Abstract
Objective
The efficacy and safety of BST-CarGel®, a chitosan scaffold for cartilage repair was compared with microfracture alone at 1 year during a multicenter randomized controlled trial in the knee. This report was undertaken to investigate 5-year structural and clinical outcomes.
Design
The international randomized controlled trial enrolled 80 patients, aged 18 to 55 years, with grade III or IV focal lesions on the femoral condyles. Patients were randomized to receive BST-CarGel® treatment or microfracture alone, and followed standardized 12-week rehabilitation. Co-primary endpoints of repair tissue quantity and quality were evaluated by 3-dimensional MRI quantification of the degree of lesion filling (%) and T2 relaxation times. Secondary endpoints were clinical benefit measured with WOMAC (Western Ontario and McMaster Universities Osteoarthritis Index) questionnaires and safety. General estimating equations were used for longitudinal statistical analysis of repeated measures.
Results
Blinded MRI analysis demonstrated that BST-CarGel®-treated patients showed a significantly greater treatment effect for lesion filling (
Conclusions
BST-CarGel® was shown to be an effective mid-term cartilage repair treatment. At 5 years, BST-CarGel® treatment resulted in sustained and significantly superior repair tissue quantity and quality over microfracture alone. Clinical benefit following BST-CarGel® and microfracture treatment were highly significant over baseline levels.
Introduction
The search for a solution to problematic articular cartilage lesions continues despite decades of orthopedic experience in the knee. None of the current repair procedures, which include bone marrow stimulation, cultured cell-based therapies, and grafting, have been studied sufficiently, particularly in the mid to long term (5-10 years), to fully understand which factors dictate longer term outcomes for this troublesome pathology.1-3 Nonetheless, the fundamental goal of any cartilage repair treatment is to avoid the progression to secondary osteoarthritis4,5 by achieving structural repair that is comparable to native hyaline cartilage, and thereby assuring long-term durability, joint function, and pain relief.
Microfracture (MFX), the
BST-CarGel® (Piramal Life Sciences, Bio-Orthopaedics Division) was therefore developed to stabilize the MFX-based blood clot by dispersing a soluble polymer scaffold containing chitosan throughout whole blood and implanting the mixture over marrow access holes in a cartilage lesion. Chitosan is an abundant glucosamine polysaccharide found in the exoskeleton of crustaceans and has many desirable biomaterial properties.20-22 BST-CarGel® is prepared as a cytocompatible liquid chitosan solution with physiological pH, 23 which does not interfere with normal whole blood coagulation, but quantifiably reinforces the implanted clot by impeding its retraction. 19 By physically maintaining critical blood factors above marrow access holes,14,19,24 BST-CarGel® has been shown to increase the quantity and the quality of repair cartilage14,19,25,26 as a result of specific modifications in the repair sequence compared with bone marrow stimulation alone.24,27,28
An international, multicenter randomized controlled trial (RCT) comparing BST-CarGel® treatment of symptomatic cartilage lesions of the femoral condyle to MFX alone was conducted. 26 The primary efficacy analysis was based on the co-primary endpoint of repair cartilage quantity and quality at 1 year as measured by quantitative 3-dimensional MRI, and a secondary endpoint was clinical benefit measured with the Western Ontario and McMaster Osteoarthritis Index (WOMAC; visual analogue scale [VAS] version), 29 questionnaire. The 1-year RCT was used as the basis for BST-CarGel® marketing approval in Canada and Europe and was continued under an extension protocol for long-term follow-up for 5 years posttreatment.
Materials and Methods
The full description of the methodology used in the original 1-year multicenter RCT (https://clinicaltrials.gov; #NCT00314236) has been reported previously, 26 including patient eligibility criteria, descriptions of randomization, surgical treatment, and rehabilitation, as well as outcome measures, including details of the 3-dimensional quantitative MRI analyses and self-administered questionnaires. The same follow-up procedures and outcome methodologies were applied in the 5-year extension protocol (https://clinicaltrials.gov; #NCT012-46895) and performed in accordance with guidelines for Good Clinical Practice.
Study Design and Participants
The initial 1-year trial 26 enrolled 80 patients at 26 clinical sites. Eligible male and female patients were 18 to 55 years old with a single, focal cartilage lesion on the femoral condyles and moderate knee pain (>4 on a 10 cm VAS). Patients were randomized (1:1) to receive BST-CarGel® treatment or MFX alone, and agreed to follow a 12-week standardized posttreatment rehabilitation program.
The trial was single-blind since the independent third party carrying out the analyses of primary endpoints was unaware of patient treatment. Investigators and patients were not blinded because of differences in incision size related to treatment. The extension protocol was originally designed to provide longer term follow-up at 2, 3, 4, and 5 years and followed identical outcome measures. All subjects who participated in the initial 1-year trial were asked to provide written informed consent prior to study activities to be part of this extension study, which was approved by the institutional review boards at each of the clinical sites prior to initiation of activities.
