Abstract
Objectives
Cisplatin (CP) is a most potent chemotherapeutic agent; however, CP causes nephrotoxicity that limits its therapeutic utility. Triticum aestivum called wheatgrass (WG) is a strong antioxidant and potent detoxifying herb that has not only been studied for anticancer activity but also for efficacy and safety when combined with anticancer drugs. This study investigated the efficacy of WG against CP-induced nephrotoxicity.
Methods
Male rats were allocated into five sets of six rats in each: (1) normal control (NC), (2) CP-treated (7.5 mg/kg) as positive control (PC), (3) WG-200 mg/kg alone, and (4) CP with WG-100, and (5) CP with WG-200 mg/kg groups. Hydro-alcoholic extract of WG was administered orally to the animals for two weeks, and CP was administered intraperitoneally to the respective groups on the 10th day to induce nephrotoxicity. Serum was used for kidney function and kidneys for histological examination and quantification of apoptosis markers, pro-inflammatory cytokines and oxidative stress.
Results
CP-induced nephrotoxicity was apparent from histological damage and increased levels of BUN, Cr and UA. CP also caused an increase in malondialdehyde and decreased superoxide dismutase (SOD), glutathione (GSH), catalase (CAT) activities and led to up-regulation of pro-inflammatory cytokines and apoptosis markers. WG caused a significant decrease in Cr, BUN, UA, MDA and increase in GSH, SOD and CAT activities.
Conclusion
WG ameliorated histological damage and led to a reversal of cytokines and apoptosis markers. Both doses of WG effectively ameliorated CP-induced nephrotoxicity; thus, during chemotherapy, WG could be a promising adjunct to CP.
Introduction
Cisplatin (CP) is the most broadly used anticancer agents for treating various forms of malignancies, such as head, neck, breast, lung, esophagus, ovarian, and cervical cancers.1–3 The efficacy of CP used to treat testicular cancer achieves more than 90% cure rates. 4 However, the major unwanted effect of CP is irreversible kidney damage (nephrotoxicity), which limits its clinical utility. 5 Despite protective measure, around 30% to 40% of patients undergoing CP chemotherapy often suffer from a significant decline in renal function after the initial dose.6,7 Cisplatin-induced nephrotoxicity is manifested by high accumulation of CP and its metabolites within kidneys especially in S1 and S3 segment of proximal tubules (PT) causing renal tubular injury, diminish glomerular filtration and leading to chronic renal failure.8,9 Because of the lack of alternative choices, clinicians usually stop the use of CP in patients to prevent chronic renal failure. 10 However, CP is still the most preferred anti-cancer drug and its clinical utility cannot be abandoned in the present situation. 11 Hence, an urgent need to discover a new adjunct that mitigates the CP-related toxicity without affecting its anticancer efficacy exists.
CP induces nephrotoxicity via multiple signaling pathways such as oxidative stress, generation of pro-inflammatory cytokines, activation of apoptosis triggers, and autophagy.2,12 It is now evident that CP causes renal tubular injury by generation of reactive oxygen species (ROS) that leads to an imbalance in antioxidant defense, affects the activity of mitochondrial complex, causes lipid peroxidation, and promotes DNA damage.2,13–17 Additionally, the pro-oxidant capacity of CP plays a pivotal role in activation of NF-ĸB that subsequently leads to up-regulation of pro-inflammatory cytokines, like tumor necrosis factor alpha and interleukins beta and 6 (TNF-α, IL-1β, and 6, respectively), thus causing apoptosis.7,18–20 Recent studies also focus on CP-induced apoptosis via mitochondrial (intrinsic), death receptor (extrinsic) and endoplasmic reticulum (ER) stress pathways and discuss the role of TNF-α receptors, Bcl-2-associated X protein/B-cell lymphoma (Bax/BcL) ratio and oxidative stress in signaling of caspase cascades.2,10,21
In the search for potent adjuvants of CP, Wheatgrass ([WG] Triticum aestivum Linn., family Gramineae), a common food supplement in the kitchen, was selected (Figure 1). WG is a significant source of vitamins (ascorbic acid, α-tochopherol, β-carotene), enzymes (cytochrome oxidase and superoxide dismutase), and minerals such as calcium, magnesium, and potassium. Phytochemical investigations of WG performed via HPLC and GC-MS revealed the presence of phenolics such as gallic, ferulic, syringic, cholorogenic, p-coumaric and caffeic acids and a number of flavonoids such as apigenin, caempferol acid, and luteolin etc Besides, the chlorophyll content in WG is ∼70%.22–25 In vitro and in vivo studies have revealed the antioxidant potential of WG against clastogen induced DNA damage and cytotoxicity.26,27 WG is an excellent detoxifying agent that prevents metabolic activation of several carcinogens and facilitates removal of toxins from blood, liver, and kidney.28–33 Scientific evidence has established the anticancer potential of WG in the treatment of leukemia, hepatic, gastric, lung, colon, breast, and laryngeal cancers.23,34–39 WG has shown synergistic benefits in cancer chemotherapy and attenuation in chemotherapy-induced side effects, such as secondary organ damage and immunomodulation.40–42 WG has been shown to possess strong anti-inflammatory activity via suppression of NF-kB-induced generation of pro-inflammatory cytokines, such as TNF-α, IL-1β, and IL-6 and anti-apoptosis action via attenuation of Bax/-BcL mediated caspase-3. 25 WG supplementation has shown enormous benefits in cancer chemotherapy when given as an adjuvant to chemo drugs, has been proven to alleviate the activation of several carcinogens and possesses strong antioxidant potential. These properties inspired us to evaluate the capability of WG to modulate CP-induced nephrotoxicity, which is the major hurdle to the use of CP in chemotherapy.

