Abstract
Objective:
The aim of this study was to evaluate the results of autologous bone marrow cell stimulation and allogenic chondrocyte implantation using 3-dimensional gel-type fibrin matrix in an animal model.
Design:
Eighteen rabbits were divided into 2 treatment groups. One group was treated with a microfracture and covering of it with gel-type fibrin (AutoBMS; n = 9), and the other group was treated with allogenic chondrocytes mixed gel-type fibrin at the cartilage defect (AlloCI; n = 9). The control group was untreated cartilage defect at the other side knee of each object. Twelve weeks after treatment, the cartilage was evaluated using the International Cartilage Repair Society (ICRS) scoring system, immunohistochemical staining, and modified O’Driscoll grading system.
Results:
The ICRS scores were similar in the AutoBMS (9.44 ± 2.44) and the AlloCI (9.33 ± 1.67) groups (P < 0.05). Immunohistochemical staining confirmed higher expression of cartilaginous collagen for both groups. The average difference (AutoBMS, 31.89 ± 6.54; AlloCI, 32.89 ± 5.25) in the modified O’Driscoll scores appeared to be nonsignificant (P > 0.05); however, both treatment groups showed significantly higher scores with respect to their control group (18.45 ± 1.65; 18.97 ± 1.58) (P < 0.05).
Conclusion:
This experimental study suggests autologous bone marrow cells stimulation and implantation of allogenic chondrocytes are both useful methodologies for regenerating hyaline-like cartilage in full-thickness cartilage defects in animal model.
Introduction
Damage to the cartilage tissue from trauma or pathologic conditions is common and often results in deterioration of the patient’s quality of life. 1 The articular cartilage has limited healing potential due to poor vascular supply and lack of self-regenerating properties of chondrocytes. 2 Cellular therapy is considered to be a pivotal component in biotechnological evolution and therapeutic strategies in the field of articular cartilage repair.3,4 Autologous chondrocyte implantation (ACI) has been widely performed clinically and has shown to yield superior tissue repair outcomes. 1 Despite the fact that ACI served as a successful treatment modality for the past 2 decades, its limitations have included donor-site morbidity, a limited supply of chondrocytes, phenotypic alternations during ex vivo expansion, cellular dedifferentiation during in vitro cultivation, and the need for a preceding surgery for culture.5,6
In our animal study, we had a choice of 2 alternative cellular therapies, the autologous bone marrow stimulation as a source of mesenchymal stem cells and the allogenic chondrocyte implantation. Allogenic chondrocyte implantation was performed in a single-step procedure, using allogenic chondrocytes harvested from other young New Zealand white rabbits. In addition, autologous bone marrow cell stimulation, the combination of a microfracture, and application of a scaffold was used. It is a more convenient and cost-effective procedure and does not require a preceding surgery for tissue cultivation and in vitro cellular proliferation and differentiation. 7
The objective of our study was to evaluate the results of autologous bone marrow cell stimulation and allogenic chondrocyte implantation using 3-dimensional gel-type fibrin matrix in an animal model, by analyzing the gross morphology and histology.
Methods
Eighteen male New Zealand white rabbits, weighing approximately 2500 g and between 5 and 6 months of age, were used for the animal study. The rabbits were randomly assigned into 2 treatment groups: the allogenic chondrocyte implantation group (AlloCI; n = 9) and the autologous bone marrow cell stimulation group (AutoBMS; n = 9). A control group was also randomly assigned one side knee of each object. A control group was set for the opposite knee of each treatment group, as these defects were left without any procedure. The study was approved in accordance with the policy of the University Hospital Clinical Research Center Animal Study Committee and regulations regarding animal experiments (INHA-IACUC No.: INHA 110503-92) prior to performing the study.
