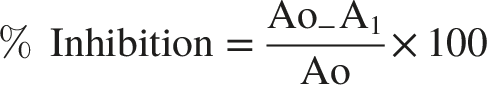Abstract
Introduction
Oxidative stress, resulting from excessive production of reactive oxygen species and thereby playing a critical role in the development of chronic diseases, derives a growing interest in medicinal plants as a source of natural antioxidant phytochemicals. Acokanthera schimperi (A. DC.) Schweinf., is a traditionally important medicinal plant that has been poorly studied for the phytochemical composition and antioxidant potential of its stem bark.
Objectives
The aim of the present study was to evaluate the phytochemical constituents using gas chromatography‒mass spectrometry (GC‒MS) analysis and determine the antioxidant activity of the stem bark of A. schimperi crude extracts.
Methods
The powdered stem bark was sequentially extracted with ethyl acetate and methanol using a Soxhlet apparatus. Phytochemical screening tests were employed by standard protocols to qualitatively identify phytochemicals. The crude extract was analyzed by GC‒MS, and its antioxidant activity was assessed using the DPPH assay.
Results
Phytochemical screening of stem bark extracts of A. schimperi showed the presence of phenols, alkaloids, steroids, terpenoids, and glycosides in the methanolic extract, while the ethyl acetate extract contained terpenoids, glycosides, and steroids. From GC‒MS analysis of ethyl acetate extracts identified trans-2-methyl-4-n-pentylthiane, S,S-dioxide (31.06%), trans-2-Methyl-4-n-butylthiane, S,S-dioxide (30.57%) and N-(5-azidopentyl)-4-methyl-4-vinylazetidin-2-one (6.82%); and n-hexadecanoic acid (5.01%) as a major components. The methanolic extract was dominated by Myo-Inositol, 4-C-methyl (49.40%); 2-Azido-2,4,4,6,6-pentamethylheptane (8.96%); and Catechol (4.041%). The methanol extract exhibited strong antioxidant activity, achieving 96.57±0.01% inhibition at a concentration of 100 μg/mL, comparable to the control ascorbic acid (97.10±0.01%), whereas the ethyl acetate extract showed moderate antioxidant activity with 48.14±0.10% inhibition.
Conclusions
This finding provides the scientific evidence on the natural antioxidant phytochemical constituents of A. schimperi stem bark extracts.
1. Introduction
Highly reactive free radicals and other reactive oxygen species with physiological activity are naturally present in the cells. However, if the formation of these reactive compounds is greater than that of antioxidants have negative effects on the immune system. An imbalance favoring oxidants over antioxidants leads to oxidative stress, which is a key cause of several diseases, such as cancer, cardiovascular disease, Alzheimer’s or Parkinson’s disease, or cell aging.1,2 Phytochemicals from medicinal plants are used as alternative therapeutic agents to treat a range of diseases caused by oxidative stress. 3 Mainly, natural antioxidant-phenolic compounds are effective as antioxidant agents. 4 Therefore, there has been growing interest in identifying new sources of antioxidant potential from medicinal plants. 5
Ethiopia is a significant source of medicinal plant species and serves as the hub of diversity for such plants. Research indicates that medicinal plants play a crucial role in providing healthcare in Ethiopia. It is worth noting that 70% of the human population and 90% of the livestock population rely on traditional medicine in Ethiopia, much like other developing countries in sub-Saharan Africa. 6 Medicinal plants and knowledge of their usage are vital for human and animal healthcare in Ethiopia, making them highly sought after due to cultural traditions. 7
Acokanthera schimperi (A. DC.) Schweinf., which belongs to the Apocynaceae family, is a popular African arrow poison plant found in Ethiopia. It is called “Merenz” in Amharic and is used in traditional Ethiopian medicine for treating hepatitis (leaves and roots), hemorrhoids (leaves) and wounds (leaves). 8 Its young leaves are also crushed and applied as a treatment for skin cancer. 9
