Abstract
Objective
Pro-inflammatory mediators such as prostaglandin E-2 (PGE2) play major roles in the pathogenesis of osteoarthritis (OA). Although current pharmacologic treatments reduce inflammation, their prolonged use is associated with deleterious side effects prompting the search for safer and effective alternative strategies. The present study evaluated whether chondrocyte production of PGE2 can be suppressed by the combination of avocado/soybean unsaponifiables (ASU) and α-lipoic acid (LA).
Design
Chondrocytes from articular cartilage of equine joints were incubated for 24 hours with: (1) control media, (2) ASU, (3) LA, or (4) ASU + LA combination. Cells were activated with lipopolysaccharide (LPS), interleukin 1β (IL-1β) or hydrogen peroxide (H2O2) for 24 hours and supernatants were immunoassayed for PGE2. Nuclear factor-kappa B (NF-κB) analyses were performed by immunocytochemistry and Western blot following 1 hour of activation with IL-1β.
Results
LPS, IL-1β, or H2O2 significantly increased PGE2 production. ASU or LA alone suppressed PGE2 production in LPS and IL-1β activated cells. Only LA alone at 2.5 µg/mL was inhibitory in H2O2-activated chondrocytes. ASU + LA inhibited more than either agent alone in all activated cells. ASU + LA also inhibited the IL-1β induced nuclear translocation of NF-κB.
Conclusions
The present study provides evidence that chondrocyte PGE2 production can be inhibited by the combination of ASU + LA more effectively than either ASU or LA alone. Inhibition of PGE2 production is associated with the suppression of NF-κB translocation. The potent inhibitory effect of ASU + LA on PGE2 production could offer a potential advantage for a combination anti-inflammatory/antioxidant approach in the management of OA.
Introduction
Osteoarthritis (OA) is a chronic, painful disorder affecting movable joints, characterized by cartilage degradation, bone remodeling, osteophyte formation, and joint inflammation. 1 Both humans and animals, particularly dogs and horses suffer from OA with debilitating morbidity. 2 Among the risk factors for developing OA are advancing age, joint trauma, and obesity.3-5 Mechanical stress factors also play an important role in the pathogenesis of the disease. 6 Studies at the cellular and molecular level have established the central role of inflammation in the pathogenesis of OA.7-12 In vivo and in vitro studies have provided strong evidence for the critical involvement of pro-inflammatory molecules including prostaglandins, cytokines, and chemokines. Among the earliest pro-inflammatory mediators identified in osteoarthritic joints and regarded as key participants in OA progression are prostaglandin E-2 (PGE2), interleukins (IL-1β, IL-6), and tumor necrosis factor α (TNF-α).9-12 Produced by chondrocytes and other tissues in the joint, these pro-inflammatory molecules are thought to account for the observed pain, cartilage breakdown, bone remodeling, and osteophyte formation in OA patients.13-18
Recognition of the prominent role of inflammatory mediators in OA has led to the development of anti-inflammatory strategies particularly nonsteroidal anti-inflammatory drugs (NSAIDs). Although NSAIDs do not modify the disease, these agents help mitigate the pain and alleviate symptoms in OA patients. However, severe adverse side effects, including cardiovascular and gastrointestinal complications as well as liver and renal toxicity have been reported with prolonged drug administration.19,20 Certain cyclooxygenase inhibitors that effectively block PGE2 synthesis were withdrawn due to high risks of life-threatening cardiac pathology. NSAIDs that profoundly deplete PGE2 levels also inhibit bone healing and fracture repair.17-20 Concerns about the side effects of drugs have prompted the search for safer and effective alternative nonpharmacologic agents to alleviate the symptoms of OA. Among the nonpharmacologic agents that have been studied for the management of OA in humans and animals are glucosamine (GLU), chondroitin sulfate (CS), and avocado/soybean unsaponifiables (ASU) alone or in combination. ASU, GLU, and CS have been documented to reduce inflammation in vitro and in vivo.21-38ASU, GLU, and CS have also been reported to be beneficial in the management of OA in man and animals with minimal adverse side effects.31-38 Of the 3 compounds, ASU has more recently been studied for its anti-inflammatory, anti-catabolic, and anabolic effects on cartilage metabolism.21-24,31 It inhibits the expression and production of cytokines, chemokines, PGE2, nitric oxide, and matrix metalloproteinases (MMPs) as well as enhances the synthesis of cartilage matrix components such as collagen and proteoglycans.21-24,31 The molecular mechanisms behind its beneficial range of activities have been theorized to involve the inhibition of nuclear factor kappa B (NF-κB) activation. 23 NF-κB is a key transcription factor that when activated translocates from the cytoplasm to the nucleus which induces the expression of pro-inflammatory genes, including enzymes that degrade cartilage matrix. 23 These findings have encouraged further exploration of other nonpharmacologic agents to attenuate inflammation.
More recently, linkage between inflammation and oxidative stress has been reported.39-41 For example, stimuli that induce oxidative stress are able to activate production of pro-inflammatory molecules, including PGE2. Products of oxidative stress also induce degradative enzymes and processes that could break down cartilage. The cross-talk between inflammation and oxidative stress has opened potential new targets for OA therapy.39-41 One such candidate agent with a long record of safe use (>50 years) in man is α-lipoic acid (LA).42-48 LA is a potent, naturally occurring, low-molecular-weight antioxidant synthesized enzymatically in mitochondria from octanoic acid.42-48 It is a critical cofactor of mitochondrial decarboxylation reactions and is essential for adequate ATP production. The biological activities of LA include scavenging of reactive oxygen species, regeneration of endogenous antioxidants such as glutathione and vitamin E, metal ion chelating, and ability to repair oxidative damage in macromolecules.42-48 In addition to its unique antioxidant properties, LA has repeatedly been shown to have inhibitory effects on inflammatory markers in humans and animals.42-48 Oral supplementation of LA has been extensively studied for the management of neurological cognitive disorder, cardiovascular diseases, chronic renal disease, obesity, pain, inflammatory diseases, and aging.42-48 Reviews of these studies point to beneficial clinical effects that could be attributed to its antioxidant activity.42,43 LA has also been combined with other agents such as eicosapentaenoic acid to determine whether enhanced beneficial anti-inflammatory effects would be achieved. 45 The aim of the present study is to determine whether LA in combination with anti-inflammatory, chondroprotective ASU would provide potentiated effects. Using the equine chondrocyte culture model, we observed that the ASU + LA combination suppresses PGE2 more effectively than either ASU alone or LA alone. This inhibition is paralleled by disruption of NF-κB translocation, a key regulatory controlling event in PGE2 synthesis. Identifying agents that are safe and effective in suppressing PGE2 synthesis in the osteoarthritic joint may offer a more beneficial, multimodal therapeutic approach.