Rabbit Focal Articular Cartilage Defect
Intramuscular injection of ketamine sodium (Ketalar, Yuhan Corporation, 10 mg/kg) and xylazine hydrochloride (Rompun, Korean Bayer, 20 mg/kg) were used for anesthesia and both knee joints were surgically exposed. An anteromedial parapatella incision was used without disrupting the extensor mechanism. The knee joints were extended and the patella was dislocated to approach the trochlea. A cylindrical 3 × 3 mm-diameter and 1-mm-depth full-thickness chondral defect was created on the trochlea of the femur using a curette, surgical knife, and a diameter 3.0-mm bone biopsy equipment. Given that the cartilage thickness in rabbits is much thinner, further precautions were taken to avoid damage to the subchondral bone in the AlloCI group as osteochondral damage, which may stimulate bone marrow cells. This area of defect intermittently bore weight and contacted the articular surface when the knee joint was flexed at 70°.
Division and Cultivation of Cartilage Cells
The allogenic chondrocytes were harvested from sacrificed young (3 months old) New Zealand white rabbits. The cartilage specimens were delivered to the GMP-certified cell processing center at Sewon Cellontech, Korea, where cell isolation, cell proliferation, and harvest from primary cultures were performed. The isolated allogenic chondrocyte specimens underwent enzyme digestion process using the collagenase solution in CRM kit. Isolated cells were seeded in 25 cm2 tissue culture flasks containing Dulbecco’s modified Eagle’s medium with fetal bovine serum in CRM kit; they were then cultured for up to 14 days in the primary culture. The medium was changed every 3 days throughout the entire culture period. Cartilage cells were observed following the cultivation time with an inverted microscope. When the cartilage cell count reached 70 to 80 cell divisions in the culture medium, the cartilage cells were detached from the container using trypsin-EDTA and was cleansed with phosphate-buffered saline solution. The specimens were moved to a microtube and prepared to match 1 × 106/32 µL of the rabbits’ cartilage cells for transplantation.
Implantation of Allogeneic Chondrocytes and Autologous-Induced Bone Marrow Cells Stimulation Using Fibrin Matrix in the Defective Area
For the cartilages defect in the allogenic chondrocyte implantation group (AlloCI; n = 9), a gel-type mixture of allogeneic chondrocytes and fibrin (Chondron; Sewon Cellontech Co. Ltd., Seoul, Korea) was injected to fill the defect and the joint was held in slight extension for 5 minutes to allow the gel to harden.
For the AutoBMS groups, a hole depth of 2 mm was then made in the subchondral bone plate using a 2.0-mm drill bit at the center of the cartilage defect (3 × 3 mm) in order to ensure migration of marrow-derived cells, and the bleeding was sealed with the fibrin matrix (Chondron).
If continuous bleeding from the subchondral bone occurred, hemostasis was achieved by local application of bone wax (Bone Wax; Ethicon, Inc., Somerville, NJ) so as to prevent too much leakage from the subchondral bone, which leads to an increased instability of graft.
Five minutes after sealing with fibrin or implanting of AlloC-fibirn complex, the gel hardened, a probe was used to assess the graft hardness, and graft stability was routinely tested by 5 times cyclic bending of the knee in both groups.
The wounds was closed in layers using absorbable surgical sutures (Ethicon, Johnson & Johnson, Somerville, NJ), followed by the application of antiseptic solutions. All of the rabbits were treated with intramuscular antibiotic (cefazoline 30 mg) injections of 50 mg/kg for 3 days. The control group was set for the opposite knee of each treatment group, as these defects were left untreated, and the operation ended in the same manner.
