Abstract
Objective
Autologous chondrocyte implantation is a necessary procedure for the repair of articular cartilage defects; however, isolated chondrocyte implantation requires a 2-step procedure (for harvesting and implantation) and is limited by cytotoxicity due to enzymatic digestion. Therefore, in this
Design
Cartilage fragments were obtained from 7 female patients with knee osteoarthritis (OA) and embedded in atelocollagen gels. As a control, chondrocytes were isolated and embedded in gels in the same manner. These composites were cultured for 3 weeks, and cell proliferation and matrix production were evaluated using histology and immunochemistry.
Results
Histologically, minced cartilage showed cell migration from the cartilage fragments into the gel, with the Bern score and cell count in the minced cartilage group being significantly higher than those in the control group. Immunohistochemistry revealed that the number of Ki67-positive cells, the expression of LECT-1 and TGF-β, and the glycosaminoglycan content were significantly higher in the minced cartilage than in the control group. Minced cartilage exhibited superior cell migration, proliferation, and glycosaminoglycan content than isolated chondrocytes.
Conclusion
Our findings support that minced cartilage has a favorable potential for cell proliferation and matrix production compared with the isolated chondrocytes after enzymatic treatment.
Introduction
Although articular cartilage is an important tissue in weight bearing on a joint and ensuring smooth motion of articular surfaces, it is well known that articular cartilage has a limited capacity for self-repair once it is damaged.1,2 Damage to articular cartilage leads to osteoarthritic changes within the joint, which often result in joint dysfunction and pain. Therefore, appropriate interventions are needed to repair damaged cartilage3,4 with the goal of replacing the cartilage defect with tissue that closely approximates the histology of the surrounding native hyaline cartilage, returning the patient to previous activity and providing long term durability of the repair. To achieve these goals, cell-based repair techniques, such as autologous chondrocyte implantation (ACI), have been used to repair large diameter cartilage defects that exceed an area of 2.5 to 3.0 cm2. Good clinical results after ACI have been reported since Britteberg and colleagues first described the breakthrough method of transplanting human autologous chondrocytes to treat defects of the articular cartilage.5-8 Ochi
Recently, alternative 1-step procedures, using cartilage fragments have been developed to address these issues. Although first described in 1982, interest in 1-step procedures increased in 2006 being described in several animal studies,14,15 with satisfactory clinical results having been reported for minced cartilage implantation.16,17 In this approach, about 200 mg of hyaline cartilage is harvested from a low load-bearing area and minced into 1- to 2-mm pieces. The fragmented cartilage is dispersed onto a biodegradable scaffold with the cartilage-scaffold composite fixed by fibrin sealant. Subsequently, this composite is transferred onto the cartilage defect via mini-arthrotomy. Cole
In this study, we hypothesized that implantation of minced cartilage embedded in atelocollagen gel could have favorable properties such as chondrocytes proliferation, migration, and production of cartilage matrix in atelocollagen gel. Therefore, the purpose of this
Methods
Seven female patients (mean age 79.1 years; range, 69-88 years) who were undergoing a total knee arthroplasty were enrolled into our study for the purpose of harvesting articular cartilage. All included patients had a clinical diagnosis of primary OA, Kellgren-Lawrence grade 3 or 4, and a varus alignment of the knee. Patients with secondary OA, such as posttraumatic OA, and/or a systemic joint disease, such as rheumatoid arthritis, were not considered.
Our study was approved by the local ethical committee of our university, and informed consent was obtained from all individual participants included in the study.
Preparation of Cartilage/Atelocollagen Composite
After bone resection, articular cartilage was harvested from the patellar groove, lateral femoral and condyle, in a sterile fashion. The cartilage was washed in 0.9% sodium chloride and subsequently minced manually using a scalpel to obtain cartilage fragments of <1 mm3. To isolate chondrocytes, cartilage specimens were treated with 0.25% trypsin (Gibco, Carlsbad, CA, USA) in sterile saline for 30 minutes, followed by 0.25% collagenase type 2 (Gibco, Carlsbad, CA, USA) in Dulbecco’s modified Eagle’s medium (DMEM, Gibco, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS) (Sigma, Taufkirchen, Germany) and antibiotics (penicillin [10,000 units], streptomycin [10,000 μg/mL]) (Nacalai tesque, Japan) for 4 hours at 37°C in a culture tube, according to methods previously described. 1 The chondrocytes were washed 3 times with culture medium and then filtered through a 70-mm sterile nylon mesh (cell strainer, BD Biosciences Discovery Labware, Franklin Lakes, NJ, USA).
