Abstract
Objectives
This study aimed to explore the antioxidant, antibacterial, and anti-hemolytic activities of Atriplex halimus L., and to highlight how optimizing extraction conditions can enhance the recovery of its bioactive compounds, providing new insights into its pharmaceutical potential.
Methods
Leaves collected in El-Oued, Algeria (May 2023) were extracted with ethanol, methanol, and acetone under varying acidity (0-2 N HCl), time (30-240 min), and temperature (25-70°C). Total phenolic (TPC) and flavonoid (TFC) contents were quantified, and antioxidant activities assessed using DPPH, FRAP, ABTS, β-carotene bleaching, hydroxyl radical, superoxide, and hydrogen peroxide assays. Anti-hemolytic and antibacterial activities were also evaluated.
Results
Acetone extract had the highest TPC (393.7 mg GAE/100 g DW) and strongest antioxidant activity (DPPH IC50 = 9.39 µg/ml; FRAP = 333.7 mg AAE/100 g), while ethanol yielded the highest TFC (47.9 mg QE/100 g DW). Optimal conditions (acetone + 1N HCl, 210 min, 37°C) enhanced bioactivity. Extracts showed stronger antibacterial effects against Gram-positive strains and provided dose-dependent hemolysis inhibition.
Conclusion
A. halimus exhibits potent antioxidant, antibacterial, and anti-hemolytic activities. The optimal extraction conditions, acetone solvent acidified with 1N HCl, 210 min of extraction time, and a temperature of 37 °C, significantly enhanced phenolic and flavonoid recovery as well as antioxidant activity. These findings support its potential as a valuable natural source for pharmaceutical and nutraceutical applications.
Keywords
Introduction
Plant therapy has arisen as a fresh alternative strategy in the treatment of several diseases, leveraging the considerable effectiveness and low toxicity associated with therapies derived from plants. Plants provide a diverse array of chemicals that exhibit robust pharmacological properties, such as flavonoids, polyphenols, alkaloids, and tannins. 1 Atriplex halimus L. (Chenopodiaceae) is a halophyte that has been widely used in traditional medicine, making it an important biological resource. Ancient herbal medicine A. halimus has been employed in the setting of the sub-Saharan area of Algeria to treat a wide range of medical ailments, including digestive and genital diseases, 1 diabetes, rheumatism, and hydatid cysts.2,3 El Oued area is characterized by arid climatic conditions, which have a significant influence on its geographical and climatic characteristics. Consequently, these aforementioned circumstances give rise to the existence of a varied and plentiful botanical population, which is sometimes disregarded or undervalued.4,5
However, a considerable proportion of plant species present in this particular geographical area are experiencing a decline in population. The botanical specimens in question have notable chemical capabilities that hold promise for medicinal applications, yet their potential has not been well investigated and their significance has been little recognized. The utilization of plant-based medicines exhibits significant potential as a viable means to obtain antioxidants from natural sources. The genus Atriplex, which has 225 species, has attracted considerable interest due to its prolific presence in Ouargla's natural specimens. 6 Atriplex species are recognized for their usage as leafy green vegetables, distinguished by the presence of organic acids, flavonoids, alkaloids, and cardenolides within their foliage. 7 The extracts of A. halimus have shown a diverse array of medicinal applications, such as anti-cancer, hypolipidemic, antidiuretic, anti-allergic, and anti-diabetic effects. 8 Research into the antibacterial capabilities of medicinal herbs has received attention in light of growing worries about antibiotic resistance and the limits of traditional therapies. The antibacterial activities of multiple plant extracts have been studied extensively; nevertheless, introducing novel microbial strains demands more study. Despite its long history of employment in soil regeneration operations owing to its salt-extracting powers, there is still very little written about A. halimus reported. 9
Due to their ability to reduce oxidative damage and the accompanying progressive illnesses, polyphenols, a prominent class of dietary antioxidants, have received a great deal of investigation.10,11 Polyphenols are mostly sourced from fruits and drinks, with A. halimus gaining notoriety due to its high nutrient content and powerful antioxidant abilities. 12 There are a lot of moving parts involved in getting the phenolic chemicals out of plants, including their composition, the extraction process, particle dimension, amount of time, conditions of preservation, and the presence of chemicals that interfere. 13 Because phenolic compounds are connected with a wide variety of biological functions, there has been a recent uptick in interest about the discovery and use of these compounds in useful nutritional products. 14 The use of antioxidants derived from plants as a natural alternative to their synthetic analogues has recently become widespread in the processing of food. Phenolic substances, in particular, display a wide variety of features, notably the ability to chelate metals and to engage in redox activity. 15 Polyphenol extraction uses different solvents, however solvent and extraction process affect yield and compound integrity. 16
Algerian botanicals have tremendous potential for therapy, although biological study is scarce. This research aimed to examine how solvents type and acidity, extraction temperature and time variations affect total polyphenol content, antioxidants, and antibacterial potential in ethnopharmacological plants. The study sought to optimize extraction conditions and characterize the biological activities of phenolic and flavonoid compounds in Atriplex halimus L., providing updated insights into their pharmaceutical and nutraceutical relevance.
