Abstract
The therapeutic value of microfracture has been demonstrated in clinical patients. The rationale is that focal penetration of the dense subchondral plate exposes cartilage defects to the benefits of cellular and growth factor influx in addition to improving anchorage of the new tissue to the underlying subchondral bone and, to some extent, the surrounding cartilage. While functional outcomes have been reported, there is a paucity of data on the histological, biochemical, and molecular changes in human patients. This paper reviews 4 basic science studies of microfracture using an equine chondral defect model that gave some insight into possible mechanisms of action and also how the microfracture response could be augmented. In study I, microfracture of full-thickness chondral defects in exercised horses significantly increased the repair tissue volume in the defects at both 4 and 12 months. No adverse effects were seen. In study II, an investigation of the healing of full-thickness chondral defects during the first 8 weeks with or without microfracture demonstrated that microfracture significantly increased type II collagen expression as early as 8 weeks after treatment compared to controls; aggrecan expression was progressively increased during the first 8 weeks but was not significantly enhanced by microfracture. In study III, it was demonstrated that removal of the calcified cartilage layer provided optimal amount and attachment of repair tissue, emphasizing that careful removal of calcified layer is critical during debridement prior to microfracture. Study IV assessed the ability of IL-1ra/IGF-1 combination gene therapy to further modulate repair of microfractured chondral defects. The repair tissue in gene therapy–treated joints demonstrated increased proteoglycan and type II collagen content compared to microfracture alone.
Introduction
The goals of surgical management of articular cartilage defects in the knee are to provide pain relief and improve joint function, thus allowing patients to comfortably perform activities of daily living and potentially maintain or return to higher levels of activity. 1 Surgical options have been grouped into 3 categories: palliative (arthroscopic debridement and lavage), reparative (marrow stimulation techniques), and restorative (osteochondral grafting and autologous chondrocyte implantation). 1 The microfracture procedure is one of the marrow stimulation techniques that also include abrasion chondroplasty, 2 debridement down into subchondral bone, 3 spongialization, 4 and subchondral drilling.5,6
The arthroscopic technique of microfracture was developed and its value demonstrated in clinical patients by Steadman et al.7-11 The use of microfracture has many of the advantages associated with subchondral drilling, including focal penetration of the dense subchondral plate to expose cartilage defects to the benefits of cellular and growth factor influx, as well as improving anchorage of the new tissue to the underlying subchondral bone and to some extent surrounding cartilage,7,10,11 and its clinical benefits have been validated by other authors. 12 Clinical efficacy of the technique for articular cartilage repair in the knee has recently been subjected to an evidence-based systematic analysis (28 studies describe 3,122 patients). 13 However, while functional outcomes have been reported, there is a paucity of data on the histological, biochemical, and molecular changes in human patients. This paper reviews a number of studies in the horse examining the basic science aspects of microfracture in cartilage healing as part of a collaboration between the Orthopaedic Research Center at Colorado State University and the Steadman-Hawkins Research Foundation.
The Horse as a Model for Articular Defects in the Human Knee
In order to evaluate the equine medial femorotibial joint as a model for grade IV Outerbridge lesions
14
in the medial femoral condyle of the human knee, preliminary studies were done by the authors that included histological measurements of the thickness of noncalcified and calcified cartilage as well as subchondral bone plate in 5 locations on the femoral trochlea and medial femoral condyle of species (
Further work was then done by the second author creating full- and partial-thickness defects arthroscopically on the medial femoral condyle in equine cadaveric stifles and later examining histologically to ensure the correct depth had been achieved as well as debridement techniques (motorized and hand tools) to optimize depth of penetration as well as consistency of depth. From this work, we developed an
Study I: Short- (4 Months) and Long-Term (12 Months) Evaluation of Healing of Chondral Defects with Microfracture
The first study that was done in the horse documented an improvement in the quantity of tissue and the type II collagen content at 4 and 12 months after microfracture of full-thickness defects, through the calcified cartilage but not penetrating the subchondral bone plate.
16
The testing model was a 1-cm2 defect made arthroscopically on the medial femoral condyle of the femur. The equine medial femorotibial joint is a separate joint compartment from the lateral and femoropatellar joint; otherwise, the internal anatomy is very similar to the human knee. The arthroscope was placed into the joint using a lateral approach and a curette used to debride the cartilage down to the subchondral bone using a cranial instrument approach.
