Abstract
Background
Arsenic trioxide (ATO) is an effective chemotherapeutic for acute promyelocytic leukemia, but its clinical use is restricted due to cardiotoxic effects such as QT prolongation, arrhythmias, and myocardial injury. Natural agents with antioxidant and anti-inflammatory properties may offer cardioprotection.
Aim
To evaluate the effects of Clematis chinensis Osbeck ethanolic extract (CCE) against ATO-induced cardiotoxicity in rats.
Materials and Methods
Thirty Sprague-Dawley rats were randomized into five groups: Control, ATO (5 mg/kg), Carvedilol (10 mg/kg) + ATO and CCE (250 and 500 mg/kg) + ATO. All treatments were administered for 7 days. Feed intake, water intake, body weight, hemodynamic parameters, electrocardiography (ECG), serum cardiotoxic biomarkers, oxidative stress, and histopathology of heart tissue were assessed.
Results
ATO administrations caused significant weight loss, reduce food intake, water intake, and increased blood pressure and mean arterial pressure, and decreased heart rate. ECG abnormalities (prolonged QT, PR, QRS intervals, and ST elevation) were also evident. Serum cardiotoxicity markers (LDH, CK-MB) are elevated, and TBARS level increase, while antioxidant enzyme GSH levels decrease. Histology showed cytoplasmic vacuolization, edema, and myocardial fiber disruption. Treatment with CCE, at a higher dose (500 mg/kg), significantly improved cardiac function by restoring hemodynamics, normalizing ECG, reducing LDH and CK-MB, lowering TBARS, elevating GSH, and attenuating histopathological damage.
Conclusion
CCE cardioprotective effects against ATO-induced toxicity, primarily via antioxidant and anti-inflammatory mechanisms. Its efficacy is comparable to carvedilol, highlighting its potential as a supportive therapeutic strategy.
Introduction
Chemotherapeutics agents are very effective for the treatment of most of cancers, but there are many long-term complications associated with the treatment and it is either the advancement of secondary malignancies or cardiotoxicity. 1 The cardiotoxic effects of anticancer drugs may include structural damage that leads to cardiomyopathy, in addition to functional abnormalities such as arrhythmias or impaired systolic and diastolic function. 2 Heart failure, also referred to as left ventricular dysfunction, is one of the most dangerous cardiac side effects associated with certain anticancer treatments. 3 Antitumor therapies also likely affect non-cardiomyocytes (NCMs) in the heart. This direct action on NCMs may account for some of the drug’s cardiotoxicity; for instance, endothelial cells lose their barrier function as a result of increased permeability and myocardial damage. 4
Arsenic is a naturally occurring metalloid found in soil, water, and air, 5 was used in traditional Chinese medicine but its application is limited due to its toxicity. 6 Arsenic trioxide (ATO) used to treat acute promyelocytic leukemia (APL), and also used to treat both newly diagnosed and all trans-retinoic acid (ATRA)-resistant patients. 7 ATO’s clinical use has been restricted due to the cardiac toxicity it causes, which includes QT interval prolongation, Torsades de Pointes, ventricular tachycardia, and sudden cardiac death. 5 The primary mechanisms behind its harmful effects are the induction of oxidative stress and the production of reactive oxygen species (ROS). This oxidative stress causes detrimental changes in important macromolecules such as proteins, lipids, and DNA. Additionally, ROS can affect critical transcription factors like nuclear factor kappa B (NF-κB), activator protein 1, and nuclear factor erythroid 2–related factor 2 (Nrf2), which are vital to the cellular antioxidant defense system. 8
To prevent the post-treatment side effects, the world is now rushing towards the traditional medicines which are basically herbal in origin. Herbal drugs attracted attention as health beneficial foods and as source material for drug development. These are prescribed widely because of their effectiveness, fewer side effects and relatively low cost. 9 Based on the pharmacological properties, CCE is proposed for this study. Clematis Chinensisone of the ancestors of Clematidis Radix et Rhizoma, Osbeck is a significant medicinal plant. Southern China is home to a large population of Clematis Chinensis Osbeck (Ranunculaceae). 10 Its dried roots and rhizomes, referred to as “Wei ling xian” in traditional Chinese medicine, have long been utilized for their purported antitumor, analgesic, and anti-inflammatory properties. 11
Numerous bioactive components, such as organic acid 12 triterpene saponins, alkaloids, flavonoids, lignans, steroids, coumarins, macrocyclic compounds, and phenolic glycosides 13 have been identified by phytochemical investigations on Clematis Chinensis Osbeck. 11 Studies have shown that the ethanol extract contains these bioactive components, 13 and yellow wine treatments engendered complex pore structure to increase the contents of four active triterpenoid saponins of Clematis Chinensis Osbeck. 14 Furthermore, the active compounds and characteristic metabolites of Clematis Chinensis Osbeck after wine treatment are more abundant, and the enriched targets also include more targets that inhibit the development of inflammation compared to raw Clematis. 12 These studies suggest that the ethanol extract of Clematis has a better effect on inhibiting the development of inflammation and pain transmission.
