Abstract
Objective
To explore the mechanism of Liu-Jun-Zi Tang (LJZT) in treating Obstructive Sleep Apnea Hypoventilation Syndrome (OSAHS) via network pharmacology, molecular docking, and experiments.
Methods
(1) Active ingredients of LJZT were screened from TCMSP (oral bioavailability OB > 30%, drug-likeness DL > 0.18). (2) Targets of ingredients and OSAHS-related targets were obtained from databases; intersection targets were identified via Venny 2.1. (3) Protein-protein interaction (PPI) network and core targets were analyzed by STRING and Cytoscape. (4) Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment were performed. (5) Molecular docking was validated via AutoDock Tools/Vina. (6) LJZT's chemical composition was analyzed by LC-MS/MS. (7) A hypoxic OSAHS cell model (HUVECs) was used; CCK-8 was screened drug concentration, and ELISA was detected inflammatory factors (IL-6, TNF-α, IL-10), HIF-1α, and pathway proteins (EGFR, SRC, STAT3).
Results
Nine core ingredients (eg, naringenin) and six core targets (eg, SRC, EGFR) were identified. KEGG enrichment highlighted pathways like HIF-1α signaling. Molecular docking showed favorable binding (binding energy < −4 kcal/mol). LC-MS/MS identified 82 components in LJZT. In hypoxic models, LJZT reduced IL-6, TNF-α, HIF-1α, inhibited EGFR/SRC/STAT3, and increased IL-10 (P < 0.05).
Conclusion
LJZT treats OSAHS by regulating the IL-6/EGFR/SRC/STAT3/HIF-1α pathway to alleviate inflammation and hypoxia-induced injury.
Keywords
Introduction
Obstructive sleep apnea hypopnea syndrome (OSAHS) is a common sleep breathing disorder with symptoms such as snoring, excessive dreaming, abnormal sleep movements, headache, and hypertension. 1 OSAHS is characterized by recurrent closure or narrowing of the upper airway, resulting in intermittent hypoxia, sleep fragmentation, and recurrent nocturnal awakenings, 2 and is characterized by hypercarbia, sleep apnea, micro-arousals, and sleep fragmentation characterization. OSAHS is a heterogeneous syndrome with multiple pathophysiological mechanisms, clinical manifestations and consequences of respiratory events. 3 According to epidemiologic statistics, the prevalence of OSAHS is 10–17% and 3–9% in adult males and females, respectively. 4 Of these, 10%-21% have moderate to severe OSAHS, and up to 49% in the elderly. 5 The number of middle-aged adults suffering from OSAHS has reached 425 million. 4 In the past, little attention has been paid to OSAHS because of the lack of awareness of its dangers. In recent years, with increasing research, various complications of OSAHS have received attention. OSAHS has been reported to increase the incidence of cardiovascular disease and many other respiratory diseases. 6 OSAHS is closely associated with hypertension, 7 diabetes mellitus, 8 cardiovascular and cerebrovascular diseases, and tumors. Therefore, OSAHS has become a hotspot of multidisciplinary medical research worldwide.
Modern medicine believes that the causes of OSAHS are complex, including obesity, abnormal upper airway anatomy, family history, and the combination of chronic underlying diseases, in addition to poor lifestyle habits; this disease will have a lot of negative impact on the patient's sleep quality, and the apnea and hypoventilation caused by the disease will lead to carbon dioxide retention in the body, which will cause other diseases and aggravate the difficulty of disease treatment. 9 Modern medicine is still unclear about the pathogenesis of this disease, and the treatment methods are limited to continuous positive airway pressure ventilation (CPAP) treatment, ENT surgery, oral orthopedic surgery, and there is no drug treatment program with precise efficacy, so the clinical efficacy is limited. In recent years, traditional Chinese medicine (TCM) has a greater advantage in the treatment of OSAHS.
