Abstract
Objectives
Bryophyllum pinnatum is a medicinal plant frequently used in the treatment of anxiety and of preterm labor; however, placebo-controlled studies evaluating its efficacy are currently lacking. Our goal was to characterize clinical outcomes needed to investigate the efficacy of Bryophyllum 50% chewable tablets (à 350 mg; made from leaf press juice of Bryophyllum pinnatum plants grown in Brazil; Weleda AG Arlesheim, Switzerland) on the treatment of preterm labor-related anxiety. Here, data collected at study discontinuation (n = 16) and learnings are described.
Methods
Patients (pregnancy week 22 to 34) suffering from signs of preterm birth, after 48-h standard tocolysis, without indications for imminent delivery were recruited for this placebo-controlled double blinded study. Primary endpoint was anxiety (at day 1, 7 and 14, assessed with standard questionnaires). Sleep quality, gestational age at birth and hospitalization days were secondary outcomes.
Results
Study was discontinued after 16 patients had been randomized with 10 of them completing the study (5 per group). At day 1, anxiety level was intermediate, and sleep quality was good; corresponding scores changed minimally in time, no tendencies for differences between the two groups were detected. Nine out of the 10 patients delivered at term, ie, after completing 37 weeks of pregnancy. In the verum (herbal medication) and placebo groups, delivery occurred after median 2 (range 2-6) and median 12 (range 2-56) days in an in-patient facility, respectively.
Conclusion
Our approach did not seem to capture anxiety related to preterm labor, due to either selection bias or inappropriateness of the questionnaires used. However, the pragmatic approach of counting the days spent in an in-patient facility as a proxy for successful preterm labor treatment appears promising for future studies.
Study registration at clinicaltrials.gov NCT05110599, November fourth 2021
Keywords
Introduction
Premature birth is one of the most common causes of neonatal morbidity and mortality. Two thirds of preterm births are associated with premature contractions (with or without preterm premature rupture of membranes; PPROM); therefore, inhibition of labor by tocolysis is the main approach in the prevention of preterm birth. The calcium channel blocker nifedipine (off label use) and the oxytocin receptor antagonist atosiban (approved for the indication) are the most commonly used drugs for the pharmacological treatment of premature contractions and recommended as first line therapy in current guidelines. 1 Due to significant side effects associated with conventional therapies and a lack of sufficient evidence indicating improved maternal and fetal outcomes with maintenance tocolysis, its administration should not be continued beyond 48 h in most cases.1–3 Several studies have shown that psychosocial stress and anxiety generally have a negative influence on the outcome of pregnancy, but particularly on the outcome of preterm labor. 4 Therefore, drugs that would reduce anxiety and inhibit uterine contractility are especially promising. One such drug could be the herbal medication Bryophyllum pinnatum.
B. pinnatum (Lamarck) Oken [syn.: Kalanchoe pinnata (Lamarck) Persoon, syn.: Bryophyllum calycinum Salisbury] grows in many parts of tropical regions, including Brazil, and is used for various indications in its countries of origin.5,6 In Europe, B. pinnatum was first introduced into anthroposophic medicine in the twentieth century. The evaluation of a German database (EvaMed) showed that Bryophyllum products were most commonly prescribed for “mental disorders” including anxiety 7 and a survey among physicians and nurses working at an anthroposophic hospital in Switzerland arrived at the same conclusion. 8 A recent prospective, single-arm study revealed marked improvements in anxiety-related symptoms during treatment with chewable tablets made from leaf press juice of Bryophyllum pinnatum plants grown in Brazil (à 350 mg; Weleda AG Arlesheim, Switzerland). 9 In line with results from additional studies on the treatment of sleep disorders with the same preparation,10–12 concomitant improvement in the sleep quality was reported among the patients with anxiety-symptoms. 9 Despite a detailed phytochemical characterization of the B. pinnatum leaf press juice,13,14 relatively little is known about the mechanism of action in the central nervous system when B. pinnatum is used as a sedative or anxiolytic. However, there are results from rodent studies showing that leaf extracts of B. pinnatum prolong the pentobarbital-induced sleep duration and possibly lead to an increase in the concentration of GABA in the brain, which indicates a CNS depressant effect. 15 More recently, B. pinnatum was shown to attenuate anxiety-like behaviors in zebrafish. 16 Current investigations suggest an activation of GABA receptors as a mechanism of action for B. pinnatum preparations. 17
