Abstract
Keywords
Introduction
Articular cartilage injuries have been observed with increasing frequency in collegiate, professional, and world-class football players and may be associated with the increased rate of joint degeneration and disability in the football population.1-6 To assure continued sports activity without risking joint degeneration and long-term disability, durable articular cartilage resurfacing is critically relevant for the football athlete with articular cartilage injury. Successful repair of articular cartilage lesions of the human knee by autologous chondrocyte implantation (ACI) was first reported by Brittberg in 1994 7 and has demonstrated excellent results after up to 20 years in the general population.7,8 Because of the relative invasiveness of the first-generation technique, its long rehabilitation time, and complications resulting from the use of periosteum, researchers have aimed to reduce these limitations through continued improvement and development of this technique and its postoperative rehabilitation. This article provides an overview of the gradual evolution of ACI since its first introduction and describes how these innovations may affect postoperative sports participation in football players with articular cartilage defects.
Technical Evolution of ACI
First-Generation Technique
With this original ACI technique, cartilage harvesting was performed in a primary procedure, followed by cell isolation and expansion
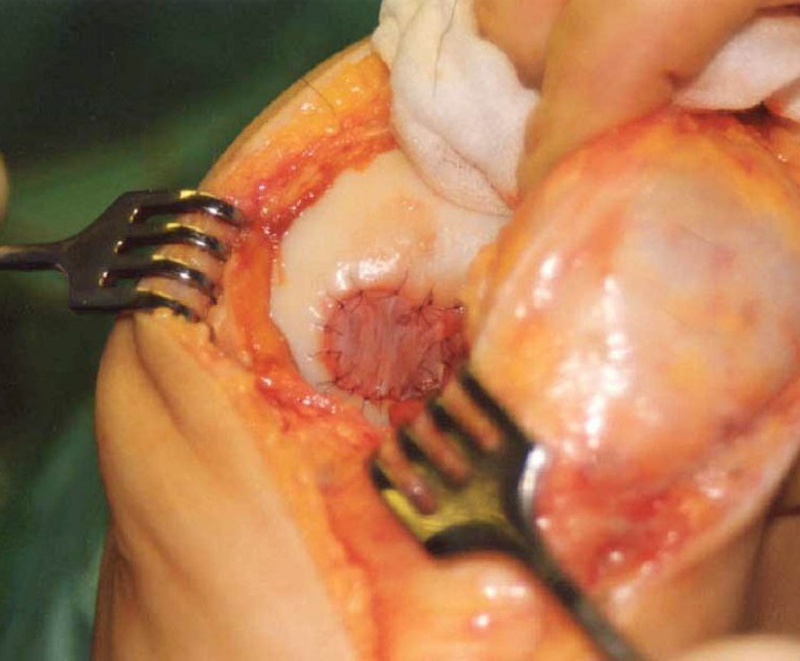
Intraoperative image of a football player treated with first-generation autologous chondrocyte implantation, demonstrating a full arthrotomy and sutured periosteal patch.
