Abstract
Objective
Previously, we demonstrated the importance of transforming growth factor-β (TGFβ)-activated SMAD2/3 signaling in chondrogenesis of bone marrow–derived mesenchymal stem cells (BMSCs). However, TGFβ also signals via the SMAD1/5/9 pathway, which is known to induce terminal differentiation of BMSCs. In this study, we investigated whether other SMAD2/3-activating ligands, Activin and Nodal, can induce chondrogenic differentiation of BMSCs without inducing terminal differentiation.
Design
Activation of SMAD2/3 signaling and chondrogenesis were evaluated in human BMSCs (N = 3 donors) stimulated with TGFβ, Activin, or Nodal. SMAD2/3 activation was assessed by determining phosphorylated-SMAD2 (pSMAD2) protein levels and SMAD2/3-target gene expression of SERPINE1. Chondrogenesis was determined by ACAN and COL2A1 transcript analysis and histological examination of proteoglycans and collagen type II.
Results
Both Activin and TGFβ enhanced pSMAD2 and SERPINE1 expression compared to the control condition without growth factors, demonstrating activated SMAD2/3 signaling. pSMAD2 and SERPINE1 had a higher level of expression following stimulation with TGFβ than with Activin, while Nodal did not activate SMAD2/3 signaling. Of the 3 ligands tested, only TGFβ induced chondrogenic differentiation as shown by strongly increased transcript levels of ACAN and COL2A1 and positive histological staining of proteoglycans and collagen type II.
Conclusions
Even with concentrations up to 25 times higher than that of TGFβ, Activin and Nodal do not induce chondrogenic differentiation of BMSCs; thus, neither of the 2 ligands is an interesting alternative candidate for TGFβ to induce chondrogenesis without terminal differentiation. To obtain stable cartilage formation by BMSCs, future studies should decipher how TGFβ-induced terminal differentiation can be prevented.
Introduction
Bone marrow–derived mesenchymal stem cells (BMSCs) are promising for treatment of cartilage damage, because they can differentiate into chondrocytes. Unfortunately, BMSC-derived chondrocytes do not resemble the stable phenotype of articular cartilage–derived chondrocytes, because they undergo terminal chondrocyte differentiation.1,2 As this will result in mineralization of implanted tissue, 3 better insight into the molecular mechanisms that regulate BMSC differentiation is required.
Transforming growth factor-β (TGFβ) is commonly used to induce chondrogenic differentiation of BMSCs. 4 TGFβ signals via complexes of type II and I serine/threonine kinase receptors: TGFBR2 and TGFBR1 (or Activin receptor-like kinase 5 [ALK5]). Following complex formation, SMAD2/3 signaling is activated, which regulates transcription of downstream target genes. 5 We previously demonstrated the necessity of SMAD2/3 signaling in chondrogenesis, because inhibition of TGFβ-activated SMAD2/3 signaling with chemical inhibitors blocked chondrogenic differentiation of BMSCs. 2 Besides the canonical SMAD2/3 pathway, TGFβ signals via the SMAD1/5/9 pathway.2,6 Interestingly, blocking SMAD1/5/9 signaling in chondrogenically differentiated BMSCs inhibited terminal differentiation and mineralization without affecting cartilage matrix production, whereas blocking SMAD2/3 signaling in BMSC-derived chondrocytes inhibited further matrix deposition. 2 These findings suggested that preventing TGFβ-activated SMAD1/5/9 signaling in BMSC-derived chondrocytes could prevent terminal differentiation. Therefore, we questioned whether other ligands known to activate SMAD2/3, but not SMAD1/5/9 signaling, could induce chondrogenesis without subsequently inducing terminal differentiation.
Like TGFβ, Activin and Nodal activate SMAD2/3 signaling, but via different receptor complexes: Activin via ACVR2A and ACVR1B (ALK4), 7 and Nodal via ACVR2B and ACVR1C (ALK7). 8 Activin and Nodal were reported to be incapable of activating SMAD1/5/9 signaling in multiple cell types8,9; however, their capacity to induce chondrogenesis has not been clearly established. Activin was shown not only to enhance chondrogenesis but also to inhibit cartilage matrix production in limb bud cells.10,11 In addition, Activin suppressed chondrocyte differentiation of murine chondroprogenitor cells. 12 So far, no study has reported whether Nodal induces chondrogenesis. Our aim was to determine whether Activin and Nodal could activate SMAD2/3 signaling and induce chondrogenic differentiation of BMSCs without inducing terminal differentiation, like TGFβ.
