Abstract
Objective
The aim of this study was to evaluate the chondroprotective effect of chemically decellularized meniscal allografts transplanted into the knee joints of adult merino sheep.
Methods
Lateral sheep meniscal allografts were chemically processed by a multistep method to yield acellular, sterile grafts. The grafts were transplanted into the knee joints of sheep that were treated by lateral meniscectomy. Joints treated by meniscectomy only and untreated joints served as controls. The joints were analyzed morphologically 6 and 26 weeks after surgery by the macroscopical and histological OARSI (Osteoarthritis Research Society International) score. Additionally, the meniscal grafts were biomechanically tested by cyclic indentation.
Results
Lateral meniscectomy was associated with significant degenerative changes of the articular cartilage of the lateral joint compartment. Transplanted lateral meniscal allografts retained their integrity during the observation period without inducing significant synovitis or foreign body reactions. Cellular repopulation of the grafts was only present on the surface and the periphery of the lateral meniscus, but was still completely lacking in the center of the grafts at week 26. Transplantation of processed meniscal allografts could not prevent degenerative changes of the articular cartilage in the lateral joint compartment. Compared with healthy menisci, the processed grafts were characterized by a significantly reduced dynamic modulus, which did not improve during the observation period of 26 weeks in vivo.
Conclusion
Chemically decellularized meniscal allografts proved their biocompatibility and durability without inducing immunogenic reactions. However, insufficient recellularization and inferior stiffness of the grafts hampered chondroprotective effects on the articular cartilage.
Keywords
Introduction
The menisci perform an important biomechanical function in the knee joint. They contribute to the congruity of the articulating joint surfaces, and thereby distribute load to a larger area. A number of clinical and biomechanical studies have demonstrated that major loss of meniscal tissue by trauma or meniscectomy increases contact pressure, joint instability, and associated shear stress, which in turn promotes progressive degeneration of the articular cartilage predominantly within the tibiofemoral compartment of the knee joint.1-6 For this reason, durable and functional reconstitution of the joint anatomy represents the primary aim, which has been clinically implemented by transplantation of fresh meniscal allografts for a couple of years.7-10 Transplantation of meniscal allografts has been shown to improve the clinical outcome regarding pain and joint function parameters, indicating the delay or reduction of articular cartilage degeneration.7,9-11
However, fresh meniscal allografts from human cadavers are not readily available in many countries because of concerns about potential disease transmission and immunological issues. 12 Therefore, biologic or synthetic scaffold materials have extensively been investigated as potential substitutes. However, their application has been limited to lesions with a preserved peripheral meniscal rim but not for replacing the complete meniscus.11,13-15
The objective of this study was to search for a more suitable meniscal graft that would combine the material properties of allografts with the safety of artificial scaffolds. Therefore, we focused on decellularized meniscal allografts processed by an established protocol.16,17 So far, the chemical process for decellularization has proven its suitability for a number of soft and hard tissues,16-19 but has not been employed for meniscal tissue yet. This method preserves the collagenous matrix with the original structural tissue anatomy and provides sterile grafts free from pathogens and lacking immunogenic or cytotoxic effects.16,17 We hypothesized, that such chemically processed meniscal allografts are well tolerated and maintain their integrity after transplantation in an in vivo model and protect the articular cartilage from early degeneration.
This is the first study to evaluate the chondroprotective effect of transplanted chemically processed decellularized meniscal allografts in vivo. We focused on an ovine model which regularly develops osteoarthritis following meniscectomy similar to the human knee.2-4
Methods
All procedures were carried out in accordance with the guidelines of the University Hospital of Erlangen-Nürnberg (Germany) and approved by the Institutional and Governmental Review Board for Animal Studies (Ref. 54-2532.1-27/09).
Chemical Processing of Meniscal Allografts
A total of 20 lateral menisci were dissected under sterile conditions from the right and left knee joints of 10 female adult donor sheep aged 3 to 4 years with body weights of 70 to 80 kg using an established protocol that has been optimized for cartilaginous tissues. 17 These processed menisci were assigned for biochemical (n = 4), microbiological analysis (n = 4), biomechanical analysis (n = 4), and for implantation (n = 8). The meniscal tissues were collected from euthanized sheep from the slaughterhouse and stored in 0.9% NaCl at −20°C until further processing. The donor animals and the animals used for this study were matched according to their body weight. Adherent synovial membrane and fibrous horn attachments were removed.
First, meniscus samples were osmotically pretreated with deionized H2O for 24 hours at room temperature (RT). Samples were then transferred to 1 N NaOH for 4 hours at RT for inactivation of pathogens, denaturation of DNA/RNA as well as for removal of cells, debris and noncollagenous components. All intermediate washing steps were carried out for 20 to 24 hours at RT, with the washing solution (deionized H2O) being exchanged for several times. After subsequent washing, samples were defatted with 70% ethanol for 3 hours at 40°C. For extraction of DNA, samples were transferred to 1 M guanidine hydrochloride (GndHCl) containing 0.05 N sodium acetate solved in an incubation buffer and incubated at 4°C for 96 hours. After another washing step, samples were treated with 5% H2O2 solution for 48 hours at 4°C protected from light. After subsequent washing, samples were treated again with 1 N NaOH solution for 4 hours and finally washed thoroughly for 48 hours with sterile endotoxin-free deionized H2O.
Prior to the animal study, 8 out of the 20 identically treated meniscal grafts were subjected to chemical (n = 4) and microbiological (n = 4) analysis to approve the processing procedure. Following processing, the percentage of denatured collagen in relation to native collagen (%) was assessed in biopsies to define the quality of the grafts. The biopsies were isolated from the red-white zone of the central third of the processed menisci using a 6-mm punch. First the biopsies were treated by selective digestion with α-chymotrypsin according to a modified method of Bank et al., 20 resulting in a fraction of denatured collagen and a fraction of residual (native) collagen. Then, the amount of denatured collagen in relation to native collagen was determined by the hydroxyproline assay as described previously 17 and was in the range of 6.2% ± 1.15%. The amount of endotoxins within the processed menisci was determined using the ToxinSensor Chromogenic LAL Endotoxin Assay Kit (Genscript, Piscataway, NJ, USA) and was limited to 2.6 ± 1.69 EU/device, which is far below the critical value for medical products (20 EU/device). Microbiological analysis approved sterility and complete removal of germs as evaluated according to the guidelines of ISO 11737-1:2006–2008 (data not shown) and lack of cytotoxicity of the processed allografts was examined as described previously in detail 17 (data not shown).