Material and Methods
Study Design, Location, and Duration
This was an in vitro experimental study designed to evaluate the bioactive properties of Atriplex halimus L. extracts under different extraction conditions. Fresh plant material was collected in May 2023 from Taghzout, El-Oued, South Algeria (33.356° N, 6.863° E). Laboratory analyses, including extraction, phytochemical determination, antioxidant, antibacterial, and anti-hemolytic assays, were carried out between May and December 2023 at the Process Engineering Laboratory, Kasdi Merbah University (Ouargla, Algeria), in collaboration with the Laboratory of Applied Chemistry and Environment, University of Hamma Lakhdar El-Oued, Algeria.
Materials
Chemicals
Biochem, Chemopharma (Montreal, Quebec) generously supplied the Folin-Ciocalteu reagent. The sodium carbonate (Na2CO3), acetone (from Prolabo), disodium hydrogen phosphate (Na2HPO4), ethanol (from Prolabo), methanol and sodium dihydrogen phosphate (NaH2PO4) (from Prolabo) were all sourced from the European Economic Area (CE). quercetin and gallic acid all came from Biochem-chemopharma (UK), as did ferric chloride (FeCl3,6H2O) and potassium ferricyanide (C6N6FeK3). Sigma-Aldrich (located in Steinheim, Germany) supplied the 1,1-diphenyl-2-picrylhydrazyl (DPPH).
Plant material
During May 2023, the leaves of A. halimus were gathered at Taghzout, which is located in El-Oued, South Algeria. Atriplex halimus L. was identified by Pr. Chehma A. (Ouargla University) and a voucher specimen (AH-09) was placed in the Laboratory of Process Engineering, Faculty of Applied Sciences, Kasdi Merbah University, Ouargla, 30000, Algeria.
After being cleaned, the leaves were left in the dark to dry at room temperature. After removing the limbs and the thick veins, the leaves were crushed into a fine powder. The dried and powdered leaves of Atriplex halimus L. were stored in airtight dark glass containers at 4 °C until further use in order to minimize degradation of phenolic and flavonoid compounds.
Methods
Plant Extraction
According to the method described by (Azwanida, N. N et al, 2015), 17 a glass vial was used to contain 500 mg of dry botanical powder, which was then subjected to extraction using 15 mL of solvent. During the extraction process, the mixture was placed in a water-bath shaker at various temperatures and for different durations. The resulting mixture was then centrifuged at 5000 rpm for 20 min using a Nuve NF 200 centrifuge (Turkey). Subsequently, the supernatant was filtered through Whatman filter paper. The extraction procedure was performed in duplicate.
