Abstract
Introduction
According to the heart disease and stroke statistics report, there was a rise in heart disease by 14.5% from 2006 to 2016, which resulted in about 17.6 million deaths globally. 1 Yearly deaths related to cardiovascular disease (CVD) are more common than the combination of cancer and lung disease, representing 46.2% of noncommunicable disease deaths. 2 Globally, environmental exposure is one of the main reasons for CVD, which accounts for 23% of cases. 3 Nowadays, research reports indicate that pesticide exposure results in several diseases of the nervous system, kidney, and heart, as well as being a cause of cancer.4,5 Of numerous environmental pollutants, cypermethrin (Cyp) is the most commonly used insecticide. It has been used for commercial and household purposes because of its high insecticidal properties. 6 Cyp has a cynao group at the α-carbon of the alcohol group, so belongs to pyrethroids type-II. 4
Most of the Cyp is absorbed into our body by absorption through the digestive tract. Both cis and trans isomers of Cyp are broken down into cyclopropanecarboxylic acid and phenoxybenzoic acid. 7 After that, Cyp displays a classical mechanism of action that is associated with binding to the sodium channel, disrupting its inactivation. Enhanced exposure to Cyp causes the heightening of the late sodium current, which impairs the action potential waveform and can cause severe cardiac arrhythmias and oxidative stress in the heart, which further promotes myocardiac tissue injury.8,9
Cyp has high efficacy against a variety of insects, with good biodegradability and low human toxicity. However, there are some reports of mammalian toxicity because of accumulation in various tissues like kidneys, adrenal glands, and ovaries, as well as in soil.10,11 The toxic effect of Cyp and its residues has been documented on epilepsy, liver, and kidney dysfunction.12,13 Because it can cross the blood–brain barrier, Cyp causes somatic growth depression, neuritis and testicular cancer, and a decrease in motor activity, as well as increases in immunotoxic and neurotoxic effects.14–16 The toxic properties of Cyp are mostly mediated through the generation of reactive oxygen species (ROS). 17 These create an imbalance in oxidative stress by increasing lipid peroxidation (LPO), and decreasing enzymatic (superoxide dismutase [SOD], catalase) and nonenzymatic (glutathione [GSH]) functions. 18 These reactive oxygen species react with cellular biomolecules and make them unstable. Another mechanism of toxicity is apoptosis through caspase 3 and 9, bax, and apoptotic protease activating factor (Apaf). 19 Inflammatory markers (Nf-kβ, interleukin 1β [IL-1β], and interleukin 6 [IL-6]) also play a major role in Cyp toxicity. 20 Inflammation is a complex process elicited by ROS and reported in the initiation and development of coronary heart disease.21,22 NF-κB is a primary transcription factor enhanced due to ROS during myocardial infarction (MI) which induces and activates various other proinflammatory genes, including IL-1β and IL-6. 23 Recently, researchers have been working on natural products and medicinal plants for the treatment of free radical toxicity because they are less expensive and with less harmful side effects as compared to modern medicine. 24
In the present study, we chose piperine, an active component of black pepper having different pharmacological effects, such as antioxidant, bioenhancer, anti-inflammatory, antifertility, antiarthritic, hepatoprotective, and immunomodulatory activities.25–28 The antioxidative activity of piperine is connected with the 5-membered ring structure. It has been shown that piperine during metabolism undergoes demethylation of its carbon-oxygen 5-membered rings, which produce bisphenol hydroxyl metabolites. 29 Furthermore, bisphenol hydroxyl seems to have enhanced antioxidant activity. Recently, with the advancement of technology, researchers are using nanoencapsulation that displays high potential as carriers of bioactive substances because of their small size, which enables easy uptake within the cell components, making the compound stable against degradation.29,30 The utilization of nanotechnology is to overcome the problem of less bioavailability and solubility. Earlier reports are also available regarding the use of piperine in nanoformulations for treating Alzheimer's disease, with very promising results, although by choosing a dose 20-fold less than the normal. 31
Therefore, in the present study, we hypothesized that nano-piperine (NP) would prevent Cyp-induced cardiotoxicity by ameliorating oxidative stress, inflammation, and histological alteration. So, in this work, we assayed the cardiotoxicity of Cyp by measuring various biochemical parameters, cardiac marker enzyme activity, antioxidant status, inflammatory cytokines, and histopathological changes concerning heart damage.
