Abstract
Objectives/Background
Sensitive scalp (SSc) is a skin condition characterized by various unpleasant reactions. Plant extracts, particularly Lamiophlomis rotata (Benth.) Kudo (L. rotata), are used for their natural antioxidant, antibacterial and anti-inflammatory properties in the treatment of sensitive scalp.
Methods
We extracted the active ingredient in L. rotata and used it to improve SSc. We recruited 30 volunteers with SSc using a 10Q questionnaire. The volunteers were asked to apply a 0.5% extract of L. rotata to their affected scalps on a regular basis. The volunteers’ scalp microbiome was analyzed by high-throughput sequencing, and non-invasive physiological indices were assessed.
Results
After two weeks (tw2) of treatment, epidermal water loss (TEWL), temperature and pH decreased significantly, while scalp moisture (CM) and sebum (SM) levels remained stable. Furthermore, analysis of the scalp microbiome revealed a decrease in the ratio of Malassezia restricta to Malassezia globosa from 14.57 to 3.65, and an increase in the ratio of Cutibacterium acnes to Staphylococcus capitis from 0.93 to 1.82.
Conclusion
L. rotata extracts were found to be effective in repairing the scalp barrier and modulating the microbiota, thereby alleviating symptoms such as itching, burning, tightness and redness associated with bacterial and fungal activity.
Introduction
Sensitive scalp (SSc) is characterised by an abnormal and unpleasant sensory response of the scalp to environmental stimuli, manifesting as itching, tingling, tightness, pain and burning.1–5 Associated external manifestations include scalp redness, erythema and dandruff. 6 The primary external factors contributing to SSc include heat, scalp care products, pollution, emotional stress, and environmental conditions.2,7,8 Intrinsic factors encompass impaired barrier function, imbalances in water and sebum levels, inflammation, microbial composition and microbial dysbiosis.5,9,10 Physiological indicators of the scalp may show decreased stratum corneum moisture content (CM), increased transepidermal water loss (TEWL), changes in pH, increased temperature, increased porphyrin fluorescence and increased scalp sebum content (SM).5,11–13
Dysbiosis in SSc has been characterised by increased proportions of Malassezia restricta (M. restricta) and Malassezia globosa (M. globosa) in Malassezia spp. (M. spp.), increased levels of Staphylococcus capitis (S. capitis) and decreased levels of Cutibacterium acnes (C. acnes).14–17 The association of Staphylococcus with epidermal barrier damage, Malassezia with pruritus scores and disease severity, and Cutibacterium with scalp CM, SM and porphyrin levels have been well documented.18,19 Previous studies have highlighted the potential role of Pseudomonas in scalp diseases. Choi et al 20 found that the relative abundance of Pseudomonas was significantly higher in the severe group compared to the mild group. Additionally, Park et al 21 identified Pseudomonas aeruginosa as a potential pathogenic factor, particularly in conditions such as dandruff, folliculitis, and alopecia (including both male and female pattern hair loss), suggesting its possible involvement in the onset and progression of these diseases. Improving scalp barrier function and the healthy environment of scalp microorganisms is imperative for ameliorating the symptoms of SSc.
Lamiophlomis rotata (Benth.) Kudo (L. rotata) is a plant belonging to the Lamiaceae family, and is a medicinal herb that is used in traditional Tibetan medicine. In Tibetan, it is referred to as Da Ba, Da Bu Ba, and Ga Guo La”. 22 In China, L. rotata is mainly distributed in western Sichuan, Qinghai, Gansu and Tibet. 23 A comprehensive chemical analysis of L. rotata has yielded at least 223 chemical constituents, including 79 volatile oils and 21 trace elements, with the predominant constituents comprising phenylethanol glycosides, flavonoids, cyclic enol ether terpenoids, and various volatile oils. 24 The plant has a wide range of pharmacological activities, including analgesic, haemostatic, anti-inflammatory, wound healing promoting, antimicrobial and antioxidant effects.25–28 Li et al 24 stated that L. rotata is used clinically to activate blood circulation and remove blood congestion, reduce swelling and relieve pain.
A study by Lei et al 29 demonstrated that L. rotata induced M2-type macrophage polarisation and suppressed inflammatory cytokines during the early stages of wound healing, with a mechanism of action involving the RAS/p38 MAPK/NF-κB signalling pathway. Furthermore, Geng et al 30 found that L. rotata suppressed oxidative stress and inflammation through the NRF2/COX2 signalling pathway in a mouse model. However, to date, few studies have explored the effects of L. rotata on the scalp.
In this study, the active compound total flavonoids from L. rotata were enriched and incorporated into a scalp essence formulation, which was applied to individuals with sensitive scalps for two weeks. The improvement in scalp physiological parameters in this group was then assessed. High-throughput sequencing of the bacterial 16S rRNA gene and fungal ITS region was employed to evaluate the effects of the L. rotata-containing scalp essence (LCSE) on the microbial communities of sensitive scalps. This study explored the correlation between changes in scalp physiological indicators and the diversity and distribution of the scalp microbiome. The aim is to provide scientific evidence for its application as an active ingredient in sensitive scalp care products. The experimental process is shown in Figure 1.