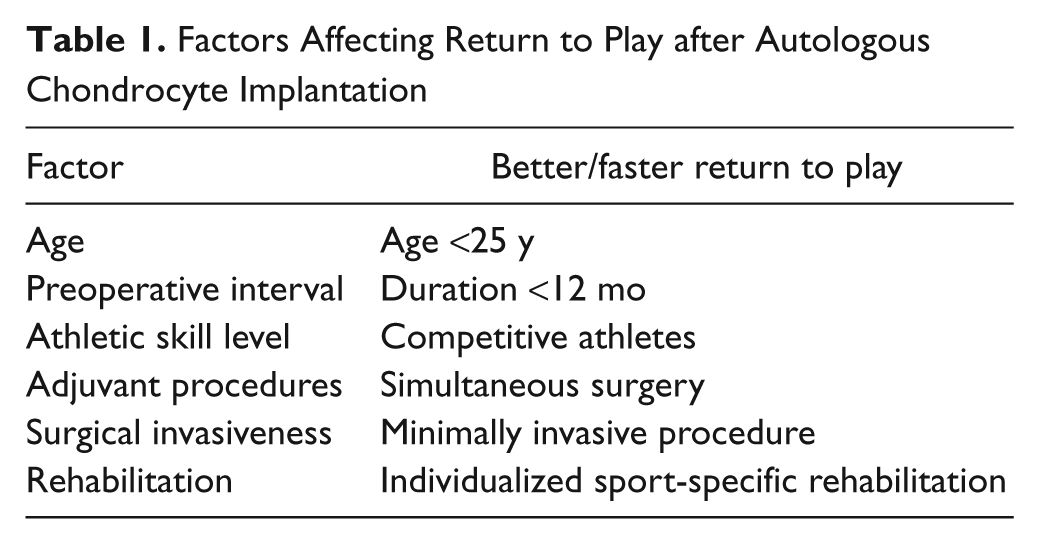
Return to football was significantly better in players with single cartilage lesions of the femur, younger age (≤25 years), and players who underwent ACI early (<12 months after injury). In fact, the ability to return to playing football was 4 times higher when ACI was performed within 1 year after injury (Table 1). No significant association was found between return to football and gender, defect type, defect location, defect size, or number of prior surgeries. Concomitant procedures such as ligament reconstructions or osteotomies did not decrease the ability to return to football, as 56% of players with osteotomies were able to return to play football. Interestingly, postoperative participation in athletic activities improved the long-term functional results after first-generation ACI. 11 The failure rate was 13%, with half of the failures resulting from traumatic delamination of hypertrophic grafts.
Factors Affecting Return to Play after Autologous Chondrocyte Implantation
Characterized Chondrocyte Implantation
The
Second-Generation Techniques
To avoid the frequent complication of graft hypertrophy and the associated risk from delamination or requirement for surgical chondroplasty of the hypertropic graft, bioabsorbable collagen membrane was developed as an innovative temporary cover of the treated defect instead of the autologous periosteal patch. This so-called collagen-covered ACI (CACI) had the advantage of reducing the invasiveness of the procedure; however, it still required an arthrotomy and suturing of the collagen membrane into the defect ( Fig. 2 ). Clinical results were similar to first-generation ACI, but this technique was able to reduce the incidence of graft hypertrophy to 6%. 16
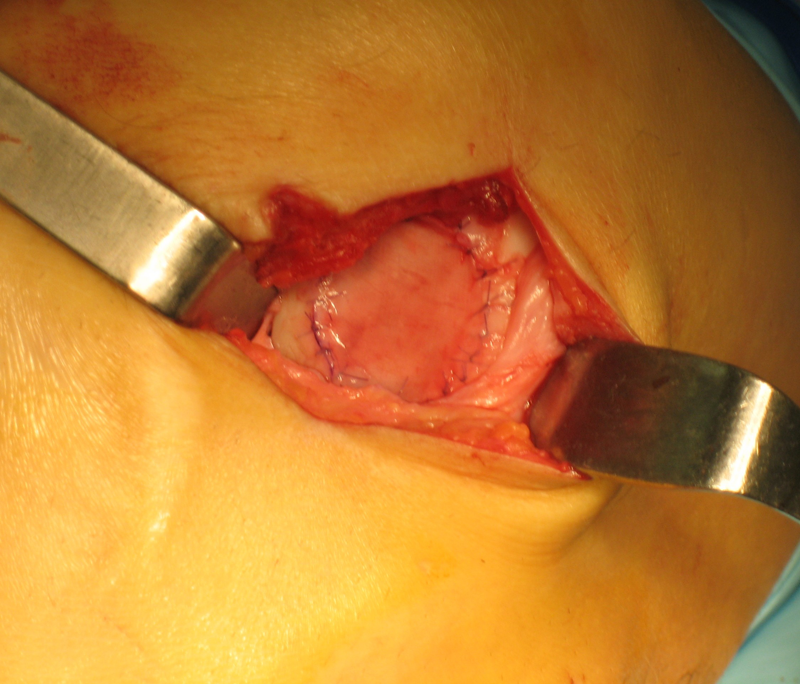
Intraoperative image of collagen membrane autologous chondrocyte implantation (CACI) using mini-arthrotomy and suture fixation of the collagen membrane.
