Abstract
Insulin-like growth factor binding protein 5 (IGFBP-5) has been proposed to promote cartilage anabolism through insulin-like growth factor (IGF-1) signaling. A proteolytic activity towards IGFBP-5 has been detected in synovial fluids from human osteoarthritic (OA) joints. The purpose of this study was to determine if protease activity towards IGFBP-5 is present in the rat medial meniscal tear (MMT) model of OA and whether inhibition of this activity would alter disease progression. Sprague-Dawley rats were subject to MMT surgery. Synovial fluid lavages were assessed for the presence of IGFBP-5 proteolytic activity. Treatment animals received intra-articular injections of vehicle or protease inhibitor peptide PB-145. Cartilage lesions were monitored by India ink staining followed by macroscopic measurement of lesion width and depth. The MMT surgery induced a proteolytic activity towards IGFPB-5 that was detectable in joint fluid. This activity was stimulated by calcium and was sensitive to serine protease inhibitors as well as peptide PB-145. Significantly, intra-articular administration of PB-145 after surgery protected cartilage from lesion development. PB-145 treatment also resulted in an increase in cartilage turnover as evidenced by increases in serum levels of procollagen type II C-propeptide (CPII) as well as synovial fluid lavage levels of collagen type II neoepitope (TIINE). IGFBP-5 metabolism is disrupted in the rat MMT model of OA, potentially contributing to cartilage degradation. Inhibition of IGFBP-5 proteolysis protected cartilage from lesion development and enhanced cartilage turnover. These data are consistent with IGFBP-5 playing a positive role in anabolic IGF signaling in cartilage.
Introduction
Cartilage homeostasis is largely controlled by the actions of chondrocytes embedded within the extracellular matrix. Chondrocytes synthesize cartilage-specific matrix proteins such as type II collagen and proteoglycans that are essential for maintaining the structural integrity of cartilage while balancing catabolic forces in order to maintain functional cartilage over long periods of time. 1 This homeostasis is disrupted in joint diseases such as osteoarthritis (OA) in which catabolic forces are favored over anabolic signals, resulting in net cartilage degeneration. 2 The primary anabolic growth factor for cartilage is insulin-like growth factor (IGF-1), which stimulates proteoglycan synthesis by chondrocytes and also induces chondrocyte proliferation. 3,4 Hepatic production provides one source of IGF-1, which is transported to target tissues including cartilage and stimulates growth. 5 IGF-1 is also produced by chondrocytes in response to growth hormone (GH) stimulation, and this locally produced IGF-1 is important for stimulating cartilage growth during development. 6 It is likely that IGF-1 plays a key role in maintaining cartilage homeostasis during adulthood.
Studies with human articular cartilage indicate that chondrocyte responsiveness to IGF-1 progressively decreases with age. 7,8 Additionally, chondrocytes from arthritic cartilage show decreased responsiveness to IGF-1, and this may contribute to cartilage loss during disease. 9,10 The mechanism by which chondrocytes lose their responsiveness to IGF-1 is not entirely clear, but IGF-binding proteins (IGFBPs) have been suggested to play a role. 11,12 IGFBPs are a family of extracellular proteins that interact with and modulate the activities of IGFs including performing an important storage function for IGF-1 in the joint. 13 IGFBPs are synthesized by articular chondrocytes during normal growth as well as during repair after injury. 14-16 In OA, the levels of IGFBP-3, -4, and -5 are increased in diseased cartilage as are degradation products of IGFBP-3 and -5. 14,15,17,18 IGFBP-3 has been shown to inhibit the stimulatory effect of IGF-1 on proteoglycan synthesis in chondrocytes, suggesting that increased expression of IGFBP-3 in OA may account for decreased IGF-1 responsiveness. 11 Conversely, IGFBP-5 has been proposed to play a positive role in cartilage anabolism through IGF-1 signaling. 19-22 Overexpression of IGFBP-5 enhanced IGF-1 activation of the PI-3 kinase pathway and promoted IGF-1–enhanced chondrogenic differentiation. 19 These findings, taken together, suggest that under certain situations, IGFBP-5 can act as both a reservoir for IGFs in cartilage and synovial fluid as well as a positive regulator of IGF-1 signaling. Therefore, factors that control the level of intact IGFBP-5 may alter the ability of cartilage to respond to IGF-1.
One variable that regulates the concentration of the intact form of IGFBP-5 is proteolysis. Some studies have reported increased amounts of IGFBP protease activity in joint fluid during the development of arthritis. 20,21,23-25 Significantly, inhibition of proteolytic cleavage of IGFBP-5 was shown to limit the amount of cartilage destruction in a canine model of OA. Inhibition of IGFBP-5 proteolysis was associated with a significant increase in the amount of IGF-1 in the synovial fluid as well as an increase in intact IGFBP-5. 20 The IGFBP-5 protease identified in model canine OA joints and in human OA synovial fluid is the complement protease C1s. 20,26 C1s is considered highly specific, cleaving the C2 and C4 components of the complement cascade. 27 Aside from IGFBP-5, C1s has been shown to cleave only a few substrates outside the complement cascade, although the physiological relevance is uncertain. 28-30
Because IGFBP-5 protease activity has been previously characterized in canine as well as human OA joint fluid, these studies were undertaken to determine if a similar activity was present in a rat model of OA and to test whether inhibition of this activity would have a protective effect on cartilage degeneration. Medial meniscal tear (MMT) in rats results in joint instability and progressive development of OA-like characteristics. 31,32 The rat MMT model is widely employed to evaluate potential therapeutic agents for OA, and it is therefore important to confirm that common metabolic pathways exist between this model and the dog model as well as human OA. To evaluate whether inhibition of IGFBP-5 proteolysis could alter disease progression in this model, we utilized peptide PB-145, which has been shown to inhibit C1s directly, 33 as well as the IGFBP-5 proteolytic activity derived from model canine OA joints. 20 It is important to note that PB-145 does not inhibit a panel of matrix metalloproteinases including MMP-1, -2, -3, -9, and -13 (Sverdrup, unpublished data) and therefore does not directly inhibit collagenolytic activity. Ex vivo, PB-145 inhibited the proteolytic activity towards IGFBP-5 found in rat MMT joints. Intra-articular administration of PB-145 beginning 7 days after MMT resulted in reduced tibial cartilage lesion severity and increased cartilage thickness. These data are consistent with a proanabolic consequence of inhibiting IGFBP-5 proteolytic activity and increasing IGF-1–mediated cartilage homeostasis.
