Abstract
Background
Anxiety and depressive disorders are increasingly associated with oxidative stress and neurotransmitter imbalance. Limitations of current pharmacotherapies have intensified interest in plant-derived agents with multi-target activity.
Objective
This study investigated the phytochemical composition, antioxidant capacity, and anxiolytic- and antidepressant-like effects of the n-butanol fraction of Caralluma dalzielii (BUF), alongside exploratory in silico analysis of potential molecular interactions.
Methods
Phytochemical profiling was performed using HPLC. Antioxidant activity was evaluated using DPPH radical scavenging, ferric reducing antioxidant power (FRAP), hydrogen peroxide scavenging, total antioxidant capacity (TAC), and total phenolic content (TPC) assays. Behavioural effects of BUF (10, 50, and 100 mg/kg, p.o.) were assessed in naïve mice using the elevated plus maze (EPM), open field test (OFT), tail suspension test (TST), and forced swim test (FST). Post-behavioural biochemical analyses included modulation of oxidative stress markers, triacylglycerol levels, and acetylcholinesterase activity. Molecular docking was conducted against selected neurobiological targets.
Results
BUF exhibited strong antioxidant capacity (DPPH IC50 < 0.2 mg/mL; TPC 537.75 ± 3.06 mg GAE/g extract). In the EPM, 100 mg/kg significantly increased open-arm entries and time spent in open arms (p < 0.0001), while in the OFT, central crossings were significantly elevated (p < 0.0001), indicating anxiolytic-like activity. In behavioural despair paradigms, immobility time was significantly reduced at 100 mg/kg in both the TST (p < 0.001) and FST (p < 0.001). Biochemically, BUF significantly reduced malondialdehyde levels (p < 0.001), increased catalase and superoxide dismutase activities (p < 0.0001), reduced triacylglycerol levels (p < 0.0001), and inhibited acetylcholinesterase activity (p < 0.0001). Docking analyses indicated favourable binding of key flavonoids, particularly naringenin, to serotonergic, monoaminergic, and GABAergic targets.
Conclusion
These findings provide experimental evidence that the BUF exerts significant anxiolytic- and antidepressant-like effects in validated behavioural models, likely mediated through integrated antioxidant, monoaminergic, and cholinergic mechanisms.
Introduction
Anxiety and depressive disorders represent a major global public health challenge, affecting hundreds of millions of individuals and contributing substantially to disability, impaired quality of life, and socioeconomic burden worldwide.1,2 The World Health Organisation estimates that more than 300 million people currently suffer from depression, with anxiety disorders frequently co-occurring and exacerbating disease severity and chronicity. 2 Recent global stressors, including the COVID-19 pandemic, have further intensified this burden, resulting in a marked increase in the prevalence of mood and anxiety disorders across diverse populations. 3 Beyond their clinical consequences, these disorders impose profound economic costs, with cumulative global losses projected to exceed USD 16 trillion between 2011 and 2030.4-6
Pharmacological management of anxiety and depression relies predominantly on benzodiazepines, selective serotonin reuptake inhibitors (SSRIs), and related agents. While these drugs are effective in many patients, their long-term clinical utility is often limited by delayed onset of action, tolerance, dependence, adverse effects, and incomplete therapeutic response. These challenges are particularly pronounced in low- and middle-income countries, where limited access to mental healthcare, high treatment costs, and social stigma contribute to a substantial treatment gap. 7 Consequently, there is growing interest in alternative and complementary therapeutic strategies that are affordable, safer, and capable of modulating multiple neurobiological pathways involved in mood regulation.
Natural products derived from medicinal plants remain a rich source of structurally diverse bioactive compounds with established relevance to central nervous system (CNS) disorders. Accumulating evidence indicates that phytochemicals such as flavonoids, phenolic acids, glycosides, and triterpenoids can influence key neurotransmitter systems implicated in anxiety and depression, including serotonergic, GABAergic, and monoaminergic pathways. In parallel, many of these compounds exert antioxidant and neuroprotective effects, suggesting that modulation of oxidative stress may represent an important mechanism underlying their neuroactive properties.8,9 The integration of phytochemical characterisation with functional and mechanistic evaluation has therefore become central to contemporary natural-product-based neuropsychopharmacological research.
Caralluma dalzielii N.E. Brown, a succulent plant of the family Apocynaceae (formerly Asclepiadaceae), is indigenous to sub-Saharan Africa and holds considerable ethnomedicinal significance. In West African traditional medicine, the plant is used in the management of pain, inflammation, fever, and neuropsychiatric conditions. 10 Phytochemical investigations of species within the Caralluma genus have identified diverse secondary metabolites, including flavonoids, saponins, triterpenes, and phenolic compounds, many of which are associated with antioxidant and neuromodulatory activities.11,12 Despite its traditional use, however, the chemical composition and neurochemical basis of the purported CNS effects of C. dalzielii remain incompletely characterised.
Limited pharmacological evidence suggests that crude aqueous extracts of C. dalzielii may exert anxiolytic effects, although data supporting antidepressant activity are sparse and mechanistic insights are lacking. 11 Fractionation of crude plant extracts represents an important strategy for enriching bioactive constituents and improving resolution of structure–activity relationships. In particular, moderately polar fractions such as n-butanol extracts are known to concentrate phenolic and glycosidic compounds with documented antioxidant and neuroprotective potential. In a preliminary investigation conducted in our laboratory, the n-butanol fraction of C. dalzielii demonstrated superior neurobehavioural activity compared with hexane and ethyl acetate fractions, providing a rationale for its selection for detailed evaluation.
Oxidative stress has emerged as a key contributor to the pathophysiology of anxiety and depression, influencing neuronal integrity, neurotransmitter metabolism, and synaptic plasticity. Elevated lipid peroxidation and impaired antioxidant defenses have been consistently reported in both clinical and experimental models of mood disorders, supporting a link between redox imbalance and behavioural dysfunction. Consequently, compounds capable of restoring oxidative homeostasis may exert anxiolytic- and antidepressant-like effects through complementary redox-modulating and neurotransmitter-related mechanisms. In addition, in silico molecular docking offers a valuable exploratory approach for probing interactions between phytochemicals and CNS-relevant molecular targets, thereby supporting experimental findings and aiding mechanistic interpretation.
In light of these considerations, the present study aimed to investigate the phytochemical profile and evaluate the anxiolytic- and antidepressant-like effects of the n-butanol fraction of Caralluma dalzielii using validated behavioural paradigms in naïve mice. High-performance liquid chromatography (HPLC) was employed for phytochemical characterisation, while antioxidant capacity was assessed using established in vitro assays. Behavioural evaluations in mouse models of anxiety- and depression-like states were conducted alongside analyses of brain oxidative stress markers to elucidate neurochemical correlates of behavioural effects. Finally, molecular docking studies were performed on HPLC-identified compounds against selected neurobiological targets to provide mechanistic support. Collectively, this study seeks to contribute to the neurochemical understanding and scientific validation of C. dalzielii as a source of bioactive natural products with potential relevance in oxidative stress-associated anxiety and depression.
Materials and Methods
Drugs and Chemicals
Diazepam and imipramine hydrochloride, used as reference anxiolytic and antidepressant drugs, respectively, were obtained from Sigma-Aldrich (St. Louis, MO, USA). Analytical-grade organic solvents, including n-hexane, ethyl acetate, and n-butanol, were purchased from Loba Chemie Pvt. Ltd. (Mumbai, India). All other chemicals and reagents used in the study were of analytical grade and were used as received without further purification.