Results
NP was successfully prepared by the hot homogenization method using lipids and surfactants.
Particle Size, Polydispersity Index, and Zeta Potential
The shape of NP, observed by TEM, was found to be round, as shown in Figure 1. Particle size distribution, zeta potential, and polydispersity index (PDI) values were analyzed by Zetasizer. The average size of NP was found to be 136.6 ± 20.4 nm (n = 3). The PDI was found to be 0.297 ± 0.062 (n = 3) (Figure 2). The zeta potential determines the charge on the surface of the NP. The zeta potential of NP was determined to be—27.9 ± 9.18 mV (n = 3).

Transmission electron microscopy micrograph of NP. Abbreviation: NP: nanopiperine.
Serum Biochemical Assay
In comparison with the control, a significant increase (P < .001) in the level of cardiac troponin I (cTnI) and the activities of creatine kinase-myoglobin binding (CK-MB), creatine kinase B (CK-B), and lactate dehydrogenase (LDH) enzymes were found in group-III treated with Cyp alone (group I), while co-treatment with varying concentration of NP (125, 250, and 500 µg/kg/day) + Cyp in group (IV-VI), respectively, showed a significant decrease in levels of LDH, cTnI, CK-MB and CK-B (P < .05; P < .01 and P < .001) as compared to the Cyp group (III). The activities of these indices in the NP group (II) were the same as in the control (Table 1).
Effects of NP against Cyp-induced cardiotoxicity.
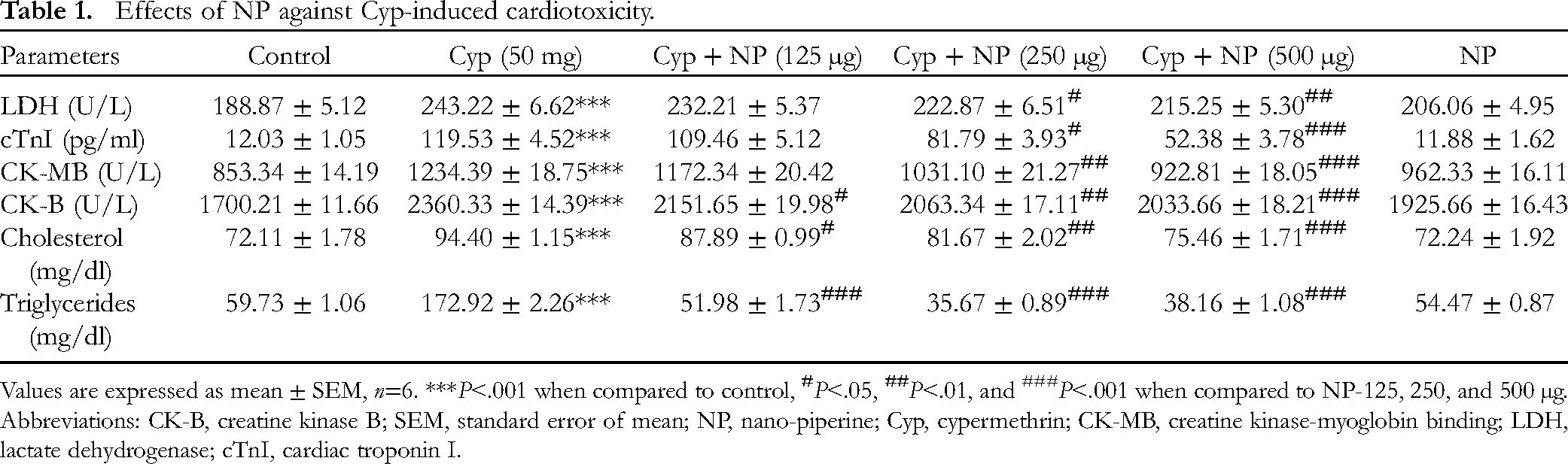
Values are expressed as mean ± SEM, n=6. ***P<.001 when compared to control,
Effect on Cholesterol and Triglycerides
A significant elevation in serum cholesterol and triglyceride levels (P < .001) was seen in the Cyp group III versus the control group (group-I). Furthermore, co-treatment with varying concentrations of NP (125, 250, and 500 µg/kg/day) + Cyp (50 mg/kg) in group (IV-VI), respectively, provided a significant decrease in values of cholesterol and triglycerides (P < .05; P < .01, and P < .001) activities in comparison with the Cyp group III (Table 1). The levels of these parameters in the NP group-II were the same as in the control.
