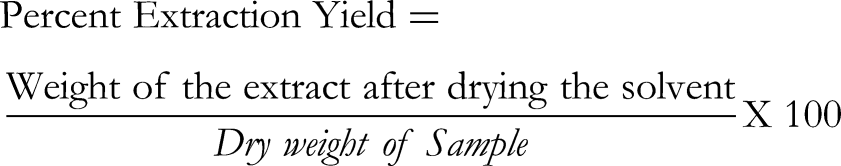Abstract
Objectives
Cisplatin, a commonly utilized chemotherapeutic drug, is linked to dose-limiting nephrotoxicity caused by oxidative stress, inflammation, and apoptosis. This research assessed the phytochemical profile and nephroprotective efficacy of Caralluma subulata (Forssk.) Decne, a traditional medicinal plant from Jazan Province, Saudi Arabia, in mitigating cisplatin-induced renal damage in rats.
Methods
The aqueous-ethanolic extract of C. subulata was examined against cisplatin induced nephrotoxicity. Rats were categorized into control group, cisplatin group only (7 mg/kg bwt), treatment group with cisplatin combined with C. subulata (100 and 200 mg/kg bwt), and C. subulata (200 mg/kg bwt) only groups.
Results
The treatment of cisplatin (7 mg/kg bwt) markedly increased serum creatinine levels (305%) and blood urea nitrogen (BUN) levels (389%) in comparison to the control group while oxidative stress markers MDA and inflammatory cytokines also significantly increased. Pretreatment with C. Subulata (CS100 and CS200 mg/kg bwt. for 15 days) significantly mitigated cisplatin-induced renal damage by reducing the creatinine by 36.68% to 64.39%, BUN by 53.38% to 74.12%, MDA by 26.08% to 77.87% as compared to cisplatin induced nephrotoxicity. Pre-treatment with C. subulata (CS100 and CS200 mg/kg bwt) also enhances the antioxidant enzymes GSH by 6.48% to138.40%, SOD by 6.81% to137.93% and CAT by 1.86% to 121.50%. Similarly, C. subulata treatment also significantly decreases the pro-inflammatory cytokines TNF-α by 6.40% to 61.23%, IL-6 by 0.62% to 48.09% and apoptotic markers like caspase-3 reduced by 1.83% to 61.02%. These biochemical improvements were supported by histopathological evidence of reduced tubular necrosis and inflammation.
Conclusion
The studies demonstrate that C. subulata alleviates cisplatin nephrotoxicity via antioxidant, anti-inflammatory and anti-apoptotic mechanisms, so validating its traditional use and highlights its potential as an adjuvant therapy to attenuate chemotherapy-induced renal damage.
Introduction
A platinum based chemotherapeutic medication called cisplatin is essential for treating several solid tumors such as bladder, ovarian, and lung cancers. 1 However, dose limiting nephrotoxicity, which affects up to 30% of patients even with preventative hydration measures, significantly limits its therapeutic usefulness. 2 There are several factors that contribute to cisplatin induced kidney damage, including oxidative stress brought on by the production of reactive oxygen species (ROS), inflammation brought on by pro-inflammatory cytokines (TNF-α and IL-6) and apoptosis caused by caspase activation and mitochondrial dysfunctions.3–5 Antioxidant and anti-inflammatory medications have shown promise in preclinical studies, but their use in the clinical practice is limited, underscoring the need for novel, naturally occurring adjuvants to mitigate kidney damage caused by cisplatin. 6
Historically medicinal plants have served as source of bioactive compound with anti-inflammatory antioxidant and anti-apoptotic properties. 7 Because of its therapeutic effectiveness against inflammatory and metabolic diseases, species of the genus Caralluma (family Apocynacease) have long been used in dry regions. 8 Caralluma species are extensively utilized in traditional medicine throughout India, Pakistan, Iran, and China for the treatment of diabetes, rheumatism, and gastrointestinal disorders. Certain tribes utilized C. fimbriata and C. tuberculata to decrease appetite during periods of hunger. 9 Current in vitro and in vivo research substantiates certain antioxidant, antibacterial, and anti-inflammatory characteristics, associating these effects with substances such as alkaloids and steroidal glycosides, flavonoids etc 10
In Jazan Saudi Arabia Caralluma subulata (locally known as “Ghabgaish”) is traditionally well known for its medicinal properties such as treating diabetes, supressing appetite and quenching thirst, addressing stomach disorder and abdominal pain, treating skin alignment, and a remedy for snake bite etc The stem extract is typically boiled in water and consumed for its purported hypoglycaemic and kidneys effects. The succulent species Caralluma subulata (CS), which is indigenous to the Saudi Arabia's Jazan Provenance, is used locally for its purported health benefits; nevertheless, little is known about its phytochemical make up and pharmacological characteristics especially renal protection. 11 Glycosides, flavonoids, and terpenoids have been identified as important component with bioactive potential in preliminary studies on related Caralluma species, suggesting that C. subulata may contain similar compounds. 12
We propose the first local GC-MS evolution of this plant extract. This analysis aims to identify the active constituents that help alleviate the renal damage by targeting oxidative stress, inflammation and apoptotic pathways. The selection of C. subulata is influenced by its local prevalence, historical usage, and the unique climatic conditions of Jazan which may impact its secondary metabolite production.13–15 This pioneering work connects traditional ethnobotanical knowledge with rigorous evidence-based pharmacology, aiming to develop a natural adjuvant that improves the safety of cisplatin cancer treatment and enhances patient quality of life.