Third-Generation Techniques
The so-called third-generation ACI techniques use biodegradable membranes or scaffolds to temporarily support the chondrocytes until they are replaced by neocartilage matrix components synthesized from the implanted cells. These techniques involve attachment or “seeding” of the cultured chondrocytes into type I/III collagen membranes or 3-dimensional scaffolds and avoid the need for the periosteum. Scaffolds and membranes can be based on carbohydrates (polylactic/polyglycolic acid, hyaluronan, agarose, alginate), protein polymers (collagen, fibrin, gelatin), artificial polymers (carbon fiber, hydroxyapatite, Teflon, polybutyric acid), or composite polymer matrices.
Matrix-induced ACI (MACI) uses a biomatrix seeded with chondrocytes and further reduces surgical invasiveness, perioperative morbidity, and risk for complications such as scarring and arthrofibrosis 17 ( Fig. 3 ). It also has the theoretical advantages of less chondrocyte leakage and more homogeneous chondrocyte distribution. This technique can be performed through a mini-arthrotomy using fibrin glue for fixation of the cell-seeded membrane into the defect. Clinical results have shown significant increases in KOOS sports and recreation subscores and Tegner activity scores at 2-year follow-up.17,18 Specific information on return to sport in the athletic population or football players is not available. The MACI technique has been associated with a reduction of graft hypertrophy to 7%. 16 Biopsy at 12 months showed evidence of hyaline-like tissue, and magnetic resonance imaging (MRI) showed good defect filling. Arthroscopic MACI has been described with a hyaluronic acid–based scaffold and showed significant improvement of knee function in 90% of the athletic population after 5 years19,20 ( Fig. 4 ). Significant improvement of Tegner activity scores was observed during the first 2 years, with 100% durability of the functional improvement until 5 years after implantation. The athlete’s age significantly correlated with the ability to resume sports activity. Concomitant procedures like anterior cruciate ligament (ACL) reconstruction were associated with the same incidence and durability of the functional improvement and same rate of resumption of sports activity. The graft hypertrophy rate was the lowest with the arthroscopic technique, with a reported rate of 4%, but the rate of delamination has not been reported.
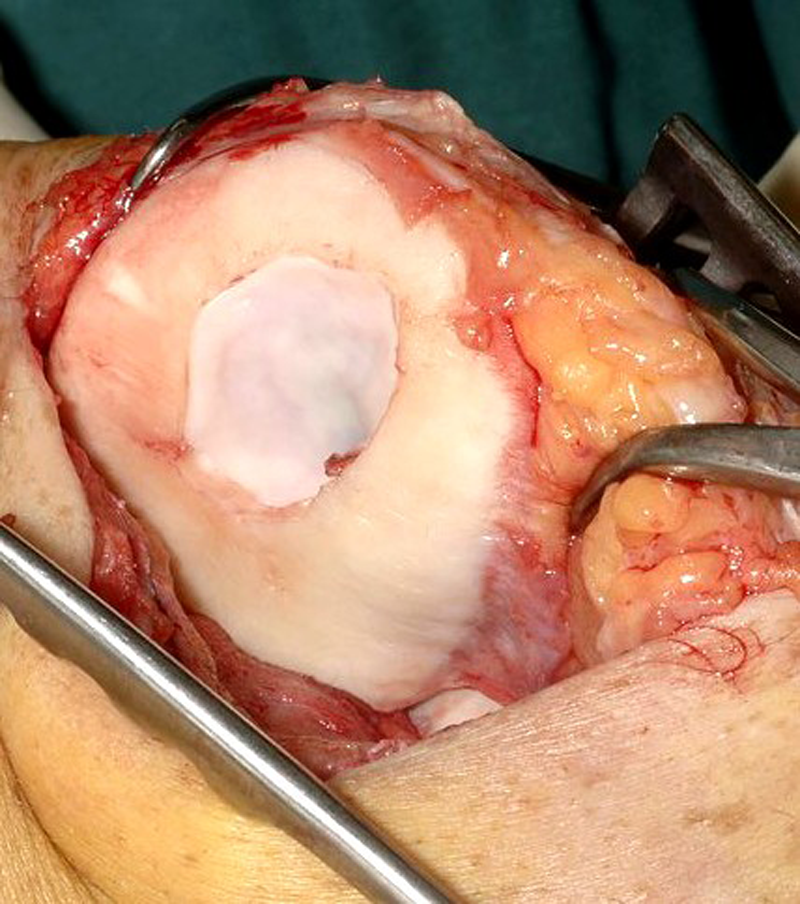
Intraoperative image of matrix-induced autologous chondrocyte implantation (MACI) using a minimally invasive technique with fibrin glue fixation of the chondrocyte-seeded matrix.
