Abstract
Objective
The objective of this study was to assess potential risk factors, including time delay until implantation, for knee cartilage defect expansion or new high-grade defect formation between biopsy and Autologous Chondrocyte Implantation (ACI) or Matrix Autologous Chondrocyte Implantation (MACI).
Study design
Consecutive knee ACI and MACI cases by a single surgeon (n = 111) were reviewed. The relationship between time between biopsy and staged implantation and (1) progression in primary cartilage defect size and (2) development of a new high-grade (Outerbridge grade ≥3) cartilage defect were determined with adjustment for demographics, body mass index, smoking status, coronal alignment, initial cartilage status, and prior surgery.
Results
Average size of the primary defect at time of biopsy was 4.50 cm2. Mean time to chondrocyte implantation was 155 days. Defect expansion increased 0.11 cm2 (standard error = 0.03) per month delay to implantation (P = 0.001). Independent predictors of defect expansion were male sex, smaller initial defect size, and delay to implantation (adjusted mean = 0.15 cm2 expansion per month). A total of 16.2% of patients (n = 18/111) developed a new high-grade defect. Independent predictors of a new secondary defect were Outerbridge grade 2 changes (vs. 0-1) on the surface opposing the index defect and delayed implantation (per month increase, adjusted odds ratio = 1.21, 95% confidence interval: 1.01-1.44; P = 0.036).
Conclusions
Patients undergoing 2-stage cell-based cartilage restoration with either ACI or MACI demonstrated long delays between stages of surgery, placing them at risk for expanding defects and development of new high-grade cartilage defects. Patients who were male, had smaller initial defect size, and longer time between surgeries were at greater risk for defect expansion.
Level of Evidence
III, retrospective comparative study.
Keywords
Introduction
First-generation Autologous Chondrocyte Implantation (ACI) and third-generation Matrix Autologous Chondrocyte Implantation (MACI) have been effective cell-based cartilage restorative techniques used to address symptomatic, full-thickness cartilage defects in the knee. Cell-based cartilage techniques have evolved since first-generation ACI (marketed as Carticel, no longer available in US as of 2017), but studies have shown the efficacy of both ACI and third-generation scaffold techniques such as MACI. Long-term reports of patients undergoing ACI have shown improved clinical and functional outcomes 10 years to 20 years post ACI.1-3 Furthermore, a recent randomized trial comparing microfracture to MACI demonstrated superior Knee Injury and Osteoarthritis Outcomes Scores (KOOS) at 5 years in MACI patients. 4 With this success, there has been an increase in cell-based repair techniques performed nationally with a recent database study noting a 626.6% increase in ACI procedures performed over a 7-year period. 5 With a growing population of active adults, knee preservation is paramount to maintaining function and delaying arthroplasty in young active patients.
Both ACI and MACI involve a 2-stage surgical approach with the initial step involving a thorough arthroscopic assessment of the knee joint and the defect in question. Arthroscopic evaluation of lesion size is critical given post-debridement lesions are on average 1.04 cm2 larger than estimates on magnetic resonance imaging. 6 Arthroscopic measurements of the defects are obtained, and a cartilage biopsy is performed at a non-weightbearing aspect of the trochlea. The biopsy cells are sent to the laboratory and expanded in vitro. 7 The second stage involves implanting the cultured cells into the defect and securing the cells with periosteum (first-generation technique) or collagen patch (second generation), or cell-seeded collagen membrane (third generation).
Unfortunately, there are several reasons for delay between biopsy and implantation, including chondrocyte processing, graft preparation, insurance approval, patient factors (preference for timing, deductibles, life events) and surgeon scheduling. Cartilage defects experience high rim stresses, which could theoretically result in progressive degeneration of the defect margins and resulting defect expansion. 6 In our practice, we have observed cases of symptomatic high-grade (Outerbridge grade ≥3) cartilage defects that, for various reasons, had a significant time delay between cartilage biopsy and definitive treatment with ACI or MACI and had either significant expansion of the primary defect or development of one or more secondary high-grade defects. Large defects and multiple knee cartilage defects can influence outcomes after chondrocyte implantation defect.8,9 A knowledge of which patients are at higher risk for expanding defects or development of new defects would assist surgeons’ preparation for these more difficult cases, as well as provide data to support expeditious treatment in high-risk patients.10,11 The purpose of the current study is to assess potential risk factors, including time delay until implantation, for knee cartilage defect expansion or new high-grade defect formation between biopsy and ACI or MACI. It is hypothesized that increasing time delay until second-stage surgery will result in either lesion expansion or new high-grade defect formation.