Methods
Use of the animals in these studies was reviewed and approved by the Pfizer Institutional Animal Care and Use Committee. The animal care and use program is fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
MMT Surgery
The MMT model of OA in rats has been described. 31,32 For characterization of synovial fluid protease activity in the model, 6 animals per time point were used for obtaining synovial fluid lavages at 1, 2, and 3 weeks after surgery. For intra-articular treatment with inhibitory peptide and subsequent analysis of joint cartilage integrity, 15 animals were used per group. Sprague-Dawley male rats (10 weeks old, ~300 g) (Charles River Laboratories, Wilmington, MA) were anesthetized with isoflurane, a skin incision was made over the medial aspect of the right knee, and the medial collateral ligament was exposed and transected. A full-thickness cut was made through the medial meniscus to simulate a complete tear. The skin was closed in one layer with surgical staples.
Synovial Fluid Lavages
Synovial fluid lavages were obtained after sacrifice by intra-articular injection of 100 uL phosphate buffered saline (PBS) into the operated (right) knees, repeated flexing (10x) of the injected joint, and withdrawal of fluid (typically 25-50 uL). Control lavages were taken from the nonoperated (left) knees for comparison. For analysis of proteolytic activities, lavages were taken 1, 2, and 3 weeks after surgery in untreated animals (n = 6).
Protease Activity Assays
IGFBP-5 protease activity in joint fluid lavages was assessed by incubating 150 ng recombinant human IGFBP-5 (Austral Biologicals, San Ramon, CA) with 1 uL lavage in a total volume of 20 uL in assay buffer (50 mM Tris, 2 mM CaCl2, pH 7.4). 34 After 30 minutes of incubation at 37 °C, samples were electrophoresed on polyacrylamide gels and blotted with a 1:1000 dilution of antisera to IGFBP-5 (AF875, R&D Systems, Minneapolis, MN) in TBS-Tween with 5% milk. Chemiluminescent detection was achieved with a peroxidase-conjugated secondary antibody (#705-036-147, Jackson ImmunoResearch Laboratories Inc., West Grove, PA) and the ECL kit (Amersham Biosciences, Piscataway, NJ). For inhibitor studies, inhibitors from the Protease Inhibitor Set (Roche Applied Science, Penzberg, Germany) were included: antipain (250 µg/mL), E-64 (60 µg/mL), leupeptin (2.5 µg/mL), pepstatin (3.5 µg/mL), phosphoramidon (1.65 mg/mL), Pefabloc (5 mg/mL) (Roche Applied Science), EDTA-Na2 (4 mg/mL), and aprotinin (10 µg/mL). These concentrations were chosen to be 5 times higher than that needed to efficiently inhibit proteases of the relevant classes, as detailed in literature included with the inhibitor set. In the case of phosphoramidon, this concentration is 25 times higher and may have resulted in nontarget class inhibition.
Treatments and Scoring System
Treatment animals (15 rats per group) received biweekly intra-articular (IA) injections (0.05 mL) of either vehicle (PBS) or peptide PB-145 33 (100 or 500 ug in PBS) in the surgery knee beginning 7 days after MMT and continuing for 3 weeks. One day after the last injection, animals were sacrificed and the joints lavaged to obtain synovial fluid. In order to evaluate many potential therapeutic agents for OA, we required a method that was both higher throughput and more time- and cost-effective than histology. Thus, we have employed a quantitative scoring system based on cartilage appearance after India ink staining as well as the thickness of cartilage and depth of lesions. The surgery joints were disarticulated and the tibial plateaus stained with India ink. To quantify the severity of cartilage lesions, a scoring system was devised that included a measure to reflect the apparent thicker cartilage in PB-145–treated joints. The tibial surface was first imaged horizontally using MetaMorph, v6.1 (Molecular Devices Corporation, Downingtown, PA). The area of India ink staining was outlined manually for the total area of lesions. The depth of lesions was evaluated by physical probing. Full-thickness lesions were recorded when the probe hit solid bone. Because the total area of cartilage lesions was not necessarily decreased in PB-145 samples, a new measure designated “skyline” depth or concavity was generated. For this, the medial tibial plateau was viewed from the anterior to posterior direction until a maximum concavity was detected and the image captured. Using this image, a line was drawn from the highest point on the inside (nearest the cruciate ligaments) to the highest point on the outside of the tibial plateau. This represents zero concavity. From this line, a perpendicular line was drawn to the deepest part of the curve, which was recorded as the “skyline depth” in pixels. In order to compare the combined data from all measurements, a total score was calculated that included surface irregularity X area of India ink staining + staining intensity + “skyline” concavity.
TIINE and CPII Assays
TIINE (type II collagen neoepitope) was quantitated utilizing BioVeris technology (BioVeris Corporation, Gaithersburg, MD). Synovial lavages were not successfully obtained from all 15 animals in each group, but a minimum of 11 samples from each group were analyzed. Briefly, 25 uL rat synovial fluid lavage, 25 uL streptavidin beads (0.4 mg/mL), 25 uL each antibody at 1 ug/mL (biotin-conjugated mouse 5109 anticapture and BVTAG-labeled 9A4 antineoepitope), and 100 uL assay buffer (DPBS, 0.1% BSA, 0.05% Tween 20, pH 7.4) were incubated for 2 hours at room temperature before reading on the BioVeris M384 analyzer. Values were calculated from a standard curve prepared from rat 30-mer TIINE peptide (0.313-20 ng/mL).
CPII (procollagen II C-propeptide) was measured by competitive immunoassay (IBEX Technologies Inc., Montreal, Quebec, Canada). Briefly, ELISA plates were coated overnight with bovine CPII. Separately, CPII standards or unknowns (n = 15/treatment group) were mixed with CPII antibody. The mixture was added to the prewashed coated CPII plate and incubated for 2 hours. After washing, goat antirabbit horseradish peroxidase was added for 1 hour. Tetramethylbenzidine was added and blue color development measured. The OD is inversely proportional to the amount of epitope present.
Data Analysis
All values are presented as mean ± standard deviation (except Fig. 7A , mean ± standard error of the mean). Statistical analyses are based on a 1- or 2-sided t test for independent samples as indicated in figure legends.
Results
In order to characterize the rat MMT model, we first examined whether a proteolytic activity towards IGFBP-5 would be detectable in joint fluid lavages of rats at various time points after MMT surgery. Samples of MMT synovial fluid lavage were incubated with recombinant IGFBP-5 and proteolysis determined by Western analysis of intact versus fragmented IGFBP-5. As shown in Figure 1 , a potent proteolytic activity is present in fluids from surgery knees 2 weeks after MMT that is not present in the contralateral control knee fluids. Specific proteolytic fragments of IGFBP-5 common only to samples incubated with MMT synovial fluid are evident. This activity was similarly detected in and specific to surgery knee synovial fluid lavages from animals at 1 and 3 weeks after MMT (data not shown).