Since the launch of “100 ancient classic prescriptions” in China in 2018, people's research on traditional Chinese medicine has entered a climax period, and after constantly reviewing related literature, the classic prescription of LJZT was found in the “Medical Zheng Zhuan” for the treatment of OSAHS. This formula originates from the Song Dynasty's Taiping Holy Prescriptions for Universal Relief serving as a classic representative of Traditional Chinese Medicine's “qi-tonifying and spleen-strengthening” category. It was later included and further promoted,10,11 Its composition adheres to the “sovereign, minister, assistant, and messenger” principle: Panax ginseng acts as the sovereign herb, profoundly supplementing the qi of the spleen and stomach; Atractylodes macrocephala (Bai Zhu) as the minister, fortifying the spleen and drying dampness to enhance P. ginseng's qi-tonifying effect; Poria cocos (Fu Ling) as the assistant, strengthening the spleen and draining dampness, which synergizes with A. macrocephala to address both symptoms and root causes of dampness; Citri Reticulatae Pericarpium (Chen Pi) and Pinellia ternata (Ban Xia) as assistants, regulating qi, fortifying the spleen, drying dampness, and transforming phlegm to resolve phlegm-turbidity arising from spleen-stomach qi stagnation; Glycyrrhiza uralensis (Gan Cao) as the messenger, tonifying qi, supplementing the middle burner, and harmonizing the actions of all herbs.12–14 The six herbs in this formula are exquisitely combined to achieve the core efficacy of ‘tonifying qi, strengthening the spleen, drying dampness, and transforming phlegm.’ Clinically, it is not only frequently used for spleen-stomach deficiency, poor appetite, loose stools, and cough with copious phlegm caused by qi deficiency and phlegm stagnation, but can also be modified based on pattern differentiation to treat various somatic disorders arising from qi deficiency, phlegm-damp obstruction of collaterals, and impaired qi circulation. Its efficacy has been validated through centuries of clinical practice, establishing it as one of the core formulas in traditional Chinese medicine for regulating qi deficiency with phlegm-damp patterns.15,16
There is no disease name corresponding to OSAHS in traditional Chinese medicine, and it can be attributed to snoring, snoring sleep and other categories according to the patient's clinical symptoms. 17 It is pointed out in the General Treatise on Causesand Manidestions of all Diseases that snoring is caused by the deficiency of spleen qi and lack of power to transport and transform, the water and dampness gather internally, and phlegm and turbidity grow internally, and they are deposited in the muscle fascia under the tongue through the meridian, and over time, the qi and blood are stagnant, and phlegm, turbidity and stagnant blood are agglutinated with each other, which leads to the muscles collapsing and blocking the airway, and the air is obstructed while sleeping and snoring is caused by the obstruction of breath in and out of the body, so its etiology and pathogenesis are mostly qi deficiency, phlegm and stagnant blood, and mutual conjugation. 18 LJZT has the efficacy of drying dampness, resolving phlegm, benefiting qi and strengthening spleen, which is suitable for the treatment of qi deficiency, phlegm and blood stasis and stagnant bean knot type of diseases. 19 While the application of LJZT in clinical research is more, the laboratory research is almost zero. So we studied its mechanism of action, but due to the fact that LJZT has more active ingredients, more targets of action, etc, because of its mechanism of action for the treatment of OSAHS is not clear. So in this study, we used the network pharmacology approach to predict the active ingredients, core targets, and signaling pathways of LJZT in OSAHS, and explore initially its mechanism of action.20,21 The system flowchart of this work is shown in Figure 1.

Schematic workflow of the integrated study on LJZT for OSAHS treatment (this flowchart outlines the multi-dimensional research strategy combining network pharmacology, molecular docking, and cellular experiments to elucidate the mechanism of Liu-Jun-Zi Tang (LJZT) in treating obstructive sleep apnea hypoventilation syndrome (OSAHS)).
Materials and Cell
The following materials and instruments were used in this study: Poria cocos (Schw.) Wolf (Anhui Yukang Traditional Chinese Medicine Drinking Tablets Co., Ltd, Nanchong, Sichuan, 220301); Pinelliae ternata (Thunb.) Breit.(Anhui Yukang Traditional Chinese Medicine Drinking Tablets Co., Ltd, Nanchong, Sichuan, 220401); Citri reticulatae Blanco (Anhui Yukang Traditional Chinese Medicine Drinking Tablets Co., Ltd, Nanchong, Sichuan, 220501); Panax ginseng C.A.Mey. (Anhui Youxin Pharmaceutical Co., Ltd, Shijiazhuang, Hebei, 220901); Atractylodes macrocephalae Koidz. (Anhui Youxin Pharmaceutical Co., Ltd, Fushun, Liaoning, 210401); Glycyrrhizae uralensis Fisch (Anhui Huadingtang Chinese Medicine Drinking Tablets Technology Co., Ltd, Wuwei, Gansu, 20220701); HUVEC (iCell Bioscience Inc. Shanghai, China. ICell - h110); minimum essential medium (MEM, 11095080); fetal bovine serum (FBS,10099141C); phosphate-buffered saline (PBS, 10010023); penicillin / streptomycin (P/S, 15070063) (Gibco BRL Gaithersburg, MD, USA); and cell counting kit 8 (CCK-8) (Elabscience Biotechnology Hubei, China. E-CK-A362). Interleukin (IL)-6 (F3066-A), IL-10 (F3071-A); tumor necrosis factor (TNF)-α (F40565-A); EGFR (F3646-A); SRC (F21353-A); STAT3 (F40162-A); HIF-1a (F3266-A); COX-2 (F8509-A); VEGF (F40076-A) kits (Shanghai Kexing Trading Co., China).
Methods
Screening of Active Ingredients, Target Prediction, and Identification of Drug-Disease Intersection Targets
The active ingredients of the six constituent herbs in LJZT (Screening of Active Ingredients, Target Prediction, and Identification of Drug-Disease Intersection Targets
The active ingredients of the six constituent herbs in LJZT (P. Cocos (Schw.) Wolf Sclerotium, P. ternata (Thunb.) Breit., C.reticulatae Blanco, P. ginseng C.A.Mey., A. macrocephalae Koidz., and G. uralensis Fisch) were screened from the TCMSP database based on criteria: oral bioavailability (OB) > 30%, drug-likeness (DL) > 0.18,22–24 and compliance with Lipinski's Rule of Five. Their corresponding SMILES were retrieved from PubChem and imported into Swiss Target Prediction (species: Homo sapiens) to predict targets, which were filtered by probability > 0 and cross-validated with TCMSP; targets without gene names or duplicates were excluded. For OSAHS targets, databases OMIM and GeneCards were searched using keywords (“obstructive sleep apnea-hypopnea syndrome”, “obstructive sleep apnea syndrome”, “obstructive sleep apnea”), and duplicates were removed. The intersection of LJZT active ingredient targets and OSAHS targets was identified using Venn 2.1.