17
Defects were made without difficulty under arthroscopic control, and the angle of the 35° tapered awl (Linvatec, Largo, FL; Arthrex, Naples, FL) allowed accurate control of microfracture penetrations, which were performed in one randomly selected defect. The other defects were not microfractured and served as a control. Microfracture holes were placed 3 to 4 mm apart along the periphery of the defect and then brought in towards the center in a similar fashion to that recently described clinically in human patients.
18
The angle of the bone awl allowed easy access to all regions of the defect sites, and 1 or 2 taps of the awl with a mallet allowed correct penetration of the subchondral bone. When intra-articular fluid pressure was released, blood was seen emanating from the subchondral bone in all defects and also from the microfracture sites and treated defects. Fat droplets were also observed emanating from the microfracture sites in the femoral condylar lesions. A group of horses were sacrificed (
Horses (
Subjective gross examination at necropsy (2 independent evaluators unaware of treatment assignment) revealed that the appearance of the repair tissue filling both treated and control defects did not change between 4 and 12 months. There was significantly more repair tissue on treated (74% ± 5%) versus control (45% ± 5%) defects 16 ( Fig. 1 ). This difference was more pronounced in femoral condyles (where the articular cartilage is significantly thicker) than in radial carpal bone defects (the latter defects were made to emulate an equine lesion in the carpus as part of a larger study).
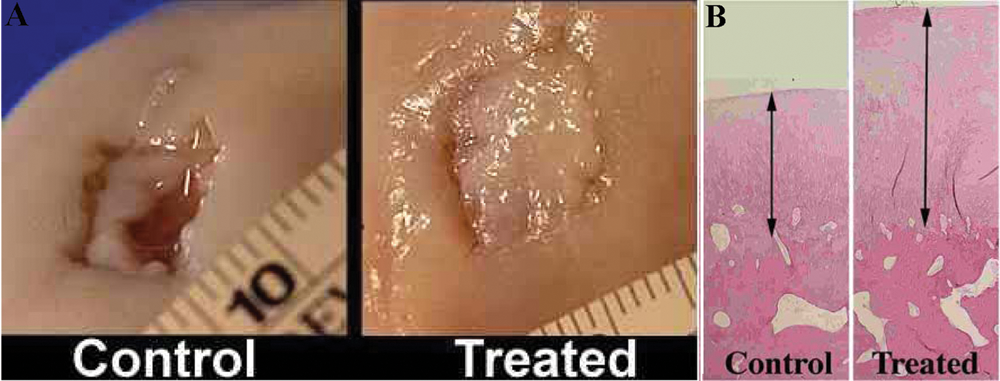
Improvement shown in the amount of tissue on (
An increase in synovial membrane vascularity scores was seen when samples were compared at 4 versus 12 months. No significant differences were seen when synovial membrane was compared between control and treated joints at either time point. There were no other significant changes in other synovial membrane indices (intimal hyperplasia, subintimal fibrosis, and edema). Using a computerized digital analysis software package (Bioquant, version 3.10; R&M Biometrics, Nashville, TN), the total area of defect, percentage of repair tissue filling defect, percentage of fibrous tissue, percentage of fibrocartilage, percentage of hyaline cartilage, and percentage of calcified cartilage that comprised the repair tissue, it was found that there was significantly more repair tissue in microfractured defects than control defects (
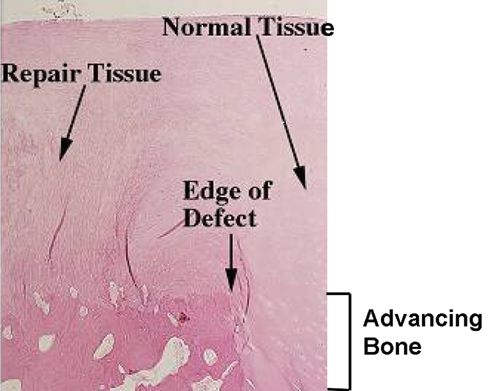
Image (10x magnification) of osteochondral section stained with H&E. Newly formed bone extends above the defect in the lower part of the repair tissue. The normal subchondral bone margin below normal tissue on the right of the diagram is just below the bottom of the picture.
