Abstract
Objective
The limits of the microfracture (MFX) treatment in terms of lesion size and long-term tissue functionality makes it necessary to investigate different alternatives to repair focal cartilage lesions. The present study aims at evaluating the efficacy of a minimally invasive approach against the conventional MFX to repair a chondral defect in rabbits. An injectable scaffold of BMP-2 pre-encapsulated in PLGA microspheres dispersed in a Pluronic F-127 solution is proposed as support of cells and controlled delivery system for the growth factor.
Design
MFX was compared versus the injectable system seeded with mesenchymal stem cells (MSCs), both without BMP-2 and under controlled release of BMP-2 at 2 different doses (3 and 12 µg/scaffold). The different treatments were evaluated on a 4-mm diameter chondral defect model using 9 experimental groups of 4 rabbits (8 knees) each, throughout 24 weeks.
Results
Histologically, all the treated groups, except MFX treated, responded significantly better than the control group (nontreated defect). Although no significant differences were found between the treated groups, only BMP(12), MSC-BMP(12), and MFX-BMP(3) groups showed nonsignificant differences when compared with the normal cartilage.
Conclusions
The hydrogel system proposed to control the release rate of the BMP-2 was safe, easily injectable, and also provided good support for cells. Treatments with MSCs or BMP-2 repaired efficiently the chondral lesion created in rabbits, being less invasive than MFX treatment.
Introduction
Cartilage injuries are a common clinical problem and their treatment continues to be a challenge for orthopedic surgeons. Focal chondral defects are seen in up to 63% of patients undergoing arthroscopy of the knee, 1 and the overall prevalence of focal chondral defects in the knee is 36% among all athletes, according to Flanigan et al. 2
With the ageing of the population worldwide, the predisposing genetic factors and modern-life environmental factors, the onset and development of chronic degenerative diseases such as osteoarthritis will become one of the leading clinical problems faced by orthopedic surgeons.3,4 In fact, it has been forecast that 25% of the adult population, or >50 million people in the United States alone, will be affected by osteoarthritis by the year 2020. It will be a major cause of morbidity and physical limitation among individuals of over 40 years of age, with an accompanying significant financial burden. 5
Articular cartilage is a hypocellular, avascular, alymphatic tissue with a dense collagen and proteoglycan matrix that provides a low-friction and highly durable wear-resistant surface. 6 Under physiological conditions, cartilage is adapted to mechanical signals and has a very low coefficient of friction, which makes it resistant to shear and compressive forces. 7 However, the avascular nature of cartilage together with the inability of chondrocytes to migrate to the site of injury are thought to be the main reasons for the limited self-healing capacity of injured cartilage. 8 Defects that do not breach the subchondral plate do not heal spontaneously, and without treatment they usually progress onto the degeneration of the articular surface. 9
Currently, a broad spectrum of surgical options has been developed and used in clinic. 7 The ideal treatment would reestablish the low-friction properties of cartilage with the ability to resist wear over time by repopulating a lesion with chondrocytes able to produce a hyaline matrix that is fully integrated with surrounding host cartilage. This will theoretically restore the articular surface, improve joint mechanics repair, and ultimately delay or even stop osteoarthritic progression.8,10,11 The treatment must be individualized, depending on patient factors (age, activity level) and lesion aspects (size, number, and location).
One of the techniques for the treatment of chondral defects is bone marrow stimulation, based on the perforation of subchondral bone to permit chondroprogenitor cells access. The microfracture (MFX) is a reproducible single-staged, minimally invasive, and relatively cost-effective technique widely used with good short-term results, although outcomes deteriorate after 18 months after surgery.8,12
The osteochondral transfer is another procedure that consists of filling the defect with multiple osteochondral plugs harvested from the patient’s femur or from a cadaver. 8 In this case, the lack of integration with the margins of defect, donor site morbidity in autograft and chondrocyte viability, and disease transmission from allogenic tissue are the main disadvantages.8,13
Tissue engineering has provided a large variety of techniques based on seed cells, scaffolds, and growth factors. 7 The autologous chondrocyte implantation uses arthroscopically harvested chondrocytes that are cultured, resuspended, and finally implanted into the defect, and then protected by a sutured periosteal patch. 14 The autologous chondrocyte implantation–like techniques incorporate the use of growth factors in chondro-conductive or -inductive scaffolds, as well as stem cells from different sources or gene therapy, to produce chondrocytes. 8 The clinical use of stem cells from bone marrow and/or adipose tissue is promising and is gaining popularity because it is accessible, reproducible, and reduces patient morbidity when compared with the autologous chondrocytes. The role of growth factors is crucial in encouraging the differentiation of stem cells, maintaining chondrocyte phenotype, and promoting collagen type II synthesis.15,16 In fact, the use of BMP in cartilage injuries is still controversial, but its osteogenic and chondrogenic activities are well documented in the literature.17,18
Supported by the previous experience of our group in the use of BMP-2 in treatment of osteochondral defects, we have designed a study based on an animal model to investigate the cartilage repair in a focal chondral defect with a minimal invasive approach. We hypothesized that an adequate defect environment, provided by implanted cells, guided and controlled use of growth factors (BMP-2), timely correlated from a scaffold that mimics the architecture and composition of native tissue may achieve in situ cartilage repair. An injectable scaffold of PLGA microspheres dispersed in a Pluronic F-127 solution is proposed as a support and delivery system for growth factor and cells. This study compares the chondral defect repair in rabbits on the practice of MFX, as a well-defined and extended treatment, versus a mesenchymal stem cells (MSCs)-seeded injectable system, both without BMP-2 and under controlled release of BMP-2 at 2 different doses (3 and 12 µg/scaffold) pre-encapsulated in PLGA microspheres.
Materials and Methods
Material was processed under aseptic conditions and all products except BMP-2 were sterilized before use. The poly(lactic-co-glycolic acid) (PLGA) used was Resomer RG504 (Evonik, Darmstadt, Germany). Pluronic F-127 was purchased from Sigma-Aldrich (St. Louis, MO). The BMP-2 (lot: B2BCO3.1A; Noricum, Madrid, Spain) showed an ED50 = 1.2 µg/mL measured by its ability to induce alkaline phosphatase production by C2C12 cells.