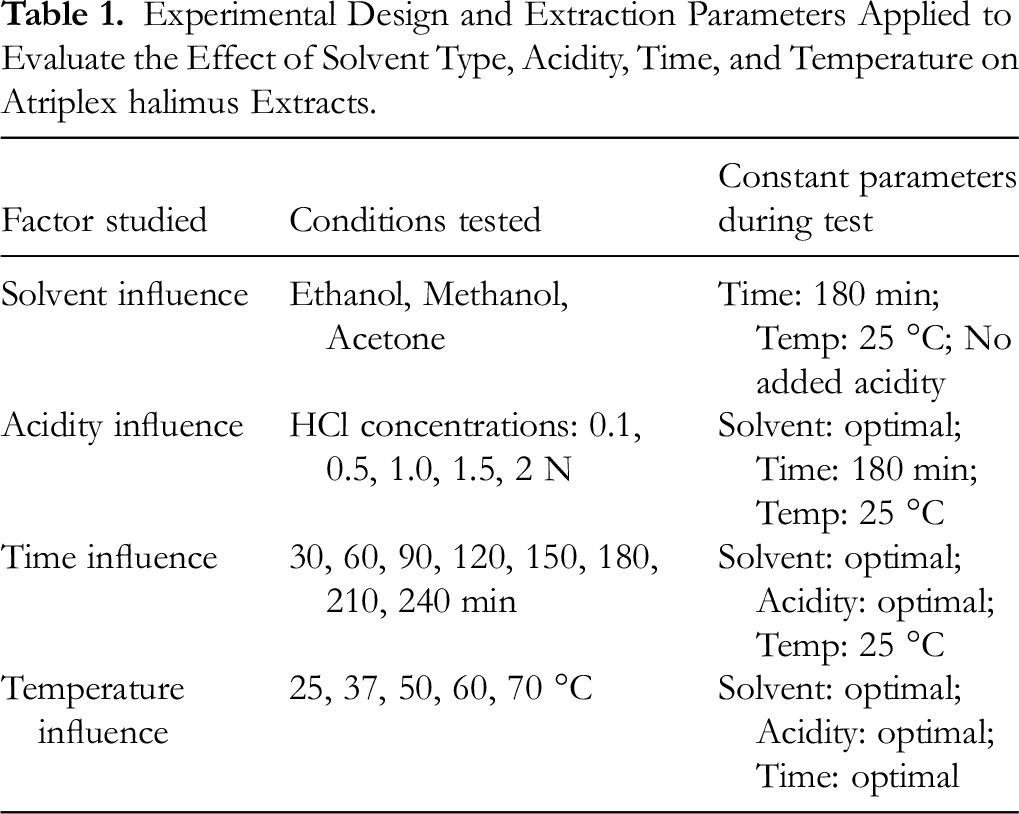
Experimental Design
The experimental design used in this study is an in-house standardized protocol established and optimized within the Laboratory of Applied Chemistry and Environment, Faculty of Exact Sciences, University of Hamma Lakhdar El-Oued. All tested variables were internally selected and optimized.
The extraction of antioxidant phenolic components from A. halimus leaves was investigated using single-factor trials. The effects of varying one factor at a time, including extraction solvent (ethanol, methanol, and acetone), solvent acidity (0 to 2 N), extraction time (30 to 240 min), and extraction temperature (25 to 70 °C), were examined while keeping all other parameters constant. The measurements of TPC, TFC, DPPH-RSA, and FRAP were employed to determine the most effective extraction conditions.
The extraction times and temperatures for the A. halimus leaf samples were 180 min and 25 degrees Celsius, respectively, using ethanol, acetone, and methanol.
The leaves of A. halimus plant were extracted using the optimal solvent. The extraction procedure was conducted using various doses of hydrochloric acid (HCl), namely 0.1, 0.5, 1, 1.5, and 2 N. The specified values for the extraction period were 180 min and the temperature was preset at 25 °C.
The optimal solvent and optimal solvent acidity were used to extract leaves from A. halimus samples. There were 8 different extraction times tested: 30, 60, 90, 120, 150, 180, 210 and 240 min. The extraction process always occurred at a constant 25 degrees Celsius.
halimus leaves were subjected to extraction at four various temperatures (25, 37, 50, 60 and 70 degrees Celsius) utilizing the previously determined optimum factors, including type and acidity of solvent and extraction time (Table 1).
Experimental Design and Extraction Parameters Applied to Evaluate the Effect of Solvent Type, Acidity, Time, and Temperature on Atriplex halimus Extracts.
Content Determination
Total Phenolic Content
The quantification of the total phenolic content (TPC) of the extract was conducted using the Folin-Ciocalteu's colorimetric technique, with gallic acid serving as the reference compound. In a succinct way, a total volume of 100 μl of each sample and standard (1 mg/mL in methanol), which have been previously dissolved in methanol, are combined with 200 μl of Folin-Ciocalteu reagent and 900 μl of water. After a duration of 5 min, a volume of 1000 microliters of sodium carbonate (Na2CO3) with a concentration of 7% is introduced. Subsequently, the resulting solution is diluted by the addition of distilled water until the final volume reaches 2.5 milliliters. The combination was subjected to vortexing for a duration of 15 s and thereafter left undisturbed for a period of 90 min at ambient temperature, while maintaining a light-free environment. The spectrophotometer was used to measure the absorbance at a specific wavelength of 750 nm, with the control solution serving as the blank. The control solution was generated in a similar manner, The extract was replaced with distilled water, save for one alteration. The quantification of the total phenolic content was conducted by measuring the amount of Gallic Acid Equivalent in milligram per 100 g DW of extract (mg GAE/100 g DW). The results are presented as the average value accompanied by the standard deviation, derived from three unique and independent research. 18
Total Flavonoid Content
Aluminum trichloride (AlCl3) was used to ascertain flavonoid concentration by the development of complex between flavonoids and aluminum. 19 In brief, a 5 ml amount of the sample and the standard, both substances were produced using methanol as the solvent, are combined with an equal volume of AlCl3 solution (2% concentration in methanol) and well mixed. After a 10-min period of incubation at room temperature, the measurement of absorbance is performed at a wavelength of 415 nm. The experiment was conducted with a total of three replicates. Quercetin was used to build a calibration curve; the subsequent step included the expression of flavonoid content (TFC) as milligrams of Quercetin per 100 gram dry weight of extract (mg QE/100 g DW). The data is given in the form of the mean and standard deviation obtained from three separate tests.