Methods
Isolation of Equine Chondrocytes
Articular cartilage was harvested from the radiocarpal and tibiotarsal joints from apparently healthy horses of unreported age, sex, or breed. Equine joint tissues used in this study were obtained from a commercial source (Celsis In Vitro Technologies, Baltimore, MD) and Dr. Rashmir–Raven, an equine veterinarian (coauthor). Animals were euthanized for reasons unrelated to this study in compliance with Michigan State University’s Institutional Animal Care and Use Committee. All harvested articular cartilage appeared smooth, glassy, and without macroscopic abnormalities. Cartilage was aseptically harvested, cut into 1 to 5 mm2 pieces, and digested with type II collagenase medium (10,000 U/L, Gibco, Invitrogen, Carlsbad, CA, USA) at 37°C, 5% CO2 for 12 to 18 hours. The cellular suspension was strained through a 0.4-µm filter (BD Biosciences, Franklin Lakes, NJ, USA) into 50-mL polystyrene conical tubes (BD Biosciences) and centrifuged at 1000 rpm for 10 minutes. The resulting cell pellet was washed 4 times with Hank’s Balanced Salt Solution (HBSS, American Type Culture Collection, Manassas, VA, USA).
Monolayer Culture Technique and Experimental Design
Cells were propagated in monolayer culture as previously described and were used at passages 2 to 4. 28 Chondrocytes were seeded in 6-well plates (5 × 106 cells/well) overnight at 37°C, 5% CO2. Before stimulation, chondrocytes were pre-incubated with: (1) control media, (2) ASU (NMX1000®, 8.3 µg/mL, Nutramax Laboratories, Inc., Edgewood, MD, USA), (3) LA (Sigma-Aldrich, St. Louis, MO, USA, concentrations ranging from 1 to 100 µg/mL), or (4) the combination of ASU (8.3 µg/mL) and LA (concentrations ranging from 0.625 to 2.5 µg/mL) for 24 hours. The concentration of ASU (8.3 µg/mL) used in this study was previously reported to have significant anti-inflammatory effects and is clinically relevant.21,22 Chondrocytes were then stimulated/activated for 24 hours with lipopolysaccharide (LPS; 1 ng/mL, Sigma-Aldrich), IL-1β (5 ng/mL, R&D Systems, Minneapolis, MN, USA), or hydrogen peroxide (H2O2, 500 µM, Sigma-Aldrich). Supernatant were collected and stored at -80°C. Viability check previously described as determined by Trypan blue dye uptake was 100%. 22
Phenotype Analysis: Collagen and Aggrecan Immunofluorescence
Chondrocytes were seeded at 1 × 104 cells/well overnight on 8-well chambered slides (Fisher Scientific, Waltham, MA, USA) at 37°C, 5% CO2. Cells were fixed with 10% v/v formalin (Thermo, Fisher Scientific) for 20 minutes and washed 4 times with 1× phosphate buffered saline (PBS; Gibco, Invitrogen). To prevent non-specific binding, a blocking solution (Gibco, Invitrogen) was applied and incubated for 30 minutes, followed by a 5-minute wash using PBS. Fixed cells were double stained for type I and type II collagen by incubation overnight with a cocktail containing a goat anti-type I antibody (Southern Biotechnology Associates, Birmingham, AL, USA) diluted 1:500 and a mouse monoclonal anti-type II (Calbiochem, La Jolla, CA, USA) diluted 1:50 in PBS containing 0.05% Triton X-100.
For aggrecan immunostaining, chondrocytes were incubated overnight at 4°C with mouse monoclonal anti-aggrecan (U.S. Biological, Swampscott, MA, USA) diluted 1:20 in PBS. Cells were washed 3 times with PBS then incubated with Alexa Fluor 488 labeled donkey anti-mouse IgG at room temperature with gentle agitation. Slides were washed 3 times with PBS and nuclei were stained with DAPI (Gibco Invitrogen) for 30 minutes at room temperature. Stained cells were analyzed using a Nikon epifluorescence TE 200 microscope and images were captured with a digital camera (Nikon Spot Camera, USA).
NF-κB Immunohistochemistry and Western Blot Analysis
Chondrocytes were seeded on 8-well chamber slides and were incubated at 37°C, 5% CO2 overnight. Prior to stimulation, cells were incubated with: (1) control media or (2) ASU (NMX1000, 8.3 µg/mL) and LA (2.5 µg/ml). Cells were next activated for 1 hour with IL-1β and were subsequently incubated with rabbit anti-NF-κB (Santa Cruz Biotechnology, Santa Cruz, CA, USA) then incubated overnight at 4°C. Cells were washed with PBS and incubated with Alexa Fluor-488 labeled donkey anti-rabbit IgG antibody (Invitrogen) for 2 hours. Cells were analyzed by Nikon epifluorescence TE 200 microscope. Images were captured by digital camera (Nikon Spot Camera, USA).
Nuclear translocation of NF-κB was analyzed by immunohistochemistry as previously described using rabbit anti-NF-κB specific to the p65 subunit of NF-κB (Santa Cruz Biotechnology, Santa Cruz, CA), which was earlier determined to cross-react with equine chondrocytes.28,29 System controls including normal serum as substitute for the primary antibody showed only background staining. Slides were viewed using a Nikon Eclipse TE200 inverted fluorescence microscope (Nikon Instruments, Melville, NY) equipped with a Nikon Spot Camera (Nikon Instruments).