Nowadays in Switzerland, B. pinnatum is frequently used in obstetrics, primarily as an add-on tocolytic.18–20 The roots of its use for tocolysis date back to the 1970s, when it was initiated at an anthroposophic hospital in Germany because of the calming and sedative properties. 21 Initial retrospective studies showed that B. pinnatum has good effectiveness and very good tolerability in patients with premature contractions.21–23 A subsequent matched-pair study supported previous findings on the use of B. pinnatum and showed much better tolerability than β-adrenoreceptor blockers. 24 Finally, a randomized trial for the acute treatment of premature contractions compared to nifedipine was initiated. Although this trial had to be terminated prematurely due to slow recruitment of participants, the results at discontinuation were promising, as they showed a reduction in the number of contractions in the first four hours both in the nifedipine as in the B. pinnatum group. 25 With basis on this latter study, B. pinnatum was among the candidates with medium potential for preterm birth treatment in clinical development identified by the Accelerating Innovations for Mothers database. 26 The effectiveness of B. pinnatum remedies in the treatment of preterm labor is well supported by various in vitro studies.27–29 The class of substances known as bufadienolides, which occurs in the species Bryophyllum, seems to be mainly responsible for the inhibition of contractility, 30 even if synergies among various types of molecules are required for inhibitory effects at the cell signaling level. 31
Up to now, no placebo-controlled randomized trial was conducted to investigate the clinical efficacy of B. pinnatum in the treatment of preterm labor and such evidence is urgently needed to justify its further use in clinical practice. Moreover - and despite the known influence of psychosocial aspects on preterm labor - previous efforts to generate the necessary clinical evidence for B. pinnatum use in tocolysis did not consider the possible contribution of anxiolytic effects. The aim of this pilot study was to explore various instruments for the characterization of efficacy and safety of the add-on treatment of preterm labor with B. pinnatum. Both effects on anxiety and on preterm labor were to be investigated. In addition, longitudinal changes and differences between the two study groups were to be studied.
Subjects and Methods
Study Participants
The study was authorized by the ethical committee of Zurich as well as by the Swiss agency for therapeutic products (Swissmedic) and was conducted according to good clinical practice (trial registration NCT05110599, November fourth, 2021). All study participants gave written informed consent. Patients admitted to the University Hospital Zurich due to signs of preterm birth (premature contractions, prenatal bleeding, and/ or preterm cervix maturation) were screened for eligibility. Patients fulfilling the following inclusion criteria were asked for their consent to participate in the study: at least 18 years old, signs of preterm birth, gestational age between 22 + 0 and 34 + 6, good German or English language skills. Patients excluded from this study were those who: presented with PPROM, showed signs of imminent delivery (preeclampsia, suspect cardiotocography (CTG), signs of infection, body temperature above 38 °C, cervix length below 5 mm), had a contraindication for B. pinnatum, lactose intolerance or wheat protein allergy, had taken either Bryophyllum 50% tablets or powder 48 h before study recruitment, or psychiatric drugs (benzodiazepines, antidepressants, neuroleptics), suffered from diagnosed psychiatric diseases; had participated in an interventional trial during the last 4 weeks, or acknowledged drug or alcohol abuse. The inclusion criteria differed at the beginning of the trial but were later amended twice, due to lack of successful recruitment. In brief, the aims of the first amendment were to allow the participation of women who had taken Bryophyllum 50% in the previous month if they had not taken it in the last 48 h, and/or continued to receive tocolysis after the initial tocolysis cycle of 48 h, and/or had a cervical length between 10 and 5 mm. The aims of the second amendment were to further allow the participation of women with multiple pregnancies, and with earlier and later gestational ages. Initial and amended participation criteria as accepted by the ethics committee and the Swiss agency for therapeutic products can be found in the supplementary information.
Study Design
In the study design, several methodological elements were incorporated to ensure precision and objectivity, including a randomized, double blind, and placebo-controlled approach. These properties aimed to eliminate biases and placebo effects. Participants were allocated manually and randomly to the two study groups with a variable block size from 4 to 8 and a 1:1 ratio. The randomization list was generated by the company providing the study medication (Weleda AG) and neither patients, treating physicians nor the study team were aware of the allocated study group of the patient. A copy of the randomization list was kept by the hospital pharmacy to enable de-blinding in case of emergency. Only after the study had been discontinued, was the group allocation of each patient revealed to the researchers.