Graphical summary. (A), Schematic illustration of the preparation process of L. ratoto extract and the overall workflow of the volunteer test, including active ingredient extraction, safety assessment, subject screening, intervention, and Evaluation criteria; (B), Summary of the analytical results, including scalp physiological parameter assessments and microbial community profiling using ITS and 16S rRNA sequencing. Arrows indicate treatment-related changes: ↑ increase, ↓ decrease, → no significant change, and ↔ numerical variation.
Materials and Methods
Material Information
The Tewameter® TM 210, Skin-pH-Meter PH 905, Sebumeter® SM 815, and Corneometer® CM 825 were obtained from Courage + Khazaka Electronic GmbH (Germany). The thermal cycler (PCR instrument) was purchased from Eppendorf AG (22331 Hamburg, Germany). An analytical balance (CP224S) was supplied by Sartorius (Shanghai) Trading Co., Ltd An electronic balance (ML3002/02) and a water bath (SB-1200) were acquired from Beijing Zhongxing Weiye Co., Ltd Pipettes were purchased from Eppendorf (Germany).
L. rotata was procured from Anguo Traditional Chinese Medicine Market. Absolute ethanol, rutin, sodium nitrite, aluminum nitrate, and sodium hydroxide were purchased from Shanghai Aladdin Biochemical Technology Co., Ltd Disposable sterile cotton swabs, disposable polyethylene gloves, powder-free latex gloves, and dry ice were obtained from Haimen Yangzi Medical Devices Co., Ld
Extraction Conditions Optimization
Extraction Methods
Extraction was performed as described by Wu and Tang with some modifications.31,32 The material-liquid ratio of L. rotata was 1:30 (m/V). The plant material was crushed and extracted with 60%, 75%, 90% ethanol and water by Ultrasound-Assisted extraction (UAE) (and Reflux Extraction, RE) for 2.0 h. Subsequently, the mixture was cooled to 25 °C and filtered using a 0.45 µm filter plate. The extract was then concentrated 20-fold under reduced pressure and freeze-dried (Table 1).
Extracted Conditions.

Enrichment Methods
Enrichment was performed as described by Yuan with some modifications. 33 The aqueous extract was subjected to ethanol precipitation, followed by ethanol-water precipitation for purification (W-E). The aqueous extract was vacuum concentrated and stirred slowly while ethanol (70-80%) was gradually added to reach the required ethanol concentration. The mixture was sealed and refrigerated for 24 h, then filtered. Ethanol was recovered, and the precipitate was washed with ethanol to obtain the refined liquid, which was then dried to yield the extract.
Similarly, the ethanol extract was vacuum concentrated, and ethanol was recovered. Ethanol was added slowly to the mixture while stirring, followed by gradual addition of water. The solution was sealed and refrigerated for 24 h, then filtered, and the precipitate washed with water to obtain the refined liquid, which was then dried (Table 2).
Enrichment Conditions.

Purification Methods
Group G1 refers to the treatment group that received the L. rotata extract after it was purified using D101 macroporous resin (Table 3). Following the method described by Yue et al with some modifications.34,35 D101 macroporous resin was washed and soaked in 2 volumes of 95% ethanol for 24 h. It was then rinsed with 95% ethanol until the effluent was colorless. The resin was subsequently soaked in 2 volumes of 5% hydrochloric acid (w/v) for 4 h, washed to neutrality, and further treated with 2 volumes of 5% sodium hydroxide (w/v) for 4 h, again washing to neutrality. The sample concentration was adjusted to 3.0 mg/mL, and the flow rate was set at 1.0 mL/min. After dynamic adsorption equilibrium, the resin was washed with distilled water until the effluent was colorless. Total flavonoids were eluted using a 75% ethanol solution. The reaction endpoint was determined by total flavonoid content analysis.
Purification Conditions.

Total Flavonoids Content (TFC)
TFC was determined as described by Zou. 36 With some modifications, a mixture of 1 mL of extract, 4 mL of distilled water and 0.3 mL of 5% NaNO2, was prepared and allowed to react for 6 min. then 0.3 mL of 10% Al(NO3)3; solution was added and mixed stirred thoroughly. After 6 min, 2 mL of 1 mol/L NaOH solution was added and allowed to stand for another 15 min. The absorbance of the mixture was measured at 510 nm versus prepared blank solution. Rutin was used as a standard for the quantification of TFC. Results were expressed in mg of rutin equivalents/g (mg/g) of extract.
In Vitro Antioxidant Activity
DPPH Assay
The scavenging activity by the DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging method according to Ohinishi.
37
With some modifications, a 1 mL of diluted extract and 1 mL of DPPH (0.2 mM) were mixed, and after 30 min the absorbance was measured at 517 nm. The scavenging activity was measured as the decrease in absorbance of the samples versus DPPH standard solution. Results were expressed as radical scavenging activity percentage (%) of the DPPH according to the formula:
ABTS Assay
The scavenging activity by the ABTS (2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)) radical cation decolorization assay was performed according to Müller.
38
With some modifications, a 0.2 mL of diluted extract was mixed with 0.8 mL of ABTS (the stock solution was diluted with phosphate-buffered saline (PBS) to yield an absorbance of 0.70 ± 0.05 at 734 nm) working solution, and after 30 min the absorbance was measured at 734 nm. The scavenging activity was measured as the decrease in absorbance of the samples versus the control. Results were expressed as radical scavenging activity percentage (%) according to the formula:
Cyclooxygenase-2 Inhibitory Assay
The COX-2 inhibitory activity was determined following the method of Andreani.
