 10 cm2 at up to 7.5 years follow-up.
Three trials evaluating accelerated WB after MACT were included in our review.10,17,22 All 3 studies concluded that accelerated return to WB is noninferior to standard return to WB with return to weight bearing as soon as 6 weeks postoperatively and follow-up times ranging from 2 to 5 years. Of note, in all 3 studies, there were no major differences in magnetic resonance imaging (MRI) findings postoperatively with the exception of graft border integration and surface contour scoring better in the accelerated (6-week) weight-bearing group in Ebert et al. 10
We found that MACT is superior to microfracture for equivalent sized defects (up to 10 cm2). 35 The evidence clearly favors MACT with regard to patient pain and functional scores. The limitation regarding these trials is that long-term outcome of MACT versus microfracture is still relatively unknown; the longest follow-up time included in our analysis was 7.5 years, with a weighted mean of 3.1 years. We believe that if MACT has similar durability to ACI-P or ACI-C (which has been shown to last well past 10 years36-39), then it should be durable enough that the outcomes of MACT versus microfracture at longer follow-up times will be similar to what has been reported. There is also evidence against microfracture as a first-line treatment for isolated cartilage defects of the knee; several studies37,40,41 have shown that second-line ACI after failed microfracture has inferior outcomes (higher failure rates, lower KOOS scores, higher rate of re-intervention) compared to when ACI is performed first.
Current high-level evidence is limited comparing MACT to first-generation ACI, MSC, and mosaicplasty. All of the trials that fit our inclusion criteria for these questions were small single-institution studies that may not be powered adequately to draw accurate conclusions. In all 3 comparison categories, there is insufficient evidence and power to conclude whether one is superior to the other. However, there is evidence in the literature to suggest that mosaicplasty is inferior to first-generation ACI in the treatment of larger cartilage defects 42 (average of 440.9 mm2). Ultimately, longer-term and adequately powered studies are needed.
Niemeyer et al. 19 concluded that there are no differences in outcomes between low, medium, and high chondrocyte dosing for MACT. Again, this single study is not enough to draw conclusions regarding dosing, but we believe that the answer to this question is most likely to be dependent on the specific commercial product. The chondrocyte dosing depends on the nature of the scaffold and delivery system, so different products will have different dosing requirements. Still, there is likely a threshold value of chondrocyte concentration below which graft maturity or fill will not occur, but currently this value is unknown.
The quality of this review is limited by the quality of studies analyzed. The mean Coleman score for our studies was 59.4, which falls into the “fair” category. However, 5 of the 13 trials had scores below 54, placing them in the “poor” category in terms of quality as determined by Cowan et al. 26 In addition, several of our questions could not be adequately answered due to the small number of trials and small number of patients in those trials. Unfortunately, this is likely due to the fact that orthopedic clinical trials tend to be small, and often underpowered. In sports medicine trials, Khan et al. 43 found a mean Fragility Index of 2, indicating reported significant results would become nonsignificant if the outcomes of 2 or more patients in the trial were to be changed. Additional adequately powered studies are needed to answer some questions proposed by our review.
Compared to microfracture, MACT has no increased risk of clinical failure or reoperation and has superior improvement in patient-reported outcome scores. Compared to MACT with standardized postoperative weight-bearing protocols, accelerated weight-bearing protocols have no increased risk of clinical failure but show superior improvement in patient-reported outcome scores. There is limited evidence regarding MACT compared to first-generation ACI, mosaicplasty, and MSC without compelling differences in outcomes. Evidence regarding the effect of chondrocyte dose at implantation is limited.
Footnotes
Appendix A
Appendix B
Modified Coleman Methodology Score 23 .