Gross and Histological Examination of the Regenerating Tissue from Defects
The rabbits were randomly sacrificed at the 12th week after implantation. For macroscopic gross evaluation, both knee joints were surgically removed and evaluated. The specimens were examined under direct light microscopy by 2 independent experienced orthopedic surgeons who were blinded to the sample groups. The surgeons were asked to grade the samples according to the International Cartilage Repair Society (ICRS) evaluation score. 8
The specimens were then fixed in 10% neutral buffered formalin (Sigma, UK) and were decalcified with formic acid and nitric acid. Fixed and decalcified specimens were then embedded in paraffin blocks, and 5-µm sections were prepared. Hematoxylin-eosin stain was used for general morphological evaluation, and safranin O dye (0.5% in 0.1 M sodium acetate, pH 4.6, for 30 seconds) was used to detect proteoglycan. Masson’s trichrome staining was performed to evaluate the general status of collagen deposition and immunohistochemistry for type II collagen was performed according to the manufacturer’s protocol (Abcam, UK). Blinded to the group status, 2 independent experienced pathologists analyzed the repaired cartilage specimens for histologic and histochemical assessments using the modified O’Driscoll grading system.9,10
Statistical Analysis
Statistical analysis was performed using the SPSS statistical computer program (version 13.0, SPSS, Inc., Chicago, IL). The ICRS and the modified O’Driscoll score values for all the groups were presented with standard deviations. Comparisons of variables between the 2 treatment groups were performed using the parametric 2-sided independent t test. Differences were considered statistically significant at P < 0.05.