Surgical Procedure
Twelve female healthy adult merino sheep aged 3 to 4 years with body weights of 70 to 80 kg were used in this study. The experimental treatment scheme is summarized in Table 1 . The study included 1 control group (n = 4 knee joints) and 4 surgical groups. Group “LMX 6” (n = 4 knee joints) and “LMX 26” (n = 4 knee joints) were treated by lateral meniscectomy (“LMX”) with a follow-up period of 6 weeks or 26 weeks, respectively. Groups “LMA 6” (n = 4 knee joints) and “LMA 26” (n = 4 knee joints) were treated by lateral meniscectomy followed by subsequent transplantation of a processed lateral meniscal allograft (“LMA”) with a follow-up period of 6 weeks or 26 weeks, respectively. The animals were randomly allocated to the experimental groups.
Experimental Treatment Scheme.
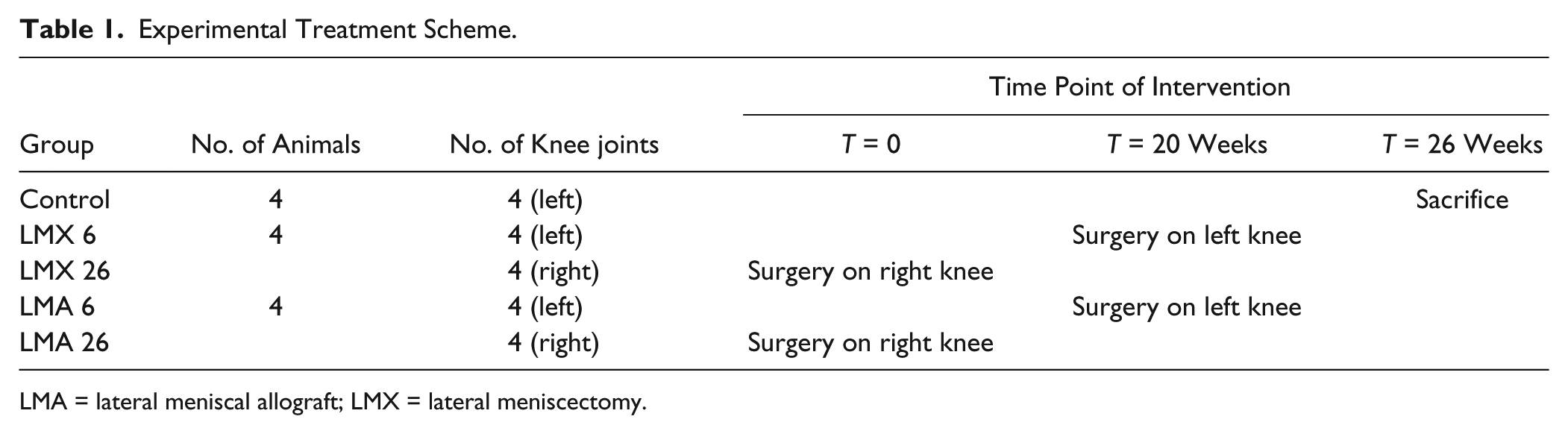
LMA = lateral meniscal allograft; LMX = lateral meniscectomy.
The surgery was first performed on the right knee joints. After 20 weeks, the animals were operated on their left knees according to the previous treatment method chosen for the right knee and were sacrificed another 6 weeks later. This treatment scheme allowed 2 different follow-up periods with 6 weeks for the left knee joints (“LMX 6” and “LMA 6”) and 26 weeks for the right knee joints (“LMX 26” and “LMA 26”). This scheme was chosen for ethical reasons to reduce the total number of animals as well as to reduce postoperative convalescence that would have arisen from simultaneous bilateral surgery. The animals were kept in an animal facility within appropriate stalls (2 sheep per 8 m2) as well as separated open-air enclosures. A temperature of 20°C to 25°C, a relative humidity of 40% to 55%, and a light–dark cycle of 12 hours were maintained. The animals were fed a standard diet ad libitum and welfare-related assessments were regularly performed. The operations were performed in special veterinary operation rooms.
The animals were induced for anesthesia by midazolam (0.4-0.8 mg/kg intravenously) and ketamine (2-5 mg/kg intravenously). Following endotracheal intubation, anesthesia was maintained by isoflurane. Perioperative pain was treated by buprenorphine (0.01 mg/kg intravenously or intramuscularly). After prepping and draping, the knee joint capsule was opened by lateral incisions anterior and posterior to the lateral collateral ligament, which was left intact. The lateral meniscus was removed by transection of the anterior and posterior meniscal horns at their bony attachment. For the sheep receiving processed allografts, transosseous tunnels were drilled from the anterior and posterior insertion sites of the lateral meniscus in projection to the medial aspect of the proximal tibia using an anterior cruciate ligament drill guide and a 2-mm Kirschner wire followed by overdrilling with a cannulated drill. The proper size of the allograft was confirmed by comparison with the explant, which was warranted by using animals with a matching body weight and age between donor and recipient. The processed meniscal allografts were attached at their anterior and posterior horns with nonresorbable Number 2 Mersilene Sutures (Ethicon, Norderstedt, Germany) in a Mason-Allen configuration. The sutures were captured by pull-out pins and transferred to the medial aspect of the tibia. The menisci were anatomically reduced and the transosseous sutures tied to the anteromedial tibia. The lateral collateral ligament remained intact. The joint capsule and the incision were closed in multiple layers with Vicryl sutures (Ethicon). Postoperative analgesia was generally adapted to the individual signs of discomfort and physical stress. Buprenorphine (0.01 mg/kg) was administered intravenously or intramuscularly at 1 or 2 doses for the first 2 to 3 postoperative days, followed by carprofen (4.0 mg/kg) for the first postoperative week.
Following sacrifice, the knee joints were dissected and first assessed macroscopically according to the OARSI (Osteoarthritis Research Society International) histopathology initiative for articular cartilage and osteophyte formation. 21 The total macroscopic score for articular cartilage was separately analyzed for each compartment (medial tibial plateau, medial femoral condyle, lateral tibial plateau, lateral femoral condyle) ranging from “0” (normal) to “3” (large erosions down to subchondral bone). The total macroscopic osteophyte score is the addition of the score values of the 4 compartments ranging from “0” (no osteophyte) to “3” (large osteophyte >4mm). Afterward, samples were prepared for biomechanical and histological analysis as described below.