GC-MS of Triticum aestivum (WG) with total ion chromatogram.
Material and Methods
Drugs and Chemicals
Renal function estimation kits for blood urea nitrogen, uric acid, and creatinine (BUN, UA, and Cr, respectively) were obtained from Randox (UK). The TNF-α, IL-1β, IL-6, and caspases-9 and -3 enzyme-linked immunosorbent test (ELISA) kits come from Abcam (UK). Sigma (St. Louis, USA) was used to buy cisplatin (CAS# 15663-27-1) and other analytical-grade chemicals were bought from same company. Wheatgrass plant was locally collected from Jazan and its powder was used for this study.
Extract Preparation
Wheatgrass powder (100 gm) was subject to Soxholet extraction with 350 ml of hydro-alcoholic solvent (ethanol plus distilled water in the 70:30 v/v ratios) for 72 h. The solvent extract was then evaporated to dryness under reduced pressure. The resulting dried extract (8.2% w/w) was stored at less than 4 °C until further use.
Sampling for GC-MS Analysis
The extract was filtered through 0.45 µm Whatman filter paper and subjected to vacuum distillation at 40 °C until a thick semisolid material was obtained. The resulting extract (1 mg) was dissolved in 1 mL of methanol, filtered through 0.45 µm nylon syringe filter and 2 μL was injected into the GCMS.
Animals
Male rats (200-250 g) were obtained from MRC, Jazan University KSA and transferred to the animal house of college of pharmacy for adaptation. For adaptation and experimentation, ideal lab conditions such as regulated room temperature of 22 ± 3 °C, standard humidity, and 12-h light/ dark cycle, were consistently maintained. Animals were provided free access to standard autoclaved food and drinking water. The study procedure was sanctioned by Jazan University Scientific Research Committee (approval no. REC-43/06/112). The ARRIVE guidelines 2.0 were followed carefully for conducting the animal research. 43
Experimental Design
Animals were randomly assigned to five groups, each involving of six rats: (i) Group 1, normal control (NC), receiving only vehicle distilled water; (ii) Group 2, nephrotoxic positive control (PC) receiving distilled water for two weeks followed by CP 7.5 mg/kg; (iii) Group 3, WG-200 mg/kg; (iv) Group 4, WG-100 44 +CP; and (v) Group 5, WG-200 45 +CP. The WG extract was suspended in distilled water along with 5% dimethyl sulfoxide (DMSO and pre-treated via an oral route 30-40 min before administration of CP, once daily for 14 days. To induce nephrotoxicity 1 ml/ 100 gm doses of CP were prepared by mixing the CP in 5% dimethyl sulfoxide (DMSO)-saline and administered on the tenth day via i.p. route. 46
Sample Collection and Preparation of Tissue Homogenate
At the end of the experiment no mortality was noticed in any group of rats and all animals were weighed and given light ether (diethyl ether) anesthesia to obtain the blood samples via retro-orbital puncture. 47 Blood samples were centrifuged at 3000 xg to collect the serum samples. The serum samples were refrigerated at −20 °C for further analysis of renal biomarkers.
After blood sampling, animals were sacrificed, and kidneys were dissected out for histological inspection and quantification of apoptosis markers, pro-inflammatory cytokines and oxidative stress. Prior to homogenization each kidney weight was determined. Kidney homogenates were prepared by mixing tissue sample in 10% (w/v) 10 mM chilled phosphate buffer (pH 7.4) along with protease inhibitor (10 μl/ml) in an Ultra Turrax T-25 homogenizer (IKA, Germany). The resulting homogenate was then centrifuged at a speed of 800 xg, at 4 °C for 5 min to separate cell debris. Now PMS (post mitochondrial supernatant) was then separated from the above sample by centrifuging the mixture at 10,500 xg, at 4 °C for 15 min.
Renal Histopathology
Ten percent formalin were used to fixed kidney tissues for two days and then dehydrated by soaking in alcohol and processed via xylene permeabilization. Tissue was then rooted in paraffin and 4-µm thick sections were ready. Finally, sections were stained with H&E (hematoxylin-eosin stain) as per the standard method and subsequently observed under light microscope (at 40 X) for histopathological examination. Tubular damage was examined, scored and presented as percentage of tubules showing epithelial damage in renal cortex: 0-normal, 1 if <10%, 2 for 10%-30% damage, 3 for 26%-75%, and 4 for >75%. Tissue damage scoring was based on lesions in proximal, tubular dilation, and distal convoluted tubules (PCT and DCT), degeneration in glomerular boundaries and tubular cell necrosis. 17
Assessment of Renal Function
Serum BUN, Cr, and UA concentrations were measured by commercially available kits, according to the manufacturer's guidelines. Sample absorbances were measured spectrophotometrically (Shimadzu UV-1601-Japan) at 490, 580, and 520 nm for Cr, BUN, and UA, respectively.
Determination of Renal MDA, SOD, GSH and CAT Activity
Kidney tissue homogenate was used for the estimation of protein content by Lowry et al (1951). 48 Determination of MDA levels were performed as per method described by Utley et al (1974). 49 SOD concentrations were assessed using method of Stevens et al (2000) while GSH and CAT activities were measured by Ashafaq et al (2020).50,51 Absorbances of all the reaction products were measured spectrophotometrically using a model UV-1601 Shimadzu spectrophotometer (Japan).