Materials and Methods
Human BMSC Isolation and Expansion
From 3 patients (age 10-12 years), pieces of iliac crest bone and marrow that were left over after cleft palate reconstruction surgery (approved by the Erasmus MC Medical Ethical Committee; MEC-2014-106) were added to α-MEM containing 1.5 µg/mL fungizone, 50 µg/mL gentamicine, 10% fetal calf serum (all from Gibco, Waltham, MA), 25 µg/mL ascorbic acid-2-phosphate (Sigma-Aldrich, St. Louis, MO), and 1 ng/mL FGF-2 (AbD Serotec, Kidlington, UK) for isolation of BMSCs by plastic adherence. When cells neared confluence, they were detached with 0.05% trypsin and re-seeded at 2,300 cells/cm2. After 4 passages, cells were characterized by flow cytometry, confirming >97% positivity for BMSC markers CD73, CD90, CD105, and CD166, and <0.5% for hematopoietic cell marker CD45 (data not shown). BMSCs expanded for 4 to 5 passages were used for experiments.
SMAD2/3 and SMAD1/5/9 Activation Following TGFβ, Activin, or Nodal Stimulation
Monolayer-cultured BMSCs were serum-starved overnight and maintained in serum-free medium (unstimulated control) or stimulated for 1 or 24 hour(s) with recombinant human TGFβ1 (10 ng/mL = 0.78 nM), Activin A (10, 100, or 250 ng/mL = 0.77, 7.70, or 19.3 nM), or Nodal (10, 100, or 250 ng/mL = 0.78, 7.81, or 19.5 nM). All ligands were purchased from R&D Systems (Minneapolis, MN). Activation of SMAD2/3 and SMAD1/5/9 signaling was determined by western blot analysis of pSMAD2 and pSMAD1/5/9 in 1-hour-stimulated BMSCs and SMAD2/3-target gene expression of SERPINE1 in 24-hours-stimulated cells.
Chondrogenic Differentiation
BMSC pellets were generated by centrifuging 200,000 cells at 200g for 8 minutes. Pellets were cultured in chondrogenic medium consisting of DMEM-high-glucose, 1.5 µg/mL fungizone, 50 µg/mL gentamicin, 1 mM sodium pyruvate (Gibco), 1:100 ITS+ (BD Biosciences), 40 µg/mL L-proline, and 100 nM dexamethasone (Sigma-Aldrich), supplemented with either TGFβ1, Activin, or Nodal (all from R&D systems; same concentrations as above) or without supplementation (unstimulated control). Pellets were cultured for 10 days, renewing medium every 2 to 3 days.
Gene Expression Analysis
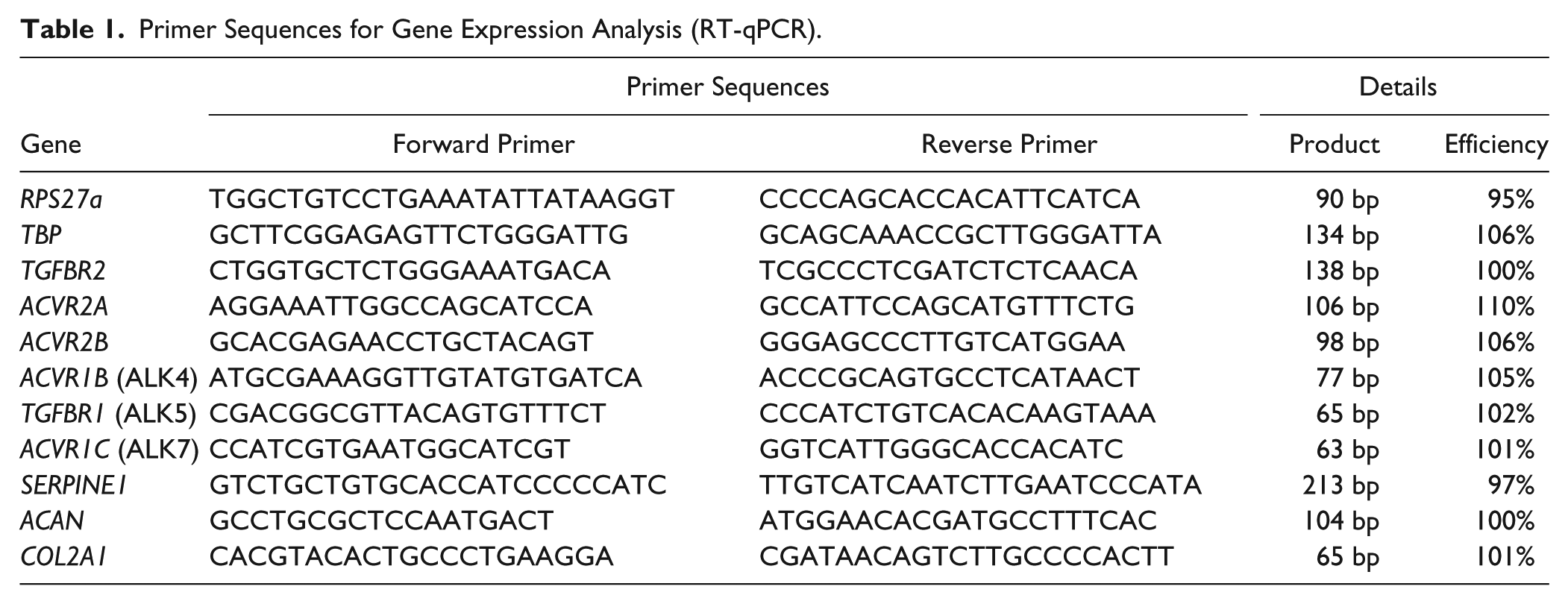