Chondrocyte nuclear fractions for Western blot analysis were isolated according to protocol using the Active Motif Nuclear Extraction Kit (Active Motif, Carlsbad, CA, USA). Total protein (10 µg protein/lane) was separated by 4% to 15% Mini-PROTEAN TGX gradient gels (BioRad) and transferred to polyvinylidene difluoride membranes (BioRad). Membranes were incubated with rabbit anti-NF-κB antibody (1:3,000; Santa Cruz Biotechnology) and transferred proteins visualized with secondary horseradish peroxidase-conjugated goat anti-rabbit IgG antibody (1:50,000; Santa Cruz Biotechnology) followed by chemiluminescence detection using ECL Plus (GE Healthcare, Piscataway, NJ, USA). Data were quantitated using the NIH ImageJ program (http://rsb.info.nih.gov/ij/).
Measurement of PGE2 Production Using ELISA
PGE2 concentrations were determined in culture supernatant using enzyme-linked immunosorbent assay (ELISA) kits (Prostaglandin E2 Parameter Assay Kit, R&D Systems). All assays were run according to manufacturer’s instructions and a standard was run in parallel to samples during each assay. Optical density was measured immediately using the SpectraMAX 340 microplate reader (Molecular Devices, Sunnyvale, CA, USA) at 450 nm with wavelength correction at 540 nm.
Statistical Analysis
Pairwise multiple comparisons were performed using 1-way analysis of variance, Tukey post hoc using SigmaStat statistical software (Windows Version 3.11), where P < 0.05 was considered statistically significant.
Results
Chondrocytes in Monolayer Culture
Articular cartilage obtained from 3 adult horses exhibited smooth, glassy appearance devoid of lesions. Retrieved chondrocytes propagated for 2 to 4 passages were 100% viable with doubling times of 3 to 5 days. Approximately 80% to 90% of chondrocytes immunostained for type II collagen and about 10% for type I collagen or both types across passages (