Assay of Pro-inflammatory Cytokines
Levels of TNF-α, and IL-1β and -6 were measured using Abcam (UK) ELISA kits according to manufacturer's protocol. These assays used a simple sandwich ELISA to measure the cytokine levels. Absorbances of duplicate sets for each sample were measured at 450 nm via the ELx 800TM BioTek ELISA microplate reader (USA).
Assay for Caspase-3 and -9
Caspases-3 and -9 were quantified using the manual included in the Abcam kits (USA). The caspase reactions were measured using an ELx 800TM BioTek ELISA reader that detected pNA light emission at 405 nm. The concentrations of both caspases were estimated using a standard curve.
GC-MS Analysis
The phytochemical screening of the WG extract was performed by Gas chromatography technique (ThermoScientific). 1 mL of WG extract was diluted to 10 mL of with methanol and 2 μL was injected using the autosampler at 250 °C. The separation was achieved in the capillary column (TR-5MS; 30 m length, 0.25 mm diameter and 0.25 μm particle size) with in house ramping protocol. The oven was initially set to 60 °C and was ramped subsequently to 100 °C, 140 °C, 180 °C, 220 °C and 250 °C with holding time five minutes at each stage. The mobile phase, helium gas, with a rate of flow of 1.2 mL/min was used to have an optimum separation with a run time of 70 min. The detector was set to a delay time 5 min and spectra was recorded to recognize the molecular masses ranging from 60-550 amu. At 270 °C for the source line and 250 °C for the transfer line, the temperatures were kept the same.
Statistical Analysis
The data analysis was achieved by using Version 8.4.3 of GraphPad Prism software (USA) through one-way-ANOVA, by multiple comparison (Tukey's) test. Statistical significance was defined as a p value of less than 0.05.
Results
GC-MS Analysis
The chemical constituents of wheatgrass (WG) abstract were determined using the software tools such as matching and reverse matching factors (SI and RSI) with a set value of above nine hundred. Xcalibur software was employed to calculate the percent relative area peak, by dividing its peak area by absolute area of all the peaks. Peak areas were estimated without an internal standard and are incorrect. All the discovered components are enlisted in Table 1 along with their percentage relative contents, retention duration, molecular formula and molecular weight.
Chemical Compounds of Wheatgrass Extract by gas Chromatography Mass Spectrometry (GCMS).

The WG extract appeared as a dark green, odorless semisolid. GC-MS analysis identified various compounds, including esters (43.4%), diterpenoids (17.44%), fatty acid derivatives (9.35%), sesquiterpenoids (7.58%), benzoic acid derivatives (6.01%), aldehyde derivatives (4.13%), oxaspiro compounds (1.8%), steroids (0.98%), glucoside (0.86%), and diphenyl derivatives (0.25%). In total, twenty-five compounds were identified, accounting for 93.58% of the total peak area based on mass balance. Peaks with match factor (SI) and reverse match factor (RSI) below 900 points were excluded to compensate for the lack of internal standard corrections (Figure 1).
Methyl linolenate (21.28%) was the most abundant compound in the extract. It is well-known for inhibiting the oxidation and peroxidation of unsaturated fatty acids, providing antioxidative and antimelanogenic effects. 52 Phthalic acid di(2-propylpentyl) ester (15%) was another predominant component, noted for its allelopathic, antimicrobial, insecticidal, and other biological activities. 53 The extract also contained a significant amount of ingenol (8.42%), a tetracyclic diterpenoid with a cyclic terpene ketone structure. Ingenol mebutate, a derivative of ingenol, is reported to have potent antibacterial, antifungal, and anticancer activities, particularly in treating actinic keratoses.54,55 Another sesquiterpenoid, fumagillol (4.55%), known for its antimicrobial properties, has documented antifungal and antigiardiasis activities.56,57 Salicylacetic acid (5.59%), a benzoic acid derivative, is described to have analgesic activities and anti-inflammatory. 58 Oleamide, a fatty acid amide derivative, has been shown to induce sleep in some animals. Lecocarpinolide H (1.64%), a sesquiterpene lactone, exhibits antibacterial activity. Additionally, aromatic phenolic aldehydes, steroids, and oxaspiro compounds were detected. The nature and content of chemical components in the WG extract differed significantly from those reported in the literature.23,59 The details of all 25 chemical compounds are given below in Table 1.
Result of WG on Body Weight and Kidney Weight
Figure 2(A and B), represents WG's impact on body weight and relative kidney weight. After 5 days of CP administration, final body weight of each animal was recorded, and percentage of weight loss/ gain was calculated. In CP treated PC group, we observed a 14.23% body weight loss as compared to initial body weight (zero day). However, WG (100 and 200 mg/kg) significantly (P < .0001) attenuated the CP-induced body weight loss as compared to PC. Similarly, relative kidney weight in CP treated PC animals was significantly (P < .0001) increased as compared to NC. Treatment with WG at different dose levels significantly attenuated the increase in relative kidney weights as compared to PC (P < .0001). While studying, no deaths were detected in either CP and/ or WG treated animals. During the study period all group animals were healthy, survived till the end of experiment and their food intake was normal except CP-treated PC animals in which recorded mortality was 16.66%.