RNA was isolated with TRIzol and treated with DNAse according to manufacturer’s protocol (Invitrogen, Waltham, MA). Purity and concentration of isolated RNA were measured by spectrophotometry and samples with 260/280<1.6 were excluded. A total of 200 to 500 ng RNA was converted into cDNA (Invitrogen). Reverse transcriptase quantitative polymerase chain reaction (RT-qPCR) measurements were performed with SYBR Green MasterMix on a StepOnePlus system (Applied Biosystems) using primers listed in Table 1. Arbitrary Ct value of 40 was assigned to samples where no signal was detected. Gene expression was normalized to 2 reference genes; TBP and RPS27a. −ΔCt values were used for statistical analyses.
Primer Sequences for Gene Expression Analysis (RT-qPCR).
Histology
Pellets were fixed in 4% formalin, embedded in paraffin, and sectioned (6 µm). Proteoglycans were stained with 0.4% aqueous Thionin. For immunohistochemical staining of collagen type II, sections were pretreated with 0.1% pronase and 10 mg/mL hyaluronidase (Sigma-Aldrich), and incubated with 0.4 µg/mL mouse-anti-collagen type II (#II-II6B3, DSHB, Iowa City, IA) or 0.4 µg/mL mouse-IgG1 (#X0931, Dako, Glostrup, Denmark). Following incubation with alkaline phosphatase (AP)-conjugated secondary antibody (1:50, #HK-321-UK, Biogenex, San Ramon, CA), AP activity was visualized with new-fuchsin substrate and positive staining appeared as a magenta color. Sections were counterstained with hematoxylin.
Western Blot
Cells were lysed with lysis buffer (Cell-Signaling-Technology, Danvers, MA) containing 1% protease inhibitor (Roche, Mannheim, Germany) and sonicated on ice. Per lane of a 10% bisacrylamide gel, 12 µg protein was loaded. After SDS-PAGE, proteins were transferred to a nitrocellulose membrane by wet transfer. Membranes were incubated overnight with rabbit-anti-pSMAD2 (1:1000, #3101L, Cell Signaling Technology) or rabbit-anti-pSMAD1/5/9 (1:1000, #9511L, Cell Signaling Technology), followed by incubation with anti-rabbit HRP-linked antibody (1:1500, #P0448, Dako). Proteins were visualized by enhanced chemiluminescence. As loading control, GAPDH was detected by the Odyssey CLx system (LI-COR, Lincoln, NE) after incubation with mouse-anti-GAPDH (1:10,000; #G8795, Sigma-Aldrich) and donkey-anti-mouse-IRDye®680RD (1:10,000, #925-68072, LI-COR).
Statistical Analysis
Statistical analyses were performed using SPSS version 22 (IBM, Armonk, NY). Normal distribution was tested using the Shapiro-Wilk test. Data were analyzed with the generalized mixed models procedure considering “conditions” as fixed parameter and “donors” as random factor. Pairwise comparisons based on estimated marginal means were used. Multiple comparisons between unstimulated control and other conditions were corrected using Bonferroni’s procedure. P < 0.05 was considered statistically significant.