Outcome Measures
Primary Outcome
Repair tissue structure, defined as both the quantity and quality of new tissue, was assessed as the primary outcome. Standardized 1.5-T MRI scans were obtained for each patient with dedicated transmit–receive knee coils at prequalified and trained MRI clinics for the initial trial at pretreatment, 1 month posttreatment, and 1 year posttreatment, and for the extension study, at enrolment and 2, 3, 4, and/or 5 years depending on individual patient follow-up status. Customized high–spatial resolution pulse sequences specific for morphological or T2 relaxation time analyses of regions of interest were used. For morphological analyses of cartilage, cartilage lesions and bone, both coronal and sagittal 3-dimensional fat-suppressed spoiled gradient echo (SPGR), and sagittal 3-dimensional gradient echo (GRE) sequences were used. Sagittal fat-suppressed dual echo fast spin echo sequences were used for transverse relaxation time (T2) analyses. All blinded scans were sent to imaging core labs for centralized scan quality review and storage (VirtualScopics, Rochester, NY) and blinded quantitative analysis (Qmetrics Technologies, Rochester, NY) using validated techniques. The quantification of lesion and repair biomarkers used proprietary, semiautomated (radiologist-corrected) morphological segmentation with a programmed anatomical atlas for all knee bone and cartilage structures. A musculoskeletal radiologist with expertise in cartilage repair manually traced the lesion boundaries on the 1-month posttreatment scan, which provided the reference for co-registration with 1-, 3-, 4-, and 5-year scans. Debrided lesions quantified using 1-month posttreatment scans represented baseline values for lesion surface area and volume. The segmented 3-dimensional volume of new tissue at each time point was then evaluated for %Fill and T2 relaxation time. (Further details and illustrations in Stanish
Secondary and Tertiary Outcomes
Clinical benefit was evaluated as a secondary outcome at initiation, 2, 3, 4, and 5 years posttreatment using the WOMAC questionnaire consisting of 3 subscales: Pain, Stiffness, and Physical Function.
Safety was assessed through recording of all adverse events (AEs) up to 5 years posttreatment. The safety definitions used during this trial conformed to international regulatory norms for clinical trials investigating medical devices.
The tertiary endpoint was the Medical Outcomes Study 36-Item Short-Form Health Survey version 2 (SF-36), 30 which includes 2 aggregate measures, the physical and mental components, derived from 8 subscales.
All questionnaires were provided to patients during on-site study visits or by mail as needed.
Statistical Analysis
Sample size determination for the 1-year trial was previously reported. 26 All treated participants who enrolled in the extension study were included in the efficacy analyses, which were performed according to a preapproved statistical analysis plan. General estimating equations were used for longitudinal analysis of repeated correlated measures using baseline (1 month posttreatment) lesion volume as a prespecified covariate for lesion %Fill and T2 MRI parameters. The longitudinal models for change from baseline for WOMAC and SF-36 comparisons were adjusted for baseline values.
To account for potential enrolment bias, patients who did not enroll in the extension study were compared with those who enrolled, looking at baseline demographic variables of gender, smoking status, previous arthroscopies, activity level, age, body mass index (BMI), number of physiotherapy sessions after study treatment, and treated lesion size as well as 1-year MRI outcome variables of lesion %Fill and mean T2 MRI values. The bootstrap method was used to account for sample size differences.
Data were analyzed using the Statistical Analysis System software (version 9.3, SAS Institute, Cary, NC). All reported
Results
Enrollment and Baseline Characteristics of the Patients
Screening and enrollment for the initial 1-year trial took place from May 2006 to January 2009, and 1-year follow-up was concluded in February 2010. Screening and enrollment into the extension study took place from March 2011 to October 2013 and the 5-year follow-up was concluded in February 2014. The extension study suffered patient loss to follow-up at all planned time points due to several factors, including extremely protracted enrolment periods for both the initial 12 month trial and the extension study, compounded by financial bankruptcy of the original trial sponsor (BioSyntech Canada Inc.) and a period of transition for the current trial sponsor (Piramal Life Sciences, Bio-Orthopaedics Division). The delayed initiation of the extension study regretfully allowed all but 4 patients to surpass their 2-year follow-up time point, and many others to pass their 3-, 4-, and 5-year time points prior to enrolling into the extension study. Ultimately, a total of 67 (84%) of the initial 80 patients consented to participate in the extension study, but their enrolment was distributed across the 5-year study period, and data were available from only 4 patients (5%) at 2 years, 32 patients (40%) at 3 years, 47 (59%) at 4 years, and 60 (75%) at 5-year follow-up. Only 2 patients had complete data for 1, 2, 3, 4, and 5 years. Because of excessively small group sample sizes, analyses in this report are thus limited to patients with 5 year follow-up data, which for BST-CarGel® and MFX, respectively, included 33/41 and 26/39 for %Fill measurements, 29/41 and 22/39 for T2 relaxation times, and 33/41 and 26/39 for all 3 WOMAC subscales. A statistical comparison of baseline characteristics of those patients who did not enroll into the extension enrolment with those who did, found that enrolled patients had significantly higher BMIs, larger treated lesion areas, and attended more posttreatment physiotherapy sessions than those that did not enroll, but were similar for all other parameters.