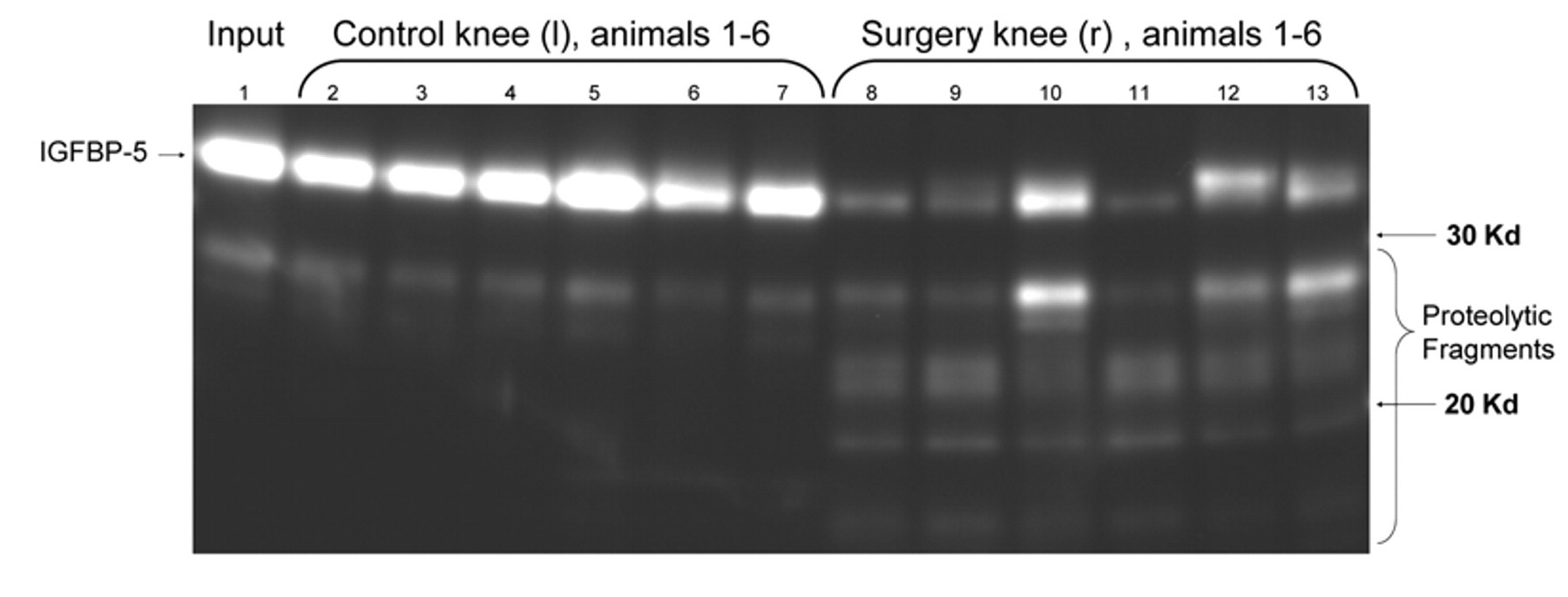
Evaluation of protease activity towards IGFBP-5 in synovial fluid lavages from rat knees after medial meniscal tear (MMT). The right knees of 6 rats were subject to MMT surgery. Two weeks after surgery, synovial fluid lavages were obtained from both the surgery (right, r) and control (left, l) knees. Aliquots of synovial fluid lavage were incubated with purified recombinant IGFBP-5 for 30 minutes. The reactions were then subject to SDS-PAGE and blotted with antisera to IGFBP-5 in order to determine the levels of intact and degraded IGFBP-5.
To further characterize the IGFBP-5 proteolytic activity, samples of MMT synovial fluid lavage were incubated with recombinant IGFBP-5 in the presence of high concentrations of various protease inhibitors. As shown in Figure 2A , the serine protease inhibitors Pefabloc SC (5 mg/mL) (Roche Applied Science) and antipain (250 ug/mL) completely inhibited the proteolytic activity (lanes 9 and 4, respectively). The activity was also inhibited by disodium EDTA (4 mg/mL), indicating a requirement for a divalent cation ( Fig. 2A , lane 10). Although there was significant inhibition with a high concentration of phosphoramidon (1.65 mg/mL) ( Fig. 2A , lane 8), the activity is not due to a metalloproteinase because 1,10-phenanthroline (1 mM) had no effect ( Fig. 2B ). The activity was insensitive to E-64, leupeptin, pepstatin, and aprotinin ( Fig. 2A , lanes 5, 6, 7, and 11, respectively). To further explore the cation dependence of the proteolytic activity, the reaction was carried out in the absence and presence of calcium. The activity was significantly enhanced in the presence of calcium ( Fig. 2C , lane 2). The activity was not enhanced in the presence of divalent zinc, magnesium, or manganese (data not shown). These data are consistent with a calcium-dependent serine protease being responsible for IGFBP-5 proteolysis in rat synovial fluid.