Culture of Cartilage/Atelocollagen Composite
Isolated chondrocytes (2.0 × 105 cells) were dispersed and mixed in 100 μL of atelocollagen gel (Koken, Tokyo, Japan) to form the chondrocyte group (isolated chondrocyte; group IC). In 2 other groups, 12.5 mg (group M1) or 25 mg (group M2) of minced cartilage were mixed in 100 μL of atelocollagen gel. All cell mixtures were placed in 6-well culture plates and incubated in a mixture of 5% CO2 and 95% air at a temperature of 37°C for 30 minutes to form atelocollagen composites ( Fig. 1 ). A culture medium containing 5% FBS and antibiotics was subsequently added into each well. The mixture was then incubated, again, in a mixture of 5% CO2 and 95% air at 37°C for 3 weeks. The culture medium was changed every 3 days, and L-ascorbic acid (50 μg/mL) was added every 2 days. After 3 weeks, each composite was divided into 2 parts, with one half of each sample fixed in 4% paraformaldehyde (PFA), and the other half used for biochemical analysis.
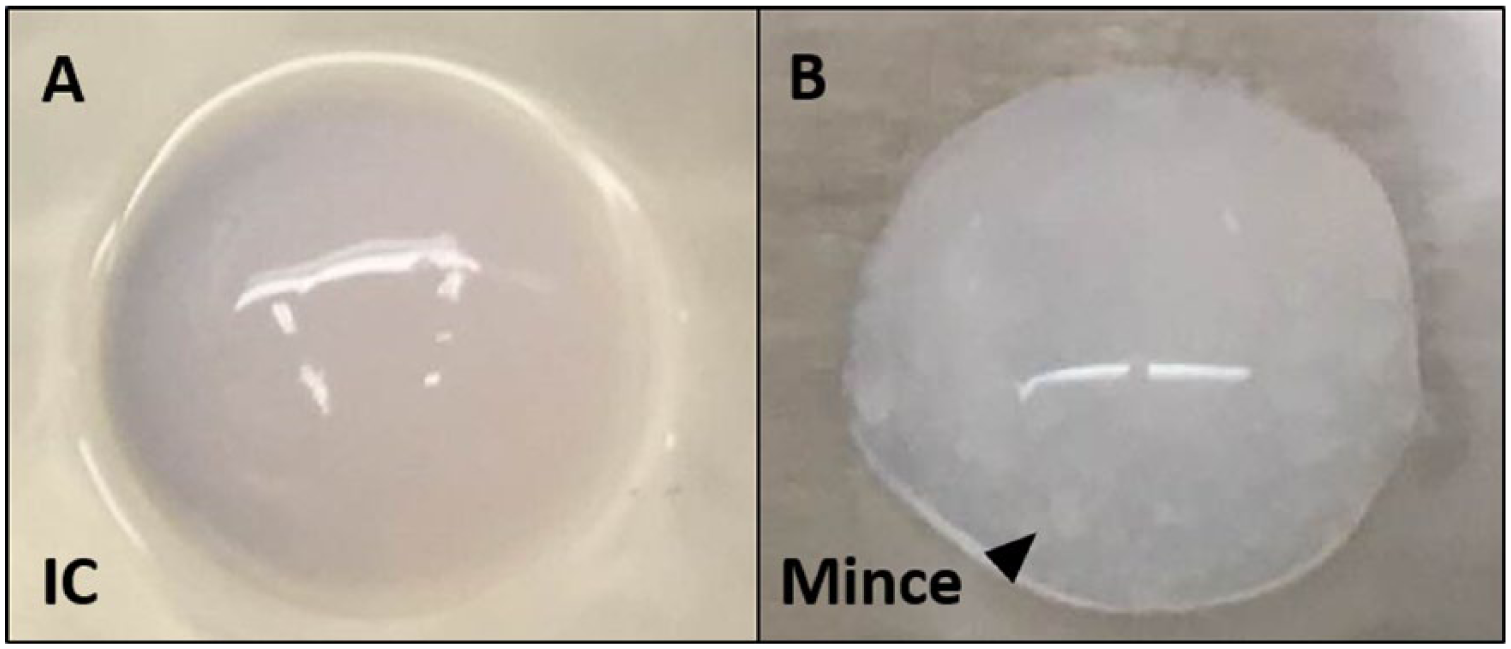
(
Histological Analysis
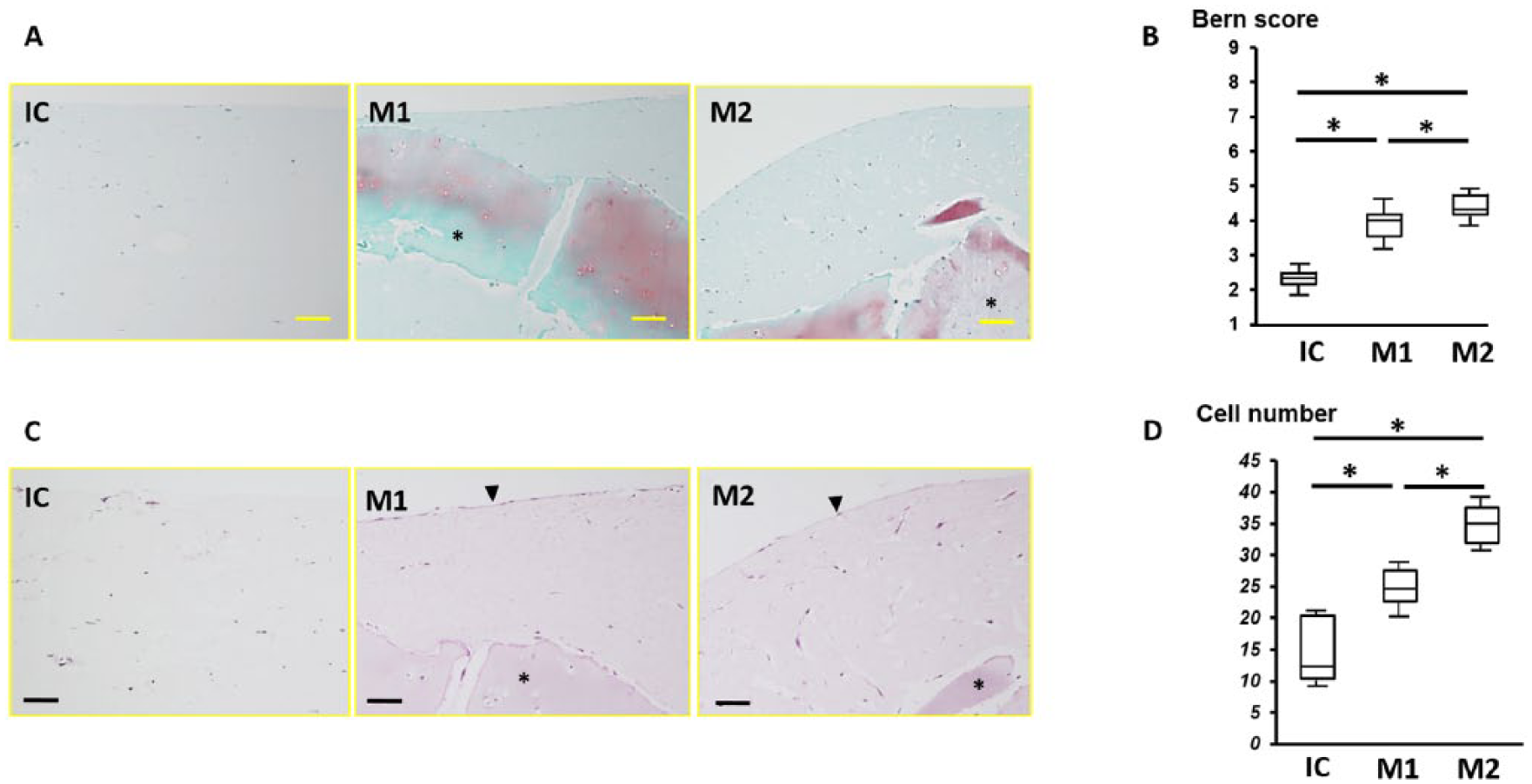
Samples for histological analysis were fixed in 4% PFA, at 4°C overnight and subsequently embedded in paraffin. Four micrometer sections were prepared and stained using hematoxylin and eosin (H&E) and safranin-O Fast-Green. Each sample was evaluated using the Bern score, according to the methods previously described. 18 The Bern score (minimum score, 0; maximum score, 9) is based on 3 items—uniformity and intensity of safranin-O staining, distance between cells/amount of matrix produced, and cell morphologic characteristics—with each item scored from 0 to 3.
For the evaluation of chondrocyte migration and proliferation in the atelocollagen gel, 6 areas (500 μm × 500 μm grid) were randomly selected in each section and chondrocytes were counted under a magnification of ×400.