Plant Material Collection, Extraction, and Fractionation
The whole plant of Caralluma dalzielii N.E. Brown was collected in September 2025 from Wamako Local Government Area, Sokoto State, Nigeria. Mr. Musa Magaji authenticated the plant material at the Herbarium of the Department of Pharmacognosy and Ethnomedicine, where a voucher specimen (Pcg/UDUS/Asdy/001) was deposited for future reference. The collected material was air-dried under laboratory conditions for four weeks and subsequently pulverized into a coarse powder.
Approximately 3 kg of the powdered plant material was macerated in 70% (v/v) methanol–distilled water for 72 h with intermittent agitation. The mixture was filtered, and the filtrate was concentrated under reduced pressure using a rotary evaporator at 45 °C. The resulting extract was further dried on a water bath maintained at 50 °C to obtain the methanol crude extract (MCE).
Sequential solvent–solvent partitioning was employed to fractionate the MCE using solvents of increasing polarity. Briefly, the MCE was reconstituted in distilled water and partitioned with n-hexane for 24 h. The n-hexane layer was separated and filtered, after which the remaining aqueous phase was successively partitioned with ethyl acetate for 24 h and then with n-butanol under similar conditions. Each solvent fraction was concentrated to dryness on a water bath at 50 °C to yield the n-hexane fraction (HF), ethyl acetate fraction (ETF), and n-butanol fraction (BUF), respectively. 13 Preliminary comparative screening of the HF, ETF, and BUF was conducted based on in vitro antioxidant activity and exploratory behavioural observations (data not shown). Among these, the BUF demonstrated comparatively higher antioxidant potential and more consistent behavioural effects. Consequently, the BUF was selected for detailed pharmacological investigation in the present study. Its chemical composition was subsequently characterised using high-performance liquid chromatography (HPLC). 14
High-Performance Liquid Chromatography (HPLC) Analysis
HPLC analysis of the BUF was performed using a reverse-phase HPLC system equipped with a quaternary pump, autosampler, column oven, and UV–visible detector. Chromatographic separation was achieved on a C18 column (250 mm × 4.6 mm, 5 µm particle size). The mobile phase consisted of solvent A (0.1% formic acid in water) and solvent B (acetonitrile) under gradient elution conditions. The flow rate was maintained at 1.0 mL/min, the injection volume was 20 µL, and detection was carried out at 280 nm. The column temperature was maintained at 30 °C. Identification of compounds was achieved by comparing retention times of peaks in the sample chromatogram with those of authenticated reference standards analyzed under identical chromatographic conditions. Quantification was performed using external calibration curves constructed from serial dilutions of each reference compound. Concentrations were calculated from peak areas and expressed as µg/mL of extract.
In Vitro Antioxidant Studies
DPPH Radical Scavenging Assay
The free radical scavenging activity of the BUF was evaluated using the 1,1-diphenyl-2-picrylhydrazyl (DPPH) assay, a widely employed method for assessing antioxidant capacity, with minor modifications.
15
Test solutions of BUF were prepared in analytical-grade methanol at concentrations of 0.2, 0.4, 0.6, 0.8, and 1.0 mg/mL. L-ascorbic acid was used as the reference antioxidant and prepared at corresponding concentrations. For the assay, 1.0 mL of each test solution was mixed with 0.5 mL of freshly prepared 0.3 mM DPPH solution in methanol. The reaction mixtures were vortexed briefly and incubated at room temperature for 15 min in the dark to prevent photodegradation. Control solutions consisted of DPPH solution mixed with methanol, while blank samples contained the test solution mixed with methanol without DPPH. L-ascorbic acid served as the positive control under identical experimental conditions. Following incubation, absorbance was measured at 517 nm using a UV–visible spectrophotometer. All measurements were performed in triplicate, and results were expressed as mean values. The percentage DPPH radical scavenging activity was calculated using the following equation:
ABSs = the absorbance of the sample.
The concentration required to inhibit 50% of the DPPH radicals (IC50) was determined from a plot of percentage radical scavenging activity against sample concentration.
Ferric Reducing Antioxidant Power (FRAP) Assay
The FRAP assay was employed to evaluate the reducing capacity of the BUF based on its ability to reduce ferric ions (Fe3+) to the ferrous form (Fe2+), as previously described with minor modifications. 16 Test solutions of BUF were prepared in methanol at concentrations of 0.2, 0.4, 0.6, 0.8, and 1.0 mg/mL. A phosphate buffer solution (0.2 M, pH 6.6) was freshly prepared, while potassium ferricyanide (1% w/v), trichloroacetic acid (10% w/v), and ferric chloride (0.1% w/v) solutions were prepared in deionised water. For the assay, 1.0 mL of each test solution was mixed with 2.5 mL of phosphate buffer and 2.5 mL of potassium ferricyanide solution. The reaction mixtures were incubated at 50 °C for 20 min to facilitate ferric ion reduction. The reaction was terminated by the addition of 2.5 mL of trichloroacetic acid, followed by centrifugation at 3000 rpm for 10 min to obtain a clear supernatant. An aliquot of the supernatant (2.5 mL) was mixed with 2.5 mL of distilled water and 0.5 mL of ferric chloride solution, resulting in the formation of a coloured complex.
Absorbance was measured at 700 nm using a UV–visible spectrophotometer. A control solution containing all reagents except the test sample was prepared and treated under identical conditions. All measurements were performed in triplicate, and results were expressed as mean values.
Ferric reducing activity was expressed as percentage FRAP and calculated relative to the control using the following equation:
ABSs = the absorbance of the sample.
Total Antioxidant Capacity (Phosphomolybdenum Assay)
The TAC of the BUF was determined using the phosphomolybdenum method, which is based on the reduction of Mo(VI) to Mo(V) and the subsequent formation of a green phosphate–molybdenum complex under acidic conditions, with minor modifications. 17 The reagent solution was freshly prepared by combining sulfuric acid (0.6 M), sodium phosphate (28 mM), and ammonium molybdate (4 mM). For the assay, 0.2 mL of appropriately diluted BUF solution was mixed with 1.8 mL of the reagent solution in a capped test tube. The reaction mixtures were incubated in a boiling water bath at 95 °C for 90 min to allow complete reduction of molybdenum. Following incubation, the tubes were allowed to cool to room temperature. Absorbance was measured at 695 nm using a UV–visible spectrophotometer. A blank solution containing all reagents except the test sample, which was replaced with the corresponding solvent, was prepared and treated under identical conditions. All measurements were performed in triplicate. Total antioxidant capacity was quantified using an ascorbic acid calibration curve generated under the same experimental conditions, and results were expressed as percentage activity.
Total Phenolic Content (TPC)
The TPC of the BUF was determined using the Folin–Ciocalteu colorimetric method, which is based on the reduction of the Folin–Ciocalteu reagent by phenolic compounds under alkaline conditions, with minor modifications. 18 Briefly, an aliquot of BUF solution (1 mg/mL) was mixed with distilled water, followed by the addition of Folin–Ciocalteu reagent (2 N). After a brief reaction period, sodium carbonate solution (2% w/v) was added to alkalinize the mixture. The reaction mixtures were incubated at room temperature in the dark for 3 h to allow complete color development. Absorbance was measured at 760 nm using a UV–visible spectrophotometer. A blank containing all reagents except the test sample, which was replaced with the corresponding solvent, was prepared and treated under identical conditions. All determinations were performed in triplicate. TPC was quantified using a gallic acid calibration curve prepared under the same experimental conditions, and results were expressed as milligrams of gallic acid equivalents per gram of extract (mg GAE/g).
Hydrogen Peroxide (H2O2) Scavenging Activity
The H2O2 scavenging activity of the BUF was evaluated based on its ability to scavenge hydrogen peroxide under buffered conditions, as previously described with minor modifications. 19 Test solutions of BUF were prepared and assessed at concentrations corresponding to those of the reference standard.