The screening procedure occurred shortly after hospitalization, enabling early identification of eligible participants by physicians of the prenatal ward. After study inclusion and randomization, patients filled out anxiety questionnaires on days 1, 7 and 14, allowing for a longitudinal evaluation of the treatment impact. To ensure compliance and monitor safety, participants were contacted every three to four days throughout their involvement in the study. This proactive approach facilitated both adherence to the study protocol and prompt identification of any potential adverse events. For discharged patients, contact was maintained via telephone to ensure continuity in data collection. Shortly after the expected date of birth, the study participant was contacted again to gain information about the birth (if it did not take place at the University Hospital Zurich).
Because no data on the distribution of the anxiety scores in women under preterm labor were published at the time the study was designed, no formal sample size planning was performed, and the initial goal was to recruit 60 patients for this pilot study. Data of this pilot study should have provided necessary data to enable the planning of sample sizes for future studies. However, due to unforeseen multifactorial delays in recruiting patients meeting the initial criteria, the target number was not achieved before medication expiry, resulting in study termination for administrative reasons.
Study Medication
Bryophyllum 50% chewable tablets are approved by the Swiss Agency for Therapeutic Products (Swissmedic) without indication by means of notification and are already used in everyday clinical practice at the University Hospital Zurich. Two tablets of the study medication were taken four times daily over two weeks of study participation. The dosage is based on USZ guidelines for the use of B. pinnatum as a tocolytic agent. 32 Weleda AG (Arlesheim, Switzerland) produced the study medication by drying fresh pressed leaf juice with lactose (composition of the active ingredient: 5 g leaf press juice processed in 10 g of lactose) and according to good manufacturing practices (GMP). A voucher specimen (no. ZSS29717) of the Brazilian plants used for leaf collection has been deposited at the Zurich Succulent Plant Collection, Switzerland. Lactose tablets were used as a placebo, as lactose is also the primary ingredient in the Bryophyllum 50% chewable tablets. The color and taste of the placebo tablets were adapted to that of the investigated product. In the present manuscript, the investigated product – Bryophyllum 50% chewable tablets – is often referred to as verum and the product not containing any active ingredient is named placebo.
Endpoints
The questionnaires 1, 2 and 3, filled out by the participants during the two weeks of study medication intake, consist of three validated instruments for assessing
As
Statistical Analysis
Baseline characteristics and outcomes of study participants were summarized as means with corresponding standard deviations (Table 1). Due to the low number of participants, data were analyzed only descriptively using IBM® SPSS® Statistics (Version 26.0 for Windows). Importantly, we decided to forgo the calculation of the statistical significance of the differences between the outcomes in the two treatment groups, as in the absence of enough patients for a multivariate analysis with the important covariates, this could have led to false conclusions.
Baseline Characteristics and Outcomes of Study Participants Treated with B. pinnatum or Placebo. Data are Expressed as Mean ± SD, or Number of Patients.

*multiple medications possible **no information in 3 cases.
Results
A total of 109 patients were screened during the recruitment phase of the study (first patient screened on 09.11.2021, last patient on 14.06.2023), of whom 82 were ineligible for study participation. Patient eligibility was strongly improved after adaptation of inclusion and exclusion criteria with the two amendments (see Figure 1). Accordingly, the majority of eligible patients was informed, and most participants were recruited during the last five months of the trial, following the final version of the study protocol (24 out of 26, and 15 out of 16, respectively). Before the last and successful amendment, 42 patients were screened, but only two were eligible and one was recruited. Most patients considered ineligible were already taking Bryophyllum 50% products at the time of screening. Almost half of the patients informed of their eligibility were not willing to participate in the trial.