39
With some modifications, a 10 μL of heme, 10 μL of a extract sample, 200 μL of assay buffer, and 10 μL of COX-2 enzyme were combined in a 96-well plate. After incubation at room temperature for 5 min, 10 μL of chemiluminescent substrate and 50 μL of arachidonic acid were added. The absorbance was immediately measured at 535 nm. Indomethacin (10 mM) was used as a positive control. The inhibition rate was calculated as follows:
Antimicrobial Activity
The minimal inhibitory concentration (MIC) of aqueous L. ratoto extracts was determined using the micro broth dilution method, according to Clinical and Laboratory Standards Institute (CLSI) guidelines with some modifications. 40 Bacterial suspensions of Staphylococcus aureus(S. Aureus), Pseudomonas aeruginosa (P. aeruginosa), and C. acnes were adjusted to an optical density (OD₆₀₀) of 0.5, while M. globosa was adjusted to OD₆₀₀ of 1.0, and subsequently diluted with fresh medium.
In a sterile 96-well plate, 100 µL of the diluted bacterial suspension was mixed with 100 µL of the sample at various concentrations. Control wells containing 200 µL of bacterial suspension (negative control), blank culture medium (blank control), and positive controls (Ketoconazole for M. globosa and penicillin sodium for the other bacteria) were included.
The plates were incubated under appropriate conditions: 37 °C for 12 h (S. aureus, Pseudomonas aeruginosa), 24 h (C. acnes), or 30 °C for 72 h (M. globosa). Bacterial growth was assessed by measuring the optical density at 600 nm. The inhibition rates were calculated using the following formulas: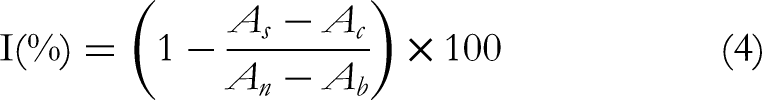
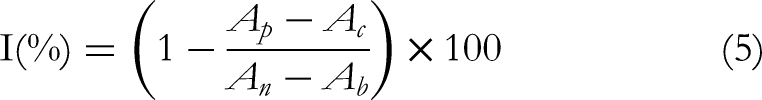
L. rotata-Containing Scalp Essence (LCSE) Preparation and Treatment
The formulation was prepared as follows: Phase A components were accurately weighed and heated to 80 °C under continuous stirring until complete dissolution was achieved. The mixture was then cooled to below 25 °C. Subsequently, PEG-40, p-hydroxyacetophenone, and L. rotata extracts were incorporated with thorough mixing to ensure homogenization. Finally, the formulation was brought to a final weight of 100 g with deionized water and filled as designated (Table 4).
Ingredient List of Scalp Spray.

Subjects applied 5 mL of LCSE to the designated dry erythematous scalp area twice daily: The first application was after shampooing in the morning, followed by allowing the scalp to dry for 1 h; The second application was 1 h before bedtime in the evening. Each application was followed by a 20-min massage, and the intervention continued for 14 days.
During this period, to minimize confounding variables from external products, participants were instructed to: (1)Use a standardized shampoo provided by the study (pH-neutral, fragrance-free, antimicrobial-free shampoo), limited to once daily (morning) during the intervention period; (2) Additionally, avoid washing the scalp for 2 days prior to the sampling procedure; (3) Conditioners or other scalp care products (eg, serums, essential oils) are prohibited; (4) Refrain from using concurrent products containing antimicrobial agents (eg, ketoconazole, salicylic acid), steroids, or alcohol-based formulations, as these ingredients may interfere with microbial analysis or skin barrier function; (5) Record hair and scalp care activities in a daily diary, documenting shampoo usage (specify the exact product if personal use is permitted), scalp massage duration, and any deviations from the protocol (eg, missed applications, additional product use); (6) Forbid special hair treatments including permanent hair coloring, perming, chemical styling (eg, thermal straightening, ionic curling), or use of styling products containing chemical fixatives (eg, formaldehyde-releasing agents, polyvinylpyrrolidone).
Efficacy Verification
Volunteer Recruitment and Study Design
The study was conducted according to the principles of the Declaration of Helsinki. All subjects provided written informed consent in person prior to the study and ethical approval was obtained from the Ethics Committee of Beijing Technology and Business University (reference BTBUECSR2024031, 21st February, 2024).
SSc is defined as the occurrence of abnormal and unpleasant sensory reactions such as pruritus, stinging, tightness, pain, and burning in response to environmental stimuli in the absence of obvious clinical signs of inflammation. 41 Scalp condition was assessed using the validated 10Q questionnaire. 42 This instrument comprises 10 questions designed to evaluate the perception of symptoms such as itching, prickling, tightness, pain, or burning. For each question, subjects indicated the presence and intensity of each symptom, with each symptom scoring 1 point if present. A score greater than 1 for any symptom was considered scalp SSc. 42 Consequently, each question had a maximum score of 5 points, yielding a total possible score of 50 for the entire questionnaire; a higher score indicates greater scalp sensitivity (Table S1).
