Abstract
Objective
Rats are an early preclinical model for cartilage tissue engineering, and a practical species for investigating the effects of aging. However, rats may be a poor aging model for mesenchymal stem cells (MSCs) based on laboratory reports of a severe decline in chondrogenesis beyond young adulthood. Such testing has not been conducted with MSCs seeded in a scaffold, which can improve the propensity of MSCs to undergo chondrogenesis. Therefore, the objective of this study was to evaluate chondrogenesis of middle-aged rat MSCs encapsulated in agarose.
Design
MSCs from 14- to 15-month-old rats were expanded, seeded into agarose, and cultured in chondrogenic medium with or without 5% serum for 15 days. Samples were evaluated for cell viability and cartilaginous extracellular matrix (ECM) accumulation. Experiments were repeated using MSCs from 6-week-old rats.
Results
During expansion, middle-aged rat MSCs demonstrated a diminishing proliferation rate that was improved ~2-fold in part by transient exposure to chondrogenic medium. In agarose culture in defined medium, middle-aged rat MSCs accumulated ECM to a much greater extent than negative controls. Serum supplementation improved cell survival ~2-fold, and increased ECM accumulation ~3-fold. Histological analysis indicated that defined medium supported chondrogenesis in a subset of cells, while serum-supplementation increased the frequency of chondrogenic cells. In contrast, young rat MSCs experienced robust chondrogenesis in defined medium that was not improved with serum-supplementation.
Conclusions
These data demonstrate a previously-unreported propensity of middle-aged rat MSCs to undergo chondrogenesis, and the potential of serum to enhance chondrogenesis of aging MSCs.
Introduction
Animal models play an essential role in the development of novel therapies for articular cartilage repair. When transitioning from in vitro experiments to live animal studies, rats offer several advantages as an early preclinical model, including low costs, availability of syngeneic, transgenic, or knockout animals, and an extensive volume of commercially available rodent antibodies and reagents.1,2 Furthermore, the relatively short life span of rats is favorable for studying the effect of aging.
In rats, healing of articular cartilage focal defects has been evaluated using an osteochondral model, and have included tissue engineering approaches using bone marrow mesenchymal stem cells (MSCs).3-5 In vivo rat studies typically involve young adult (<12 weeks old) subjects, although the availability of aging rats from commercial or federally funded programs offers the opportunity to evaluate older populations that better represent middle-aged humans for whom there is a significant need for cartilage tissue engineering. 6 However, for MSCs the chondrogenic propensity of rat cells has been reported to greatly diminish beyond young adulthood,7,8 to a much greater extent than aging human MSCs. 9 These data suggest that rats are not an appropriate species for modeling aging of human MSCs for cartilage tissue engineering.
To date, changes in the chondrogenic propensity of rat MSCs with aging have been investigated in in monolayer 7 or cell aggregate 8 cultures. However, these data may not be reflective of MSC-seeded scaffolds that are used for tissue engineering as 3-dimensional culture has been shown to improve extracellular matrix accumulation,10,11 or enhance expression of chondrogenic genes12,13 relative to aggregate cultures. Furthermore, integrating a polysaccharide network into MSC aggregates restored the chondrogenic potential of aging human MSCs that failed to differentiate in conventional cell aggregate culture. 14 Therefore, we postulated that rat MSCs from donors beyond young adulthood are capable of undergoing chondrogenesis when seeded into a 3-dimensional scaffold. The primary objective of this study was to evaluate chondrogenesis of MSCs derived from 14- to 15-month-old rats, an age that correlates with middle-aged humans 15 for which there is a significant need for cartilage resurfacing strategies. For culture expansion, we demonstrated that the limited growth potential of middle-aged rat MSCs in expansion culture can be improved with modifications to the culture environment that includes short-term exposure to chondrogenic medium. Chondrogenic cultures were conducted with MSCs seeded in agarose hydrogel, a scaffold that has been used extensively to study the biology of chondrocytes or chondrogenic MSCs. In agarose, preparations of middle-aged rat MSCs contain subpopulations of cells that secrete cartilage-like extracellular matrix in conventional defined chondrogenic medium. Cell survival and extracellular matrix accumulation were enhanced with the addition of serum to chondrogenic culture. Chondrogenic cultures were repeated using MSCs from 6 week old rats. Notably, the supportive role of serum in chondrogenesis appears to be specific to older donors as chondrogenesis of young MSCs was robust in defined medium, and was not enhanced by serum.
Methods
Bone Marrow Harvest and MSC Colony-Forming Culture
Bone marrow was flushed from the femurs of 14- to 15-month-old or 6-week-old Wistar rats that were euthanized for reasons unrelated to this study. All cultures were incubated at 37°C in ambient air plus 5% carbon dioxide. The nucleated cells were seeded at a concentration of 0.75 × 106 cells/cm2 in low glucose Dulbecco modified Eagle medium (DMEM, ThermoFisher Scientific, Waltham, MA) supplemented with 15% heat-inactivated fetal bovine serum (FBS) (Atlas Biologics, Fort Collins, CO), 10 mM HEPES (ThermoFisher Scientific) and antibiotic/antimycotic solution (100 U/mL penicillin, 100 µg/mL streptomycin, and 0.25 µg/mL Gibco Amphotericin B) (ThermoFisher Scientific). Colony-forming MSCs were observed after several days, and formed nearly confluent colonies by day 5 or 6. Preliminary testing of FBS lots from 4 vendors did not indicate a difference in MSC yield among sera (data not shown).
Given that the primary bone marrow cultures consisted of a mixed population of morphologies, the following steps were taken to largely isolate the fibroblastic, colony-forming MSCs. With exposure to trypsin it was observed that colony-forming MSCs detached quickly compared to the intracolony cell population. Therefore, primary cultures were treated with trypsin for 60 to 90 seconds, lightly tapped to detach the colonies, and then quickly neutralized with medium. To further isolate MSCs, the collected cells were seeded at a concentration of 20 × 103/cm2 in α-minimal essential medium (AMEM, ThermoFisher Scientific), 10% FBS, 10 mM HEPES, antibiotic/antimycotic solution, and 5 ng/ml fibroblast growth factor-basic (Peprotech, Rocky Hill, NJ), cultured for 1 to 2 days, and then treated with trypsin as described above. As determined by visual inspection, this process resulted in a largely homogenous population of cells that morphologically resembled colony-forming MSCs. Using this process, approximately 1 × 106 to 6 × 106 cells were obtained per animal.