Biological activity studies on different extracts of A. schimperi leaves and stem bark indicated antibacterial and antifungal activities. 10 The antimalarial activity test showed that this plant had significant antiplasmodial activity, as evidenced by its ability to suppress P. berghei infection in mice in a dose-dependent manner, which may partly justify the claim of traditional practitioners about the use of this plant against malaria. 11 Furthermore, an in vitro antioxidant activity test of the ethanolic extract from leaves of A. schimperi showed maximum inhibition of DPPH at 80%. 12
Organic extracts from the leaves of A. schimperi were examined qualitatively and showed that flavonoids, terpenoids, phenolic compounds, tannins, saponins, glycosides, and phytosterols were present.12,13 Another study investigated the chemical fingerprint profile of glycosides, steroids, phenols, and tannins of the ethanolic extract of A. schimperi using a high-performance thin-layer chromatography (HPTLC) technique, which confirmed that the ethanolic extract of A. schimperi contained secondary metabolites such as glycosides, steroids, phenols, and tannins. 14
Previous studies on the isolation of compounds from leaves of A. schimperi have reported eight triterpenoids, including a new triterpenoid ester, lupan-20-ol-3(β)-yl 3-hydroxyoctadecanoate, afforded, along with seven known triterpenoids, lupeol, 28-nor-urs-12-ene-3β,17β-diol, ursolic aldehyde, 3β-hydroxyoleana-11,13(18)-dien-28-oic acid, alagidiol, oleanolic acid and ursolic acid lactone. 15 In addition, Abera et al reported that from the root of A. schimperi afforded oleic acid, lupeol, dihydroferulic acid, acovenosigenin A- 3-O-α-L-rhamnopyranoside and sucrose were identified. 16
The traditional knowledge regarding the use of this plant’s use is well-known, but there is no enough scientific research to support its medicinal claims. Therefore, it is important to further study the chemical composition and biological activity of different parts of the plant. However, existing literature does not describe the phytochemical investigations and biological activities the stem bark of A. schimperi. This report is the first time to address extracts from the stem bark of A. schimperi. The study aimed to examine the phytochemicals, GC-MS analysis, and in vitro antioxidant activity of the methanolic and ethyl acetate crude extracts from the stem bark of A. schimperi.
2. Materials and Methods
2.1. Chemicals and Instrumentations
All chemicals used for this study were methanol (Sisco Research Laboratories Pvt. Ltd, New Mumbai India), ethyl acetate (Alpha chemica, India), 2, 2-Diphenyl1-Picryl Hydrazyl (Alpha chemical, India)), sodium nitrite (Loba chemie, Pvt, Ltd, India), sodium hydroxide (Loba Chemie Pvt Ltd, Andheri, Mumbai India) and helium gas. All reagents were analytical grade. The instruments used in this study included UV–visible spectroscopy (Analytic Jena SPECORD 50, Germany), GC–MS (Agilent 8890 GC, Germany coupled with 5977B MS, USA), a refrigerator (Lec 1602, England), and a grinder (IKA-WRKE, GMBH & CO.KG D-79219 Staufen, Germany).
2.2. Collection, Identification, and Preparation of Plant Materials
The stem bark of A. schimperi was collected from Berehet District, North Shoa Zone, Ethiopia, from well-grown plants grown under natural field conditions. The plant was authenticated by a botanist by Mr. Melaku Wendaferash at the Botany Department of Addis Ababa University, and the specimen was stored (Voucher no: AW001) in the National Herbarium of Ethiopia, Addis Ababa University, Addis Ababa, Ethiopia. The bark was washed with water without squeezing to remove trash and dust particles and then air-dried at room temperature for two weeks. The air-dried stem bark was chopped into small pieces and ground using an electrical grinder.