Results
Gross Appearance of Defects
Macroscopic examination at the 12th week after implantation revealed that the defects treated with allogeneic chondrocytes and autologous bone marrow stimulation both showed good filling with the surface appearing smooth. In contrast, the untreated cartilage defects of the opposite knee (AlloCI-control and AutoBMS-control) showed incomplete filling with yellow inflammatory fibrous tissue (Fig. 1). To qualitatively evaluate the regenerating tissues found in the AlloCI and AutoBMS groups, a visual method was used to calculate the ICRS score. The AlloCI and the respective control group (AlloCI-control) had an average of 9.33 ± 1.67 and 4.33 ± 1.67, respectively. When comparing the AutoBMS group and the respective control group (AutoBMS-control) the average was 9.44 ± 2.44 versus 4.38 ± 1.62, showing that both groups had notably superior cartilage regeneration capacity compared with the untreated control knees (P < 0.01). However, no statistically significant difference was found between the AlloCI and AutoBMS groups (P > 0.05) (Fig. 2).
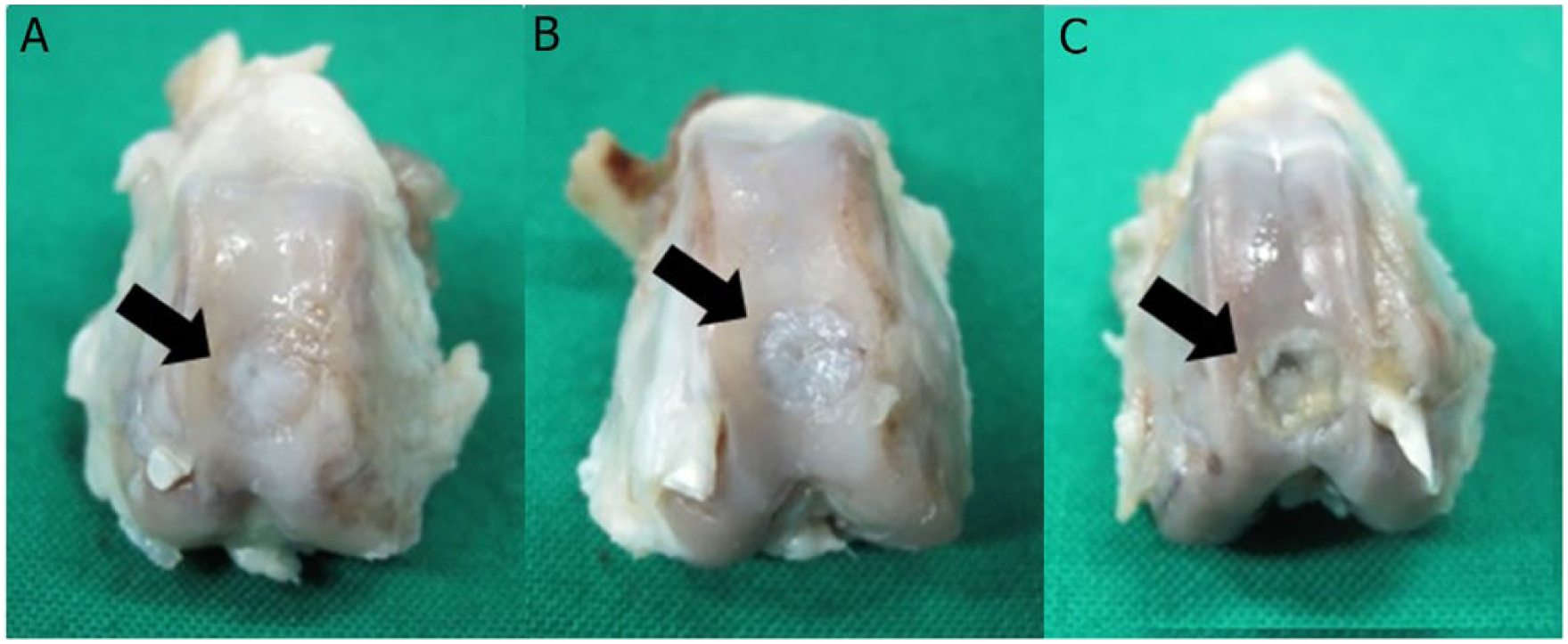
Macroscopic evaluation of the defective areas of articular cartilage from the AlloCI, AutoBMS, and control groups at 3 months after transplantation (
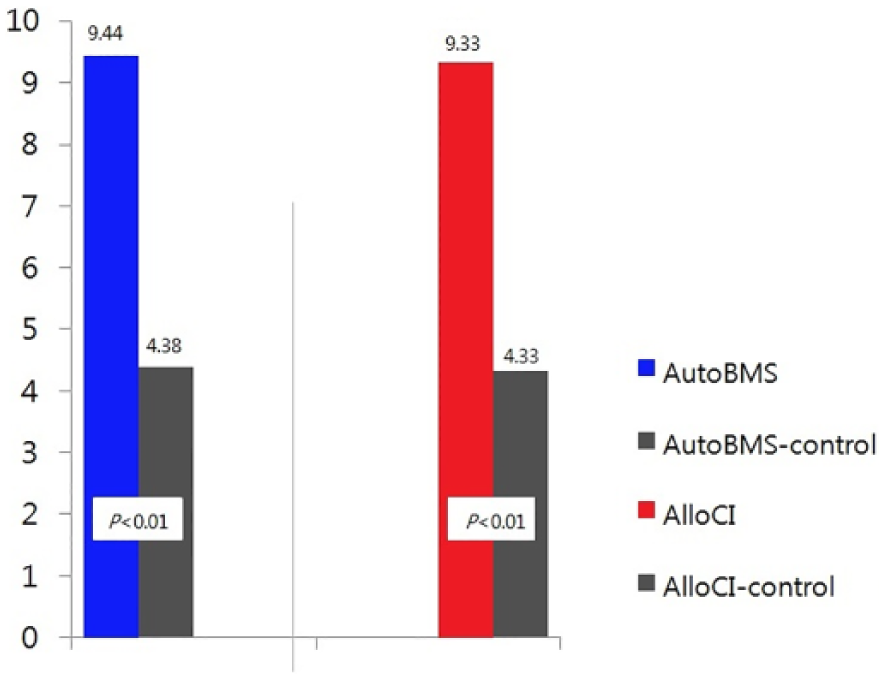
International Cartilage Repair Society evaluation scores of articular cartilage regeneration in the AutoBMS and AlloCI treatment groups were compared with their respective controls on the contralateral side 3 months after transplantation.