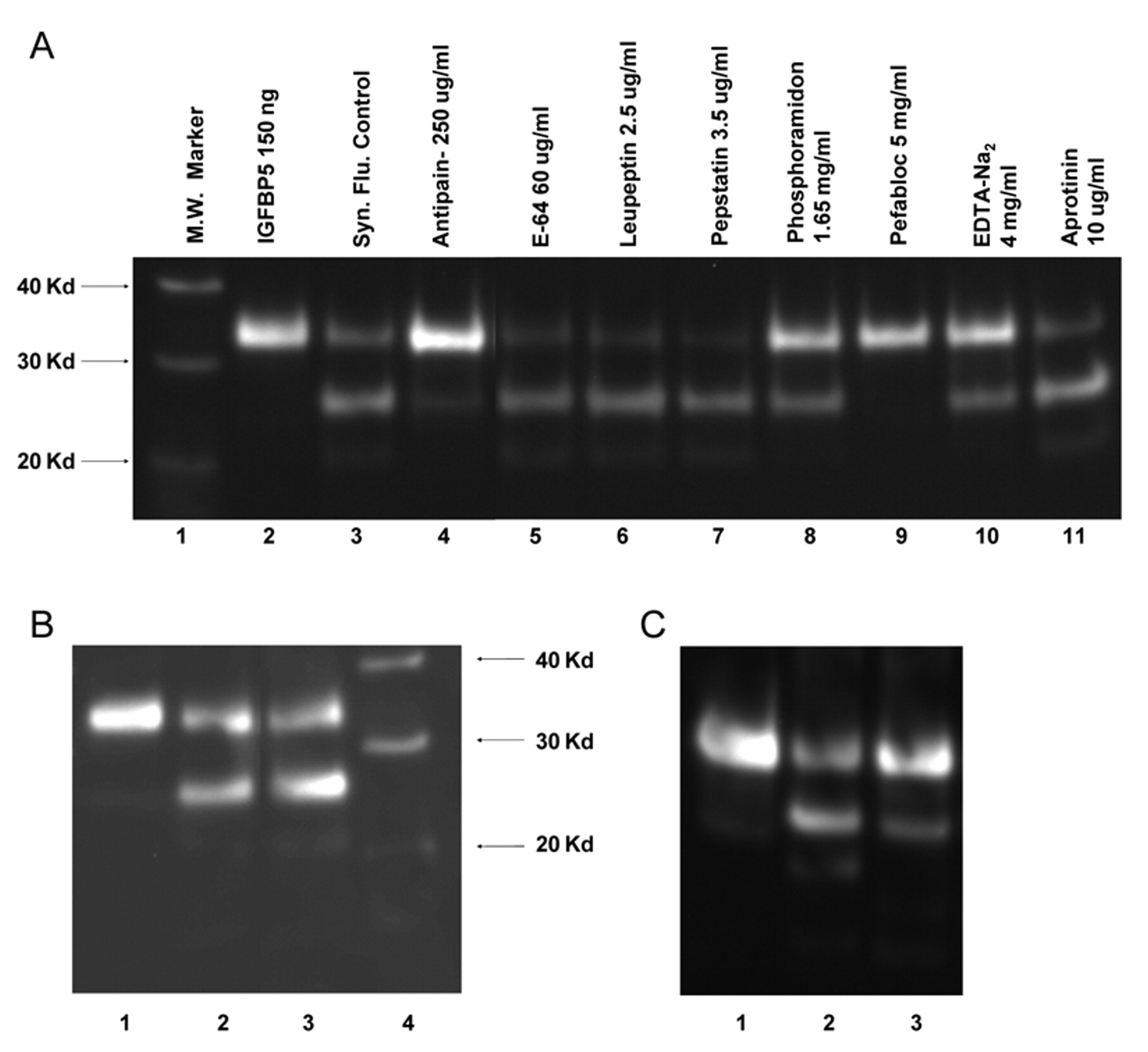
Characterization of rat synovial fluid IGFBP-5 protease activity. (
In order to test the hypothesis that inhibition of IGFBP-5 proteolytic activity would alter disease progression in the rat meniscal tear model of OA, we sought out a peptide inhibitor of the activity that would be suitable for intra-articular administration. Peptide PB-145 has been previously described to be an efficient inhibitor of the IGFBP-5 proteolytic activity found in model canine OA joints. 20,33 Furthermore, intra-articular injections of PB-145 in the canine ACLT model resulted in decreased disease progression and alterations in IGF-1 metabolism. 20 Therefore, we tested the ability of PB-145 to inhibit the IGFBP-5 proteolytic activity derived from rat MMT synovial fluid. As shown in Figure 3 , PB-145 inhibits IGFBP-5 proteolysis in the low micromolar range, consistent with its ability to inhibit canine-derived IGFBP-5 proteolytic activity. This result indicated that the inhibitory activity of PB-145 crossed species and that the peptide would be useful as an in vivo tool in the rat.
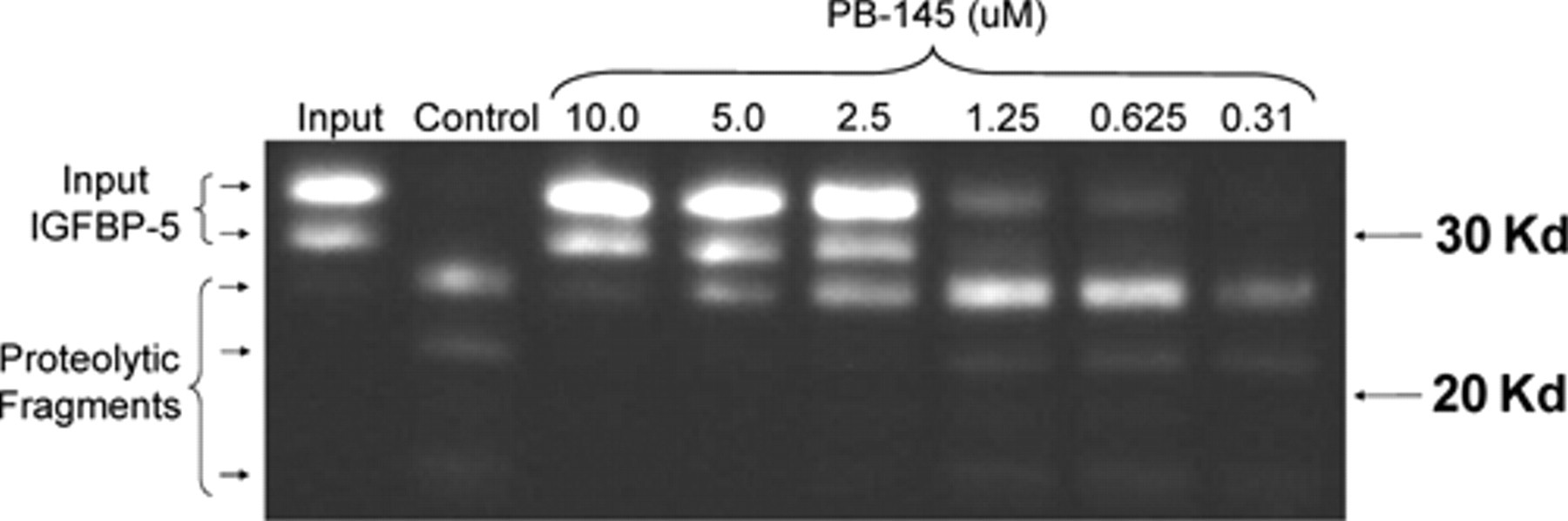
Inhibition of rat synovial lavage protease activity by peptide PB-145. Recombinant IGFBP-5 was incubated with synovial lavage from rat medial meniscal tear (MMT) knee joints in the absence (control) or presence of the indicated concentrations of peptide. Samples were then processed as in Figure 1 .
We next determined if PB-145 would protect cartilage from lesion development in the rat MMT model of OA. Beginning 1 week after MMT, rats were given twice-weekly intra-articular injections of PB-145 or saline as a vehicle control. An additional group of animals did not receive intra-articular injections (no treatment controls). Figure 4A shows India ink staining of an entire tibial plateau 4 weeks after MMT with no treatment. The darkest areas of staining on the medial tibial plateau (left side) indicate deep focal lesions that span nearly the full depth of the cartilage to the underlying subchondral bone. The lighter stained areas represent less severe cartilage degeneration. Note the smooth cartilage and lack of any staining on the lateral tibial plateau (right side), which is typical of this model. Figure 4B depicts the medial aspects of the tibial plateaus from 2 representative animals in the vehicle (saline) treated group. Again, deep focal lesions with an extended area of staining and fibrillation are present. Treatment with PB-145 reduced the depth and severity of lesions. Figure 4C shows medial tibial plateaus from the low-dose PB-145 group (100 ug/injection). Focal lesions are still present, but there are no full-thickness lesions, and the fibrillated area is shallower in depth, and staining with India ink is less intense. Figure 4D shows medial tibial plateaus from the high-dose PB-145 group (500 ug/injection). Instead of focal lesions, there is a thick layer of cartilage with a rough, slightly fibrillated surface and faint India ink staining.