Preparation and Characterization of the Microsphere Suspension
Microspheres were prepared by a double emulsion (w/o/w) method 19 modified to reduce the microsphere size, 90 µg or 360 µg of BMP-2, depending on the dose to be administered, dissolved in 200 µL of an aqueous solution of 1% poly-vinyl alcohol (PVA) were vortexed with 2 mL of a PLGA methylene chloride solution (50 mg/mL) for 3 minutes. Then, to obtain small particles, the emulsion was poured into 100 mL of a 0.5% PVA (w/v) solution under stirring at 1000 rpm for 1 minute (Silverson Homogenizer L4RT, Chesham, UK). Afterwards, the suspension was maintained (1.5 hours) under magnetic agitation for organic solvent evaporation. Microspheres were filtrated and freeze-dried. Microspheres were observed with a scanning electron microscopy (SEM, Jeol JSM-6300, Tokyo, Japan) and sized using laser diffractometry (Mastersizer 2000, Malvern Instruments, Malvern, UK).
Microspheres (7 ± 0.5 mg) were injected dispersed in 300 µL of Pluronic F-127(1.5%) in saline solution. The suspension of microspheres loaded in the syringe was kept at 4°C during 90 minutes before administration, except when MSCs were pre-seeded in the microspheres suspension.
BMP-2 was labeled with 125I using the iodogen method as previously described 20 to determine encapsulation efficiency and suspension injectability (20-G needle). Concretely, to assess dose uniformity, 5 syringes of 1 mL were loaded with 125I-BMP-2 microsphere suspension. The initial radioactivity was measured in each syringe using a gamma counter (Cobra II, Packard, Downers Grove, IL), then the remaining radioactivity in the syringe after the unloading of each dose was monitored. The injected dose was calculated by the difference between the 2 measurements.
125I-BMP-2 microspheres were also used to carry out the in vivo release assay. 21 Briefly, the detector (Captus, Capintec Inc., Ramsey, NJ) was placed onto the defect site of 5 sedated rabbits and the remaining radioactivity was measured at 27 keV. The initial measure (time = 0) was considered the given dose (100%).
Cell Seeding
Mesenchymal stem cells were isolated as previously described, 22 from bone marrow of healthy New Zealand white rabbits of the same age (6 months) as the animals to be implanted. The isolated cells were expanded and subcultured to approximately 80% confluence, separated, and kept frozen. Before implantation, the MSCs were thawed and pre-cultured once again. All animal experiments were carried out in conformity with the E.C. (2010/63/UE) and Spanish (R.D. 53/2013) guidelines on care and use of animals in experimental procedures. Furthermore, the animal experiments were previously approved by the local committee of the University of La Laguna.
Once the microsphere suspension was loaded into the syringe, 20 µL of a cell suspension (1.25 × 107 cells/mL, passage 2) in phosphate buffer (pH 7.4) were dropped and mixed manually with the syringe plunger. Then, the preparation was incubated at 37°C and 5% CO2 under orbital stirring (100 rpm, Orbital shaking platform POS-300, Grant-bio, Grant Instruments, Cambridge, UK) for 90 minutes before administration to the animals, to allow cell adhesion. XTT colorimetric assay (Roche, Madrid, Spain) was used to check cell adhesion. Cells were incubated in calcein AM (Sigma-Aldrich, St. Louis, MO) to visualize viable cells (Zeiss, Axiovert 40 CFL inverted microscope, Jena, Germany).
Animal Surgery
The animal experimental design is detailed in Table 1 .
Experimental Groups of 4 Rabbits (8 Knees) Each for Histological Evaluation After Euthanasia at 12 and 24 Weeks Post-Treatment.
MFX = microfracture; MSC = mesenchymal stem cells.
The surgery procedure was carried out in aseptic conditions. Briefly, both hind legs (72 knees) of 36 anesthetized New Zealand male rabbits of 6 months of age (3-4 kg) were shaved and disinfected. The drugs used during the surgery and the post-surgery period were ketamine (35 mg/kg) and xylazine (5 mg/kg) as anesthetics, enrofloxacin (5-15 mg/kg) for antibiotic prophylaxis, and buprenorphine (0.05-0.1 mg/kg, s.c.) and ketoprofen (2-5 mg/kg, s.c.) for analgesia.
The cartilage defect was performed in the frontal area of the intercondylar space of distal femur (femoral trochlea), on mature, male New Zealand rabbits. Briefly, a parapatellar incision was made in the knee and a dislocation of the patella was performed to access the femoral condyles. First a circular area was delimited using a 4-mm biopsy punch. Then with the punch and a 20-G needle the cartilage was carefully removed avoiding damage to the subchondral bone and avoiding bleeding. Finally, the wound was sutured and bandaged. After recovery from the surgery (20-30 minutes), the animals were allowed free movement in their cages, food, and water. According to the different experimental groups (see Table 1 ), the microsphere suspension both with and without MSCs was administered by intra-articular injection 1 week later.
In the case of animals that underwent bone marrow stimulation, before suturing the wound, the center of the lesion was drilled to a depth of 4 mm with a 0.9 mm gauge needle coupled to a low power motor (max 300 rpm) simulating a MFX. Consequently, bleeding occurred and MSCs were expected to access the injured joint. The wound was sutured and in the selected groups (see Table 1 ) the microsphere suspension was then intra-articularly injected.
Histology and Immunohistochemistry
Nine experimental groups of 4 rabbits (8 knees) were considered for histological evaluation ( Table 1 ). In addition, in the groups of animals used as control (nontreated defect), the healthy cartilage adjacent to the defect was histologically evaluated to be used as normal cartilage reference.
The histological findings (light microscopy, LEICA DM 4000B, Leica Microsystems, Wetzlar, Germany) were scored by 2 independent evaluators using an established scoring system described by Tokuhara et al. 23 The evaluated parameters were cell morphology (scores 0-4), matrix staining (scores 0-3), surface regularity (scores 0-3), cartilage thickness (scores 0-2), integration of donor to host adjacent cartilage (scores 0-2), and thickness of subchondral bone (scores 0-4). Therefore, this system establishes a maximum total score of 18 that would correspond to a cartilage and subchondral bone of high quality.
Animals were sacrificed at 12 and 24 weeks post-treatment by anesthetic overdose. At each time, 4 samples of un-decalcified cartilage specimens were prepared for histological analysis. 24 Von Kossa and Masson-Goldner’s trichrome staining and aniline blue staining were used for cartilage repair evaluation and scoring.