Material and Methods
Chemicals and Reagents
Cisplatin, Ethanol (70% aqueous), Folin-Ciocalteu reagent, Gallic acid and quercetin standards, Aluminium chloride (AlCl3), Dragendorff reagent, Shinoda test reagents, Salkowski reagent, Neutral buffered formalin (10%), Paraffin wax, Haematoxylin and eosin (H&E) stains, from Sigma Aldrich St. Louis, Mo, USA. Serum creatinine and BUN kits purchased from crescent diagnostic KSA, GSH, SOD and CAT, TNF-α, IL-6, and Caspase-3 kit was purchased from Beijing Solarbio Science and Technology Co., Ltd Building 85, Beijing China.
Plant Material and Extract Preparation
Caralluma subulata (Forssk.) Decne was gathered from Jazan Province, Saudi Arabia (geographic coordinates: 17°29'N, 42°42'E) in the blossoming season (March–April 2023). A botanist from Jazan University authenticated the plant, and a voucher specimen (JZU-HB-2023-015) was deposited in the herbarium. The fresh aerial components were shade-dried, pulverized, and subjected to maceration in 70% aqueous ethanol (1:10 w/v) for 72 h. The filtrate was concentrated at reduced pressure (40 °C) utilizing a rotary evaporator (Buchi R-300) and subsequently freeze-dried to provide a crude ethanolic extract, which was used for all in vivo experiments
Determination of extraction yield was carried out by the following formula:
For the GC-MS analysis, only the volatile and semi-volatile portion of the extract was required. Since the crude ethanolic extract contains many non-volatile constituents that cannot be analysed directly by GC-MS, a small portion of the crude extract was suspended in water and partitioned sequentially with petroleum ether and ethyl acetate. These two solvents help isolate compounds of low to medium polarity that are suitable for GC–MS. The combined petroleum ether/ethyl acetate layer was dried and used as the GC–MS sample, while the crude ethanolic extract was kept unchanged for dosing in the animal study.
Phytochemical Profiling
Qualitative assays were conducted to identify alkaloids (Dragendorff's reagent), flavonoids (Shinoda test), terpenoids (Salkowski reaction), phenolics (Ferric chloride test), and saponins (foam test).16–18
Bioactive Compound Quantification
The Folin-Ciocalteu method 18 was used to determine the total phenolic content (TPC) at 765 nm λ, which is represented in milligrams of gallic acid equivalents (GAE)/gram of dry extract. The aluminium chloride assay is used to measure the total flavonoid content (TFC) at 510 nm λ, which is then reported as milligrams of quercetin equivalents (QE) per gram of dry extract. 17 Total alkaloids content (TAC) was measured at 470 nm λ which is represented in milligram of Atropine Equivalent (AE)/ gram of dry extract. 19
Free Radical Scavenging Capacity by DPPH
Total free radical scavenging capacity of C. extracts was determined using Brand Williams, 1995 DPPH procedure at 517 nm. Ascorbic acid was used as standard antioxidant and DPPH % scavenging was calculated by following formula:
Where Ab of control: Absorbance of the DPPH solution without the test sample.
Ab of sample: Absorbance of DPPH solution after reacting with the test sample.