Type I and II collagen ratios were assessed using gel permeation high-performance liquid chromatography (HPLC) of the peptides released by a cyanogen bromide digestion of the tissues. More type II collagen was found in femoral condylar defects versus distal radial carpal bone defects; more type II collagen was found in samples at 12 months compared to 4 months and in repair tissue from microfractured femoral condylar defects at 4 months but not at 12 months ( Fig. 3 ) when compared to nonmicrofractured control defects.
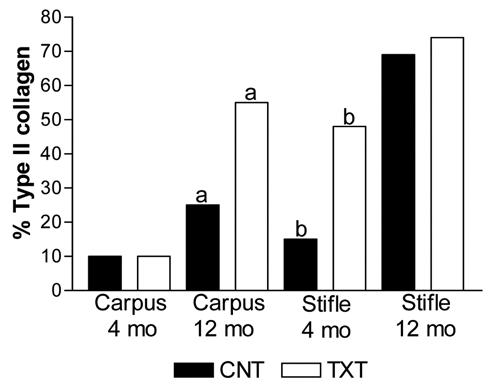
Percentage of repair tissue staining positive for type II collagen plotted by treatment group, joint, and time period in repair tissue following microfracture compared to controls. Like letters represent statistical difference (
Study II: Early Events in Cartilage Repair after Subchondral Bone Microfracture
This study investigated healing of large full-thickness articular cartilage defects during the first 8 weeks with or without subchondral bone microfracture in the previously described equine model of cartilage healing.
20
Cartilage defects of the weightbearing portion of the medial femoral condyle were made bilaterally; one defect in each horse was microfractured, whereas the contralateral leg served as the control. Repair tissue was examined histologically and for expression of cartilage extracellular matrix components (type I and II collagen and aggrecan) using reverse transcriptase coupled polymerase chain reaction (rtPCR),
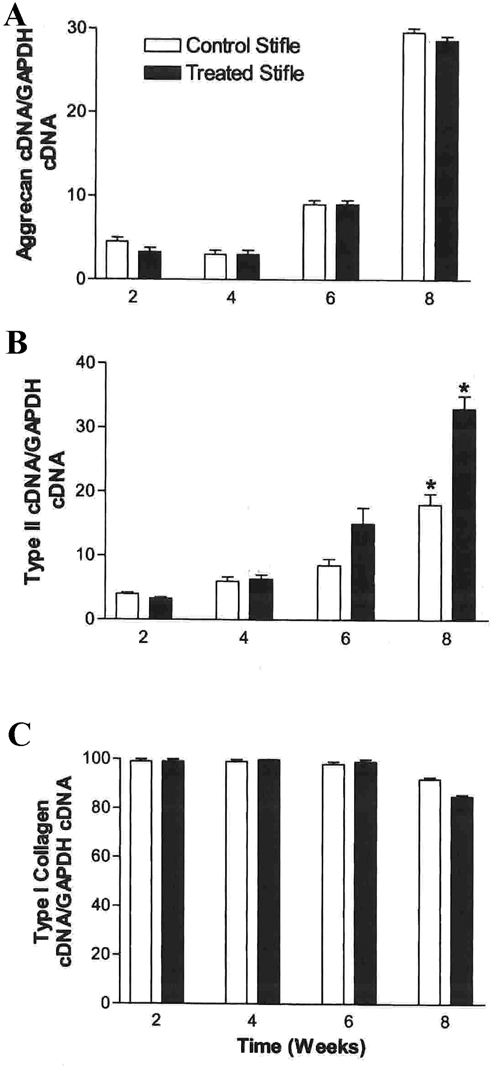
The results of reverse transcription coupled polymerase chain reaction (rtPCR) of repair tissue for key extracellular matrix components: (