Effects on Lipid Peroxidation and Reduced Glutathione
Exposure to Cyp produced significant adverse effects on the oxidative status of the heart, as shown by a significant increase in the malondialdehyde (MDA) level (Figure 3A) and a decrease in GSH (Fig 3B), as compared to the control. However, supplementation with NP along with Cyp significantly (P < .05; P < .01) ameliorated the LPO and GSH levels as compared to the Cyp-treated group-II rats.
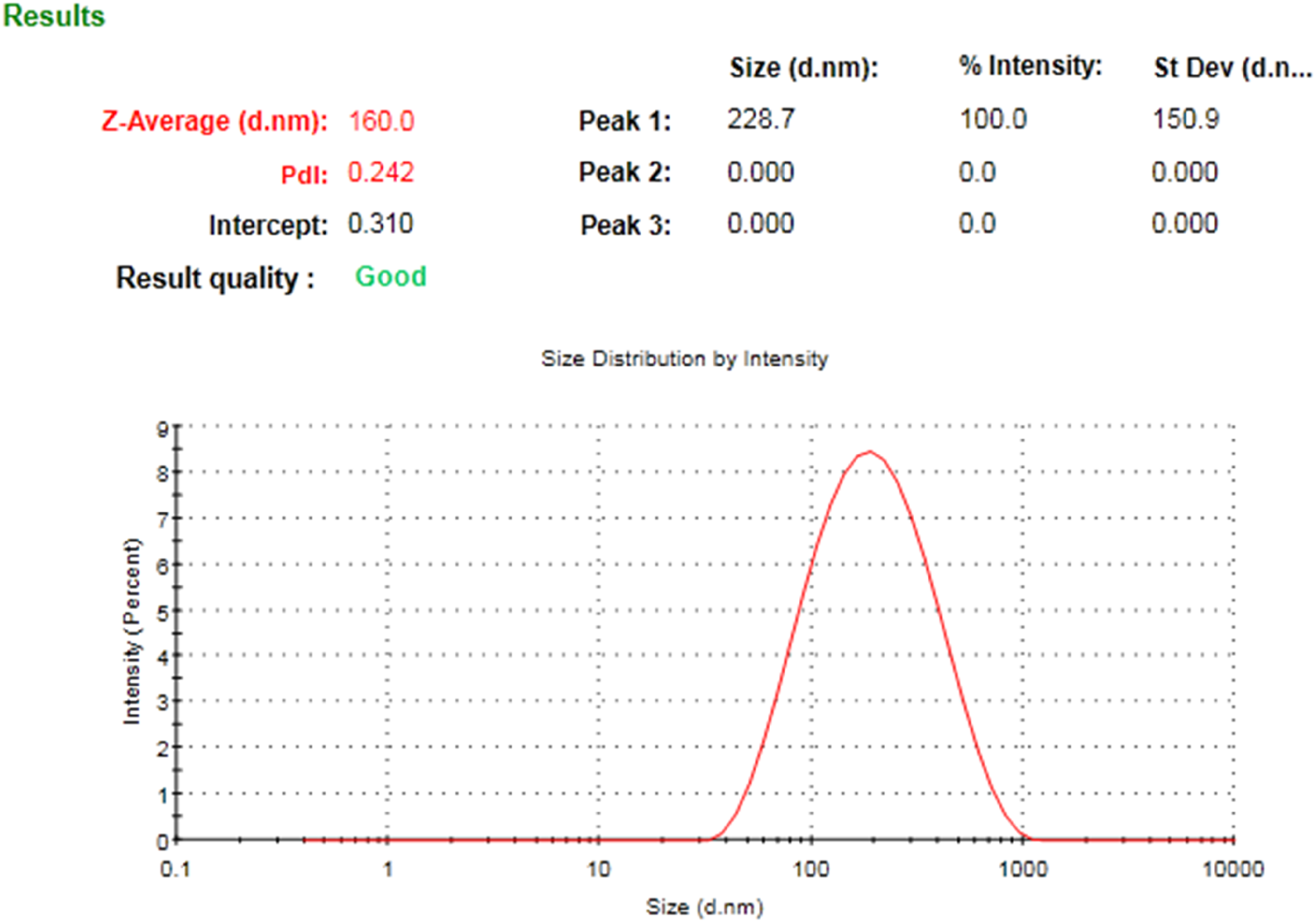
Particle size distribution of NP. Average particle size distribution of one of three (n = 3) optimized formulations of NP. The size of the particle was 160 nm with narrow size distribution (< 0.5).