MSC Expansion
Mesenchymal stem cells were seeded on tissue culture plastic (TCP), or TCP coated with fibrinogen precipitated from equine plasma, as previously described. 16 Cultures were maintained in AMEM expansion medium, or AMEM expansion medium plus 6.25 µg/mL insulin, 6.25 µg/mL transferrin, 6.25 ng/mL selenious acid, 1.25 mg/mL bovine serum albumin, and 5.35 µg/mL linoleic acid (ITS+ Premix, BD Biosciences, Bedford, MA). In some cases, monolayer cultures were maintained in chondrogenic medium (high-glucose DMEM, 1% ITS+ Premix, 10 mM HEPES, antibiotic/antimycotic solution, 0.1 mM non-essential amino acid solution (ThermoFisher Scientific), 37.5μg/mL ascorbate-2-phosphate (Wako Chemicals, Richmond, VA), 100 nM dexamethasone (Sigma-Aldrich, St. Louis, MO), and 10 ng/mL recombinant human transforming growth factor-β1 (Peprotech) 17 plus 5% FBS.
Encapsulation of MSCs in Agarose and Chondrogenic Culture
Low melting agarose was dissolved in phosphate buffered solution and sterile filtered. Casting molds were created by transferring 2.4 mL of warm 1% (w/v) agarose to 35 mm petri dishes and cooled to initiate gelation, and 6 mm diameter wells were created using a biopsy punch. Culture-expanded MSCs were suspended in warm 1.5% (w/v) agarose gel at 10 × 106 cells/mL, which was transferred to the casting molds and then cooled at room temperature. Cell-seeded agarose samples were cultured in defined chondrogenic medium, or chondrogenic medium containing 5% FBS.
Quantification of Cell Viability
MSC-seeded agarose samples were evaluated for cell viability using a commercial kit CellTiter-Blue assay (CTB; Promega, Madison, WI) and AMEM expansion medium without FGF2 in a cell culture environment. Samples were equilibrated in expansion medium for 2 hours, and then cultured in expansion medium containing CellTiter-Blue for 1 hour and 15 minutes. The reaction was stopped by adding 3% SDS (sodium dodecyl sulfate) and incubating at room temperature for 15 minutes, and the medium was analyzed according to the manufacturer’s instructions. Data were normalized to sample wet weight.
Quantification of Extracellular Matrix Accumulation
MSCs-seeded agarose samples were digested in proteinase K (Sigma-Aldrich) in 50 mM Tris HCl, 1 mM CaCl2 solution at 60°C overnight. Total accumulated sulfated glycosaminoglycan (GAG) and hydroxyproline were quantified by dimethylmethylene blue and dimethylamino benzaldehyde dye binding assays, respectively. Extracellular matrix accumulation data were normalized to the sample wet weight or CTB absorbance.
Histological Staining
MSCs-seeded agarose samples were fixed in 10% formalin for 48 hours, paraffin-embedded, sectioned, and mounted on slides. Immunohistochemical staining: Sections were deparaffinized and rehydrated, and then incubated with proteinase K at 37°C for 15 minutes. Sections were exposed to mouse anti-collagen type II IgG primary antibody using undiluted supernatant (Hybridoma Bank, Iowa City, IA), followed by donkey anti-mouse IgG secondary antibody conjugated with peroxidase at a 1:500 dilution (Jackson Immunoresearch, West Grove, PA). Antibody detection was performed using VECTOR NovaRED (Vector laboratories, Burlingame, CA). Additional sections were incubated with normal mouse serum at equal concentration to that of the primary antibody as a negative control. Mature cartilage was analyzed in parallel as a control. Toluidine blue: Sections were stained with 0.04% toluidine blue solution (Electron Microscopy Sciences, Fort Washington, PA).
Adipogenesis and Osteogenesis
MSCs were seeded at a concentration of 25 × 103 cells/cm2 in 48-well plates and cultured in AMEM expansion medium for 24 hours. Osteogenic cultures were maintained in high glucose DMEM, 10% FBS, 5 mM β-glycerol phosphate, 10 nM dexamethasone, and 37.5 µg/mL ascorbate-2-phosphate. Adipogenic cultures were maintained in StemPro adipogenic medium (ThermoFisher Scientific). Control cultures were maintained in AMEM expansion medium. Adipogenic cultures were maintained for 6 days, fixed in 10% formalin solution, and then stained with 1% oil red O. Osteogenic cultures were maintained for 15 days, fixed in 10% formalin solution, and then stained with 0.1% alizarin red.
Statistical Analysis
Data were evaluated for normality using the Shapiro-Wilk test. Data were analyzed using a paired t test, or for analysis of variance with mixed model using animal as a random effect. Pairwise comparisons were analyzed using the least squares means procedure. Statistical tests for normal distribution and paired t-test were performed using GraphPad Prism 7, while analysis of variance was performed using SAS 9.3 software. Data are presented as mean ± standard error.
Results
Expansion of Middle-Aged MSCs on Fibrinogen Surfaces
MSCs from 4 animals (2 male, 2 female) were used to compare proliferation on fibrinogen-coated surfaces (FS) to TCP. Cultures were seeded at 5000 cells/cm2. After 3 days, the cells were harvested and counted as both TCP and FS contains localized areas in which MSCs had grown to near-confluence. On day 3, the number of population doublings on FS was 2.3 ± 0.1, which was significantly higher than TCP (1.9 ± 0.1) (P < 0.05, data not shown). After reseeding into new flasks the rate of proliferation dropped dramatically. On day 7 the cumulative number of population doublings in FS cultures was 2.6 ± 0.1, and 1.9 ± 0.1 in TCP cultures (P < 0.05). Given the modest increase in population doublings, FS was used in all subsequent experiments.
