Abstract
Objective
Quercetin (Que), a bioflavonoid, is both anti-inflammatory and antioxidative. Que has been used as an oral supplement for osteoarthritis (OA) with inconsistent findings because of its low bioavailability. We encapsulated Que in a mPEG-polypeptide thermogel to prolong its bioactivity. The efficacy of this formulation was evaluated in a posttraumatic OA rat model.
Design
Methoxy-poly(ethylene glycol)-
Results
In vitro studies showed that chondrocytes were viable after 72 hours of incubation with mPEG-PA, and the release of Que could be sustained for >28 days. Among all OA rats, the limb idleness index (LII) were significantly increased at 24 weeks post-ACLT. Rats that received hydrogel with Que (50 μg) showed the most reduction in LII at both 4 and 8 weeks post-treatment. The Osteoarthritis Research Society International score of rats received hydrogel with Que (50 μg) was significantly lower than the control group. All rats suffered from low-grade synovitis (Krenn score: 2-4).
Conclusion
This study suggests that a sustained delivery of Que (50 μg) could provide symptom relief and also delay the progression of OA in the knee.
Introduction
Osteoarthritis (OA) is a degenerative joint disease and one of the major causes of disability that severely affects the quality of life in patients. OA is characterized by cartilage loss, synovial inflammation and subchondral bone remodeling, which leads to pain and joint stiffness. 1 There is currently no effective cure for OA, which the recommended pharmacological treatments include oral administration of analgesics, nonsteroidal anti-inflammatory drugs (NSAIDs) and intra-articular injections of corticosteroids. However, these treatments can only provide temporary symptomatic relieve and have their associated deleterious side effects such as irritation of the gastrointestinal tract and increase risks of cardiovascular diseases. 2 Oxidative stress plays a role in OA, which leads to chondrocyte apoptosis, senescence and autophagy, and affects the homeostasis of the articular cartilage extracellular matrix.3,4 Furthermore, oxidative stress has been shown to contribute to the pro-inflammatory phenotype of OA by upregulating redox-sensitive transcription factors and disrupt various signaling pathways that leads to both synovial inflammation and subchondral bone dysfunction. 5
Symptomatic slow-acting drugs for OA (SYSADOAs) are a class of chondroprotective bioactive compounds that can provide symptomatic relieve and delay the progression of OA with minimal secondary effects. 6 Quercetin (Que), one of the most abundant bioflavonoids, is found to be both antioxidative and anti-inflammatory. In arthritis, Que has been demonstrated to promote apoptosis in arthritic fibroblast-like synoviocytes and protects chondrocytes against oxidative stress in vitro.7,8 In arthritic animal models, Que could reduce symptoms and arthritis by reducing inflammation and oxidative stress while improving the antioxidant defence.9-13 Clinically, Que was able to improve clinical symptoms, synovial fluid properties and reduce the concentration of inflammatory factors in serum of patients with arthritis.14-16 However, no obvious improvements with the use of Que in treating arthritis have also been reported.17,18 This inconsistency has been attributed to the low bioavailability of Que through oral administration. 19 Intra-articular (IA) therapy provides a direct route of drug administration to a diseased joint, which some of its many advantages include increased bioavailability and minimized potential systemic side effects. 20 Previously, we investigated the effect of multiple IA injections of an antioxidant formulation in an OA rat model, which showed short term symptomatic relieve. 21 However, repeated injections were required and is undesirable in clinical settings.
To prolong drug actions and delay clearance within the IA space of a knee, various drug delivery systems have been developed for IA therapy to deliver therapeutics at a controlled and sustained manner.
20
Thermosensitive, biodegradable polymers-based in situ–forming hydrogels, including polyesters, polycarbonates, and polyphosphazenes, have been studied to deliver potential drugs for OA. For example, hydrogels with drugs or hybrid systems of microparticles in thermogels have been developed and studied its effectiveness against OA both in vitro and in vivo.22-25 However, some of the degradative products of these materials, such as PLGA—poly(lactic-co-glycolic acid)—polymer, could increase the local acidity and contribute to inflammation in tissues.