The following proteins were identified at the defect sites: collagen II (Col II) for mature functional hyaline cartilage, and aggrecan, a cartilage-specific proteoglycan. Collagen X (Col X) was also identified as typical hypertrophic cartilage and endochondral ossification marker. For this purpose, sections were deplastified with anti-Col II (Millipore, Barcelona, Spain; Catalog No. AB746P) and anti-Col X polyclonal antisera (Millipore, Barcelona, Spain; Catalog No. 234196) and with an anti-aggrecan monoclonal antibody (Millipore, Barcelona, Spain; Catalog No. MAB2015 clone EFG-4). Briefly, sections were deplastified and rehydrated in TBS buffer (pH 7.4, 0.1 M), which was also used for all further incubation and wash steps. The sections underwent antigen retrieval in Tris-EDTA buffer (pH 9.0, 10:1 mM) at 65°C for 20 minutes and were then blocked in FBS at 2% in a TBS-Triton X-100 solution. The indirect immunohistochemical procedure was carried out by incubating the sections overnight at 4°C with the mentioned antisera and antibody (1/100). After rinsing, sections were sequentially incubated with biotin-SP conjugated F (ab′) fragment donkey anti-rabbit (Millipore, Barcelona, Spain) (1/1000) and biotin-SP conjugated F (ab′) fragment donkey anti-mouse (Millipore, Barcelona, Spain) (1/1000) for 60 minutes and streptavidin-peroxidase complex (Millipore, Barcelona, Spain) (1/1000) for another 60 minutes. Peroxidase activity was revealed in Tris-HCl buffer (pH 7.6, 0.05 M), containing 0.04% 4-chloro-1-naphtol (Sigma, Poole, UK) and 0.01% hydrogen peroxide. Immunolabeling specificity was controlled by replacing the specific antiserum by normal serum.
Statistical Analysis
Data were statistically analyzed comparing separately the different treated groups with both the control group (nontreated defect) and with normal cartilage.
Statistical analysis was performed with SPSS.21 software using one-way analysis of variance (ANOVA) with a Tukey multiple comparison posttest. Significance was set at P < 0.05. Results are given as means ± SD.
Power analysis was performed using R software (R Foundation for Statistical Computing, version 3.5.1, 2018, Vienna).
Results
System Characteristics
The mean volume diameter of the microspheres was 55 µm, and 80% of particles were in the range of 31 to 81 µm. BMP-2 encapsulation efficiency was 70.5 ± 6.8%.
With respect to the dose uniformity, the results indicated that the injected dose was 70 ± 10% of the initial radioactivity loaded in each syringe. Therefore, the doses of BMP-2 administered to the rabbits were 3.1 ± 0.44 µg as low dose and 12.4 ± 1.8 µg the high dose.
Calcein AM staining images and XTT analysis revealed that most (95 ± 7%) of the 2.5 × 105 MSCs initially seeded were viable in the suspension of microspheres after 1.5 hours of incubation.
Finally, the BMP-2 release profile showed a high burst effect, and after 2 days of the intra-articular injection approximately 75% of the dose was already released. About 90% to 95% of the total amount of BMP-2 disappeared from the defect in 10 days, showing a mean residence time in the area of 2 days. The fast release rate might be due to the small size of the microspheres required for easy injection.
Histological and Histomorphometrical Evaluation
Histological analysis was performed at 12 and 24 weeks postsurgery.
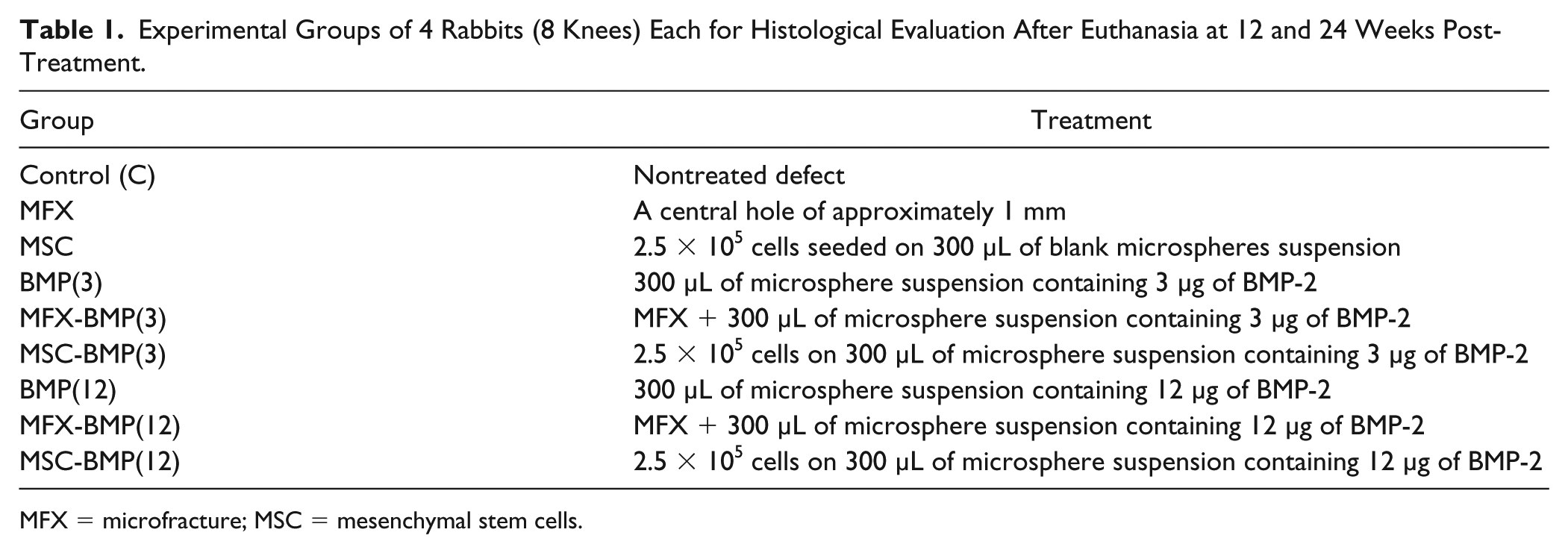
In all specimens of the control group, the defect was perfectly localized during the experimental period (Fig. 1a-d). However, mild signs of repair were seen in some animals at 24 weeks ( Fig. 1e ).