Immunohistochemistry
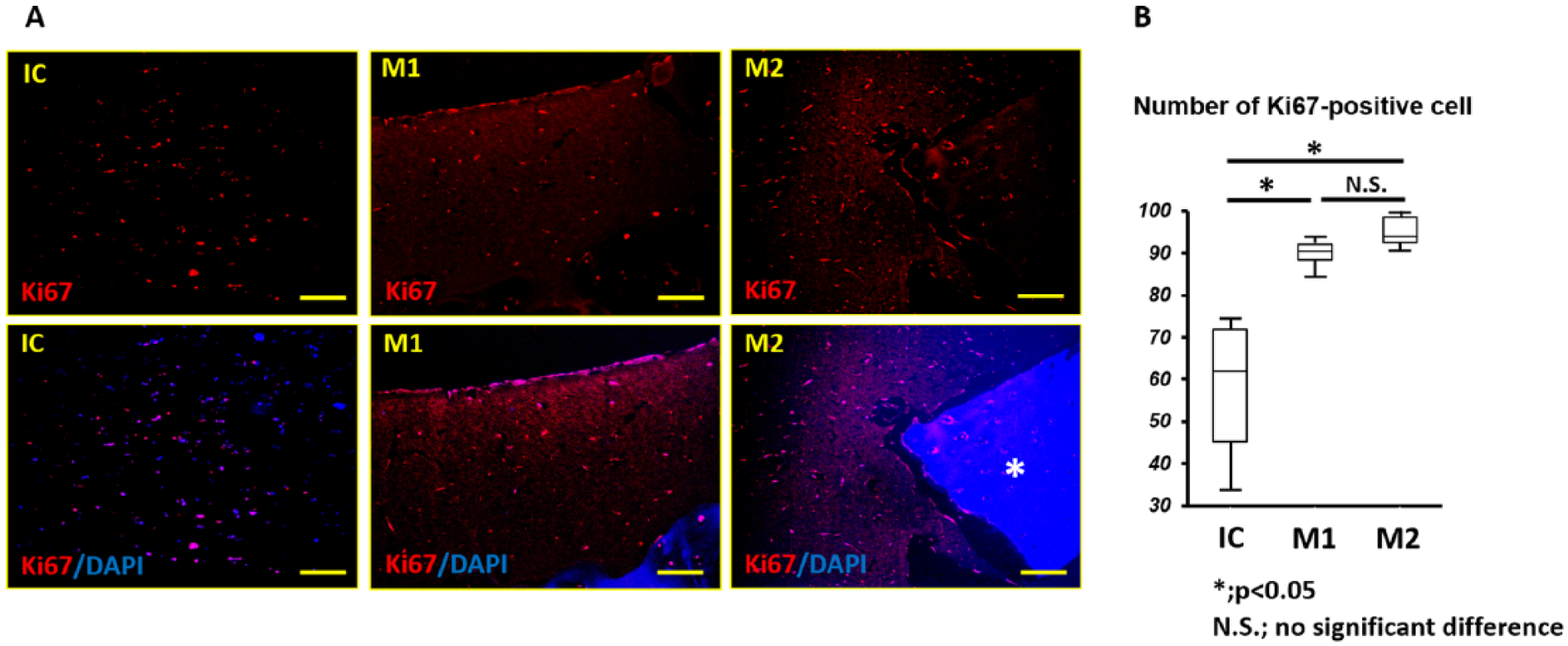
Immunostaining of each section was performed using anti-Ki67 antibody (Richard-Allan Scientific, Kalamazoo, MI, USA), anti-LECT1 antibody (Abcam, Cambridge, UK), anti-transforming growth factor (TGF)-β antibody (Abcam, Cambridge, UK), anti-collagen type I antibody (Novus Biologicals, Littleton, CO, USA), and anti-collagen type II antibody (Kyowa Pharma Chemical, Toyama, Japan). As the secondary antibody for Ki67, we used Alexa Flour 568-conjugated anti-rabbit IgG (Molecular Probes, Invitrogen, Carlsband, CA). Alexa Flour 488-conjugated anti-rabbit IgG was used for LECT1 and TGF-β. The negative control was treated with IgGs isotypes to replace primary antibodies. A DAPI (4′,6-diamidino-2-phenylindole; Dojindo Laboratories, Kumamoto, Japan) solution was applied for nuclear staining. For collagen types I and II, the sections were visualized using the avidin-biotin system (Vectastain Elite ABC Mouse IgG kit, Vector Laboratories, Inc., Burlingame, CA) and 3,3′-diaminobenzidine (Peroxidase Substrate Kit, Vector Laboratories, Inc.), according to the manufacturer’s instructions. For the evaluation of cell proliferation ability, the ratio of Ki-67 positive cells to the total cell count was calculated in each section.
Evaluation of Glycosaminoglycan Contents
After the 3-week period incubation, half of the composites for each group were used for biochemical assays for glycosaminoglycan (GAG)/DNA quantification, using the Blyscan Glycosaminoglycan assay kit (Biocolor, Carrickfergus, UK), according to the manufacturer’s protocol.