Baseline demographic characteristics of the patients with 5-year data were generally similar between treatment groups (
Table 1
), with the exception that BST-CarGel® patients compared with MFX patients were significantly younger (34.3 ± 9.7 vs. 40.0 ± 10.0 years;
Baseline Characteristics of Patients with 5-Year Data. a
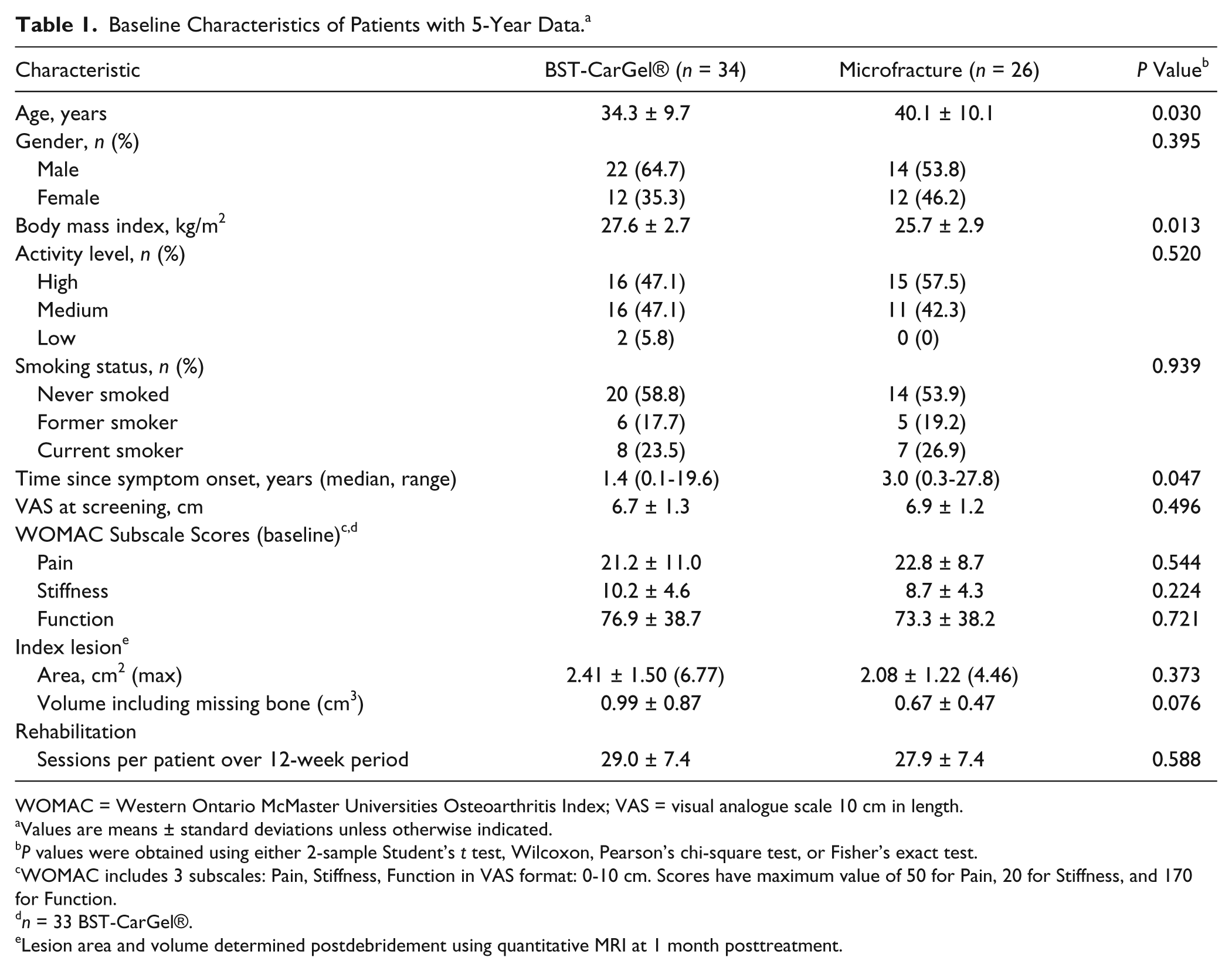
WOMAC = Western Ontario McMaster Universities Osteoarthritis Index; VAS = visual analogue scale 10 cm in length.
Values are means ± standard deviations unless otherwise indicated.
WOMAC includes 3 subscales: Pain, Stiffness, Function in VAS format: 0-10 cm. Scores have maximum value of 50 for Pain, 20 for Stiffness, and 170 for Function.
Lesion area and volume determined postdebridement using quantitative MRI at 1 month posttreatment.