2.3. Extraction of the Stem Bark of A. schimperi
20 grams of air-dried and powdered stem bark were placed in a thimble and extracted sequentially with ethyl acetate (300 ml) twice for 8 hours using a Soxhlet apparatus. The resulting extract mixture was concentrated in a rotary evaporator. The remaining marc was then extracted with methanol using a similar procedure. The extracts were concentrated using a rotary evaporator, dried until a constant weight was reached, and stored at 4 °C in a refrigerator for further analysis. 17
2.4. Phytochemical Screening
Phytochemical screening tests were performed to determine the class of compounds present in the crude stem bark extract of A. schimperi following standard protocols. Qualitative screening of ethyl acetate and methanolic extracts of stem bark of A. schimperi was performed for the identification of various classes of active chemical constituents, such as phenols, alkaloids, saponins, flavonoids, glycosides, and terpenoids, in the plant.
2.4.1. Test of Phenols
1 mL of the extracts, 3-4 drop of 5% FeCl3 (w/v) were added. Formation of bluish black color indicates the presence of phenol. 18
2.4.2. Test of Saponins
3 mL of the extract in a test tube was mixed with 5 mL of distilled water and shaken vigorously for 2 minutes. The formation of a stable form or froth established the presence of saponins. 19
2.4.3. Test for Flavonoids
2 mL of extract in a test tube was mixed with 2-3 drops of 2% NaOH to form an intense yellow color. Then, a few drops of dilute HCl were added dropwise, which caused the intense yellow color to become colorless, indicating the presence of flavonoids. 18
2.4.4. Test for Alkaloids
2 mL of extracts in test tube was mixed with 2 drops of 1.5% HCl and filtered. 2 ml filtrate of plant extracts; 2 ml of Wagner’s reagent was mixed. The formation of a reddish brown precipitate indicated the presence of alkaloids. 18
2.4.5. Test for Steroids
A red color was produced in the lower chloroform layer when 2 mL of the extract dissolved in 2 mL of chloroform and 2 mL of concentrated sulphuric acid was added to the test tube, indicating the presence of steroids. 18
2.4.6. Test for Glycosides
2 mL of extract was treated with 1 mL of glacial acetic acid and two drops of 5% (w/v) FeCl3. Then, 2 mL of concentrated H2SO4 was carefully dispensed along the walls of the test tube. The development of a reddish-brown colored ring at the intersection of the two layers indicated the presence of glycosides. 19
2.4.7. Test for Terpenoids
The test for terpenoids was conducted by mixing 3 ml of the extract with 2 mL of CHCl3 in a test tube. Thereafter, 2 mL of concentrated H2SO4 was added gently to form a ring layer that interfaced with a reddish-brown color, thus indicating the presence of terpenoids. 18
2.5. Gas Chromatography‒Mass Spectrometry Analysis
Gas chromatography mass spectrometry (GC–MS) was used to identify the phytochemical components of A. schimperi extracts, using a previously reported method. 20 The chemical composition of the photochemical constituents of A. schimperi was analyzed by gas chromatography (Agilent 7890B) with a mass spectroscopic detector (Agilent 5977MDS) equipped with an HP-MS capillary column (30 m × 0.25 mm; 0.2 μm film thickness with 5% phenylmethyl silicon). The oven temperature was programmed from 50 °C to 200 °C at 4 °C/min and held for 5 min. Helium (99.99%) was used as the carrier gas at a constant flow rate of 1.0 mL/min. Samples (1 µL) were injected in split mode (1:20) after a solvent delay of 5 min. The injector and detector temperatures were set at 250 °C. Mass spectra were recorded under electron ionization (70 eV) with a transfer line temperature of 240 °C, ion source temperature of 230 °C, and a mass scan range of 45–500 amu. The identification of components was based on NIST libraries, as well as a comparison of their retention indices. The constituents were identified after comparison with those available in the computer library (NIST) attached to the GC‒MS instrument, and the results obtained were tabulated.