Statistical Analysis
The data were expressed as a mean and standard deviation (SD). One-way analysis of variance, followed by Turkey’s
Results
In the IC group, dispersed chondrocytes were observed in the atelocollagen gel, with many cells accumulated at the edge of the gel. In the M1 and M2 groups, chondrocytes outside of the cartilage fragments were observed, indicative of cell migration from the cartilage fragment. The cartilage fragments were stained with safranin-O in groups M1 and M2 (
Fig. 2
). The mean ± SD number of chondrocytes in the atelocollagen gel in groups IC, M1, and M2 was 15.2 ± 10.8, 24.9 ± 5.4, and 35.4 ± 17.8 cells, respectively, which was significant between all groups (
(
Positive Ki67 cells, indicative of the proliferation capacity of chondrocytes in atelocollagen gel, were identified in all groups (
Fig. 3
). The average proportion of Ki67-positive cells in groups IC, M1, and M2 was 58.1% ± 30.9%, 89.6% ± 11.4%, and 94.9% ± 6.0%, respectively. This proportion was significantly difference just between IC and M1, as well as IC and M2 (
(
Immunohistochemistry for TGF-β and LECT-1 was performed to examine whether these three groups had anabolic factors. LECT-1 was expressed in all groups, with greater intensity in groups M1 and M2 than in group IC, as well as greater intensity in group M2 than in group M1 ( Fig. 4 ). Similarly, TGF-β expression was greater in groups M1 and M2 than IC, as well as in group M2 than M1 ( Fig. 5 ). In groups M1 and M2, TGF-β was intensely expressed at the edge of the gel.
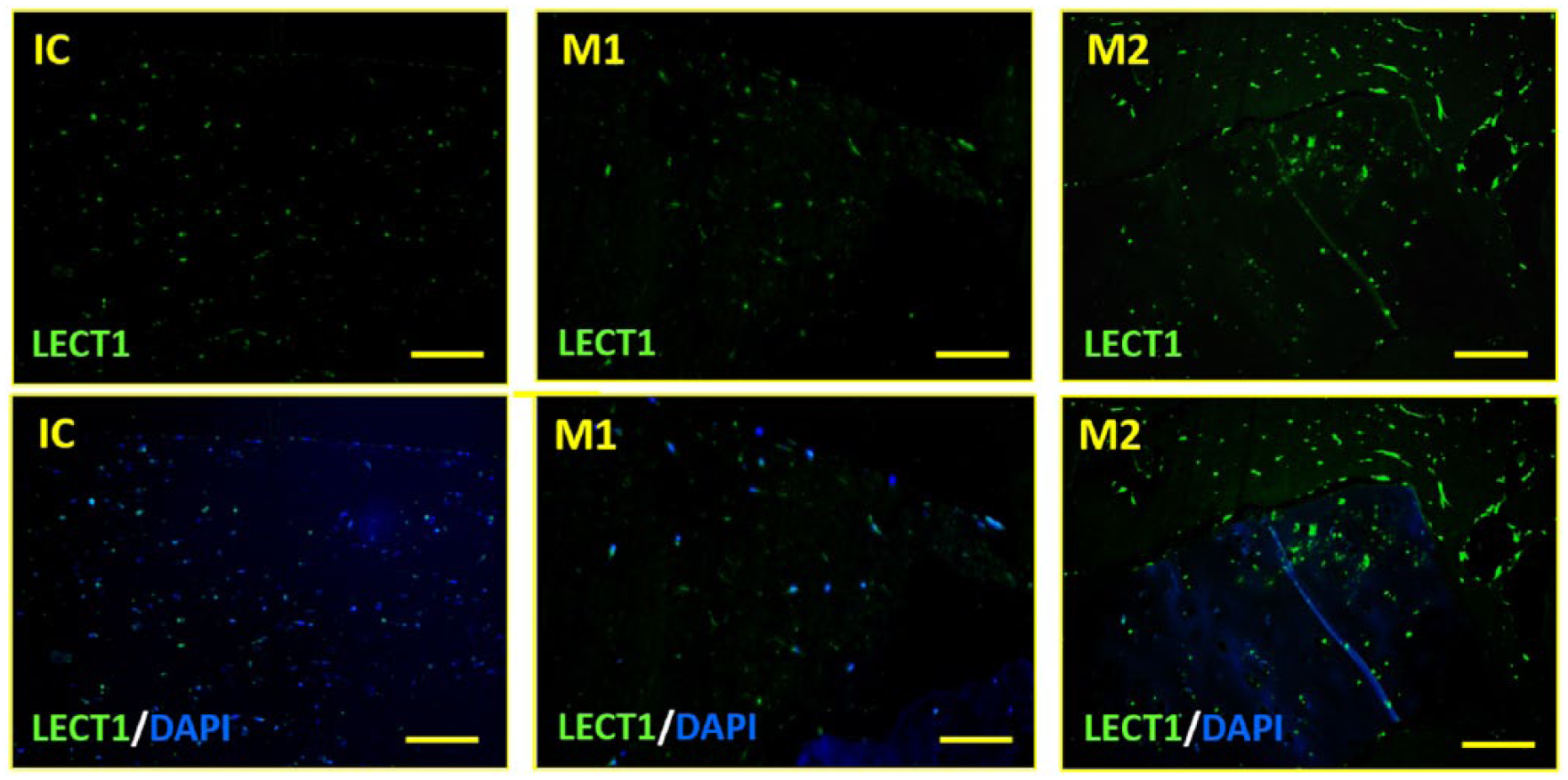
Immunohistochemistry of LECT-1; bar = 100 μm. LECT-1 was expressed to a greater intensity in groups M1 and M2 than in group IC (isolated chondrocyte).