Antioxidant Activity Assays
1,1-Diphenyl-2-Picrylhydrazyl (DPPH) Radical Scavenging Activity Assay
The extracts and standards underwent testing using the methodology outlined in the reference,20,21 to determine their ability to scavenge DPPH stable radicals. The DPPH solution, with a concentration of 0.1 mM in methanol, was introduced to samples of different concentrations. These samples, consisting of 1 mL each, were created by reconstituting in solvents that corresponded to their respective concentrations. The mixture was then vortexed. Following a reaction period of 20 min at a temperature of 25 degrees Celsius, the quantification of absorbance was performed at a wavelength of 517 nm using a spectrophotometer. The reference solution for establishing a blank was methanol. As a comparison, 5 mL of methanolic DPPH solution without inhibitors was employed. The quantification of the extract's capacity to scavenge DPPH radicals was assessed by determining its half inhibitory concentration (IC50) Here is how we determined how much of DPPH was quenched:
Ferric Reducing Antioxidant Power (FRAP) Assay
The potassium ferricyanide-ferric chloride method was used to figure out how well A. halimus leaf extracts reduce ferric. 22 The reduction of potassium ferricyanide (Fe+3) to potassium ferrocyanide (Fe+2) occurs when exposed to reducing agents. Subsequently, the potassium ferrocyanide is further reduced to ferric chloride (Fe+2) by ferric chloride (FeCl3). The aforementioned chemical process yields a complex consisting of ferric and ferrous ions, which displays a distinct peak in absorbance at a wavelength of 700 nm. 23 The sample was diluted with a phosphate buffer (0.2 M, pH 6.6) and ferricyanide of potassium (1% weight/ volume) to a final volume of 1 mL. Following a duration of 20 min at a temperature of 50 °C, a volume of 2.5 mL of trichloroacetic acid with a concentration of 10% by volume (w/v) was introduced into the mixtures. A volume of 5.0 mL comprising 2.5 mL of distilled water and 0.5 mL of a 0.1% FeCl3 (w/v) solution were introduced into the mixture for the purpose of dilution. At 700 nm, blue-green color intensity was measured on a white background. The amount of ascorbic acid equivalents (mg AAE/100 g DW) used to quantify ferric reduction activity. The numbers are shown as the average of three separate calculations.
Linoleic Acid /β-Carotene Bleaching Assay
The antioxidant power of a plant extract was tested using a version of Pratt's β-carotene whitening method that had been changed.
24
In a solution of 0.5 ml chloroform, 0.2 mg of β-carotene and 20 mg of linoleic acid were combined to create a mixture, and 200 mg of Tween 40 (polyoxyethylene sorbitan monopalmitate). The chloroform was subjected to evaporation using a rotary evaporator operating at a temperature of 45 degrees Celsius and under vacuum conditions. After thoroughly combining the ingredients, they were mixed with 10 ml of water that had been triple-distilled and agitated for another minute or so. The total volume of the emulsion was then brought to 50 ml by adding oxygenated distillate water. This emulsion was divided into 0.2 ml aliquots and placed in separate test tubes. To facilitate evaluation, ascorbic acid was used. To serve as a comparison, a control was prepared using 0.2 mL of the equivalent solvent and 4 mL of the aforementioned emulsion. The tubes underwent heating in a water bath maintained at a temperature of 50 degrees Celsius. The absorbances of all samples at a wavelength of 470 nm were first measured at time zero (t = 0), then thereafter at 15-min intervals until a total period of 180 min (t = 180 min). As a control, we made a similar combination without the β-carotene. All counts were done three times for accuracy. Extracts’ antioxidant activity (AA) was calculated by measuring their ability to bleach β-carotene.
Anti-Hemolysis Assay
The assessment of anti-hemolysis activity was conducted by the quantification of the concentration of released plasma hemoglobin.