Samples Pretreatment for GC
100 µl of extract petroleum ether/ethyl acetate extract was mixed with 900 µL methanol (MS grade) and vortexed for 1 min and filtered through 0.45 µm syringe filter and directly into injected into GC-MS for analysis.
GC-MS Analysis of Caralluma subulata
Gas chromatography was performed on Shimadzu instrument (GCMS-QP2010 ultra, Shimadzu, Japan) fitted with AOC-20S autosampler, AOC 20i autoinjector, GC-2010 GC chamber, and GCMS 50 detector was used for identification of components present in the Caralluma subulata. The injection port was set at 2700C and filtered 2 µL of diluted sample injected in split-less mode into a capillary column (TraceTM TR-5MS, 30 M length, 0.25 µ film thickness, 0.25 mm internal diameter, Thermo Scientifics, USA). Helium was used as a carrier gas with a flow rate of 1.2 mL/min. The initial oven temperature was 70 °C followed by 15 °C ramping rate to 290 °C and held for 20 min. The total run time is 34 min. The ion source and interface temperatures were 230 °C and 300 °C, respectively. The mass analyser was running on scan mode for m/z 40 to m/z 500 mass range. LabSolutions GCMS (Shimadzu, Japan) was used for data processing and analysis.
Experimental Animals and Scheme
Male Wistar rats (180-220 g) were obtained from the Jazan University Animal Facility and maintained under regular conditions (25 ± 2 °C, 12-h light/dark cycle) with unrestricted access to food and water. All operations adhered to the Institutional Animal Care guideline of Jazan University for the Care and Use of Laboratory Animals and received approval from the Institutional Animal Ethics Committee (REC-47/01/1557), Jazan University, Saudi Arabia. The ARRIVE guidelines 2.0 were followed carefully for conducting the animal research. 20
The details of grouping are given blow:
Control: Administered saline orally for a duration of 15 days. Cisplatin (CP): Administered intraperitoneally (i.p.) at a dosage of 7 mg/kg bwt on day 10.
21
CP combined with C. subulata (Cisplatin + CS100): Pre-treated with extract (100 mg/kg bwt, orally) for 15 days followed by cisplatin on day 10. CP combined with C. subulata (Cisplatin + CS200): Pre-treated with extract (200 mg/kg bwt. orally) for 15 days followed by cisplatin on day 10.12,13 Extract of C. subulata alone (CS200): Pre-treated with extract (200 mg/kg bwt, orally) for 15 days to control for potential extract-related toxicity.
Biological Sample Preparation
Blood and kidney tissues were obtained 72 h following cisplatin administration at the end of experiments. Blood and kidney tissue were collected after giving a anaesthesia (ketamin (100 mg/kg bwt) and xylazin(10 mg/kg bwt) mixture 1:10 v/v). Blood sample allowed to coagulate at room temperature after that sample was centrifuged at 3 K RPM for 10 min and serum was collected to analyse the renal markers.
For oxidative stress indicators (GSH, CAT, SOD, and MDA) testing, kidney homogenate (10%) was homogenized in phosphate-buffered solution (0.1 M and pH 7.4). Next, the homogenized material was centrifuged at 800× g for 15 min at 4 °C to separate the supernatant. It's also utilized for MDA, CAT, and SOD tests. The PMS for GSH assay was obtained by centrifuging the residual homogenate at 10,500× g for 15 min at 4 °C. 22
Biochemical and Molecular Assessments
Renal Function Indicators
Serum creatinine and blood urea nitrogen (BUN) were quantified utilizing commercial kits (Cresent diagnostic, KSA).
Indicators of Oxidative Stress
Malondialdehyde (MDA): Measured using the Thiobarbituric acid reactive substances (TBARS) assay by Ohkawa et al 1979. 23 Glutathione (GSH), superoxide dismutase (SOD), and catalase (CAT) were evaluated utilizing kits from Solarbio China.
Inflammatory and Apoptotic Biomarkers
Pro-inflammatory cytokines (TNF-α, IL-6) and Caspase-3 were quantified with ELISA kits from Solarbio China.