Participants who met the following criteria were included in the study: (1) 20 to 27 years of age; (2) the tester observed with dermo scope that the subject's scalp had obvious symptoms of redness or erythema; (3) subjects reported itching or burning of the scalp; (4) provided written informed consent willing to participate in complete test process; (5) no hair perming or coloring in the last two months and promised no hair perming or coloring during the study period; (6) the last shampoo was performed 48 ± 2 h before the formal test; (7) use the assigned products regularly; (8) abstain from using any other products with similar intended purposes. Subjects were excluded if they (1) Scalp surgery within the past month; (2) Current pregnancy, breastfeeding, or plans to become pregnant during the study period; (3) A history of autoimmune diseases, cancer, renal or hepatic dysfunction, or any other significant systemic conditions that may interfere with the study outcomes.
A total of 136 volunteers participated in this study, of whom 102 were diagnosed with SSc symptoms based on the 10Q questionnaire. From the cohort of 102 subjects with sensitive scalp symptoms, 30 participants were selected based on the severity of their symptoms. These 30 participants (6 males and 24 females; mean age 23.83 ± 1.15 years, range 20-27) were chosen to assess the efficacy of LCSE. All participants did not have any underlying scalp disorders such as seborrheic dermatitis, psoriasis, or eczema, ensuring that the observed symptoms were specifically related to scalp sensitivity rather than pre-existing scalp conditions.
Scalp Microbial Sample Collection
The study was conducted during the autumn months of September and October. The sampling sites were selected from three erythematous areas of the scalp. Each sampling location was fixed and moistened with sterile 0.9% NaCl solution prior to sampling. Two sterile cotton swabs were used for each site, resulting in a total of six sterile cotton swabs for sampling. The swabs were immediately stored at −80 °C, and fungal (bacterial) DNA was extracted as soon as possible.
Scalp Physiological Parameter Measurements
The physiological measurements were performed under a controlled environment with relative humidity between 40%–60% and temperature at 25 °C. All subjects stayed in this environment for 30 min before the measurements. Additionally, participants were instructed to refrain from washing their hair for 48 ± 2 h prior to the measurements to minimize potential variations.
TEWL (Tewameter TM210), pH (skin-pH-meter PH905), SM (Sebumeter SM815) and were measured in the same area of the scalp to characterize the scalp barrier function and sebum secretion.
Statistical Analysis of Bioinformatics
The genomic DNA of the sample was extracted using TGuide S96 Magnetic Soil/Stool DNA Kit (Tiangen Biotech (Beijing) Co., Ltd) according to manufacturer's instructions. The V1-V9 hypervariable regions of the 16S rRNA gene were amplified usingprimers(27F:AGRGTTTGATYNTGGCTCAG;1492R:TASGGHTACCTTGTTASGACTT). The amplicons were quantified, after which the normalized equimolar concentrations of amplicons were pooled and sequenced on the PacBio Sequel II platform (Beijing allwegene Technologies Co., Ltd, Beijing, China).
Data analysis: The qualified sequences with more than 97% similarity thresholds were allocated to one operational taxonomic unit (OTU) using USEARCH (version 10.0). Taxonomy annotation of the OTUs/ASVs was performed based on the Naive Bayes classifier in QIIME2 using the SILVA database (release 138.1) with a confidence threshold of 70%.43,44 Alpha was performed to identify the complexity of species diversity of each sample utilizing QIIME2 software. Beta diversity calculations were analyzed by principal coordinate analysis (PCoA) to assess the diversity in samples for species complexity. One-way analysis of variance was used to compare bacterial abundance and diversity. Linear discriminant analysis (LDA) coupled with effect size (LEfSe) was applied to evaluate the differentially abundant taxa. 45
Statistical analyses were performed using GraphPad Prism 9.5 software using ordinary one-way analysis of variance (ANOVA), with p < 0.05 being considered a statistically significant difference. All tests were performed three times in parallel and the data were expressed as mean standard deviation.
Result
TFC of the Extracts
The efficiency of various extraction and enrichment strategies was evaluated based on the TFC. As summarized in Tables 5 and 6, the TFC of the extracts exhibited significant variation depending on the solvent concentration and post-extraction treatment. Among the crude extracts obtained via UAE, the E90 demonstrated the highest TFC of 117.54 ± 1.71 mg RE/g, which, after enrichment, reached 143.92 ± 1.34 mg RE/g, significantly higher than that of the E75, E60, Water (Table 5). A similar trend was observed in the RE (Table 6). Considering the TFC, extraction yield, and processing costs, the 75% ethanol extract obtained via RE was selected for subsequent purification. This extract was processed using D101 macroporous resin, resulting in the purified fraction E75-D with a TFC of 584.92 ± 3.02 mg RE/g. Based on these results, the following samples derived from the RE method were selected for further bioactivity experiments: E75, E90, E75-W, E90-W, and E75-D. The group numbers are R3, R4, F3, F4 and G1.
TFC of UAE Results.

TFC of RE Results.

Antioxidant Activity of the Extracts
All L. ratoto extracts displayed notable DPPH radical scavenging activity, which progressively increased as the concentration of the samples rose (Figure 2a). Among these, the E75-W and E90-W extracts surpassed 90% scavenging at concentrations exceeding 0.4 mg/mL. In contrast, the D101-purified E75-D extract also demonstrated over 90% scavenging, but at a slightly lower concentration of 0.3 mg/mL. Notably, the D101-purified extract outperformed the ethanol extracts in scavenging activity. The IC50 values for DPPH radical scavenging, determined for the L. ratoto samples R3, F3, R4, F4, and G1, were found to be 0.17, 0.13, 0.16, 0.11, and 0.07 mg/mL, respectively, with G1 exhibiting the highest scavenging potency, followed by F3, R3, F4, and R4.