Representative images from the control group (C) 12 (
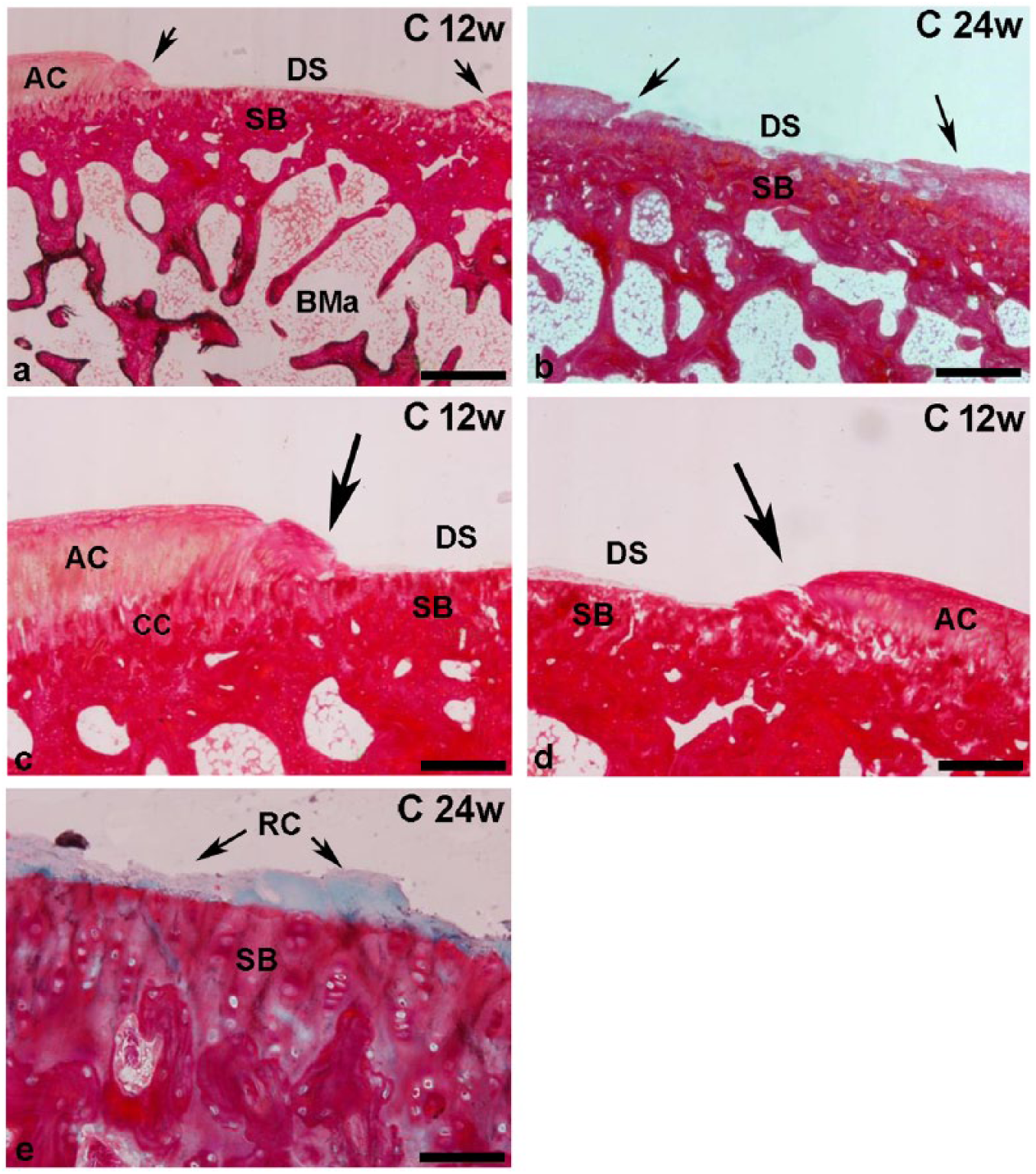
Twelve weeks postimplantation, treated animals showed signs of cartilage repair ( Fig. 2 ). A higher degree of repair was observed in animals of the BMP(12), MFX-BMP(3), MFX-BMP(12), MSC-BMP(3), and MSC-BMP(12) groups (Fig. 2f-j) compared with the MFX, MSC, and BMP(3) groups (Fig. 2c-e).
Representative images of the chondral defect showing the degree of repair in the different experimental groups (
Furthermore, in the MFX ( Fig. 3b ), MSC ( Fig. 3c ), and BMP(3) ( Fig. 3d ) groups, the defect limits were clearly observed and also the repaired tissue presented an irregular surface with extremely thin areas in comparison with the normal cartilage ( Fig. 3a ). Contrarily, the tissue observed in BMP(12), MFX-BMP(12), MSC-BMP(12) ( Fig. 3e , g , h ), and MSC-BMP(3) groups showed a thicker and more homogeneous surface than the other groups but still thinner than normal cartilage ( Fig. 3f ). Overall, most of the experimental groups showed differences in tissue architecture and cell distribution ( Fig. 3g and h ), with normal cartilage ( Fig. 3f ). Despite the fact that after 12 weeks cell density was still low, some chondrocytes looked reactive and formed clusters ( Fig. 3g and h ) that suggested active repair processes.