Chondrogenesis of Middle-Aged MSCs in Defined Medium
Chondrogenesis was evaluated in conventional defined medium using MSCs from 7 male and 7 female animals. MSCs from colony-forming culture were seeded at 5000 cells/cm2 on FS, cultured in AMEM expansion medium for 3 days, and then seeded into agarose. MSC-seeded agarose was cultured in defined chondrogenic medium for 15 days, and then evaluated for GAG and hydroxyproline accumulation. Independent of sex, mean GAG and hydroxyproline accumulation was 1.1 and 0.11 µg/mg wet weight, respectively. GAG and hydroxyproline accumulation was not significantly different between male and female donors (P = 0.43 and P = 0.81, Fig. 1 ).
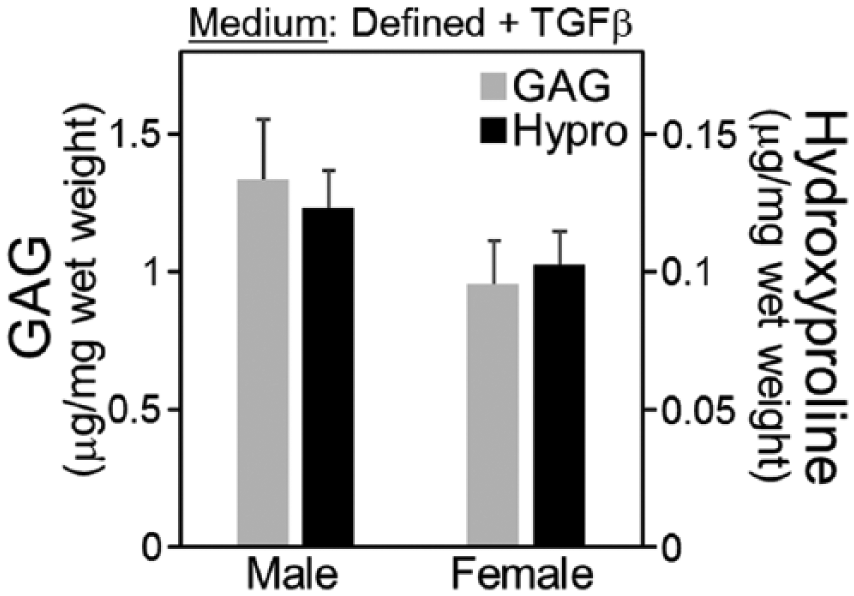
Extracellular matrix accumulation for middle-aged mesenchymal stem cells (MSCs) seeded in agarose and cultured in defined chondrogenic medium for 15 days. Experiments were conducted using MSCs from 7 male and 7 female donors (N = 7).
Chondrogenesis of Middle-Aged MSCs in Serum-Supplemented Medium
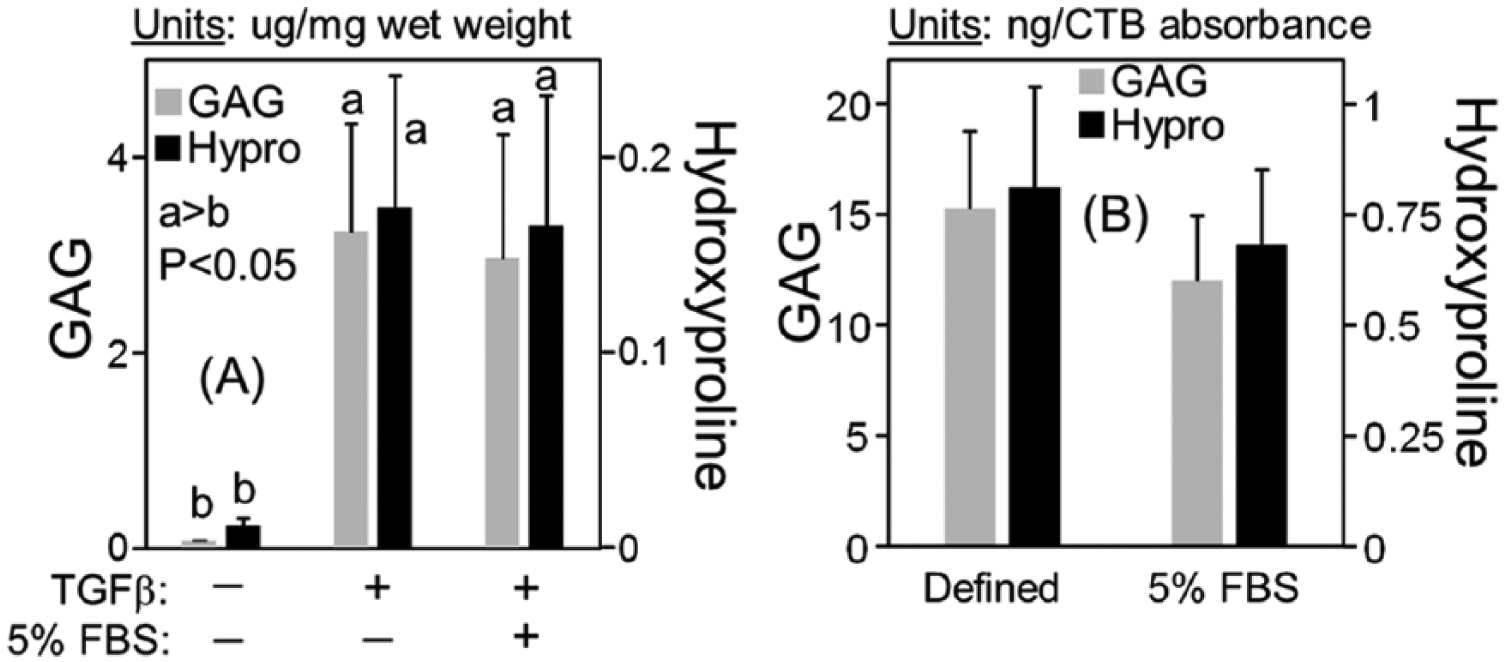
Chondrogenesis in medium containing 5% serum was compared to defined medium using MSCs from 4 animals ( Fig. 2 ). In this experiment, chondrogenic cultures were compared with negative control conditions (defined medium without transforming growth factor-β [TGFβ]). After 15 days of culture, GAG accumulation in negative controls was below the detection limit of the DMMB (dimethylmethylene blue) assay, while hydroxyproline accumulation was extremely low (~0.005 µg/mg wet weight). Extracellular matrix accumulation in defined, TGFβ cultures was comparable to Figure 1 , with hydroxyproline accumulation more than 20-fold higher than negative control cultures (P < 0.01). With serum supplementation, GAG and hydroxyproline accumulation was 2.7- and 3.4-fold higher than defined, TGFβ cultures, respectively (P < 0.002).