Methods
After receiving institutional review board approval (#2014H0101), a retrospective chart review of 111 consecutive ACI and MACI cases by a single surgeon over a 10-year period (2008-2018) was performed. Indications included full-thickness symptomatic cartilage defects without signs of tricompartmental arthritis. Meniscal pathology and malalignment were addressed as necessary prior to or at time of ACI implantation. Patients were designated as ACI or MACI patients by operative notes and year of surgery. The primary surgeon adopted MACI following Food and Drug Administration approval in the United States in late 2016, after which first-generation ACI was removed from the US market in mid-2017. Baseline demographic data were obtained, including age, gender, weight, height, activity level, and smoking status. Full-length lower extremity films were reviewed with coronal alignment measured using hip knee ankle (HKA) angle and percent lateralization of the mechanical axis at the tibiofemoral joint line. Operative notes and arthroscopic images were reviewed to record defect location, dimensions, and orientation. All characteristics of cartilage defects were recorded at time of biopsy and time of implantation (see Figs. 1 and 2 for case example). Defects were measured with a ruler at time of biopsy and at time of implantation surgery. During arthroscopic assessment at final stage of implantation, any new high-grade cartilage defects were evaluated and noted.
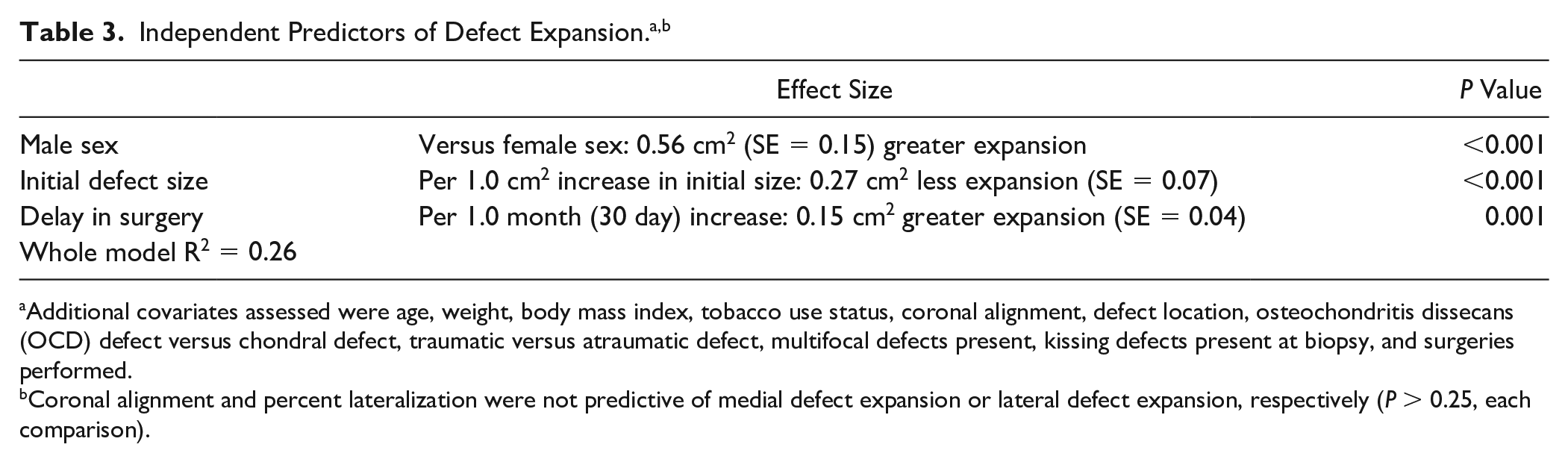
Case Illustration of a 31-year-old male with prior microfracture to medial and lateral femoral condyles undergoing diagnostic arthroscopy at time of cartilage biopsy: (
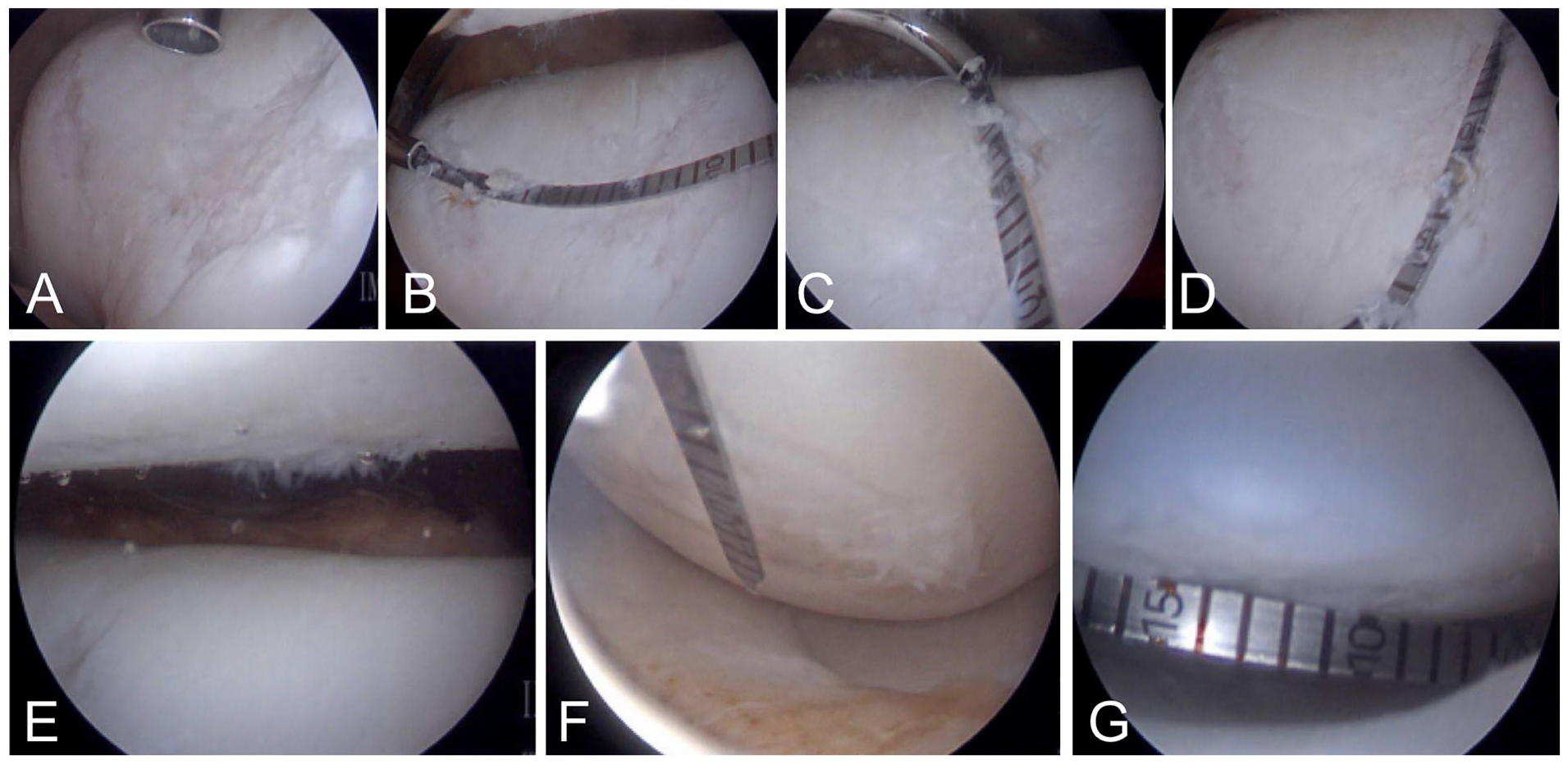
Photos of patient at time of implantation 224 days from biopsy: (
Statistical Analysis
Statistical analysis was performed with a standard software package (JMP 14.2, SAS Institute, Cary, NC). Descriptive statistics were first generated for the entire sample. Bivariate analyses were performed to determine the association between time delay from biopsy to implantation and change in defect size via linear regression and risk of new high-grade defect formation via logistic regression, respectively. To determine independent risk factors for defect expansion and new high-grade defect formation, multivariate linear and logistic regression models were created, respectively. A backward selection method was utilized with an exit criterion of P > 0.05. Variables assessed for potential inclusion in the multivariate models were age, sex, weight, body mass index, tobacco use status, coronal alignment, initial defect size, defect location, osteochondritis dissecans (OCD) defect versus chondral defect, traumatic versus atraumatic defect, multifocal high-grade defects present at biopsy, kissing defects present at biopsy, and prior surgeries performed.