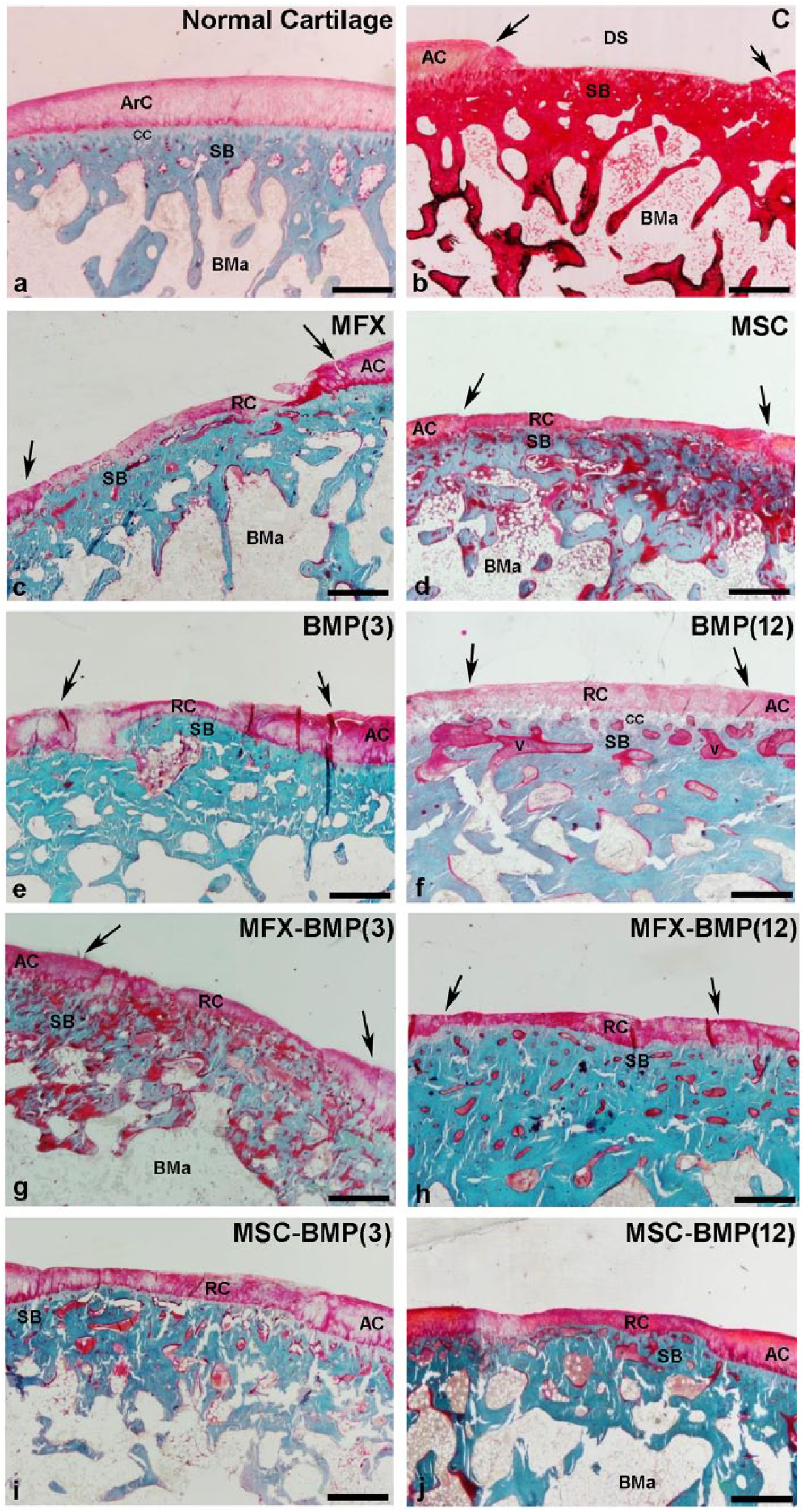
Details at different magnifications of normal articular cartilage (ArC) showing the calcified cartilage (CC) and the tidemark (arrowheads) in order to compare with the repaired cartilage in the experimental groups (
Twenty-four weeks postimplantation, the repair cartilage was more homogeneous in almost all experimental groups (
Fig. 4
). However, poor repair was observed in MFX, MSC, and BMP(3) groups, excessively thin cartilage was observed (
Fig. 4c
and
e
), thinner than normal cartilage (
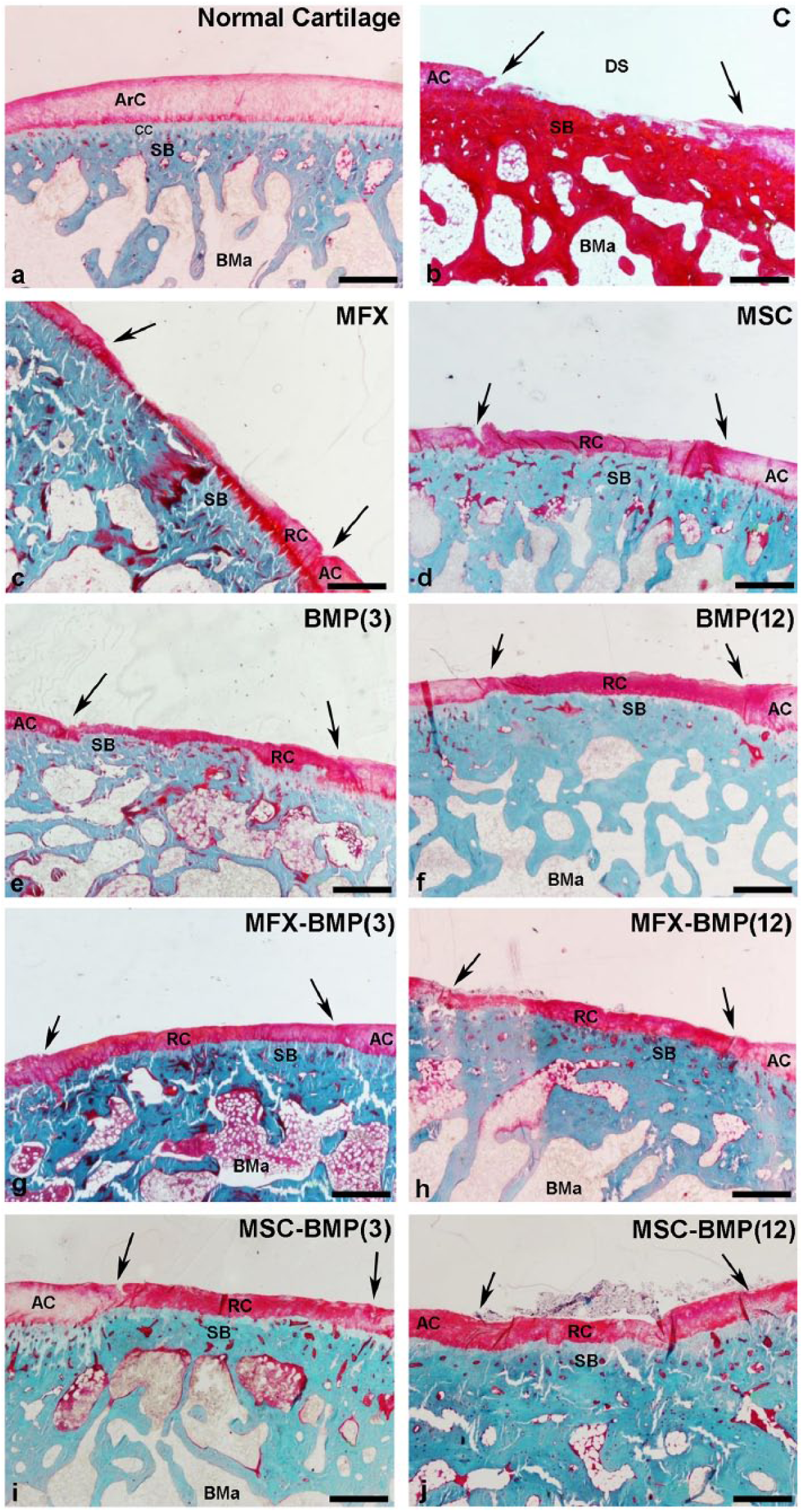
Representative images of the chondral defect showing the repair degree in the different experimental groups (
Migration of the tidemark toward the joint surface was observed in some animals ( Fig. 3d and e ) mainly 12 weeks postsurgery, which could be a consequence of ossification processes produced in the area of calcified cartilage as well as reparative phenomena that take place in the subchondral bone. The absence of articular cartilage in the area of the defect would allow the bone, which has greater proliferative capacity than the cartilage, to expand toward the articular surface occupying part of the chondral region as has been observed by other authors with different experimental models of chondral lesion.25-27