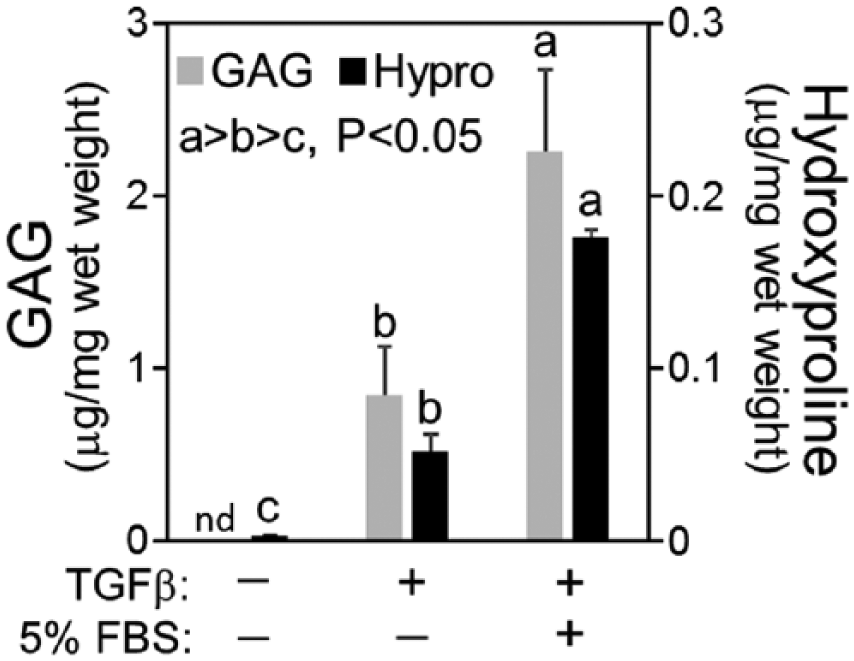
Extracellular matrix accumulation for middle-aged mesenchymal stem cells (MSCs) seeded in agarose and cultured in defined or serum-supplemented chondrogenic medium. Negative control cultures were maintained in defined chondrogenic medium without transforming growth factor-β (TGFβ). Cultures were maintained for 15 days prior to analysis. Experiments were conducted using MSCs from 4 donors (N = 4).
Expansion of Middle-Aged MSCs With Transient Exposure to Chondrogenic Medium
Given the limited capacity of middle-aged MSCs to proliferate in monolayer culture, 2 experiments were conducted to evaluate whether proliferation could be enhanced with exposure to chondrogenic medium. Experiments were conducted using MSCs from 4 animals. First, MSCs were seeded at 5000 cells/cm2 on FS, in AMEM expansion medium or chondrogenic medium containing 5% FBS. After 2 days, the cultures that were established in chondrogenic medium were switched to AMEM expansion medium. On day 3, the cultures that were established in chondrogenic medium had undergone 3.0 ± 0.1 population doublings, which was 0.8 population doublings more than AMEM cultures (P < 0.05, Fig. 3A ). However, temporary exposure to chondrogenic medium did not sustain a high level of proliferation as growth slowed to approximately half of a population doubling between days 3 and 7, in a manner that resembled AMEM cultures ( Fig. 3A ). Next, MSCs were seeded at a density of 2000 cells/cm2 on FS as the chondrogenic medium cultures in Figure 3A were nearly confluent on day 3. In this experiment, cultures were maintained in chondrogenic medium for 2 days, and were then switched to AMEM expansion medium as performed for Figure 3A , or AMEM expansion medium plus ITS+. Cultures maintained in AMEM expansion medium underwent 3.2 ± 0.1 population doublings by day 3, while ITS+ supplementation significantly increase proliferation to 3.7 ± 0.1 population doublings (P < 0.005, Fig. 3B ). With additional time in culture AMEM/ITS+ cultures experienced a decrease in proliferation rate, with a cumulative number of population doublings of 5.2 ± 0.3 on day 7 ( Fig. 3B ). For the second passage in AMEM/ITS+ medium was variable among donors, with population doubling times of 1.7, 2.5, 3.0, or 6.7 days.