Effects on SOD and Catalase
In the hearts of the Cyp-treated group-II rats (Figure 4) there was a significant decrease in SOD (Figure 4A) and catalase activity (Figure 4B) (P<.001) in comparison to the controls. Co-treatment of rats with NP and Cyp significantly ameliorated the oxidative enzyme (SOD and catalase) activities near to those of the control, in a dose-dependent manner.

Effect of NP treatment on LPO in term of MDA (A) and GSH (B) in contrast to heart toxicity produced by Cyp. Data stated as mean±SE (n=6). ***P<0.001 vs. control, #P<0.05, ##P<0.01, vs. Cyp group.
Cytokine Assay
Cyp induced a raised expression of cytokines (IL-1b, IL-6) in only the Cyp group-III (P<.001). Moreover, cotreatment with NP significantly ameliorated the expression of cytokines in the Cyp + NP (250 and 500) groups (P<.001, P<.01, P<.05) (Figures 5A and B).

Impact of NP treatment on SOD (A) and catalase (B) in contrast to Cyp induced heart toxicity. Data presented as mean±SE (n=6). ***P<0.001 vs. control, #P<0.05, ##P<0.01, ###P<0.001 vs. Cyp group.
Immunohistochemical Studies
Upon Cyp treatment, a profound expression of 4-hydroxynonenal (4-HNE) (Figure 6A-D), nuclear factor-κB (NF-kB) (Figure 6E-H), Bax (Figure 7A-D), and apoptotic protease activating factor-1 (Apaf-1) (Figure 7E-H) were reported in heart tissues (group-III). However, co-treatment with NP showed a marked amelioration of this expression with varying doses. Tissue with the highest dose of NP (500 µg) (Figure 6D, H, 7D, and H) showed a nearly similar pattern as that of the control (group-VI) (Figures 6 and 7). However, the lowest dose (125 mg) did not show any observable change when compared to the Cyp group (data not shown).
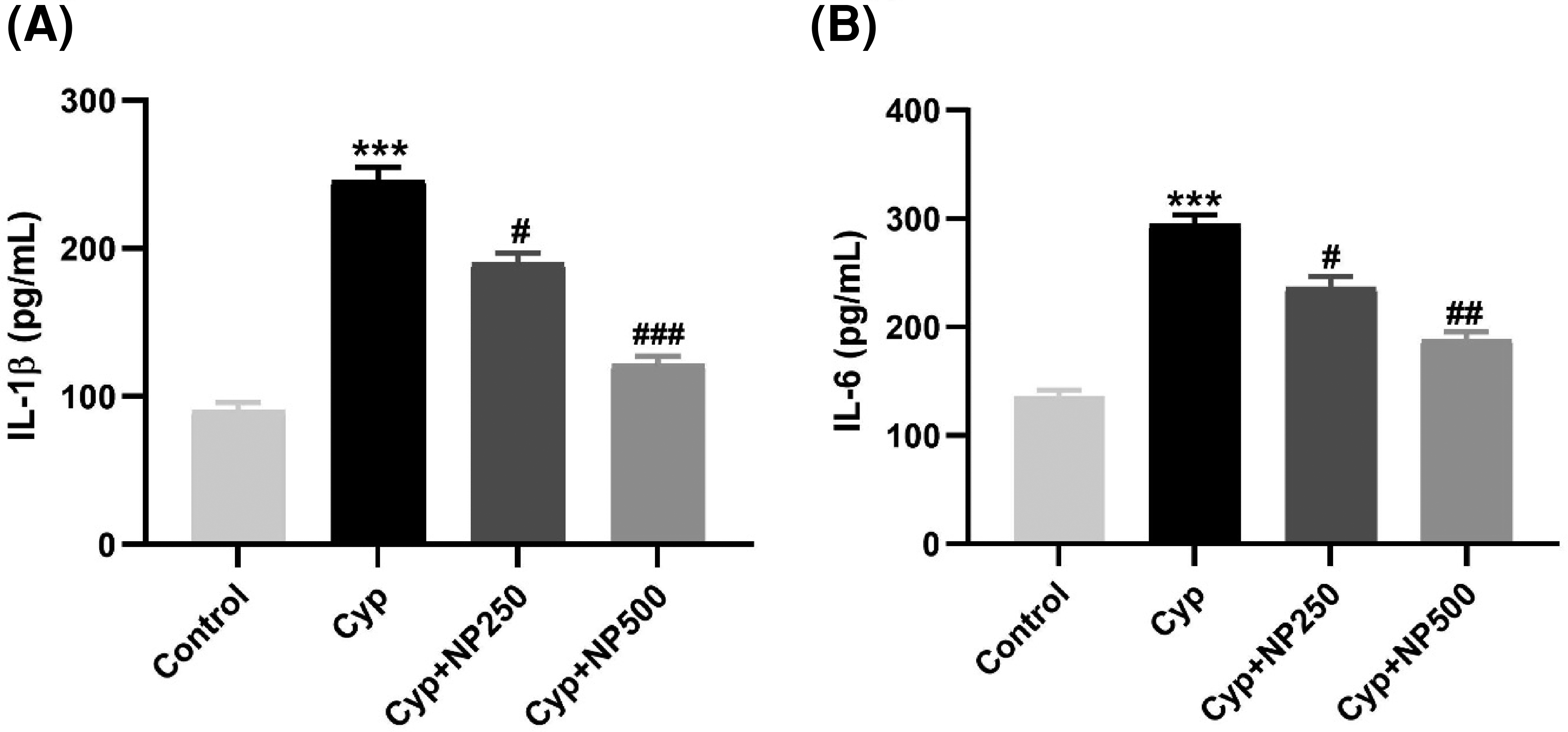
Effect of NP treatment on IL-1 β (A) and IL-6 (B) expression induced by CP in heart tissue. Data expressed as mean±SD (n=6). ***P<0.001 vs. control, #P<0.05, ##P<0.01, ###P<0.001 vs. Cyp group.