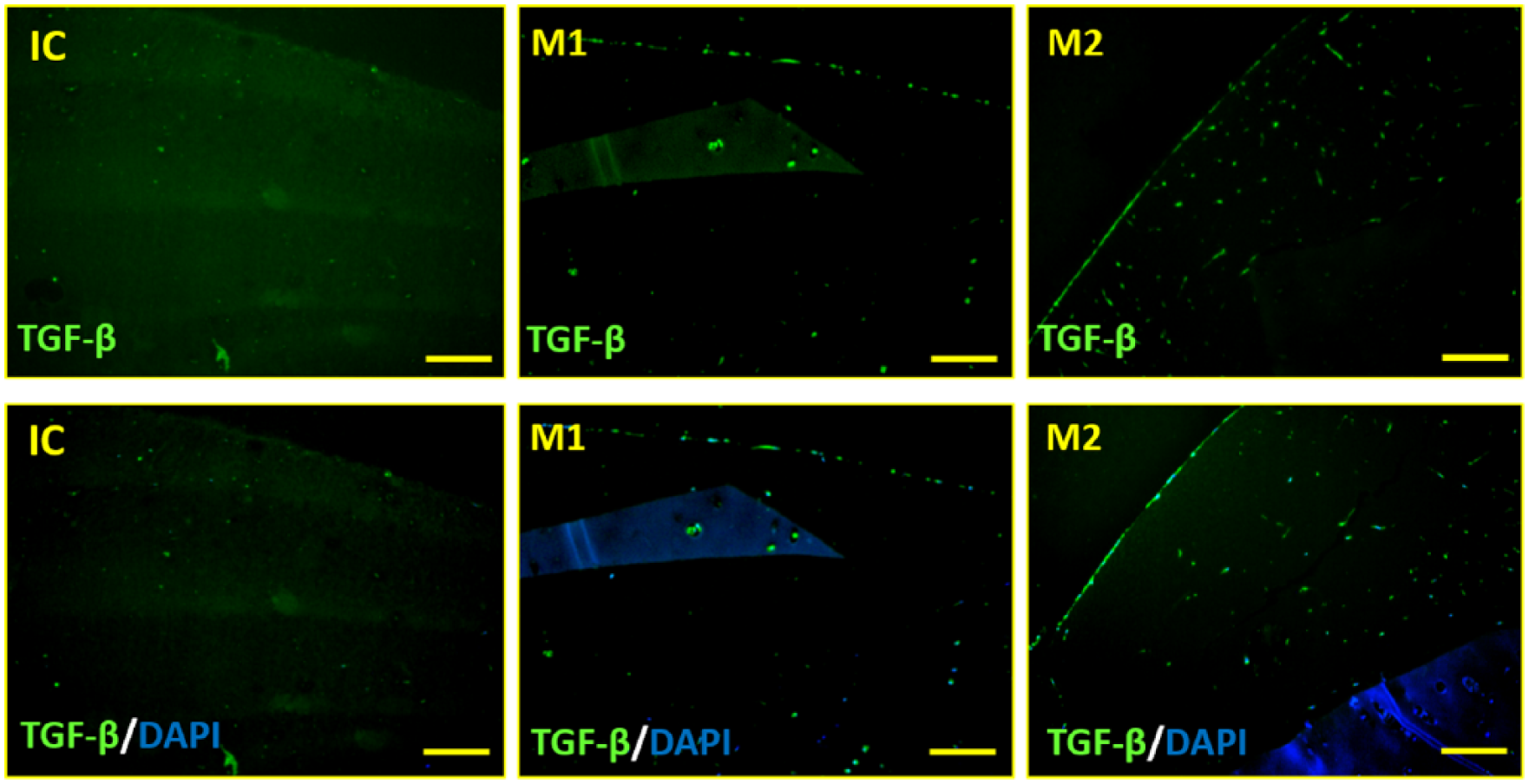
Immunohistochemistry of TGF-β; bar = 100 μm. TGF-β was expressed to a greater intensity in groups M1 and M2 than in group IC (isolated chondrocyte).