For the assay, 0.2 mL of the test solution was mixed with 0.3 mL of hydrogen peroxide solution (40 mM) and 1.2 mL of phosphate buffer (40 mM, pH 7.4). The reaction mixtures were vortexed gently and incubated at room temperature. Ascorbic acid (100 mg/dL) was used as the reference standard and treated under identical experimental conditions. A control solution containing hydrogen peroxide and phosphate buffer without the test sample was prepared concurrently.
Following incubation, absorbance was measured at 230 nm using a UV–visible spectrophotometer. All measurements were performed in triplicate, and results were expressed as mean values.
Hydrogen peroxide scavenging activity was expressed as antioxidant capacity (mg/dL) and calculated relative to the reference standard using the following equation:
Experimental Animals
Male and female Swiss albino mice weighing 25-30 g were obtained from the Animal Facility of the Department of Pharmacology and Therapeutics, Ahmadu Bello University, Zaria, Nigeria. The animals were housed under standard laboratory conditions at a temperature of 24–26 °C and relative humidity of 55–60%, with a 12 h light/dark cycle. Mice were allowed free access to standard laboratory feed and clean drinking water throughout the experimental period. All animal handling and experimental procedures were conducted in accordance with the National Institutes of Health (NIH) guidelines for the care and use of laboratory animals. The reporting of this study conforms to ARRIVE 2.0 guidelines for reporting animal research. 20 Ethical approval for the study was obtained from the Animal Ethics Committee of Usmanu Danfodiyo University, Sokoto, Nigeria (Approval No.: NHREC/UDU-HREC/25/06/23-PGP-13).
Acute Toxicity Testing
Acute oral toxicity of the BUF was evaluated in female Swiss albino mice in accordance with the Organisation for Economic Co-operation and Development (OECD) guideline 425 (Up-and-Down Procedure). A total of five animals were used for the toxicity assessment. The study was initiated by administering BUF dissolved in distilled water at a starting dose of 175 mg/kg body weight to the first animal via oral gavage. Following a 48 h observation period without mortality, a second animal received a dose of 500 mg/kg body weight. Subsequently, the remaining three animals were dosed sequentially with 2000 mg/kg body weight, each administered only after confirmation of survival of the preceding animal. All treated animals were observed individually for signs of toxicity, behavioural changes, and mortality during the first 24 h after administration, and were monitored daily thereafter for a total period of 14 days.
Experimental Procedure
All drugs and the BUF were freshly reconstituted in distilled water before oral administration. Where necessary, gentle vortexing was performed to ensure uniform suspension. A total of thirty (30) mice were randomly allocated into five experimental groups, with six animals per group (n = 6). Dose selection (10, 50, and 100 mg/kg) was based on the outcome of the acute toxicity study (LD50 > 2000 mg/kg) and guided by standard pharmacological practice of selecting doses representing approximately 1/200th to 1/20th of the maximum tolerated dose to evaluate graded biological responses.
The experimental groups were treated as follows: • • • • •
All treatments were administered orally once daily for seven consecutive days. Following the final administration, animals were subjected to behavioural assessments appropriate to each experimental model, and their responses were carefully observed and recorded. Separate cohorts of animals were used for anxiolytic and antidepressant behavioural assessments to prevent behavioural interference and carryover effects between tests.
Behavioural Tests
Elevated Plus Maze (EPM)
The anxiolytic-like activity of the BUF was evaluated using the EPM paradigm, following a standard validated procedure with minor modifications. 22 Diazepam (1 mg/kg) was used as the reference anxiolytic drug. Animals in the treatment groups received BUF orally at doses of 10, 50, or 100 mg/kg, while the normal control group received distilled water. Behavioural assessments were conducted once, 30 min after the final administration on day 7. Thirty minutes after drug administration, each mouse was individually placed at the centre of the maze facing an open arm and allowed to explore freely for 5 min. Behavioural testing for all groups was conducted under identical experimental conditions. After each trial, the maze was thoroughly cleaned with 70% ethanol to eliminate olfactory cues. The parameters recorded included the number of entries into the open arms (NEOA), the number of entries into the closed arms (NECA), time spent in the open arms (TSOA), and time spent in the closed arms (TSCA).
Open Field Test (OFT)
The OFT was employed to assess locomotor activity and anxiety-related behaviour in mice, following a previously established protocol with minor modifications. 23 Diazepam (1 mg/kg) was used as the reference anxiolytic drug. Animals in the treatment groups received the BUF orally at doses of 10, 50, or 100 mg/kg, while the normal control group received distilled water. Thirty minutes after drug administration, each mouse was individually placed at the centre of the open field arena and allowed to explore freely for 5 min. Behavioural assessments were conducted under identical experimental conditions. The open field apparatus was thoroughly cleaned with 70% ethanol between trials to eliminate olfactory cues. The parameters recorded included the number of peripheral crossings (NPC) and the number of central crossings (NCC).
Tail Suspension Test (TST)
The antidepressant-like activity of the BUF was evaluated using the TST, performed according to a previously described and validated protocol with minor modifications. 24 The TST was employed as a validated behavioural despair paradigm for screening antidepressant-like activity in naïve rodents and does not require prior induction of depressive pathology. Imipramine (10 mg/kg) was used as the reference antidepressant drug. On the day of the experiment, animals were transported to the behavioural testing room in their home cages and allowed to acclimatise to the environment for 2 h before testing. Animals in the treatment groups received BUF orally at doses of 10, 50, or 100 mg/kg, while the control group received distilled water. Each mouse was individually suspended by the tail using adhesive tape placed approximately 1 cm from the tip of the tail, with the suspension point positioned 50 cm above the floor. Care was taken to ensure that animals were acoustically and visually isolated from one another during testing to prevent external interference. The total duration of immobility was recorded manually during a 6 min test period. Immobility was defined as the absence of initiated movements, except those required for respiration.
Forced Swim Test (FST)
The FST was employed to evaluate antidepressant-like activity in mice using a well-established protocol with minor modifications. 25 The FST was similarly used as a standard screening model for antidepressant-like activity in naïve animals. Imipramine (10 mg/kg) served as the reference antidepressant drug. Before experimentation, mice were transported from the animal facility to the behavioural testing room in their home cages and allowed to acclimatise for 2 h. Animals in the treatment groups received the BUF orally at doses of 10, 50, or 100 mg/kg, while the control group received distilled water.
For the pre-test session, each mouse was individually placed in a cylindrical glass chamber (25 cm height × 15 cm diameter) containing water to a depth of 15 cm, maintained at room temperature, and allowed to swim for 5 min. Animals were then removed, gently dried, and returned to their home cages. Twenty-four hours later, the test session was conducted. Each mouse was reintroduced into the same apparatus and, following an initial 1 min acclimatisation period, behavioural activity was recorded for 5 min. The duration of immobility was recorded as the primary outcome measure. Immobility was defined as the absence of active escape-directed behaviours, with only minimal movements required to keep the head above water. In addition, time spent swimming during the test session was also recorded.
Sample Collection for In Vivo Biochemical Assessment
Following completion of behavioural testing, animals were humanely euthanised under deep anaesthesia induced by intraperitoneal administration of ketamine (80 mg/kg) and xylazine (10 mg/kg). The brains were immediately excised, rinsed in ice-cold normal saline to remove residual blood, and briefly immersed in cold saline for approximately 10 min to facilitate partial tissue solidification. Each brain was then transferred to a graduated vessel and homogenised in phosphate-buffered saline (PBS, pH 7.4) to obtain a 10% (w/v) tissue homogenate using a chilled homogeniser. The homogenates were centrifuged at 10,000 rpm for 15 min at 4 °C, and the resulting supernatants were carefully collected. The supernatants were kept under cold conditions and used for subsequent biochemical analyses.