| Item | Points |
|---|---|
| Inclusion Criteria | |
| Not described | 0 |
| Described without percentages given | 3 |
| Enrollment rate <80% | 6 |
| Enrollment rate >80% | 9 |
| Power | |
| Not reported | 0 |
| >80%, methods not described | 3 |
| >80%, methods described | 6 |
| Alpha error | |
| Not reported | 0 |
| <0.05 | 3 |
| <0.01 | 6 |
| Sample size | |
| Not stated or <20 | 0 |
| 20-40 | 3 |
| 41-60 | 6 |
| >60 | 9 |
| Randomization | |
| Not randomized | 0 |
| Modified/partial | |
| Not blinded | 2 |
| Blinded | 4 |
| Complete | |
| Not blinded | 6 |
| Blinded | 8 |
| Follow-up | |
| Short-term (<6 months) | |
| Patient retention <80% | 0 |
| Patient retention 80% to 90% | 2 |
| Patient retention >90% | 4 |
| Medium-term (6-24 months) | |
| Patient retention <80% | 2 |
| Patient retention 80% to 90% | 4 |
| Patient retention >90% | 6 |
| Long term (>24 months) | |
| Patient retention <80% | 4 |
| Patient retention 80% to 90% | 6 |
| Patient retention >90% | 8 |
| Patient analysis | |
| Incomplete | 0 |
| Complete | 3 |
| Complete and intention-to-treat-based | 6 |
| Blinding | |
| None | 0 |
| Single | 2 |
| Double | 4 |
| Triple | 6 |
| Similarity in treatment | |
| No | 0 |
| Similar co-interventions | 3 |
| No co-interventions | 6 |
| Treatment description | |
| None | 0 |
| Fair | 3 |
| Adequate | 6 |
| Group comparability | |
| Not comparable | 0 |
| Partially comparable | 3 |
| Comparable | 6 |
| Outcome assessment | |
| Written assessment by patient with assistance | 0 |
| Written assessment by patient without assistance | 2 |
| Independent investigator | 4 |
| Recruited patients | 6 |
| Description of rehabilitation protocol | |
| Not reported | 0 |
| Not adequately described | 2 |
| Well described | 4 |
| Clinical effect measurement | |
| Effect size | |
| Not reported | 0 |
| <50% | 2 |
| 50% to 75% | 4 |
| >75% | 6 |
| or Relative risk reduction | |
| Not reported | 0 |
| <25% | 3 |
| >25% | 6 |
| or Absolute risk reduction | |
| Not reported | 0 |
| <10% | 3 |
| >10% | 6 |
| Number of patients to treat | |
| Not reported | 0 |
| Reported | 4 |
Appendix C
Methodological Index for Non-Randomized Studies (MINORS) 24 .

| Item | Description |
|---|---|
| 1 | A clearly stated aim: The question addressed should be precise and relevant in the light of available literature. |
| 2 | Inclusion of consecutive patients: All patients potentially fit for inclusion (satisfying the criteria for inclusion) have been included in the study during the study period (no exclusion or details about the reasons for exclusion). |
| 3 | Prospective collection of data: Data were collected according to a protocol established before the beginning of the study. |
| 4 | Endpoints appropriate to the aim of the study: Unambiguous explanation of the criteria used to evaluate the main outcome which should be in accordance with the question addressed by the study. Also, the endpoints should be assessed on an intention-to-treat basis. |
| 5 | Unbiased assessment of the study endpoint: Blind evaluation of objective endpoints and double-blind evaluation of subjective endpoints. Otherwise the reasons for not blinding should be stated. |
| 6 | Follow-up period appropriate to the aim of the study: The follow-up should be sufficiently long to allow the assessment of the main endpoint and possible adverse events. |
| 7 | Loss to follow-up less than 5%: All patients should be included in the follow up. Otherwise, the proportion lost to follow up should not exceed the proportion experiencing the major endpoint. |
| 8 | Prospective calculation of the study size: Information of the size of detectable difference of interest with a calculation of 95% confidence interval, according to the expected incidence of the outcome event, and information about the level for statistical significance and estimates of power when comparing the outcomes. |
| 9 | An adequate control group: Having a gold standard diagnostic test or therapeutic intervention recognized as the optimal intervention according to the available published data. |
| 10 | Contemporary groups: Control and studied groups should be managed during the same time period (no historical comparison). |
| 11 | Baseline equivalence of groups: The groups should be similar regarding the criteria other than the studied endpoints. Absence of confounding factors that could bias the interpretation of the results. |
| 12 | Adequate statistical analyses: Whether the statistics were in accordance with the type of study with calculation of confidence intervals or relative risk. |
Each item was assigned a score of 0, 1, or 2—0 (not reported), 1 (reported but inadequate) or 2 (reported and adequate).
Appendix D
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: David C. Flanigan is a consultant for Depuy Mitek, Inc., Smith & Nephew, vericel, Ceterix Orthopaedics, Conmed, Histogenics Corporation, and Zimmer Inc. The other authors have no conflicts of interest to disclose.