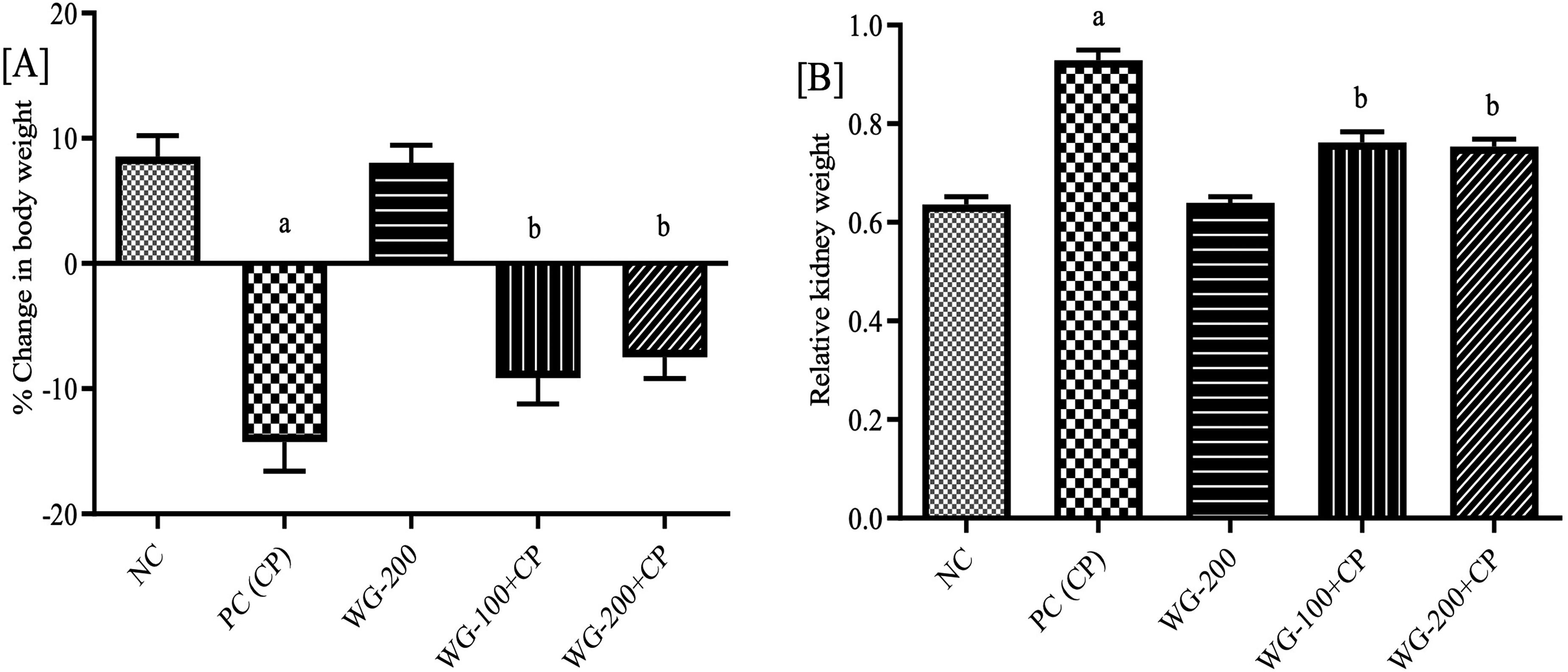
Effect of wheatgrass treatment on body weight [A], and relative kidney weight [B] in cisplatin treated rats. Data expressed as mean±SD (n =6). Abbreviations: NC, normal control; PC, positive control; CP, cisplatin; WG, wheatgrass. aP < .0001 versus NC; bP< .0001; versus PC.
Renal Histopathology
Microscopic sections exhibited the regular shaped Bowman's capsule with normal histological structure of nephron in the NC kidneys (Figure 3A). Examination of CP-treated animals revealed necrosis, capillary congestion, and desquamation of epithelial cells with hemorrhagic edema in the glomerular basement membrane, proximal convoluted tubule and distal convoluted tubules (PT and DT, respectively) as shown in Figure 3B. Treatment with various doses of WG (100 and 200 mg/kg) caused significant attenuation of renal tubular damage and CP-induced degenerative changes (Figure 3D-F) in which glomerular degeneration and tubular epithelial cell injury were found lower than in the CP group. Assessment of tubular damage score revealed that WG significantly ameliorated the CP-induced lesions in PT and DT, and degeneration and necrosis within the renal tubules. However, no significant lesions were observed in the WG-200 mg/kg treated group, and kidney architecture closely resembled the NC kidney structure (Figure 3C).

Hematoxylin and eosin (H&E) staining represents. [A] NC with clear glomerular filtration (GF) and normal renal tubules (proximal convoluted tubules-PT; distal convoluted tubules-DT). [B] CP treated PC showing toxicity with necrosis in the PT & DT with debris in lumen and GF basement layer degradation. [C] WG-200 mg/kg, no significant changes in the PT & DT and GF basement layer [D] CP with WG-100 mg/kg, and [E & F] CP with WG-200 mg/kg showing improvement in PT, DT and GF basement layer. Assessment of the tubular and glomerular damage score from H & E-stained slide. Data expressed as mean±SD (n=6). Abbreviations: NC, normal control; PC, positive control; CP, cisplatin; WG, wheatgrass. aP <.0001 versus NC; bP< .0001 versus PC.