Assessment of Oxidative Stress Markers
Oxidative stress markers in brain tissue were evaluated using standard biochemical methods. Lipid peroxidation was assessed by quantifying malondialdehyde (MDA) levels using the thiobarbituric acid reactive substances (TBARS) assay, with minor modifications. 26 Briefly, 500 μL of the brain tissue supernatant was transferred into labelled test tubes, while control tubes contained 500 μL of 50 mM Tris–HCl buffer (pH 7.4). Each tube was treated with 250 μL of 20% trichloroacetic acid (TCA) and 500 μL of 0.67% thiobarbituric acid (TBA), sealed, and heated in a water bath at 90 °C for 10 min to allow formation of the MDA–TBA complex. The mixtures were cooled to room temperature and centrifuged at 3000 rpm for 15 min. Absorbance of the resulting supernatant was measured at 530 nm using a UV–visible spectrophotometer, with the reagent blank serving as baseline. MDA concentration was calculated using the Beer–Lambert law with an extinction coefficient of 1.56 × 105 M-1 cm-1 and expressed as micromoles of MDA per gram of tissue.
Catalase (CAT) activity was determined by monitoring the decomposition of hydrogen peroxide, as previously described. 27 An aliquot of 0.1 mL of the brain supernatant was mixed with 1.9 mL of 50 mM phosphate buffer (pH 7.0), and the reaction was initiated by the addition of 1.0 mL of freshly prepared hydrogen peroxide solution (30 mM). The decrease in absorbance due to hydrogen peroxide decomposition was monitored at 240 nm using a UV–visible spectrophotometer. Catalase activity was calculated from the rate of absorbance decline and expressed as units per milligram of protein.
Reduced glutathione (GSH) levels were quantified based on its reaction with 5,5′-dithiobis-(2-nitrobenzoic acid) (DTNB), as described previously. 28 In this assay, 20 μL of the brain homogenate was mixed with 3 mL of DTNB reagent and incubated at room temperature for 1 h to allow complete colour development. The formation of the yellow chromophore was measured spectrophotometrically at 412 nm. GSH concentration was calculated using the Beer–Lambert law with a molar extinction coefficient of 13,600 M-1 cm-1 and expressed as micromoles per gram of tissue.
Superoxide dismutase (SOD) activity was determined using a commercial RANDOX assay kit (Randox Laboratories, UK) in accordance with the manufacturer’s instructions. The method is based on the generation of superoxide radicals via the reaction between xanthine and xanthine oxidase, which subsequently react with iodonitrotetrazolium chloride to form a red formazan dye. SOD activity inhibits formazan formation by catalysing the dismutation of superoxide radicals into less reactive species. The degree of inhibition was measured by monitoring the increase in absorbance at 505 nm using a UV–visible spectrophotometer, and enzyme activity was expressed as units per milligram of protein.
Triacylglycerol (TAG) levels in brain tissue homogenates were determined using a manual glycerol-based enzymatic method. 29 Briefly, TAGs present in the brain supernatant were hydrolysed enzymatically to release glycerol. The liberated glycerol was subsequently oxidised through a coupled enzymatic reaction, resulting in the formation of a coloured chromogen. An aliquot of the brain supernatant was incubated with the reaction reagent under standard assay conditions, and colour development was measured spectrophotometrically at the appropriate wavelength using a UV–visible spectrophotometer. TAG concentrations were calculated from a glycerol standard calibration curve prepared under identical experimental conditions and were expressed as milligrams per decilitre (mg/dL) of tissue homogenate.
Acetylcholinesterase (AChE) Activity Determination
AChE activity in mouse brain homogenates was determined using a colourimetric method based on the principle originally described by Ellman and colleagues, with minor modifications. 30 Briefly, 200 μL of brain homogenate was added to 2.2 mL of 50 mM Tris–HCl buffer, followed by the addition of 200 μL of 15 mM 5,5′-dithiobis-(2-nitrobenzoic acid) (DTNB). The mixture was incubated at room temperature for 15 min to allow baseline colour development. The enzymatic reaction was initiated by the addition of 400 μL of acetylthiocholine iodide (3.75 mM), and the change in absorbance was recorded immediately at 412 nm using a UV–visible spectrophotometer. Acetylcholinesterase activity was calculated from the rate of increase in absorbance per minute and expressed as enzyme activity per milligram of protein in the brain homogenate.
Protein Estimation
Protein concentration in brain homogenate supernatants was determined using the Bradford colorimetric method, which is based on the binding of Coomassie Brilliant Blue G-250 dye to proteins. Briefly, an aliquot of the brain homogenate supernatant was mixed with Bradford reagent and incubated at room temperature to allow colour development. Absorbance of the resulting blue complex was measured at 595 nm using a UV–visible spectrophotometer. A standard calibration curve was generated using bovine serum albumin (BSA), and protein concentrations of the samples were determined from the standard curve. The estimated protein values were used to normalise antioxidant enzyme activities and acetylcholinesterase activity, which were expressed per unit protein.
In Silico Molecular Docking
Phytochemical constituents identified from the BUF were evaluated for their potential interactions with neurobiological targets implicated in anxiety and depression using in silico molecular docking. Three-dimensional crystal and cryo-electron microscopy structures of the serotonin 2A receptor (5-HT2AR; PDB ID: 6A93), serotonin transporter (SERT; PDB ID: 6VRH), monoamine oxidase A (MAO-A; PDB ID: 2Z5X), and γ-aminobutyric acid type A receptor β3 homopentamer (GABA(A)R-beta3 homopentamer; PDB ID: 4COF) were retrieved from the Protein Data Bank. Protein structures were prepared using the Protein Preparation Wizard in the Schrödinger Suite (version 2021-2), including bond order assignment, hydrogen addition, optimisation of protonation states at physiological pH using PROPKA, removal of distant crystallographic water molecules, and energy minimisation with the OPLS2005 force field. 31
Ligands identified by HPLC, together with reference drugs diazepam and imipramine, were prepared using the LigPrep module of Schrödinger. Ligand preparation involved the generation of appropriate ionisation and tautomeric states at physiological pH (7.2 ± 0.2) using Epik, retention of defined stereochemistry, and energy minimisation using the OPLS2005 force field. 32 Receptor grids were generated around the native ligand binding sites using Maestro (version 12.8), with grid parameters optimised to allow appropriate ligand flexibility during docking. 33
Molecular docking simulations were performed using the Glide docking module in standard precision (SP) mode with flexible ligand sampling. Docked poses were ranked based on Glide docking scores, and the most energetically favourable conformations were selected for further analysis. Docking experiments were repeated three times to ensure reproducibility. Ligand–receptor interactions were visualised and analysed using Discovery Studio Visualizer (version 3.0). 34
Data Analysis
All data were expressed as mean ± standard error of the mean (SEM). Statistical analysis was performed using one-way analysis of variance (ANOVA), followed by Dunnett’s multiple comparison post hoc test to assess differences among groups. All analyses were conducted using GraphPad Prism version 8 for Windows (GraphPad Software Inc., San Diego, CA, USA). A probability value of p < 0.05 was considered statistically significant.