Construction of Protein-Protein Interaction Networks and Visualization of Multi-Level Networks
Intersection targets were uploaded to STRING to construct a PPI network (species: Homo sapiens; minimum interaction score: high confidence [0.700]; disconnected nodes hidden). The network was imported into Cytoscape (v3.10.2) via TSV files, and topological analysis using “Network Analyzer” identified the top 6 core targets by degree. Additionally, data on LJZT, active ingredients, and intersection targets were formatted into network files and imported into Cytoscape to construct a visualized drug-active ingredient-target network, with the top 8 active ingredients (by degree) selected.
Functional Enrichment Analysis and Molecular Docking Validation
GO and KEGG enrichment analyses of intersection targets were performed using Metascape, focusing on biological processes (BP), molecular functions (MF), and cellular components (CC) for GO, and significant signaling pathways for KEGG. Results were visualized as bar/bubble plots using R (v4.2.1) with ggplot2. A drug-active ingredient-target-pathway-disease network was constructed in Cytoscape, incorporating the top 20 KEGG pathways. For molecular docking, 2D structures of key active ingredients (from PubChem) were converted to PDB via PyMOL (v2.5.2); core target structures (from RCSB PDB) were processed in PyMOL to remove water/ligands. AutoDockTools (v1.5.7) and AutoDock Vina (v1.2.3) evaluated binding affinity, with results visualized in PyMOL to highlight binding sites and hydrogen bonds.
Analysis of Chemical Constituents of LJZT
The LJZT extract was separated on an Accucore aQ C18 (100 mm × 2.1 mm, 1.7 μm) at a flow rate of 0.2 mL/min and a column temperature of 35 °C. The injection volume was 3 μL. The mobile phase consisted of acetonitrile (A) and 0.2% formic acid aqueous solution (B). The gradient conditions were as follows: 0–40 min, 2%–25% (v/v) A; 40–70 min, 25%–45% (v/v) A; 70–80 min, 45%–60% (v/v) A; 80–90 min, 60%–100% (v/v) A; and 90–100 min, 100% (v/v) A. Electrospray ionization was used in both positive and negative ion modes, and the MS-ddMS2 scanning mode was applied for analysis. The mass spectrometry conditions were as follows: sheath gas flow rate, 48 Arb; auxiliary gas flow rate, 11 Arb; capillary temperature, 256 °C; gas heater temperature, 300 °C.
Cell Experiments
Cell Culture and Passaging
HUVEC cells were cultured in DMEM medium supplemented with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin, maintained in a 37 °C incubator with 5% CO₂. Cells were passaged at a 1:3 ratio, and passages 3–8 in the logarithmic growth phase were selected for subsequent experiments.
Preparation of LJZT Extract
LJZT extract was prepared using the water extraction and ethanol precipitation method. The specific procedure was as follows: Six medicinal herbs (P. Cocos (Schw.) Wolf Sclerotium, P. ternata (Thunb.) Breit., C.reticulatae Blanco, P. ginseng C.A.Mey., A. macrocephalae Koidz., and G. uralensis Fisch)were weighed according to classical formula ratios. They were soaked in 10 times their volume of distilled water for 30 min, then subjected to two reflux extractions (1.5 h each). The two extracts were combined and concentrated under reduced pressure to a crude drug concentration of 1 g/mL. Subsequently, the paste was added anhydrous ethanol to achieve a 70% ethanol concentration in the system, refrigerated at 4°C for 12 h, removed the supernatant, recovered ethanol under reduced pressure, and concentrated until no ethanol odor remains. The LJZT dry powder extracts were obtained by freeze dry and stored seally at −20°C for future use. Prior to administration, the powder extracts were dissolved in ultrapure water and sterilized by passing through a 0.22 μm sterile filter membrane.
Pre-Preparation Before Administration
A stock solution with a concentration of 100 μg/mL was prepared by dissolving LJZT dry powder extract in MEM medium (basic cell culture medium) supplemented with 10% fetal bovine serum (FBS). After vortexing until completely dissolved, a series of working solutions were prepared sequentially at concentrations of 50, 25, 12.5, 6.25, 3.125, 1.563, and 0.783 μg/mL using serial dilution, respectively.
Cell Drug Delivery
Log-phase HUVEC cells were seeded at a density of 1 × 105cells per well in a 96-well cell culture plate. Each well was added with 100 μL of complete medium and incubated at 37°C in a 5% CO2 incubator for 24 h. Drug delivery was performed once the cells reached 70%–80% confluence. The original medium in each well was aspirated and discarded. Then, 100 μL of medium containing different concentrations of LJZT working solution (concentration gradient: 100, 50, 25, 12.5, 6.25, 3.125, 1.563, 0.783 μg/mL) were added to each well, respectively. A blank control group (containing only complete medium) was also established. Six replicate wells were set for each concentration and cultured for an additional 24 h. After drug administration, 10 μL of CCK-8 reagent was added to each well. The plates were incubated in the dark for 0.5 h, then the absorbance at 450 nm was measured using a microplate reader to calculate the cell survival rate for each concentration group.