Biomechanical Assessment
Immediately following sacrifice and macroscopic analysis, cylindrical biopsies of the grafts were harvested from the central part of the “pars intermedia” using a 5-mm biopsy punch. Biopsies from both, healthy age-matched menisci and nontransplanted processed meniscal grafts served as control groups. Parallelepiped tissue sections were cut from the upper and lower meniscus surfaces to prepare samples with parallel interfaces for the mechanical tests. First, the sample thickness was estimated by an ultrasound measurement as described previously. 22 The cutting resulted in sample thickness values of 1.81 ± 0.37 mm. Then, the samples were mounted in a sample holder, immersed in phosphate-buffered saline and tested with a high-precision material testing machine (BOSE ElectroForce LM1 TestBench, Eden Prairie, MN, USA), which was equipped with a 225.5-N force sensor and a plane-ended, 1.5-mm diameter steel indenter. A dynamical compression test was performed according to the protocol described by Danso et al. 23 Briefly, 20% prestrain was applied for a few seconds. Then, 10 cycles of sinusoidal loading at 1 Hz and a strain amplitude of 2 % were used for preconditioning. The measurement was performed with 10 cycles at 1 Hz sinusoidal loading with a strain amplitude of 2%. The measured displacement and force data were converted into strain (ε) and stress (σ) values, respectively. A sinusoidal function was then fitted to the time-dependent stress and strain values to obtain the amplitudes and the phase. The phase lag φ was calculated from the phase difference between the stress and strain curves. The dynamic modulus was calculated using the correction proposed by Hayes et al. 24 :

where a is the radius of the indenter, h is the sample thickness, κ is a scaling factor derived from Zhang et al. 25 and ν = 0.5 is the Poisson’s ratio using the assumption of an incompressible material. 23
Histological Assessment
For histological analysis of the articular cartilage, the complete femoral condyle and tibial plateaus were resected from the knee joints using an oscillating saw. The osteochondral specimens, as well as the menisci and synovial tissue were fixed in 4% paraformaldehyde for at least 12 hours. Menisci were decalcified in 0.5 M ethylenediamine tetraacetic acid (EDTA) for 2 weeks, and osteochondral samples were decalcified in the same way for 3 months. After standard processing, the samples were embedded in paraffin. The specimens were scanned throughout the tissue blocks with serial transverse 5-µm sections and stained with toluidine blue or hematoxylin-eosin. The microscopic scoring of articular cartilage and synovial changes were performed blinded by 3 independent specialists according to the OARSI histopathological initiative for sheep. 21 The microscopic score of articular cartilage is the addition of the single score values for the following parameters: “structure of the articular cartilage” ranging from “0” (normal) to “10” (erosion or severe fibrillation to subchondral bone), “chondrocyte density” ranging from “0” (normal) to “4” (no cells), “cell cloning” ranging from “0” (normal) to “4” (multiple cell nests or no cells), “interterritorial toluidine blue staining” ranging from “0” (normal) to “4” (no staining), and “tide-mark/calcified cartilage/subchondral bone” ranging from “0” (intact subchondral bone plate + single tide-mark) to “3” (tide-mark penetrated by blood vessels). The microscopic score of synovial changes is the addition of the single score values of the following parameters: “intimal hyperplasia” ranging from “0” (normal, 1 cell deep) to “3” (5 or more cells deep), “inflammatory cell infiltration” ranging from “0” (normal) to “3” (marked-discreet lymphoid aggregates), “subintimal fibrosis” ranging from “0” to “3” (heavy diffuse collagenous staining), and “vascularity” ranging from “0” (0-2 vascular elements per 100× field) to “3” (more than 8 vascular elements per 100× field).
Statistical Analysis