Results
Descriptive Data
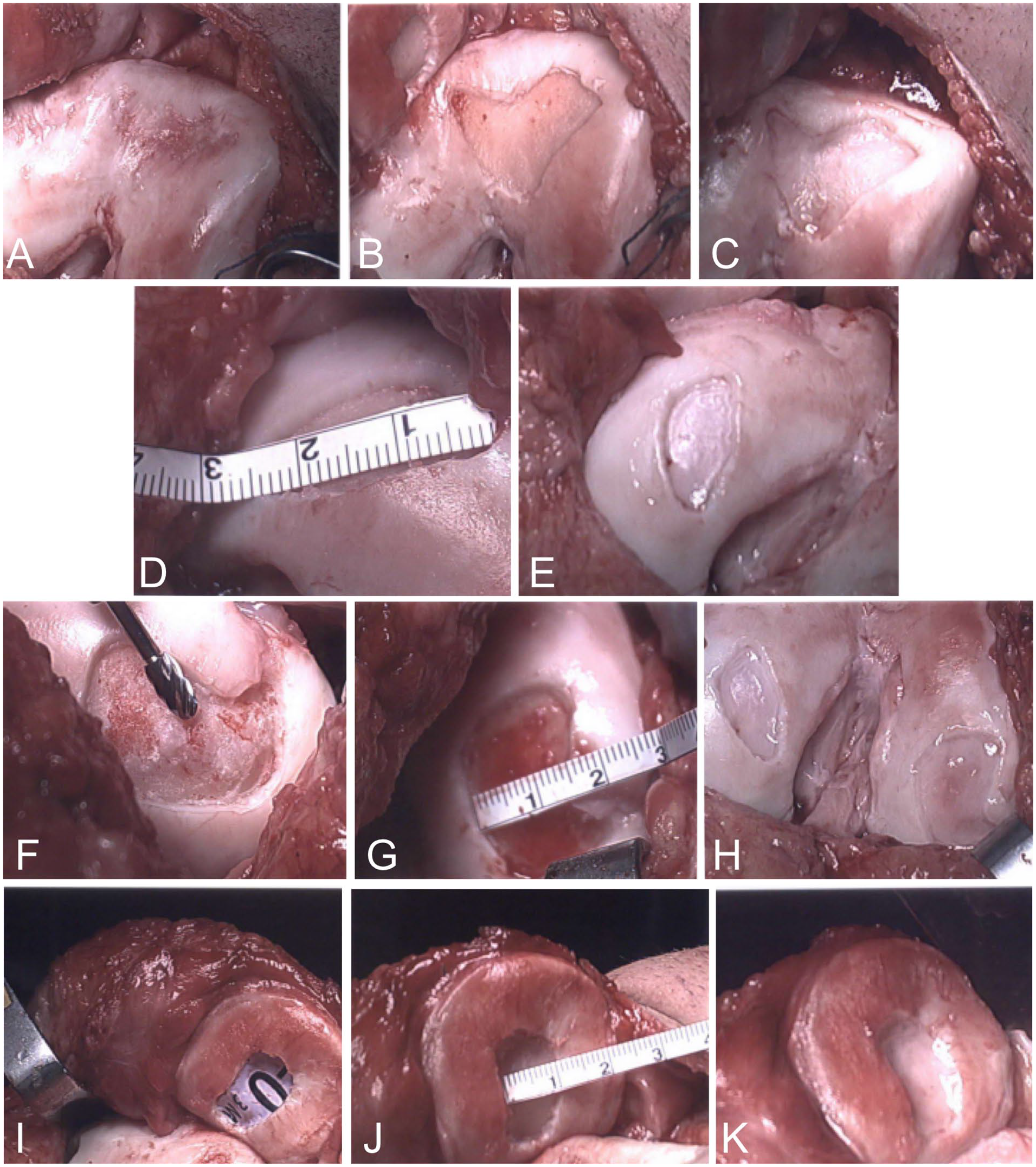
Baseline demographic data reported in Table 1 . There were 128 knees in 111 patients addressed with ACI (77 knees) or MACI (51 knees). The average size of the largest high-grade defect in each knee at the time of initial articular cartilage biopsy was 4.50 cm2 (SD = 2.05 cm2). At time of biopsy, 13.5% of patients (n = 15/111) had 2 or more high-grade (Outerbridge grades 3 or 4) defects. The mean time from biopsy to ACI or MACI was 155 days (SD = 131, 25th percentile 70, median = 117, 75th percentile 189) ( Table 2 ). Among the 128 high-grade defects seen at time