Histologic and Immunohistochemical Appearance of the Regenerated Cartilage Tissues
At the 12th week after implantation, microscopic inspection was conducted on the regenerated tissues in the 3 groups (i.e., the AlloCI group, AutoBMS group, and control groups). In the control group, the defects did not appear to undergo complete healing but were instead merely filled with fibrous soft tissue (Fig. 3C, F, I, and L). The various staining methods revealed that the defects in the treatment groups were filled with regenerated cartilage at the treated sites, although some areas appeared to be infiltrated by small amounts of fibrovascular tissue. The regenerating cartilage of the 2 treatment groups showed a mixture of hyaline cartilage and fibrocartilage, which was well connected to the surrounding normal cartilage (Fig. 3A and B). With immunohistochemical staining, higher expression of type II collagen was verified compared with the control groups (Fig. 3D and E). Even with Masson’s trichrome staining, a clearer configuration and distribution of chondrocytes and collagen were visible (Fig. 3G and H). To verify the glycosamionoglycan (GAG) substance, the main component of cartilage tissues, safranin O staining was used. Compared with the control group, the 2 treatment groups showed higher concentrations of GAG substance (Fig. 3J and K).
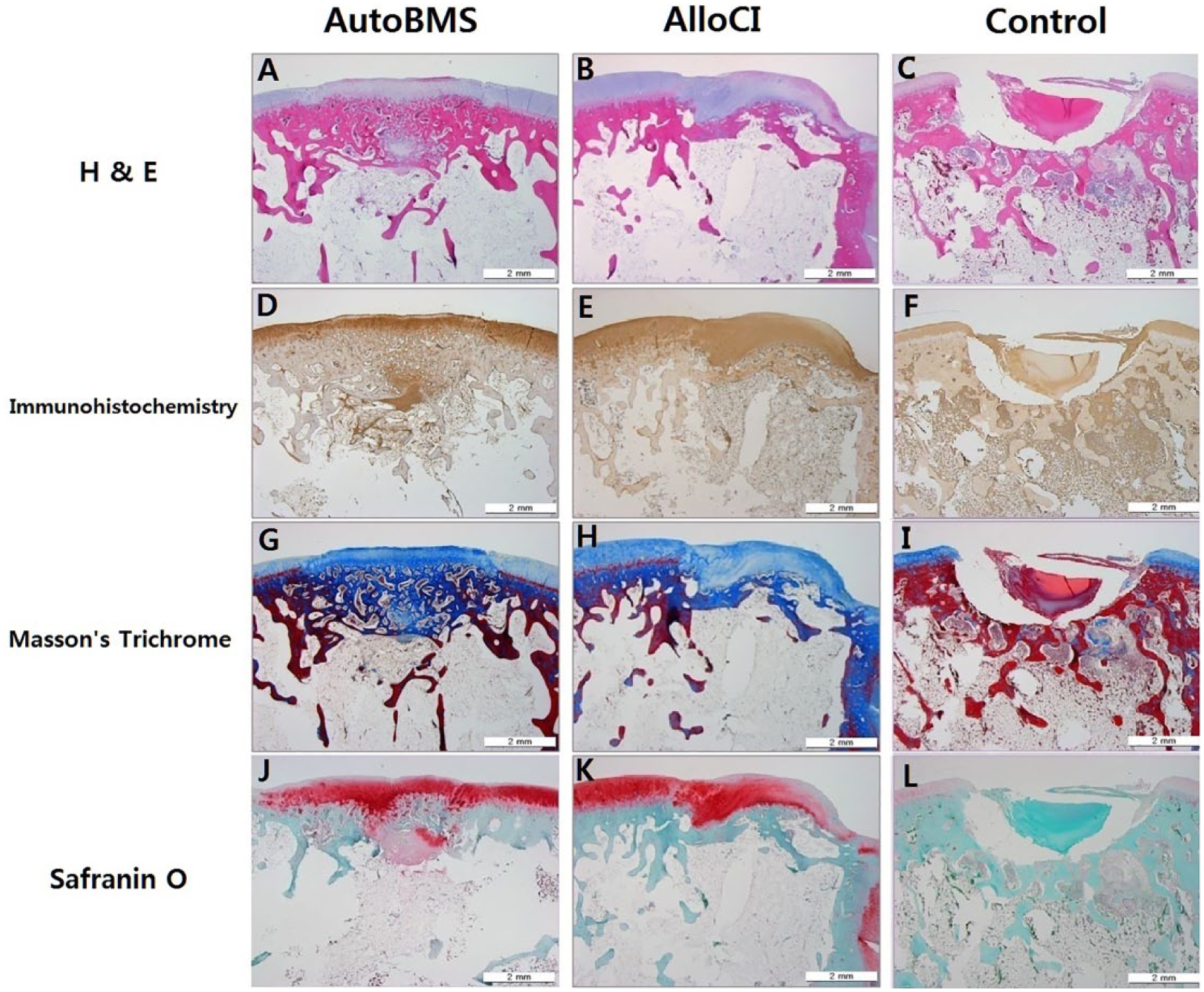
Histology (hematoxylin-eosin) of the defect site of a rabbit’s knee in AutoBMS-treated group
Using the modified O’Driscoll grading system, the histologic outcomes were quantitatively analyzed. The morphological features of the regenerated tissue were similar to those of normal hyaline-like cartilage in both the treatment groups, and showed more than 90% defect fillings. However, union with the surrounding tissue was better incorporated in the AlloCI group than in the AutoBMS group.