Histopathological Analysis
Kidney tissues were preserved in 10% formalin, embedded in paraffin, sectioned to 5 μm, and stained with haematoxylin and eosin (H&E). A blinded pathologist assessed tubular necrosis, inflammation, and glomerular injury. 24 Tubular damage was assessed, quantified, and expressed as a percentage of tubules exhibiting epithelial damage, tubular dilitation, degeneration of tubular cell, and cell death in the renal cortex: 0-normal, 1 for < 10%, 2 for 10–30% damage, 3 for >30–75%, and 4 for >75%. Tissue damage assessment was predicated on lesions in the proximal convoluted tubules (PCT), tubular dilatation, and distal convoluted tubules (DCT), as well as degeneration and necrosis of tubular cells. 25
Statistical Examination
Data are presented as mean ± standard deviation (SD). Group differences were assessed using one-way ANOVA, complemented by Tukey's post hoc test (GraphPad Prism 9.0). A p-value of less than 0.05 was deemed statistically significant (Alam et al
Results
Extraction and Yields
The ethanolic extraction of Caralluma subulata was performed to obtain a crude extract. The process resulted in a yield of 18.2% w/w, indicating that 18.2 grams of extract were obtained for every 100 grams of the starting plant material. This substantial yield suggests that ethanol is an efficient solvent for extracting a significant portion of the bioactive compounds from this plant species. Further ethanolic extract was sequentially liquid- liquid petitioned with solvents of increasing polarity like petroleum ether followed by solvent intermediate polarity like ethyl acetate for GC-MS analysis.
GC-MS Analysis
The components of Caralluma subulata were identified by using software generated match similarity factor having thresholds of 75% and above. The relative content (%) of each component was calculated by dividing the peak area of each component to the total peaks area using LabSolutions software. The peaks area was calculated without any internal standard and are uncorrected. The details of all identified compounds with their retention time, molecular weight, molecular formula, and relative content (%) and chemical class are represented in Table 1S.
The ethyl acetate/petroleum ether extract of plant Caralluma subulata appeared as a greenish-yellow semisolid material with no odor. The GC-MS analysis revealed the presence of straight chain alkanes (16.55%; 18 compounds), terpene derivatives (9.02%; 10 compounds), branched chain alkanes (3.80%; 15 compounds), naphthalene derivatives (3.20%; 6 compounds), biphenyl derivatives (0.98%; 5 compounds), phenolic-like (0.63%; 2 compound), esters (1.0%; 5 compounds), fatty alcohols (1.37%; 4 compounds), toluene derivatives (0.76%; 4 compounds), oxaspiro compound (0.33%; 1 compound), cycloalkane derivatives (0.35%; 2 compounds), monoglycerides (0.34%; 1 compound) and acetal (0.38%, 1 compound), fatty aldehyde (0.89%; 1 compound).
A total of seventy-five compounds were identified in preliminary GCMS screening accounting for 39.60% based on mass balance with respect to total peak area (Figure 1). Peaks below less than 75% similarity points and those with high noise ratios were not considered for the studies to compensate the internal standard corrections.

GC-MS total ion chromatogram of Caralluma subulata.
Tetra tetracontane at retention time of 19.87 min appeared to be most prominent peak (6.69%) and can be considered as a major component of the extract. It is a 44-carbon atom unbranched alkane serve as a human metabolite. This has been reported in various species of Ruta graveolens, Cymbopogon citrates, and Terminalia chebula.26–28 It has been reported to be associated with antioxidant and antimicrobial properties. 29 Overall, a total of eighteen alkanes and fifteen branched chain alkanes have been found with 4,5-Dipropyloctane (1.78%) as the major branched alkane. Lupeol, a pentacyclic triterpenoid was found to be the next most abundant peak (4.12%). It has been reported in seeds and latex of various plants and edible fruits and has anti-inflammatory, anticancer and various other activities associated with it (Saleem, 2009). 30 Among the triterpenes, Olean-18-ene (2.16%), norolean-12-ene (0.75%), phytane (0.45%), pristane (0.43%), norpristane (0.42%) were among the major constituents. (Ageta & Arai, 1983). 31 Triterpenes have also been reported to have beneficial biological properties. Similar type of components has been reported earlier in this plant extract Caralluma subulata with some variations. 32 Some detected peaks, such as phthalate esters and certain long-chain hydrocarbons, may originate from laboratory plasticware, solvents, or column bleed. Although blank injections were run, minor contamination cannot be completely excluded. The GC–MS analysis was performed on a separate volatile/semi-volatile fraction prepared exclusively for chemical profiling and should not be interpreted as representing the crude ethanolic extract administered in the animal study.