Additionally, Clematichinenoside 15 and AR-6 is a triterpene saponin isolated from the root of Clematis Chinensis Osbeck. 16 Triterpene saponins, especially those derived from hederagenin and oleanolic acid, are thought to play a major role in the plant’s pharmacological activities, which include anti-oxidant, 17 anti-inflammatory, analgesic, antitumor, cardioprotective effects 11 and hypouricemia effect. 18 The anti-inflammatory properties of solvent-extracted triterpene saponin from Clematis chinensis have been shown to effectively suppress the inflammatory-related transcription factor NF-κB in rats. 19 Its therapeutic potential is further enhanced by the presence of indole alkaloids, particularly in the area of inflammation reduction. The understanding of Clematis Chinensis’s therapeutic value has grown as a result of ongoing research into its phytochemistry and biological activities. 20
The selection of CCE for this study is justified by its broad spectrum of pharmacological activities, underscoring its potential as a cardioprotective agent. To date, few studies have examined the role of CCE in mitigating chemotherapeutic agent–induced cardiac dysfunction. Therefore, the present study aims to evaluate its protective effects against ATO-induced cardiac dysfunction in rats and to compare its efficacy with carvedilol, a standard cardioprotective agent with antioxidant properties. 21
Material and Methods
Experimental Animals
Procured thirty adult (6-week-old) either gender Sprague Dawley rats weighing 150 ± 20 g from a register breeder. The Institutional Animal Ethical Committee provided the animal study protocol, which was assigned the approval number ASCB/IAEC/19/24/189. In accordance with Committee for Control and Supervision of Experiments with Animals (CCSEA) requirements in New Delhi, India, the rats were kept in a regular laboratory setting with a temperature of 23 ± 2 °C, a humidity of 40 ± 10%, and a natural light-dark cycle of 12 hours each. They were fed a standered chow pellet diet and provided unrestricted access to water. A week of acclimatization was given to the rats prior to the start of the tests. The rats were then divided into 5 study groups at random.