2.6. Antioxidant Activity
To evaluate the radical scavenging activity of the stem bark of A. schimperi, a 0.2 mmol solution of DPPH in methanol was prepared. The extracts from the stem bark and leaves were diluted to concentrations of 500, 250, 125, and 62.5 µg/mL. Then, 1 mL of each extract solution was added to 4 mL of DPPH solution (prepared in methanol) to obtain final concentrations of 100, 50, 25, and 12.5 µg/mL. For the control, 1 mL of the DPPH solution was added to 4 mL of methanol, while 4 mL of methanol was used as a blank. The mixtures were shaken and left to stand for 30 minute in the dark at room temperature, and the absorbance was recorded at 517 nm using a double beam UV‒Vis spectrophotometer. Ascorbic acid was used as a reference standard at the same concentrations. The percentage inhibition of DPPH radical was determined using the following equation:
2.7. Statistical Analysis
Data were expressed as means ± standard deviation (n =3). The statistical analysis was carried out using GraphPad Prism 10.6.1 version statistical software. Two-way ANOVA was applied to the sample. The IC50 values were obtained by nonlinear regression for the purpose of interpolating values from standard curves.
3. Results
3.1. Extract Yields
Extractive Values of Different Extracts of A. schimperi Stem Bark

3.2. Phytochemical Screening Test
Phytochemical Screening of Stem Bark Extracts

(+) indicates present (-) indicates absent.
Phytochemical analysis of A. schimperi stem bark revealed both methanolic and ethyl acetate extracts contained terpenoids and glycosides, while saponins and flavonoids were absent. The methanol extract additionally contained phenols and alkaloids. Steroids were detected only in ethyl acetate extract (Table 2).
3.3. GC‒MS Analysis
GC‒MS Spectral Analysis of the Ethyl Acetate Crude Extract of the Stem Bark of A. schimperi