Effect of NP on 4-HNE (A-D) and NF-kB (E-H) expression induced by Cyp. Elevated expression was observed in the sections of the Cyp treated groups (B and F), as compared to the control (A and E), while the Cyp+NP treated groups (250 and 500 μg) showed moderate immunostaining of 4-HNE (C, D) and NF-kB (G, H). However, the control group showed nearly insignificant staining. Photomicrographs were taken by lightmicroscope at 40×magnification.
Histopathology
Staining the cardiac tissue sections from the various groups with hematoxylin and eosin (H&E) is shown in Figure 8. The control group showed all the normal morphological features (Figure 8A). Group (II), treated with Cyp only, showed alteration, as shown in Figure 8B, manifested by myolysis, loss of striation, and hemorrhages, indicating heart toxicity. However, cotreatment with NP showed the reversal of the histopathological alteration in a dose-dependent pattern (Figure 8C and D).

Effect of NP on Bax (A-D) and Apaf-1 (E-H) expression induced by Cyp. Elevated expression was observed in the sections of the Cyp treated groups (B and F), as compared to the control (A and E), while the Cyp+NP treated group (250 and 500 μg) showed moderate immunostaining of Bax (C, D) and Apaf-1 (G, H). However, the control group showed insignificant staining. Photomicrographs were taken by light microscope at 40 × magnification.

NP treatment protects against histopathological changes induced by Cyp. Control group showed all the normal morphological features (A), while Cyp treatment represents myolysis, loss of striation, and hemorrhages indicating heart toxicity, as compared to the control group (B). However, cotreatment with NP showed the reversal of histopathological alteration in a dose-dependent pattern (C, D). Abbreviations: NP, nano-piperine; Cyp, cypermethrin.