26
Polypeptide-based hydrogels are temperature-sensitive hydrogels, which undergoes sol-gel transition at physiological temperature, allows for low gelation concentration and encapsulation of hydrophobic molecules. Polyalanine conjugated to poly(ethylene glycol) have been used to encapsulate cells for skin wound and investigated as a 3-dimensional scaffold for tissue engineering applications, which demonstrated its biocompatibility and biodegradability.27-29 In this study, in order to increase the bioavailability and provide a sustained treatment for OA, Que was encapsulated in a polypeptide-based hydrogel, methoxy-poly(ethylene glycol)-
Materials and Methods
Synthesis and Characterization of Monomethoxy-Poly(Ethylene Glycol)-l -Poly(Alanine) (mPEG-PA)
A thermosensitive hydrogel, mPEG-PA, was synthesized via ring-opening polymerization of N-carboxy anhydride of
Cell Viability Assay
The collection of human samples was approved by the Joint Chinese University of Hong Kong–New Territories East Cluster Clinical Research Ethics Committee Clinical Research Ethics Committee (Reference number: 2013.248). Human chondrocytes were collected and cultured from patients who underwent total knee arthroplasty according to previously published protocol. 32 Chondrocytes at passage 1 were seeded onto 96-well plate overnight and treated either with medium (Dulbecco’s modified Eagle medium: Nutrient Mixture F-12, 10% fetal bovine serum, 1% penicillin-streptomycin-neomycin (PSN) antibiotic mixture or medium supplemented with various concentrations of mPEG-PA hydrogel (4 or 0.4 mg/mL) for 24, 48, and 72 hours. At 4 hours prior to complete incubation, 10 µL of alamarBlue reagent (Bio-Rad, Hercules, CA, USA) was added to each well. The absorbance at wavelengths 570 and 600 nm were measured (μQuant, BioTek Instruments, Inc., Winooski, VT, USA).
In Vitro Release of Que-Loaded mPEG-PA Hydrogel
Quercetin dihydrate (Sigma Aldrich, Cat No.: Q0125, 1 mg/mL, figure 2a insert) suspension was prepared in phosphate buffered saline (PBS) at 4°C and mixed with mPEG-PA polymer. The mixture was vortexed until all mPEG-PA polymer was dissolved. The Que loaded mPEG-PA was gelated at 37°C in a 96-well plate (n = 6). Prewarmed PBS was added to each well and replaced at predetermined time intervals (Time: baseline, 4 hours, 1 day, 2 days, 3 days, 7 days, 14 days, and 28 days). At each sampling time point, an aliquot of the supernatant (50 µL) was mixed with ethanol (100%, 50 µL). The absorbance was measured at 380 nm (μQuant, Biotek Instruments, Inc.). 33
Osteoarthritis Induction
All animal experiments were approved by the Animal Ethics and Experimentation Committee of the Chinese University of Hong Kong (Reference No: 13/015/GRF). Rats were kept at a 12-hour light/dark cycle. A standard diet and water were available ad libitum. Unilateral post-traumatic OA was surgically induced in the right knees of thirty-two 12-week-old, female Sprague-Dawley rats (average body weight: 213 ± 15 g). 34 Briefly, under ketamine (75 mg/kg) and xylazine (10 mg/kg intraperitoneally) anesthesia, the right knee of each rat was shaved and disinfected. A medial para-patellar incision was made, and the patella was dislocated to expose the intra-articular space. The anterior cruciate ligament (ACL) was transected with microscissors and confirmed by anterior drawer test. The patella was relocated, and the joint capsule and skin were closed sequentially with absorbable suture (Ethicon US, Somerville, NJ, USA). Free cage activity was allowed after the operation.
Intra-Articular Injections
Rats were randomly assigned to 4 different groups for an IA injection (average body weight: 281 ± 23 g; n = 8 per group), when OA had been well developed (24 weeks post-ACLT) and the limb idleness index was significantly increased. 35 A Que suspension (1 or 10 mg/mL) was freshly suspended in sterile, PBS (pH 7.4) and mixed with mPEG-PA polymer at 4°C. Under isoflurane (2%) anesthesia, each rat received a 50 µL IA injection of either 0.9% saline, mPEG-PA or mPEG-PA with Que dihydrate (50 µg or 500 µg of Que) in the operated knee. 21 IA injections were performed through the patellar tendon using a 50 μL syringe with a 26G removable needle (Hamilton Company, Reno, NV, USA) and monitored using ultrasound (Vevo 770, FUJIFILM VisualSonics Inc., Toronto, Ontario, Canada). Three-dimensional ultrasound scans of the OA knees were taken before and after IA injections each time (see Supplemental Material available in the online version of the article).