All data are presented as mean ± SD. For the evaluation of ordinally scaled morphological parameters nonparametric Kruskal-Wallis analysis of variance followed by post hoc Dunn’s multiple comparison tests was used to determine treatment-specific differences. The biomechanical data were analyzed by analysis of variance followed by Bonferroni post hoc multiple comparison tests. All statistical results were considered significant for P values <0.05.
Results
All animals tolerated the surgical procedure well and regained full weightbearing on the operated limb within the first 2 postoperative weeks. In all surgical groups, there was postoperative lameness for 1 to 3 days, followed by protective limping for about 1 week. Subjective differences were not apparent in the gross gait pattern between the different treatment groups during the course of the observation period of this study.
Macroscopic Assessment
Gross observation of the knee joints of untreated healthy adult animals showed no or only minimal degenerative changes of the articular cartilage ( Figs. 1a and 2a ). Instead, lateral meniscectomy (“LMX”) induced macroscopically visible degenerative changes of articular cartilage, which reached the level of significance in the lateral tibial plateau at week 6 (P = 0.05) and week 26 (P = 0.01) ( Fig. 1a ). The joint surfaces in the medial compartment were not affected by lateral meniscectomy.
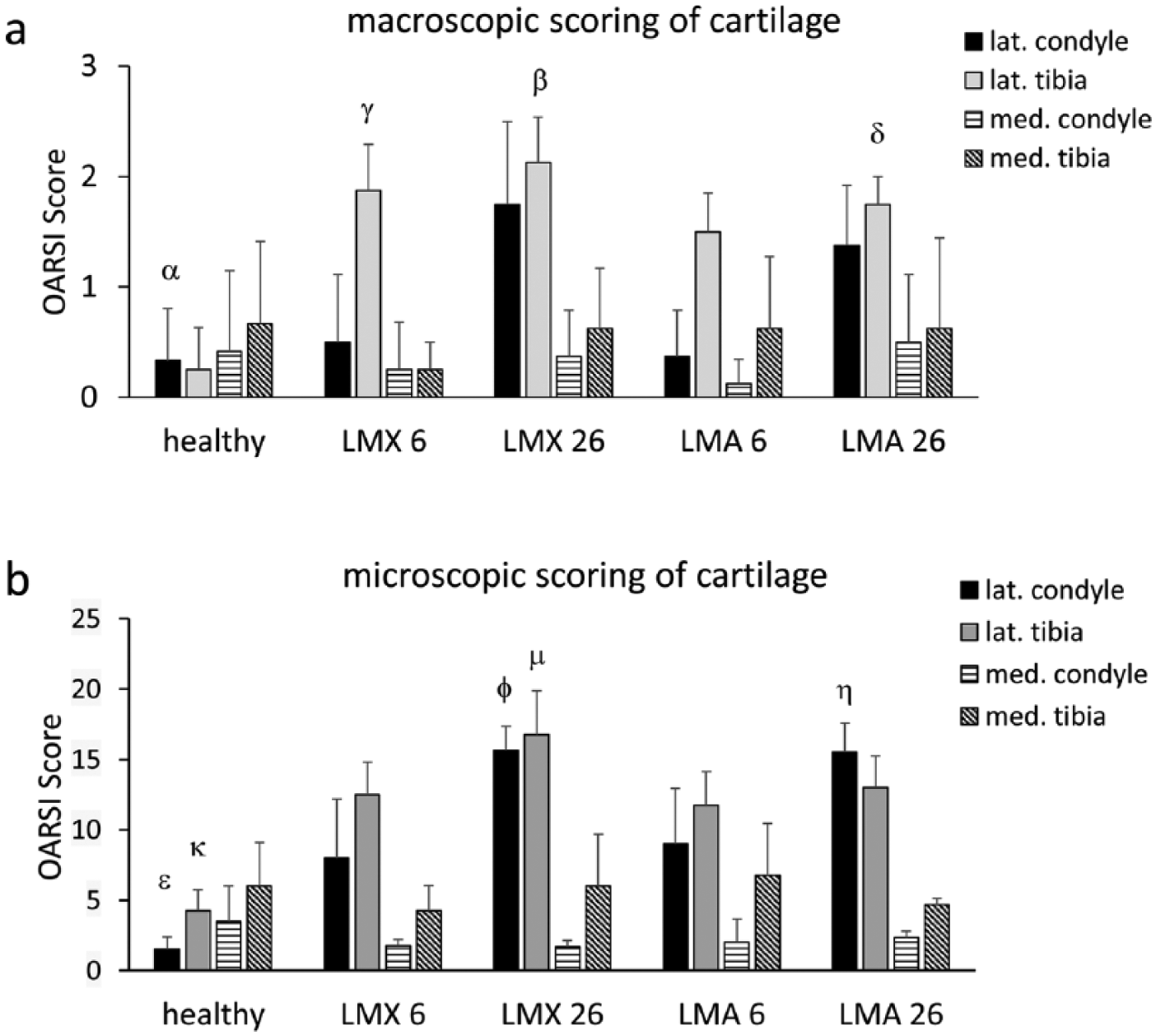
Macroscopic (
Transplanted lateral meniscal allografts (“LMA”) retained their integrity without any shrinkage throughout the observation period. No fraying or signs of resorption of the grafts were visible at 6 or 26 weeks ( Fig. 2b and c ). Transosseous suture fixation of the anterior and posterior horns remained tight and kept the grafts in their anatomical position throughout the study period. Synovial or fibrous tissue merged with the anterior and posterior attachment of the grafts and covered the suture material. Pannus formation was not observed. The macroscopic comparison of LMX- and LMA-treated animals revealed that LMA-treatment could not prevent the progression of degenerative changes of the articular cartilage in the lateral tibial plateau in the time course between 6 (LMA 6) and 26 weeks (LMA 26). After 26 weeks, the degenerative changes in LMA-treated joints (lateral tibia: 1.75 ± 0.25; lateral condyle: 1.375 ± 0.54) were not significantly different compared with LMX-treated joints (lateral tibia: 2.125 ± 0.41; lateral condyle: 1.75 ± 0.75) ( Fig. 1a ). There were no significant degenerative changes of the articular cartilage in the medial compartment at 6 or 26 weeks, regardless of LMX- or LMA-treatment ( Fig. 1a ).
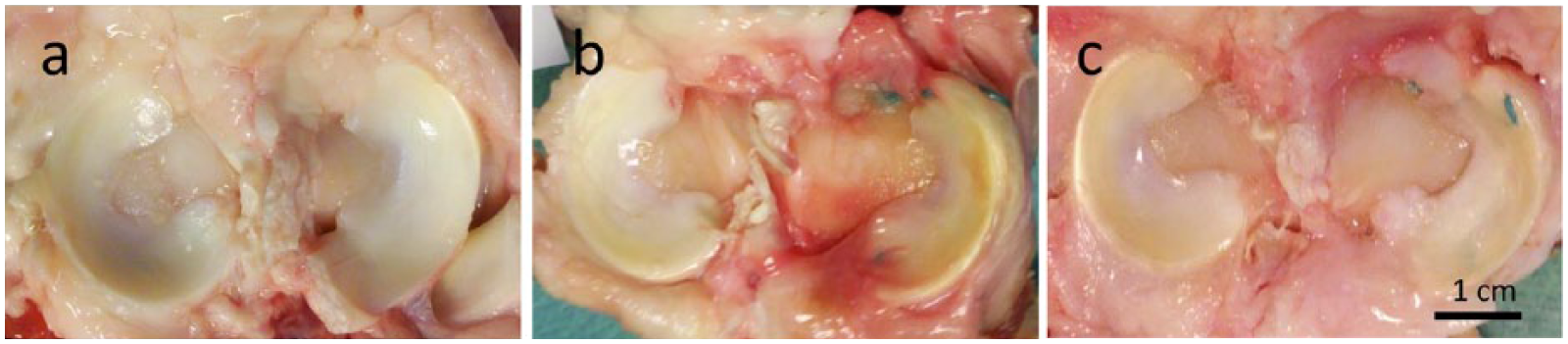
Macroscopic appearance of the tibial plateau of nontreated healthy animals (
Assessment of Meniscal Allografts
In the nontransplanted native status, the processed meniscal allografts are characterized by a preserved 3-dimensional collagenous network, lack of proteoglycans and by complete acellularity ( Fig. 3b , f , and j ). Following transplantation, only a limited repopulation with fibrochondrocyte-like repair cells could be observed. After 6 weeks, a thin cell layer could be detected on the upper and lower surface and at the lateral basis of the allografts ( Fig. 3c , g , and k ). Migration of the cells into the matrix of the allograft, leading to an even cell distribution, could not be observed at this stage. The allografts were still devoid of proteoglycans at week 6 ( Fig. 3c , g , and k ). After 26 weeks, the cell layer on the allograft surface gained in thickness forming a proteoglycan-rich matrix of up to 80 to 100 µm ( Fig. 3d and h ). However, the cells from the superficial cell layer still did not migrate through the dense superficial collagenous network into the center of the allografts. Instead, cellular ingrowth of the grafts was rather evident at the lateral meniscal basis with cell repopulation along former vascular channels from the meniscal basis toward the center of the allografts ( Fig. 3d and l ). The ingrowing cells were characterized by spindle-shaped morphology generating a locally restricted proteoglycan-rich secondary matrix ( Fig. 3l ). Repopulation of the grafts was still completely lacking in the central part of the graft after 26 weeks with no staining for proteoglycans in the acellular regions.