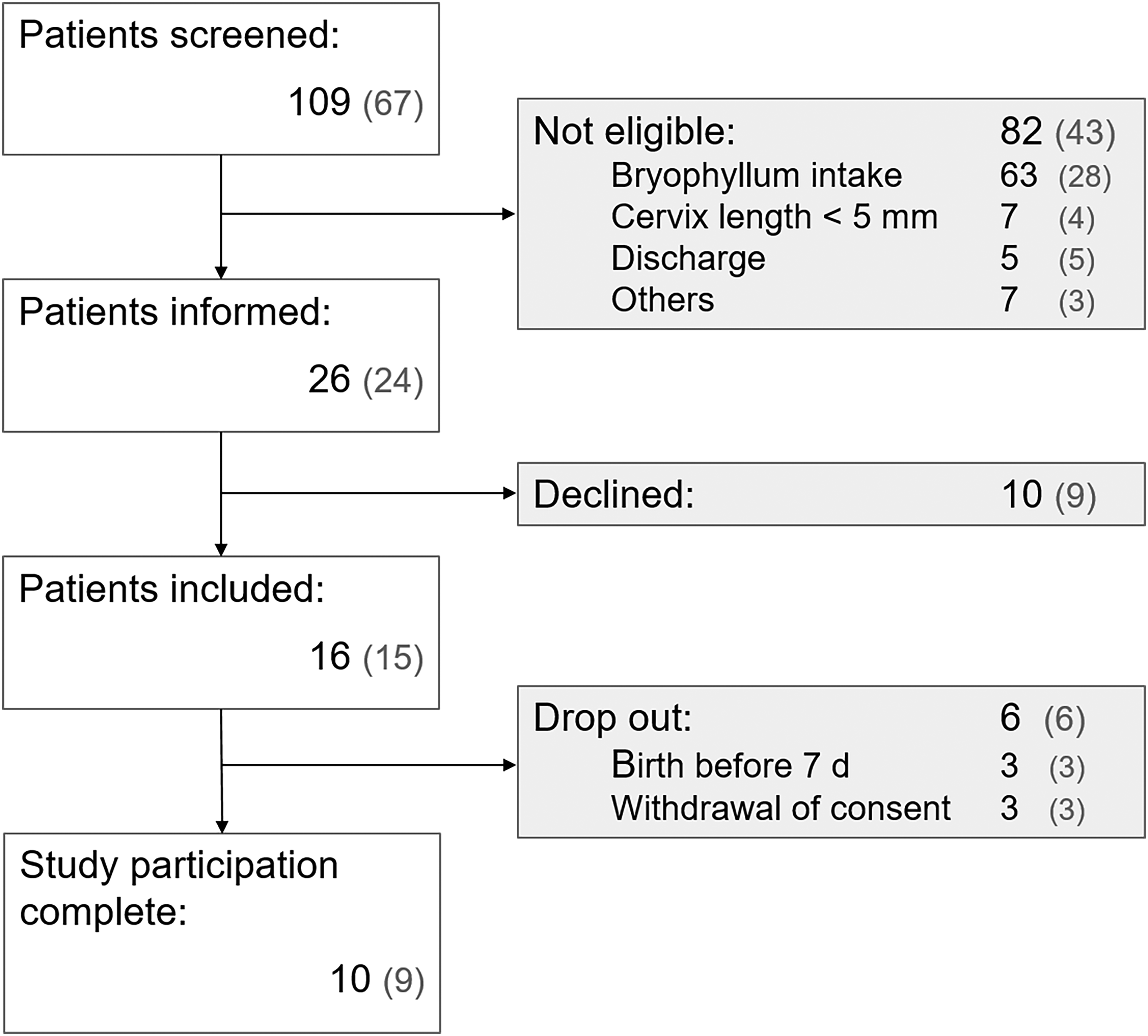
Flow chart of patient recruitment process showing total number of patients in each step.
Of the 16 recruited patients, ten had completed study participation at study discontinuation, five per group. The baseline characteristics of the patients included in the study were comparable in the two study groups. However, one parameter differed between patient groups at the time point of randomization: the gestational age for patients in the Bryophyllum group was on average four to five weeks higher than in the placebo group. Furthermore, the average cervix length measured 5 mm longer in the Bryophyllum group. All patients received standard tocolytic treatment in addition to the study medication (Table 1).
At day 1, the anxiety level was of low to moderate intensity in most patients (STAI-S, 20-54; STAI-T, 25-51; PRAQ-R2, 13-31; see Figure 2 for exact scores) and was overall stable for all three evaluated scores over the course of the two-week treatment (Figure 2 A, B, C). Patients indicated good sleep quality on the instrument used on all three assessment days (Figure 2).

Anxiety-related outcomes.
Of the 10 patients who completed study participation, nine delivered at term. Among all 16 recruited patients, 5 patients delivered their baby prematurely between week 24 + 0 and 36 + 6 (2 in the Bryophyllum group, 3 in the placebo group; 3 are unknown; Figure 3A). Patients were hospitalized between 2 to 56 days, of whom four out of ten stayed at the hospital for longer than one week (all from the placebo group; Figure 3B).

Obstetric-related outcomes.
No adverse reactions to study medication were reported during the trial.