Result of WG on Renal Function
Renal function test data are depicted in Table 2. CP caused significant renal dysfunction as observed in kidney parameters in which CP treatment led to a significant increase in serum Cr (4.97 ± 0.76), BUN (153.51 ± 9.04), and UA (2.39 ± 0.45) levels as compared to NC (Cr 1.29 ± 0.21, BUN 14.54 ± 5.01, UA 0.78 ± 0.09). WG caused significant attenuation in a dose-dependent way for all the markers of kidney injury. WG-100 mg/kg produced a significant reduction in nephrotoxicity as shown by decreased levels of Cr (3.27 ± 0.51), BUN (108.32 ± 12.56), and UA (1.29 ± 0.1) which was around 34% to 46% recovery from the induced nephrotoxicity as compared to CP-treated PC. WG-200 (designated as WG-200 + CP) was the most protective dose against CP as it caused significant decrease in serum Cr (2.15 ± 0.37), BUN (69.74 ± 7.76), and UA (0.92 ± 0.08) levels (around 55% to 61% recovery) compared to the PC group. Treatment with WG-200 mg/kg alone did induce the significant variations in renal function parameters compared to NC group animals and proved the safety of WG on kidney function.
Effect of Wheatgrass Treatment on serum Cr, BUN and Uric Acid Against Nephrotoxicity Induced by Cisplatin.

Data expressed as mean±SD (n=6). Abbreviations: NC, normal control; PC, positive control; CP, cisplatin; WG, wheatgrass; Cr, creatinine; BUN, blood urea nitrogen.
aP< .0001 versus NC.
bP< .0001 versus PC.
Result of WG on Renal MDA, SOD, GSH and CAT Activity
Results of antioxidant activity revealed that CP (in PC group) caused significant decrease in GSH (14.96 ± 2.45), SOD (115.98 ± 6.13), and CAT (15.54 ± 1.61) activities while causing an increase in lipid peroxidation (measured as MDA content, 61.46 ± 8.01) compared to NC (P < .0001) as shown in Table 3. Meanwhile, dose combinations of WG and CP caused significant attenuation of CP-induced oxidative stress. After 14 days of continues administration of WG-100 mg/kg, significant increases in the activities of GSH, SOD, CAT (21.33 ± 3.53, 189.08 ± 13.72, and 22.04 ± 3.42, respectively) and reduction in MDA (46.28 ± 3.41) were observed compared to PC (ranging from P < .05 to P < .0001), while similar but dose dependent protection was observed with WG-200 + CP compared to PC (P < .0001).
Effect of Wheatgrass Treatment on Cisplatin Induced Oxidative Stress in Renal Tissue.

Data expressed as mean±SD (n=6). Abbreviations: MDA, malondialdehyde; GSH, glutathione; SOD, superoxide dismutase; CAT, catalase.
aP< .0001 versus NC.
bP < .0001.
cP < .001.
dP < .01.
eP < .05 versus PC.
Result of WG on Cytokines
The results of WG's effects on pro-inflammatory cytokines are summarized in Figure 4[A-C]. The TNF-α level (364.89 ± 9.39) was significantly higher (P < .0001) in CP-treated animals compared to the NC (259.01 ± 6.09). Moreover, compared to NC, the levels of IL-1β and -6 in the CP group were significantly elevated to 1537.67 ± 21.48 and 2378.33 ± 40.73, respectively (P < .0001). Interestingly, WG caused significant amelioration of the increase in levels of TNF-α, and IL-1β and -6 against CP compared to PC in a dose-dependent manner (P < .0001). The recovered level of pro-inflammatory cytokines in WG-200 + CP treated group were close to values seen in the NC group. Though, no major changes were noticed between NC and WG-200 mg/kg treated group for pro-inflammatory cytokines.

Effect of wheatgrass treatment on renal IL-1β [A], IL-6 [B], and TNF-α [C] levels induced by cisplatin. Data expressed as mean±SD (n=6). Abbreviations: NC, normal control; PC, positive control; CP, cisplatin; WG, wheatgrass; IL-1β, interleukin-1β; IL-6, interleukin-6; TNF-α, tumor necrosis factor-α. aP < .0001 versus NC; bP <.0001 versus PC.
Effect of WG on Caspase-3 and -9
Compared to NC, CP caused up-regulation of the expression of caspase-9 (746.83 ± 18.01) that subsequently caused rise in caspase-3 (765.17 ± 14.7). The expression of caspase-3 and -9 was three-fold higher compared to NC (P < .0001). Administration of WG-100 + CP led to a significant decrease in caspase-3 (308.67 ± 5.85), and -9 (362.17 ± 14.49) by nearly 50% compared to CP (PC) and with WG-200 mg/kg. Moreover, compared to NC, no significant differences were observed with WG-200 mg/kg treated group, indicating that WG did not produce apoptotic effects on normal healthy renal tubular cells [Figure 5(A, B)].

Effect of wheatgrass treatment on renal apoptotic marker caspase-3 [A] and caspase-9 [B] up-regulated by CP. Data expressed as mean±SD (n=6). Abbreviations: NC, normal control; PC, positive control; CP, cisplatin; WG, wheatgrass. aP <.0001 versus NC; bP < .0001 versus PC.
Discussion
This study explored the importance of WG in CP-mediated nephrotoxicity in a rat model for the first time. The outcomes displayed that CP caused a significant increase in the serum markers of renal function and, increase in the expression of cytokines and apoptosis while primary to a decreased in antioxidant defense in kidney tissue, which were inhibited by WG treatment. The nephroprotective effects of WG obtained from the conclusions of biochemical estimation were further sustained by histological examination of kidney.