GC‒MS chromatography of the ethyl acetate stem bark extract of A. schimperi
An analysis of the ethyl acetate stem bark of A. schimperi using GC‒MS revealed the presence of trans-2-methyl-4-n-pentylthiane, S,S-dioxide (31.05%), trans-2-methyl-4-n-butylthiane, S,S-dioxide (30.56%), N-(5-azidopentyl)-4-methyl-4-vinylazetidin-2-one (6.82%), n-hexadecanoic acid (5.01%), Methyl 12,13-tetradecadiethenoate (2.03%), 4-n-Hexylthiane, S,S-dioxide (0.90%), Hexadecanoic acid, methyl ester (0.89%), Propanoic acid, ethyl ester (0.85%), and Phenol, 2,6-bis(1,1-dimethylethyl)-(0.43%). Structure of major components of methanolic stem bark extract of A. schimperi, as shown in Figure 2. Structure of major components of ethyl acetate stem bark extract of A. schimperi
GC‒MS Spectral Analysis of the Methanol Crude Extract of the Stem Bark of A. schimperi

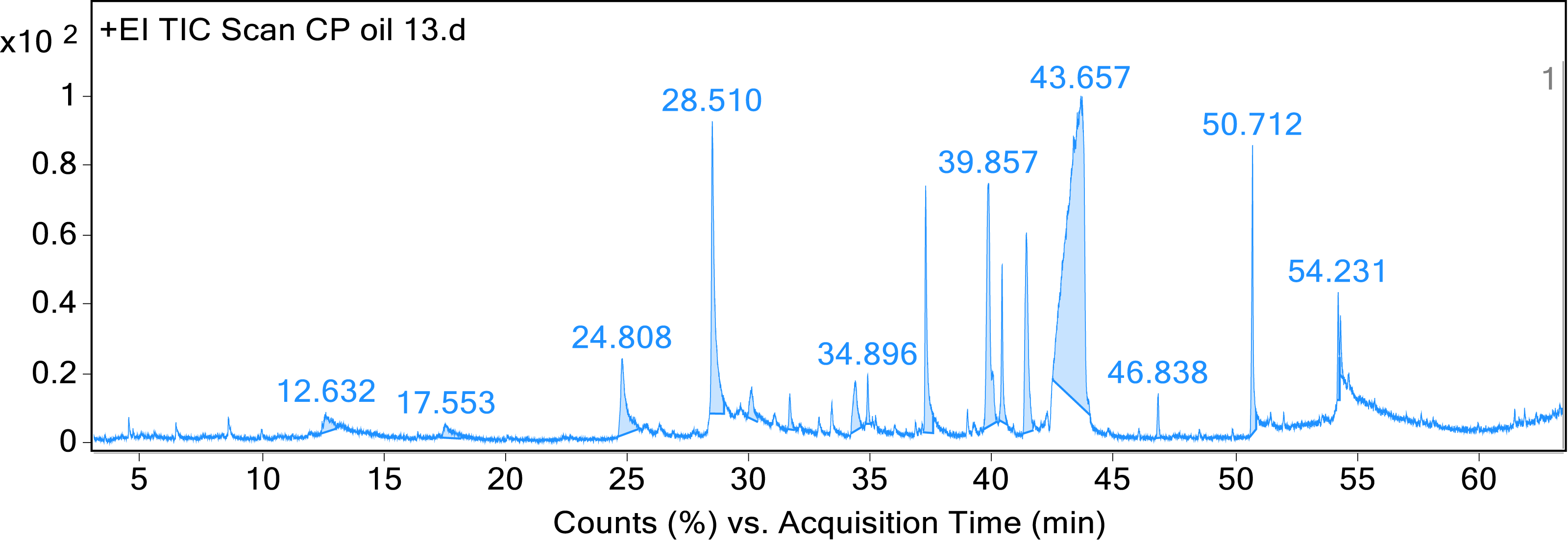
GC‒MS chromatography of the methanolic stem bark extract of A. schimperi
GC–MS analysis of the methanolic extract of stem bark extract of A. schimperi showed the presence of major volatile compounds viz. Myo-Inositol, 4-C-methyl- (49.40%); 2-Azido-2,4,4,6,6-pentamethylheptane (8.96%); n-Hexadecanoic acid (4.26%); and, Catechol (4.04%). The other minor components are also including; Methyl 12,13-tetradecadienoate (1.34%); Phosphonic acid, (p-hydroxyphenyl)-(1.19%) 2-Amino-3-methylpyridine-N-oxide (1.06%) and Phenol, 2,6-dimethoxy-(0.70%). Major components of methanol stem bark extract of A. schimperi are shown in Figure 4. Structure of major components of methanolic stem bark extract of A. schimperi
3.4. Antioxidant Activity
Free Radical Scavenging Activity of A. schimperi Stem Bark Extracts
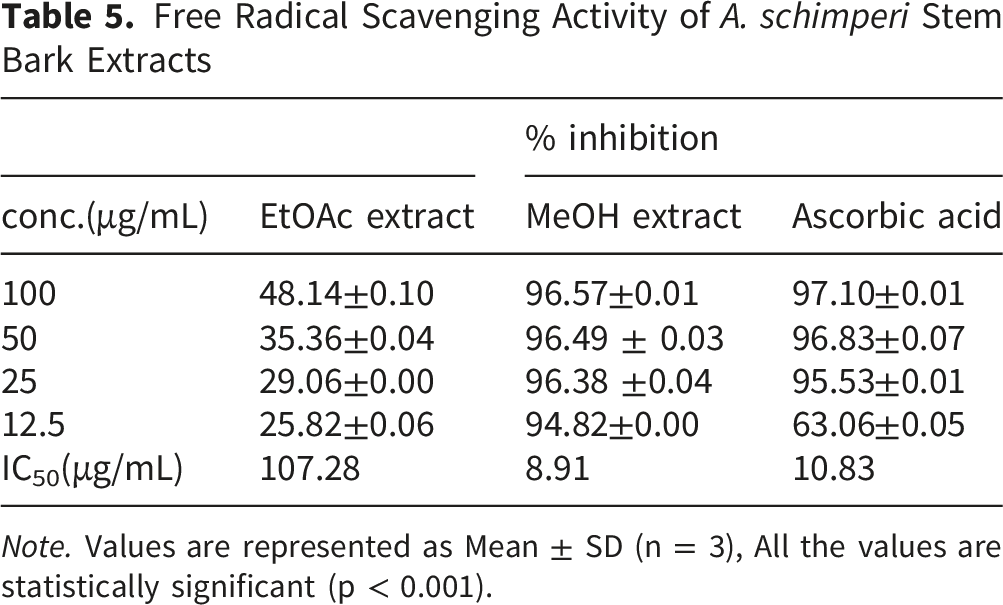
Note. Values are represented as Mean ± SD (n = 3), All the values are statistically significant (p < 0.001).