Phytochemical Analysis of C. subulata Extract
The aqueous-ethanolic extract of C. subulata demonstrated elevated concentrations of phenolics (28.4 ± 0.31 mg GAE/g) and flavonoids (4.75 ± 0.523 mg QE/g), alkaloid (2.81 ± 0.24 mg AE/g) contents. Preliminary screening also identified the presence of terpenoids, semi terpenoids and saponins but not quantified while free radical scavenging capacity was also analysed and found 81.99% in Ethanolic extract (Table 1).
Identification of Bioactive Compounds in C. subulata.

QE = Quercetine equivalent, GAE = Gallic acid equivalent, AE = Atropin equivalent
C. subulata Mitigates Cisplatin-Induced Renal Impairment
The treatment of cisplatin (7 mg/kg) markedly increased serum creatinine levels (3.96 ± 0.11 mg/dL) and blood urea nitrogen (BUN) levels (88.5 ± 5.1 mg/dL) in comparison to the control group (0.97 ± 0.08 mg/dL and 18.25 ± 0.41 mg/dL, respectively; p < 0.001). Pretreatment with C. subulata at doses of 100 and 200 mg/kg resulted in a dose-dependent reduction in creatinine (2.5 ± 0.40 and 1.41 ± 0.18 mg/dL) and BUN (41.62 ± 1.07 and 23.10 ± 0.40 mg/dL), with the higher dosage approaching normality (Figure 2A–B). The extract alone (200 mg/kg) produced no detrimental effects.
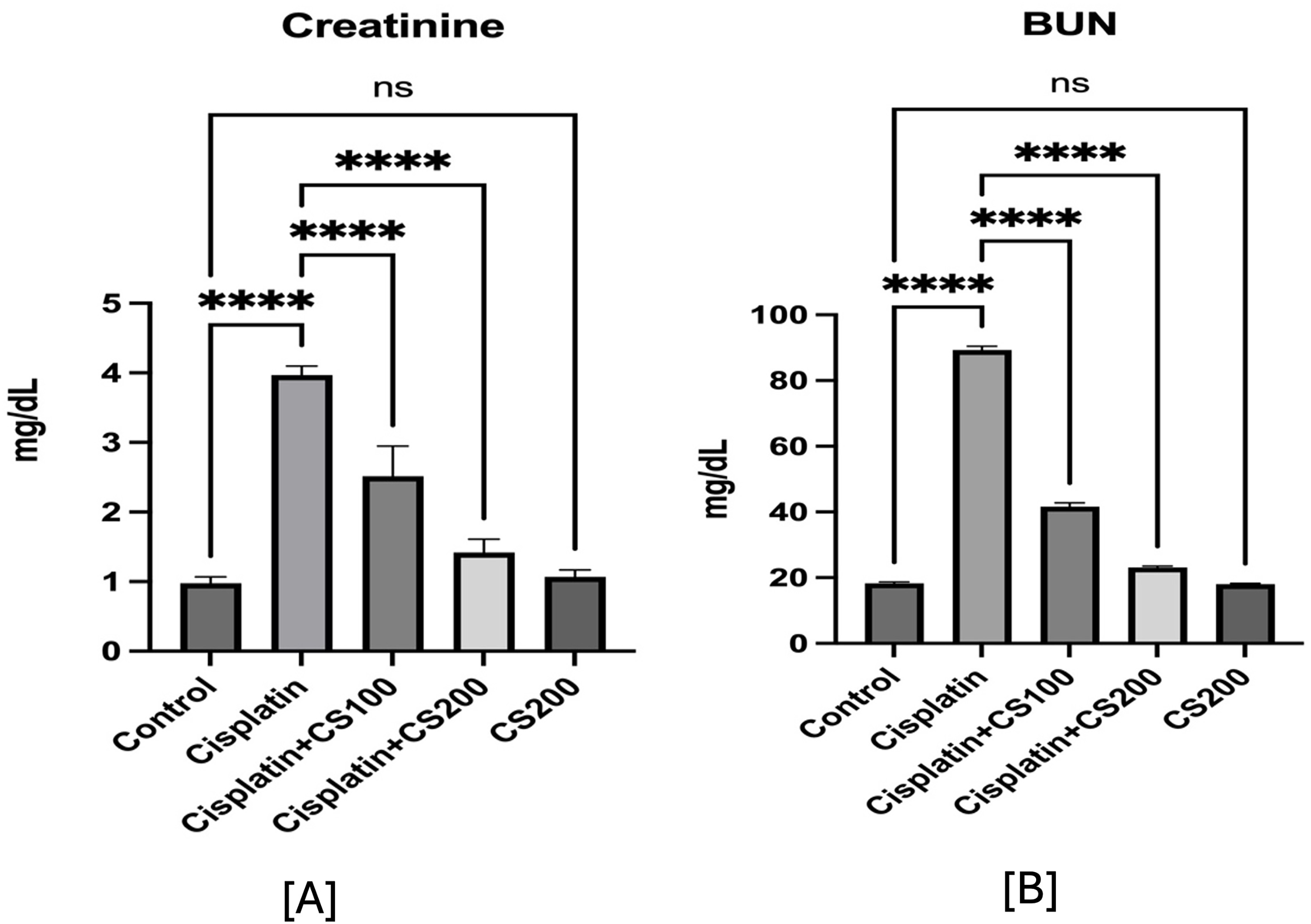
Effects of C. Subulata on Serum creatinine and BUN levels across groups.