Histomorphometric analysis showed score values of 5.25 ± 2.63 in the control group while the score in the treated groups was between 8.25 ± 2.50 of the MFX group and 12.33 ± 0.58 and 12.33 ± 4.62 of the MSC-BMP(3) and MSC-BMP(12) groups, respectively, 12 weeks postimplantation. Significant differences were detected at this time point in all experimental groups except in MFX and BMP(3) groups with respect to the control group ( Fig. 5a ). Twenty-four weeks postimplantation, the scores were 6.5 ± 0.58 in the control group, and ranged from 9.25 ± 2.63 in the MFX group to 14.33 ± 0.58 in the BMP(12) group. At this time point, the score of the MFX group remained not significantly different from the control group ( Fig. 5a ). The scores obtained in the evaluation of each of the chondral parameters analyzed are reflected in Figure 5b to g .
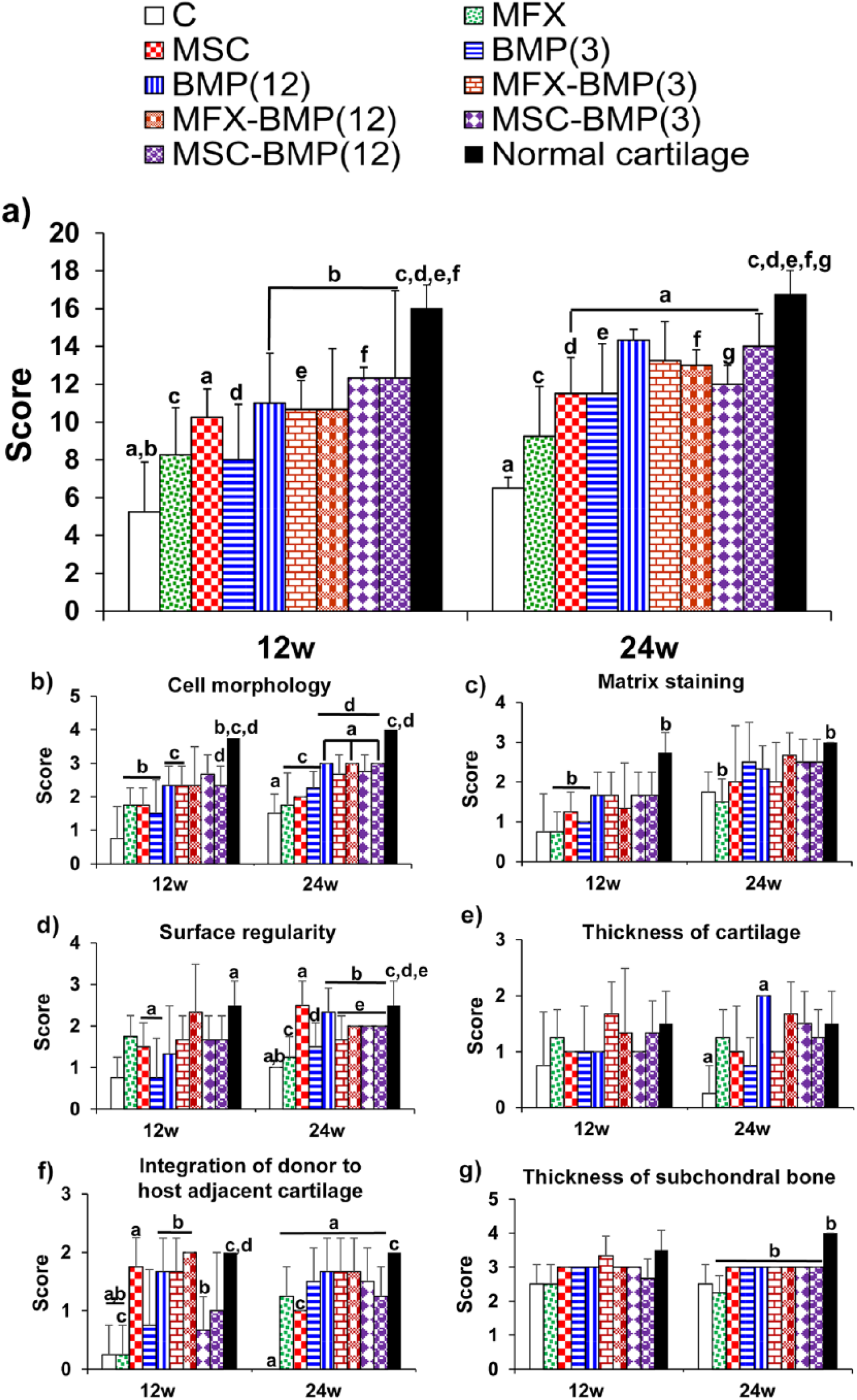
(
Normal cartilage adjacent to the defect in the control groups at 12 and 24 weeks postsurgery showed scores of 16.0 ± 1.44 and 16.75 ± 1.26, respectively. In the statistical analysis of the different treated groups with respect to the normal cartilage, nonsignificant differences between BMP(12), MSC-BMP(12), and MFX-BMP(3) groups and the normal cartilage were found.