Results
HPLC Profiling of the BUF
List of Compounds in the n-butanol Fraction of C. dalzielii Identified by HPLC
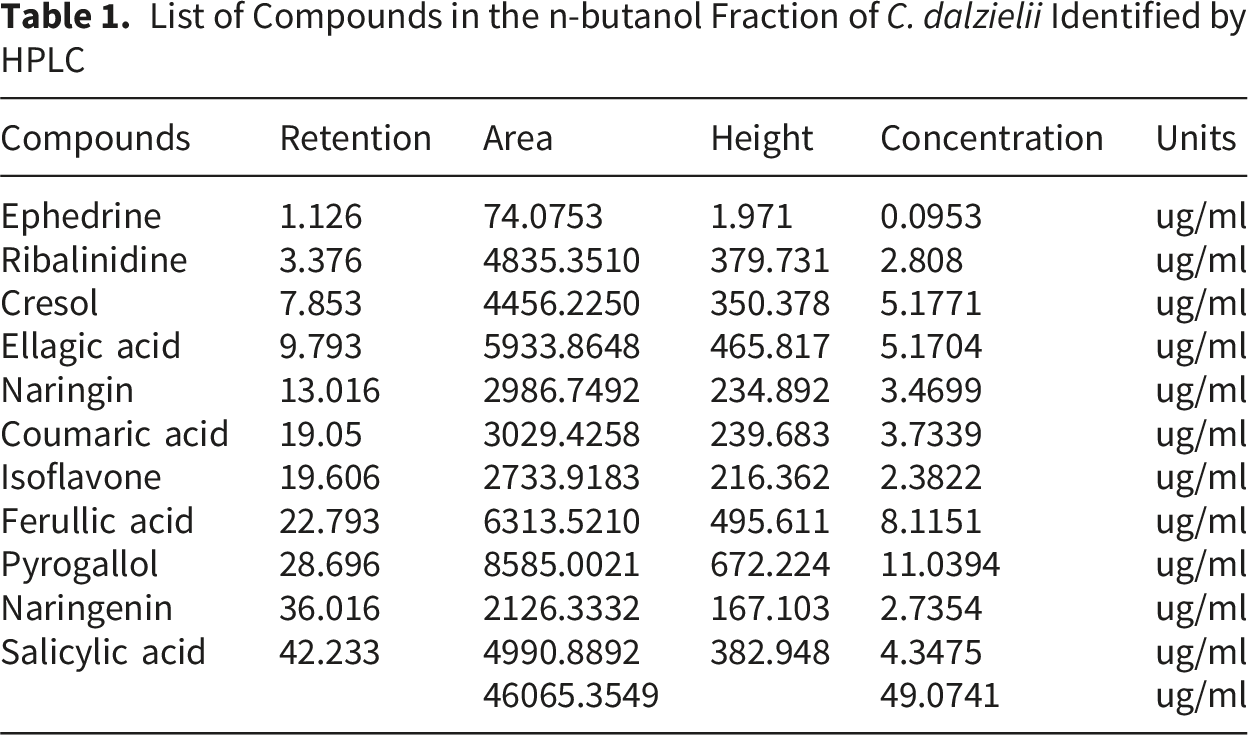
Antioxidant Activity of the BUF
The BUF and vitamin C exhibited marked, concentration-dependent DPPH radical scavenging activity across the tested concentration range (Figure 1A). At the lowest concentration evaluated (0.2 mg/mL), both BUF and vitamin C produced greater than 50% radical scavenging activity. Due to the high level of inhibition observed at this concentration, the IC50 values for both BUF and vitamin C were estimated to be less than 0.2 mg/mL (Table 2). The FRAP of BUF increased progressively with increasing concentration, demonstrating a clear concentration-dependent reducing capacity (Figure 1B). Based on the concentration–response relationship, the EC50 value, defined as the concentration required to achieve 50% of maximal FRAP activity, was approximately 0.53 mg/mL (Table 2). The TAC, assessed using the phosphomolybdenum assay, revealed an overall antioxidant activity of 95.43 ± 0.76%. The TPC of BUF was determined to be 537.75 ± 3.06 mg gallic acid equivalents per gram of extract. Hydrogen peroxide scavenging activity was recorded as 77.47 ± 1.84% scavenging capacity (Table 2). Antioxidant activity of the n-butanol fraction of Caralluma dalzielii (A) DPPH radical scavenging activity of BUF compared with ascorbic acid (standard) across concentrations of 0.2–1.0 mg/mL. (B) Ferric reducing antioxidant power (FRAP) of BUF across the same concentration range, expressed as absorbance at 700 nm. Data are presented as mean ± SEM (n = 3). BUF represents the n-butanol fraction of Caralluma dalzielii Antioxidant Activity of the n-Butanol Fraction of C. dalzielii

Acute Toxicity Testing
No mortality or observable signs of acute toxicity were recorded in mice treated with the BUF at doses of 175, 500, and 2000 mg/kg during the 14-day observation period. Animals remained active and exhibited no abnormal behavioural changes throughout the study duration. Based on these findings, the LD50 of BUF was considered to be greater than 2000 mg/kg under the conditions of this study.
Effects of BUF on Anxiety- and Depression-Related Behaviour
EPM and OFT
The BUF produced significant, dose-dependent anxiolytic-like effects in the EPM. At 50 mg/kg, BUF significantly increased the NEOA (p < 0.05) and significantly reduced NECA (p < 0.05) compared with the control group (Figure 2A). These effects were markedly enhanced at 100 mg/kg, where NEOA increased significantly (p < 0.0001), and NECA decreased significantly (p < 0.001), indicating reduced anxiety-related avoidance behaviour. Similarly, TSOA increased in a dose-dependent manner, with the 100 mg/kg dose producing a highly significant increase relative to control (p < 0.0001). Correspondingly, TSCA was significantly reduced at 50 mg/kg (p < 0.05) and more prominently at 100 mg/kg (p < 0.0001) (Figure 2B). The magnitude of these behavioural changes at 100 mg/kg approached that observed with diazepam (1 mg/kg), supporting a statistically robust anxiolytic-like effect of the fraction. It also produced dose-dependent increases in the NCC, with significant elevations observed at 50 mg/kg (p < 0.05) and a highly significant increase at 100 mg/kg (p < 0.0001) compared with the control group. Increased central zone exploration is indicative of reduced anxiety-related behavioural inhibition (Figure 2C). Similarly, the NPC, a measure of general locomotor activity, was significantly increased at 50 mg/kg (p < 0.05) and 100 mg/kg (p < 0.001) relative to control. The concurrent increase in both NCC and NPC suggests that the anxiolytic-like effect was not attributable to sedation or locomotor suppression. At 100 mg/kg, the behavioural pattern approached that observed with diazepam (1 mg/kg), further supporting a statistically significant anxiolytic-like profile of the fraction. Effects of the n-butanol fraction (BUF) of Caralluma dalzielii on anxiety-like behaviour in mice.
TST and FST
Treatment with the BUF produced significant antidepressant-like effects in both the TST and FST. In the TST, immobility time was significantly reduced at 10 mg/kg (p < 0.05), 50 mg/kg (p < 0.05), and more markedly at 100 mg/kg (p < 0.001) compared with the control group, demonstrating a clear dose-dependent behavioural response when treated with BUF (Figure 3). Similarly, in the FST, BUF significantly reduced immobility time at 50 mg/kg (p < 0.05) and 100 mg/kg (p < 0.001) relative to control. The reduction observed at 100 mg/kg approached that produced by the reference antidepressant imipramine (10 mg/kg), indicating a statistically robust antidepressant-like effect. Decreased immobility in both paradigms reflects enhanced active coping behaviour and reduced behavioural despair, supporting the antidepressant potential of the fraction. Effects of the n-butanol fraction of Caralluma dalzielii on depressive-like behaviour in mice.
Effect of BUF on Oxidative Stress Markers
Effect of BUF on Oxidative Stress Markers

Values = mean ±S.E.M. (n=6); *p<0.05; **p<0.001; ***p<0.0001 compared to normal control. One-way ANOVA followed by Dunnett test. BUF represents the n-butanol fraction of Caralluma dalzielii; D/W represents the distilled water control.