Modulation of Oxidative Stress and Antioxidant Defences
Cisplatin induced a 19-fold increase in renal MDA (39.14 ± 0.54 nmol/mg protein) compared to controls (2.26 ± 0.3 nmol/mg. C. subulata (200 mg/kg) reduced MDA (4.91 ± 0.49 nmol/mg) to near-control levels. Depleted GSH (15.25 ± 0.48 μmol/g) and suppressed SOD (35.14 ± 0.55 ↓) and CAT (14.44 ± 0.43↓) activities in the cisplatin group were restored by C. subulata (200 mg/kg: GSH (36.38 ± 0.58) μmol/g, SOD 83.47 ± 0.40↑, CAT 32.09 ± 0.89↑; p < 0.0001 versus cisplatin) (Table 2).
Effects of C. subulata on Oxidative Stress and Antioxidant Markers.

Data represent mean ± Sd,(n = 8), *P < 0.001 CS100 versus Cisplatin(GSH), **P < 0.0001 Cisplatin versus Control, CS100 versus Cisplatin, CS200 versus Cisplatin and nsP>0.05 is not significant CS200 versus Control.
Inhibition of Inflammation and Apoptosis
Cisplatin induced a 3.02-fold elevation in TNF-α (462.75 ± 10.92 pg/mg) and a 2.93-fold increase in IL-6 (1516.5 ± 37.52 pg/mg) relative to controls (152.87 ± 11.77 and 517.12 ± 3.37 pg/mg; p < 0.0001). C. subulata (200 mg/kg) decreased TNF-α (179.35 ± 5.26 pg/mg) and IL-6 (786.87 ± 24.46 pg/mg) by 58.79% and 47.47%, respectively (p < 0.001). The activity of caspase-3 escalated 3.52-fold in the cisplatin group (894.37 ± 14.39) compared to controls (253.8 ± 7.83), but C. subulata (200 mg/kg) inhibited it by 60.96% (Figure 3A–C).

(A-C). Effects of C. subulata on Inflammatory cytokines and apoptosis markers.
Histopathological Enhancements
Cisplatin induced significant tubular necrosis, glomerular congestion, and interstitial inflammation (histopathology score: 3.8 ± 0.4). C. subulata (200 mg/kg) diminished these lesions (score: 1.2 ± 0.2; p < 0.001), maintaining renal architecture akin to controls (Figure 4A–E).

(A-E). Effects of C. subulata on kidney histopathology (H&E staining). A represents Control, B represents cisplatin, C represents treatment with CS100, D represents the treatment with CS200 and E represents the treatment with CS200 only along with kidney damage score graph.
Safety Profile
No significant changes in renal or hepatic markers were observed in the C. subulata-alone group, confirming its non-toxic nature at the tested doses.