Discussion
This study demonstrated that Cyp induced severe cardiotoxicity at a dose of 50 mg and its crucial mechanism of cardiotoxicity. However, NP ameliorated the toxic effects due to its potent antioxidant and anti-inflammatory properties.
Many factors are responsible for achieving the therapeutic goal. The antioxidant and anti-inflammatory properties and the nanoparticle size are considered to be the most important among them. The last of these is thought to be a significant factor in achieving transport through biological barriers. The higher permeation is achieved by smaller-sized particles. 32 The smaller value of PDI indicates a narrow size distribution. The surface charge on the nanoparticles predicts the stability of colloidal dispersion. The zeta potential value is considered to be an assessment of surface charge. The electrostatic stabilization of the formulations is achieved with a zeta potential value of >20. 33 Thus, the prepared NP is considered to be a physically stable nano-system.
Heart-related diseases represent an extensive general health problem with an increased frequency worldwide. Many studies have found a link between CVDs and environmental contaminants.4,34 Extensive exposure to pesticides is among the reasons related to cardiac complications, including MI, electrocardiogram perturbation, and histological changes. 35 In the present study, we have investigated the possible mechanism of action of Cyp in the induction of cardiac toxicity in rats and the cardioprotective effects of NP.
In the present study, an increase in the levels of cTnI, CK-MB, CK-B, and LDH were primarily markers of cardiac injury; all of these were elevated in the Cyp treatment group as compared to the control group of rats. Thus, it may be stated that the increase in levels of all cardiac-specific serum markers was due to increased cellular leakage into the circulation, most probably due to the loss of membrane architecture and an increase in the number of necrotic cells in the tissues, as previously reported.36–38
Moreover, this study showed that treatment with Cyp for 15 days was enough to induce hyperlipidemia, such as hypercholesterolemia and raised levels of triglycerides. The literature reports that hyperlipidemia is associated with increased cardiac oxidative stress, but the precise molecular mechanisms by which this induces hyperlipidemia in the heart are not entirely clear. Also, a number of preclinical studies have demonstrated that classical antioxidants administered alone or in combination with other drugs or in multivitamin preparations could improve serum lipid profiles and myocardial oxidative stress in hypercholesterolemia.39,40 NP treatment inhibited these specific markers in the co-treated groups mainly by regulating membrane integrity by their antioxidant potential, which was proved by an earlier report. 41 However, the NP-alone treated group exhibited no observable changes in comparison to the control group.
CVD is also associated with a heightened lipid profile in serum. 42 Hypertriglyceridemia and hypercholesterolemia play important roles in the development and pathogenesis of MI. 43 So, from our study, we can summarize that Cyp treatment significantly elevates the levels of cholesterol and triglycerides. However, NP reverses these lipid levels and this finding is in accord with a previous report. 44 NP alone did not produce any significant changes when compared to the control.