Effect of BUF on AChE Activity
Effect of BUF on Acetylcholinesterase (AChE) Activity

Values = mean ±S.E.M. (n=6); *p<0.05; **p<0.001; ***p<0.0001 compared to normal control. One-way ANOVA followed by Dunnett test. BUF represents the n-butanol fraction of Caralluma dalzielii.
Molecular Docking Analysis
Docking Scores of Compounds From the Butanol Fraction of Caralluma dalzielii
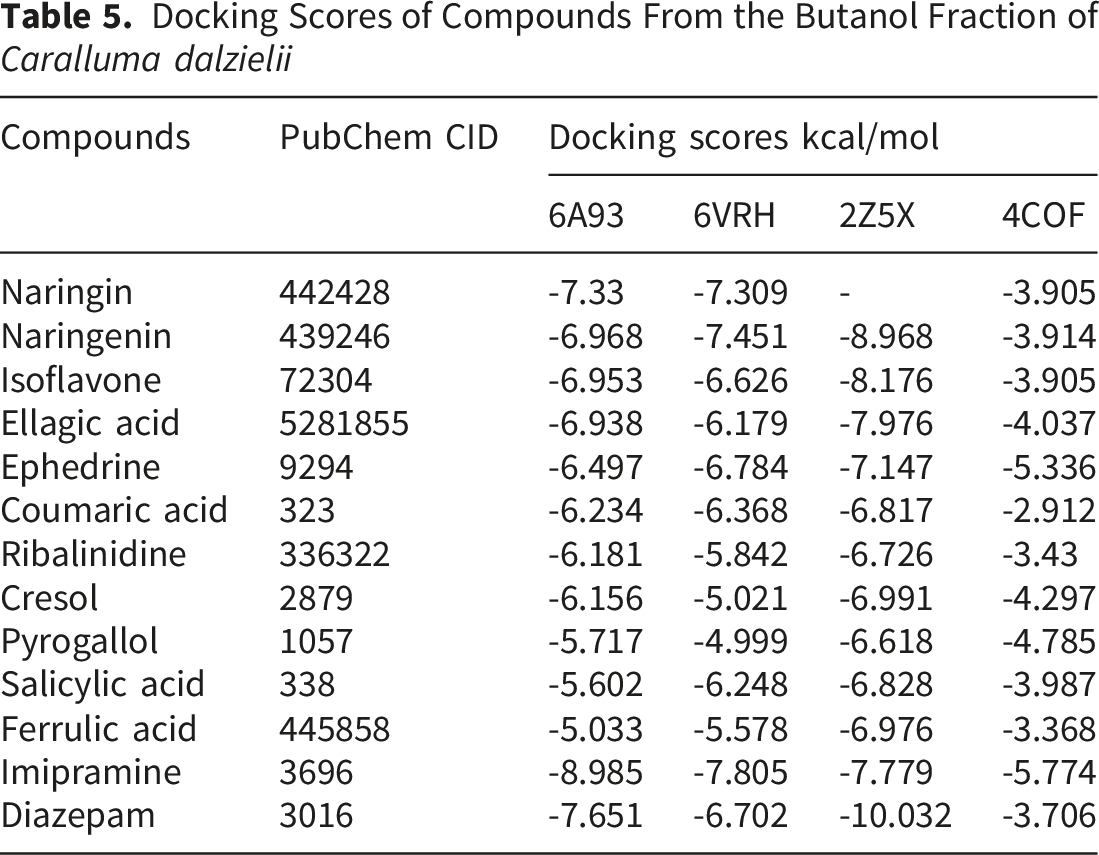
The two-dimensional and three-dimensional binding conformations of the three highest-ranking phytochemicals for each protein target are presented in Figure 4A-D, while detailed ligand–residue interactions are summarized in Table 6. The chemical structures of all docked phytochemicals, as well as the reference drugs, are shown in Figure 5. Among the identified constituents, naringenin consistently ranked among the top three ligands across most protein targets, except for the GABA(A)R-beta3 homopentamer. Molecular interaction of amino-acid residues of top three compounds on (A) 5-HT2AR (PDB ID: 6A93) i. Naringin ii. Naringenin iii. Isoflavone, (B) Serotonin transporter (PDB ID: 6VRH) i. Naringenin ii. Naringin iii. Ephedrine (C) Monoamine oxidase A (PDB ID: 2Z5X) i. Naringenin ii. Isoflavone iii. Ellagic acid, and (D) Gamma-aminobutyric acid receptor (PDB ID: 4COF) i. Ephedrine ii. Pyrogallol iii. Cresol Interaction of the Top Three Compounds of Butanol Fractions of Caralluma dalzielii Against Target Proteins 2D structures of the three highest-ranking compounds from BUF and the standard drugs (A): Naringin; (B): Naringenin; (C): Isoflavone; (D): Ephedrine; (E): Ellagic acid; (F): Pyrogallol; (G): Cresol; (H): Diazepam; (I): Imipramine
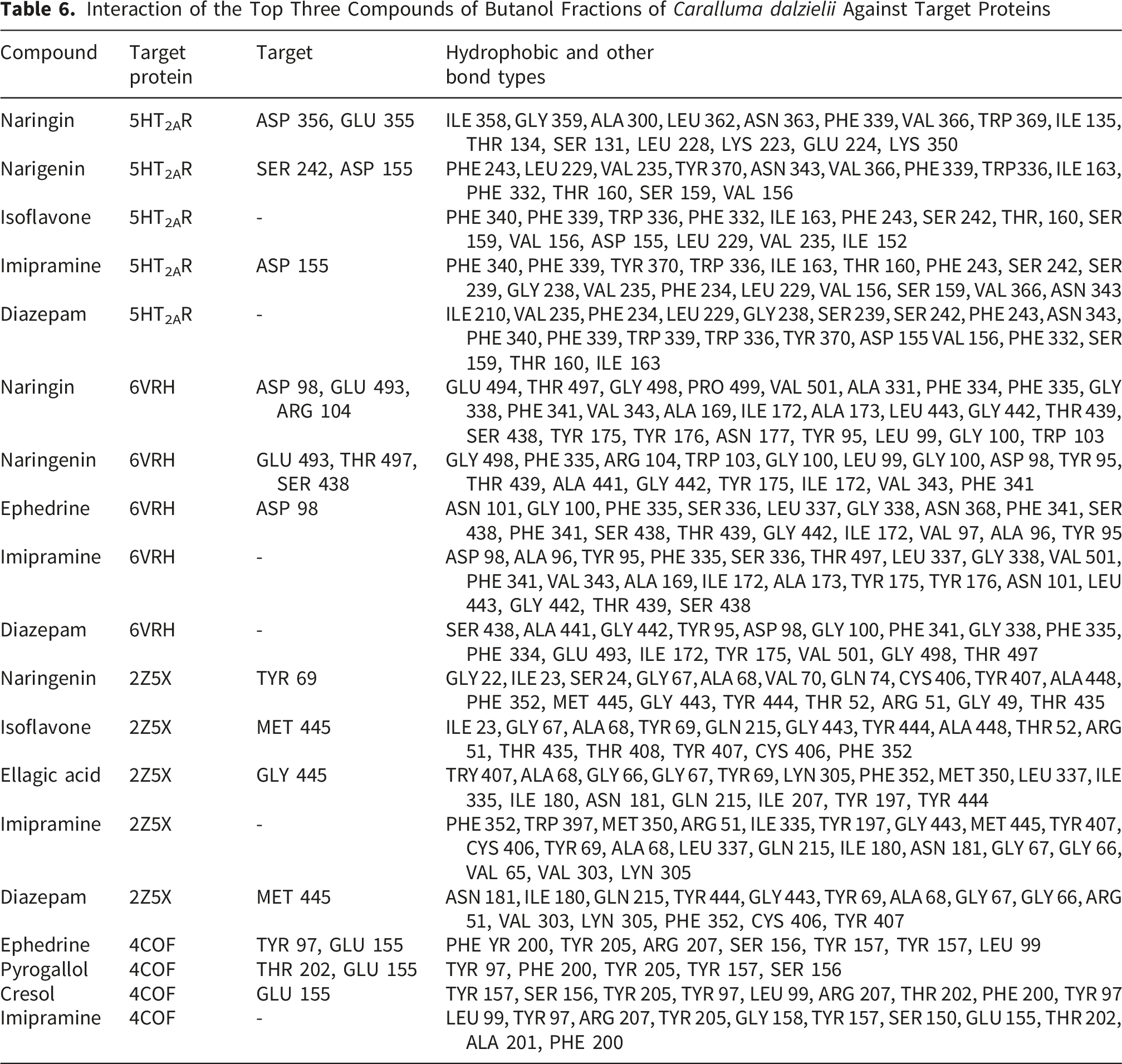

Against the serotonin 2A receptor (PDB ID: 6A93), naringenin formed hydrogen bond interactions with SER242 and exhibited π–π stacking with PHE340 and ASP155. Docking to the serotonin transporter (PDB ID: 6VRH) revealed hydrogen-bond interactions with SER438, THR497, as well as π–π stacking with TYR175 and GLU493. In the MAO-A complex (PDB ID: 2Z5X), naringenin formed displayed π–π stacking interactions with TYR444 and TYR69.