Induction of Cardiac Dysfunction and Study Plan
Thirty Sprague-Dawley rats of either sex were randomized into five groups (n=6) in each group, Group 1: Normal control (CONT, saline, p.o.); Group 2: negative control (Arsenic trioxide ATO, 5 mg/kg, i.p.); Group 3: positive control (Carvedilol CVD, 10 mg/kg, p.o. + ATO); Group 4: Clematis chinensis Osbeck ethanolic extract (CCE-I 250 mg/kg +ATO); Group 5: (CCE-II 500 mg/kg). In order to induce cardiac disorders, Arsenic trioxide (ATO, purity ≥99%, Cat# A1013, Sigma-Aldrich, USA) was dissolved in distilled water and administered intraperitoneally at 5 mg/kg once daily for 7 days to induce cardiac dysfunction. Carvedilol (CVD, Cat# C3993, Sigma-Aldrich) was dissolved in saline and given orally at 10 mg/kg. The ethanolic extract of Clematis chinensis Osbeck extract (CCE) was prepared by macerating dried roots in 70% ethanol for 72 h, followed by filtration and evaporation under reduced pressure; the yield was 12.5% (w/w). CCE was suspended in 0.5% carboxymethylcellulose and administered orally at 250 or 500 mg/kg. All treatments (CVD or CCE) were given 6 h before ATO injection each day for 7 consecutive days. On a daily basis closely assess the animal’s water, food consumption and changes in body weight and also observed their hemodynamic parameters, including blood pressure (BP), mean arterial pressure (BP), heart rate (HR). After 7 days, the rats were anesthetized with 0.75 to 1.5 g/kg Urethane (Cat# U2500, Sigma-Aldrich). The electrocardiogram (ECG) was performed in anesthetized rats. At the end of the final treatment, the rats were put on overnight fasting after blood sample were taken for biochemical analysis. Animals were sacrifice, and the heart was separated for antioxidant and histological investigation.
Measurement of Hemodynamic and Electrocardiogram (ECG)
Rats were acclimatized and placed in restrainers for blood pressure measurement. A tail-cuff was fitted at the base of the tail with a pulse transducer positioned distally. The cuff was inflated to occlude blood flow and then slowly deflated, with systolic blood pressure determined at the point when the pulse reappeared. Readings were obtained using the CODA® system(Kent Scientific, Torrington, CT, USA) connected to the pulse transducer and related apparatus.
The rats were anesthetized, and then three needle electrodes were attached to the right arm, left arm and left leg of the rats, and the ECG patterns were recorded using dual bioamplifier (AD Instrument, Power Lab) to monitor changes in ECG heart rate.
Evaluation of Hematological Parameters (LDH and CK-MB)
Following the normal procedure, the serum was separated from the blood by centrifugation, and the Lactate Dehydrogenase (LDH) levels in the serum were subsequently determined using Reckon Diagnostics kits(Cat# CKMB-025, AGD Biochemicals, India). The auto analyser used the rate at which the LDH enzyme catalyses the conversion of lactate to pyruvate to determine the LDH concentration.
The standard procedure was followed to assess the serum’s Creatine kinase-MB (CK-MB) levels using AGD Biochemical’s kits. By monitoring the rate at which creatine phosphate is transformed into creatine, which is catalysed by the enzyme CK-MB, the auto analyser was able to determine the CK-MB content.
Tissue Antioxidant Level in Rat Heart (MDA and GSH)
A useful test for lipid oxidation is the measurement of malondialdehyde (MDA), a byproduct of the breakdown of polyunsaturated fatty acids in cell membranes. In this procedure, 2 ml of cold 1.15 M KCl buffer pH 6.0 were used to homogenize the cardiac samples was used to measure lipid peroxidation. 22 MDA levels were determined by thiobarbituric acid reactive substances (TBARS) method.
Reduced glutathione (GSH) is an important endogenous antioxidant and non-protein thiol that balances damage caused by free radicals. The tissue homogenate (in 0.2 M phosphate buffer pH 7.6) was obtained in order to measure the GSH level. Spectrophotometer and GSH level were ascertained using the standard curve of reduced GSH, which was achieved with GSH concentrations of 0.2, 0.4, 0.6, 0.8, and 1.0 nm. 23
Histopathology of Heart
Histopathology of the heart was done to determine the tissue damage inflicted by the inducing substance in the animals from the study groups. After the animals were sacrificed, the heart was removed and fixed for at least six hours in a 10% neutral buffered formalin solution. Heart tissue was prepared on slides using hematoxylin and eosin staining. A photomicroscope(Olympus BX53, Tokyo, Japan) was used to capture images of the pre-made slides for analysis.Scale bars (50 μm) were added to all images using ImageJ software to indicate magnification (×400).
Statistical Analysis
The data were present as mean ± SEM and the data from each group was analysed using GraphPad Prism (Software, version 10.5.0) using one-way ANOVA followed by Tukey’s test and two-way ANOVA followed by Bonferroni post-hock tests. A significant p-value was defined as p<0.01.