Results
BMSCs Express the Type II and Type I Receptors of TGFβ, Activin, and Nodal
Since complexes of type II and type I receptors are required for activation of SMAD2/3 signaling, 5 we evaluated their expression in BMSCs (N = 3 donors). Both the type II (TGFBR2, ACVR2A, ACVR2B) and type I receptors (TGFBR1, ACVR1B, ACVR1C) were detected with small variation between donors ( Fig. 1A-C ), indicating that the required receptors were present in BMSCs prior to chondrogenic induction and therefore not a limiting factor for Nodal, Activin, and TGFβ signaling.
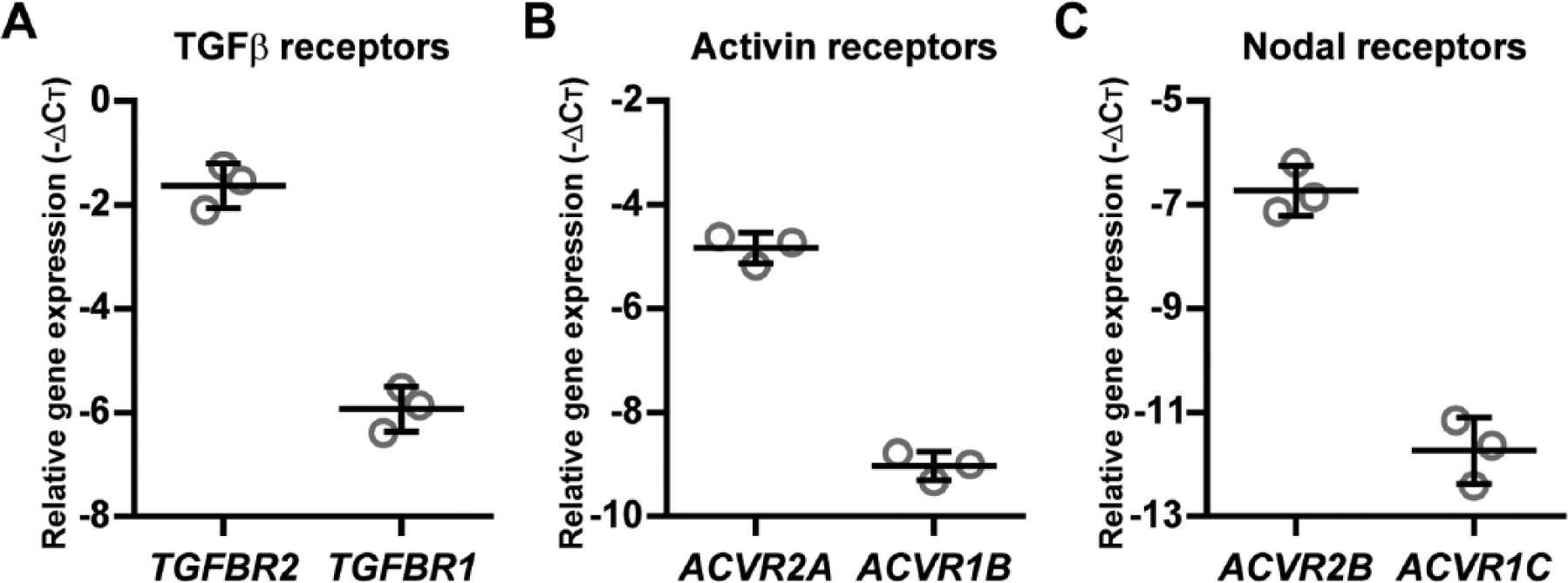
The receptors of TGFβ, Activin, and Nodal are expressed in BMSCs. In in vitro–expanded human BMSCs (N = 3 donors), gene expression of the receptors of (
TGFβ and Activin Activate SMAD2/3 Signaling, whereas Nodal Does Not
To determine whether TGFβ, Activin, and Nodal activated SMAD2/3 signaling in BMSCs, we analyzed expression of phosphorylated-SMAD2 protein (pSMAD2) and SERPINE1 mRNA. Since the effective dose of Activin and Nodal to activate SMAD2/3 signaling in BMSCs was unknown, we evaluated the effect of 10, 100, and 250 ng/mL.