The reference drug diazepam did not form hydrogen bond interactions with the serotonin 2A receptor but exhibited π–π stacking with TRP336. No hydrogen bonding or π–π stacking interactions were observed between diazepam and the remaining protein targets. Imipramine showed π–π stacking with PHE352 in the MAO-A complex and additional π–π and π–cation interactions with residues including PHE200, TYR157, TYR205, and GLU155 across other targets, without hydrogen bond formation. The molecular structures of diazepam and imipramine are shown in Figure 5.
Discussion
The antioxidant profile demonstrated by the BUF provides a strong biochemical foundation for its observed neuropharmacological effects. Oxidative stress is increasingly recognised as a central contributor to the development and progression of anxiety- and depression-like states, where excessive generation of ROS disrupts neuronal membranes, neurotransmitter metabolism, and intracellular signalling pathways. 35 Within this context, the antioxidant capacity of BUF offers a plausible mechanistic framework for understanding the behavioural and biochemical effects observed in vivo. The pronounced free-radical scavenging and reducing activities of BUF indicate the presence of phytochemicals capable of donating electrons or hydrogen atoms to neutralise reactive intermediates before initiation of oxidative chain reactions. Such redox activity is characteristic of polyphenolic compounds, which stabilise free radicals through resonance and structural rearrangement. 35 In agreement with this, previous studies on Caralluma species and related medicinal plants have linked antioxidant efficacy to high phenolic and flavonoid content, reinforcing the central role of these compounds in redox modulation. 36 Beyond direct radical scavenging, the ability of BUF to reduce ferric ions and detoxify hydrogen peroxide highlights its capacity to interfere with multiple oxidative pathways. Transition metals such as iron catalyse the formation of highly reactive hydroxyl radicals via Fenton-type reactions, while hydrogen peroxide readily diffuses across cellular membranes and contributes to lipid peroxidation and mitochondrial dysfunction when inadequately cleared. The ability of BUF to act at these distinct oxidative nodes suggests a broad-spectrum antioxidant profile rather than a single-mechanism effect. The high phenolic burden of BUF further supports this interpretation, as phenolic compounds are known not only for their direct antioxidant actions but also for their ability to modulate endogenous defence systems, including antioxidant enzymes and redox-sensitive transcription factors. 37 Such dual functionality may explain why plant-derived fractions often exert more sustained biological effects than isolated antioxidant molecules. Importantly, oxidative stress in neuropsychiatric disorders extends beyond oxidative damage alone and is closely intertwined with neuroinflammation, neurotransmitter dysregulation, and impaired synaptic plasticity. By attenuating oxidative pressure, BUF may indirectly preserve monoaminergic neurotransmission, reduce stress-induced neuronal dysfunction, and stabilise behavioural responses. Similar mechanistic links between antioxidant capacity and anxiolytic or antidepressant outcomes have been reported for other flavonoid- and phenolic-rich plant extracts. 38
Anxiolytic and antidepressant agents exert their effects primarily through modulation of central neurotransmitter systems that regulate emotional and stress-related behaviours. Benzodiazepines enhance GABA-mediated inhibitory neurotransmission at GABAA receptors, leading to neuronal hyperpolarisation and reduced excitability, 39 while serotonergic dysfunction underlies many anxiety and depressive disorders and forms the basis for monoaminergic therapies. The use of multiple behavioural models in the present study, therefore, enabled a comprehensive evaluation of the anxiolytic- and antidepressant-like properties of BUF within established neurochemical frameworks.
The EPM exploits rodents’ innate aversion to open and elevated spaces, with increased open-arm exploration reflecting reduced anxiety-like behaviour.40,41 The enhancement of open-arm exploration following BUF administration suggests modulation of neural circuits governing anxiety responses. This pattern aligns with previous reports on Caralluma species, supporting the view that phytochemicals within this genus possess central nervous system activity relevant to emotional regulation. 42 Findings from the OFT further corroborated the anxiolytic-like profile of BUF. Increased exploration of the central zone, alongside preserved locomotor activity, is indicative of reduced anxiety rather than sedation. 43 The concurrent increase in central and peripheral crossings observed in this study suggests attenuation of anxiety-driven behavioural inhibition, consistent with earlier neuropharmacological reports on Caralluma dalzielii extracts.11,44
Antidepressant-like activity was evaluated using the TST and FST, both of which model behavioural despair in rodents. The TST and FST are widely used screening models for antidepressant-like activity in naïve rodents and do not require prior induction of depressive pathology. These paradigms assess passive coping behaviour in response to an acute inescapable stressor and are predictive of monoaminergic antidepressant efficacy. Accordingly, the present findings reflect antidepressant-like behavioural effects rather than reversal of experimentally induced depressive states. Reduction in immobility time reflects a shift toward active coping strategies and is a hallmark of antidepressant activity. The significant reduction in immobility observed following BUF treatment, therefore, indicates antidepressant-like effects comparable to those of the reference drug. Given the established role of monoaminergic dysfunction in depression, these behavioural outcomes suggest involvement of serotonergic and noradrenergic pathways. The concurrent anxiolytic- and antidepressant-like effects are notable and reflect the substantial clinical overlap and shared neurobiological substrates of these disorders.