With regard to immunohistochemistry for type I collagen, the atelocollagen gel was well stained in all groups, with no staining of cartilage fragments. In contrast, cartilage fragments were well-stained for type II collagen. Chondrocytes in the gel also had positive staining for type II collagen ( Fig. 6 ).
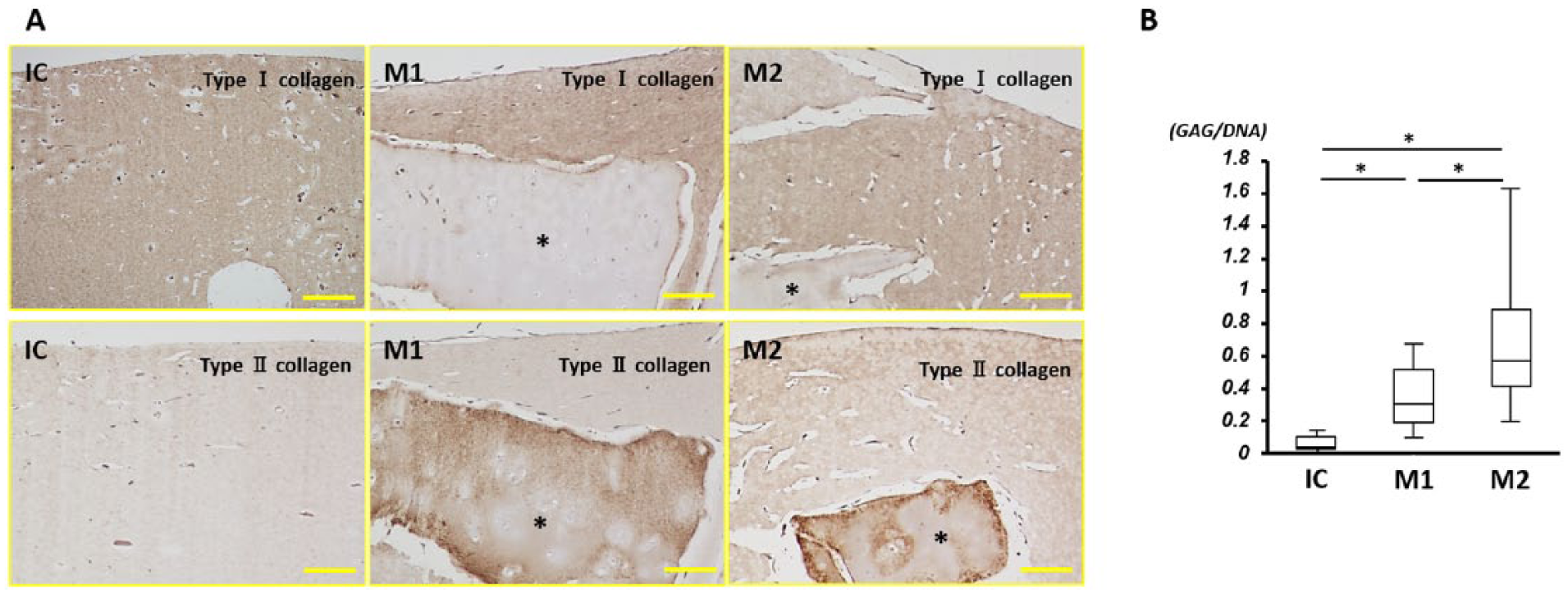
(
On biochemical analysis, the GAG-to-DNA ratio in group M2 was the highest among the 3 groups, as well as being higher in group M1 than group IC, with a significant difference in the ratio between all groups (
Discussion
In this study, we found that implantation of minced cartilage embedded in atelocollagen gel could be more effective for chondrocytes proliferation, migration, and production of cartilage matrix in atelocollagen gel than isolated chondrocytes in atelocollagen gel.