of biopsy (in 111 patients), the location was on the patella in 18.0% (n = 23/128), trochlea in 24.2% (n = 31/128), medial femoral condyle (MFC) in 32.0% (n = 41/128), and lateral femoral condyle (LFC) in 25.8% (n = 33/128) ( Fig. 3 ).
Patient Demographics and Knee Alignment.
BMI = body mass index; ACI = Autologous Chondrocyte Implantation; MACI = Matrix Autologous Chondrocyte Implantation.
Time between Biopsy & Implantation and Defect Characteristics at Time of Implantation.
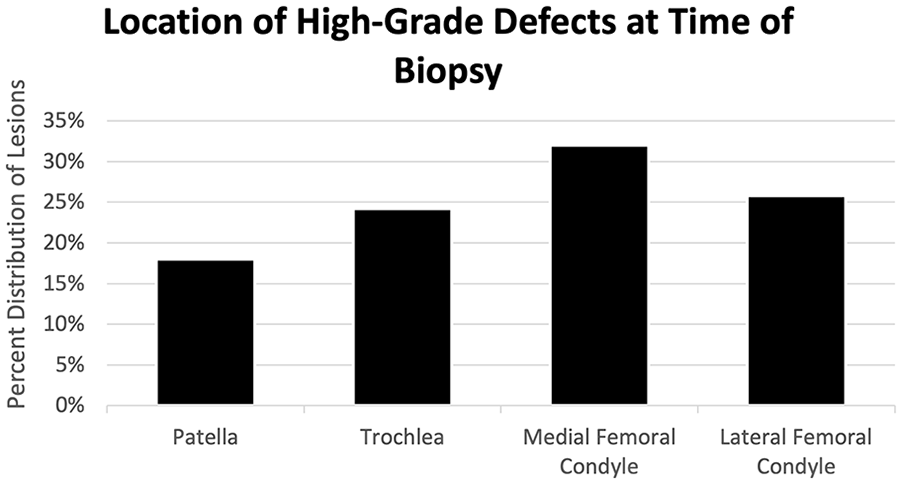
Graph illustrating the distribution of high-grade defect locations identified and measured at initial biopsy stage.