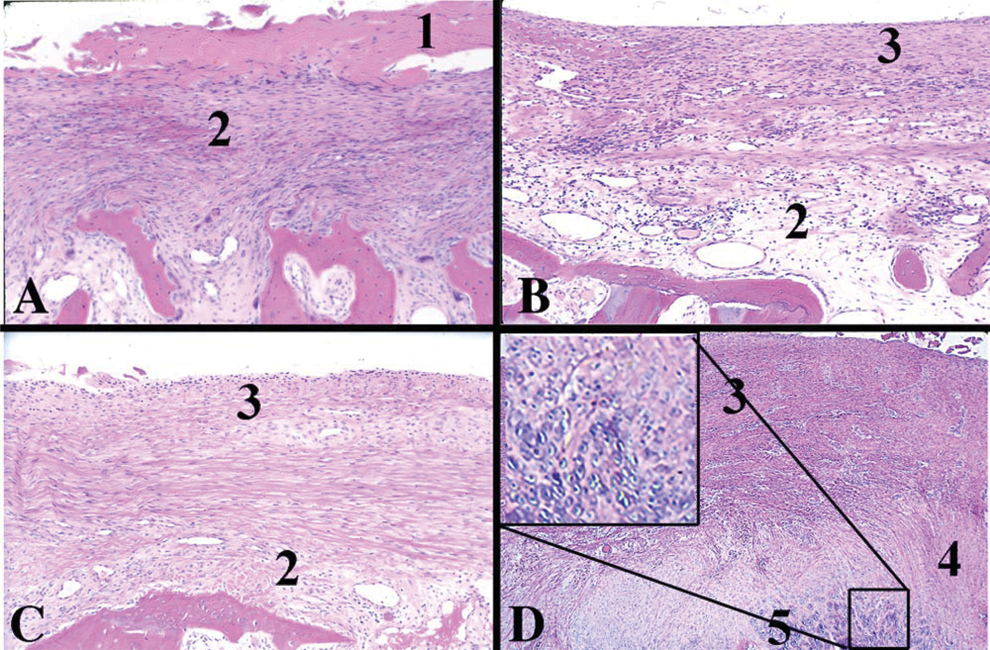
Changes in repair tissue in early time periods. Photomicrographs of osteochondral sections stained with H&E are shown, including repair tissue in the underlying bone at (
Study III: Effects of Calcified Cartilage Removal or Retention on Healing of Chondral Defects Treated with Microfracture
During study I, it was noted that if there was retention of any calcified cartilage, inferior healing was obtained. This finding had also been reported in the dog as well as earlier equine studies from the Orthopaedic Research Center at Colorado State University, but no controlled work had been done to define the actual difference. Hence, a third study was done with the hypothesis that removal of the calcified cartilage with retention of subchondral bone enhances the amount of attachment of the repair tissue compared with retention of the calcified cartilage layer (CCL). This was a randomized, blocked experimental study, again involving 1-cm2 articular cartilage defects made in 12 skeletally mature horses on the axial weightbearing portion of both medial femoral condyles. 21 Using a custom measuring device and direct arthroscopic observation of the subchondral bone beneath the calcified cartilage layer, we either removed or retained the calcified cartilage layer in one defect of each horse. The repair was assessed with arthroscopy, clinical and radiographic examinations, biopsy at 4 months, gross and histopathological examinations at 12 months, as well as mRNA immunohistochemical evaluations ( Fig. 6 ). The horses were exposed to the same athletic exercise regimen as described in study I.