Gait Analysis
At pre-ACLT, 24 (pretreatment), 28 (4 weeks posttreatment), 32 (8 weeks posttreatment), and 36 weeks (12 weeks posttreatment) post-operation, gait analysis was performed using Catwalk XT 9.0 system (Noldus Information Technology, Wageningen, the Netherlands). Rats were placed onto a glass walkway and allowed to walk voluntarily back and forth. The OA-associated pain was analyzed and indicated by the limb idleness index (LII) according to our published protocol. 35 The LII measures the gait adaptation to OA pain in rats with unilateral knee OA by taking into the account on three parameters, the ratio of target print (hind limbs) intensity, anchor print (front limbs) intensity and swing duration of the hind limbs.
Histopathological Assessment
All rats were sacrificed at 36 weeks post-operation (12 weeks post-treatment). The hind limbs were collected for histopathological assessments. In brief, knee samples were fixed in 10% buffered formalin solution for 3 days and decalcified in 9% formic acid for 4 weeks. Joints were cut into 2 approximately equal halves along the medial collateral ligament in the frontal plane. The anterior and posterior halves were embedded in paraffin. Sections of 5 µm at 200 µm interval were cut and a minimal of 3 intervals were collected for toluidine blue and hematoxylin and eosin staining. The OA severity of the most severe section was evaluated using the Osteoarthritis Research Society International (OARSI) score (OARSI score: 0-24). 36 Briefly, the histological sections were scored on 2 criteria: grade (0-6.5) and stage (0-4). The grade is an indicator of OA severity by considering the depth of OA progression into the articular cartilage. Grades 1 to 4 describe changes only in the articular cartilage, progressing from intact cartilage surface, surface discontinuity, vertical fissures to erosion. Grades 5 and 6 describe changes in the subchondral bone with denudation and joint deformation. The stage considers the extent of the articular cartilage surface that is involved with OA, with stage 1 is less than 10% involvement and stage 4 is over 50% involvement. The total OARSI score is a product of the grade and stage, which is a representation of the OA severity and extent in the knee. The synovial inflammation was evaluated using the Krenn scoring system (Krenn score: 0-9) at the lateral compartment of the knee at sites with most pathological changes. 37 The Krenn score is the total sum of 3 parameters, the thickness of the synovial lining (score: 0-3), density of resident cells (score: 0-3), and presence of inflammatory infiltrate (score: 0-3). Histological scoring was performed by 2 raters (nonblinded [SWM] and blinded [YCC]). In cases of discrepancies, discussions were held until consensus had been reached.
Statistical Analysis
All statistical analysis was performed using IBM SPSS Statistics 24 (IBM, Armonk, NY, USA). All data were checked for normality using the Shapiro-Wilk test. Parametric tests were used for normally distributed data, otherwise, a nonparametric equivalence was used. Repeated-measure analysis of variance was used to analyze cell viability. Friedman test was used to compare gait data with respect to temporal changes. To compare the difference in LII between pre- and post-treatment, and histological scoring between the control and treatment groups, Kruskal-Wallis H test with pairwise comparison was used. Significance values were adjusted with Bonferonni correction for multiple testing. The interrater reliability of histological scoring between the 2 raters were evaluated by calculating the intraclass correlation coefficients (ICCs). All statistical significance was accepted at α = 0.05.
Results
Among the 32 rats, 2 rats that were randomly assigned to the saline group were very light at pre-ACLT (weight: 172 and 178 g), pretreatment (weight: 203 and 209 g), and the study endpoint (weight: 213 and 213 g). Based on their body weights at the aforementioned time points, these rats were outliers (
Ranges of the Body Weights of Rats Included in the Study.

ACLT = anterior cruciate ligament transection; IQR, interquartile range.