Defect Expansion
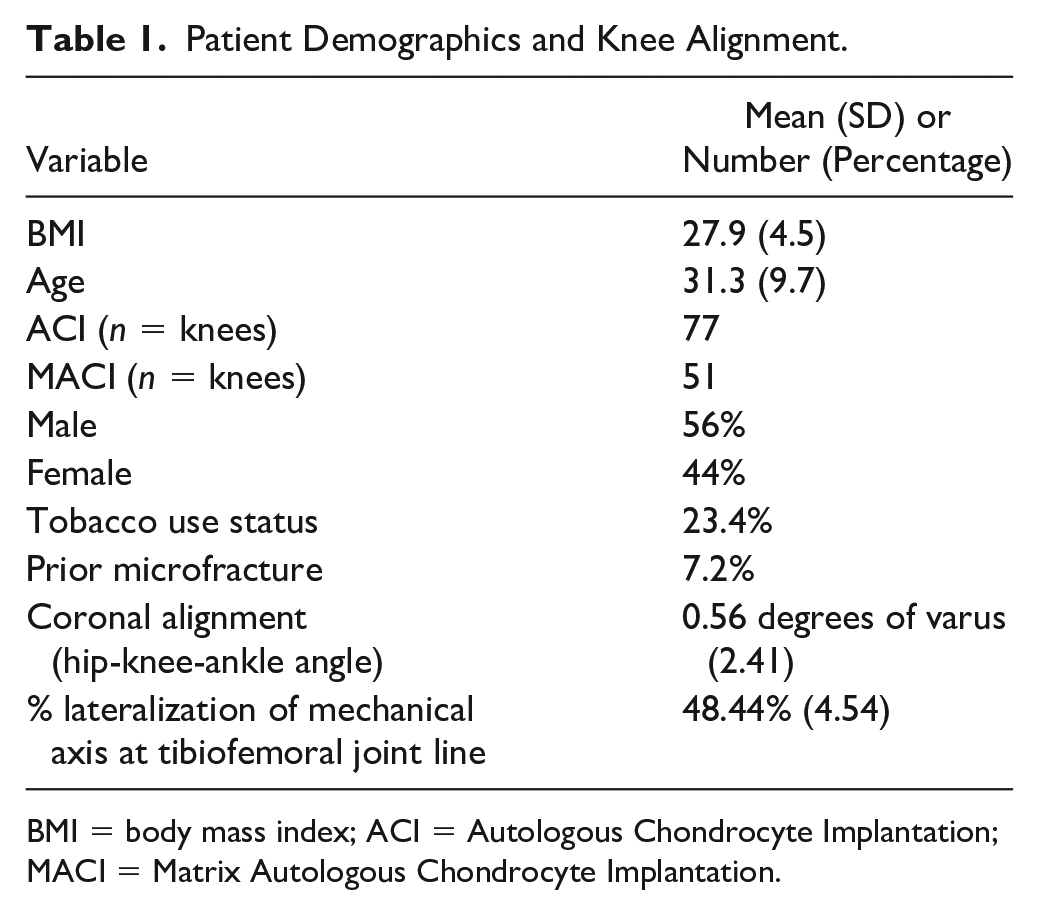
In the bivariate linear regression analysis, without adjusting for potential confounders, defect expansion increased an average 0.11 cm2 (standard error [SE] = 0.03) per month delay from biopsy to implantation (P = 0.001; R2 = 0.085). In the multivariate linear regression analysis, independent predictors of defect expansion were male sex, smaller initial defect size, and greater time delay from biopsy to implantation (adjusted mean = 0.15 cm2 per month delay) ( Table 3 ).
Additional covariates assessed were age, weight, body mass index, tobacco use status, coronal alignment, defect location, osteochondritis dissecans (OCD) defect versus chondral defect, traumatic versus atraumatic defect, multifocal defects present, kissing defects present at biopsy, and surgeries performed.
Coronal alignment and percent lateralization were not predictive of medial defect expansion or lateral defect expansion, respectively (P > 0.25, each comparison).
No difference was seen in defect expansion between biopsy and implantation for ACI or MACI patient in the univariate analysis (P = 0.75) or after controlling for sex and initial defect size (P = 0.16). This was also true when controlling for sex, initial defect size, and delay of surgery (P = 0.31). Variables found to be non-predictive of defect expansion and ultimately excluded from the final multivariate model were age, weight, body mass index, tobacco use status, coronal alignment, defect location, OCD defect versus chondral defect, traumatic versus atraumatic defect, multifocal high-grade defects present at biopsy, kissing defects present at biopsy, and ACI versus MACI (P > 0.05, each comparison).