Discussion
Our initial goal was to characterize clinical outcomes needed to investigate the efficacy of the herbal medication Bryophyllum 50% in the treatment of preterm labor-related anxiety and associated outcomes in an exploratory placebo-controlled study. Due to the paucity of comparable data on the study population investigated, a power calculation could not be performed, and the decision was taken to recruit 30 patients per group. Due to several necessary but time-consuming adjustments of inclusion criteria and the limited storage life of the medication, the study was terminated prematurely, when 16 patients had been recruited and 10 had finished the study by protocol. In the following, the most important learnings and results are discussed as a contribution to facilitating the design and implementation of similar future studies and to avoiding publication bias.
Evaluated anxiety scores and the sleep quality index indicate that (a) patients suffered from (merely) moderate anxiety, and that (b) anxiety levels only changed minimally over the course of the study trial. One reason for this could be that the anxiety questionnaires chosen – STAI-S, STAI-T and PRAQ-R2 – do not capture clinically relevant anxiety in the patient population studied. This could be particularly true in the case of STAI-S and STAI-T, which were indeed not developed for use in pregnant patients. However, they had been used previously to investigate the effect of various complementary therapies during pregnancy, 38 and in a systematic review they were identified as the best available assessment tools for the evaluation of anxiety during pregnancy. 39 PRAQ-R2, on the other hand, is a questionnaire specifically intended for monitoring pregnancy-related anxiety. 35 Importantly, neither STAI-S/T nor PRAQ-R2 had been used previously to investigate anxiety related to preterm labor. A second reason could be that patients with more anxiety (who were in a more vulnerable state) chose not to participate in the study in the first place. In other words, an unwanted selection of relatively relaxed patients might have occurred, which appears possible especially if the rather difficult recruitment process with many ineligible patients is considered. This would explain why the study patients – against our expectations – were not strongly anxious, or at least not anxious to a clinically relevant extent. The patients’ good sleep quality (both at recruitment and during the study) is in line with a lack of strong anxiety and seems to support this latter explanation. Interestingly, in a recent prospective, single-arm study on treatment of anxiety-related symptoms with Bryophyllum 50% chewable tablets that was conducted with patients with severe anxiety symptoms, marked improvements were observed after two weeks. 9
When looking at the obstetric-related outcomes in our (small) patient collective, namely prolongation of gestation and hospitalization days, a different picture emerges. The gestational age at which contractions started was clearly within a pathological range, and its prolongation varied markedly among the patients. In addition, the number of hospitalization days between the initiation of tocolysis and birth differed substantially among the patients, with those in the placebo group experiencing a pronouncedly longer duration of hospitalization compared with patients in the verum group. Moreover, our data show that in the study population of ten or 16 patients, those in the placebo group seemed to have a higher tendency for preterm delivery than those in the verum group. While this suggests better obstetric-related outcomes in the verum group, it should be noted that patients in the placebo group were in a riskier state due to the earlier gestational age and slightly more pronounced cervical shortening, which could result in unfavorable obstetric outcomes. Disentangling the two highly interrelated groups of outcomes – anxiety and obstetric-related outcomes – remains challenging. Adopting a pragmatic and economically relevant method of tracking the duration of inpatient stays shows promise for future research endeavors. Economical aspects are particularly pertinent in the management of preterm labor, as preterm birth rates are higher in low-income countries. 40 Our observations suggesting that B. pinnatum might enable the treatment of (some) women with preterm contractions at home are not only possibly relevant for low-income countries in which the access to hospitals might be limited, but also in high-income countries in general, as a measure to try to attenuate the current increases in health-care costs.
Earlier studies aiming to prove the effectiveness of tocolysis therapy with B. pinnatum faced similar recruitment problems as the present study. Pregnant women represent a difficult patient population for clinical study recruitment in general, because the safety of the unborn child also must be assured. In our case, the main problem was the widespread application of Bryophyllum 50% products in our patient collective. An earlier retrospective analysis from obstetric clinical practice at the Department of Obstetrics at the University Hospital Zürich (patients with a single pregnancy, undergoing tocolysis between 01.01.2009 and 31.12.2014, n = 216) revealed that 62% of participants had taken Bryophyllum 50% as an add-on therapy, 41 ongoing retrospective analysis of tocolysis management points to even higher percentages of women taking Bryophyllum 50% (data not shown). Moreover, in a survey published in 2021, almost 40% of pregnant women in canton Zürich indicated having taken Bryophyllum 50% at some point in their pregnancy for any indication. 42 Our initial intention to reduce the routine use of Bryophyllum 50% during the study duration turned out to be difficult to implement. To be successfully implemented and completed in a reasonable period, a study on the efficacy of B. pinnatum should probably be carried out in a country where B. pinnatum is unknown (or at least not usually used for the indication of preterm labor and available as an over-the-counter medicine). Furthermore, an enrichment in patients with high-risk pregnancies in the study center during the study duration could only be counteracted by allowing the participation of patients with multiple pregnancies and with wider gestational ages. It is nevertheless important to highlight that the adaptations in the study protocol led to successful recruitment. With additional time, through either an extended shelf life or replacement of the study medication, our recruitment goals could have been achieved. Assuming a steady recruitment rate similar to the period between the third amendment and study end (10 recruited patients from January to June 2023 completed the study), this would have corresponded to up to two and a half additional years.