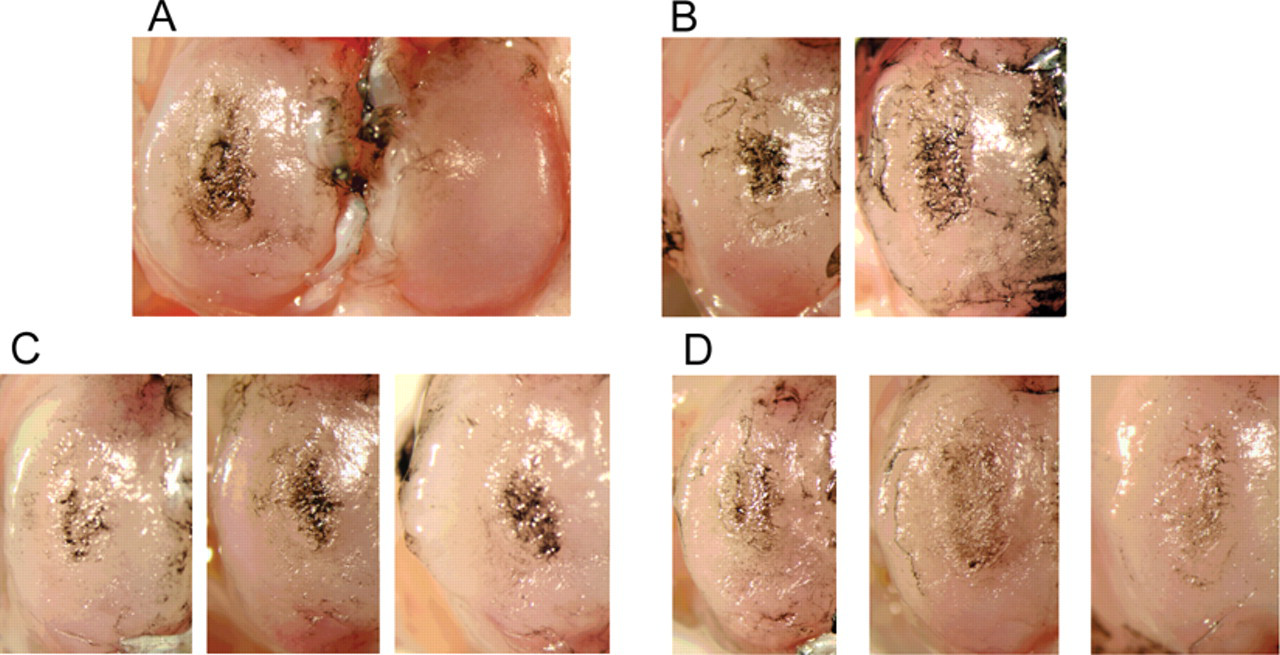
India ink staining of rat tibial plateaus 4 weeks after medial meniscal tear (MMT) surgery. Representative photographs are shown for each group and depict either the entire tibial plateau (group 1) or medial tibial plateaus (groups 2-4). (
To quantify the severity of cartilage lesions, a scoring system was devised that included a measure to reflect the apparent thicker cartilage in PB-145–treated joints (see Methods for details). The tibial surface was first imaged horizontally, and the area of India ink staining was outlined manually for the total area of lesions. The depth of lesions was evaluated by physical probing. Because the total area of cartilage lesions was not necessarily decreased in PB-145 samples, a new measure designated “skyline” depth or concavity was generated from viewing the medial tibial plateau from the anterior to posterior direction. A line drawn from the highest point on the inside (nearest the cruciate ligaments) to the highest point on the outside of the tibial plateau represents zero concavity. From this line, a perpendicular line was drawn to the deepest part of the curve, which was recorded as the “skyline” depth in pixels ( Fig. 5A ). Representative skyline photographs from group 2 (vehicle) ( Fig. 5B ) and group 3 (PB-145 100 ug) ( Fig. 5C ) show the reduced skyline depth in PB-145–treated animals. The skyline data from all animals in groups 1 to 4 ( Fig. 5D ) indicate a dose response reduction in the depth of lesions with PB-145 treatment. In order to compare the combined data from all measurements, a total score was calculated that included multiplying the surface irregularity score by the area of India ink staining and then adding the staining intensity and “skyline” concavity scores ( Fig. 6A ). The surface irregularity score ( Fig. 6B ), staining intensity score ( Fig. 6C ), and the total tibial score ( Fig. 6D ) all indicate improvement of gross tibial cartilage morphology with PB-145 treatment compared to vehicle.