Antioxidant activity assay result. (a), DPPH; (b), ABTS; (c), COX-2.
Similarly, the ABTS radical scavenging assay revealed a corresponding increase in scavenging activity with rising concentrations (Figure 2b). Both the E75-W and E90-W extracts showed over 90% ABTS scavenging above 0.2 mg/mL, with the E75-D extracts reaching this threshold at the same concentration. The IC50 values for ABTS scavenging, recorded for the L. ratoto samples, ranged from 0.05 to 0.11 mg/mL, with G1 again showing the most robust activity, followed by F3, R3, F4, and R4. Remarkably, the ABTS scavenging ability was found to be superior to that observed in the DPPH assay.
Furthermore, the COX-2 inhibition assays confirmed the presence of dose-dependent inhibitory effects across all extracts (Figure 2c). The F4 and G1 extracts exhibited the most potent COX-2 inhibition, achieving over 80% suppression at a concentration of 0.2 mg/mL. The IC50 values for COX-2 inhibition in the L. ratoto extracts were determined to be 0.06, 0.05, 0.04, 0.03, and 0.01 mg/mL for R3, F3, R4, F4, and G1, respectively, with G1 demonstrating the highest inhibition potency. The potency order for COX-2 inhibition was thus G1 > F3 > R3 > F4 > R4.
Antimicrobial Activities of the Extracts
The antibacterial effects of L. ratoto extracts against C. acnes were evaluated (Figure 3a). No inhibitory activity was observed for any of the extracts at concentrations below 5 mg/mL, regardless of the treatment method used.

Antimicrobial Activities assay result. (a), C. acnes; (b), S. aureus; (c), P. aeruginosa; (d), M. globosa.
Regarding S. aureus, all extracts exhibited weak antibacterial activity, with inhibition increasing as the concentration rose (Figure 3b). The E75-W, E90, and E90-W extracts showed over 50% inhibition at concentrations exceeding 5 mg/mL. In contrast, the E75-D extract achieved over 50% inhibition at 1 mg/mL. The MIC50 values for S. aureus inhibition were 5.0, 5.0, 5.0, 3.4, and 1.0 mg/mL for the R3, F3, R4, F4, and G1 samples, respectively. No bactericidal activity was detected below 5.0 mg/mL for any of the extracts.
The extracts demonstrated considerable inhibition against P. aeruginosa, with a 90% inhibition rate observed at higher concentrations (1-3 mg/mL) (Figure 3c). The bactericidal activity (MBC) for the L. ratoto extracts R3, F3, R4, F4, and G1 against P. aeruginosa was determined to be 3.0, 3.0, 5.0, 3.0, and 1.0 mg/mL, respectively.
Weak inhibitory effects were noted against Malassezia, with 50% inhibition achieved at concentrations above 3 mg/mL for some extracts (Figure 3d). The MIC50 values for Malassezia inhibition were 5.0, 3.0, 3.0, and 1.48 mg/mL for the R3, F3, R4, F4, and G1 samples. No bactericidal activity was observed at concentrations below 5.0 mg/mL for any extract.
At equivalent concentrations, the E-W extracts exhibited higher inhibition than their ethanol counterparts, while the D101 purified extracts showed the most potent activity across all treatments.
Validation of LCSE Efficacy
Scalp Questionnaire Research
A total of 136 healthy subjects between the ages of 20–27 years old participated in the study according to the scoring criteria of the 10Q questionnaire (Table 7). Of these, 102 (75%) of the volunteers scored 1 or more and were thus classified as having SSc. This result suggests a relatively high prevalence of SSc symptoms in the study's questionnaire population. The results also indicated that itching was the top symptom associated with SSc, reported by 54.41% of volunteers. Tightness and burning accounted for 34.55% and 27.94%, respectively.
10Q Questionnaire Results.

The efficacy of L. rotata was evaluated by using a 10Q questionnaire to screen 30 volunteers with SSc for the use of a product containing 0.5% L. rotata. The basic demographic characteristics of these 30 volunteers, as well as their baseline scalp severity scores, are shown in Table 8. Additionally, their scalp physiological parameters and microbiological status were assessed.
Sample Information and Physiological Characteristics of Recruited Volunteers.

Scalp Physiological Parameters Acquisition Results
The study measured physiological parameters of reddened regions of the scalp, including CM, TEWL, pH, Temperature and SM (Figure 4). A total of 30 SSc patients aged between 20–27 years old participated in the experiment. Following a fortnight of administration of the extract containing L. rotata, a reduction in TEWL values was observed in 23 of the volunteers, with 14 of these demonstrating a decrease of more than 30.99% (p < 0.05) compared to the initial baseline value (Figure 4b). Furthermore, a decline in scalp pH was observed in 28 participants, with 21 demonstrating a decrease of more than 15.19% (p < 0.001) (Figure 4c). Additionally, all 30 volunteers exhibited a reduction in scalp temperature, with 19 showing a decrease of more than 1.09% (p < 0.001) (Figure 4d). However, no significant change was observed in CM and SM.