Concentration Screening and Model Establishment
HUVEC cells in the logarithmic growth phase were seeded into 96-well plates at 1 × 105 cells/well, incubated at 37 °C with 5% CO₂ for 24 h. After discarding the supernatant, 100 μL of medium containing different LJZT concentrations (100, 50, 25, 12.5, 6.25, 3.125, 1.563, 0.783 μg/mL) was added (6 replicates per concentration) and incubated for another 24 h. Then, 10 μL of CCK-8 was added to each well, incubated for 0.5 h, and absorbance was measured at 450 nm to evaluate the effect of LJZT on cell viability.
For hypoxia model establishment, cells at 60%–70% confluence were washed twice with PBS, switched to sugar-free and serum-free medium, and exposed to 1%, 3%, or 5% O₂ in a hypoxic incubator for 4 h. After washing twice with PBS, complete medium was added, and cells were reoxygenated at 37 °C with 5% CO₂ for 2 h, repeated twice. The optimal oxygen concentration for modeling was screened by CCK-8 absorbance measurement.
Experimental Grouping, Drug Administration, and Index Detection
Using the optimal modeling conditions, cells were divided into four groups: control group (CON), model group (MOL), low-dose LJZT group (LJZT-L), and high-dose LJZT group (LJZT-H). Except for CON, all groups underwent modeling, followed by drug administration and continuous incubation for 24 h. Cell supernatants were collected to measure the following indices according to the kit instructions: ① Model evaluation and pharmacodynamic indices: IL-6, IL-10, TNF-α, NO, SOD, ROS, HIF-1α; ② Pathway-related proteins: EGFR, SRC, STAT3, COX-2, and VEGF levels were detected using ELISA kits.
Data Analysis
Data analysis was conducted utilizing SPSS 28.0. One-way analysis of variance was used to test the homogeneity of variance; one-way analysis of variance was utilized to compare the variations among groups. Statistical significance was determined by observing differences with P < 0.05.
Results
Screening and Prediction of LJZT Active Ingredients and Targets
The active ingredients of six herbs in LJZT were screened via the TCMSP database using criteria: OB > 30%, DL > 0.18, and AlogP<5. A total of 120 active ingredients were obtained: 10 from P. ginseng C.A.Mey., 5 from A. macrocephalae Koidz., 13 from P. Cocos (Schw.) Wolf Sclerotium, 4 from C. reticulatae Blanco, 6 from P. ternata (Thunb.) Breit., and 82 from G. uralensis Fisch (Supplementary Table 1). SMILES of these ingredients were retrieved from PubChem and imported into Swiss Target Prediction, integrating with TCMSP to obtain 835 drug-related targets. OSAHS targets were acquired from OMIM and Gene Cards, yielding 3570 unique targets after integration. VENNY 2.1 was used to map intersections between LJZT drug targets and OSAHS targets, identifying 317 potential overlapping targets (Figure 2A).

Network pharmacology analysis results of LJZT against OSAHS (
PPI Network, Core Targets, and Pathway Analysis
The 317 intersecting targets were imported into STRING to construct a protein-protein interaction (PPI) network with 317 nodes, 1668 edges, an average node degree of 10.5, and an average local clustering coefficient of 0.462. After importing the TSV file into Cytoscape for topological analysis, core targets with Degree>60 were identified, including AKT1, STAT3, SRC, HSP90AA1, EGFR, and IL-6 (Figure 2B). A drug-active ingredient-action target network was visualized in Cytoscape (Figure 2C). As licorice serves as a guide drug to harmonize various herbs, it was not included in the screening of active compounds. Active components were ranked by their Degree values, and the top 9 active ingredients (as shown in Table 1) were screened out. They are naringenin, Girinimbin, kaempferol, Celabenzine, 5,7-dihydroxy-2-(3-hydroxy-4-methoxyphenyl) chroman-4-one, Citromitin, 3beta-Hydroxy-24-methylene-8-lanostene-21-oic acid, Cerevisterol, and trametenolic acid, which play key roles in the treatment of OSAHS. GO enrichment analysis via R language revealed 3649 entries (P < 0.05): 3167 in biological processes (BP, eg, cellular calcium ion homeostasis, MAP kinase activity regulation), 173 in cellular components (CC, eg, membrane rafts, caveola), and 309 in molecular functions (MF, eg, protein tyrosine kinase activity, growth factor binding) (Figure 2D). KEGG pathway analysis identified 178 pathways (P < 0.05), with the top 20 including calcium signaling, EGFR tyrosine kinase inhibitor resistance, HIF-1, AGE-RAGE, and MAPK pathways (Figure. 2E).
Top 9 Active Ingredients by Degree Value.

Network Construction and Molecular Docking
A drug-active ingredient-target-pathway-disease network was built using Cytoscape, integrating the top 20 KEGG pathways (Figure 3A). Molecular docking was performed for 9 active ingredients and 6 core targets (SRC, HSP90AA1, STAT3, AKT1, EGFR, IL-6). Binding energies > −4 kcal/mol indicated good docking, with those > -5 kcal/mol considered better (Table 2). Based on the core targets and screened pathways, the core targets included in the pathways were selected for visualization. Docking results for EGFR, IL-6, SRC, and STAT3 with selected active molecules were visualized (Figure 3B-E).