Primary Outcomes
BST-CarGel®-treated patients showed a significantly greater treatment effect over the 5-year follow-up (
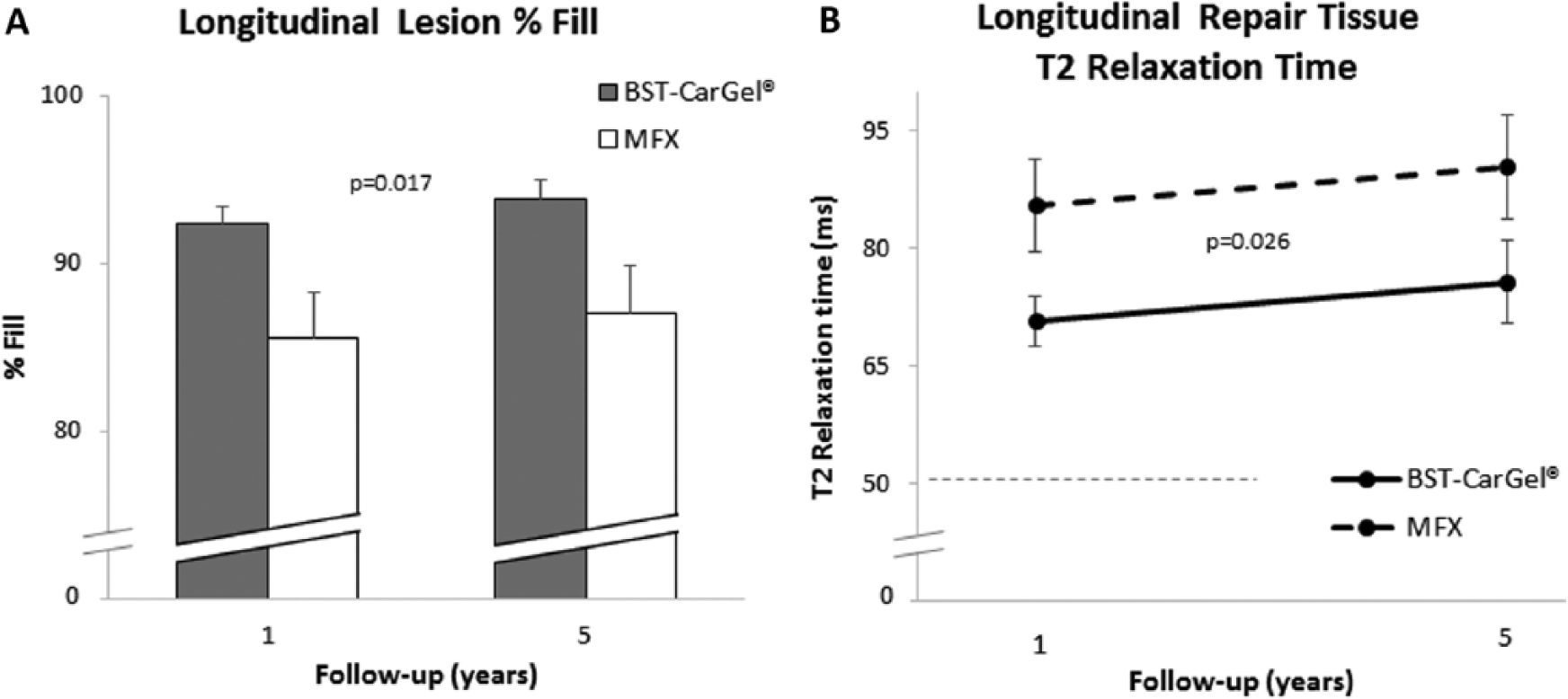
Longitudinal analysis of repeated measures using general estimating equations (GEE) for the quantity and quality of repair cartilage over 5 years posttreatment. Values represent least squares means adjusted for lesion volume and the standard error. (
Model-Based Primary, Secondary, and Tertiary Outcomes at 5 Years. a
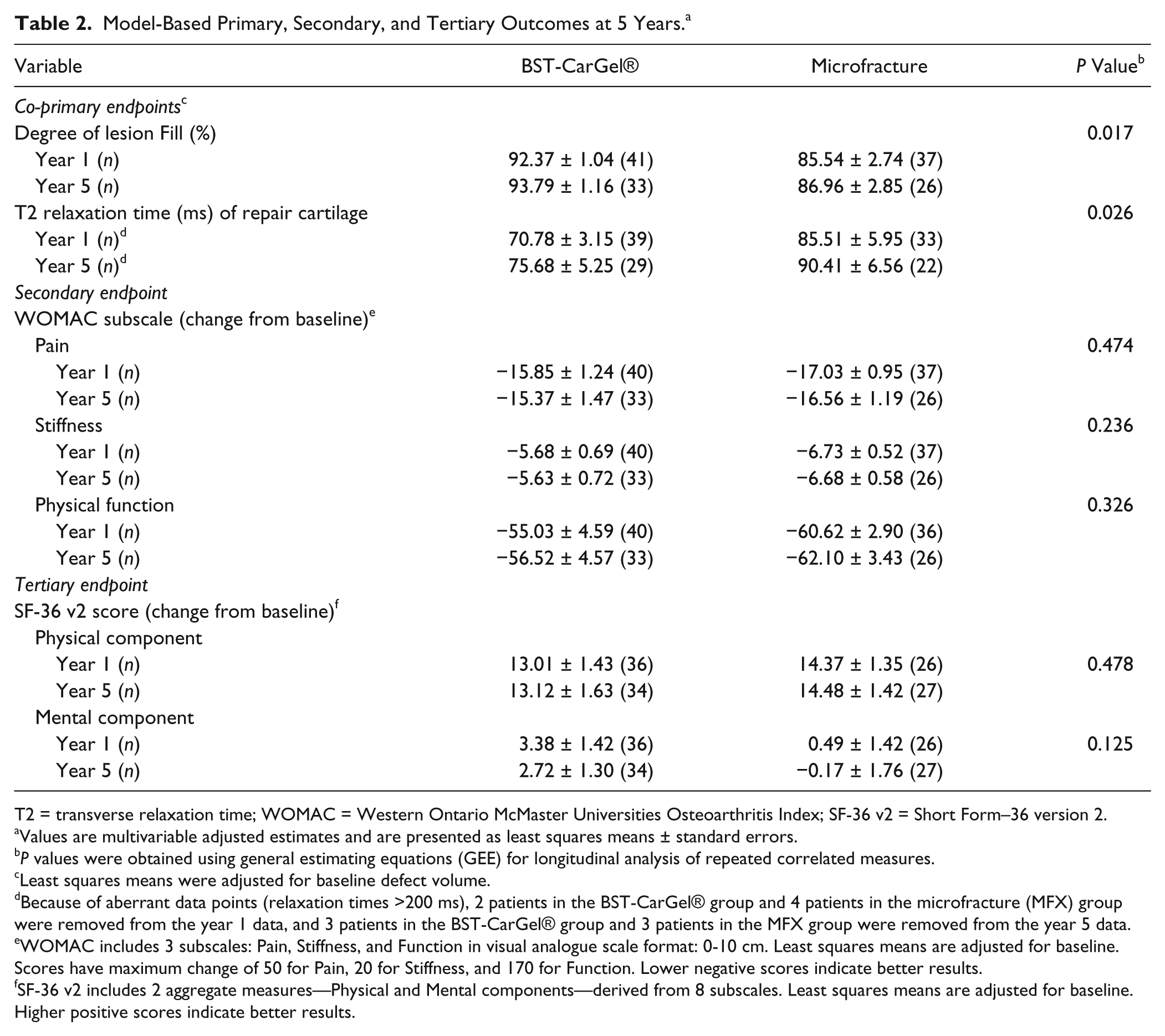
T2 = transverse relaxation time; WOMAC = Western Ontario McMaster Universities Osteoarthritis Index; SF-36 v2 = Short Form–36 version 2.
Values are multivariable adjusted estimates and are presented as least squares means ± standard errors.
Least squares means were adjusted for baseline defect volume.
Because of aberrant data points (relaxation times >200 ms), 2 patients in the BST-CarGel® group and 4 patients in the microfracture (MFX) group were removed from the year 1 data, and 3 patients in the BST-CarGel® group and 3 patients in the MFX group were removed from the year 5 data.
WOMAC includes 3 subscales: Pain, Stiffness, and Function in visual analogue scale format: 0-10 cm. Least squares means are adjusted for baseline. Scores have maximum change of 50 for Pain, 20 for Stiffness, and 170 for Function. Lower negative scores indicate better results.
SF-36 v2 includes 2 aggregate measures—Physical and Mental components—derived from 8 subscales. Least squares means are adjusted for baseline. Higher positive scores indicate better results.
A significantly greater treatment effect over the 5-year follow-up period (
Secondary and Tertiary Outcomes
BST-CarGel® and MFX groups both showed significant improvement from pretreatment baseline at 5 years for all 3 WOMAC subscales (
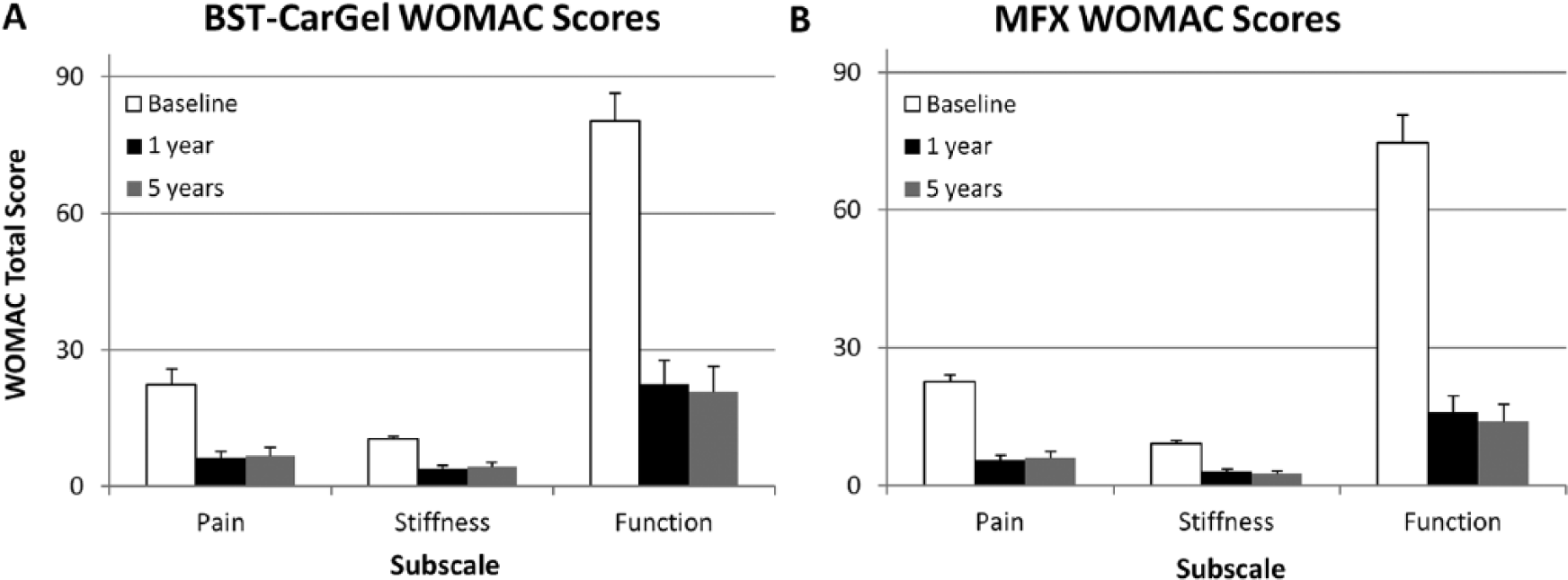
Improvement in clinical outcomes following (
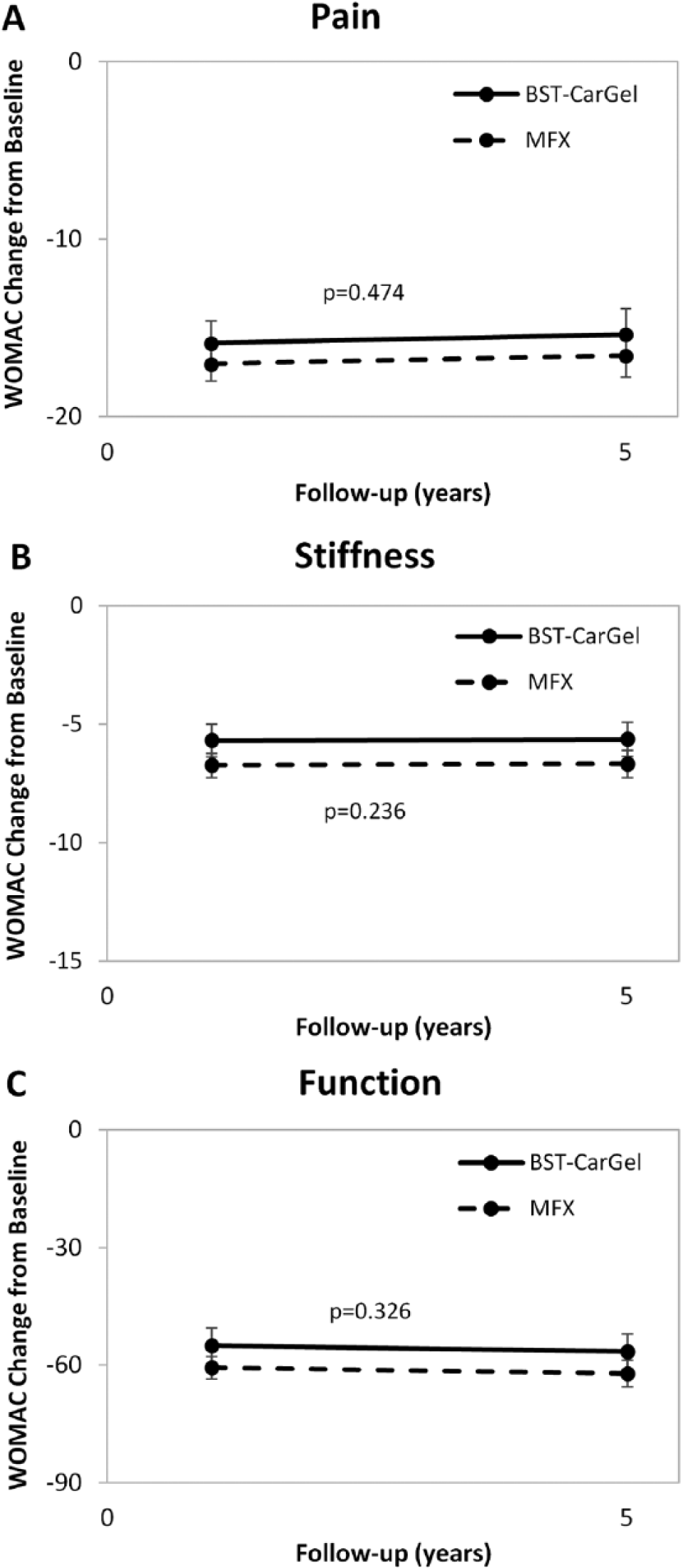
Longitudinal analysis of repeated measures using general estimating equations (GEE) for Western Ontario McMaster Universities Osteoarthritis Index (WOMAC) scoring over 5 years posttreatment. Values represent mean change from baseline adjusted for baseline and standard errors. Similar improvements for both BST-CarGel® and microfracture (MFX) groups were observed for subscales (