CP's nephrotoxicity is dose-dependent and cumulative. This is due to high accumulation of CP (∼5 times greater than blood) in the kidney. According to rat model studies, single i.p. injection (between 4 to 10 mg/kg) reaches to systemic circulation then rapidly distributes to almost all organs then mainly eliminate via kidney. As per previous reports, on single i.p administration in rats, about 50% of the CP eliminate via urine in the first 24 h, 60% to 76% in 48 h, and ∼90% in 72 h. Within kidney, the highest concentration of CP is found in the inner cortex and corticomedullary region (region of PT and DT). It was also reported that 72 h after CP administration, the maximum level of CP was noticed in mitochondria followed by cytosol, nuclei and microsomes (37, 27, 22% and 14%, respectively as observed within 5 days of single i.p. injection of CP). 60 CP severely affects renal excretion and lead to accumulation of protein metabolism waste products such as Creatinine, blood urea nitrogen, and uric acid causes acute tubular necrosis and glomerular basement layer degeneration.3,10,61,62 Kidney tubular damage refers to injury to the tubular structures of the kidney, which are responsible for reabsorbing water, electrolytes, and other essential substances from the filtrate. When these tubules are damaged, their function is impaired, leading to the leakage of substances such as creatinine, uric acid, blood urea nitrogen and albumin into the blood or urine that serve as markers of kidney injury.
In the present study, CP caused 3.75, 3.69 and 3-fold increases in serum Cr, BUN, and UA, respectively, which were consequently reflected in the histological examination. While WG administration counteracted the effects of CP and led to a significant improvement in renal function and tissue architecture in a dose-dependent way. Moreover, higher protection was provided by the higher dose of WG (WG-200 mg/kg), which led to a reversal in the CP-induced renal function parameters and tubular damage score by more than 50%. This finding is in accordance with WG's earlier reports that demonstrated the protective role of WG against lead-, mercuric chloride- and alcohol-induced nephrotoxicity.33,63
CP disrupts renal physiology by targeting key molecule production via multiple signaling pathways, generating ROS, and causing an imbalance in antioxidant defense (Figure 6). 64 Within PT cells, CP is bio-transformed by glutathione-S-transferase to form glutathione-S-platinum conjugates that cause intracellular depletion of GSH and lead to higher ROS production.65,66 Once a platinum conjugate passes through the lumen, it is cleaved to cysteinyl-glycine and then to cysteine conjugates at which point is further transported into the PT and again metabolized by cysteine-S-conjugate β-lyase to convert it into a highly potent nephrotoxic thiol.65,67,68 High accumulation of CP in the kidney leads to impairment of the mitochondrial respiratory chain by disrupting enzyme complexes I-IV, leading to loss of mitochondrial –SH proteins, inhibition of calcium uptake, and changes in membrane potential. In addition, depletion of ATP and other co-factors are also key events in over-production of ROS and cellular damage.7,11,66,69 CP-induced ROS formation causes lipid peroxidation, damages lipid components, and changes the structure and permeability of cellular membrane.17,70 CP targets endoplasmic reticulum (ER) of renal proximal tubular cells (PTECs), induces ROS production via the microsomal CYP450 enzymatic system (subfamily E/ CYP2E1), causes prolonged ER stress (ERS) and hypoxic injury, which are the leading causes of apoptosis and necrosis.10,66,71 CP-induced oxidative stress mainly involves ROS, like superoxide anions, H2O2 radicals, and very reactive hydroxyl radicals that cause DNA double strand breakages and eventually lead to cell death.10,27,72,73 In this study, significant CP-induced elevations in MDA is a reflection of ROS-induced lipid peroxidation, while decreased level of GSH demonstrate the high affinity of CP to form the CP-SH complexes that lead to GSH depletion. At the same time, the decrease in SOD and CAT activities reflect the capacity of CP to induced superoxide anions, hydrogen peroxide, and other radicals. 33
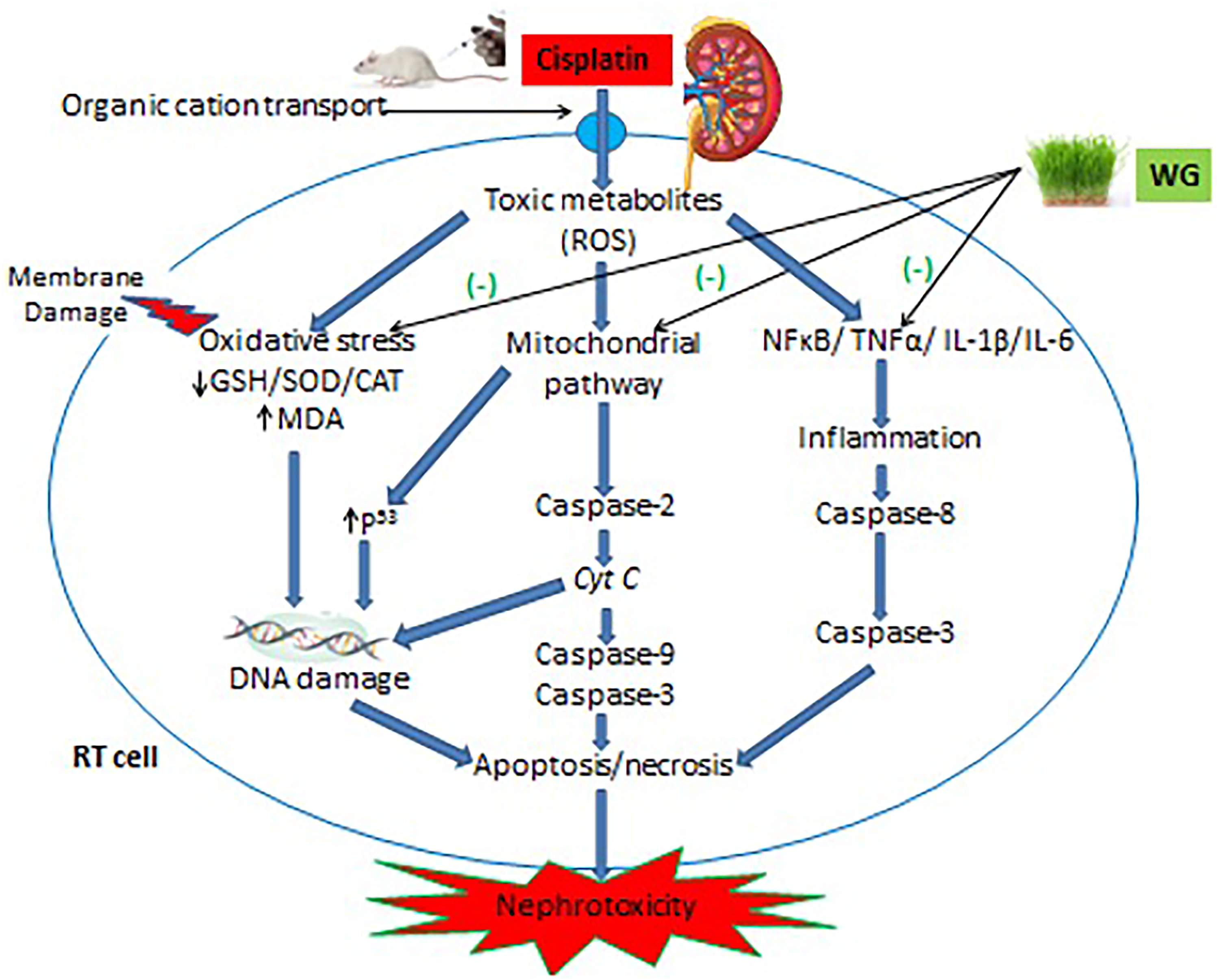
CP based nephrotoxicity management and its reversal by wheatgrass: mechanistic approach.