Risk of New High-Grade Defect Formation
A total of 16.2% of patients (n = 18/111) had a new high-grade defect at time of re-implantation (n = 22 defects in 18 patients) which occurred in the patella in 10/22, trochlea in 5/22, MFC in 4/22, and LFC in 3/22 ( Fig. 4 ). In the bivariate logistic regression analysis, without adjusting for potential confounders, risk of a new high-grade defect did not increase with time (per 1.0 month [30 day] increase, odds ratio [OR] = 1.08, 95% confidence interval [CI]: 0.93, 1.26; P = 0.32). In the multivariate linear regression analysis, independent predictors of development of a new high-grade defect were presence of grade II changes opposite or “kissing” a high-grade defect and increased time from biopsy to implantation (per 1.0 month [30 day] increase, adjusted OR = 1.21, 95% CI: 1.01, 1.44; P = 0.036) ( Table 4 ).
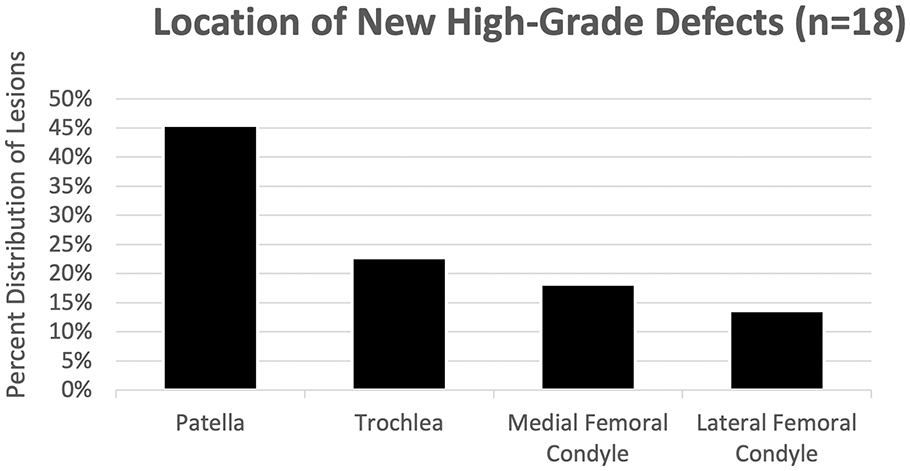
Graph illustrating the location distribution of new high-grade defects identified during arthroscopy in second-stage implantation.
Independent Predictors of a New High-Grade Defect at Time of Implantation. a

CI = confidence interval; AUROC = area under receiver operator curve.
Additional covariates assessed were age, sex, weight, body mass index, tobacco use status, coronal alignment, defect location, osteochondritis dissecans (OCD) defect versus chondral defect, traumatic versus atraumatic defect, multifocal high-grade defects present at biopsy, and prior surgeries performed.
No difference was seen in rate of new defects between biopsy and implantation for ACI or MACI in the univariate analysis (P = 0.92) or after controlling for kissing defects (P = 0.75) or kissing defects and delay of surgery (P = 0.55). Variables found to be non-predictive of new defect formation and ultimately excluded from the final multivariate model were age, sex, weight, body mass index, tobacco use status, coronal alignment, defect location, OCD defect versus chondral defect, traumatic versus atraumatic defect, multifocal high-grade defects present, and ACI versus MACI (P > 0.05, each comparison).
Discussion
Orthopedic surgeons in the United States are performing cartilage procedures in increasingly higher volumes. Specifically, autologous chondrocyte implantation procedures have increased by 626.6% from 2010 to 2016. 5 This increasing trend of 2-stage cartilage repair techniques and the reliance on cartilage defect size for preoperative planning necessitates that cartilage surgeons identify the average turnaround time between stages, barriers to expeditious implantation, and patients with risk factors for poor outcomes due to longer intervals between stages. Reported data in clinical studies of ACI show a variable treatment time from biopsy to implantation from 6 weeks to 2 years. 12 However, to our knowledge, no published report exists describing the exact interval between stages, what factors affect this interval, and how the health of the knee is impacted by delaying implantation in cell-based cartilage techniques.