For the assessment of quality of life, the improvement of the physical and mental components of the SF-36 following BST-CarGel® treatment was maintained over 5 years and there were no statistically significant differences between the BST-CarGel® and MFX groups from 1 to 5 years (
Table 2
). However, MFX patients demonstrated worse mental component scores that fell below baseline at 5 years compared with BST-CarGel® patients (
Safety
Overall, both trial treatments were well tolerated and the safety profiles were considered comparable. During the 5-year follow-up period, 54 AEs were reported in 31 individual patients, 13 (19.4%) BST-CarGel® patients and 18 (26.9%) MFX patients. Most AEs (>90%) were considered mild to moderate in severity. The most frequently observed AE in both groups was knee pain (11% vs. 17% for BST-CarGel® and MFX, respectively). For the BST-CarGel® group, 2 unanticipated procedure-related AEs occurred for 1 patient (1.5%) and 1 anticipated AE was reported by 1 patient (1.5%), all that were mild to moderate in severity and ongoing at the 5-year follow-up. For the MFX group, 2 anticipated procedure-related AE occurred for 2 patients (3%) which were mild in severity and ongoing at the 5-year follow-up. No unanticipated AEs were reported for the MFX group. For the BST-CarGel® group, 2 unanticipated device-related AEs occurred for 1 patient (1.5%) that were mild to moderate in severity and ongoing at the 5-year follow-up. There were no anticipated device-related AEs for the BST-CarGel® group. No anticipated or unanticipated device-related AEs were reported for the MFX group. One (1) serious AE (SAE) was reported by 1 subject in the MFX group, which was moderate in severity and not related in any way to the study treatment or index knee but required surgery and radiotherapy. Limited information was released by the patient except that the SAE was ongoing at the time of the 5-year follow-up period. No patient in either treatment group was discontinued from the study because of an AE, SAE, or incident. There were no deaths over the 5-year period of the study.
Discussion
This study has demonstrated that the more voluminous, higher quality repair cartilage found at 1 year