In our study, enhanced levels of MDA were directly indicated in cardiac tissues after Cyp treatment, with membrane damage and cellular leakage, 45 along with changes in physiochemical assets, membrane fluidity manipulating signal transduction, and ion exchange properties, finally leading to membrane remodeling.46,47 Redox status in the cells is preserved by the high intracellular content of the thiol-containing reducing agent glutathione. 48 Depletion of GSH content is well documented in conditions where the disease progresses through the generation of ROS, followed by oxidative stress, which stresses the functional and structural integrity of cells and subsequently causes their death. 49 In this research, short levels of GSH content were measured after Cyp treatment may have 3 possible reasons: (i) GSH is a key substrate of Cyp uptake, (ii) having thiol binding property, and (iii) elevated oxidized glutathione (GSSG) by Cyp-mediated oxidation. 50
However, dose-dependent treatment with piperine efficiently abridged LPO in terms of MDA and raised GSH levels. The antioxidant activity of piperine potentially decelerates lipid peroxidation generation, thus preventing harm to the cells. 51 Since piperine has membrane permeability improvement properties, it may increase the influx of GSH into cells, thus improving the intracellular antioxidant status. 52
Cyp-mediated cardiac toxicity is also manifested by an increase in the levels of interleukin cytokine (IL-1b, IL-6).53,54 Moreover, oxidative imbalance elicits inflammation and is more provoked to cardiac pathogenesis. Macrophages are activated by inflammatory cytokines causing the release of reactive radicals leading to oxidative stress. Our study showed that Cyp treatment leads to a large increase in interleukins (IL-1b, IL-6) as compared with the control. However, NP significantly attenuated the increased cytokines in the treated group, whereas NP alone did not show any significant effect compared to the control. Our findings are consistent with earlier reports of NP having anti-inflammatory activity. 55
Histopathological studies showed distorted cardiac muscle fibers in the Cyp-treated rats with deeply stained nuclei (pyknotic) and highly thickened and elongated arterial walls, which revealed hemorrhage due to hemolyzed RBCs, loss of striation, myocardial tissue separation, and myolysis. All these changes are in accord with earlier studies. 56 However, NP treatment effectively ameliorated all the histological changes near the values of the control, as reported earlier. 57
An alternative mechanism of Cyp-induced cardiac grievances is also accompanied by inflammation. 58 Elevation in the levels of tissue pro-inflammatory markers (NF-kB), oxidative stress marker (4-HNE), and apoptosis markers (Bax and Apaf) are all related to oxidative imbalance and expression in stress conditions. In the present study, the increase in the expression of Cyp toxicity is clearly seen by immunohistochemistry. However, upon concurrent treatment with NP in a dose-dependent manner, it reversed the expression of 4-HNE, NF-kB, Bax, and Apaf. Our results are similar to those in an earlier report. 59
Conclusions
The present study explored the ameliorative effect of NP against Cyp-induced cardiac toxicity mediated through oxidative stress, inflammation, and apoptosis. Furthermore, the nano-formulation of piperine offers cardiac protection via antioxidant, anti-inflammatory, and anti-apoptotic activities. Based on the present evidence, piperine nano-formulation can be used as a potential therapeutic agent to improve health and prevent Cyp-induced cardiac toxicity.
Materials and Methods
Chemicals
In this study, commercially available analytical chemicals of the highest grade were procured from Sigma Chemicals, USA. All kits were purchased from Randox (UK) and antibodies from Abcam (UK).
Animals
Male Wistar rats (200-250 g) were acquired from the medical research center of Jazan University. Ethical approval was granted by the Institutional Research Review and Ethical Committee (IRREC), College of Pharmacy, Jazan University. All experiments were conducted in accordance with the standard Guidelines of the Ethical Committee (Letter No.: 6010/2903/1442). Animals were kept and fed in standard laboratory conditions during the study. Animals were acclimatized a week before the dosing and the whole study was conducted according to the institutional ethical committee.
Nano Formulation of Piperine (NP)
Nano formulations containing piperine (NP) were prepared by the hot homogenization method. These lipid nanoparticles are composed of solid lipid (stearic acid), liquid lipid (sesame oil), and surfactants (tween-80 and sodium dodecyl sulfate). A clean beaker containing an accurately weighed amount of stearic acid (SA) (500 mg) was taken and kept on a hot plate for melting. A measured volume of sesame oil (SO) (250 µL) was transferred to the beaker and mixed (lipid phase). An accurately weighed amount of piperine (100 mg) was mixed with the lipid phase. Another beaker containing purified water (25 mL) was mixed with tween-80 (T80) (50 µL) and sodium dodecyl sulfate (SDS) (25 mg) and heated to 70° C (aqueous phase). The aqueous phase was transferred and mixed with the lipid phase using a homogenizer (HG-15D, WiseTis, Germany) for 20 min at a speed of 6000 rpm. 60
Particle Size, PDI and zeta Potential
The size of the nanoparticles and PDI were measured by photon correlation spectroscopy using a Malvern Zetasizer (Nano ZS, UK). The surface charges of nanoparticales, called zeta potential, were measured using a Malvern Zetasizer.
Dose Selection
Cyp, freshly prepared by dissolving in normal saline, was given orally for 15 days, at a dose of 50 mg/kg/day.61,62 NP were administered orally for 10 days in 3 doses (125, 250, and 500 µg/kg/day) and started 5 days after Cyp. NP was administered for 10 days and a time gap of 1 hour was maintained between doses.