Cell Viability
The percent reduction of alamarBlue across time and between chondrocytes treated with or without hydrogels (0.4 or 4 mg/mL) were insignificant (P = 0.082, repeated-measures analysis of variance). Cells were viable and proliferated after 72 hours of incubation with mPEG-PA hydrogel (
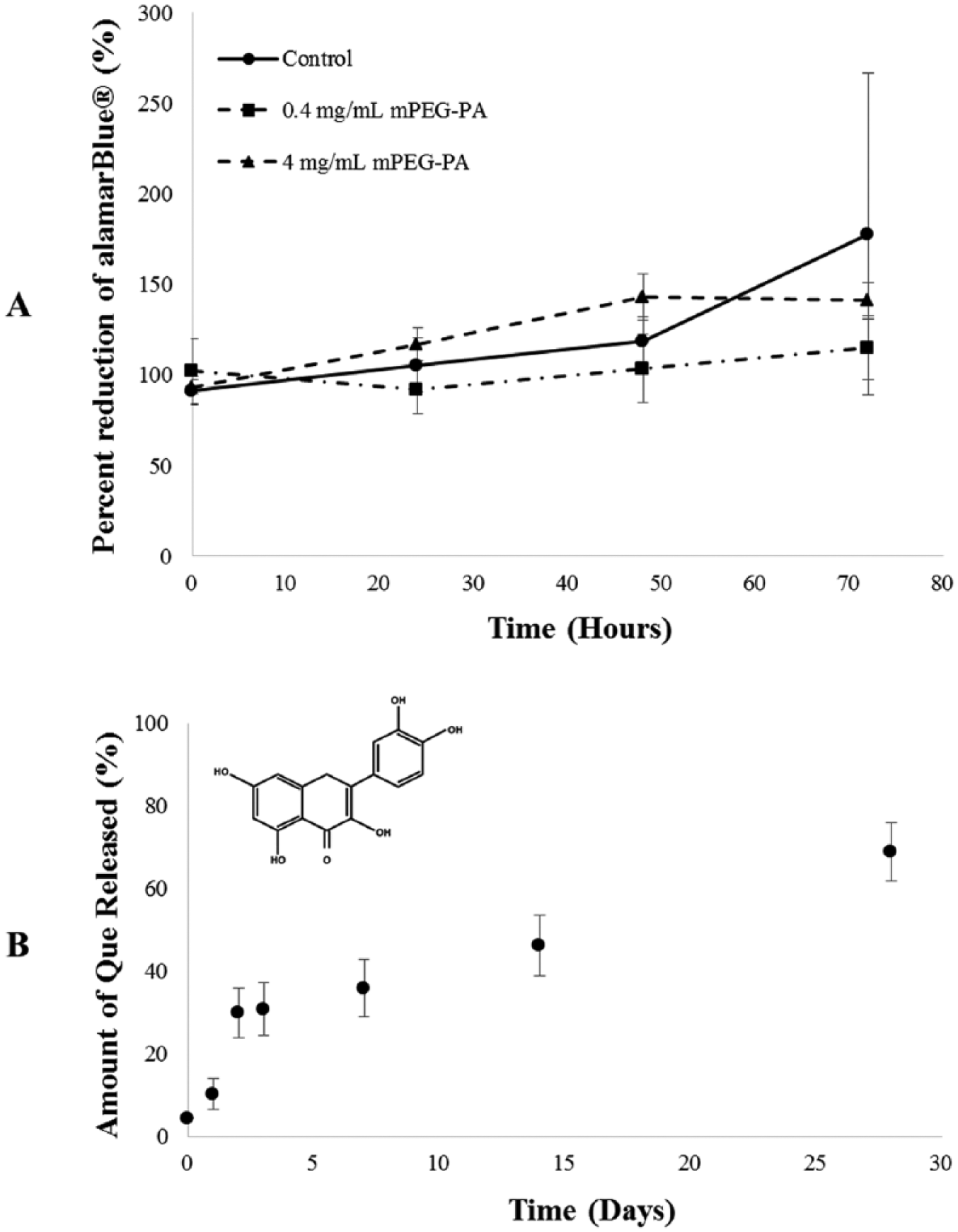
(
In Vitro Release
The release of Que from mPEG-PA hydrogel could be sustained for over 28 days, which means approximately 30% of Que remained in the hydrogel on day 28 (