26
following BST-CarGel® treatment of isolated full-thickness lesions was sustained over 5 years, supporting the hypothesis that BST-CarGel® increases the consistency of cartilage repair and enhances long-term structural superiority compared with MFX. Improvements in pain, stiffness, and function were highly significant over baseline (
The baseline characteristics for the patients included in this 5 year analysis were generally well-balanced, except for a few notable exceptions: (1) The BST-CarGel® patient population had higher baseline BMIs (
Unique to this trial was the new level of evidence brought by the use of validated 3-dimensional quantitative MRI, which assessed the structural outcomes of repair tissue quantity and quality over 5 years with a high level of standardization and precision not previously achieved in a Good Clinical Practice–compliant RCT for cartilage repair. The superior quantity of new repair cartilage in BST-CarGel® lesions at 5 years (93.79%) was sustained from that found for BST-CarGel® over MFX (
Quantitative MRI also demonstrated higher quality repair tissue by T2 for the BST-CarGel® group over 5 years in this study. T2 (or transverse) relaxation time is well known to be sensitive to, and highly dependent on, the extracellular cartilage matrix and particularly the collagen network structure, orientation, as well as macromolecular concentration, and tissue hydration.36-40 Since the quality of articular tissue can be assessed by the closeness of measured T2 values to that found in normal articular cartilage for the same joint compartment, 41 the significantly different and lower BST-CarGel® mean T2 values at both 1 and 5 years compared with that of the MFX group indicates an improved level of tissue quality sustained over time. This interpretation for T2 MRI and its relationship to collagen organization is substantiated by a previous statistical correlation between T2 and polarized light microscopy scoring of 38 repair tissue biopsies retrieved at 1 year posttreatment in this same study. 42 This scoring found significantly better zonal organization and collagen characteristics for the BST-CarGel® biopsies over MFX biopsies.43,44 Improved collagen content and zonal organization are necessary components for long-term durability of repair cartilage since collagen breakdown is considered to be a critical step in the progression of osteoarthritis.45,46