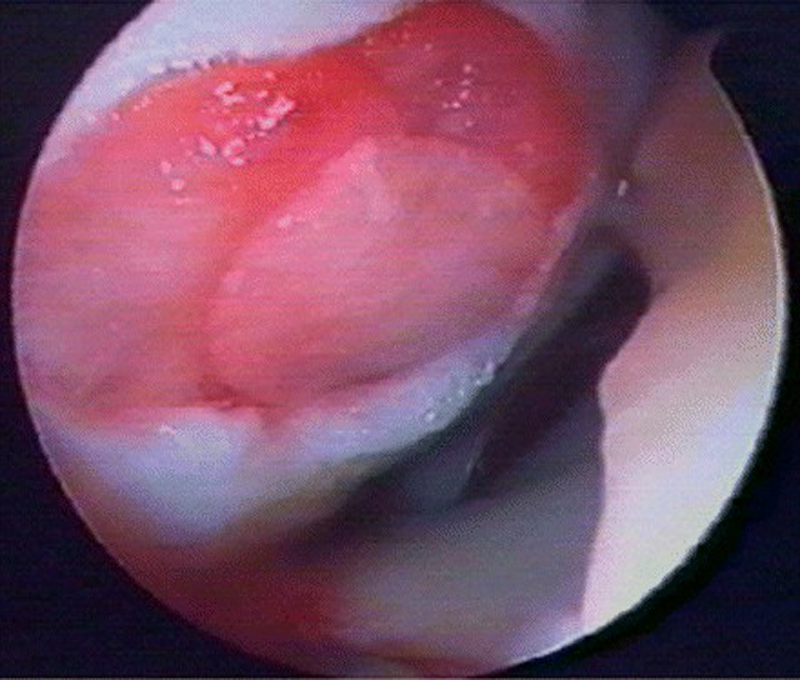
Intraoperative image of arthroscopic autologous chondrocyte implantation using a 3-dimensional hyaluronic acid scaffold seeded with chondrocytes covering the cartilage defect. From Kon
Advances in ACI Rehabilitation
Postoperative rehabilitation has traditionally been prolonged long after chondrocyte implantation with empirically based duration of joint protection and load progression following the biological phases of cartilage healing and maturation. 21 While early rehabilitation protocols have focused on protection, more recent evidence has demonstrated that accelerated rehabilitation with early limited loading improves cartilage healing, knee function, and sports participation after chondrocyte implantation.11,22,23 Decreased invasiveness of the newer-generation ACI allowed for faster postoperative joint mobilization, accelerated neuromuscular recovery, and restoration of joint biomechanics. The addition of individualized rehabilitation programs with stepwise on-field rehabilitation and reintegration into the sport-specific movement and joint loading patterns has been successful in reducing the average time to return to sport from 18 months with the first-generation ACI technique to an average of 11 months with the arthroscopic third-generation technique without compromising the return rate or durability even in highly competitive players. 22
Discussion
ACI has been described as a successful technique for the restoration of full-thickness articular cartilage lesions in the knee by several investigators.7,8,16 Recent data indicate excellent long-term durability of articular cartilage restoration and improved knee function up to 20 years postoperatively.
8
This technique has been successfully used for articular cartilage repair in athletes and specifically football players.