The highest detected level of pSMAD2 was observed in BMSCs stimulated with TGFβ ( Fig. 2A ). Whereas Activin dose-dependently enhanced pSMAD2 expression, Nodal had no effect on pSMAD2 as compared to control ( Fig. 2A ). Consistent with these findings, both TGFβ and Activin enhanced SERPINE1 expression ( Fig. 2B ; P = 6.27E-26 for TGFβ; P = 5.64E-4 for 10 ng/mL Activin; P = 6.70E-9 for 100 ng/mL Activin; P = 3.56E-10 for 250 ng/mL Activin), whereas Nodal did not ( Fig. 2B ; P = 1.00 for all doses). Although Activin induced SERPINE1 expression, it never reached the same level of TGFβ-induced SERPINE1 ( Fig. 2B ). In addition, we show that, in contrast to TGFβ, Activin and Nodal did not enhance phosphorylation of SMAD1/5/9 proteins ( Fig. 3 ).
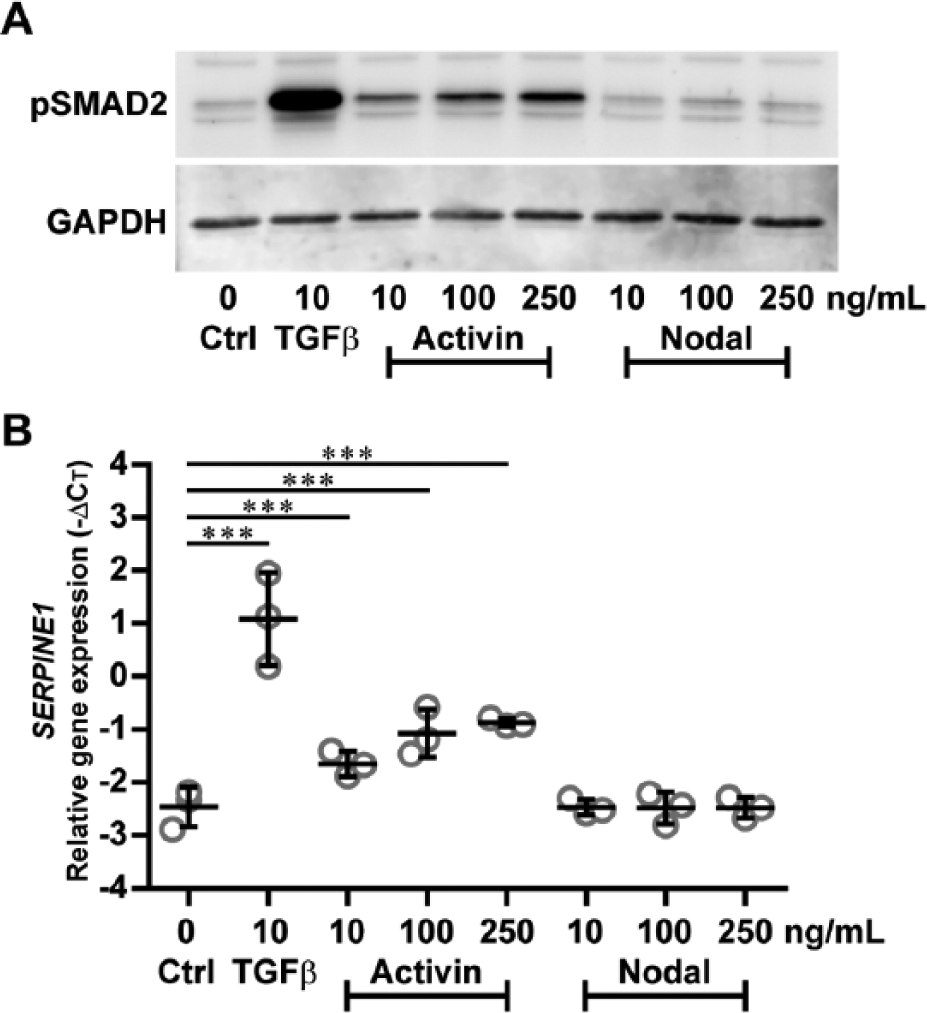
TGFβ and Activin, but not Nodal, induce SMAD2 phosphorylation and SMAD2/3-target gene expression. SMAD2/3 activation was determined in human BMSCs (N = 3 donors) that were either not stimulated (Ctrl) or stimulated with TGFβ, Activin, or Nodal at indicated concentrations. After 1 hour, cells were lysed for Western blot analysis of pSMAD2 using GAPDH as loading control (
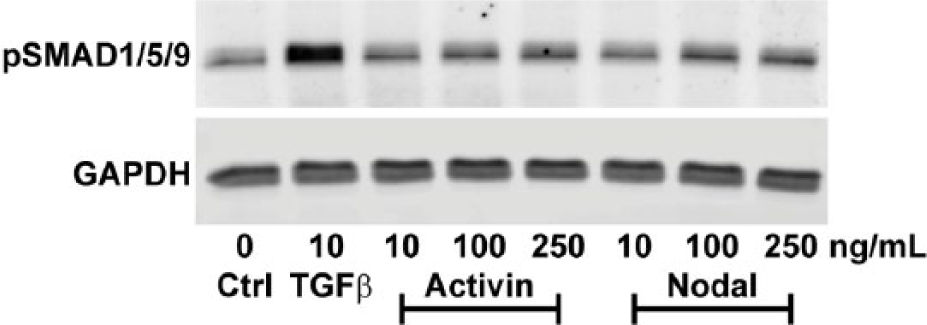
Unlike TGFβ, Activin and Nodal do not activate SMAD1/5/9 signaling in BMSCs. Activation of SMAD1/5/9 signaling was determined in human BMSCs (N = 3 donors) that were either not stimulated (Ctrl) or stimulated with TGFβ, Activin, or Nodal at indicated concentrations for 1 hour. Cells were lysed for Western blot analysis of pSMAD1/5/9 using GAPDH as loading control. Results of one representative donor (out of 3) are shown.