The most important finding of this study is that lesions can progress and new lesions form as time between cartilage biopsy and implantation increases. Reasons for delay between stages were multifactorial and not readily available for reporting, but include asymptomatic period after biopsy, patient preference due to time of year, economics, insurance, or surgeon scheduling. This study showed a mean time of 155 days from cartilage biopsy until chondrocyte implantation. Concerningly, each additional month between stages led to a mean defect expansion of 0.11 cm2. Furthermore, 16.2% of patients had a new high-grade defect at the time of chondrocyte implantation. Although suspected in prior studies, this report is the first to demonstrate that untreated symptomatic defects continue to grow with time. 13
Several risk factors were identified for worsening cartilage injury progression in patients with cartilage defects. Expansion of existing cartilage defects was predicted by male sex, initial defect size, and time delay to implantation. We found no significance in lesion expansion or lesion development between patients undergoing ACI versus MACI. Development of a new high-grade defect treatable by ACI or MACI was predicted by presence of a grade II kissing defect and time delay to implantation. Identifying why these variables are risk factors is challenging, but the observations that smaller defects and Outerbridge grade II kissing defects were at higher risk for advancement to a high-grade defect, and yet that defect location was not predictive of expansion or new defect formation, suggest a potential manifestation of a degenerative pathway when cartilage defects are left untreated. A possible explanation is the result of a complex interplay between macroscopic and microscopic factors. Altered rim stress on defect diameters greater than 10 mm may increase pressure on cartilage defects leading to expansion, and when lesions expands to greater than 2 cm2 there is loading of the subchondral bone. 14 As this process occurs, the release of cartilage breakdown products and subsequent inflammatory response may persist, leading to a cycle of inflammation with degenerative mediators conducive to defect breakdown and loose body formation.3,15 Overtime, cartilage delamination, fragmentation, and loose body formation may play a role in mechanical wear of articular cartilage resulting in expansion and formation of secondary defects.
Further importance of avoiding unnecessary defect expansion is demonstrated by studies noting larger cartilage defects and multifocal defects with poorer clinical outcomes. At least one prior long-term study on ACI have noted patients with defects over 4.5 cm2 have worse clinical outcomes compared with patients with smaller defects. 12 In addition, defect expansion may lead to situations where more sophisticated surgical techniques are necessary. Larger defects require more graft, which must be ordered and prepared prior to surgery. Larger uncontained defects may not fit standardized templates requiring free hand grafts that may not efficiently utilize the surface area of the grafts. Furthermore, uncontained defects may require sandwich techniques or use of anchors to create a barrier for the graft. Larger defects may require larger incisions to help with exposure, which may require more invasive techniques such as meniscal takedown with subsequent repair. Iatrogenic injury is also increased with defects expanding into more challenging areas of the knee including the posterior aspect of the femoral condyle. All these examples pose a significant problem for cartilage repair surgeons.
The information provided by our study assists the surgeon in preoperative planning, setting expectations with the patient, and leveraging insurance companies during the approval process. Our data offer a compelling argument to address cartilage defects expeditiously to prevent further deterioration of the knee joint and ultimately obtain better outcomes for our patients. These results assist the surgeon in counseling patients on the timeliness of implantation when undergoing MACI, especially when the patient exhibits important predictive factors. Furthermore, surgeons performing MACI should be prepared to address larger surface areas due to defect expansion or new defects that may have developed since initial biopsy.
Limitations
This present study is not without limitations. The study is retrospective in nature, based on one surgeon’s results, and was not blinded. Some of the observed defect expansion between initial sizing and implantation is due to debridement of the defect edges at the time of implantation to a healthy margin. However, this should not bias the observed relationship between time delay and greater defect size as the debridement technique and goal of healthy cartilage margins is the same for each case regardless of chronicity.
Conclusion
Patients undergoing 2-stage cell-based cartilage restoration with either ACI or MACI demonstrated long delays between stages of surgery, placing them at risk for expanding defects and development of new high-grade cartilage defects of the knee. Patients who were male, had smaller initial defect size, and longer time between surgeries were at greater risk for defect expansion. Surgeons, patients, insurance, and industry should coordinate efforts to expedite the interval between biopsy and implantation in 2-stage cartilage restoration procedures.
Footnotes
Authors’ Note
This study was approved by the Biomedical Institutional Review Board of The Ohio State University.
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: David C. Flanigan has received consulting payments from Vericel, Smith and Nephew, Linvatec, and DePuy.