When comparing these groups by quantification, the AlloCI treatment group had an average of 32.89 ± 5.25 compared with the AlloCI-control group, which had 18.97 ± 1.58 (P = 0.008). The AutoBMS treatment group and the AutoBMS-control group showed 31.89 ± 6.54 and 18.45 ± 1.65 (P = 0.024), respectively. However, the results between the treatment groups were not statistically significant (P > 0.05) (Fig. 4).
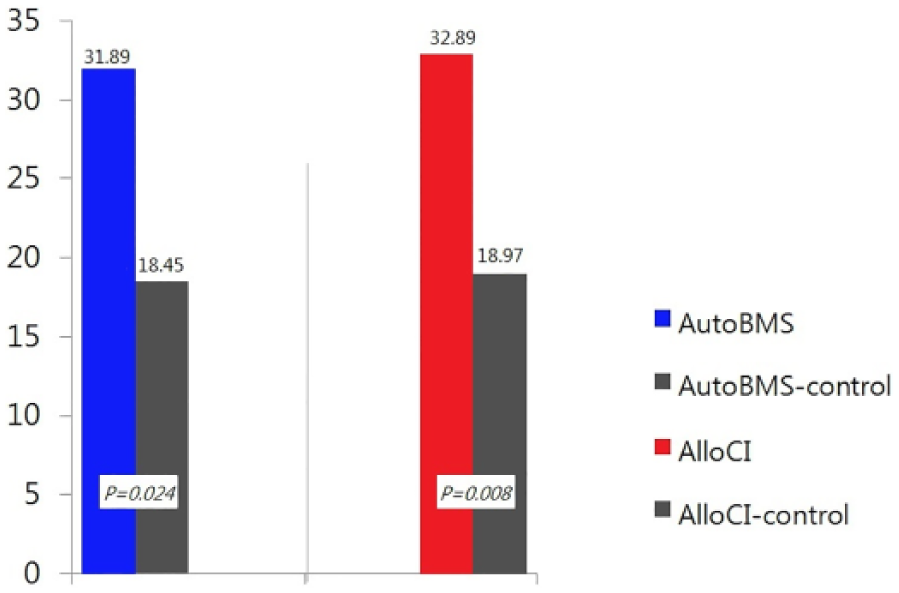
Quantitative histologic evaluation of the regenerated cartilage using modified O’Driscoll grading system scores at 3 months after treatment, with the AutoBMS and AlloCI groups together with their respective controls.