Taken together, these findings show that SMAD2/3 signaling and subsequent downstream SERPINE1 expression in human BMSCs were activated by TGFβ and Activin, but not by Nodal.
Activin and Nodal Do Not Induce Cartilage Formation
We next investigated whether Activin and Nodal were capable of inducing chondrogenic differentiation of BMSCs. Even though the selected concentrations of Nodal did not activate SMAD2/3 signaling, we tested its chondrogenic induction capacity as it had not been investigated previously.
TGFβ highly upregulated gene expression levels of ACAN ( Fig. 4A ; P = 8.01E-20) and COL2A1 ( Fig. 4B ; P = 6.02E-28). Activin slightly enhanced ACAN expression, but only at the highest dose of 250 ng/mL ( Fig. 4A ; P = 1.03E-2). Furthermore, 10 ng/mL as well as 250 ng/mL Activin increased COL2A1 mRNA levels ( Fig. 4B ; P = 3.19E-4 for 10 ng/mL; P = 1.93E-2 for 250 ng/mL). None of the 3 Activin doses induced ACAN and COL2A1 expression as high as TGFβ did. Compared to control, Nodal had no effect on expression of ACAN ( Fig. 4A ; P = 1.00 for all doses) and COL2A1 ( Fig. 4B ; P = 1.00 for all doses).
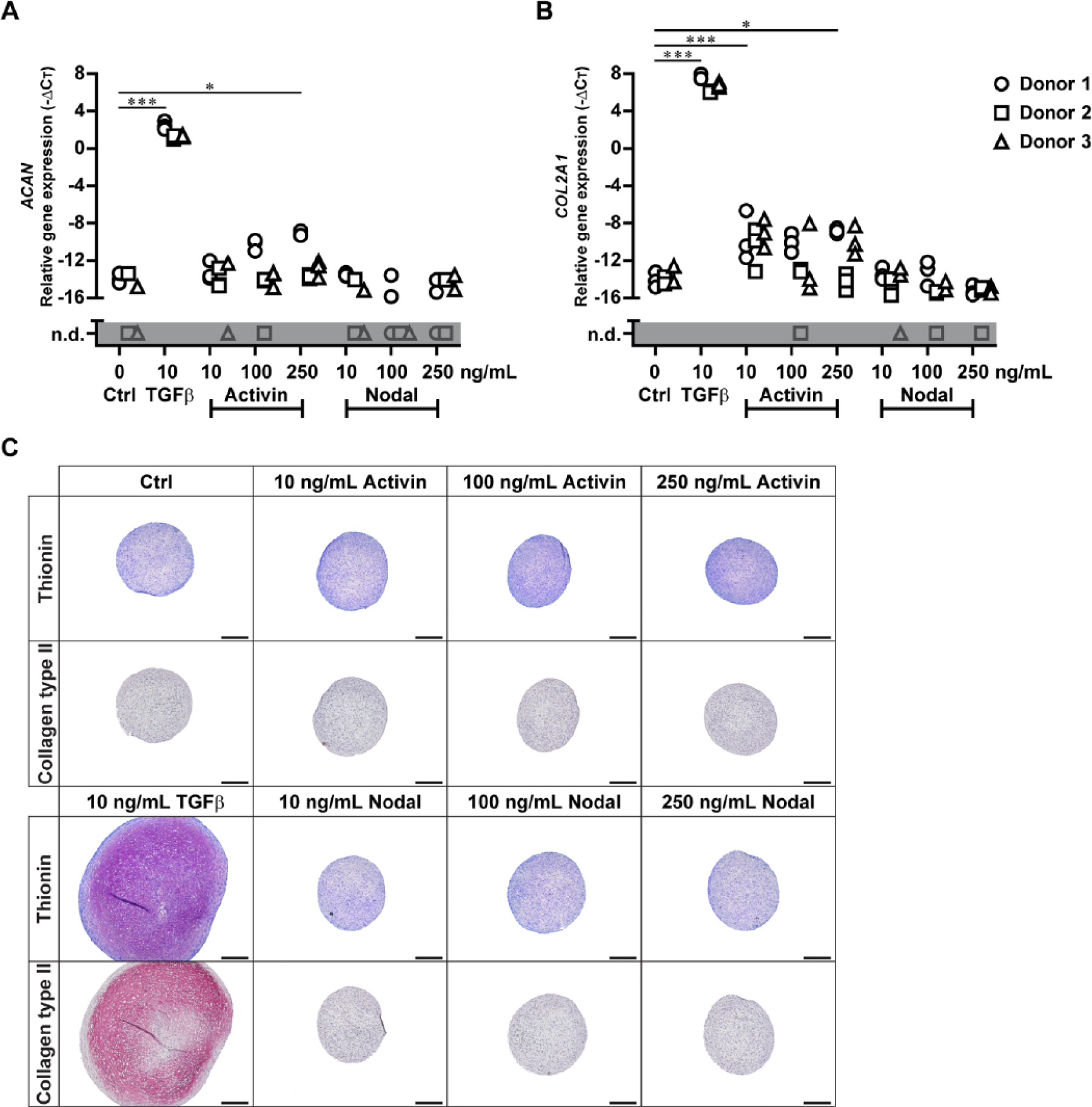