Statistical analyses performed (9 experimental groups, sample size of N = 4) taking a significance level of 0.05 and considering the variance intra-groups (from the one-way ANOVA) gave a post hoc power analysis higher than 0.85 in all cases. The minimum effects difference to be detectable was 2.4 standard deviations. These minimum detectable effects were found around a score of 6.2 and 4.3 (maximum score 18) for the statistical analyses at 12 and 24 weeks, respectively.
To confirm the histomorphometrical data, the expression of markers of chondral maturity and stability was determined 24 weeks postimplantation ( Fig. 6 ). The results showed the same trend as in the histomorphometric analysis. Significant differences in the number of Col II and aggrecan immunoreactive cells in all experimental groups compared with the control group were detected, even in the MFX group, although the differences in this group (MFX) were less evident ( Fig. 6b ). Analysis of the expression of Col X, a marker of chondral stability, showed that in all groups the repaired cartilage exhibited little or no expression of Col X ( Fig. 6b ), which was restricted mainly to the lower area of articular cartilage and calcified cartilage ( Fig. 6a ).
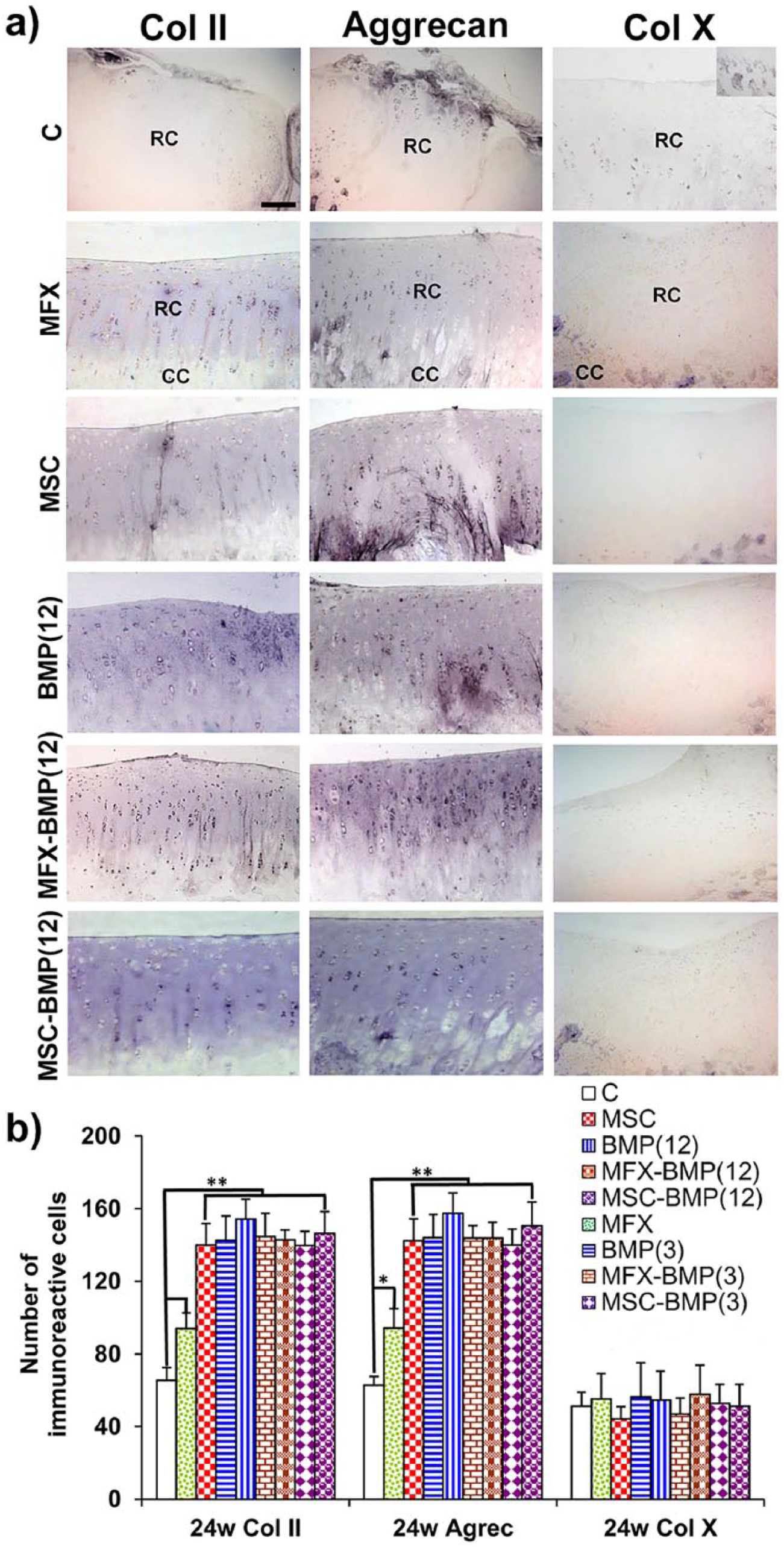
(
Discussion
The goal of the present study was to contribute to the repairing of focal cartilage lesions using noninvasive techniques. The effect of BMP-2 formulated in a controlled delivery system was compared against MSCs (previously aspirated from bone marrow) pre-seeded on the injectable scaffolds or MSCs provided by bone marrow stimulation, simulating a MFX. The combination of MSCs obtained by both procedures combined with microspheres of BMP-2 were also evaluated versus microspheres of BMP-2 alone.