Various findings have correlated the role of antioxidants in prevention of CP-induced nephrotoxicity. In the current investigation, the role of oxidative stress in the augmentation of CP-induced nephrotoxicity by WG was investigated. Surprisingly, these events were significantly reversed with the 2-week continues administration of WG thus confirming the antioxidant potential of WG. WG supplementation provides superior protection against lipid peroxidation as shown by our results (specifically those with MDA) and agree with the human trials. 74 Besides, chlorophyll, which is most effective constituents of WG, reported to suppress the metabolic activation of several carcinogens and increase the kidney concentration of GSH against CP in mice. 75 At the same time, WG caused an elevation in the concentrations of SOD and CAT, demonstrating the role of WG in augmentation of superoxide anions and hydrogen peroxide radicals formation induced by CP. 76 In addition, WG contains SOD enzyme hence elevation of SOD can also correlate to WG's SOD mimetic action that played important role against CP-induced nephrotoxicity.4,77
Signaling pathways activating pro-inflammatory cytokines are considered as the key regulators of nephrotoxicity (Figure 6). In PTECs, CP activates the NF-κB pathway via phosphorylation of NF-κB. 60 Activated NF-κB then translocates toward the nucleus in which it promotes transcription of TNF-α and other cytokines, like IL-1β and -6. 78 The present study a significant rise in concentration of TNF-α and IL-1β and -6 in CP-treated PC animals compare to NC was noted. Enhancement of TNF-α and IL-1β and -6 genes contribute to nephrotoxicity. 79 Previous research has shown that higher levels of TNF-α and IL-1β cause more adhesion molecules to be made by renal endothelial cells. This leads to more circulating leukocytes entering the inflamed renal tissue causing more profound inflammation and kidney damage.60,80,81 On the other hand, production of IL-6 in renal tubular epithelium (RTE) cells promote the renal tubular atrophy, collagen-I generation and enhance tubulointerstitial fibrosis. 82 We observed that management with WG produced significant reduction in TNF-α and subsequently IL-1β and -6. Our findings agree with Avisar et al (2020) claimed that supplementation of WG with oxaliplatin (a platinum-based chemotherapeutic agent) suppressed chemotherapy-induced expression of cytokines in cancer patients. 43 Besides, direct evidence has proven that WG causes suppression of up-regulation of IL-6 and inhibited of NF-κB and IL-1β expression in immune-compromised mice.25,83 An increase in cytokine levels is also related to the imbalance between pro-/-antioxidant mechanisms, leading to generation of ROS and loss of antioxidant defenses. Many of such reactive molecules directly activate the signaling cascade that results in the production of TNF-α. 84 Luyen et al (2014) demonstrated that phenolic components of WG can produce a reduction in TNF-α expression during inflammatory processes and hence become useful in the treatment of inflammatory diseases. 44
CP has been reported to cause apoptosis even at the lowest concentration of 8 µM in RTE cells. In this study, CP caused a significant rise in the expression of caspases-3 and -9, which were measured as the markers of apoptosis. CP-induced apoptosis has been associated with several pathways, including intrinsic (mitochondrial), extrinsic (death receptor), and ERS pathways. 3 Intrinsic and ERS pathways are linked to ROS-induced oxidative stress. 10 Once CP enters RTE cells and induces oxidative stress, translocation of pro-apoptotic Bax (a pore- destabilizing protein) toward mitochondria occurs followed by activation of caspase-2 that results in the release of cytochrome C (Cyt c). Apoptosis-protease activating factor 1 (APAF-1) is mediated by Cyt c needed for photolytic progress of initiator caspase-9 and executioner caspase-3 for apoptosis.2,7 In the present study, WG led to a significant down-regulation of caspases-3 and -9 expressions in the kidney against CP. This down-regulation can be correlated to the capacity of WG to quench ROS. The antioxidant potential of WG may result in mitochondrial blockade of events involved in apoptosis, such as up-regulation of caspases-9 and -3. 72 Moreover, the mitochondrial pathway also involves caspase-mediated apoptosis as a result of CP-induced DNA damage in kidney tubular cells through activation of p53, leading to Bax-mediated mitochondrial membrane permeabilization and release of Cyt c.2,7,16,66 WG also prevents chemotherapy-induced DNA damage and therefore prevent the activation of p53-mediated caspase cascades in renal tubular cells. 