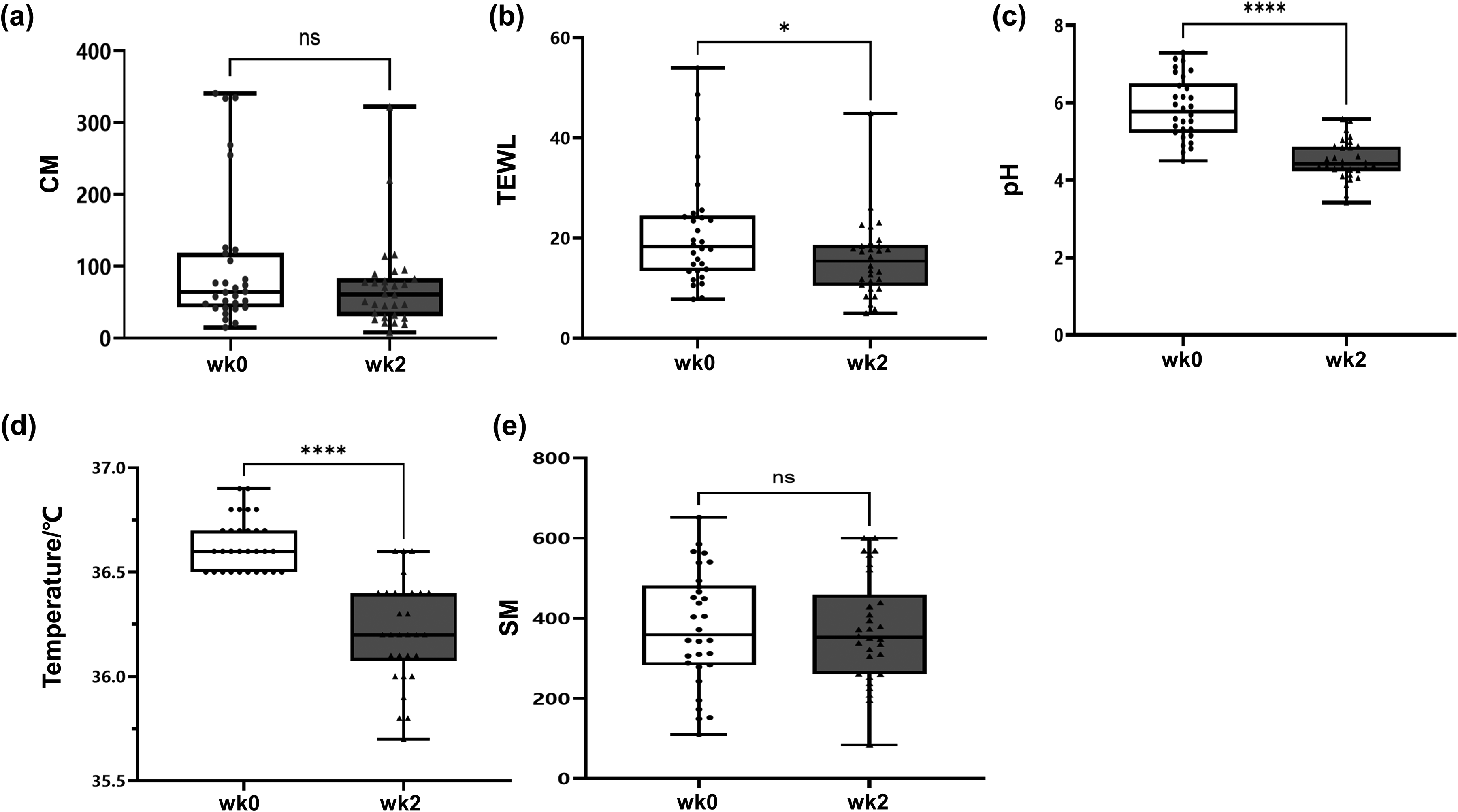
Comparison of results of scalp physiological parameters. (a), CM; (b), TEWL; (c), Ph; (d), temperature; (e), SM.
The Condition of the Scalp Microorganisms
The Alpha diversity analysis was performed using QIIME software to compare the similarity of species diversity. The Shannon index curves (Figure 5a and b) and species accumulation curves (Figure 5c and d) for fungal and bacterial communities in the Alpha diversity index of each sample are shown in Fig. A higher Shannon index indicates a higher number of species and richer species diversity, which means that the samples covered most of the microbial species present. The flattening of the species accumulation curve indicates that the amount of sequencing data is sufficient and that the number of detected species is unlikely to increase significantly with further sequencing (each curve represents one sample, marked with a different colour). The Shannon exponential curve is almost flat, indicating that the amount of sequencing data is sufficient and the number of species detected is unlikely to significantly increase with further sequencing.

Analysis of variance in alpha diversity index. Sample Shannon Index curves, (a), fungal; (b), bacterial. Species accumulation curves at genus level, (c), fungal; (d), bacterial.