Improvements in pain, stiffness, and function were highly significant over baseline (
To further understand the equivalence found between the WOMAC subscales for each group at 5 years, post hoc sensitivity analysis was carried out to determine if specific patient characteristics or the structural variables of %Fill or T2 contributed to 5-year clinical outcomes. No single factor was identified that correlated with WOMAC pain, stiffness, and function. Ultimately, the determination of what factors are predictive of clinical outcome following cartilage repair will be multivariate, considering the numerous patient-specific and cartilage lesion–specific variables. Patient age,31,50,55,56 BMI,32,57 time from onset of symptoms,34,56 gender, lesion size, and location are all relevant, 58 although other procedure-based parameters such as technical aspects of the surgical treatment and postoperative rehabilitation58,59 could also play critical roles. Consequently, the correlation between repair tissue structure and clinical outcomes has been elusive. Several studies have reported relationships between structural assessments and long-term clinical outcomes, but these studies suffer from small sample sizes, statistical rigor, and subjective scoring of both the structural and the clinical components.50,56,60,61 A recent cohort study following MACI treatment used multivariate models to assess predictive factors, including a subjective MRI composite score, but no correlation with KOOS (Knee injury and Osteoarthritis Outcome Score) sports/recreation or SF-36 was identified. 58 Reviews of the topic agree that there are major inconsistencies between MRI structural outcomes, repair procedures, and their resulting clinical benefits,62,63 and that analysis of longer term studies are complicated by the potential for progression of degenerative changes in the knee or new unrelated knee pathology. Previous studies have been limited to subjective semiquantitative MRI analysis using MOCART 64 or Henderson scores 65 and none used quantitative 3-dimensional analysis for structural assessments as described in this study.