Discussion
This study shows that Caralluma subulata (C. subulata) extract provides substantial protection against cisplatin-induced nephrotoxicity in rats, mainly due to its antioxidant, anti-inflammatory, and anti-apoptotic effects. These findings correspond with the plant's historical application and offer mechanistic insights into its reno-protective capabilities, as corroborated by both biochemical and histological evidence
Cisplatin-induced nephrotoxicity is caused by increased reactive oxygen species (ROS) formation, leading to the depletion of endogenous antioxidants (glutathione, superoxide dismutase, catalase) and the promotion of lipid peroxidation (malondialdehyde) and nitric oxide overproduction.33,34 This study demonstrated that C. subulata pretreatment mitigated cisplatin-induced oxidative damage by reinstating GSH levels (36.38 ± 0.58 μmol/g compared to 15.25 ± 0.48 μmol/g in the cisplatin group) and augmenting SOD (88% increase) and CAT (91% increase) activities. The plant contains high levels of phenolic (28.4 ± 0.31 mg GAE/g) and flavonoid 2.81 ± 0.24 mg QE/g) compounds, which are known for their ability to scavenge reactive oxygen species and bind redox active metals. 35 The ameliorative effects of C. subulata observed here—marked by reduced serum creatinine, blood urea nitrogen (BUN), and malondialdehyde (MDA), alongside restored glutathione (GSH) and antioxidant enzymes (SOD, CAT)—strongly suggest its capacity to counteract oxidative damage. These results corroborate existing literature on natural antioxidants, such as flavonoids and phenolics, which mitigate cisplatin toxicity by scavenging reactive oxygen species (ROS) and bolstering endogenous antioxidant defences.6,7
Literature review shows that cisplatin stimulates NF-κB and MAPK signalling pathways, resulting in the overproduction of pro-inflammatory cytokines such as TNF-α and IL-6, hence aggravating kidney damage.5,36 In this study, C. subulata decreased TNF-α (179.35 ± 5.26 pg/mg) and IL-6 (786.87 ± 24.46 pg/mg) by 59% and 47%, respectively, in comparison to cisplatin-treated rats. The suppression of pro-inflammatory cytokines (TNF-α, IL-6) and apoptotic markers (caspase-3) further underscores C. subulata's multi-targeted mechanism. TNF-α and IL-6 are pivotal mediators of cisplatin-induced renal inflammation, activating pathways that exacerbate tissue damage. 37 By attenuating these cytokines, C. subulata likely disrupts the inflammatory cascade, thereby preserving renal architecture, as evidenced by reduced tubular necrosis and inflammation in histopathological analyses. Similarly, the downregulation of caspase-3 aligns with studies highlighting the role of apoptosis in cisplatin nephrotoxicity and the protective effects of anti-apoptotic agents.4,38
The anti-inflammatory action may originate from its antioxidant properties, which have demonstrated the ability to suppress TLR4/NF-κB activation in like other Caralluma species which helps to reduce the production of pro-inflammatory cytokines like TNF-α and IL-6. 39 The association between decreased cytokine levels and enhanced histology (eg, reduced interstitial inflammation; Figure 3) further substantiates this process. Previous study reported that cisplatin induces apoptosis through mitochondrial malfunction, shown by caspase-3 activation.3,38 In this study C. subulata administration also inhibited caspase-3 activity by 60.96%.
The nephroprotection reported is likely due to synergistic interactions among many bioactive substances. Flavonoids augment antioxidant enzyme activity, whereas terpenoids and saponins alleviate inflammation and apoptosis. 40 The multi-target effects are superior to single-agent therapy, as they tackle the multifactorial aspects of cisplatin nephrotoxicity. 41 Other chemical induced nephrotoxicity and its protection with Beta vulgaris root extract also investigated for the nephroprotective role in rats. 42 The distinctive phytochemical composition of C. subulata from Jazan Province may indicate adaptations to its arid habitat, which frequently promotes secondary metabolite synthesis in succulent plants. This corroborates ethnobotanical accounts from Jazan regarding the utilization of Caralluma species for inflammatory conditions14,43 hence reinforcing the plant's traditional use. Furthermore, the antioxidant properties of various species of Caralluma, attributed to their flavone and polyphenolic content, underpin other therapeutic activities such as hepatoprotection and wound healing. 44
Phytochemical profiling revealed elevated phenolics, flavonoids, and terpenoids in C. subulata, compounds widely associated with antioxidant, anti-inflammatory, and anti-apoptotic activities. 45 Phenolics and flavonoids, for instance, are known to inhibit ROS generation and NF-κB signalling, while terpenoids modulate apoptotic pathways.46,47 These bioactive constituents likely synergize to confer renal protection, validating the plant's traditional use. However, while the ethanolic extract demonstrated efficacy, the specific compounds responsible for these effects remain unidentified, necessitating further isolation and characterization studies. Further research is necessary to assess the active compounds effects on human cell lines and its pharmacokinetic integrations with cisplatin. This study has primary limitation for rat model, and it cannot be directly extrapolated to human without subsequent clinical trial.