Fig. 1
,
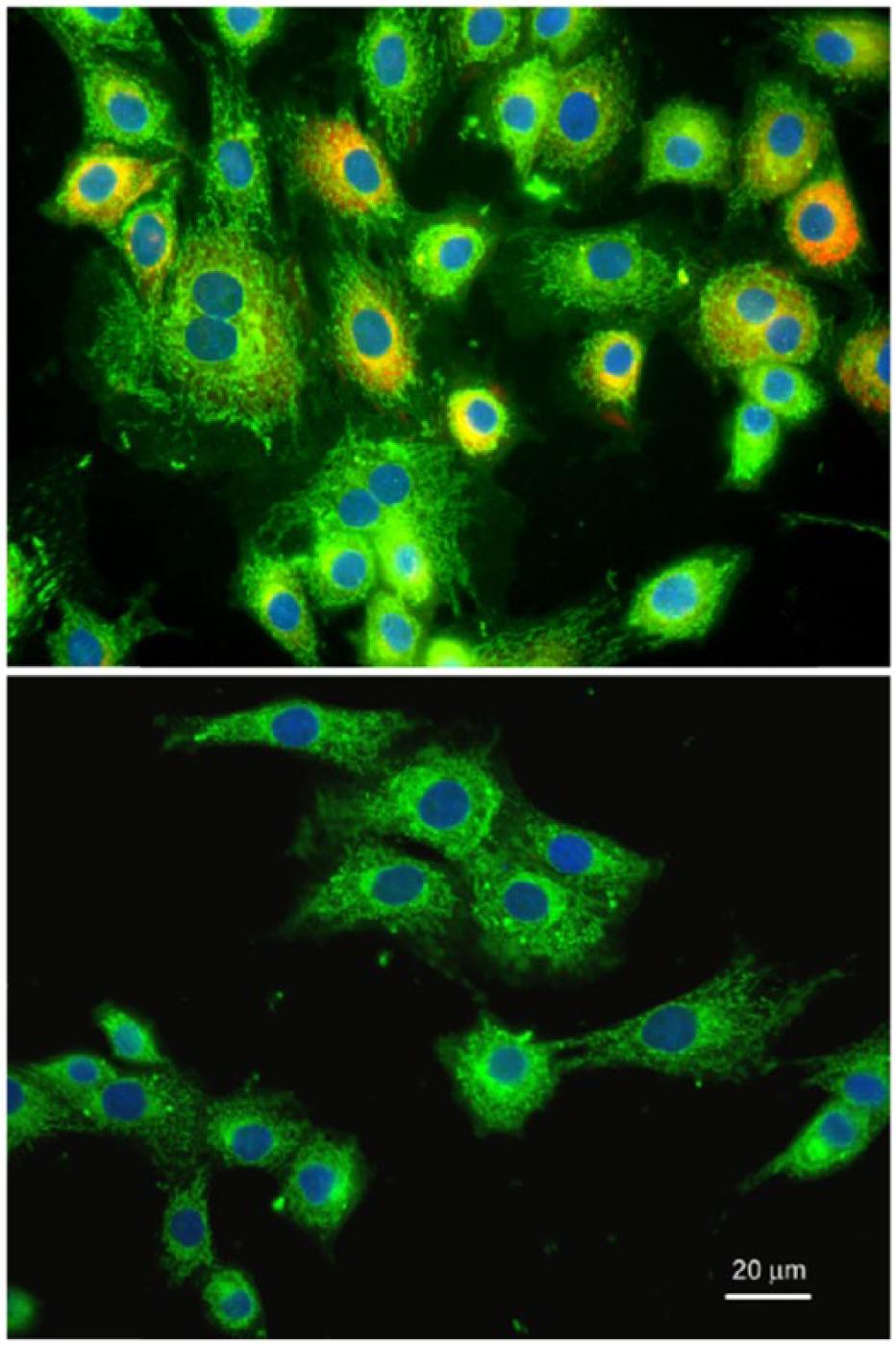
Representative immunofluorescent staining of collagen and aggrecan in equine chondrocytes at passage 3 monolayer culture. Equine chondrocytes stained green for type II collagen and red for type I collagen (
LA Dose Response Inhibition of LPS and IL-1β Induced PGE2 Production in Chondrocyte Cultures
To determine the concentrations of LA for combination studies with ASU, a dose titration was performed. The inhibition of LPS or IL-1β induced PGE2 production showed a dose dependency ( Fig. 2A and B ). With both pro-inflammatory activators, PGE2 production was suppressed to about 25% of activated controls with as low as 2.5 µg/mL of LA. LPS and IL-1β are commonly used to induce inflammation in vitro and in vivo. For the subsequent combination studies with ASU, LA concentrations of 2.5 µg/mL and below were used to ensure detection of maximal combined inhibitory effects. From a similar ASU dose titration of LPS-induced PGE2 production in our previous study, the lowest inhibitory concentration of 8.3 µg/mL was selected. 22
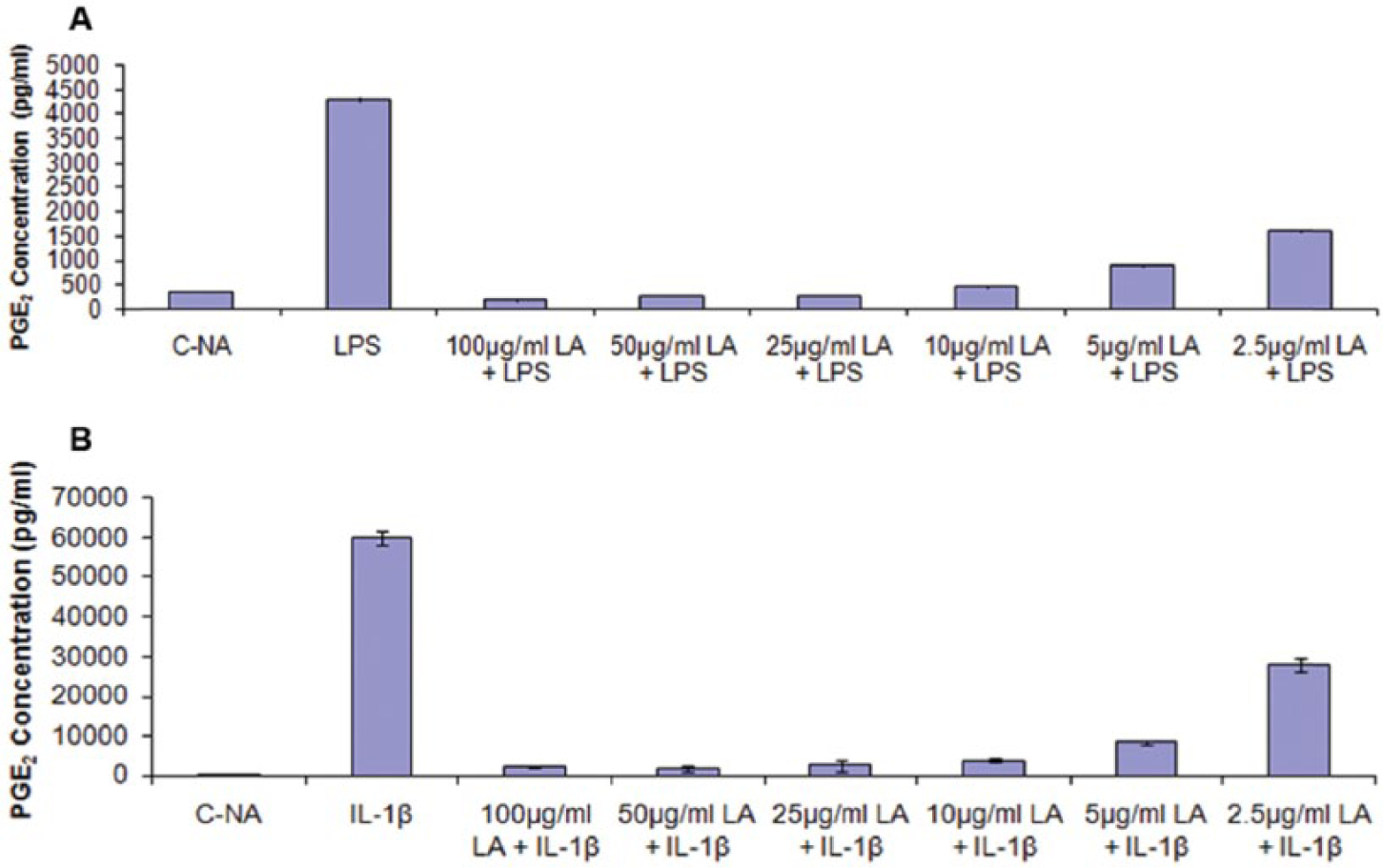
Effect of various concentrations of α-lipoic acid (LA) on lipopolysaccharide (LPS) (
LA Enhances the ASU Inhibition of LPS, IL-1β, and H2O2-Induced PGE2 Production
Chondrocytes activated with LPS significantly increased PGE2 production ( Fig. 3A-C , P < 0.001) and pretreatment with ASU, LA, or ASU + LA significantly inhibited this increase ( Fig. 3A-C ). The combination of ASU with the 3 concentrations of LA: 0.625 µg/mL ( Fig. 3A ), 1.25 µg/mL ( Fig. 3B ), and 2.5 µg/mL ( Fig. 3C ) reduced PGE2 production significantly (P < 0.001) more than either agent alone. The combination product decreased PGE2 production by about 75% compared with about 30% inhibition by either ASU or LA alone.