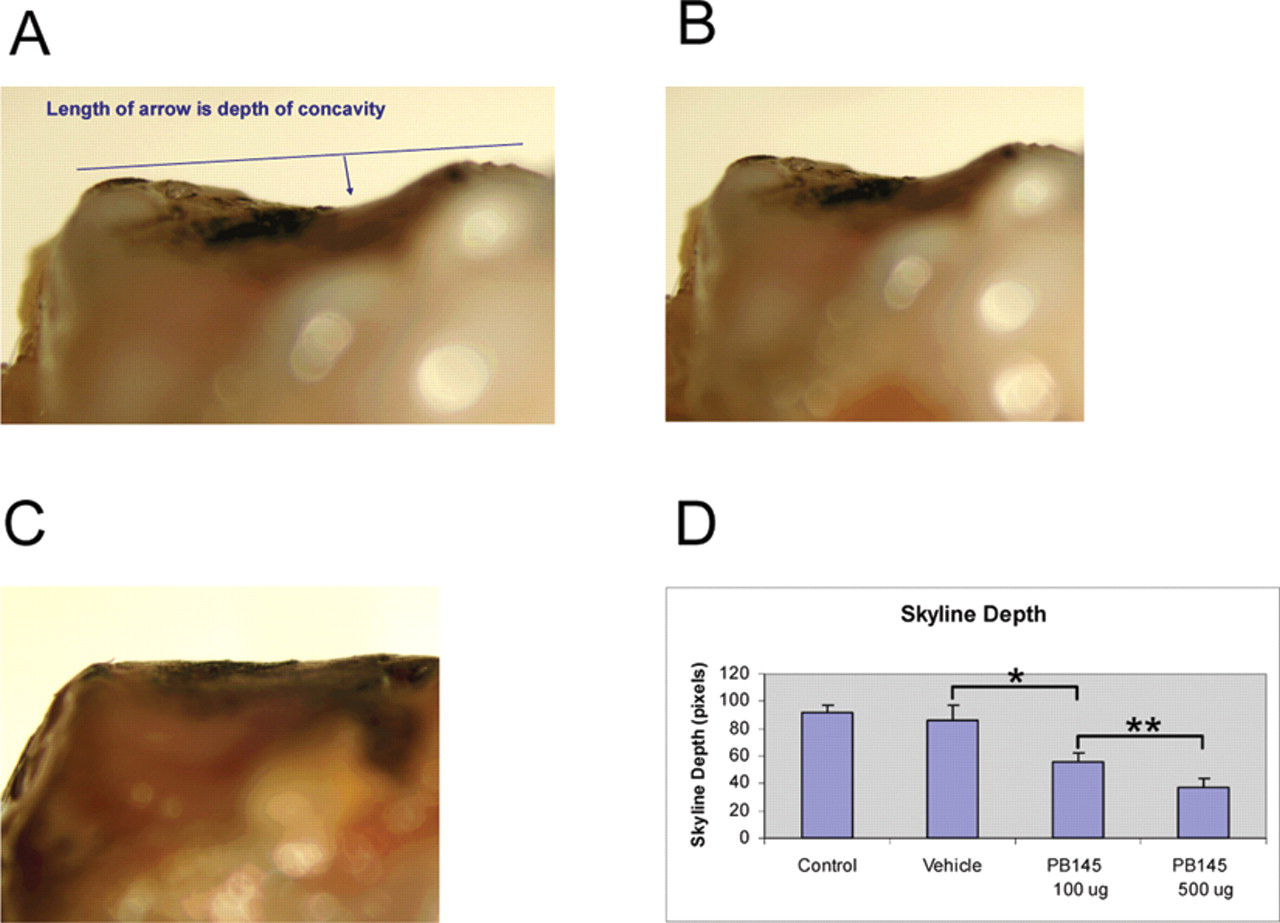
Skyline concavity measurement of the medial tibial surface. (
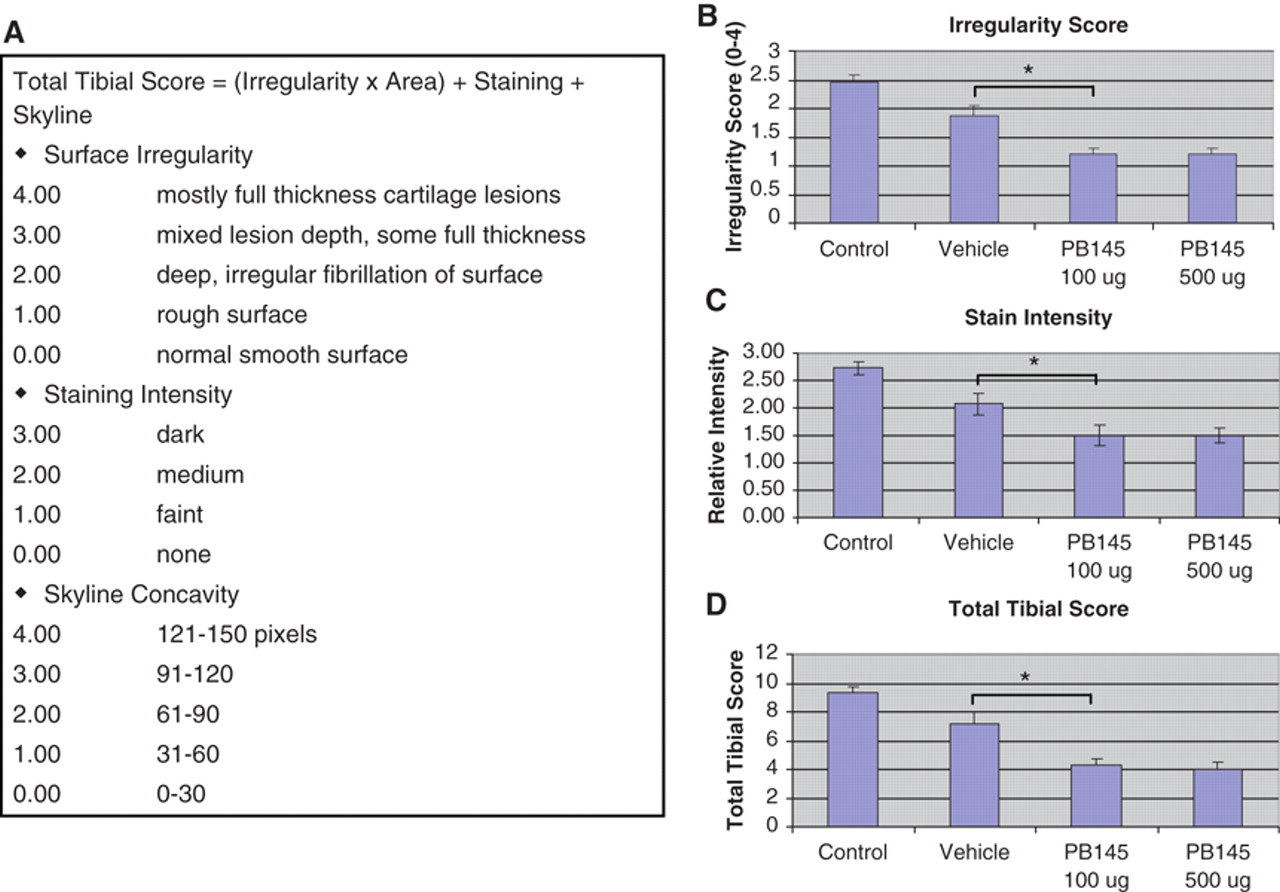
Scoring of joints for cartilage lesion severity. (
Biomarkers of cartilage metabolism were assayed in both the limited amount of synovial fluid lavage that was obtained (collagen type II neoepitope [TIINE]) as well as in serum (procollagen type II C-propeptide [CPII]). TIINE in the synovial fluid or urine is a marker of cartilage breakdown. 35 Four weeks and 1 day after MMT, TIINE levels were elevated in the surgery (right) knee lavages as compared to nonsurgery (left) knee lavages in the control group (untreated), indicating that MMT surgery resulted in the breakdown of type II collagen ( Fig. 7A ). It is important to note that animals in the control group did not receive intra-articular injections during the course of the experiment that otherwise may have perturbed and/or diluted out the concentration of TIINE in the joint. Indeed, the levels of TIINE in the vehicle-treated animal synovial lavages were decreased as compared to controls. There was a significant (60% ± 15%, P = 0.016) increase in TIINE levels in animals in the high-dose (500 µg PB-145) treatment group as compared to vehicle, indicating an increase in the amount of type II collagen breakdown product ( Fig. 7A ). CPII is a marker of new type II collagen synthesis. 35 Due to the limited amount of synovial fluid lavage sample, we were unable to look at CPII levels in this compartment. However, we did look at CPII levels in serum. Although not statistically significant, there was a trend towards increased CPII levels in the sera from PB-145–treated animals ( Fig. 7B ). These data are consistent with increased collagen II synthesis and breakdown in the joints of PB-145–treated rats as compared to vehicle-treated rats.