(A) Drug-ingredient-target-pathway-disease network. (B-E) Molecular docking visualizations of core ingredients with key targets. (B) EGFR-naringenin binding; (C) IL-6-Celabenzine binding; (D) SRC-Girinimbin binding; (E) STAT3-kaempferol binding. Binding energy ≤ −4 kcal/mol indicates stable binding, and ≤ −5 kcal/mol indicates excellent binding.
Molecular Docking Binding Energy.

Note: A:3beta-Hydroxy-24-methylene-8-lanostene-21-oic acid、B:5,7-dihydroxy-2-(3-hydroxy-4-methoxyphenyl)chroman-4-one、C: Celabenzine、D: Cerevisterol、E: Citromitin、F: Girinimbin、G: kaempferol、H: naringenin、I: trametenolic acid
Analysis of Chemical Constituents of LJZT
A total of 82 compounds were identified from LJZT by LC-MS/MS (Supplementary Table 2), total ion chromatography for component identification (Figure 4) covering triterpenoids, flavonoids, organic acids, nucleosides, and other components. Specifically, 20 components were detected in P. Cocos (Schw.) Wolf Sclerotium, dominated by triterpenic acids (eg, 3-hydroxypachymic acid, pachymic acid A/B), accompanied by fatty acids (palmitic acid, lauric acid) and flavonoids (naringenin). P. ternata (Thunb.) Breit. contained 18 components, including nucleosides (guanosine, thymidine), volatile oils (β-elemene, citral), and amino acids (threonine). C. reticulatae Blanco yielded 9 components, characterized by flavonoids (nobiletin, tangeretin) and alkaloids (synephrine). Ten saponins (eg, ginsenosides Re, Rg1, Rb1) were identified in P. ginseng C.A.Mey.. A. macrocephalae Koidz. showed 12 components, mainly sesquiterpenoids such as atractylenolide I/II/III. Thirteen components were found in G. uralensis Fisch, including triterpenoids (ammonium glycyrrhizinate) and flavonoids (liquiritin). Mass spectrometry analysis showed that triterpenoids were predominantly detected as [M-H]⁻ in negative ion mode (eg, ginsenoside Rg1, m/z 799.48), while fatty acids exhibited [M + H]⁺ peaks in positive ion mode (eg, palmitic acid, m/z 257.25).

Total ion chromatogram for LJZT component identification (top: negative ion chromatogram, bottom: positive ion chromatogram).
Screening of LJZT Administration Concentration and Oxygen Concentration for Cell Model
As shown in Figure 5A, when the LJZT concentration was 25 μg/mL, cell viability decreased to less than 50%, indicating that this concentration exerted cytotoxic effects on cells. By contrast, cells showed high viability within the concentration range of 0.783–12.5 μg/mL. Therefore, 3.125 μg/mL (LJZT-L) and 12.5 μg/mL (LJZT-H) were selected as the subsequent experimental concentrations. Through detecting the effect of different oxygen concentrations on the viability of HUVEC cells (Figure 5B), it was found that cell viability at 3% O2 concentration was significantly higher than that at 1% and 5% (P < 0.05). Thus, 3% O2 was determined as the oxygen concentration for modeling.

Pharmacodynamic effects and mechanism of LJZT on OSAHS cell model (A: effects of different drug concentrations on cell viability; B: effects of different O2 concentrations on cell viability; C: detection indices for successful modeling (oxidative stress, hypoxia, inflammatory indices); D: inflammatory detection indices for pharmacodynamic effects; E: oxidative stress detection indices for pharmacodynamic effects; F: hypoxia detection indices for pharmacodynamic effects; G: ELISA detection of related protein expressions (EGFR, SRC, STAT3, COX-2, VEGF)). Data are presented as mean ± SD (n = 6).
Successful Verification of OSAHS Cell Model
As shown in Figure 5C, compared with the normal control group (CON), the model group (MOL) showed significantly increased levels of inflammatory factors IL-6 and TNF-α (P < 0.05), and a significant decrease in the anti-inflammatory factor IL-10 (P < 0.05). The oxidative stress indicators, including reactive oxygen species (ROS) levels and superoxide dismutase (SOD) activity, were both elevated (P < 0.05). Additionally, the content of the hypoxia marker HIF-1α was significantly increased (P < 0.05). These results indicated that the 3% O2-induced cell model successfully mimicked the inflammatory, oxidative stress, and hypoxic states associated with OSAHS.
Pharmacodynamic Effects of LJZT on OSAHS Cell Model
Compared with the MOL group, the LJZT-L group significantly decreased the level of TNF-α (P < 0.05) and increased the level of IL-10 (P < 0.05). The LJZT-H group significantly reduced the levels of NO, IL-6, and TNF-α (P < 0.05) and increased the level of IL-10 (P < 0.05), suggesting that LJZT exerts therapeutic effects by regulating the inflammatory network (Figure 5D). The LJZT-L group significantly decreased SOD activity (P < 0.05), while the LJZT-H group significantly reduced both ROS and SOD levels (P < 0.05), indicating that LJZT alleviates cellular oxidative damage (Figure 5E). Both LJZT-L and LJZT-H groups significantly reduced the content of HIF-1α (P < 0.05), suggesting that LJZT improves the cellular microenvironment by downregulating the hypoxic pathway (Figure 5F).