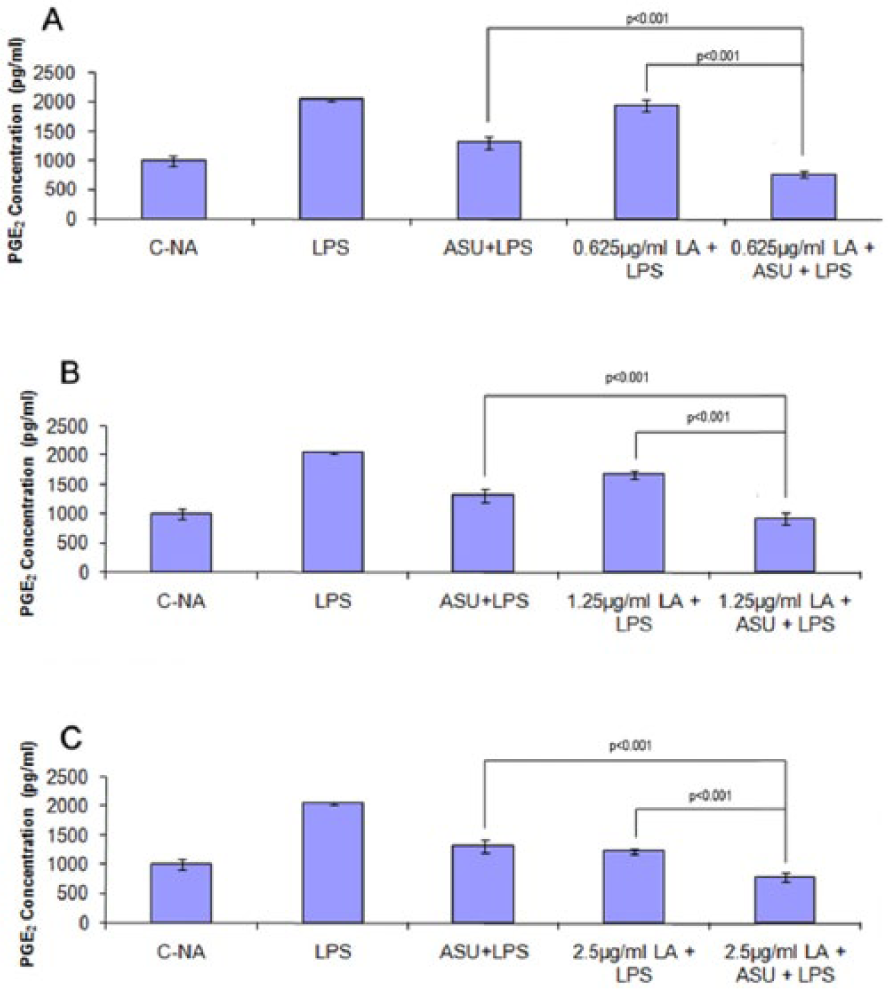
Effect of avocado/soybean unsaponifiables (ASU), α-lipoic acid (LA), or ASU + LA on lipopolysaccharide (LPS)-induced prostaglandin E-2 (PGE2) production. PGE2 levels were measured in cell culture supernatant following incubation with LPS for 24 hours. PGE2 levels significantly (P < 0.001) increased following LPS stimulation compared with control nonactivated cells (C-NA,
To further examine the potentiated anti-inflammatory effect of ASU + LA, chondrocytes were activated with IL-1β ( Fig. 4A-C ). PGE2 production by IL-1β stimulated cells significantly increased (P < 0.001) and was significantly suppressed (P < 0.001) by ASU or LA at 0.625 µg/mL ( Fig. 4A ), 1.25 µg/mL ( Fig. 4B ), or 2.5 µg/mL ( Fig. 4C ). Similar to the observation using LPS activated chondrocytes, the reduction in IL-1β induced PGE2 production was potentiated by the combination ASU + LA. The inhibitory effect was greater than that of ASU alone (P < 0.001) or LA alone (P < 0.001) ( Fig. 4A-C ).
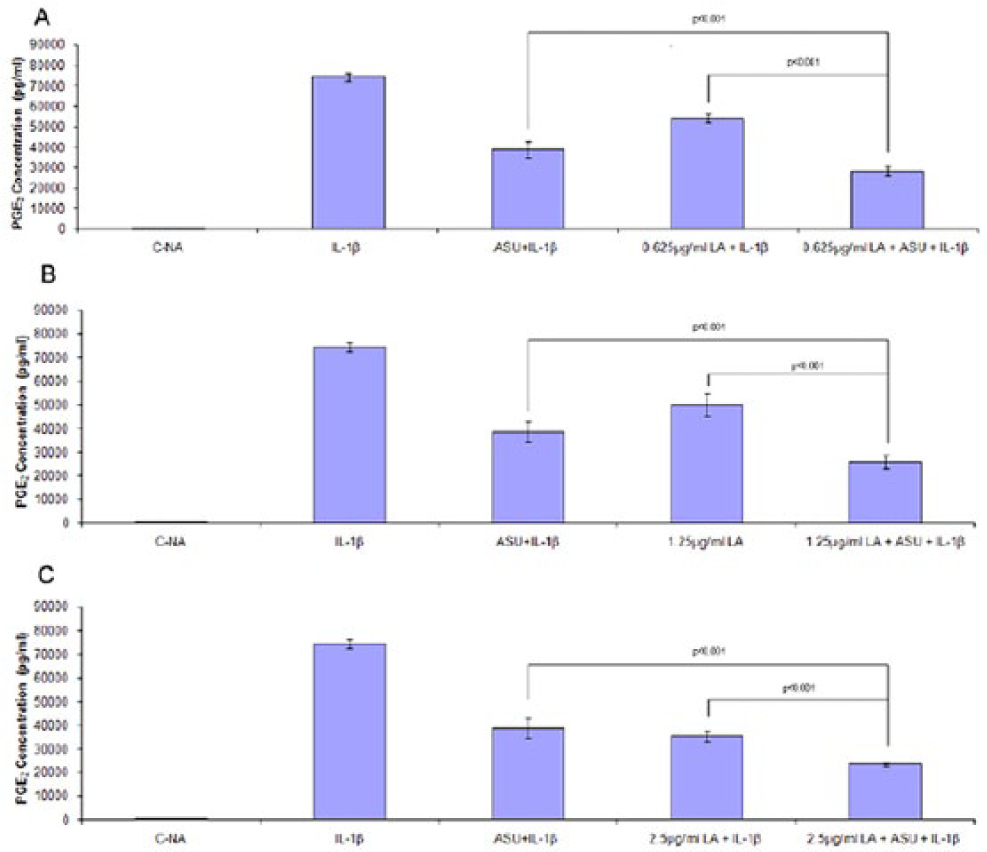
Effect of avocado/soybean unsaponifiables (ASU), α-lipoic acid (LA), or ASU + LA on interleukin-1β (IL-1β)-induced prostaglandin E-2 (PGE2) production. PGE2 levels were measured in cell culture supernatant following incubation with IL-1β for 24 hours. PGE2 levels significantly (P < 0.001) increased following IL-1β stimulation compared with control nonactivated cells (C-NA,