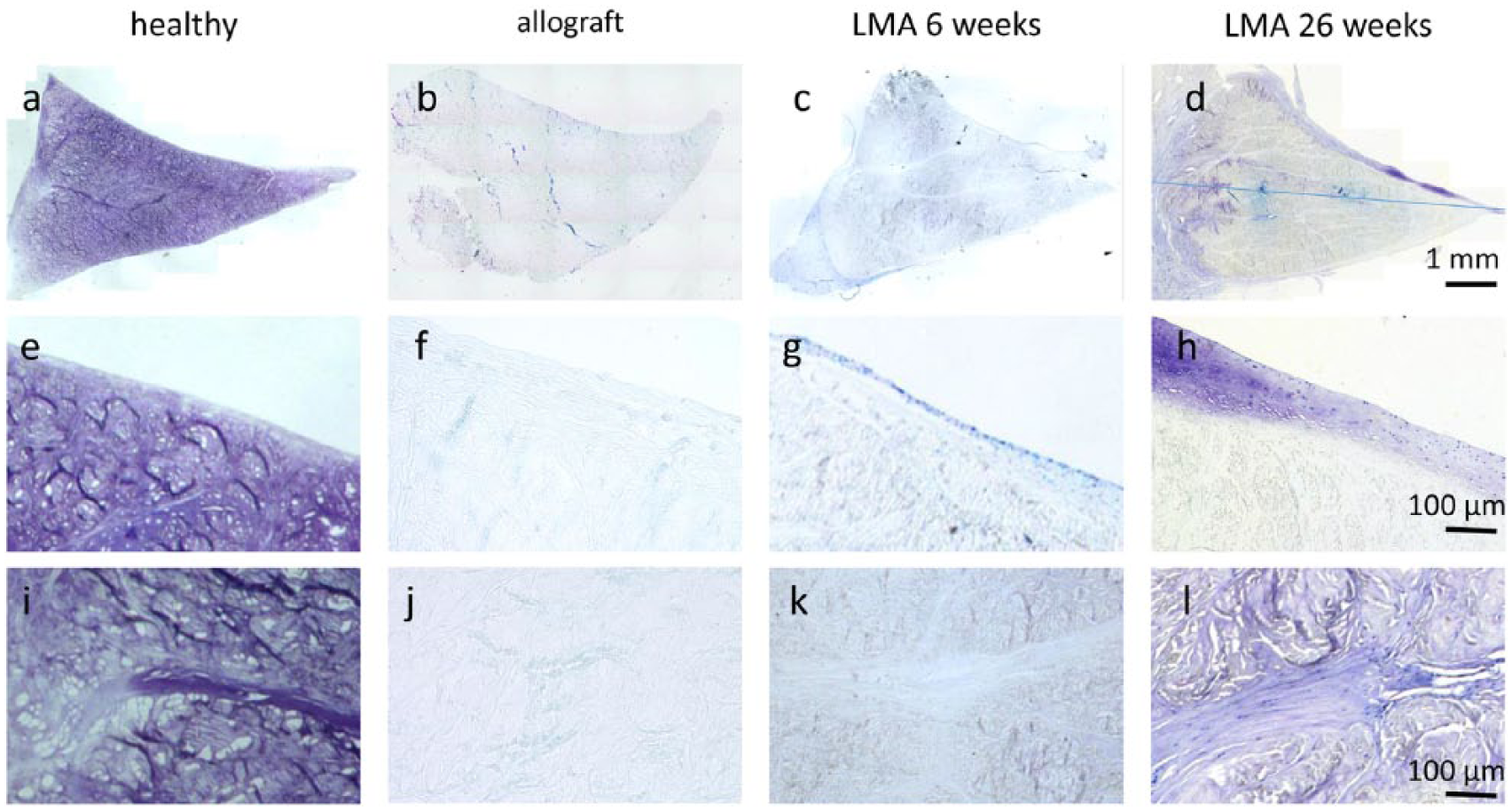
Histological analysis of meniscal tissue. Toluidine blue staining of healthy lateral meniscus of untreated control animals (
Histological Assessment of Articular Cartilage
The articular cartilage of the medial and lateral femoral condyle and tibial plateau was microscopically assessed using the scoring system according to the OARSI histopathology initiative. 21 Healthy untreated joints were characterized by no or only minimal degenerative changes, which were present in the medial than in the lateral compartment ( Figs. 1b , 4a and f ). Instead, lateral meniscectomy (“LMX”) induced moderate degeneration of the articular cartilage in the lateral condyle (8.0 ± 4.1) and tibial plateau (12.5 ± 2.3) at week 6 ( Figs. 1b , 4b and g ). The degenerative changes induced by meniscectomy had further progressed at week 26 (LMX 26), leading to severe osteoarthritis characterized by erosion and loss of proteoglycans from the surface to the mid/deep zone both in the lateral condyle and the lateral tibial plateau ( Figs. 1b , 4c and h ). Transplantation of lateral meniscal allografts (LMA) could not protect articular cartilage from developing degenerative changes in the lateral condyle (8.0 ± 4.18) or tibial plateau (12.5 ± 2.3) in the first 6 weeks ( Figs. 1b , 4d and i ). At 26 weeks, degenerative changes in LMA-treated joints further progressed. At that time point; the degree of the degenerative changes in the lateral tibial plateau or lateral femoral condyle did not differ significantly between in LMA-treated joints (13.0 ± 2.23; 15.5 ± 2.06) compared with LMX-treated joints (16.75 ± 3.1; 15.6 ± 1.7), respectively ( Figs. 1b , 4e and j ). The articular cartilage of the medial knee compartment remained completely unaffected by the transplantation procedure during the whole observation period ( Fig. 1b ).
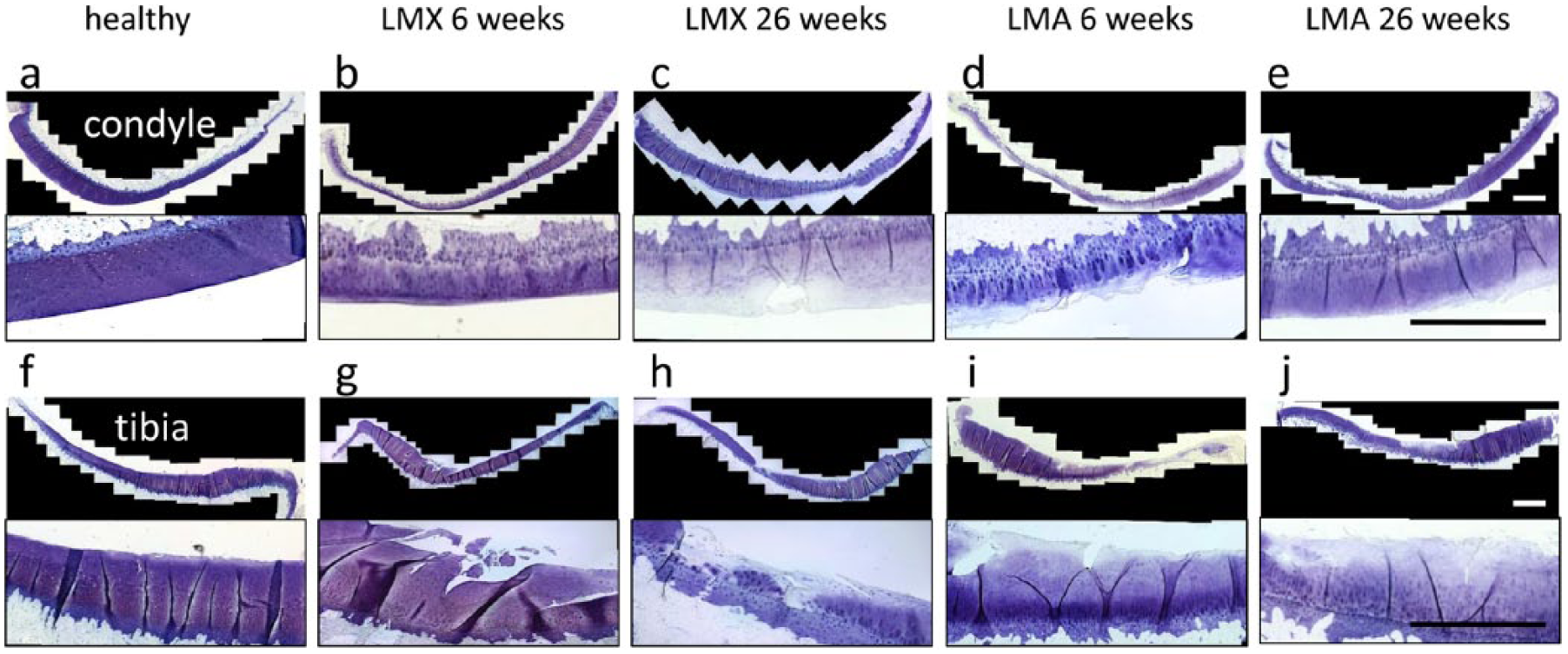
Histological analysis of articular cartilage of the lateral femoral condyle (
Macroscopic Analysis of Osteophytes
In both, LMX- and LMA-treatments, the formation of osteophytes was observed, which was most prominent in the lateral femoral condyle and in the lateral tibial plateau. Macroscopically, osteophytes appeared as whitish cartilaginous outgrowths at the joint margins of the lateral femoral condyle ( Fig. 5a ) and the lateral tibial plateau ( Fig. 5c ) forming a contrast to the yellowish articular cartilage. Histologically, osteophytes included a superficial zone of hyaline-like cartilage with an osseous outgrowth underneath. In contrast to the adjacent articular cartilage, osteophytes typically lack a continuous tide-mark and a nonstratified cellular arrangement ( Fig. 5b , d , and e ). Interestingly, LMA-treatment led to more severe osteophyte formation compared with LMX-treatment, which was most obvious after 26 weeks (P = 0.001) ( Fig. 5a-f ).