Prediction of Action Mechanism: Regulation of EGFR/SRC/STAT3 Signaling Pathway
As shown in Figure 5G, compared with the CON group, the protein expressions of EGFR, SRC, STAT3, COX-2, and VEGF in the MOL group were significantly increased (P < 0.05). However, both LJZT-L and LJZT-H groups significantly inhibited the expressions of the above proteins (P < 0.05), suggesting that LJZT may exert anti-inflammatory, anti-oxidative, and hypoxia-ameliorating effects by inhibiting the EGFR/SRC/STAT3 signaling pathway and downregulating the expressions of COX-2 and VEGF.
Discussion
OSAHS belongs to the category of snoring in Chinese medicine. 25 From the perspective of the correlation between TCM theory and molecular pathways, the core efficacy of LJZT, “tonifying qi and strengthening the spleen, drying dampness and transforming phlegm”, are highly aligns with the molecular mechanisms revealed in this study. Traditional Chinese medicine's “spleen deficiency” corresponds to immune-metabolic disorders in the body. While LJZT's effects of upregulating IL-10 and downregulating IL-6/TNF-α essentially represent “tonifying qi” to regulate immune homeostasis.14,26 The pathogenesis of “phlegm-dampness obstructing the airways” corresponds to hypoxic inflammatory injury of the airway epithelium in OSAHS. The inhibitory effect of LJZT on the EGFR/SRC/STAT3 pathway,27,28 and the decrease in HIF-1α content29,31 can improve theoxic microenvironment and dissipate the airway phlegm stasis, realizing the TCM therapeutic goal of “drying dampness and resolving phlegm, and dging the airway.”The pathogenesis of OSAHS is mainly based on qi deficiency, phlegm and stasis, and the treatment should be based on benefiting qi and strengthening the spleen, promoting qi, eliminating phlegm and dispersing stasis. OSAHS is usually characterized by severe systemic or local inflammatory reactions, and the increase of certain inflammatory factors may cause damage to airway reactivity and airway diameter, 30 and may lead to complications such as hemorrhage, cardiovascular and cerebral vascular accidents, upper airway infarction, pharyngeal stenosis, and palatopharyngeal closure abnormalities and articulation.31–34 At present, there is no good treatment for OSAHS, and with the vigorous development of traditional Chinese medicine in China, the classic formula is the first choice for our research direction, so we selected the more clinically used LJZT for the treatment of OSAHS. It has a clear therapeutic efficacy, but the mechanism of action and the active ingredients are not yet known. Therefore, we utilized network pharmacology to predict its mechanism of action and active ingredients.
Through the results of network pharmacology, we obtained nine active ingredients and six core targets SRC, HSP90AA1, STAT3, AKT1, EGFR, IL-6, as well as EGFR tyrosine kinase inhibitor resistance obtained by KEGG enrichment analysis, HIF-1α signaling pathway and VEGF signaling pathway, which may be the active components, core proteins, and related pathways in the treatment of OSAHS by LJZT. The regulatory mechanisms of nine active components was as follows: Naringin targets STAT3 and IL-6, exerting anti-inflammatory effects by inhibiting STAT3 phosphorylation and pro-inflammatory factor release. Kaempferol simultaneously binds to EGFR and HIF-1α, blocking EGFR/SRC pathway activation while downregulating HIF-1α expression under hypoxic conditions, thereby achieving dual anti-inflammatory and anti-hypoxic effects. Genipin significantly inhibits SRC kinase activity, blocking downstream STAT3 signaling at its source to mitigate airway endothelial cell damage. See Table 3 below for details.
Target Sites, Regulatory Pathways, and Mechanisms of Action for 9 Key Components.

For the KEGG-enriched pathways: HIF-1α pathway: the intermittent hypoxia caused by recurrent nocturnal airway obstruction in OSAHS patients induces massive activation of HIF-1α. On one hand, it upregulates VEGF expression, driving abnormal vascular proliferation and structural remodeling in airway tissues, thereby aggravating airway narrowing.38,39 On the other hand, it promotes the release of pro-inflammatory factors such as IL-6 and TNF-α, forming a vicious cycle of “hypoxia-inflammation”.42,43 VEGF pathway: Hypoxia and inflammatory stimuli induce abnormal overexpression of COX-2. 48 and its metabolites can accelerate VEGF synthesis, promoting airway angiogenesis and fibrosis, further exacerbating airway obstruction.31,36 The EGFR/SRC/STAT3 pathway: The chronic inflammatory microenvironment of OSAHS can activate EGFR in airway epithelial cells, subsequently phosphorylating downstream SRC and STAT3. Upon nuclear translocation, STAT3 initiates the transcription of inflammatory genes,43,44 while also exacerbating oxidative stress damage, making it a central signaling axis driving the inflammatory progression of OSAHS.