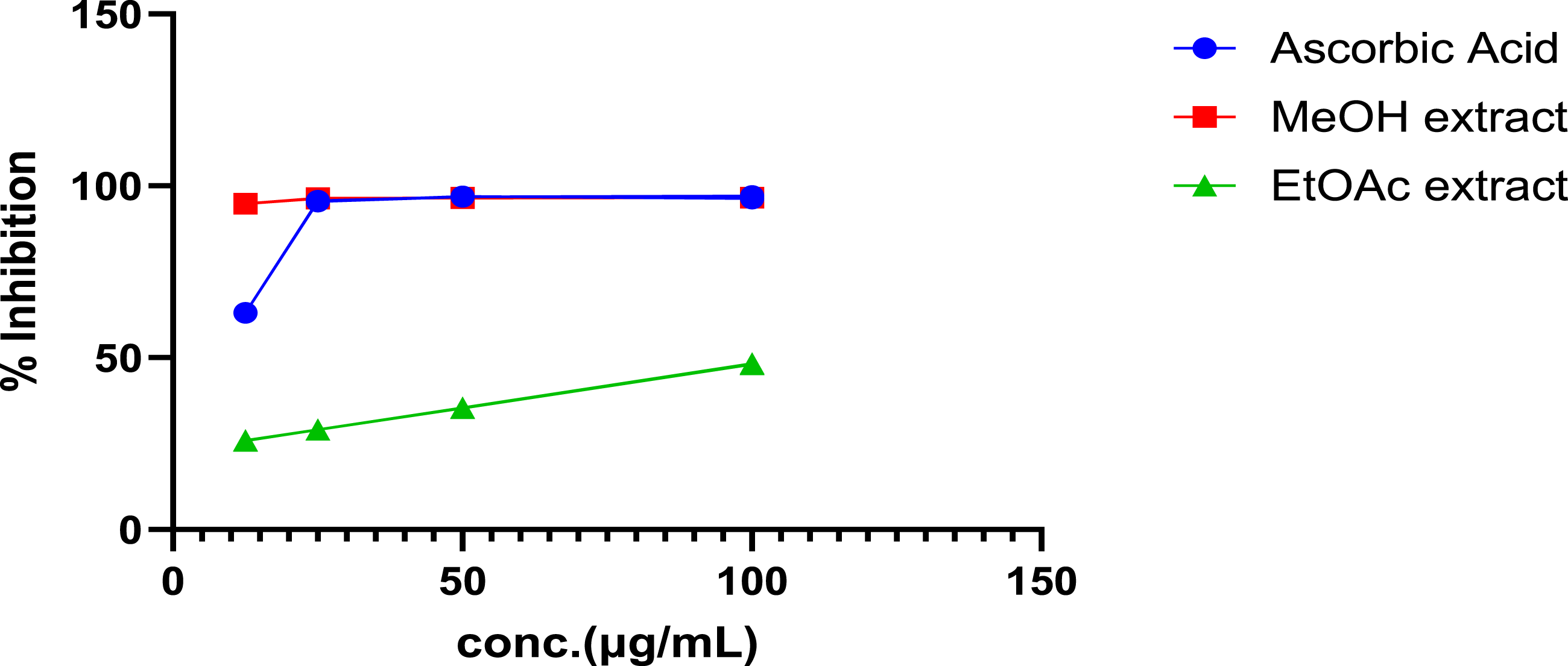
Percentage of DPPH free radical scavenging effect of A. schimperi stem bark extracts
The ethyl acetate and methanol extracts were examined for their radical scavenging activities. The DPPH radical scavenging activity of the methanolic extract was found to be 96.57±0.01%, 96.49 ± 0.03%, 96.38 ±0.04%, and 94.82±0.00% at concentrations of 100, 50, 25, and 12.5 μg/mL respectively, with IC50 value of 8.91 (Table 5). The ethyl acetate extracts radical scavenging activity was 48.14±0.10%, 35.36±0.04%, 29.06±0.00%, and 25.82±0.06% with IC50 Value of 107.28 at the same concentrations as the methanolic extract. The methanolic crude extract obtained was found to be promising compared to ascorbic acid, which was used as a positive control, with a percent inhibition of the radical of 97.10±0.01%, 96.83±0.07%, 95.53±0.01%, and 63.06±0.05% with IC50 value of 10.83 at similar concentrations of the extract.
4. Discussions
A well-known protic solvent, methanol enhances polar phytochemicals such as phenols, alkaloids, and glycosides by forming hydrogen bonds. Conversely, substances of intermediate polarity, such as terpenoids and some steroids, are typically extracted using ethyl acetate, which is moderately polar. 22 The ethyl acetate extracts showed steroids, and both extracts showed terpenoids, but the foam test revealed no saponins. Saponins are glycosides composed of sugar moieties linked to a steroidal triterpenoid aglycone. Because terpenoids and steroids might exist as free aglycones rather than glycosylated derivatives, their presence does not always imply the presence of saponins. Moreover, saponins are usually extracted using highly polar aqueous solvents. The lack of a positive foam test may be attributed to either the lack of glycosylated saponins or their low concentration in the extracts. 23
The results of this study indicate that the stem barks of A. Schimperi methanol extract contain phenols, alkaloids, terpenoids, and glycosides (Table 2). These phytochemicals are known to be therapeutically important due to their biological roles. From previously report phenols have been shown to have antioxidant activity, 24 and several in vivo and clinical investigations have reported that alkaloids have various pharmacological effects, including antibacterial, 25 antiviral, 26 anticancer, 27 and anti-inflammatory activities. 28 Terpenoids possess antioxidant and antibacterial activities, 29 and glycosides also have antioxidant activity. 30