Conclusion
C. subulata alleviates cisplatin-induced nephrotoxicity by restoring redox balance through phenolic and flavonoid-mediated antioxidant mechanisms, reducing inflammatory markers IL-6 and TNF-α, and inhibiting apoptosis via the caspase 3 pathway. This study indicates that C. subulata may mitigate cisplatin-induced nephrotoxicity and enhance patient tolerance to higher dosages. The antioxidant, anti-inflammatory, and anti-apoptotic properties underscore the importance of ethnobotanical knowledge in pharmaceutical development.
Limitation and Future Direction
This research exclusively used a rat model, and findings may not directly apply to humans due to biological difference in drug metabolism, kidney physiology and disease pathways between species. No clinical trials or human data included to validate safety or efficacy, limiting its relevance for human therapeutic use. The GC-MS table lists phthalate esters and a few long-chain hydrocarbons that may come from plasticware, solvents, or column bleed, not the plant. Blank injections reduced this issue, although contamination is always possible.
Key Findings Summarized
C. subulata exhibits phytochemical richness with high phenolic/flavonoid content.
Renal protection normalises creatinine/BUN, inflammatory cytokines and oxidative stress marker dose dependently.
Biochemical data and histological correlation show reduced necrosis and inflammation.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X261417944 - Supplemental material for Phytochemical and Nephroprotective Evaluation of Caralluma Subulata From Jazan Province Against Cisplatin-Induced Nephrotoxicity Through Oxidative Stress, Inflammation, and Apoptosis In Rats
Supplemental material, sj-docx-1-npx-10.1177_1934578X261417944 for Phytochemical and Nephroprotective Evaluation of Caralluma Subulata From Jazan Province Against Cisplatin-Induced Nephrotoxicity Through Oxidative Stress, Inflammation, and Apoptosis In Rats by Saeed Alshahrani, Mohammad Firoz Alam, Marwa Qadri, Ali Hanbashi, Santhosh Joseph Menachery, Zia Ur Rehman, Ambreen Shoaib, Hina Rashid, Ali Mohammed Ali Hazazi, Ayed A. Alshamrani, Tarique Anwer and Basher M Abdalsaed in Natural Product Communications
Supplemental Material
sj-pdf-2-npx-10.1177_1934578X261417944 - Supplemental material for Phytochemical and Nephroprotective Evaluation of Caralluma Subulata From Jazan Province Against Cisplatin-Induced Nephrotoxicity Through Oxidative Stress, Inflammation, and Apoptosis In Rats
Supplemental material, sj-pdf-2-npx-10.1177_1934578X261417944 for Phytochemical and Nephroprotective Evaluation of Caralluma Subulata From Jazan Province Against Cisplatin-Induced Nephrotoxicity Through Oxidative Stress, Inflammation, and Apoptosis In Rats by Saeed Alshahrani, Mohammad Firoz Alam, Marwa Qadri, Ali Hanbashi, Santhosh Joseph Menachery, Zia Ur Rehman, Ambreen Shoaib, Hina Rashid, Ali Mohammed Ali Hazazi, Ayed A. Alshamrani, Tarique Anwer and Basher M Abdalsaed in Natural Product Communications
Footnotes
Acknowledgment
We are thankful to Deanship of Scientific Research, Jazan University for its financial supports.
Ethical Approval
This study was approved by the Local Committee for Research Ethics (Reference no
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Author Contributions
Conceptualization, S.A., and M.F.A.; methodology, S.A., ZR and M.F.A.; software, AS, SJ, TA, AS, BMA; validation MQ, AH, HR, formal analysis…MF, AH, ZUR, AS, HR, AMAH, MQ, AAA; investigation,; resources, SJ, HA, SA, AS, TA; data curation, AH, AMAH,AAA; writing—original draft preparation, MFA, TA, S.A., writing—review and editing MFA, AAA, SA,BMA visualization, supervision SA, MFA; project administration, MFA.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the funding of the Deanship of Graduate Studies and Scientific Research, Jazan University, Saudi Arabia, through project number: RG24-M022.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
This article contains the complete data set from this research investigation.
Statement of Human and Animal Rights
All the experimental procedure involving animal were conducted in accordance with the institutional Animal care guidelines of Jazan University, Saudi Arbia and approved by the Local Committee for Research Ethics Jazan University, Saudi Arabia.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.