The impact of the data gathered in this study is limited by the small number of patients who completed the study protocol. An additional limitation is the lack of validated measurement tools for assessing anxiety in the context of preterm labor. Nevertheless, the data presented and the methodological learnings discussed may support the conceptualization and design of future studies in this field.
Conclusions
The recruitment difficulties felt at study implementation and their overcoming through two protocol amendments illustrate how critical the stringency of inclusion/exclusion criteria is. Taken together, our results suggest that the study protocol did not permit the adequate capture of clinically significant anxiety related to preterm labor within the study population. Whether this was due to an inadequacy of the questionnaires used for the research question and/or to an unintended selection of patients with only moderate anxiety cannot be stated with certainty. Importantly, the pragmatic approach of counting the days spent in an in-patient facility as a proxy for successful preterm labor treatment appears promising for future studies.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X261417922 - Supplemental material for Bryophyllum pinnatum Treatment of Anxiety Related to Preterm Labor: A Randomized, Double-Blind, Placebo-Controlled Study as Investigator-Initiated Trial IIT
Supplemental material, sj-docx-1-npx-10.1177_1934578X261417922 for Bryophyllum pinnatum Treatment of Anxiety Related to Preterm Labor: A Randomized, Double-Blind, Placebo-Controlled Study as Investigator-Initiated Trial IIT by Leonie Zurfluh, Torsten Hothorn, Christian Haslinger and Ana Paula Simões-Wüst in Natural Product Communications
Footnotes
Abbreviations
Acknowledgements
We are very grateful to all professionals in the obstetrics clinic at the University Hospital Zurich who supported the study team with the recruitment of study participants. At the University Hospital Zurich, we further thank Prof Dr Nicole Ochsenbein-Kölble, Prof Dr Roland Zimmermann (both Department of Obstetrics) and Dr Andrea Burch (Hospital Pharmacy) for important discussions and for sharing their experience. The study-nurse Alexandra Dolder-Behna is gratefully acknowledged for general support as is Dr Heather Murray for language corrections. We thank the members of the Bryophyllum study group for fruitful discussions during the last decades.
Ethical Approval
The study was authorized by the Cantonal ethical committee of Zurich (BASEC-Nr. 2021-00941, June 22nd 2021) as well as by the Swiss agency for therapeutic products (Swissmedic, 2021DR2097, June 18th 2021) and was conducted in line with the principles of the Declaration of Helsinki and according to good clinical practice. The trial was registered at ![]() , on November fourth 2021, having received the number NCT05110599.
, on November fourth 2021, having received the number NCT05110599.
Statement of Informed Consent
Written informed consent was obtained from all individual participants included in the study.
Author Contributions
L Zurfluh: Protocol/project development, study coordination, Data collection or management, Data analysis, Manuscript writing
T Hothorn: Protocol/project development, Manuscript editing
CH Haslinger: Data collection or management, Protocol/project development, Manuscript editing
AP Simões-Wüst: project administration, Protocol/project development, Data analysis, Manuscript writing/editing
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Weleda AG provided the study medication free (verum and placebo). Weleda AG and the Johannes Kreyenbühl foundation contributed to the costs.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The study was in part financially supported by Weleda AG, the company that produces Bryophyllum 50% chewable tablets. APSW has received research funding from Weleda AG over the last 5 years. Weleda AG had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results. APSW is also an employee of the hospital “Klinik Arlesheim AG”, where several Bryophyllum preparations are used.
Data Availability Statement
The complete dataset presented in this article is not readily available because data sharing and additional analysis would need an additional ethics permit. The authors would, however, support other authors in getting an ethics permit to re-use the data.
Statement of Human and Animal Rights
All procedures in this clinical study were conducted in accordance with the Cantonal ethical committee of Zurich approved protocols. This article does not contain any studies with animal subjects.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