Discussion
In our animal study, we evaluated and compared the structural, histologic, and immunohistologic characteristics of tissue regeneration in cartilage defects after 2 different types of cellular therapies: allogenic chondrocyte implantation and autologous bone marrow cell stimulation using fibrin matrix. The most critical findings of the study were that both types of cellular therapies showed better regenerating abilities than the untreated control group in full-thickness cartilage defects. In addition, both gross examination using the ICRS evaluation score and histologic examination using the modified O’Driscoll grading system indicated no statistically significant difference between the 2 treatment techniques (P > 0.05). Both treatment groups showed statistically significant results that were superior to those of the control group (P < 0.05). Immunohistochemical staining was performed to identify the types I and II collagen and evaluate the characteristics of the regenerated tissues, whether fibrous or hyaline cartilage. The majority of regenerated tissue in both treatment groups resulted in differentiation into type II collagen. The histologic findings indicated hyaline-like cartilage with mixed properties of fibrous and hyaline cartilage, which is similar to the outcomes of allogenic chondrocyte implantation and autologous matrix-induced chondrogenesis reported in previous studies. 11
It is generally recognized that cartilage contains a relatively lower density of chondrocytes and a higher density of extracellular matrix. These characteristic properties lead to limited migration of surrounding chondrocytes to the sites of the defects and have been associated with lower regenerative abilities than other tissues.3,12 Although smaller defects of cartilage can be repaired to a certain extent, they generally regenerate fibrous cartilage that has a weaker mechanical strength than the native hyaline cartilage. 12
Cellular understanding of cartilage regeneration has contributed to the development of cellular techniques in the treatment of cartilage damage. Microfracture techniques were devised to regenerate cartilage from the proliferation of bone marrow mesenchymal stem cells that leaked from the sites of subchondral damage. The subsequent production of fibrin clots resulted in a lack of mechanical stability due to a relative abundance of type I collagen and the scarceness of proteoglycan, resulting in fibrous cartilage.13,14 Due to the disproportionate fibrous and hyaline components in regenerated cartilage, structural degradation subsequently begins to occur along with the relapse of symptoms. Although pain and mobility tend to improve for up to 2 years in 60% to 70% of the patients treated with microfractures,15-17 autologous bone marrow cell stimulation using biocompatible scaffolds was able to compensate for the disadvantages and reciprocate the advantages associated with microfracture techniques.18,19 Gille et al. reported that 87% of the patients who received autologous matrix-induced chondrogenesis were satisfied with the treatment outcomes at a 5-year follow-up. 7 In addition, ACI was first introduced by Brittberg et al. in 1994 and it has proven excellent results in the regeneration of cartilage and offered better applicability, regardless of the degree of damage. 20 However, in chondrogenesis techniques, the in vitro differentiation of chondrocyte had several drawbacks because delivery in a liquid state caused difficulties in fixation at the transplantation site. 21 The use of scaffolds such as collagen, hyaluronan, and fibrin has improved cellular delivery to the target site and the structural and mechanical stability of the regenerated cartilage.22-24
Regarding the use of fibrin scaffold in the current animal study, previous studies have stated that fibrin has a 3-dimensional structure characterized by biocompatibility, absorptivity, degradability, and lack cellular toxicity.25-28 Furthermore, it was reported that within a fibrin glue, type II collagen and thrombin facilitated the differentiation of chondrocytes into hyaline cartilage and differentiated cells, respectively. 29 Kim et al. performed clinical, radiologic, and arthroscopic evaluations of 30 patients treated with autologous matrix-induced chondrogenesis using gel-type fibrin matrix and concluded that the mechanical and histologic properties of the regenerated cartilage were similar to those of normal cartilage. 30 Despite the advantages of the scaffolds, histologic variability of the cartilage after transplantation has not yet been completely resolved. Because the cellular properties and potential regenerability of the cartilage differ depending on the condition of the donor cartilage, therapeutic outcomes are anticipated to be variable. Factors such as donor sites, patient age, and physical conditions must be considered when selecting potential cells for transplantation.31,32 Verbruggen et al. reported that the quality and differentiability of harvested chondrocytes differ depending on the patient’s age. 32 In addition, juvenile human chondrocytes have greater potential for restoring articular cartilage than adult cells, and they can be transplanted with less fear of rejection. This suggests a new allogeneic approach to restoring articular cartilage in older individuals. 33
One of the most prominent downsides of the allogenic chondrocyte implantation is immunity rejection.