By drilling the subchondral bone, the MFX technique provides bone marrow cell delivery into the defect for cartilage repair. Some BMPs are involved in all phases of chondrogenesis. BMP-2 regulates the expression of chondrocyte-specific genes and also stimulates chondrocyte proliferation and differentiation as well as matrix synthesis.28-30 Therefore, an injectable formulation of BMP-2 might be a good strategy to repair focal cartilage lesion. On the one hand, previous studies have demonstrated the efficacy of BMP-2 to repair osteochondral defects.22,31-33 On the other hand, implantation of cell pellets sequentially treated with BMP-2 and Wnt5a in an articular cartilage defect in nude rats resulted in the regeneration of cartilage with hyaline-like features. 34 However, few studies have been dedicated to the in vivo evaluation of active substances or cells in scaffolds to repair chondral defects. 35
The experimental model used in the present work consisted of a 4-mm diameter full-thickness chondral defect made at the central trochlear groove. The cartilage, including the calcified cartilage layer, was carefully removed, avoiding bleeding or damage of the subjacent subchondral plate. Consequently, a lesion with low reparative potential was obtained. Resembling a MFX technique, a controlled drilling at low rpm with a 0.9 mm needle and 4 mm depth was performed. In order to maintain bone bridges between the MFX and the borders of the defect and to preserve the integrity and function of the subchondral bone plate, a single perforation at the center of the lesion was made.
We have not found any publication dedicated to the evaluation of an injectable BMP-2 controlled delivery system to repair cartilage defect in the bibliographical sources. In the present study, the formulation was fabricated with synthetic polymers. PLGA, a Food and Drug Administration–approved biodegradable polymer useful for fabrication of controlled delivery systems and an in situ gel-forming poloxamer constituted the injectable system. BMP-2 from the system was released for 10 days postimplantation into the lesions of the rabbits. We hypothesized that BMP-2 could stimulate chondrocytes remaining at the defect borders to initiate a proper cartilage repair response.30,36,37 Cell derived from the synovial membrane could also be stimulated by the presence of growth factors and act as a source of cell for chondral defect repair. 38 Furthermore, the administration of MSCs adhered to the formulation probably improves the response in this type of defect where the subchondral bone is not affected and migration of MSCs is not expected. In fact, the reparative effect of the MSCs groups was superior to that of the control group (nontreated defect) where the cells available for cartilage regeneration are limited to the chondroblasts present at the edges of the defect. According to our results, the previously extracted MSCs incorporated into the scaffold were more efficient than the endogenous MSCs from the MFX, probably due to an insufficient or irregular contribution of cells. The group which had undergone MFX exhibited the lowest effect throughout the experimental period. As MFX is one of the most popular techniques used to regenerate the damaged cartilage, the MFX model has been used in animals to test different strategies such as condrocytes, MSCs, and scaffolds.39-41 In general, better reparative effect was observed in the cartilage defects treated with combinations of cells and scaffolds than with the MFX technique. Chondrocytes in fibrin hydrogel or in methoxypolyethyleneglycol polylactide scaffold induced better cartilage repair than MFX alone. 42 Combination of MFX with autologous bone marrow concentrate 43 or MSCs combined in situ with scaffolds 44 also improved the repair observed with MFX alone in animals. Similarly, some human studies reported that the reinforcement of MFX technique with different cell-free scaffolds alone 45 or in combination with cartilage repair substances, as platelet-rich plasma in combination with a polyglycolic acid-hyaluronic acid (PGA-HA) scaffolds, 46 enhanced the results. In addition, treatment of human focal cartilage defect by combination of MFX with PGA-HA scaffolds 10 or collagen scaffolds, 11 enriched with autologous bone marrow concentrate, improved cartilage function. The previous reports coincide with the observed results in the present study. The response of MFX-BMP groups was always higher than the MFX group, although the behavior of MFX groups combined with BMP-2 was the same regardless of the dose. Similarly, in the groups treated with the combination of BMP-2 and MSCs, the effect of the dose was not reflected and the cartilage healing was the same regardless of the protein dose. Despite the fact that after 12 weeks the best response was observed in animals treated with MSCs combined with any of the BMP-2 doses, no improvement was seen in the defect 12 weeks later, at 24 weeks postimplantation. With regard to the aim of this study, we have to highlight that the synergism was only observed in the group treated with the combinations of the low dose of BMP-2, MFX-BMP(3), and MSC-BMP(3) in comparison with the response of the group treated only with a low dose of BMP-2. However, this effect was not observed when comparison was done with high dose of BMP-2. At the end of the experimental period (24 weeks), despite the fact that nonsignificant differences were found between the different treated groups, only BMP(12), MSC-BMP(12), and MFX-BMP(3) groups were not significantly different with respect to normal cartilage. From all of them, the highest cartilage repair with the lowest variability was observed in the group treated with the high dose of BMP-2. Consequently, at this point, the discussion focuses on the advantages and disadvantages of using high doses of BMP-2 versus low doses of BMP-2 combined with MFX or MSCs. However, the doses used in this study are lower than the equivalent dose approved for human use. In addition, the risk of systemic toxicity is not worrisome since BMP-2 was locally injected in a non-vascularized defect and the diffusion of the protein into the vascular system is extremely unlikely. Also, the poor systemic stability and the short half-life of BMP-2 ensure the safety of the formulation. On the other hand, the combination of BMP-2 with cells involves more manipulation steps and the procedures are more invasive. The MFX damages the subchondral bone and the number of cells is not controlled, so an irregular response could be expected. In addition, MSCs extracted from different sources require at least 2 surgery steps. Isolation and expansion procedures may also be required, without forgetting that the number of cells and the capacity of cell proliferation and differentiation depend on the age and condition of the patient and are therefore another source of variability.
Finally, it has to be mentioned that although a good correlation between histological structure and mechanical properties of cartilage have been documented,47,48 the main limitation of this study could be the lack of a biomechanical test to confirm the functionality of the repaired cartilage.
Conclusions
The system proposed, prepared with synthetic polymers containing BMP-2, was effective in repairing the lesion, was safe, and was less invasive in comparison with MFX. The injectable hydrogel system developed provided stable delivery formulation for therapeutic agents, including cells and growth factors, to treat cartilage defects. In fact, in the present study, 24 weeks after the BMP-2 microspheres suspension injection in a focal condylar cartilage defect in rabbits, approximately 85% of the area of the lesion was repaired against the 55% observed after MFX.
Footnotes
Acknowledgments and Funding
We thank Mrs. Virginia Hernández for assistance with manuscript preparation. This work was supported by the Ministry of Science and Technology (MAT2011-23819 and MAT2014-55657-R).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Ethical Approval
Ethical approval for this study was obtained from Committee of Ethics of Research and Animal Welfare of the University of La Laguna (CEIBA2011-0001).