Results
Effect of Clematis chinensis Osbeck extract (CCE) on Feed Intake, Water Intake and Body Weight of Rats
ATO-treated rats showed a significant (p<0.01) decline in feed intake, water intake and body weight by day 7 compared to the control group. Treatment with the standard drug carvedilol (CVD+ATO) significantly reversed these declines, restoring parameters toward normal levels (p<0.01 vs. ATO), whereas treatment with CCE(H) effectively restored these parameters toward normal levels, comparable to the ATO-treated group, indicating that CCE(H) provides a similar level of protection against ATO-induced wasting (Figure 1(A)-(C)). (A, B and C) effect of CCE on feed intake, water intake and body weight.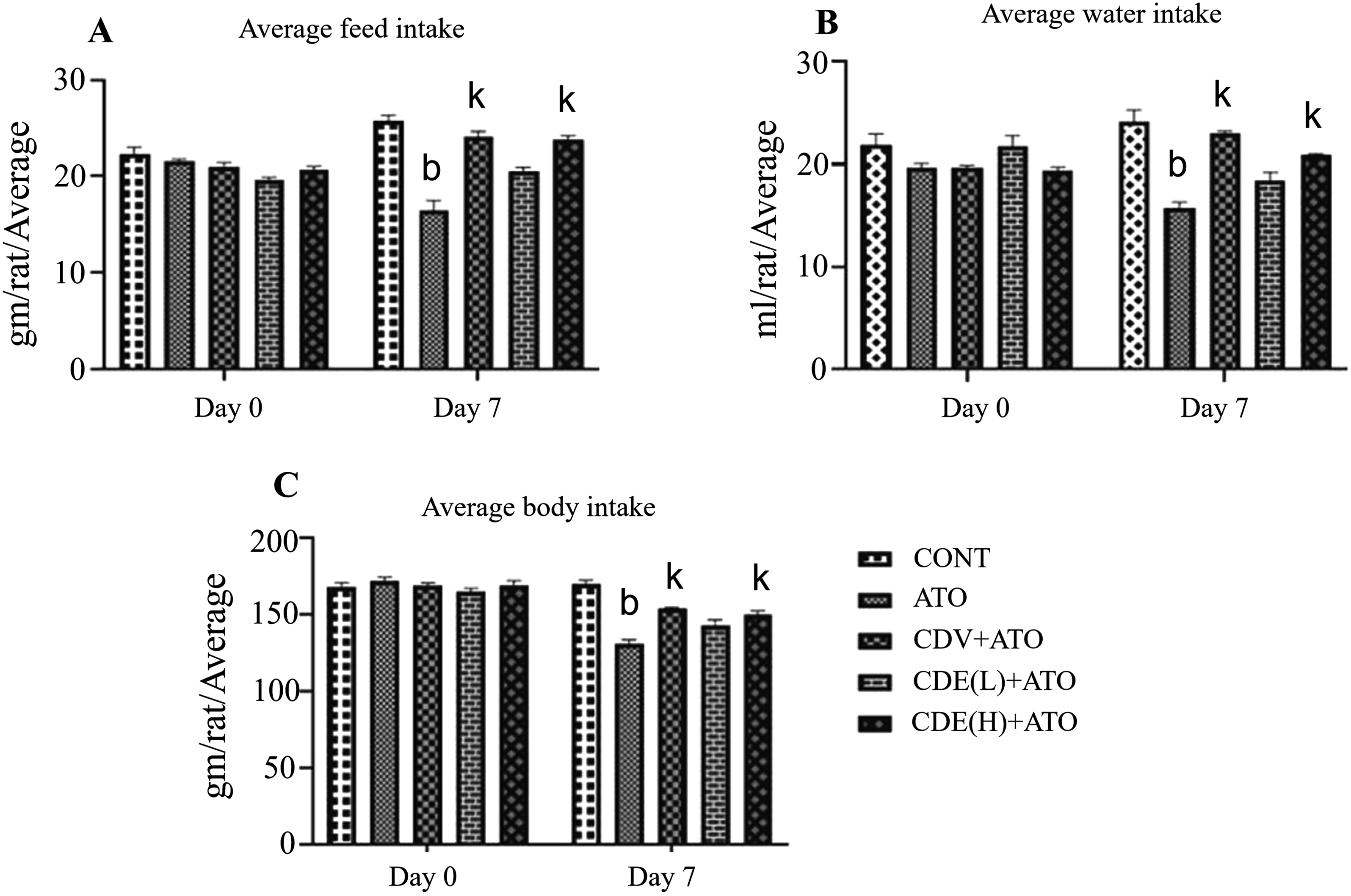
Effect of CCE on Hemodynamic Parameters of Rats
Effect of CCE Extract on Hemodynamic Parameters of Rat

Values are expressed as mean ± SEM (n=6).