The biochemical alterations observed following BUF administration provide further mechanistic insight into its behavioural effects. Oxidative stress contributes to anxiety and depression by promoting lipid peroxidation, impairing antioxidant enzyme activity, and disrupting neurotransmitter homeostasis. The reduction in MDA levels observed at higher doses of BUF indicates attenuation of lipid peroxidation and preservation of neuronal membrane integrity. Similar reductions in MDA have been reported for phenolic- and flavonoid-rich plant extracts with anxiolytic and antidepressant properties. 45
The enhancement of endogenous antioxidant enzyme activities further reinforces the neuroprotective profile of BUF. SOD and CAT constitute primary enzymatic defences against ROS, acting sequentially to detoxify superoxide radicals and hydrogen peroxide. 46 Their upregulation suggests strengthening of intrinsic antioxidant defences, limiting oxidative injury and supporting neuronal resilience. Such effects have been associated with improved stress tolerance and behavioural outcomes in experimental and clinical studies of mood disorders. 47 The selective reduction in GSH levels at the lowest dose of BUF, with preservation at higher doses, suggests dose-dependent modulation of glutathione dynamics rather than global depletion. Low-dose exposure may transiently increase GSH utilisation during detoxification of reactive intermediates, reflecting adaptive redox engagement. 48 Preservation of GSH at higher doses likely reflects reduced oxidative burden and compensatory regeneration, supported by concurrent increases in SOD and CAT activities. Similar biphasic effects on glutathione dynamics have been reported for plant-derived antioxidants. 49 The observed reduction in TAG levels further indicates an interaction between oxidative stress and lipid metabolism in the brain. Dysregulated lipid homeostasis has been implicated in neuropsychiatric disorders through effects on membrane fluidity, receptor function, and intracellular signalling. By reducing TAG levels, BUF may contribute to stabilisation of neuronal membrane composition and improved synaptic efficiency, thereby indirectly supporting its anxiolytic- and antidepressant-like effects. It is important to note that oxidative stress was not experimentally induced in the present study. Therefore, the observed changes in antioxidant enzymes and lipid peroxidation reflect modulation of basal redox status rather than protection against a defined oxidative insult. While this limits direct inference regarding therapeutic antioxidant efficacy under pathological conditions, the enhancement of endogenous antioxidant defenses suggests redox-regulatory potential that may contribute to the behavioural effects observed.
In addition to redox modulation, AChE activity represents an important biochemical interface between oxidative stress and neurotransmission. Although monoaminergic mechanisms are central to conventional antidepressant therapies, accumulating evidence indicates that cholinergic dysregulation also contributes to mood and stress-related disorders. Altered acetylcholine signalling has been implicated in emotional processing, stress responsiveness, and behavioural adaptation. AChE regulates synaptic acetylcholine availability, and excessive enzyme activity may impair cholinergic signalling relevant to both emotional and cognitive function. The observed dose-dependent reduction in AChE activity following BUF treatment suggests enhanced cholinergic neurotransmission, which may complement monoaminergic and antioxidant mechanisms in mediating the behavioural effects observed in this study. Furthermore, oxidative stress has been reported to upregulate AChE activity; thus, the concurrent reduction in lipid peroxidation and AChE activity observed here may indicate interruption of a redox–cholinergic pathological interplay. This integrated modulation is consistent with reports of plant-derived neuroprotective compounds exerting dual antioxidant and cholinergic regulatory effects.50,51
Chromatographic and computational analyses provided further insight into the molecular basis of these effects. HPLC profiling confirmed the presence of flavonoids (naringin, naringenin, isoflavones) and phenolic acids (ellagic acid, ferulic acid, p-coumaric acid), supporting a multi-target phytochemical profile. Flavonoids are known to influence monoaminergic transmission, inhibit monoamine oxidase, and interact with GABAergic systems, 45 while phenolic acids primarily contribute to antioxidant and neuroprotective actions.
Molecular docking analyses further supported these findings by demonstrating favourable interactions between BUF-derived phytochemicals and key neurobiological targets, including the serotonin 2A receptor, serotonin transporter, monoamine oxidase A, and GABAA receptor β3 subunit. Among the identified compounds, naringenin consistently ranked among the top ligands, forming stable interactions through hydrogen bonding and aromatic interactions such as π–π stacking and π–cation contacts. 52 These interactions support, but do not establish, a molecular basis for the observed behavioural effects. The relevance of naringenin is supported by previous reports documenting its antioxidant, anti-inflammatory, anxiolytic, and antidepressant properties, as well as its ongoing clinical evaluation for metabolic disorders.53,54 Other identified compounds, including ellagic acid and p-coumaric acid, have similarly been reported to exert neuroprotective and mood-modulating effects.55,56 Alkaloids such as ephedrine and ribalinidine also exhibited notable in silico interactions; however, their precise contributions to the observed behavioural effects warrant further experimental investigation.
Study Limitations
Despite the promising findings, several limitations should be acknowledged. First, the study relied on behavioural and biochemical assessments without direct receptor-binding or neurotransmitter quantification assays; therefore, mechanistic interpretations remain inferential. Second, molecular docking provides predictive insights but does not confirm in vivo receptor interactions. Third, although the BUF demonstrated significant bioactivity, the individual contributions of specific phytochemicals were not experimentally isolated. Additionally, chronic models of anxiety and depression were not employed, limiting extrapolation to long-term therapeutic contexts. Future studies should incorporate targeted neurochemical analyses, receptor-specific assays, and chronic stress paradigms to further elucidate the precise mechanisms underlying the observed effects.
Conclusion
This study demonstrates that the n-butanol fraction of Caralluma dalzielii exhibits significant anxiolytic- and antidepressant-like effects in mice, supported by robust antioxidant, neurochemical, and behavioural evidence. The fraction showed strong in vitro antioxidant activity and produced dose-dependent reductions in anxiety- and depression-related behaviours in established animal models. These behavioural effects were accompanied by attenuation of brain oxidative stress, as evidenced by reduced lipid peroxidation, enhanced antioxidant enzyme activities, modulation of glutathione dynamics, and reduced triacylglycerol levels. In addition, inhibition of acetylcholinesterase activity suggests enhanced cholinergic neurotransmission, providing further mechanistic support for the observed neurobehavioural effects. Phytochemical profiling revealed a multi-target composition dominated by flavonoids and phenolic acids, while molecular docking analyses supported favourable interactions between key constituents, particularly naringenin, and neurobiological targets implicated in anxiety and depression. Collectively, these findings provide experimental support for the traditional use of C. dalzielii and highlight the n-butanol fraction as a promising source of bioactive compounds with potential relevance in oxidative stress–associated neuropsychiatric disorders. Further studies are warranted to isolate active constituents and elucidate precise molecular mechanisms underlying these effects.
Supplemental Material
Supplemental Material - Anxiolytic and Antidepressant Activities of the n-Butanol Fraction of Caralluma dalzielii: Behavioural and Neurochemical Evidence
Supplemental Material for Anxiolytic and Antidepressant Activities of the n-Butanol Fraction of Caralluma dalzielii: Behavioural and Neurochemical Evidence by Guoguang Song, Qin Zhou, Musa Abbas, Nuhu Baba, Yusuf Ibrahim, Chinenye Ugwah-Oguejiofor in Natural Product Communications
Footnotes
Acknowledgement
This research was funded by Science and Technology Project of Xuzhou Health Commission (Project No. XWKYHT20220131), and Brain Science and Brain-like Intelligence Technology-National Science and Technology Major Project (No. 2022ZD 0209100). The authors thank Abdulmumin Ibrahim and Idris Haruna for assisting with the neurobehavioral studies.
Ethical Considerations
All animal handling and experimental procedures were conducted in accordance with the National Institutes of Health (NIH) guidelines for the care and use of laboratory animals. They complied with the ARRIVE guidelines for reporting animal research. Ethical approval for the study was obtained from the Animal Ethics Committee of Usmanu Danfodiyo University, Sokoto, Nigeria (Approval No.: NHREC/UDU-HREC/25/06/23-PGP-13).
Authors Contributions
All authors contributed and gave approval for the final manuscript.
Funding
This research was funded by Science and Technology Project of Xuzhou Health Commission (Project No. XWKYHT20220131), and Brain Science and Brain-like Intelligence Technology-National Science and Technology Major Project (No. 2022ZD 0209100).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated are included in the article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