In contrast to TGFβ, Activin and Nodal do not induce chondrogenic differentiation. Human BMSCs (N = 3 donors with 3 pellets per donor) were pellet-cultured in chondrogenic medium either without growth factor (Ctrl) or with TGFβ, Activin, or Nodal at indicated concentrations. After 10 days, triplicate pellets were harvested for cartilage-specific gene expression analysis of (
The effects of TGFβ, Activin, and Nodal on cartilage-specific gene expression were reflected by deposition of proteoglycans and collagen type II. Only BMSC pellets stimulated with TGFβ stained highly positive for proteoglycans and collagen type II ( Fig. 4C ). Although Activin slightly enhanced expression of cartilage-specific genes, neither proteoglycans nor collagen type II were detected ( Fig. 4C ). Also Nodal-stimulated pellets were devoid of cartilage matrix.
These results demonstrate that, unlike TGFβ, Activin and Nodal did not induce chondrogenesis of BMSCs.
Discussion
The aim of this study was to determine whether Activin and Nodal could activate SMAD2/3 signaling in BMSCs and, unlike TGFβ, induce chondrogenic differentiation without inducing terminal differentiation. We show that the receptors of TGFβ, Activin, and Nodal were expressed in BMSCs and that TGFβ as well as Activin activated SMAD2/3 signaling, whereas Nodal did not. In contrast to TGFβ, Activin and Nodal dosages up to 25 times higher than that of TGFβ did not induce chondrogenic differentiation of BMSCs.