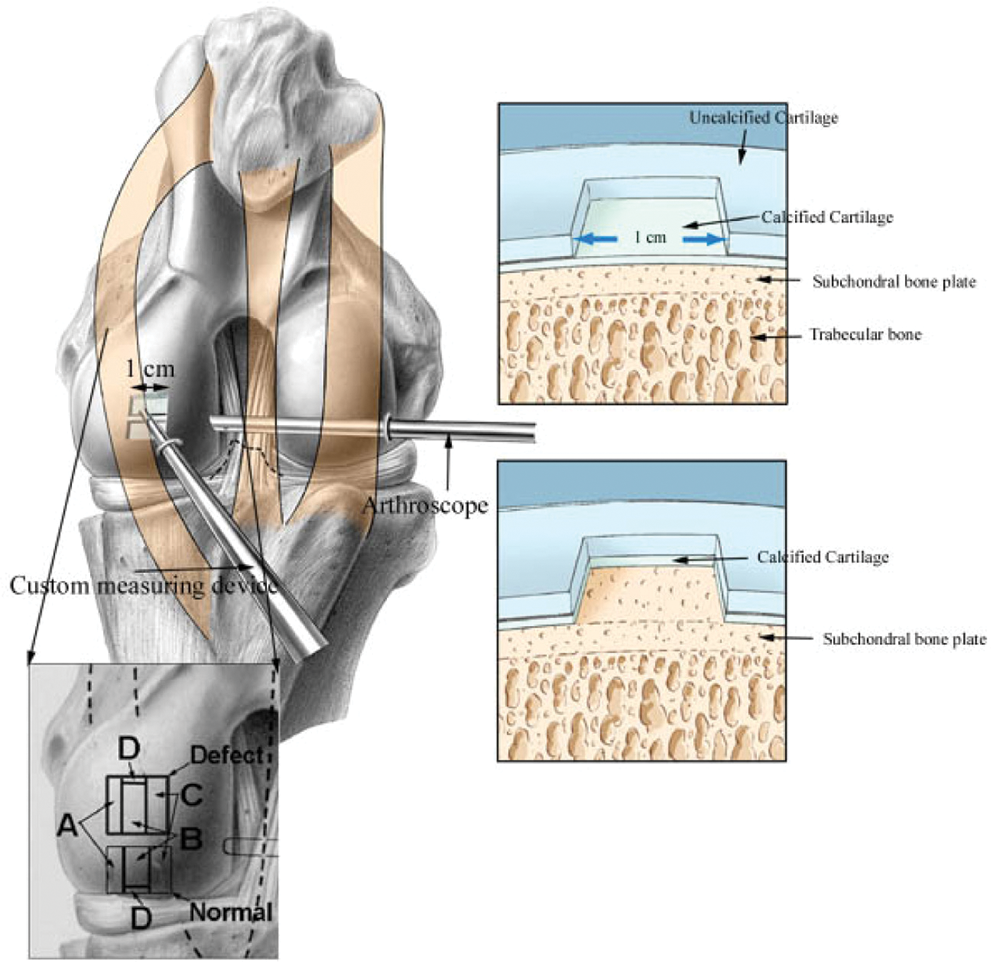
Schematic diagram demonstrating the anatomical location of the defect, defect creation, level of defect debridement, and sampling sites on the medial femoral condyle. Inset depicts respective areas within the defect and adjacent normal cartilage where tissue was collected for immunohistochemistry (
At 4 months under arthroscopic visualization, the overall repair tissue grade was significantly improved for defects that had the CCL removed compared to when the CCL remained (2.1 ± 0.2 v. 1.4 ± 0.2;
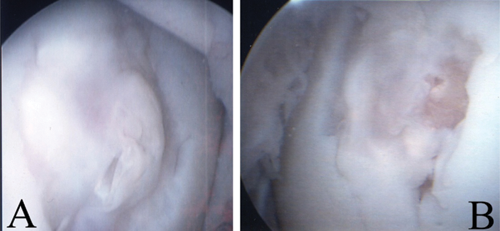
Arthroscopic images of repair tissue filling defects 4 months after surgical creation. Plate
At 12 months after defect creation, the mean percentage repair tissue area filling the defects (as assessed histologically) was significantly greater (
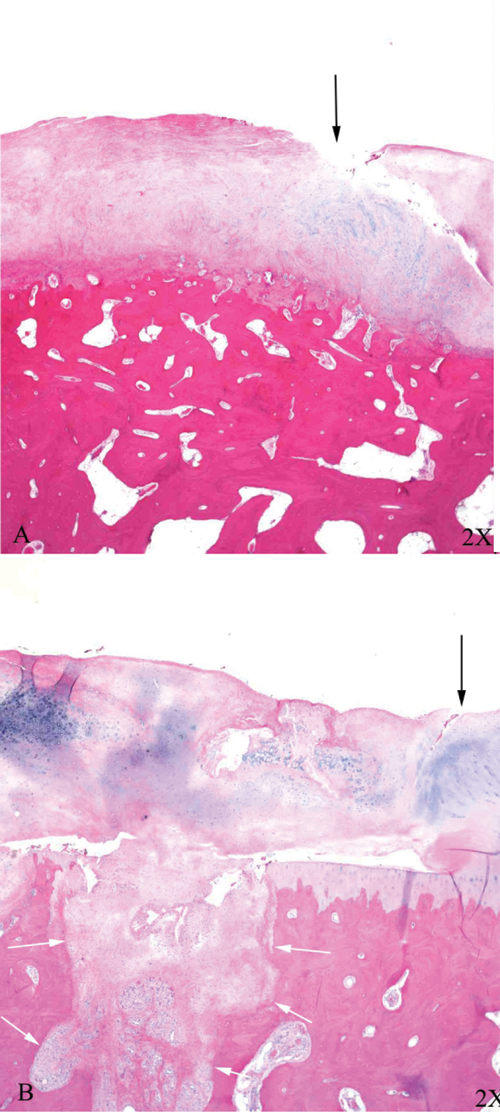
Histological specimens of defects from subject 23 stained with H&E with calcified cartilage removed (
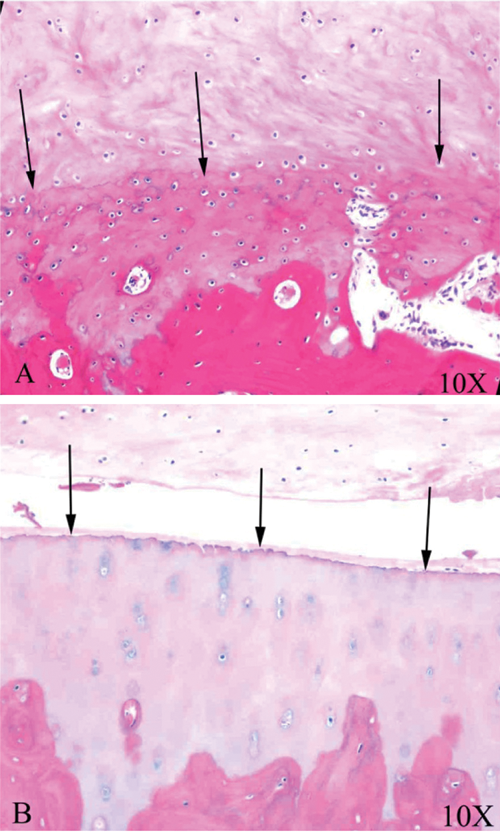
Histological specimens from subject 23 stained with H&E of defects with calcified cartilage removed (
Study IV: Assessment of IL-1ra/IGF-1 Gene Therapy to Modulate Repair of Microfractured Chondral Defects
This most recently published study was based on the premise that repair of cartilage defects involves participation of specific hormones and growth factors along with potential impairment by inflammatory cytokines.
22
We explored an
We found, following treatment with IL-1Ra and IGF-1 using gene therapy compared to untreated joints, a significantly increased proteoglycan content in treated defects along with significantly augmented type II collagen associated with substantial transgene expression of IL-1ra during the first 3 weeks. Although transgene expression of IL- 1ra was evident for 3 consecutive weeks, IGF-1 concentrations in synovial fluid were increased in all joints (including controls) without