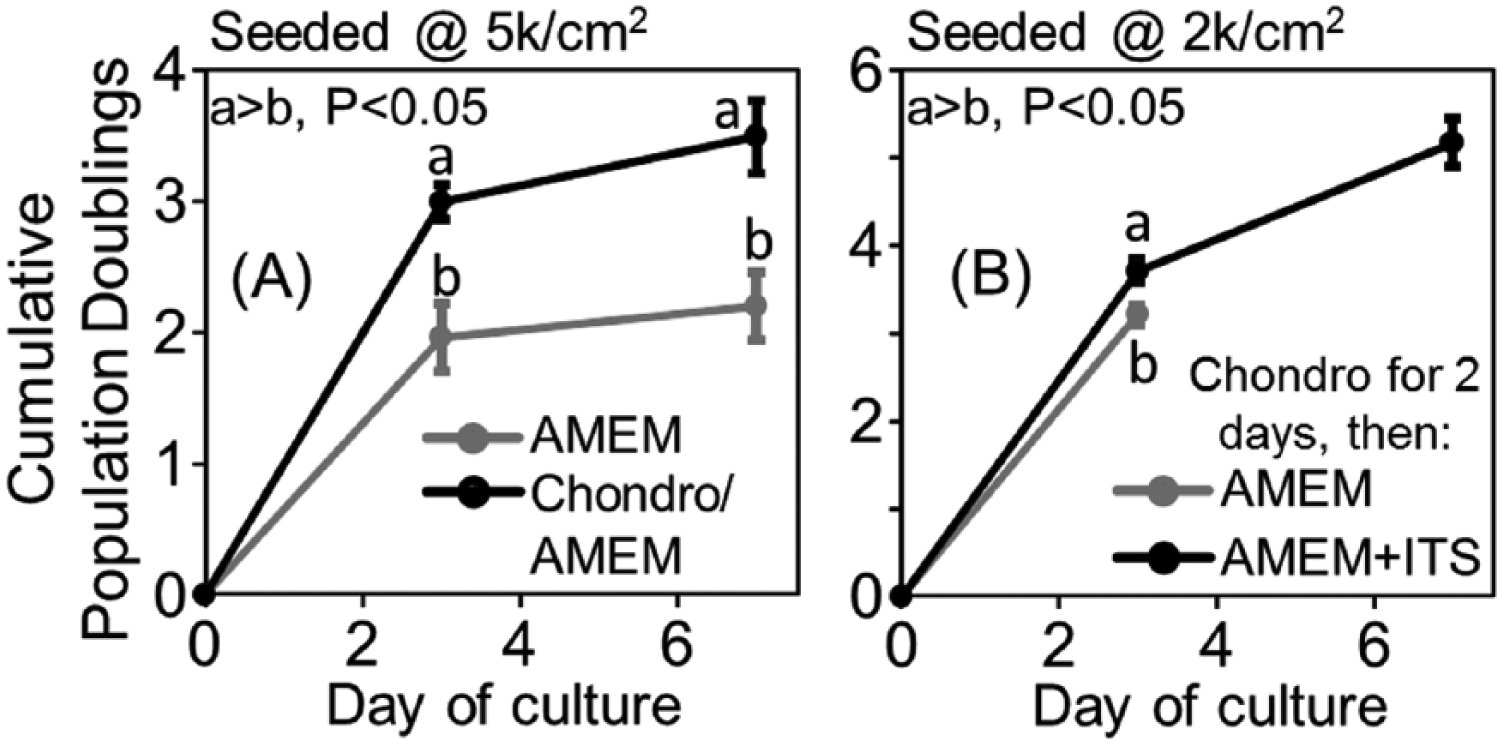
Expansion of middle-aged mesenchymal stem cells (MSCs) with short-term exposure to chondrogenic medium. (
Chondrogenesis of Middle-Aged MSCs Expanded With Transient Exposure to Chondrogenic Medium
Chondrogenesis was evaluated for MSCs expanded in chondrogenic medium and AMEM/ITS+ as performed for the first passage for Figure 3B , using MSCs from 4 animals. MSCs were seeded into agarose, and cultured in defined or serum-supplemented chondrogenic medium for 15 days. Consistent with Figure 4 , GAG and hydroxyproline accumulation normalized to wet weight in serum-supplemented cultures were 2.1- and 4.5-fold higher than defined medium cultures, respectively (P < 0.05 and P < 0.001, Fig. 4A ). In serum-supplemented cultures, cell viability (234 ± 36 CTB absorbance/mg wet weight) was 1.7-fold higher than defined medium (141 ± 34 CTB absorbance/mg wet weight) (P < 0.05, data not shown). When extracellular matrix accumulation was normalized to CTB absorbance, GAG accumulation was not significantly different between serum-supplemented and defined medium (P = 0.18), while hydroxyproline accumulation was 2.3-fold higher in serum-supplemented medium (P < 0.05, Fig. 4B ). For histological staining, toluidine blue and type II collagen staining was largely concentrated in pericellular regions ( Fig. 5 ). In both medium conditions a subset of cells did not accumulate pericellular extracellular matrix. Qualitatively, the proportion of cells that lacked pericellular staining in defined medium was higher than serum-supplemented samples.
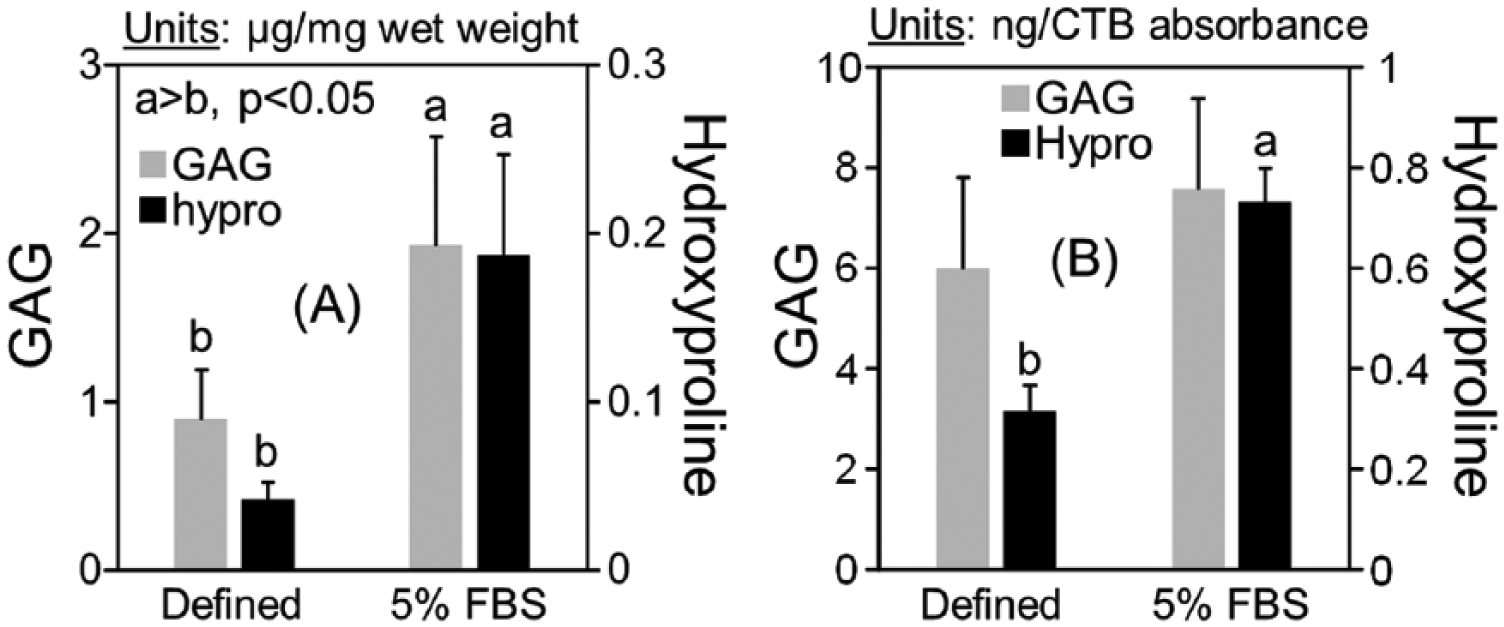
Extracellular matrix accumulation for middle-aged mesenchymal stem cells (MSCs) in agarose following short-term expansion in chondrogenic medium. Extracellular matrix data were collected for samples cultured in defined or serum-supplemented medium for 15 days, and normalized to (
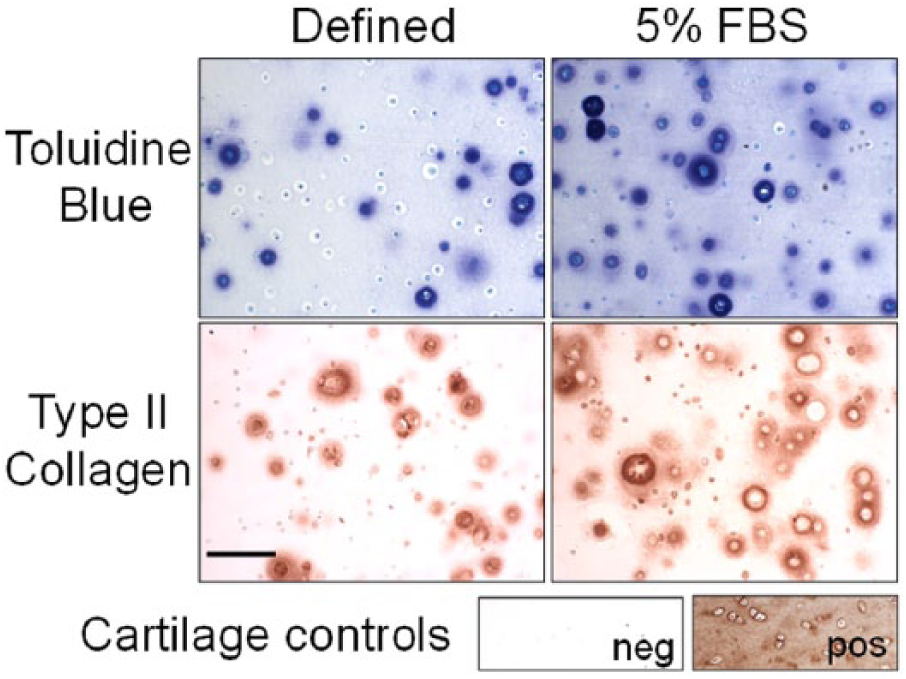
Toluidine blue and type II collagen immunohistochemical staining for middle-aged MSCs seeded into agarose and cultured in defined or serum-supplemented chondrogenic medium for 15 days. Bar = 100 µm.
Chondrogenesis of 6-Week-Old Rat MSCs