Where c(p<0.001), when compared with CONT; l(p<0.001), when compared with ATO; n(p<0.01), when compared with CVD; y(p<0.01), when compared with CCE(L) and CCE(H) (Two-way ANOVA followed by Bonferroni’s post hoc analysis).
Effect of CCE on ECG Parameters
Effect of CCE on Electrocardiogram Parameters

Values are expressed as mean ± SEM (n=6). c(p<0.001), when compared with CONT; l(p<0.001), when compared with ATO; n(p<0.01), when compared with CVD;y(p<0.01), when compared with CCE(L) and CCE(H). Using one-way ANOVA followed by Tukey’s test.

Electrocardiogram of rat: (A). (CONT) normal rat showing normal ECG; (B). (ATO) intoxicated rats showed elongation of RR interval ( ) ventricular premature beats shown by (
) ventricular premature beats shown by ( ), inversion in QRS wave shown by (
), inversion in QRS wave shown by ( ); (C). (CVD+ATO) showed normal ECG as compared to ATO group; (D). (CCE(L)+ATO) showed less ventricular premature beats as compared to ATO group; (E). (CCE(H)+ATO) showed significant decreased ventricular premature beats and no inversion In QRS wave
); (C). (CVD+ATO) showed normal ECG as compared to ATO group; (D). (CCE(L)+ATO) showed less ventricular premature beats as compared to ATO group; (E). (CCE(H)+ATO) showed significant decreased ventricular premature beats and no inversion In QRS wave
Effect of CCE on LDH, CK-MB, GSH and TBARS
Effect of Clematis chinensis Osbeck extract on Antioxidant Enzyme and Oxidative Stress Markers Level
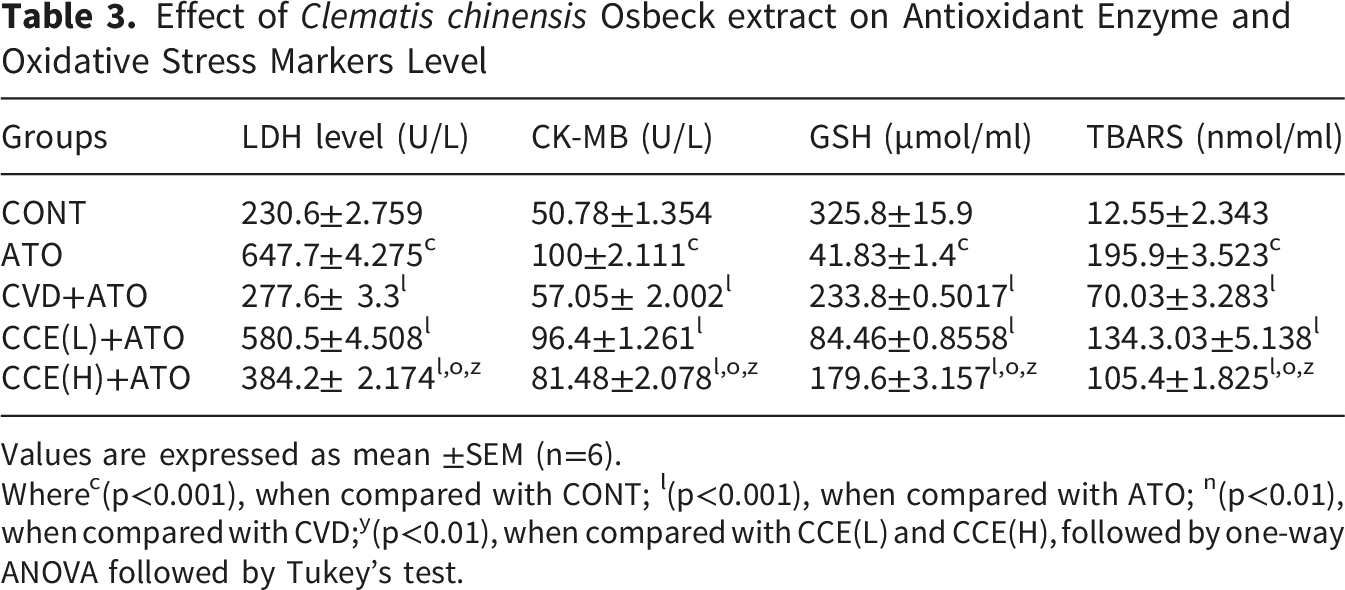
Values are expressed as mean ±SEM (n=6).
Wherec(p<0.001), when compared with CONT; l(p<0.001), when compared with ATO; n(p<0.01), when compared with CVD;y(p<0.01), when compared with CCE(L) and CCE(H), followed by one-way ANOVA followed by Tukey’s test.
TBARS activity was significantly increased in the ATO group (p < 0.001) compared with the control group. Treatment with a higher dose of CCE extract significantly reduced TBARS levels (p < 0.001) on day 7 in heart tissue compared to the ATO group (Table 3). GSH levels were markedly decreased in the ATO group, while treatment with a higher dose of CCE extract significantly (p < 0.001) restored antioxidant levels in heart tissue. CCE(H) is less effective than CVD in restoring oxidative stress indicators. Nevertheless, CCE(H) exhibits a clear dose-dependent effect and provides significant antioxidant and cytoprotective benefits (Table 3).