OA-Related Pain
The LIIs of all rats were significantly increased at 24 weeks post-ACLT (P < 0.001, Wilcoxon signed-rank test,
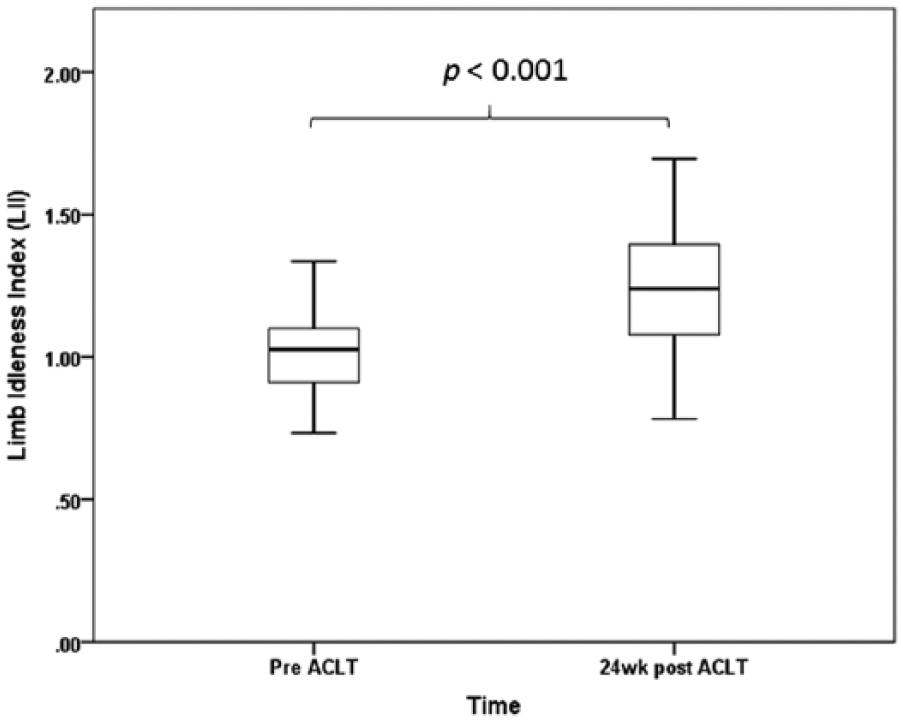
Limb idleness index (LII) at pre–anterior cruciate ligament transection (pre-ACLT) and 24 weeks post-ACLT (pretreatment). The LII of all rats significantly increased (P < 0.001, paired t test).
Difference in Limb Idleness Index (LII) Relative to Preinjection.
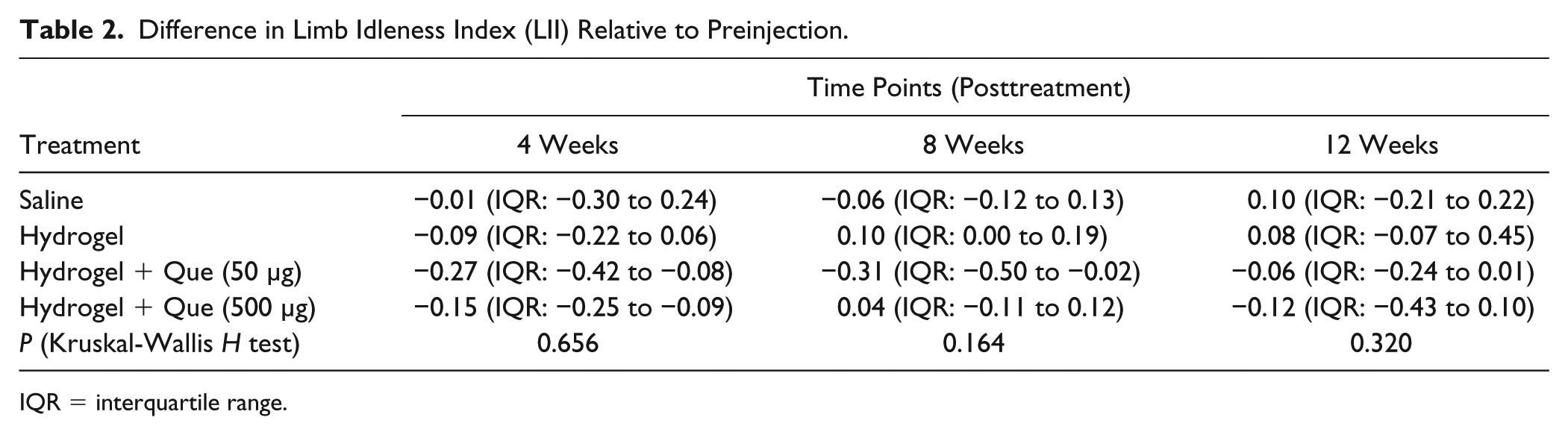
IQR = interquartile range.