MSCs from 6-week-old rats were expanded in chondrogenic medium and AMEM/ITS+ for 3 days as described for Figure 3B , seeded into agarose, and cultured in defined or serum-supplemented chondrogenic medium for 15 days. In Figure 6A , GAG and hydroxyproline accumulation are normalized to wet weight. Negative controls in defined chondrogenic medium without TGFβ accumulated small amounts of GAG and hydroxyproline. For samples cultured in defined medium containing TGFβ, GAG and hydroxyproline accumulation was ~50- and 15-fold higher than negative controls, respectively (P < 0.001). Serum-supplementation did not significantly affect GAG (P = 0.23) or hydroxyproline (P = 0.53) accumulation as a function of wet weight. In TGFβ cultures, cell viability in defined medium (199 ± 22 CTB absorbance/mg wet weight) was not significantly different than serum-supplemented cultures (222 ± 16 CTB absorbance/mg wet weight) (P = 0.63, data not shown). When extracellular matrix accumulation was normalized to CTB absorbance, GAG (P = 0.08) and hydroxyproline (P = 0.39) accumulation were not significantly different between defined and serum-supplemented culture ( Fig. 6B ).
Chondrogenic cultures of mesenchymal stem cells (MSCs) from 6-week-old rats. Extracellular matrix accumulation for young MSCs in agarose following short-term expansion in chondrogenic medium. Extracellular matrix data were collected for samples cultured in defined or serum-supplemented medium for 15 days, and normalized to (
Adipogenesis and Osteogenesis of Middle-Aged MSCs
Adipogenesis and osteogenesis was evaluated using MSCs from 4 animals. MSCs from colony-forming culture were seeded at 5000 cells/cm2 on FS, cultured in AMEM expansion medium for 3 days, and then seeded into differentiation culture.
Adipogenesis
After 6 days, negative control cultures contained fewer than 5 cells with fat vacuoles that stained with oil red O ( Fig. 7A ). Cultures maintained in adipogenic medium contained a subset of cells that stained with oil red O ( Fig. 7B ) that were approximately 3 to 4 higher in frequency than negative control cultures. The potential for spontaneous adipogenesis in expansion medium was further explored for MSCs expanded for 7 days on FS, as described for Figure 1 . Adipogenic cells were not observed during the initial 5 days of expansion; however, by day 7, all cultures contained at least a small number of cells that stained for oil red O ( Fig. 7C ).