Some studies have been conducted on some of the GC-MS-identified ethyl acetate stem bark extract compounds. Trans-2-methyl-4-n-butylthiane S,S-dioxide, which is the major component of this extract, has antiproliferative and apoptosis-inducing properties. 31 The major components of the methanolic extract of stem bark was previously reported using molecular docking analysis of the binding of myo-Inositol, 4-C-methyl- with protein (PDB ID: 1wx2), which showed an extensive hydrogen bond network with amino acid residues such as ASN 191 and ALA 202. This compound showed good binding affinities with target receptors and can be used as a lead molecule for the inhibition of tyrosinase. 32 Other components of the stem bark of A. schimperi, the methanolic extract contains the constituent 2-Azido-2,4,4,6,6-pentamethylheptane 8.96% in stem bark extracts. This bioactive compound has been identified in the ethyl acetate extracts of Ceratobasidium sp. using GC-M 33 and leaf mustard (Brassica juncea var. multiceps). 34 Chachol was previously reported that this compound has antifungal 35 and antibacterial 36 activities. N-hexadecanoic acid is a fatty acid that is found in both ethyl acetate and methanol extracts and has been said to have possible anti-inflammatory effects and cytotoxic potential.37,38
Radical scavenging activities are crucial because free radicals have harmful effects on biological systems. When the production of oxidants exceeds antioxidant defense system in certain conditions, oxidative stress occurs, which can damage important biomolecules like lipids, DNA, and proteins. 39 In present study, metanolic extract showed strong radical scavenging activity (96.57±0.01% inhibition at concentration of 100 μg/mL), while ethyl acetate extract showed moderate activity (48.14±0.10 % inhibition at concentration of 100 μg/mL). The promising antioxidant activity of the methanolic extract may be attributed to the presence of phenolic compounds, as indicated in the phytochemical results (Table 2) and the GC-MS analysis (Table 4). These phenolic compounds may be responsible for this antioxidant activity.40,41 Phenolic compounds act as antioxidants by donating hydrogen to the hydroxyls in their phenolic groups. 41 This result suggests that the stem bark of A. schemperi can be used as a natural antioxidant. However, A. schemperi is a known source of cardenolide toxins. 42 Despite the demonstration of antioxidant activity, these findings do not establish toxicological safety. Therefore, using this plant for medicinal or dietary applications is premature without a comprehensive safety evaluation.