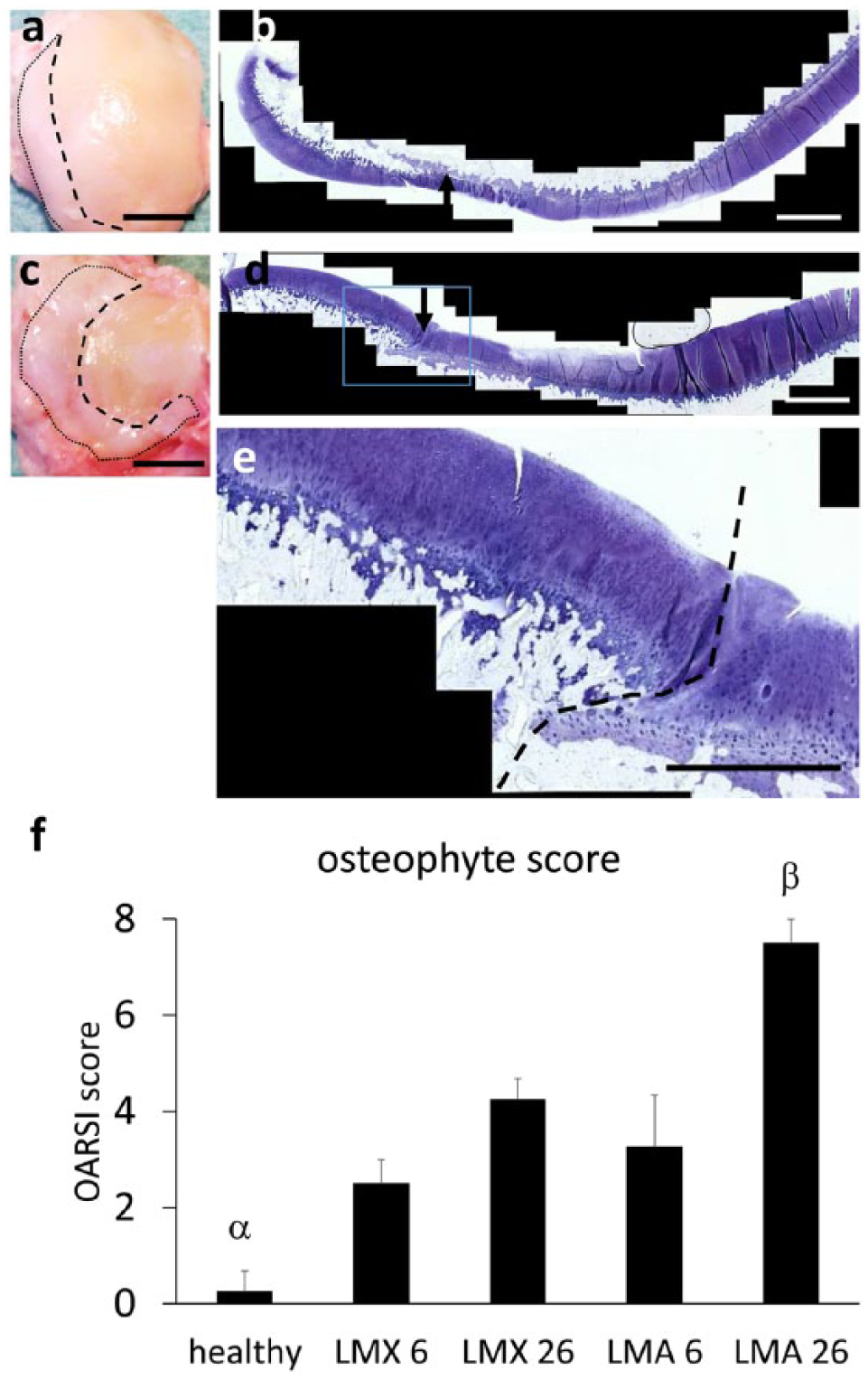
Osteophyte formation. Macroscopic appearance of the lateral femoral condyle (
Synovial Changes
The synovial membrane was assessed microscopically according to the OARSI histopathological initiative 21 to evaluate the degree of inflammation induced by LMX- or LMA-treatment. Gross observation of the joints revealed signs of mild to moderate synovitis predominantly in the inferior half of the lateral recess. All synovial samples were harvested from this location. The general score revealed only mild postoperative synovitis following LMX-treatment at week 6 (3.0 ± 1.0) with a moderate increase at weeks 26 (4.3 ± 0.4), which reached the level of significance compared with healthy joints (P = 0.03) ( Fig. 6b , c , and f ). The elevated score can be ascribed to subintima fibrosis and increased vascularity rather than intima hyperplasia and presence of inflammatory cells. LMA-treatment elicited a significant degree of synovitis at 6 weeks (4.7 ± 0.5) compared with healthy joints (P = 0.01) that declined at 26 weeks (3.3 ± 0.5) ( Fig. 6d and e ). The elevated values following LMA-treatment were primarily due to subintima fibrosis. Signs of foreign body reactions were not observed.