H2O2 has been reported to induce pro-inflammatory gene expression, including COX-2, the principal regulator of PGE2 synthesis. 49 Chondrocytes responded to H2O2 with significant increase in PGE2 synthesis (P = 0.003, Fig. 5A and B ). Pretreatment with ASU alone did not reduce PGE2 synthesis whereas LA alone inhibited significantly at 2.5 µg/mL (P < 0.001) but not at 1.25 µg/mL (P > 0.05). In contrast, the combination of ASU + LA (2.5 and 1.25 µg/mL) significantly inhibited PGE2 production greater than ASU alone or LA alone (2.5 and 1.25 µg/mL, Fig. 5A and B ). LA at 2.5 µg/mL inhibited by 44% while the combination product decreased PGE2 production by 76% ( Fig. 5B ).
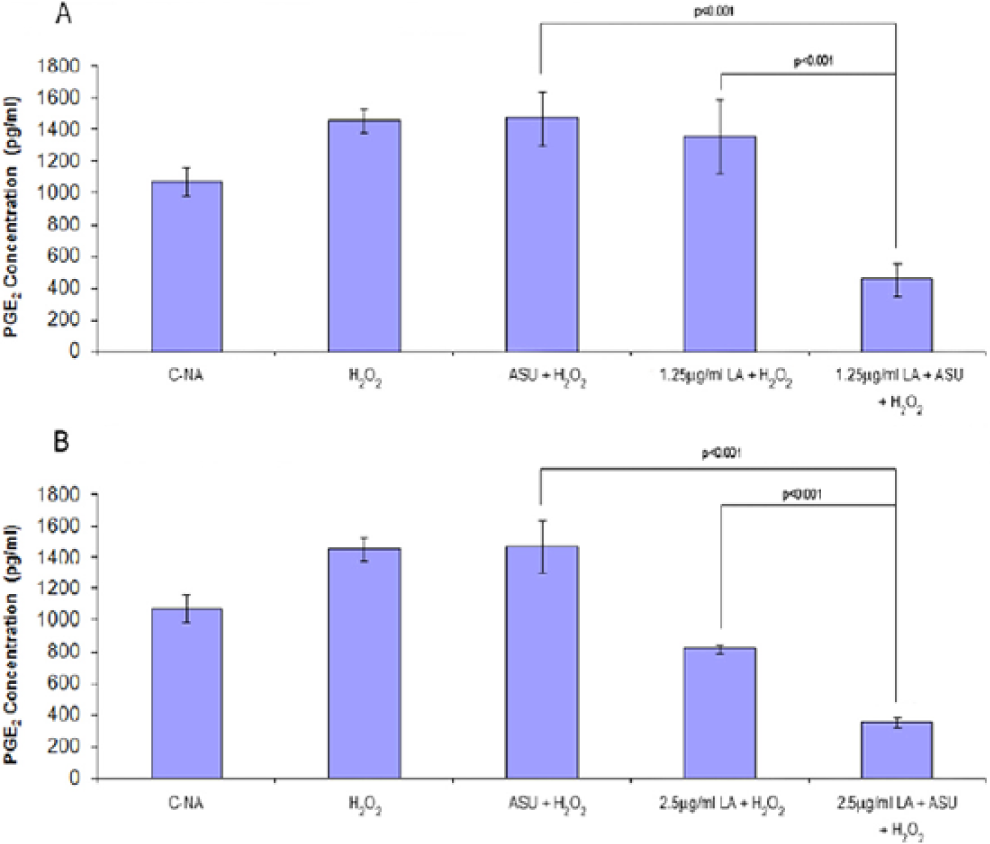
Effect of avocado/soybean unsaponifiables (ASU), α-lipoic acid (LA), or ASU + LA on H2O2-induced PGE2 production. PGE2 levels were measured in cell culture supernatant following incubation with H2O2 for 24 hours. PGE2 levels significantly (P = 0.003) increased following H2O2 stimulation compared with control nonactivated cells (C-NA,
ASU + LA Inhibits NF-κB Nuclear Translocation
Chondrocytes seeded on 8-well chamber slides were pre-treated with ASU + LA and then stimulated with IL-1β. Control cells showed only cytoplasmic fluorescent immunostaining. IL-1β stimulated cells showed intense fluorescent immunostaining in the nucleus indicative of NF-κB translocation. Pretreatment with the ASU + LA combination profoundly diminished nuclear fluorescence ( Fig. 6 ). Western blot analysis of nuclear fractions confirmed the reduced translocation/activation of NF-κB ( Fig. 7A and B ).