differences in levels between the 3 treatment groups. No macroscopic and histological treatment differences in defect healing were detected between the 2 treatment groups. The most important findings of the combined expression of IL-1ra and IGF-1 to modulate cartilage repair were the increased amounts of proteoglycan and type II collagen present in the repair tissue of the treated defects. It showed that this gene therapy protocol could further augment endogenous healing associated with subchondral microfracture. However, contrary to IL-1ra, we were unable to ascertain that increases in IGF-1 concentrations originated solely from transgene expression because increases were observed in all groups. This study was the first to report modulation of proteoglycan content through an
Summary
Basic research studies of microfracture in the horse have shown 1) microfracture of full-thickness chondral defects in exercised horses significantly increased the repair tissue volume at both 4 and 12 months, 2) microfracture significantly increased type II collagen mRNA expression as early as 8 weeks after treatment compared to full-thickness defects without microfracture; aggrecan expression was progressively increased in the first 8 weeks but was not significantly enhanced by microfracture, 3) removal of the calcified cartilage layer provided optimal amount of repair tissue as well as attachment within the defect, emphasizing that careful removal of the calcified cartilage layer is critical during debridement prior to microfracture (and this debridement requires curettage), and 4) the quality of repair of microfractured chondral defects could be further enhanced with intra-articular IL-ra/IGF-1 combination gene therapy.
Footnotes
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The authors received funding from the Steadman-Hawkins Research Foundation for studies I and IV that are reported in this article.