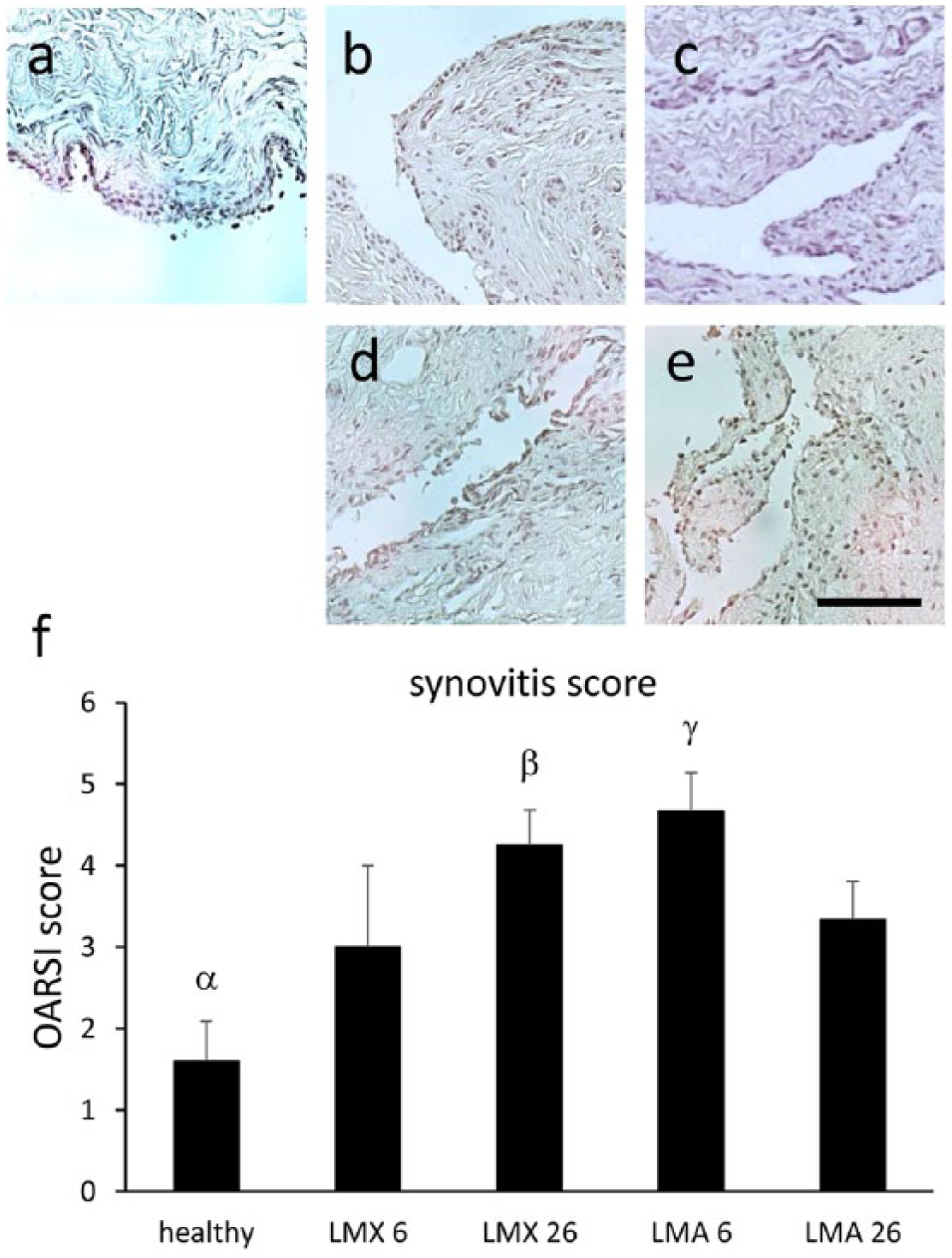
Analysis of synovial changes. Histologic sections of the synovial membrane from the lateral recesses of untreated control joints (
Biomechanics
The analysis of the dynamic modulus and phase lag revealed no significant differences between the upper and the lower parts of the menisci biopsies. Therefore, the values were averaged for each sample. The phase lag did not differ significantly between the different groups ( Fig. 7a ). However, the dynamic compressive modulus of nontransplanted allografts (T = 0) (2.6 ± 3.8 MPa), transplanted allografts after 6 weeks (LMA 6) (1.4 ± 0.2 MPa), or transplanted allografts after 26 weeks (LMA 26) (1.0 ± 0.3 MPa) were significantly lower than that of samples of the healthy control group (9.2 ± 5.9 MPa). No significant differences were observed between (nontransplanted) allografts (T = 0) and implanted allografts after 6 or 26 weeks ( Fig. 7b ).
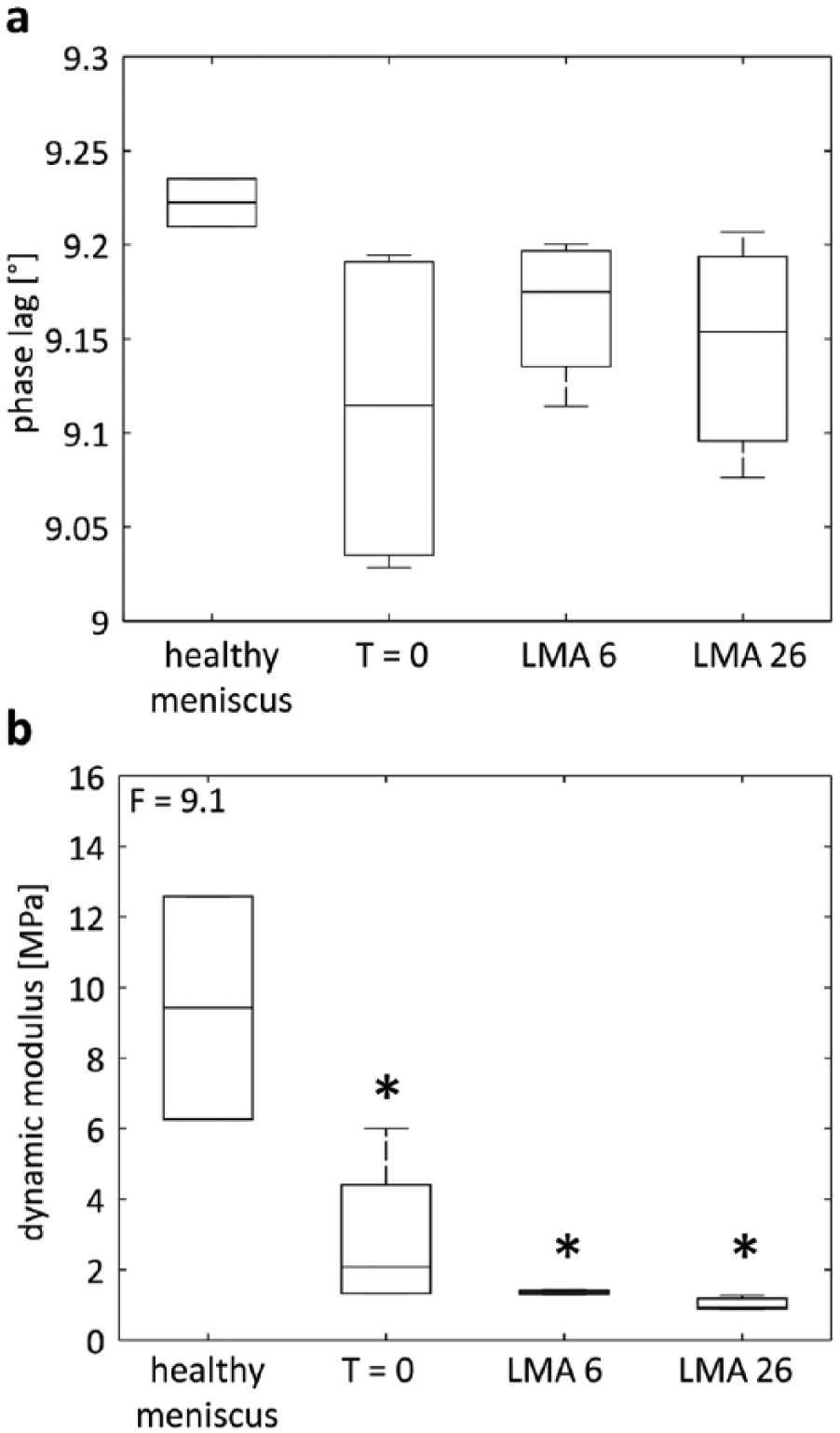
Biomechanical assessment of meniscal tissue and processed meniscal allografts. Phase lag (
Discussion
This is the first in vivo study investigating the chondroprotective effect of chemically processed decellularized meniscal allografts in an osteoarthritis sheep model. We focused on the lateral meniscus since the lateral meniscus relatively carries more joint load than the medial meniscus, 20 which results in earlier onset of degenerative changes following lateral meniscectomy compared with medial meniscectomy. 21 In this pilot work based on (1) a limited number of animals and (2) a limited observation period of 26 weeks, transplanted decellularized meniscal allografts did not significantly prevent or delay degenerative changes. In the literature, there is controversial data about the amount of chondroprotective effects achieved by meniscus transplantation. The outcomes appear to depend on the animal model or clinical setting, the follow-up period, and the method of analysis.26-32 Transplanted meniscal grafts may support joint congruity and contribute to the stability, which may be the occasion for the pain relief and favorable functional clinical outcomes described in several clinical studies.7-10 Thus, recent studies reported at least a reduction of degenerative changes following transplantation of meniscal xeno- and allografts.26,32-35 One reason for the lacking chondroprotective effect in this study can be ascribed to the inferior biomechanical properties of the processed grafts, which are characterized by a significant lower dynamic modulus compared with healthy menisci. This concern has also been observed in other studies and can be attributed to the removal of proteoglycans by the processing procedure.33,34 Other commercially available artificial acellular meniscal scaffolds made of polyurethane are also characterized by inferior biomechanical properties, 36 which has limited their clinical use to partial meniscal defects so far.14,37 The reconstitution of a proteoglycan-rich extracellular matrix would depend on the cellular synthetic activity; however, there was no sufficient repopulation of the grafts during the observation period of 26 weeks. Cellular ingrowth was largely limited to peripheral meniscal basis through former vascular channels along the collagen fibrils. Repopulation of the grafts slightly increased from 6 to 26 weeks, but will probably need much more time to occupy the entire graft. It is true that recellularization has recently been shown to be facilitated by removal of proteoglycans 17 ; however, in the processed grafts, the preserved superficial layer of the grafts with a highly dense network of thin collagen fibers may be the major factor that prevents the ingrowth of cells from the surface toward the inner parts of the graft. Nevertheless, the newly formed cellular layer of fibrocartilaginous tissue facing the articulating joint surfaces represent a beneficial effect for restoring a low-friction articulation, which in turn protects the graft from destruction. This may be an advantage over other artificial cell-free meniscal scaffolds, which were shown to undergo rapid degeneration and resorption within the joint.29,30