Cumulative relative species abundance plots are indicative of the relationship between sample size and the number of annotated species. The individual red squares thus represent the total number of species contained in the samples collected, and the cumulative curve formed by all the red squares reflects the rate at which new species appeared under continuous sampling with a wide range of sample sizes. A sharp rise in the curve indicates that large number of new species were found in the community, whereas the flatness of the curve suggests that there was no significant increase in the number of species in the environment as the sample size increased. The results suggest that the amount of data and sample size for this sequencing is reasonable, that more data volume and sample size would produce only a small number of new species (eg, OTUs), and that the data are reliable.
Species richness maps of scalp fungi (Figure 6a) and bacteria (Figure 6b) were generated from samples of 30 volunteers following a two-week period of use of extracts containing L. rotata. The relative abundance of M. restricta in the scalp was 67.59%, while M. globosa was only 4.64%. Following treatment with L. rotata extract, the relative abundance of M. restricta decreased to 60.44%, while M. globosa increased to 16.57%. The ratio of M. restricta to M. globosa was significantly reduced from 14.57 to 3.65 (p < 0.005), and was correlated with a reduction in scalp itching and dandruff. Furthermore, an increase in Saccharomyces cerevisiae from 0.93% to 7.26%, Malassezia dermatis from less than 0.01% to 1.49%, Chaetomidium leptoderma from 1.23% to 0.01% and Aspergillus heterocaryoticus increased from 0.02% to 1.13% was observed.

Scalp sample species abundance map. (a), Fungi; (b), Bacteria. (Group A: wk0; Group B: wk2).
Bacterial analyses revealed that the relative abundance of C. acnes and S. capitis in the scalp was 39.92% and 38.23%, respectively. Following the administration of L. rotata, there was an increase in the relative abundance of C. acnes to 42.94%, while the relative abundance of S. capitis decreased to 21.03%. The total value of C. acnes and S. capitis decreased from 78.14% to 63.97%. Concurrently, the percentage of unclassified Candidatus Fonsibacter spp. diminished from 3.21% to a value below 0.1%, while the percentage of Acinetobacter harbinensis and unclassified Gemmatimonadaceae augmented from less than 0.1% to over 1%. These fluctuations in bacterial composition may signify an enhancement in scalp health subsequent to treatment with the extract.
The R-based species correlation network diagram (Figure 7) illustrates the correlation between the top 50 genera. In the figure, circles represent species, with the size of the circle denoting the average abundance of the species; lines represent the correlation between two species, with the thickness denoting the strength of the correlation; and the colours, red indicating a positive correlation and green indicating a negative correlation. The results demonstrated that Malassezia spp. was the genus with the highest mean abundance and dominated the scalp fungi community.

Network diagram of species at genus level (fungi).
Discussion
Several factors contribute to the development of SSc, with the pathogenesis thought to involve interactions among the scalp microbiota, the immune system, and dysregulated sebum production in the stratum corneum.41,46 Antifungal agents like zinc pyrithione and ketoconazole are widely used to regulate the scalp microbiota and control the growth of Malassezia spp. 47 Regular shampooing and the use of antifungal shampoos can improve scalp conditions.48–50 Plant-derived bioactive compounds with antifungal, anti-inflammatory, and antioxidant properties can improve scalp health. Wang et al 51 demonstrated that treatment with 4% glycyrrhetinic acid complex gel rapidly relieved symptoms of seborrheic scalp dermatitis and resulted in significant improvement in clinical scores and patient-reported quality of life.
Pathogens of the genus Malassezia produce reactive oxygen species during metabolism, leading to scalp erythema and hair aging.22,52–54 Multiple studies that flavonoids derived from medicinal plants can reduce inflammation and oxidative stress, which can have beneficial effects on SSc. 55 Flavonoid extracts from L. ratoto have been shown to possess potent antioxidant and anti-inflammatory properties. 22 L. rotata is enriched with a number of active constituents, which can enhance scalp health. In our study, the effects of L. ratoto extracts on antioxidant and anti-inflammatory activities (Figure 2), as well as their role in modulating the scalp microbiota, were evaluated. The results confirm that L. ratoto extracts exhibit beneficial properties.
The impact of scalp microorganisms on scalp conditions is significant, and overgrowth of Malassezia spp. is associated with symptoms of SSc. 56 In this study, scalp sampling was conducted specifically in reddened regions of the scalp, which are commonly associated with scalp sensitivity and seborrheic conditions. The application of L. rotata extract in these regions showed a decrease in the percentage of Malassezia restricta and an increase in Malassezia globosa, suggesting a modulation of the fungal balance in the affected areas (Figure 6). The L. rotata extract appears to regulate the balance between these fungi on the scalp by promoting the growth of M. globosa while inhibiting M. restricta. A study by Tao Rong et al 57 using amplicon sequencing showed an increase in the relative abundance of M. restricta and a decrease in the relative abundance of M. globosa in lesion areas compared to healthy controls.
Staphylococcus spp. and Cutibacterium spp. have been identified as key bacterial species, with the former being associated with healthy scalps and the latter with SSc. 58 In the present study, the overall ratio of C. acnes to S. capitis increased significantly from 0.93 at baseline (wk0) to 1.82. Saxena et al 15 used high throughput amplicon sequencing (bacterial 16S rRNA V3 region and fungal ITS-1 region) and macro genome sequencing. Their findings revealed that M. restricta and M. globosa were the most prevalent fungal species, with M. globosa exhibiting higher abundance in healthy scalps compared to SSc scalps. Among the bacterial species, Cutibacterium spp. and Staphylococcus spp. were the most abundant, with S. capitis showing significantly higher levels in SSc. Subsequent analyses show that L. rotata modulates the scalp microbiota, alleviating sensitivity symptoms of SSc. The increase in C. acnes may result from mutual inhibition with S. capitis, indicating improved scalp health after L. rotata extract intervention.