Application of cartilage fragments can be used as a 1-step procedure for the repair of articular cartilage defects, without the need for cell culture. The intra-articular environment after implantation of minced cartilage embedded in atelocollagen gel might be beneficial, as a 1-step procedure, to promote outgrowth of chondrocytes and to induce the culture environment for chondrogenesis.19,20 This approach might be useful for the treatment of large cartilage defects which will be an important future task to be solved. To cover a large cartilage defect, a large amount of cartilage fragments is required. However, there is a limit to the extent of cartilage that can be harvested in patients due to donor site morbidity. Therefore, a suitable scaffold to support the three-dimensional proliferation of chondrocytes and matrix production is needed. Hence, in this
In previous studies regarding the use of minced cartilage for repair of a defect, minced cartilage was fixed to the area using fibrin glue.16,17 Fibrin is used in practice because of its biocompatible and biodegradable properties. However, several animal studies have demonstrated the negative effects of fibrin on cell migration and tissue repair.21,22 Moreover, there is the possibility that exogenous fibrin may induce an immune response. 23 It is to address these limitations of fibrin that we used atelocollagen gel as a scaffold, according to previous reports and our experiences.9-12
Atelocollagen, with the inclusion of type I collagen, has the capacity to remove telopeptides, which are antigenic determinants on the peptides chains of type I collagen.24,25 Previous studies have confirmed the effectiveness of atelocollagen gel in maintaining chondrocyte phenotype in 3-dimensional
Although the benefit of cartilage repair using chondrocytes has been reported, the source of chondrocytes also needs to be considered to improve chondrocyte mobility at the donor site for repair of a large cartilage defect. As such, the use of cartilage fragments has been proposed as a cell source, without isolation and culture.15,29,30 In our present study, migration of chondrocytes in the atelocollagen composites was identified even though the cell source was OA minced cartilage. Similarly, compared to composites of suspended chondrocytes from enzymatic digestion, the composites of suspended minced cartilage contained significantly more chondrocytes and more Ki67-positive cells in the gel. These findings are indicative that the enzymatic digestion procedure damaged chondrocytes. By comparison, in the minced cartilage group, trypsin and collagenase were not used for enzymatic digestion, which yielded better cell proliferation and would be helpful to cover a large cartilage defect. From our preliminary data, 100 mg of cartilage fragment, harvested from patients who were undergoing total knee arthroplasty, contained about 2 × 105 chondrocytes. The composite of isolated chondrocytes group contained 2 × 105 chondrocytes in 100 μL atelocollagen gel in a manner of previous study. 27 Therefore, the isolated chondrocytes composite contained cells isolated from 100 mg cartilage fragment in 100 μL gel. The best cell proliferation ability and Bern score were identified in the group containing 25 mg of minced cartilage in 100 μL of atelocollagen gel, which means that the amount of cartilage in the minced cartilage procedure is only one-fourth that of isolated chondrocytes. Hence, minced cartilage embedded in atelocollagen gel would have a potential to cover a cartilage defect 4 times larger than conventional ACI and need less cartilage which can repair the same size lesion with less donor site morbidity. In addition, minced cartilage obtained from normal cartilage tissue and embedded in atelocollagen gel, which does not require enzymatic digestion to isolate chondrocytes, has the advantage of preserving the cartilage matrix at the time of implantation. In fact, minced cartilage composites in our study had more abundant GAG content compared with isolated chondrocyte composites. Moreover, a recent study showed that type II collagen, which is the specific collagen type in articular cartilage, has an extremely limited turnover in an individual’s lifetime. 31 We demonstrated that it was favorable to use cartilage fragments that contain cartilage matrix, collagen, and chondrocytes for cartilage repair without digestion.
Tissue-engineered cartilage has been implanted into cartilage defects with good clinical results. However, the effect of the implanted tissue-engineered cartilage on the subchondral bone and surrounding cartilage has not been clearly evaluated. To address this issue, we evaluated the anabolic factor of atelocollagen gel. Our results showed that minced cartilage had an abundance of LECT-1 expression, which encodes chondromodulin-1. Chondromodulin-1 plays an important role in inhibiting chondrocyte hypertrophy and vascular invasion.32,33 Moreover, atelocollagen gel containing minced cartilage also had an abundance of TGF-β, compared with the chondrocytes group, indicative of the advantage of minced cartilage, embedded in the atelocollagen gel.
There were several limitations in this study that need to be acknowledged. First, only cartilage from OA patients undergoing total knee arthroplasty was used. It has been reported that the structural and mechanical properties, and even molecular aspects, of cartilage are changed by aging,34-37 with human cartilage fragments showing a slower migration rate of chondrocytes compared to juvenile cartilage.
38
Our procedure using atelocollagen gel and cartilage fragment might be advantageous for repairing larger cartilage defect, such as OA, using a smaller amount of cartilage at the donor site than required for ACI. It would be important to evaluate cell proliferation, migration, and extracellular matrix production in the atelocollagen gel using osteoarthritic cartilage. Second, in Japan, most of cases who are performed total knee arthroplasty are female. Muraki
In conclusion, minced cartilage embedded in atelocollagen gel exhibited good cell migration and proliferation, with abundant GAG, compared with the isolated chondrocytes, even if harvested from OA cartilage. These findings support that there is the possibility to repair cartilage defects in a one-step procedure using implantation of minced cartilage embedded in atelocollagen gel.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by MEXT KAKENHI Grant-in-Aid for Scientific Research (B), Grant No. 17H04314 (N.A.).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Our study was approved by the local ethical committee of our university.
Informed Consent
Written informed consent was obtained from all subjects before the study.
Trial Registration
Not applicable.