Effect of Clematis chinensis Osbeck extract on Histopathology
The results of the histopathological analysis of the myocardial tissues, the CONT group’s H&E-stained heart sections showed healthy cardiomyocyte structures, whereas the ATO-induced cardiotoxicity group’s sections showed cytoplasmic vacuolization, soft perivascular edoema, intermyocardial edoema, and muscle separation. Treatment with carvedilol (CVD+ATO) markedly attenuated these pathological changes, with sections showing near-normal architecture. Meanwhile, the treatment methods utilizing CCE(L)+ATO and CCE(H)+ATO have improved these signs of ATO-induced cardiac dysfunction. Among them,CCE(H) exhibits a potent protective effect against ATO-induced cardiac injury, similar to the effect of carvedilol (Figure 3). Representative photomicrographs of heart tissue sections (H&E, ×400, Scale Bar=20 μm)
Discussion
Arsenic trioxide (ATO) has been used in chemotherapy mainly for the treatment of Acute Promyelocytic Leukemia (APL) as one of the most effective anticancer agents. 24 Despite its efficacy, the application of Arsenic trioxide is greatly compromised by its cardiotoxicity. 25 Significant efforts have been made in the past decades aiming at prevention or attenuation of ATO- induced cardiotoxicity but so far the efficacy of these intervention is limited.7,26,27 The present study established a cardiotoxicity model in rats by intraperitoneal injections with ATO (5mg/kg).28,29
In the present study, we explored the effects of ethanolic extract of Clematis Chinensis Osbeckat the dose of 250mg/kg, 500mg/kg 30 against the detrimental effects of ATO on normal parameters, hemodynamic parameters, ECGs, histopathological alterations, serum cardiac markers, oxidative stress parameters by using a rat model of ATO-induced cardiotoxicity. The ethanolic extract of CCEcontain triterpenoid saponins, 31 phenolic glycosides, flavonoids, alkaloid, 32 and terpenoids has reported anti-inflammatory 15 and antioxidant properties, 17 as evident from various research studies. Clematichinenoside is a triterpenoid saponin isolated from the roots of Clematis Chinensis exhibited antioxidant, 16 anti-inflammatory. 11 Although detailed phytochemical profiling is not included here, our findings are consistent with previous reports attributing the cardioprotective effects to these constituents.