9
The good results in this population are encouraging because articular cartilage injuries are frequent in football players and often associated with the limited ability to play and increased risk for the development of knee osteoarthritis in this high-demand athletic population, particularly at the elite level.1,4,6,24 The increased injury rate in football players results from the high joint stresses associated with the repetitive joint impact and torsional loading seen with rapid deceleration motions, frequent pivoting, and player contact in football as well as the high incidence of ACL and meniscus injuries in this sport. The fact that chondrocyte transplantation in this population provides lasting functional improvement and return to football demonstrates the ability of the repair cartilage tissue to withstand even maximum mechanical stress over time
The percentage of good to excellent results observed after ACI in football players falls within the previously reported range of good to excellent results reported with this technique.8,16,26 Similar to the results in the mixed populations of previous studies, isolated lesions of the femoral condyle in the football population were associated with better outcomes. Significant improvement of activity scores was observed in more than 80% of players, confirming the overall functional improvements described after autologous chondrocytes in previous studies.8,15,26 The return rate was significantly higher in high-level competitive athletes than in recreational-level players. This phenomenon is also observed for other cartilage repair techniques or ACL reconstruction1,2,11,27 and can be attributed to the higher portion of acute lesions, younger age, and differences in postoperative rehabilitation, chronic deconditioning, and absence from competition. Higher motivation, fear of reinjury, and economic and social aspects will also influence the difference between the return to sport in recreational and professional athletes. 28
Return to football is significantly better if cartilage repair is performed within 12 months after injury.8-10 Delayed surgery has been associated with significantly inferior repair cartilage morphology, 27 which may result from the development of degenerative joint changes if the defect is left untreated for too long. Experimental studies have shown a change in joint hemostasis with increased glycosaminoglycan loss with delayed repair and significantly better repair cartilage quality with early repair. 29 Early surgical treatment of articular cartilage lesions is therefore recommended to optimize postoperative joint function and sports activity.
Player age affected the ability to return to football after chondrocyte transplantation and was also observed with other cartilage repair techniques. 30 This effect can be attributed to the decreasing metabolic activity of chondrocytes with increasing age as well as change in social demands.
Average time to return to football was 18 months after first-generation techniques. Newer second- and third-generation implantation techniques combined with accelerated and sport-specific rehabilitation have allowed a marked reduction of the time to return to sport to an average of 11 months. 22 Limited invasiveness of the newer implantation techniques leads to less surgical trauma to the joint with reduced disruption of joint motion, mechanoreceptors, and muscular inhibition, thereby accelerating recovery of the functional joint unit and optimizing the environment for graft stimulation and maturation. Improved understanding of the graft maturation process and the effect of gradual early graft loading has been one important aspect of this improvement.21-23
Importantly, chondrocyte implantation has shown the best durability of all cartilage repair techniques after return to sport, as 87% of returning players were still competing at a mean of 52 months postoperatively. 31 This may be because of the improved histological tissue quality seen with this technique compared to other techniques.12,13 While initial recovery with chondrocyte transplantation may be more prolonged, our data suggest that this cartilage repair technique provides excellent durability even under very high athletic demands. Some world-class players were able to rapidly return to their preinjury level of play with excellent long-term durability of more than 9 years in professional football. In contrast to other cartilage repair procedures, this positive effect does not seem to be limited to small defect size or primary cartilage repair but is equally successful in large defects and revision situations.31,32
Hypertrophy of the grafted periosteum has been described in up to 26% in first-generation ACI8,16 and was associated with an increased rate of traumatic delamination particularly in high-impact pivoting sports like football. 9 Technical modifications such as CACI and MACI have successfully reduced the rate of graft hypertrophy to 4% to 7%.16,17,33 This reduced risk likely translates into a decreased risk for traumatic delamination in athletes and facilitates more rapid activity progression and return to sport.
Concomitant injuries such as ligamentous instability are frequently associated with articular cartilage lesions. Addressing concomitant pathologies has been shown to improve cartilage repair and does not negatively influence the ability to return to sport.8,9,31 By performing all repair procedures simultaneously, repeated surgical intervention with prolonged rehabilitation and absence from competition can be avoided, and return to sport becomes more likely.
In summary, ACI has gone through a significant evolution since its first description in 1994. The first-generation technique demonstrated the efficacy of this cartilage repair procedure to return athletes to demanding sports such as football with excellent durability. Young, competitive players who undergo surgery within 12 months after injury will have the highest chance of returning to sports activity. While the initial ACI technique was associated with several limitations because of invasiveness, frequent graft hypertrophy, and long intervals until return to competition, stepwise improvements of the surgical technique and rehabilitation have successfully addressed these shortcomings with reduced morbidity, complication rate, and accelerated return to sport without compromising the return rate or ability to compete over time.
Footnotes
The authors received no financial support for the research and/or authorship of this article.
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