Experimental Design
Rats were randomly divided with six animals into six groups. Group I (control), was given normal saline for 15 days, while Groups II-V received Cyp (50 mg/kg, orally) daily for 15 days. Groups (III-V) further received a dose of NP of 125, 250, and 500 µg/kg/day, respectively, for 10 days, which started after 5 days of Cyp (50 mg/kg). In Group VI, a dose of 500 µg/kg NP was given orally daily for 10 days to check for any significant effect of NP alone as compared to the control group. Signs of toxicity were observed throughout the treatment regime. At the end of the experiment, blood samples were drawn from the retro-orbital venous plexus for separation of serum for biochemical estimations. After that, animals were euthanized by cervical dislocation and the heart was isolated for oxidative stress parameters. Ten % formalin was used to keep the heart tissue for histopathology studies. The perfused heart was also kept for immunohistochemistry studies.
Sample Preparation
Blood samples were obtained by ocular punctures from each group. The serum was extracted by centrifuging the blood samples at 3000 rpm for 10 min. After that, the animals were anesthetized using ketamine hydrochloride (30 mg/kg b/w, intramuscular injection), and euthanized by cervical dislocation. Heart tissue was quickly dissected out and 5% to 10% homogenates and postmitochondrial supernatants (PMSs) were prepared in 10 mM phosphate buffer (pH 7.4). The biochemical assay was made in homogenate/PMS by using a double-beam ultraviolet (UV)-spectrophotometer (UV-1800, Shimadzu).
Serum Biochemical Assay
The activity of LDH (1001260, Spinreact), CK-MB (1001055, Spinreact), CK-B, Troponin I (ab246529, Abcam), cholesterol (1001091, Spinreact) and triglycerides (TR201, Randox) were determined by using a spectrophotometer and microplate reader according to the protocol provided by the manufacturer.
Estimation of LPO and GSH and Antioxidant Enzyme Activity
Estimations of LPO in terms of MDA and GSH were carried out in heart tissue by preparing a 10% homogenate.63,64 An antioxidant enzyme (SOD and catalase) activity status was measured in the PMS of heart tissue. 65 The scheme of Lowry et al 66 was used to measure protein concentration.
Inflammatory Cytokines (IL-1β and IL- 6)
Inflammatory interleukin (IL-1β, IL-6) activities were estimated by the protocol provided with the ELISA Kit from FD (USA). A plate reader was used to take the absorbance at 405 nm (ELx 800TM BioTek, USA).
Immunohistochemistry of 4-HNE, NF-kB, Bax and Apaf-1
Immunohistochemistry was performed to detect the expression of 4-HNE, NF-kB, Bax and Apaf-1 proteins. Heart sections were obtained after cryo-sectioning (10-20 µm thick). Slices were washed with PBS and incubated for 1 h each with blocking solution to reduce nonspecific antibody binding. The section was treated with primary and secondary antibodies. Slides were then exposed to DAB to develop color. The sections were covered with a coverslip after dehydration and mounted in mounting media. Photomicrographs were taken by light microscope at 40× magnification. 67
Histopathological Study
The heart tissue obtained from each group was kept in 10% formalin solution. 68 Heart tissues were fixed and embedded in paraffin and tissue sections were cut for examination with H&E stain. Slides were examined under a microscope.
Statistics
Results are represented as mean ± standard error. Significance was measured by one-way analysis of variance followed by t-tests.
Footnotes
Acknowledgements
The authors wish to thank all Pharm D students (Muteb Othman Hakami, Yahya Hussin Baity, Abdulrahma Yahya Mahdli, Wael Mohammed Kabani, Amjab Omar Ogdi) for their help during the entire work for preparing the reagents and performing various assays.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors extend their appreciation to the Deputyship for Research & Innovation, Ministry of Education in Saudi Arabia for funding this research work through project number: ISP22-3.
Ethical Approval
This study was approved by the Institutional Research Review and Ethical Committee (IRREC; Letter No.: 6010/2903/1442) College of Pharmacy, Jazan University.
Statement of Human and Animal Rights
This study does not contain any studies on human subjects. The Standard Guidelines of the Ethical Committee for animals were followed.