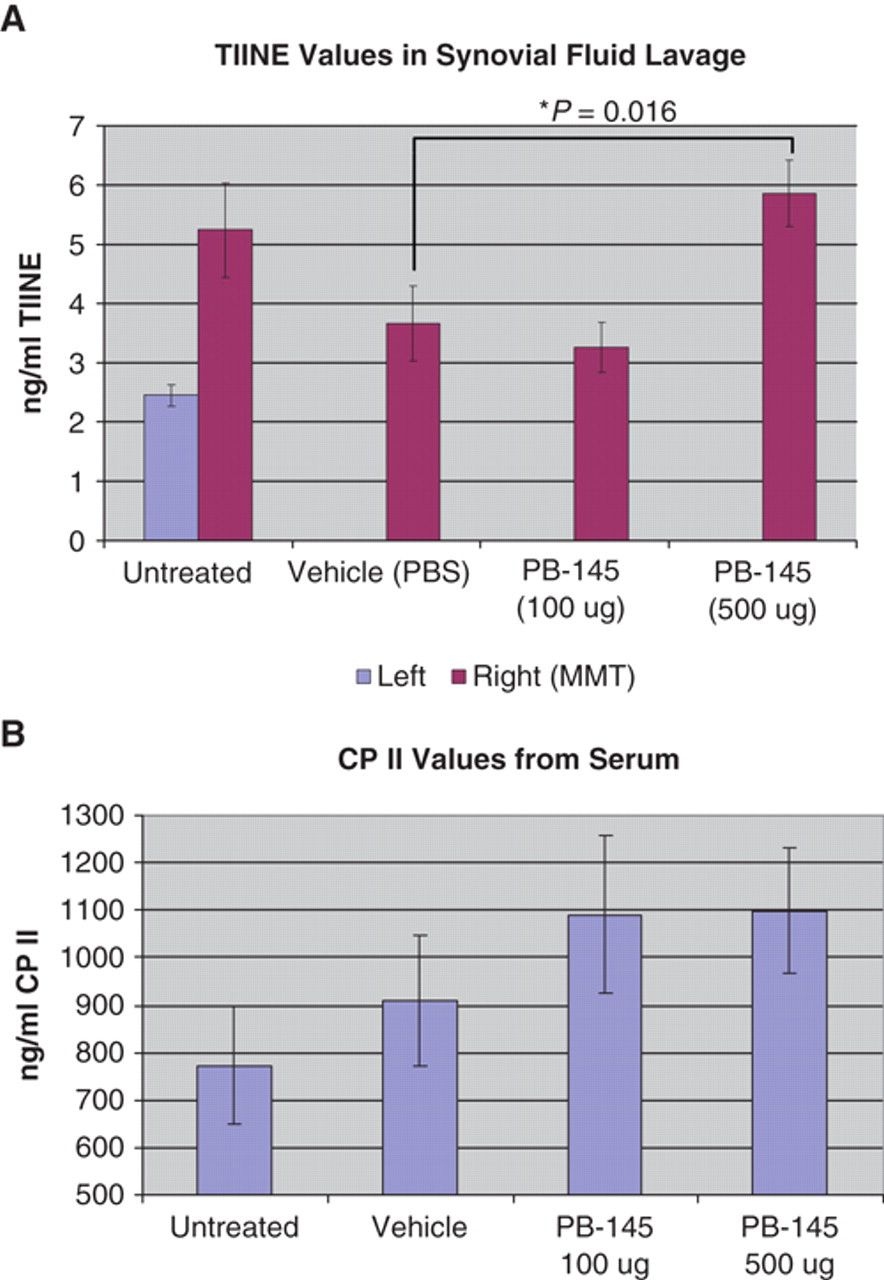
Biomarkers of cartilage turnover. (
Discussion
Prior studies have shown that IGFBP-5 is cleaved by an activity in joint fluid from dogs after ACLT surgery. 20 The data presented in Figure 1 clearly indicate that such an activity is also induced in rat synovial fluid after MMT surgery. 31 In the dog ACLT model, complement component 1s (C1s) was implicated as the protease responsible for cleaving IGFBP-5, and it was shown that C1s was present in an activated state in synovial fluid after ACLT surgery. 20 Furthermore, a peptide inhibitor of C1s (PB-145) reduced the level of cleaved IGFBP-5 in synovial fluid. However, the potential involvement of another protease that would also be inhibited by PB-145 could not be excluded. In addition to C1s, 34 several other proteases have been shown to cleave IGFBP-5 including HTRA-1, 36 Adam 9, 37 Adam 12S, 38,39 PAPP2A, 40 MMP-2, and MMP-9. 41 However, only C1s, 20 HTRA-1, 42,43 and ADAM 12 39 have been shown to be increased in arthritic disease. Characterization of the IGFBP-5 protease activity in this study suggests that the responsible rat enzyme is a calcium-dependent serine protease ( Fig. 2 ), consistent with the profile of C1s. 44-46 Unfortunately, the available antisera to C1s cross-reacted poorly with rat C1s, and we were unable to determine if there was an increase in activated C1s in the relatively dilute rat synovial fluid lavages after MMT surgery. However, C1s mRNA levels are increased in the rat MMT model of OA (Pfizer, unpublished results) as well as in the rat ACLT/MMT 47 and the rat monosodium iodoacetate-induced 48 models of OA. Additionally, in a recent study of human OA synovial fluid, C1s was identified as the human protease responsible for cleaving IGFBP-5. 25 Taken together, these findings suggest that in human OA as well as in the rat MMT and canine ACLT models of OA, activation of C1s may be a common mechanism responsible for degrading IGFBP-5.