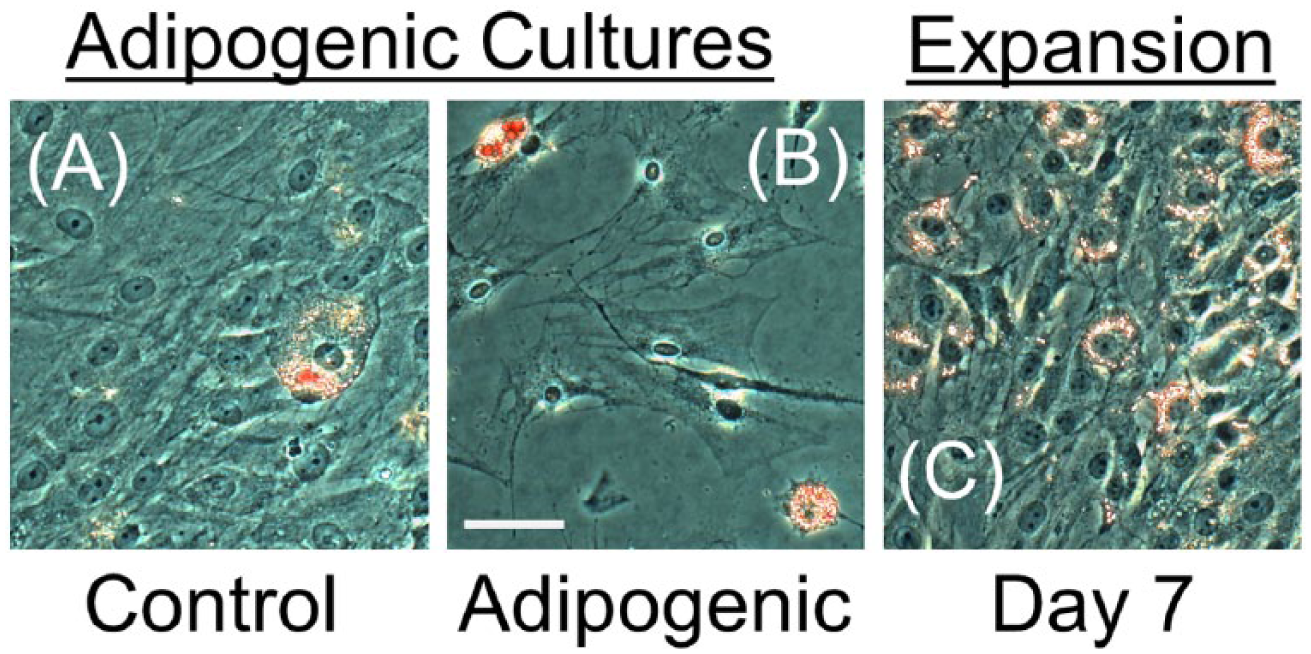
Oil red O staining for middle-aged mesenchymal stem cells (MSCs) in adipogenic culture. Staining in control (
Osteogenesis
After 15 days, alizarin red staining was nearly absent in negative control cultures ( Fig. 8A ), while osteogenic cultures contained abundant staining ( Fig. 8B ) that covered the majority of the cell monolayer. Similar to adipogenic control cultures, osteogenic controls appear to contain a small number of adipogenic cells (data not shown). Osteogenic cultures contained a much higher number of clustered adipogenic cells ( Fig. 8C ) that stained with oil red O ( Fig. 8D ).
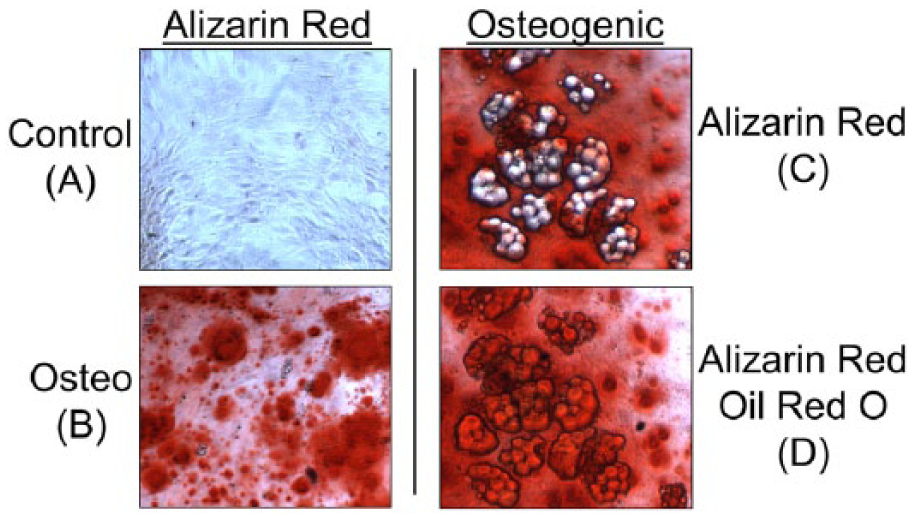
Alizarin red and oil red O staining for middle-aged mesenchymal stem cells (MSCs) in osteogenic culture. Cultures were maintained for 15 days prior to analysis. (
Discussion
The use of MSCs for cartilage tissue engineering has been motivated by evidence that chondrogenic MSCs can secrete abundant extracellular matrix in vitro. While minimal extracellular matrix accumulation has been reported for chondrogenic cultures of middle-aged rat MSCs,7,8 in the current study, agarose supported significant biochemical and histological evidence of neo-cartilage accumulation. In defined chondrogenic medium, GAG and hydroxyproline accumulation was on the order of that previously reported for middle-aged to elderly human MSCs that were cultured in a similar manner. 18 These data suggest that the 3-dimensional environment can strongly influence the chondrogenic propensity of aging rats MSCs, and supports the potential to evaluate MSC-based cartilage tissue engineering strategies in middle-aged subjects.
For many years a serum-free, defined medium containing TGFβ has been widely used to induce MSC chondrogenesis in vitro. 17 Recently, we reported that supplementing defined chondrogenic medium containing ITS+ with FBS moderately enhanced collagen accumulation in young adult equine MSC cultures. 19 For middle-aged rat MSCs, the effect of serum was more pronounced as GAG and hydroxyproline accumulation, and cell survival improved over defined medium. With serum-supplementation of middle-aged rat cultures, the accumulation of GAG (~2 µg/mg w/w) was approximately similar to young adult equine MSCs that were cultured in the same manner, while hydroxyproline accumulation in rat cultures (~0.18 µg/mg w/w) was approximately 50% of equine cultures. 19 While differences in variables such as seeding density, scaffold, or duration of chondrogenic cultures limits the extent to which comparisons to additional studies can be considered, it appears possible that with serum-supplementation the chondrogenic potential of adult rat MSCs may be comparable to MSCs from other species.