75 CP-induced apoptosis via ERS pathways mediated by CYP450 (CYP2E1) in ER membranes of proximal tubular epithelial cells causes oxidative stress and activation of initiator caspase-12 and downstream caspase-3.61,85–87 WG leads to inhibition of the expression of enzyme CYP2E1, which mediates the ERS pathway and hence prevents activation of caspase-3 and apoptosis of renal tubular cells.2,88,89 TNF-α binds to death receptors on the plasma membrane as part of the extrinsic route causes renal tubular apoptosis. Since CP increased in the TNF-α expression, the interaction of TNF-α with the TNF-receptor occurs and directly triggers activation of caspases-8 and -3, leading to apoptosis.1,2,33 As evident from current study together with earlier reports, WG inhibit the expression of TNF-α via minimization of oxidative stress and inhibition of NF-κB; hence, WG interferes with the interaction of TNF-α with death receptors thus preventing caspase-mediated apoptosis through extrinsic pathway.44,83,84
Chemotherapy is crucial for cancer treatment and in management of cancer plant-based products are gaining popularity as vital resources. In the present investigation, administration of WG alone did not induce significant toxicity in the normal healthy rat kidney; however, after, combining WG with CP, the WG caused a reduction in the nephrotoxicity induced by CP. Nephrotoxicity is the most frequent and dose-limiting side effect of CP therapy. Hence, WG as an adjuvant to CP can not only prevent chemotherapy-induced nephrotoxicity but also become a vital resource in cancer therapy. Experimental research on the ameliorating effects of Triticum aestivum (wheatgrass, WG) against cisplatin-induced nephrotoxicity often has several limitations, which can affect the interpretation and applicability of the findings in human. The effects of WG may vary significantly among individuals due to genetic, dietary, or environmental factors, which are required to addressed in preclinical studies.
Conclusion
The present study demonstrates the defensive role of WG against Cisplatin-induced nephrotoxicity. The nephroprotective effect of WG may be due to combined and synergistic effects of complex mixture of bioactive phytoconstituents present in WG. These phytoconstituents not only scavenge ROS generated during biotransformation of CP, as well as interfere in multiple signaling pathways associated with generation of pro-inflammatory cytokines, DNA damage, and caspase cascades that lead to apoptosis. WG is one of the best household anticancer herbal drugs and as an adjuvant to chemotherapy could not only prevent the severity of malignancy but also adverse chemotherapy-associated effects. However, further studies are required to yield more insight.
Summary Point
Cisplatin is commonly employed in the treatment of many malignant malignancies but, it is associated with significant adverse effects, including renal toxicity by multiple pathway (ROS, Inflammation, Apoptotic etc)
Many plant and its active ingredient reported the renal protection aaginst cisplatin due to high antioxidant, anti-inflamatory and therepuetic value but very limitted or no study have been conducted for WG against cisplatin.
In this study WG ethanolic extract was used to protect the renal toxicity by reducing the oxidative stress and inflammation through deroxyfying the free radicals generated by doxorubicine.
Biochemical effects of WG effects in rat models and improve antioxidant enzymes such as glutathione, catalase, superoxide dismutage by reducing oxidative stress. It also successfully reduces the renal specific marker in serum.
Inflammatory effects of WG dramatically reduces the important inflammatory cytokines and apoptotic markers such as IL-1b, IL-6, TNFα and Caspase-3and 9. Overall WG protection also refelected in renal histolog.
WG is one of the best household anticancer herbal drugs and as an adjuvant to chemotherapy could not only prevent the severity of malignancy but also adverse chemotherapy-associated effects.
Future direction and conclusion: The rsults suggests that WG may offer a novel therapeutic approach for renal problem and related diseaeses by adding in suplimentation with food. However, further studies are required to yield more insight.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors express their sincere gratitude for the financial support provided by the Deanship of Graduate Studies and Scientific Research at Jazan University, Saudi Arabia, under project number RG24-S0106.
Ethical Approval
The study protocol was permitted by Jazan University Scientific Research Committee (approval no. REC-43/06/112).
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the institutional animal care guideline and approved by the Scientific Research Committee of Jazan University, Jaza, Saudi Arabia (approval no. REC-43/06/112 dated 18/01/2022).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