Wide evidence exists demonstrating that the isolated allogenic chondrocyte implantation induces an immune response that gradually destroys the resulting cartilage tissue. 34
It is also indicated that after the detection of implanted allogenic chondrocytes by the host’s immune system, the cartilage was slowly destroyed over time by large infiltrations of mononuclear lymphoid cells. Particularly, macrophages accumulated around the chondrocytes immediately after implantation, while later stages of cartilage destruction showed large populations of natural killer and cytotoxic suppressor T cells. 35
A patient without previous surgery in addition to the use of young donor tissue would be most advantageous for the use of allogenic chondrocyte implantation—the most important consideration in utilizing allogenic chondrocytes is the potential for immunogenicity.
In other studies, the use of synthetic biomaterials combined with allogenic chondrocytes has been explored for tissue repair with considerable success.36,37
In one study, rabbit chondrocyte-fibrin gels were embedded within a PGA-lactic acid polymer. Cartilage defect was filled with hyaline-like cartilage tissue, and while a large number of mononuclear cells were observed around the defect 4 weeks after surgery, none of these cells infiltrated into the newly formed cartilage. The mild immune response at 4 weeks was attributed to polymer scaffold resorption, which was complete by 12 weeks when no immune response was observed. 37
More specifically, the 3-dimensional scaffold is believed to form a protective barrier around the chondrocytes, which blocks both the infiltration of host immune cells into the graft as well as the escape of immunogenic chondrocytes out of the graft.36,38
In our study, both the gross and histologic evaluations of the regenerated cartilage by allogenic chondrocyte implantation with fibrin gel and autologous matrix-induced chondrogenesis indicated that the regenerated cartilage was similar to native hyaline cartilages. Adverse effects including tissue rejection were not evident, particularly with allogenic chondrocyte implantation.
The microfracture technique has been used to promote the stimulation and recruitment of mesenchymal stem cells from the bone marrow to regenerate damaged cartilage. 13 The pitfalls of this techniques are that regenerated cartilage that has more fibrocartilaginous than hyalinocartilaginous components and that improvement in pain and articular functions have been limited to smaller articular defects. 14 However, the use of scaffolds such as collagen membrane, hyaluronan polymer, and fibrin gel has been confirmed to promote cellular differentiation and the formation of cartilage matrix and ultimately to provide better mechanical stability and cellular regeneration.30,39,40
Our animal study has limitations that must be addressed regarding the analysis. First, despite our efforts to quantify the gross and histologic results in all groups, the small sample size contributed to larger statistical deviations in the current study compared with previous reports. We could not further extend the representation of our results. Nonetheless, the study provided valuable findings in the comparisons between allogenic chondrocyte implantation and autologous bone marrow cell stimulation in various aspects of cartilage regeneration. Second, because our study design did not compare ACI to the 2 other cell therapies currently being used, areas of future research remain. Third, our animal study did not include degenerative cartilage models, that is, old age animals; it only contained young animals. There are still many risks in directly applying these techniques to human surgery although these experiments yielded good results from animal studies. From this perspective, our study is limited and further research is necessary for the degenerative arthritis animal model and clinical study.
In conclusion, full-thickness articular cartilage defects treated with allogenic chondrocyte implantation and autologous bone marrow cell stimulation using fibrin matrix produced hyaline-like cartilage that had significantly better qualities than the untreated control group. Because of several pitfalls of ACI such as the unpredictable differentiability of the transplanted chondrocytes, limited numbers of chondrocytes from donors and age-dependency of donors, allogenic chondrocyte implantation and autologous bone marrow cell chondrogenesis are expected to serve as an alternative technique for effective articular regeneration, with the additional benefit of cost-effectiveness. Because the safety and efficacy of both allogenic chondrocyte implantation and autologous bone marrow cell chondrogenesis were verified by the gross and pathologic evaluation of regenerated cartilage in our animal study, we suggest the results of this study should encourage the development of novel treatments for cartilage injury.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Academic Research Foundation of Jeju National University Institute of Medical Science in 2014.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved in accordance with the policy of the University Hospital Clinical Research Center Animal Study Committee and regulations regarding animal experiments (INHA-IACUC No.: INHA 110503-92) prior to performing the study.