In this study, rats exposed to ATO, body weight, feed and water consumption decreased significantly (p < 0.01). CCE increased these parameters (p < 0.01). CCE showed potential in enhancing body weight, feed intake and water intake.
Arsenic trioxide triggers cardiotoxicity through oxidative stress (excess ROS, reduced antioxidants) 28 which activates NF-κB/MAPK signaling, leading to inflammation, 29 mitochondrial dysfunction, and apoptosis (altered Bax/Bcl-2 ratio, caspase-3 activation). 33 It further disturbs ion channels and Ca2+ regulation, causing prolonged repolarization, arrhythmias, structural remodeling, 34 and ultimately impaired cardiac performance. Previous studies showed that rats treated with ATO develop open myocardial cell damage, elevated systolic blood pressure, diastolic blood pressure, mean blood pressure and decreased heart rate leading to cardiac dysfunction. 35 In this study ATO administration increases the systolic blood pressure, diastolic blood pressure, mean arterial pressure (p < 0.01), and decreased heart rate(p < 0.01). Treatment with CCE significantly reduces the systolic blood pressure, diastolic blood pressure, means arterial pressure (p < 0.01) and significantly increased heart rate (p < 0.01) in our rat model.
The ATO-induced myocardial damage was also evident in the present study as an alteration in the ECG pattern. QT prolongation is considered a marker of cardiac dysfunction. 35 In this study, CCE significantly (p < 0.01) reduced the ATO-induced QT prolongation. CCE also reversed ST-Height, QRS interval and PR interval. Notably, high-dose CCE (500 mg/kg) restored ECG parameters to near-normal values, with effects comparable to carvedilol, although carvedilol showed slightly superior improvement in ST segment elevation. These observations further confirm the protective effects of CCE against ATO-induced cardiac dysfunction.
The biochemical finding also revealed that CCE prevented the ATO triggered derangement of cardiac dysfunction enzymes and other parameters (e.g. serum cardiotoxicity biomarkers LDH and CK-MB while levels of antioxidant enzyme decrease, abnormal hemodynamic parameters, oxidative stress and redox imbalance). The CK-MB is an isoform of creatine kinase that is expressed in the heart muscle. The LDH is present in the heart as well as other muscles and catalyses’ conversion of pyruvate to lactate and contribute energy metabolism. 36 Elevated serum LDH is an indicator of cellular damage. Reduction of serum CK-MB and LDH (p < 0.01) by CCE indicates its cytoprotective potential.
Accumulating evidence suggests that oxidative stress serves an important role in ATO-induced cardiac damage. 37 MDA, a typical product of LPO, is formed by the oxidation of polyunsaturated fatty acids caused by ROS and serves as a directional biomarker of oxidative stress that indirectly reflects the degree of cardiomyocyte damage. 38 ATO treatment induced significant lipid peroxidation (MDA) depletion of antioxidant defenses present in heart muscles including GSH. 39 Previous studies have reported cardio protective effects of its constituents, such as Clematichinenoside, primarily in ischemia/reperfusion injury through antioxidant and anti-apoptotic mechanisms. 40 Our results broaden this scope to arsenic-induced cardiac damage and show parallels with other natural agents, including α-lipoic acid, 28 curcumin, 41 and resveratrol. 42 All of which mitigate ATO cardiotoxicity by reducing oxidative stress and regulating apoptosis. In the present study, we found that ATO raised MDA levels in myocardial tissue (p < 0.01), while CCE significantly lowered MDA (p < 0.01) and elevated GSH, indicating improved antioxidant status. These effects are likely mediated through activation of the Nrf2 pathway and inhibition of NF-κB, as suggested by previous studies on saponin-rich extracts. 17 However, further molecular investigations are needed to confirm these pathways.