TGFβ is well known to induce chondrogenesis of BMSCs. Previously, we showed that activation of SMAD2/3 signaling by TGFβ is crucial for chondrogenic induction; however, TGFβ also activated SMAD1/5/9 signaling, which is involved in terminal differentiation. 2 Since Activin and Nodal had been reported to activate SMAD2/3 signaling without activating SMAD1/5/9 signaling in multiple cell lines,7-9 we hypothesized these ligands could be alternative candidates for TGFβ to induce chondrogenesis of BMSCs without terminal differentiation. We demonstrate that, in human BMSCs, SMAD2/3 signaling was activated by Activin, but surprisingly not by Nodal. Although it was described that 250 to 4,000 ng/ml of Nodal could activate SMAD2/3 signaling in murine P19 embryonal carcinomal cells, 8 we did not observe the same effect in human BMSCs (data not shown). Although we detected mRNA of Nodal receptor ALK7, the Ct value was relatively high (about 32), whereas the Ct value of TGFβ receptor ALK5 was approximately 26. Although this can be influenced by primer efficiency, this does hint toward an actual lower expression of Nodal receptors, which can have subsequent effects on possibilities for actual signaling and downstream gene activation. Why Nodal did not induce SMAD2/3 signaling and chondrogenesis may also be explained by dependency of Nodal on co-expression of Cripto, a member of the epidermal growth factor-Cripto/FRL1/Cryptic protein family, as formerly shown in embryonic cells. 9 As we are first in determining the effects of Nodal on adult BMSCs, further investigation is necessary to gain insight into the role of Nodal in chondrogenesis of BMSCs.
To date, the chondrogenic induction capacity of Activin remained unclear. In contrast to a study showing that Activin inhibited aggrecan expression in murine ATDC5 cells (chondroprogenitor cell line), 12 we show that Activin induced aggrecan expression in human BMSCs. This discrepancy might be caused by differences between ATDC5 cells and BMSCs, for instance, ATDC5 cells could undergo chondrogenesis with only insulin supplementation, 13 whereas BMSCs could not. 4 Although the molar dose of Activin was up to 25 times higher than that of TGFβ, Activin neither activated SMAD2/3 signaling nor induced formation of cartilage proteins as intense as TGFβ. Previously, low TGFβ doses (<1 ng/mL), known to result in less intense SMAD2/3 activation than with 10 ng/mL, 14 did not induce chondrogenesis, 4 indicating that chondrogenic induction may depend on the intensity of SMAD2/3 activation. SMAD3 has been shown to be important for activation of SOX9, a master transcription factor for chondrogenesis, 15 and we observed that silencing of SMAD2/3 expression in TGFβ-stimulated BMSCs inhibits chondrogenic differentiation (unpublished data, manuscript under preparation). Moreover, a recent study demonstrated that in HepG2 cells (human hepatocarcinoma cell line) the maximum efficacy of Activin to activate SMAD2/3 signaling is 36% lower than that of TGFβ, 16 suggesting that Activin is not able to activate SMAD2/3 proteins as efficiently as TGFβ. Therefore, possibly Activin did not stimulate cartilage matrix formation, because it could not activate a similar level of SMAD2/3 signaling as TGFβ did. In addition, since we recently found that both SMAD2/3 and SMAD1/5/9 pathways seem required for TGFβ-induced chondrogenesis of human BMSCs,2,6 the absence of SMAD1/5/9 pathway stimulation by Activin ( Fig. 3 ) might be responsible for absence of chondrogenic differentiation.
This study is first in comparing the chondrogenic induction capacity between known SMAD2/3-activating ligands: TGFβ, Activin, and Nodal. As only TGFβ induced chondrogenic differentiation of BMSCs, neither Activin nor Nodal are suitable alternatives for TGFβ to induce chondrogenesis without terminal differentiation.
Footnotes
Acknowledgments and Funding
The authors would like to thank Janneke Witte-Bouma for isolation and culture of mesenchymal stem cells. The II-II6B3 monoclonal antibody against collagen type II developed by T. F. Linsenmayer was obtained from the Developmental Studies Hybridoma Bank, originally developed under the auspices of the National Institute of Child Health and Human Development and maintained by The University of Iowa, Department of Biology, Iowa City, IA. This work was financially supported by the Dutch Arthritis Foundation (11-1-404), and by the Netherlands Institute of Regenerative Medicine (grant FES0908). RN was supported by ZonMw Translational Adult Stem Cell Research (Grant 116005009).
Authors’ Note
The work described in this article was done at the Erasmus MC University Medical Center and Radboud University Medical Center.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was obtained from the Erasmus MC Medical Ethical Committee (MEC-2014-106).
Informed Consent
Informed consent was not sought for the present study because waste material from surgeries was used according to the institutional policies with permission from the Erasmus MC Medical Ethical Committee (MEC-2014-106), whereby parents were given the option to opt-out of the use of this material if they wished.