In the absence of ITS, serum supplementation has demonstrated a poor capacity to support MSC chondrogenesis.17,19,20 Therefore, in middle-aged MSC cultures it is likely that serum did not improve chondrogenesis per se, but functioned to prevent cell death and improve collagen accumulation of differentiated cells. The large (70%) difference in cell viability between defined and serum-supplemented conditions was unexpected as MSCs have generally proven to tolerate serum-free chondrogenic culture, although loss of viability of MSCs in agarose and defined chondrogenic medium has been reported for cells from numerous species,18,21-23 and has been attributed to apoptosis. 18 It is possible that the relatively low viability for middle-aged rat MSCs in defined medium was due to apoptosis that was induced by serum withdrawal. 24 Furthermore, a possible contributing factor to apoptosis is elevated levels of intracellular reactive oxygen species, 25 which have been reported to increase substantially with induction of MSC chondrogenesis,19,26 and can be lowered with serum supplementation. 19
In contrast to middle-aged rats, MSCs from 6-week-old rats experienced robust chondrogenesis in defined medium, with cell viabilities that were not significantly different between defined and serum-supplemented cultures. These data suggest that aging MSCs require chemical factors beyond conventional defined medium to optimally support cell survival and chondrogenesis. Given the undefined nature of serum it is difficult to postulate which serum components were chondro-supportive for aging rat MSCs, although based on the current study it appears that such components may correct for loss of function with aging. For example, serum may provide antioxidants that counteract the loss in endogenous antioxidants that MSC experience with chronological aging.27-29
Monolayer expansion rates of aging rat MSCs have been somewhat variable among studies. For example, for 6- to 12-month-old donors, the population doubling time was approximately 1 day over a 12-day culture period. 30 In another study, 12-month-old rat MSCs proliferated steadily over long-term culture, with a population doubling time of 2.7 days. 27 Other studies have reported a sharp decline in proliferation of aging MSCs with time in culture. For 17- to 18-month-old donors, the population double time following colony-forming culture was approximately 2.5 days during early passages, and then more than doubled with time in culture. 31 For 24- to 28-month old donors, the MSCs experienced a population doubling in less than 3 days, after which the growth rate dropped dramatically over a 10-day culture period. 32 In the current study, the pattern of proliferation with time most closely resembled 24- to 28-month-old donors in Igura et al. as early expansion on fibrinogen resulted in a population doubling time of 1.3 days, but then dramatically decreased with additional time in culture. While additional data are needed to establish trends of proliferation with aging, it appears possible that rat MSCs may experience a substantial drop in expansion potential during middle age.
The limited growth of middle-aged rat MSCs with time in expansion culture is not ideal for laboratory models of chondrogenesis, which can require millions of cells for a single experiment. Therefore, to improve the yield of MSCs from a single donor we investigated the effect of chondrogenic medium on proliferation in monolayer expansion culture. Previously, we observed that equine MSCs maintained for 2 days of chondrogenic suspension culture proliferated rapidly when returned to monolayer expansion culture. 33 In addition, in a recent study 10 days of exposure to chondrogenic medium in monolayer culture-induced markers of stemness and improved subsequent expansion and chondrogenesis of rat MSCs. 34 In the current study, 2 days of exposure to serum-supplemented chondrogenic medium stimulated MSC proliferation over AMEM expansion medium over the first passage, with the shortest population doubling time (0.81 days) achieved by adding ITS to AMEM expansion medium. When considering all expansion conditions used in this study, the combination of chondrogenic medium followed by AMEM/ITS improved the first passage cell yield by approximate 4-fold over standard AMEM culture (summarized in Table 1 ). Over the second passage, the population doubling time for MSCs maintained in chondrogenic medium followed by AMEM/ITS was 1.7 to 3.0 days for 3 out of 4 donors, which is consistent with proliferation of MSCs from younger rats.27,30 However, subject-to-subject variability may be a concern for middle-aged rats given that MSCs from 1 donor experienced little growth during the second passage.
Summary of Proliferation in Different Culture Conditions.

AMEM = α-minimal essential medium; FBS = fetal bovine serum; FGF2, fibroblast growth factor 2; PD = population doubling.
Evidence of oil red O staining in expansion and osteogenic cultures indicated a strong propensity for middle-aged rat MSCs to undergo adipogenesis. The age of the rats may have contributed to this behavior as in the bone marrow niche it is well known that aging is associated with increased adipogenesis. 35 Furthermore, for mouse MSCs spontaneous adipogenesis in expansion culture was reported to increase with donor age. 36 For osteogenic cultures, the presence of adipogenic cells was unexpected given that factors that stimulate osteogenesis generally suppress adipogenesis. 37 For example, treating MSC osteogenic cultures with a peroxisome proliferator-activated receptor gamma agonist induced adipogenesis while suppressing osteogenesis. 38 Given that trilineage potential is not necessarily uniform among individual cells within a preparation of expanded MSCs, 39 a possible explanation for the data in this study is that a subset of MSCs committed along the adipogenic lineage prior to seeding into osteogenic culture. Additional studies are needed to better delineate the extent to which expansion culture induces adipogenesis of aging rat MSCs, and the consequences for subsequent osteogenic or chondrogenic differentiation.
In summary, this study describes a previously unreported potential of middle-aged rat MSCs to undergo robust chondrogenesis, which supports the potential use of rats as an aging model for MSCs and cartilage tissue engineering. In addition, the beneficial effect of serum for middle-aged but not 6-week-old rats indicates a potential strategy for addressing age-related declines in chondrogenic propensity. For in vivo testing, the lack of significant differences between male and female donors suggests that sex is not a critical variable, and that potential differences between male and female subjects would reflect the ability of the joint environment to support cartilage regeneration as a function of sex. Given the importance of the ability of animal models to reflect humans, additional topics to consider include comparative studies with rat and human MSCs to evaluating the degree to which aging reduces chondrogenic potential, detailed phenotyping of chondrogenic rat MSCs with aging, and mechanisms by which aging affects rat MSC chondrogenesis. Furthermore, establishing the extent to which rat MSCs are consistent with cells from larger species would benefit the translation from small to large animal testing. Such research would be critical to thoroughly assess the potential of rats to serve as a model for MSC aging and chondrogenesis, and preclinical testing for cartilage tissue engineering.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: College Research Council, Colorado State University.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JDK owns shares of Advanced Regenerative Therapies and Regenerative Sciences. LRG owns shares of Advanced Regenerative Therapies. These are companies associated with cell-based therapies for orthopaedic injuries.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because the animals were euthanized for a University-approved study that was unrelated to the present study.