The limitations of this study are qualitative screening which only indicates only absence and presence of major secondary metabolite without quantitative information. The GC‒MS analysis relied on library matching for compound identifications which allow only tentative assignment. Furthermore, the antioxidant activity was evaluated only in vitro chemical assay which provide preliminary insights but may not accurately reflect in vivo antioxidant effects. Consequently, the specific compounds responsible for the observed antioxidant activity were not confirmed. Further studies involving quantitative analysis, compound isolation, structural validation using complementary techniques, and biological assays are recommended to strengthen these findings.
5. Conclusions
The present study revealed the presence of alkaloids, phenols, Terpenoids, steroids, and glycoside constituents in A. schimperi stem bark extracts. The GC‒MS analysis showed that 9 different chemical compounds were present in the stem bark ethyl acetate extract of A. Schimperi, and 8 were present in the methanolic extract. Among identified compounds, trans-2-methyl-4-n-pentylthianemethyl-4-n-pentylthiane S,S-dioxide (31.06%) and trans-2-methyl-4-n-butylthiane S,S-dioxide (30.57%) are major components in ethyl acetate extract, and myo-inositol, 4-C-methyl- (49.40%) is a major component in methanolic extract. The free radical scavenging ability of the methanolic extract of A. schimperi showed excellent antioxidant activity, which correlated with the observed phenolic content and suggests significant potential for the stem bark of this plant as a natural antioxidant. However, biological activity was assessed using a single in vitro assay, and compound identification is tentative using GC‒MS. Future work should focus on bioassay-guided fractionation to isolate the active constituents, supported by comprehensive structural elucidations and expanded biological evaluations, including in vivo studies to establish physiological relevance.
Footnotes
Acknowledgments
The authors would like to express their gratitude to Arba Minch University for granting access to laboratory resources for conducting this research. Additionally, they extend their thanks to the Botany Department of Addis Ababa University for authenticating the plant.
Ethical Considerations
Ethical Approval is not applicable for this article.
Consent to Participate
There are no human subjects in this article and informed consent is not applicable.
Author Contributions
The authors confirm their contributions to the paper as follows: study conception and design: Ayele Wondatir; data collection: Ayele Wondatir and Mitku Belew; Laboratory work: AyeleWondatir and Mitku Belew; analysis and interpretation of results: Ayele Wondatir and Mitku Belew; draft manuscript preparation: Ayele Wondatir. All authors reviewed the results and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research; Arba Minch University, covered the data collection expense with grant number: GOV/AMU/TH3/CNS/Chem/06/2021.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.