So,We focused on the inflammatory pathway and HIF-α pathway for validation. And, modern pharmacological studies have shown that the active constituents of flavonoids, alkaloids, and steroids have antioxidant, free radical scavenging, and cardiovascular protective effects as well as inhibit the expression of inflammatory factors (IL-6, TNF-α), and act as anti-inflammatory by affecting the signaling pathways, such as NF-KB, HIF-1α, VEGF, etc 52 Studies have confirmed that HIF-1α is an important factor for the body in hypoxia. 49 It has been confirmed that serum HIF-1α levels can be elevated in patients with severe OSAHS, and it has been shown that serum HIF-1α in patients with polysomnography monitoring can be used as a serologic marker for the diagnosis of OSAHS with high sensitivity and specificity. 50 The most prominent compensatory response of tissues to hypoxia is angiogenesis to increase blood supply to relieve hypoxia. Angiogenesis is a complex physiopathological process, which is controlled by both pro-angiogenic and inhibitory angiogenic factors. COX-2, an inducible isoform of cyclooxygenase, has been shown to have anti-tumor apoptotic and vasculoproliferative effects, in which the pro-angiogenic effect is more important, 51 and vascular endothelial growth factor (VEGF) has been shown to be one of the most potent pro-angiogenic factors. 52 Numerous studies have demonstrated that OSAHS patients have higher plasma concentrations of VEGF than normal subjects,53,54 and that expression is upregulated when induced by hypoxia. As OSAHS patients are in hypoxemia as well as obstructive apnea sensation during sleep, it leads to a disproportionate release of NO from endothelial cells, which exacerbates the symptoms of OSAHS. From this, it can be deduced that LJZT can modulate the IL-6 EGFR/SRC/STAT3/HIF-α signaling pathway to alleviate the symptoms of OSAHS (Figure 6).
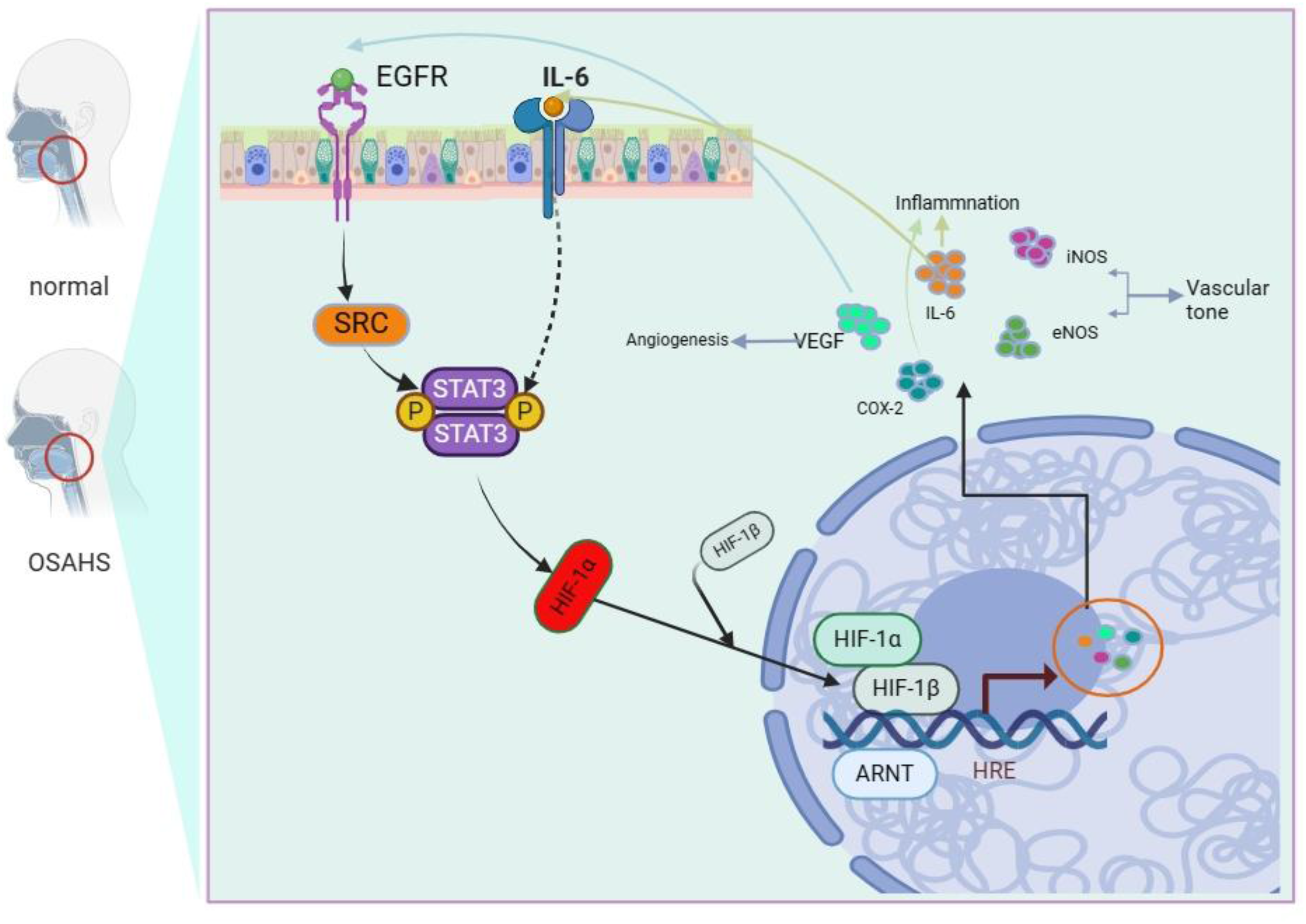
Schematic diagram of LJZT's mechanism in treating OSAHS (the pathway illustrates the key molecular events of OSAHS and LJZT's intervention sites).