The analysis of the plotting of fungal networks coefficients (Figure 7), we found that M. spp. was the most prevalent species. Previous studies have indicated that M. restricta, a lipophilic fungus, produces lipase, which hydrolyzes triacylglycerols—the primary component of human sebum—resulting in the release of unsaturated fatty acids. 59 The adherence of M. spp. to the scalp compromises the barrier function of the stratum corneum by releasing these unsaturated fatty acids, leading to scalp sensitization. 60 Additionally, some tryptophan metabolites produced by M. spp., such as indole derivatives, have been implicated in dandruff formation, which is also a manifestation of SSc. 61
Changes in scalp physiology have been shown to indicate improved scalp sensitivity, as evidenced by the findings of this study. In the tests conducted, a decrease in pH and a decrease in TEWL was observed in the majority of volunteers following the application of L. rotata (Figure 4). Elevated pH and increased TEWL are recognised as indicators of impaired SSc barrier function. 12 S. epidermidis has been identified as a disadvantageous species that thrives in environments with high pH. The use of Staphylococcus spp. products has been linked to exudates that further raise pH, thereby creating a vicious cycle. The microbiota plays a pivotal role in maintaining scalp homeostasis, thereby supporting the prevailing theories on fungal and bacterial colonisation. 62
We observed an improvement in the scalp condition after using LSCE. Due to practical and objective constraints, the study was conducted with a relatively small sample size, without a placebo or vehicle-control group, and over a short intervention period. These factors may limit the strength of causal inference and the generalizability of the findings. Future studies with larger sample sizes, longer follow-up durations, and randomized controlled designs are warranted to provide more definitive evidence regarding the effects of L. rotata extract.
In addition, the study was conducted between September and October, a period intentionally selected to minimize pronounced seasonal fluctuations in scalp physiological parameters. While this approach helped reduce variability related to environmental influences, other uncontrolled factors may still have affected the outcomes and should be more comprehensively addressed in future investigations.
Conclusion
This study demonstrated the anti-inflammatory effects of L. ratoto. The L. ratoto extract purified by macroporous resin exhibited pronounced antioxidant and antimicrobial activities. In terms of scalp physiological parameters, significant reductions in TEWL, scalp temperature, and pH were observed compared with baseline values. Furthermore, 16S rRNA and ITS sequencing analyses indicated that the application of LCSE influenced the diversity and composition of the scalp microbiota, exerting a modulatory effect on the microbial community. Specifically, the relative abundances of S. capitis and M. restricta were decreased, whereas those of C. acnes and M. globosa were increased. Overall, these findings suggest that L. ratoto extract may contribute to scalp barrier repair and microbiota regulation, thereby helping to alleviate scalp sensitivity.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X261431013 - Supplemental material for Preparation Process and Regulation on Scalp Microecology Study of Total Flavonoids from Lamiophlomis rotata (Benth.) Kudo
Supplemental material, sj-docx-1-npx-10.1177_1934578X261431013 for Preparation Process and Regulation on Scalp Microecology Study of Total Flavonoids from Lamiophlomis rotata (Benth.) Kudo by Yue Wu, Guo-long Dong, Qi-lin Li, Jiang-hong Ran, Qian-yin Pan and Li Li in Natural Product Communications
Supplemental Material
sj-pdf-2-npx-10.1177_1934578X261431013 - Supplemental material for Preparation Process and Regulation on Scalp Microecology Study of Total Flavonoids from Lamiophlomis rotata (Benth.) Kudo
Supplemental material, sj-pdf-2-npx-10.1177_1934578X261431013 for Preparation Process and Regulation on Scalp Microecology Study of Total Flavonoids from Lamiophlomis rotata (Benth.) Kudo by Yue Wu, Guo-long Dong, Qi-lin Li, Jiang-hong Ran, Qian-yin Pan and Li Li in Natural Product Communications
Footnotes
Ethical Approval
The study was conducted in accordance with the Declaration of Helsinki and informed consent was obtained from all participants, and ethical approval was obtained from the Ethics Committee of Beijing Technology and Business University (reference BTBUECSR2024031, 21st February, 2024).
Consent for Participation
Written informed consent was obtained from all participants (or their legal guardians, where applicable) prior to their inclusion in the study. Participants were fully informed of the study's objectives, procedures, potential risks, and benefits, and were given the opportunity to ask questions.
Consent for Publication
Written informed consent for the publication of patient/participant data and images (eg, patient images/data reported in a case report or case series) was obtained from all participants (or their legal guardians, where applicable).
Author Contributions
All authors contributed to the study conception and design. Y.W and GL.D. performed the research. L.L designed the research study. GL.D., QL.L. and JH.R. contributed essential reagents or tools. JH.R. and QY.P. analyzed the data. Y.W. and GL.D. wrote the paper. L.L. reviewed and edited the draft. All authors have read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
All the authors declare that they have no conflict of interest relevant to this study.
Data Availability Statement
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