In the present investigation the histopathological examination of myocardial tissue shows there is remarkable change in myocardial tissue, inflammation and separation of myocardial fibers in ATO group. With the treatment of CCE, decreases in myocardial tissue damage, inflammation and separation of myocardial fibers. Thus, the present study shows that CCE at higher dose protects heart against ATO induced Cardiac dysfunction in rats.
Although carvedilol exhibited slightly greater efficacy in some parameters, CCE at 500 mg/kg showed comparable overall cardioprotection. This finding is promising considering the natural origin, low toxicity profile, and multi-targeted action of CCE. The dose of 500 mg/kg was selected based on previous study 12 and our preliminary dose-range study, and no adverse effects were observed at this dose.
This study has limitations, the molecular mechanisms underlying CCE’s effects were not fully elucidated, future studies should examine key signaling pathways. Besides, detailed phytochemical characterization of the extract is required to identify the active compounds responsible for the observed effects. Futhermore, the healthy rats, while it appropriate for mechanistic cardiotoxicity studies, does not fully mimic the clinical scenario in cancer patients, therefore, further studies in tumor-bearing models are warranted.
Conclusion
In conclusion, the ethanol extract of Clematis chinensis demonstrated significant cardioprotective effects against ATO-induced cardiac dysfunction in rats, as evidenced by improvements in body weight, hemodynamic parameters, ECG abnormalities, serum biomarkers, oxidative stress, and histopathology. The protective effects are likely mediated through antioxidant and anti-inflammatory mechanisms, with efficacy comparable to carvedilol at the higher dose tested. These findings support the potential of CCE as a supportive therapeutic strategy to mitigate chemotherapy-induced cardiotoxicity. However, further studies are needed to identify the active constituents and elucidate the precise molecular mechanisms.
Footnotes
Acknowledgement
The authors are gratefull to the institute and management committee member of Amar Shaheed Baba Ajit Singh Jujhar Singh Memorial College of Pharmacy, BELA (Ropar) for granting the necessary learning access.
Ethical Considerations
The animal study protocol (ASCB/IAEC/19/24/189) was approved by the Institutional Animal Ethical Committee Amar Shaheed Baba Ajit Singh Jujhar Singh Memorial College of Pharmacy, Bela, Ropar, Punjab-140111, and all experiments were conducted in accordance with the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CCSEA), New Delhi, India.
Consent to Participate
This article does not include any studies involving human subjects; therefore, informed consent is not applicable.
Author Contributions
All authors contributed equally to this work. Qian Nie: Conceived the study and designed the experiments; Mingjun Han: Curated and analyzed the data; Haseeb Sattar: Revised the manuscript and interpreted the data; Jue Zhao: Performed supportive experiments and critical evaluation; Ramneet Kaur: Conducted experiments and prepared the initial draft; Ajay Singh Kushwah: Supervised the study, implemented animal experiments, and contributed to manuscript writing; Navjot Kaur: Provided experimental and analytical support; Wen Xie: Designed and supervised the experiments as co-corresponding author; and Ailing Huang: Coordinated the study, proofread the manuscript, and served as corresponding author.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Sichuan Science and Technology Program Key research and development project (2022YFS0395); Sichuan Provincial Administration of Traditional Chinese Medicine, Special Project for Scientific and Technological Research, No. 2069999. This project also funded with: Project Description of Science and Technology Research Special Project, Sichuan Provincial Administration of Traditional Chinese Medicine, No 2024MS165, 25ZDIZX024.
Declaration of Conflicting Interests
The authors declared no potential conflict of interest with respect to the research, authorship, and/or publication of this work.
Data Availability Statement
All relevant data are contained within the article. The original contributions presented in the study are included in the article and supplementary material. Further data can be provided upon request to the corresponding author.