Although this study is the first to combine network pharmacology with experimental validation, it still has the following limitations: ① The results of network pharmacology rely on pre-existing association data from public databases such as TCMSP and GeneCards, representing only in vitro correlation inferences, which cannot simulate complex processes such as in vivo and tissue-specific target binding. Compared to omics technologies like transcriptomics and proteomics, they lack direct characterization of authentic molecular interaction networks in vivo;②Active ingredient screening: This study used “OB > 30%, DL > 0.18” as the screening threshold. Although it can enrich highly effective ingredients, it missed effective ingredients with low bioavailability such as polysaccharides in P. Cocos and polysaccharides in P. ginseng that can be activated by intestinal microbiota metabolism, and did not fully cover the characteristic components of LJZT (such as 3-hydroxyporic acid in P. Cocos and Atractylenolide III in A. macrocephala).
At the cellular experimental level, the focus solely on the hypoxia-inflammation microenvironment of HUVECs fails to simulate the comprehensive pathophysiological characteristics of OSAHS. OSAHS is a disease with repeated collapse of the upper airway as its core, accompanied by systemic hypoxic inflammatory damage, while HUVEC can only reflect the damage and repair of vascular endothelium, and does not cover the pathological changes of key target tissues such as airway smooth muscle and airway epithelium. This study has not yet conducted clinical trials connecting basic research. The cellular pharmacological data of this study have not been translated to clinical populations, Consequently, it remains unconfirmed whether LJZT improves core clinical indicators such as the apnea-hypopnea index (AHI) and nocturnal blood oxygen saturation in patients with OSAHS. Furthermore, its synergistic or alternative value relative to first-line clinical treatments (eg, CPAP) has not been established, limiting the potential for clinical translation of these findings. No cross laboratory or cross cell batch validation has been conducted, and different sources of HUVEC cell lines (such as primary cells and immortalized cell lines) have not been compared. Therefore, the specificity of the experimental system cannot be completely ruled out, and its universality needs further confirmation. No reverse validation of target knockout/overexpression was conducted for the core pathway (EGFR/SRC/STAT3), and protein expression levels were only detected by ELISA. The specificity of pathway regulation was not confirmed at multiple levels such as gene transcription and protein phosphorylation, and the depth of mechanism validation was insufficient. The study fails to account for the population heterogeneity of OSAHS: OSAHS patients exhibit variations in age, gender, and underlying conditions.The cellular model used in this research does not simulate cellular response characteristics under different physiological/pathological states (eg, high-sugar, high-fat microenvironments). Consequently, it cannot predict therapeutic efficacy differences of LJZT in specific patient populations, limiting its applicability for personalized medicine.
In response to the above shortcomings, future research can be conducted in the following areas: combining transcriptome and metabolomics techniques to validate the targets and pathways predicted by network pharmacology in animal models, and improving the in vivo authenticity of mechanism interpretation; Relaxing the screening thresholds for active compounds, incorporating gut microbiota metabolic conversion models to explore the therapeutic value of low bioavailability components, while simultaneously establishing quantitative methods and safety evaluation systems for core compounds; Constructing a “TCM syndrome pattern - molecular pathway - clinical efficacy” correlation model to achieve deep integration of Chinese and Western medical mechanisms, providing a more comprehensive basis for the precise clinical application and new drug development of LJZT. Establishing a rat model of combined intermittent hypoxia and upper airway narrowing (OSAHS), evaluateing the therapeutic effects of LJZT on improving airway compliance, apnea-hypopnea index (AHI), and nocturnal blood oxygen saturation in the model rats, and simultaneously validating the drug's regulatory effects on the EGFR/SRC/STAT3 pathway, collagen deposition, and angiogenesis in airway tissues. A small-sample clinical pilot study will be conducted to enroll patients with qi deficiency and phlegm-dampness pattern of OSAHS. The study will compare the clinical efficacy of LJZT monotherapy versus combination therapy with CPAP to determine its target population and optimal dosing regimen. EGFR/SRC targets will be knocked out using CRISPR-Cas9 technology to provide reverse validation of pathway regulation specificity.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X261425487 - Supplemental material for Mechanisms of Liu Jun Zi Tang in the Treatment of Obstructive Sleep Apnea Hypoventilation Syndrome: Based on a Network Pharmacology Study
Supplemental material, sj-docx-1-npx-10.1177_1934578X261425487 for Mechanisms of Liu Jun Zi Tang in the Treatment of Obstructive Sleep Apnea Hypoventilation Syndrome: Based on a Network Pharmacology Study by Junxia Liu, Wei Luo, Xin Liu, Yue Lin and Junjun Han in Natural Product Communications
Footnotes
Acknowledgments
The authors would like to thank Jilin Agricultural Science and Technology College, School of Biological and Pharmaceutical Engineering for their support and assistance.
Ethical Approval
This paper does not involve human or animal experimentation and therefore does not require ethical approval.
Informed Consent Statement
Not applicable. No human subjects were used in this article.
Author Contributions
JL, WL and XL made substantial contributions to conception and design, and revised the manuscript critically for important intellectual content. JH revised the manuscript and gave final approval of the version to be published. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Key Discipline Program in Biology from Jilin Agricultural Science and Technology College (2025SW38).
Declaration of Conflicting Interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Data Availability
The study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