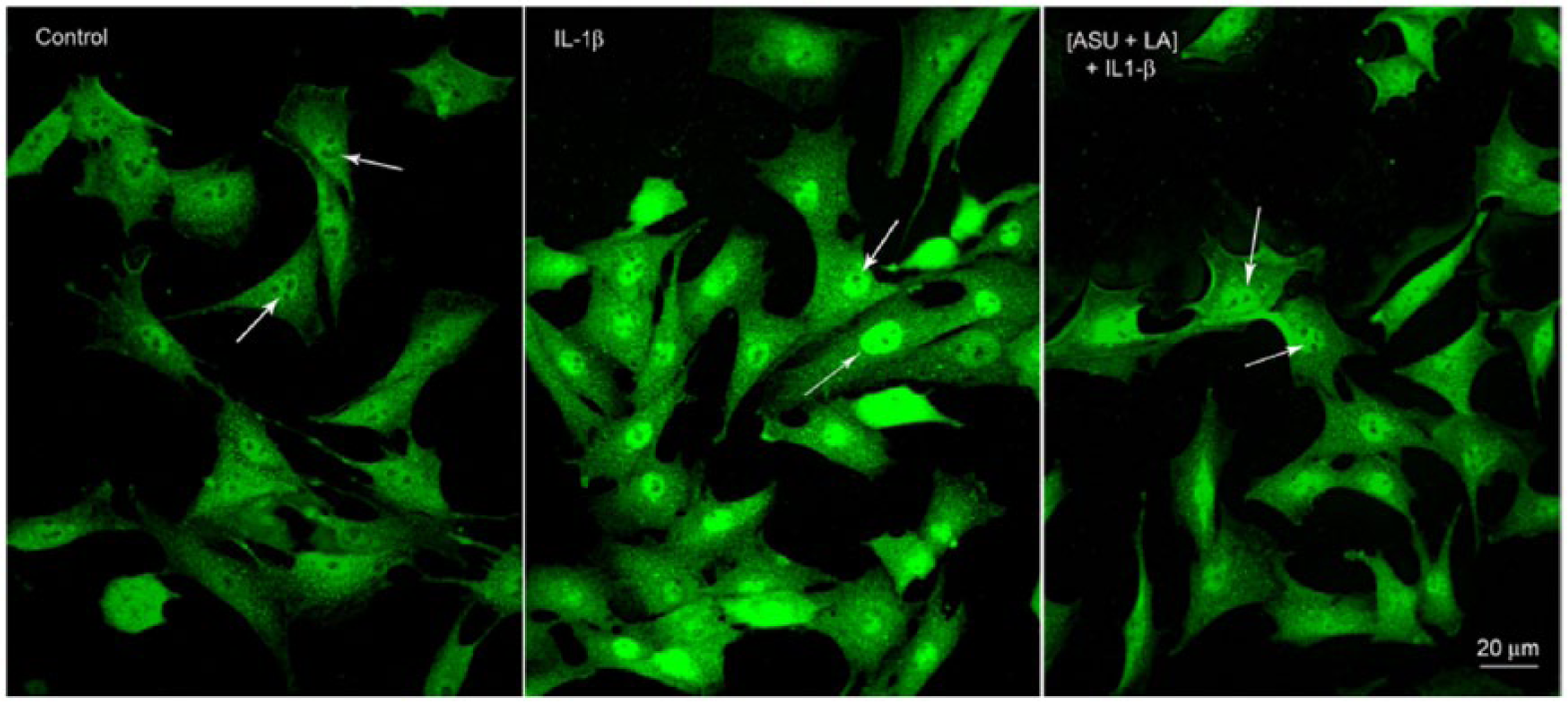
Representative nuclear factor-kappa B (NF-κB) immunofluorescence in control, interleukin-1β (IL-1β)-stimulated, and avocado/soybean unsaponifiables + α-lipoic acid (ASU + LA)–treated chondrocytes. Control cells show cytoplasmic and no nuclear fluorescence with NF-κB (
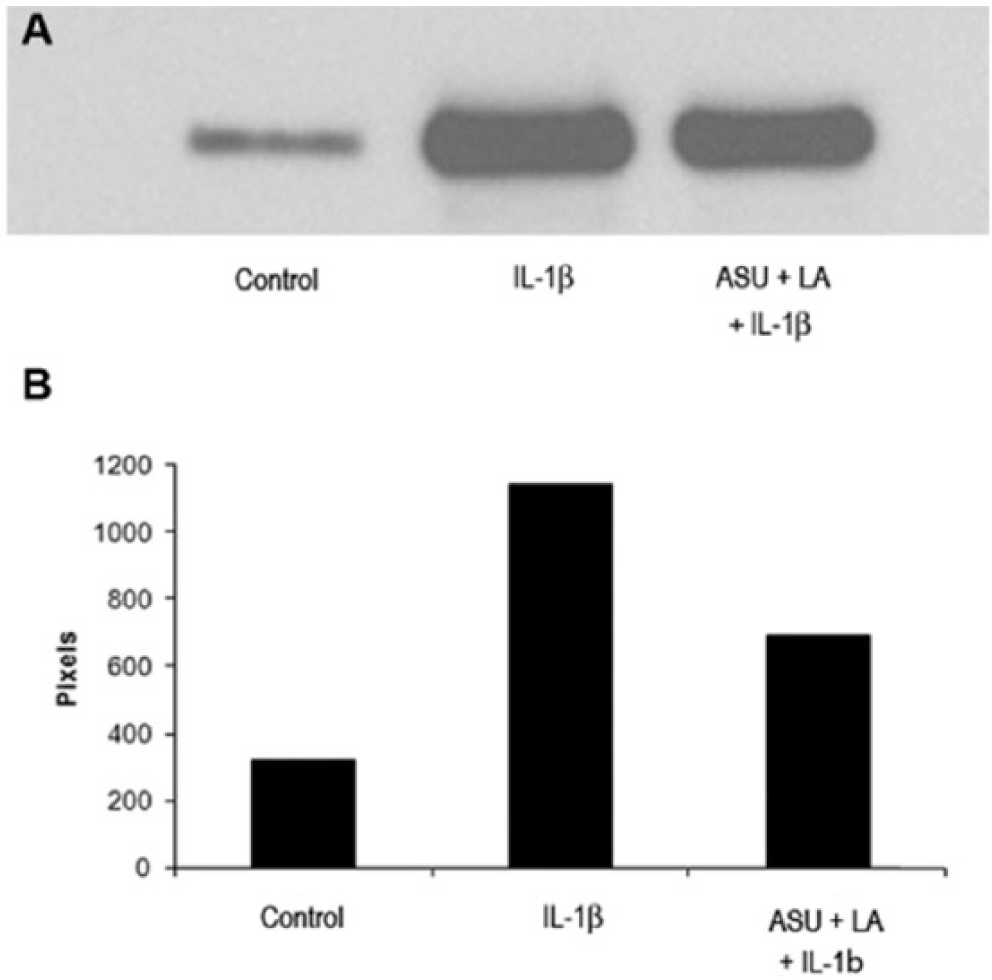
(
Discussion
The well-known adverse side effects of NSAIDs currently used in the treatment of OA have led to efforts in identifying therapeutics with potent anti-inflammatory activity but with fewer side effects. In this study, we evaluated the effect of a combination of ASU, a preparation with well-documented anti-inflammatory activities, and LA, an agent with well-characterized antioxidant activity. We used chondrocytes from equine articular cartilage since they share many cellular and molecular characteristics with human chondrocytes.50,51 Articular joints from horses and humans also have similar joint volume, structure, and biomechanical function.50,51 With these similarities, horses are commonly used as in vivo model for human OA, while their cartilage and constituent chondrocytes are used for in vitro studies. Both equine and human chondrocytes in monolayer culture similarly undergo phenotypic alterations not unlike those observed in osteoarthritic cartilage. Prominent among the phenotypic changes of chondrocytes seen in OA is the gradual shift from producing the articular cartilage phenotype marker type II collagen to the fibroblastic-osteoblastic type I collagen in osteoarthritic cartilage.52,53 Chondrocytes at passages 2 to 4 used in the present study consisted mainly of cells producing type II with a few cells producing either collagen type I alone or both ( Fig. 1 ).
PGE2 was used in this study as a well-defined marker of inflammation and as a key participant in the pathogenesis of OA.7-15 It stimulates the production of degradative enzymes and inhibits the synthesis of cartilage components particularly proteoglycans. It perpetuates the inflammatory response and damage to cartilage by inducing production of other pro-inflammatory mediators.7-15 PGE2 is also known to sensitize nociceptors resulting in chronic pain.13,14 Our observation that H2O2, a known inducer of oxidative stress, stimulated PGE2 synthesis in chondrocytes reinforces the notion that common pathways linking inflammation and oxidative stress are involved ( Fig. 5 ). Recent studies have shown that activators of oxidative stress such as H2O2 induce the expression of pro-inflammatory genes, including COX-2, a key regulatory enzyme for PGE2 synthesis.39-41,54
Our findings confirm previous reports that LA inhibits LPS, IL-1β, and H2O2 (2.5 µg/mL) induced PGE2 production ( Figs. 3 - 5 ). There was robust inhibitory activity of ASU or LA alone with LPS and IL-1β stimulation but not with H2O2. In contrast, the ASU + LA combination was potently inhibitory regardless of the stimuli used ( Figs. 3 - 5 ). That LA significantly enhanced the anti-inflammatory effect of ASU by more than an additive reduction in PGE2 production suggests synergy. 