A critical objective of BST-CarGel® treatment is to reduce pain and symptoms over long term and thus offer the patient a return to normal lifestyle, which can only be assessed through patient-reported instruments. There is currently no consensus regarding the optimal patient-based instruments for outcome assessments in cartilage repair. Some have been validated but are insensitive and incomplete. 66 This trial, using the WOMAC tool, failed to demonstrate clinical differences between the 2 groups and although it is validated and designed for patients with osteoarthritis, the 3 WOMAC constructs for pain, stiffness, and function may not be responsive enough to measure important changes between these groups. Clearly, more and longer studies are required both with structural tools and patient-reported clinical measures that are specific enough to detect improvements following cartilage repair before the relationship between clinical and structural outcomes will emerge. Thus, with clinical benefit being shown at acceptable levels for most cartilage repair therapies33,34,67,68 and for significantly long follow-up periods, superiority of one therapy or technique falls to the structure of the replaced or regenerated cartilage within the lesion, such as was found in this trial. It can be easily argued that a sufficient quantity of repair cartilage with hyaline features enables appropriate articulation, biomechanical loading, and tissue metabolism, which would be necessary for long-term durability and function. Furthermore, repair tissue structure represents a reliable clinical trial endpoint since hyaline cartilage has an exquisite structure characterized by hallmark features, including collagen content and zonal organization, glycosaminoglycan, and cell population, 69 which can be easily discriminated by highly accurate quantitative measures sensitive to early changes in cartilage structure under reasonable clinical trial time frames.
The major limitation of this study was the number of patients lost to follow up (25%). As described, this was related to several factors, including financial bankruptcy, long time frames, and patient trial fatigue, although our trial was similar to others who reported 5-year results from 77% 50 and 79% 34 of trial patients. Furthermore, the trial outcomes reported here at 5 years are likely conservative estimates since 2 negative prognosticators, higher BMIs and larger lesions, were found in the enrolled patients compared with those who did not enroll in the extension study, although neither were found to be significant statistical covariates.
In summary, it has been shown that patients treated with BST-CarGel® demonstrate significant structural superiority of repair tissue quantity and quality over MFX for a period of 5 years posttreatment. The clinical benefit of BST-CarGel® is highly significant over baseline levels of pain, stiffness, and function, illustrating that BST-CarGel® is a safe and effective treatment for symptomatic full-thickness cartilage lesions.
Footnotes
Acknowledgements
The extension study was conducted by Piramal Life Sciences, Bio-Orthopaedics Division. The critical efforts of the BST-CarGel® Clinical Study Group, including clinical site investigators and sub-investigators and their research coordinators who tirelessly contributed to the study success are warmly acknowledged. The BST-CarGel® Clinical Trial Group investigators and sub-investigators were (in Canada) William Stanish (Halifax), Nicholas Mohtadi (Calgary), Peter MacDonald (Winnipeg), Robert McCormack, Jordan Leith, Patrick Chin, and Mike Gilbart (Vancouver), Stéphane Pelet, Réjean Cloutier, Jean Lamontagne, and Sylvain Belzile (Quebec City), Don Johnson and Allan Liew (Ottawa), Paul Marks (Toronto), Michel Malo, Julio Fernandes, Pierre Ranger, Jacques Desnoyers, Patrick Lavigne, and Sébastien Guimond Simard (Montreal), Paul Zalzal and Tim Deakon (Oakville), and Frank Smith (Hamilton); (in Spain) Francisco Forriol, Felipe Lopez-Oliva, Gloria Lopez, Manuel Leyes, Javier Vaquero, Diego Garcia, Santiago Bello, Alonso Moreno, and Patricia Villanueva (Madrid), Francisco Macule (Barcelona), and Antonio Maestro Fernandez (Gijon); and (in South Korea) Myung Chul Lee, Sang-Hoon Lee, and Kyoung Ho Yoon (Seoul). MRI site qualification and management was carried out by VirtualScopics (Rochester, NY), and MRI quantitative analyses were performed by Qmetrics Technologies (Rochester, NY).
Authors’ Note
This investigation was performed at 26 clinical sites in Canada, Spain, and South Korea (complete list is given in the Acknowledgments section).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: One or more of the authors received payments, either directly or indirectly (i.e. via their institution), from a third party in support of an aspect of this work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Piramal Healthcare (Canada) Ltd. was the sole funding source and was responsible for study design, data interpretation, and manuscript publication. Data collection and blinded analyses were conducted by third parties as described herein.