It is well documented that IGF-1 directly promotes cartilage growth and repair. IGF-1 stimulates chondrocyte cell proliferation and protein synthesis including proteoglycan synthesis. 3,4,6,10,49,50 Direct injection of IGF-1 into the joint enhances cartilage development and repair. 51-53 Overexpression of IGF-1 in experimental models of OA using gene therapy approaches is protective for articular chondrocytes. 52,54,55 Decreasing cytokine levels in combination with IGF-1 therapy produces additive improvement in cartilage matrix accumulation. 56 Additionally, osteogenic protein-1 (OP-1) enhances the effects of IGF-1 on articular cartilage, and IGF-1 synthesis is upregulated during articular chondrocyte repair in several animal models of arthritis. 57 It will be interesting to determine the effects of OP-1 treatment on IGFBP-5 synthesis and degradation in animal models. These findings suggest that strategies to re-establish IGF-1 homeostasis and activity in the joint may lead to improved outcomes in OA.
The role of IGFBPs in modulating IGF-1 action in normal and OA cartilage has been less clear. For example, in a dog model of OA, IGFBP-3 and -4 were upregulated, and these changes were accompanied by increases in IGF-1. 58 Iwanaga et al. showed increased expression of IGFBP-3, -4, and -5 as well as their degradation products in OA articular cartilage as compared to control. 14 Large increases in the concentrations of IGFBPs in extracellular fluid can limit IGF-1 access to receptors. Consistent with this, pharmacological disruption of the IGF-1 association with IGFBP-3 resulted in improvement in proteoglycan synthesis in human OA cartilage, suggesting that IGFBP-3 was inhibiting IGF-1 actions. 59 However, studies by Duan et al. suggested that IGFBP-3 had an important positive modulatory role on IGF-1 activity in chondrogenesis during development. 13 Kiepe et al. reported that IGFBP-1, -2, -4, and -6 act exclusively as growth inhibitors of chondrocytes in vitro; however, they obtained more complex results with IGFPB-3 showing both potentiation and inhibition of the effects of IGF-1 under different conditions. 19 In contrast to other IGFBPs, IGFBP-5 enhanced IGF-1–stimulated growth plate chondrocyte proliferation, differentiation, and matrix production. 19,21,22 It appears that IGFBP-5 plays a positive role in IGF-1 signaling in cartilage and that protecting IGFBP-5 from cleavage should help maintain anabolic responses in disease states such as OA. However, a recent report that ADAM12 cleavage of IGFBP-5 enhances IGF-1–driven chondrocyte proliferation indicates that IGF-1 signaling in chondrocytes may be more complex. 39
In order to inhibit IGFBP-5 protease activity in the dog ACLT model of OA, Clemmons et al. utilized intra-articular injections of peptide PB-145. 20 PB-145 is a peptide that was originally designed as an inhibitor of complement activation and does indeed inhibit C1s. 33 In order to rule out the possibility that PB-145 may inhibit a more general pathway of cartilage degradation, we determined that PB-145 did not inhibit a panel of matrix metalloproteinases including MMP-1, -2, -3, -9, and -13 as well as the serine protease PACE4 (data not shown). In this study, we showed that PB-145 inhibited the protease activity towards IGFBP-5 from rat MMT synovial fluid ( Fig. 3 ), indicating this peptide would be useful as a tool to determine the consequence of inhibiting IGFBP-5 proteolysis on cartilage degeneration. Intra-articular injection of PB-145 in the rat MMT model of OA resulted in dramatic changes in the appearance of cartilage lesions as well as the thickness of cartilage. Lesions were more superficial in nature and stained less intensely with India ink. Although the area of each lesion was not necessarily smaller, the cartilage appeared thicker than in untreated or vehicle-treated controls, especially in the high-dose PB-145 group ( Fig. 4 ). In order to capture quantitatively the apparent differences in cartilage thickness, a new measurement was employed that takes advantage of the natural concavity of the medial aspect of the tibial plateau when viewed along the cartilage surface. PB-145 treatment reduced medial tibial plateau concavity in a dose-dependent manner ( Fig. 5 ), suggesting there was increased cartilage thickness in treated animals as compared to control and vehicle-treated animals. Because the cartilage showed signs of being subjected to a mechanically unstable joint (rough surface), yet was thick, having lost its natural concavity, and was protected from deep lesion formation, it is tempting to speculate that PB-145 promoted an anabolic response that was able to counteract the catabolic forces on the cartilage; however, direct evidence for this is still lacking.
Analysis of biomarkers of collagen turnover suggests that degradation of type II collagen (TIINE) was increased in the high-dose PB-145 group ( Fig. 7A ). In light of the fact that this group of animals showed the highest degree of protection from lesion development and exhibited a thick, yet rough surfaced layer of cartilage, the increase in the level of TIINE can be interpreted as the result of having an increased amount of substrate (cartilage) present at the location where mechanical instability promotes cartilage breakdown. The ability to maintain a layer of cartilage on the medial tibial plateau while catabolic forces result in the continuous degradation of type II collagen suggests that anabolism and new collagen synthesis are playing a role. Because of the limited amount of sample, we were unable to measure the type II collagen synthesis marker CPII directly in the synovial fluid lavages. However, CPII in the serum showed a trend upward with PB-145 treatment. It is likely that the high background of CPII in the serum of growing animals precluded us from getting a statistically significant difference based on events that were occurring in a single joint. Therefore, we cannot conclude definitively that PB-145 is protecting cartilage due to promoting an anabolic response to counteract the catabolic forces of mechanical instability. With respect to inhibiting IGFBP-5 proteolysis, we again were not able to measure the levels of intact IGFBP-5 and IGF-1 in the rat synovial fluid lavages due to dilution from the lavage technique and limited sample volumes. In the canine ACLT model, Clemmons et al. were able to demonstrate increases in both IGFBP-5 and IGF-1 in joint fluids from dogs treated with PB-145. This was associated with improved joint architecture, increased proteoglycan staining, and an increase in the depth of the chondrocyte layers within cartilage, consistent with a robust reparative response. 20 In order to more thoroughly understand the action of PB-145 in the rat MMT model, it may be necessary to study the association between levels of IGFBP-5 and IGF-1 and the carefully monitored levels of markers of cartilage synthesis and degradation during the course of OA disease progression.
In summary, our findings support the conclusion that inhibition of IGFBP-5 proteolysis is chondroprotective in experimental animal models of OA. Inhibiting IGFBP-5 proteolysis would be expected to increase the amount of intact IGFBP-5, thereby increasing the local IGF-1 concentration and enhancing IGF-1 actions in cell types that are present in the joint space. Combined with the recent demonstration that the IGFBP-5 proteolytic activity present in human OA synovial fluid is due to the complement protease C1s, this suggests that specific strategies to inhibit C1s activity may positively modulate the reparative response in human OA.
Footnotes
Acknowledgements
The authors thank Min Liu, Kam Fok, Larry Kahn, and Jim Zobel for providing reagents and QC of peptides; Micky Tortorella and Grace Munie for specificity testing; and Ruteja Barve for providing expression analysis.
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
The authors disclosed receipt of the following financial support for the research and/or authorship of this article: This work was fully funded by Pfizer Inc.