Histopathological Assessments
The ICCs of the OARSI and Krenn scoring between 2 raters were 0.809 and 0.789, respectively, which indicates that there was a good interrater agreement. Cartilage degradation was severe in most rats, which the most severe region was found in the posterior compartment of the medial tibial plateau in the operated knees. The stage of cartilage involvement was mostly >50% in all rats and there was no difference among treatment groups (P = 0.263, Kruskal-Wallis H test). However, the tissue reaction varied. The median of cartilage degeneration in the hydrogel and hydrogel with Que groups were lower than the saline group (grade 5 and 6.5, respectively). In the saline group, the changes in subchondral bone were evident, which deformation, including bone remodeling, microfracutre with fibrocartilaginous and osseous repair extending above the calcified cartilage could be found in most rats (
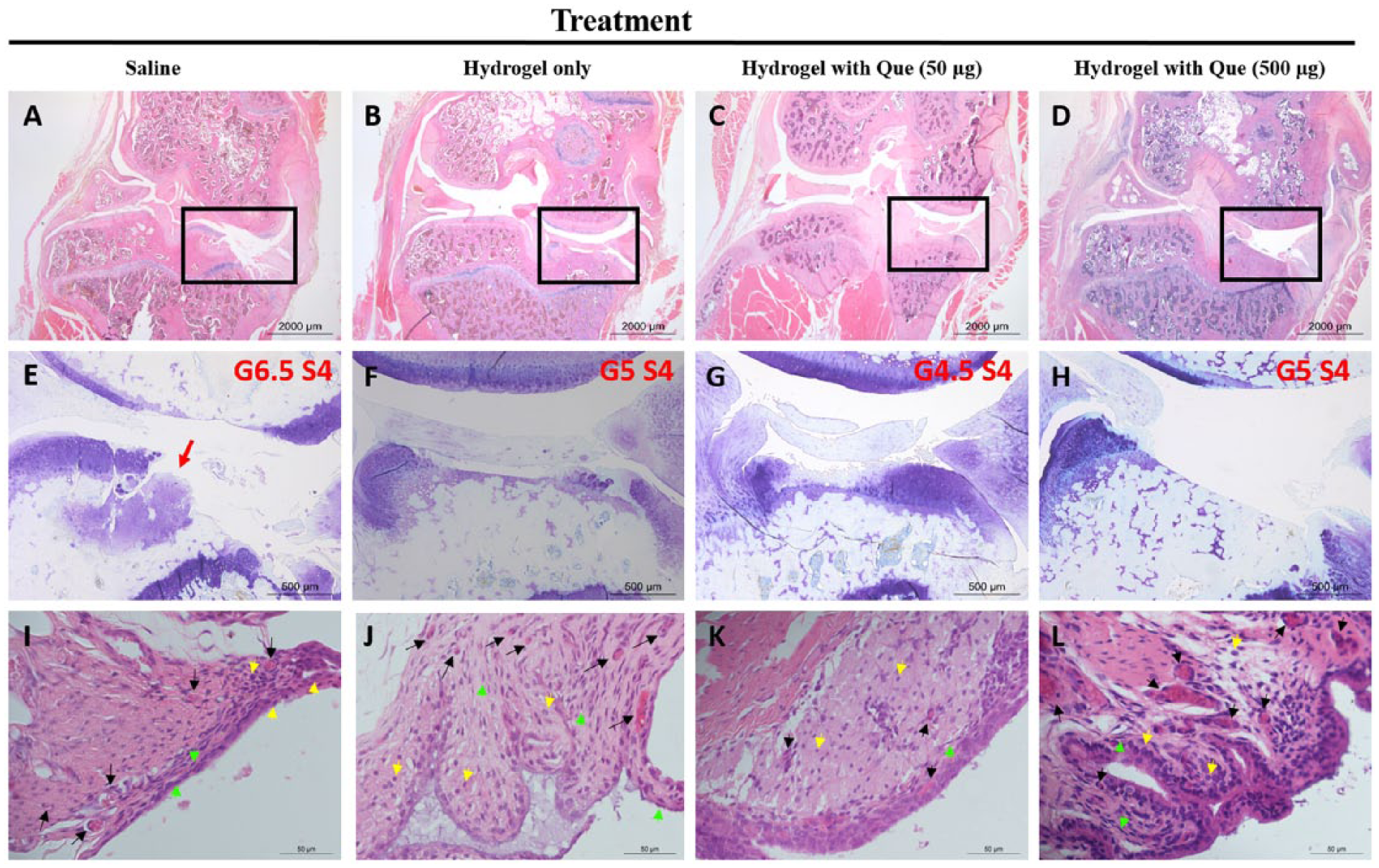
Histopathological sections of operated rat knees at 12 weeks posttreatment (36 weeks post–anterior cruciate ligament transection [post-ACLT]). Images shown represent the median of the Osteoarthritis Research Society International (OARSI) score and Krenn score in each treatment group. Frontal view of the section with most severe cartilage degradation in the knee (
Histopathological Assessments on Articular Cartilage and Synovium. a

Asterisks indicate statistical significances using Kruskal-Wallis H test with multiple comparisons: *P = 0.046 (vs. saline group); **P = 0.009 (vs. saline group).