Of course, this study could not cover the analysis of all biomechanical properties of the grafts. We primarily focused on analyzing the dynamic compressive modulus of the allografts since the decellularization procedure with loss of proteoglycans primarily affects the compressive properties rather than the tensile properties. 33 Nevertheless, future studies will also have to consider gait analyses and kinematic analyses of the whole joint or extremity to evaluate potential beneficial long-term effects of meniscal replacement on the stability and related shear stress of the entire joint, which is well-known to be affected by loss of menisci.2,38
As a side effect, the transplantation of processed meniscal allografts significantly coincided with the formation of osteophytes, which even exceeded that observed following meniscectomy alone. Thus, there must factors in addition to osteoarthritis that promote the formation of these osteochondral outgrowths at the joint margins. It has been speculated if transosseous drilling channels provoke the release of pro-osteogenic growth factors from bone marrow spaces that promote osteophyte formation.39-41
Despite the lack of chondroprotective effects, this study demonstrated excellent biocompatibility, and microbiological safety of the grafts. Meniscal allografts elicited neither severe synovial inflammation nor any foreign body reactions, which emphasizes their immunological compatibility and lack of cytotoxicity. We could not detect shrinkage of the grafts, which has been reported for tissue-engineered collagen meniscus implants. 11 The processing method allows to retain the physiological collagen network, which seems to be a prerequisite for its durability in vivo. 17 The moderate degree of synovitis following LMA was comparable to that following LMX and could be ascribed to surgical trauma and the developing osteoarthritis. This may be an important advantage over nonprocessed fresh allografts. It is true that the latter was shown to be tolerated in the short term in most clinical human cases.7-10 However, they contain potential immunogenic cells presenting class-I and class-II human leukocyte antigens. 42 In the short term, these antigens may be hidden within the matrix, but may later on be exposed due to degeneration and may be responsible for the induction of immunological responses. Indeed, histological analysis following transplantation of human fresh nonprocessed allografts detected immunoreactive lymphocytes within the synovial membrane and even within the allograft tissue itself. 16
In conclusion, this pilot study demonstrated excellent biocompatibility and durability of processed meniscal allografts in vivo. However, the inferior biomechanical properties have hampered chondroprotective effects on the articular cartilage in this sheep model for osteoarthritis. Future development will include modified processing protocols with retention of the proteoglycan-rich extracellular matrix to preserve the physiological material properties and methods to improve cellular repopulation, such as the incorporation of chemotactic agents, increasing porosity of the collagen matrix, or precultivation of the grafts with autologous cells.
Footnotes
Acknowledgments and Funding
We thank Melanie Pflügner for expert technical assistance. This work was supported by the German Research Foundation (Grant WE 4881/1-1).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from the Institutional and Governmental Review Board for Animal Studies (Ref. 54-2532.1-27/09).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