55 The observed differences in inhibitory activity could be due to their actions on separate, distinct molecular targets ( Figs. 3 - 5 ). This supports the rationale that combining the 2 agents potentiates and complements their shared as well as different biologic activities. Well known as an antioxidant, LA has a long history of safe use in the management of several disorders associated with oxidative stress, including dementia, cardiovascular disease, and renal disease.42,43 It is reported as physiologic antioxidant in the form of a conjugate base with high negative reduction potential and can induce expression of antioxidant enzymes.42,43 In comparison, ASU has been extensively evaluated in vitro, in animals and in humans for its anti-inflammatory and chondroprotective effects.21-24,31 ASU has also been shown to enhance other agents such as CS, EGCG (epigallocatechin gallate), and GLU in reducing inflammation in vitro.27-31 Studies in humans and animals demonstrated the anti-inflammatory, anti-catabolic, and anabolic activities of ASU, thus helping maintain the structural integrity of articular cartilage in osteoarthritic joints.21-24,31 Like LA, ASU has an excellent record of safety in humans and animals.31,42-48
The suppression of PGE2 production by the combination of ASU and LA is paralleled by the inhibition of NF-κB translocation and activation (
Several mechanisms have been speculated as to how NF-κB could serve as a link between inflammation and oxidative stress. Activators of oxidative stress such as H2O2 could affect pathways that stimulate the IKK complex thus facilitating phosphorylation and activation of NF-κB which turns on the production of pro-inflammatory molecules and other catabolic events affecting cartilage. 42 It has also been speculated that NF-κB inhibits the activation of Nrf2 (nuclear erythroid 2-related factor), which controls induction of anti-inflammatory and antioxidant enzyme gene expression. The ASU and LA combination could thus affect linked pathways of inflammation and oxidative stress. Considering that ASU and LA produce their effects via independent mechanisms, their use in combination has a scientific rationale as an alternative or adjunct to conventional pharmacologic treatments for the management of OA. Taken together, dosing of NSAIDs could be reduced while achieving the same therapeutic effects and minimizing deleterious complications.
Footnotes
Ethical Approval
The present study does not need institutional review board approval.
Animal Welfare
Animals were euthanized for reasons unrelated to this study in compliance with Michigan State University’s Institutional Animal Care and Use Committee.
Acknowledgments and Funding
We would like to thank Dr. Reinhard Grzanna for designing the immunohistochemistry and Western blot experiments as well as for constructive review of the manuscript. The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Carmelita G. Frondoza, Lowella Fortuno, Mark Grzanna, and Angela Y. Au are former employees of Nutramax Laboratories, Inc. but do not hold stocks or royalties. Stacy Ownby is a current employee of Nutramax Laboratories, Inc. but does not hold stocks or royalties.