For synovitis, most rats demonstrated a low-grade synovitis (Krenn score: 2-4). A slight enlargement of synovial lining (2-3 layers) with increased number of synovial fibroblasts and lymphocytes were observed. The density of resident cells was slightly increased with increased vasculature in the sublining layer. An increase in the number of lymphocytes in the perivascular regions were observed (
Discussion
There is currently no cure for OA. Medications for symptom relieve were often prescribed with side effects. Quercetin as an oral supplement for OA has been studied in both humans and animals. However, its anti-inflammatory and chondroprotective properties have been inconsistent attributed to its low bioavailability, solubility, and high rate of metabolism in the gastrointestinal tract.19,39 The current study aimed to employ mPEG-PA hydrogel as a drug delivery system to localize and prolong the effect of Que and investigate its dose effect to treat OA in the knee using a post-traumatic OA rat model.
Rats received hydrogel with Que (50 and 500 µg) injections showed a reduction of LII relative to pretreatment. The relieve of pain symptom lasted throughout the 12 weeks post-treatment, except there was a slight increase in LII in the high-dose group at 8 weeks post-treatment. The decrease in LII was the greatest in rats that received a low dose of Que (50 µg). These suggest that Que is anti-nociceptive, which is consistent with previous in vivo studies that showed Que was able to reduce diabetes-, cancer-, and arthritis-induced pain.21,40-42 Furthermore, our results suggest that by employing mPEG-PA as a drug delivery system, the effect of Que could be confined and sustained. In contrast to our previous study, multiple injections of an antioxidant formulation were required to delay the progression toward asymmetric gait in OA rats when a drug delivery system was not employed. 21 However, there was a large variation in the difference in LIIs between rats in each treatment group. This can be explained by the episodic nature of OA pain, which can be affected by factors including temperature and the activity levels of the animals. 43
The histopathological assessment on cartilage degradation showed that the OA grade was significantly lower in the hydrogel group (grade 5) than the saline group (grade 6.5). This implies that the hydrogel alone may provide beneficial effects. Hydrogels, such as hyaluronic acid, are water-swollen polymeric material that had been used as viscosupplementation. Several in vivo studies had shown that IA injection of hyaluronic acid could reduce cartilage degeneration.44,45 Hence, we can postulate that the mPEG-PA hydrogel could provide lubrication to reduce cartilage degradation in the OA knees. Furthermore, the total OARSI score was significantly lower in rats received hydrogel with a low dose of Que than the saline group. This implies that, besides the hydrogel, Que released may provide additional beneficial effects to delay the progression of OA. Previous in vitro studies have shown that Que is chondroprotective by reducing the production of molecules, including prostaglandins (PGE2), nitric oxide (NO), and matrix metalloproteinases (MMPs), in both lipopolysaccharide (LPS) and pro-inflammatory cytokine interleukin-1β (IL-1β) induced inflammatory responses in human articular chondrocytes.10,46 Physically, a recent study has demonstrated Que was able to stabilize the extracellular matrix of the articular cartilage via hydrogen bonding and hydrophobic interactions. 47 Hence, the Que released into the IA space from the hydrogel may reduce the inflammatory responses of chondrocytes and interact with the extracellular matrix of the articular cartilage to provide protection and delay the progression of OA in rats. Although the median total score in rats which received a higher dose of Que was lower than the control group, the same significant differences cannot be shown.
Besides being a well-known antioxidant, Que has been shown to reduce inflammatory responses in macrophages and human synoviocytes in vitro.10,48,49 However, we were unable to show that the current treatment could reduce synovitis in vivo. One possible explanation is that since the synovitis of these OA rats is mild, the potential anti-inflammatory effect may not be significant enough to be detected. Furthermore, we have recently investigated the in vivo retention of a fluorescently labelled mPEG-PA in healthy Sprague-Dawley rats. Our results showed that ~25% of the hydrogel remained in the intra-articular space at 4 weeks after the IA injection (see Supplemental Material). It is known that the IA clearance rate of an OA knee is much faster than a healthy knee, all injected materials can be cleared out by 4 weeks post-injection. Since the synovitis of the knee was scored at the study end point (12 weeks post-injection), we may not be able to evaluate its immediate effect on the synovium.
One of the major limitations of this study is the use of a post-traumatic animal model. ACLT leads to permanent instability in the knee that persists throughout the course of OA. Hence, the sensitivity of treatment effects using this animal model may be compromised. Dunkin Hartley guinea pigs develop OA spontaneously and are histopathologically similar to humans, which might be a better animal model to study treatment efficacies. Moreover, the IA injections were given at a time point where OA was well developed and may not be salvaged, which could explain the small differences we observed. Furthermore, the OARSI scoring system used in this study described the different degree of cartilage degeneration, however, the scoring was assessed on the most advanced grade irrespective of its extent and vice versa on the most severe section of the knee. Therefore, this can lead to an overestimation of the severity of OA. In addition, even though gait analysis allows for a longitudinal monitoring on OA pain-related asymmetric gait of the rats, histological assessments were only performed at the end point. Therefore, any potential short-term effects of Que could not be revealed.
Further studies should investigate the potential antinociceptive mechanism of Que on OA pain and chondroprotection. OA pain has been shown to be induced by inflammatory cytokines, hence, whether Que could modulate inflammatory pain by reducing inflammatory cytokines should be investigated. In addition to the preferential affinity to the cartilage matrix, Que may also be able to protect the articular cartilage by reducing reactive oxygen species (ROS) and downstream factors such as MMPs. Recently, IL-1β has been demonstrated to mediate joint inflammation by inducing the generation of ROS and upregulating the expression of NADPH oxidases. 50 As Que is a strong antioxidant, it may provide potential chondroprotection by antagonizing ROS production and NOX4 expression.
This study investigated 3 aspects of OA in terms of pain, cartilage degradation, and synovial inflammation. Despite the large variations observed, our results suggested that a single IA injection of hydrogel and hydrogel with Que was able to reduce OA pain and delay the progression of OA. The insignificant differences observed in synovial inflammation was most likely due to the low degree of inflammation of the disease and the inability of the injected materials to retain in the IA space throughout the 12-week study period. In conclusion, the current approach to delay the progression of OA by localizing and sustained delivery of a bioactive component in a drug delivery system is promising. Currently, there are other oral SYSADOA in the market which their efficacy in treating OA were inconsistent. It may be meaningful to investigate their true efficacy using a similar approach.
Supplemental Material
Supplementary – Supplemental material for Intra-Articular Delivery of Quercetin Using Thermosensitive Hydrogel Attenuate Cartilage Degradation in an Osteoarthritis Rat Model
Supplemental material, Supplementary for Intra-Articular Delivery of Quercetin Using Thermosensitive Hydrogel Attenuate Cartilage Degradation in an Osteoarthritis Rat Model by Sze-Wing Mok, Sai-Chuen Fu, Yau-Chuk Cheuk, I-Ming Chu, Kai-Ming Chan, Ling Qin, Shu-Hang Yung and Ki-Wai Kevin Ho in CARTILAGE
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Hong Kong Research Grant Council, General Research Fund (CUHK14106014).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All animal experiments were approved by the Animal Ethics and Experimentation Committee of the Chinese University of Hong Kong (Reference No: 13/015/GRF).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