25
A blood sample was collected from a healthy human individual with blood type O+ using the Malagobi preparation procedure
26
with little modification in accordance with.
27
In this experimental study, a specified amount of 40 µl of erythrocytes derived from blood was combined with 2 ml of an extracted solution at various concentrations. Subsequently, the resultant solution was subjected to incubation at an ambient temperature of 37 °C for a period of 5 min. An additional volume of 40 µl was added to the mixture, consisting of 30 ml mol of H2O2 and 80 ml mol of FeCl3. After incubating for 1 h at a temperature of 37 °C, the mixture underwent centrifugation at a speed of 700 revolutions per minute for a period of 10 min. Following this, the measurement of absorbance was conducted on the resultant supernatant at a specific wavelength of 540 nm using a UV-Vis spectrophotometer. Ascorbic acid served as the reference standard (positive control) in this investigation.
The Test for Scavenging of Superoxide Anion
The experimental procedure for measuring the activity of superoxide anion radical scavengers was facilitated by the use of the riboflavin-light-NBT system.
28
In this experimental study, a sample volume of 1 ml was obtained at different quantities. Subsequently, it was mixed with 0.5 ml of phosphate buffer solution (50 mM, pH 7.6), 0.3 ml of riboflavin (50 mM), 0.25 ml of PMS (20 mM), and 0.1 ml of NBT (0.5 mM). The reaction was begun by exposing the reaction mixture to illumination using a fluorescent light source. The absorbance at a wavelength of 560 nm was measured after a 20-min incubation period. The standard used in the experiment was ascorbic acid. The scavenging capacity of the plant extract was assessed using the following equation:
Hydroxyl Radical Scavenging Assay
Various fractions of A. halimus L. extract was evaluated to determine their ability to eliminate hydroxyl radicals. This was accomplished by submitting the fractions to the Fe+3 ascorbate-EDTA-H2O2 system, also known as the reaction of Fenton.
29
The reaction mixture consisted of 500 liters of 2-deoxyribose (2.8 millimolar concentration) dissolved in phosphate buffer (50 millimolar concentration, pH 7.4), 200 liters of a solution containing a 1:1 volume ratio of premixed ferric chloride (100 millimolar concentration) and EDTA (100 millimolar concentration), and 100 liters of hydrogen peroxide (200 millimolar concentration). The extract solution (100 liters) was optionally included in the reaction mixture. Following an incubation period of 1 h at a temperature of 37 degrees Celsius, a volume of 100 liters with a concentration of 300 millimolar ascorbate was introduced to initiate the reaction1 ml of a 2.8% w/v solution of TCA in water was added to the reaction mixture. Then, 1 ml of a 1% solution of TBA in water was added. The mixture was heated in a water bath for 15 min until it was boiling. We used a blank solution, which was the same as the test solution except that it didn't have the reagent, to measure absorption at a wavelength of 532 nm. This measurement was performed subsequent to the cooling of the combined solutions. The following analysis presents a computation of the hydroxyl radical scavenging activity:
The Action of Scavenging Hydrogen Peroxide
A 50 mM solution of phosphate water (pH 7.4) was used to make a 2 mM solution of hydrogen peroxide. After putting 0.1 ml samples of each part into its own test tube, the rest of the space was filled with 50 mM phosphate buffer solution with a pH of 7.4. Following the addition of 0.6 ml of hydrogen peroxide solution, the tubes were subjected to vigorous shaking for a duration of 10 min. then, the absorbance of the hydrogen peroxide was quantified at a wavelength of 230 nm and then compared to a blank sample30,31 . Using the following calculation, the ability to get rid of hydrogen peroxide was figured out:
2,2’-Azino-bis (3-Ethylbenzothiazoline-6-Sulfonic Acid) ABTS Assay
The ABTS test used the methodology proposed by Arnao et aland Aouadi et al32,33 ; but with some modifications. The stock liquids consisted of a 7.4 mM solution of ABTS and a 2.6 mM solution of potassium persulfate. Subsequently, the viable resolution was achieved by the amalgamation of equimolar quantities of the two initial solutions, followed by a 12-h incubation period at ambient temperature and under light-restricted conditions. Subsequently, the solution was diluted by the process of combining 1 mL of ABTS solution with 60 mL of methanol. The process was continued until the spectrophotometer indicated an absorbance value of 0.7 at a wavelength of 734 nm. A novel ABTS response was generated for every individual test conducted. A total volume of 150 mL of samples was combined with 2850 mL of the ABTS solution under conditions of darkness for a duration of 2 h. Subsequently, the spectrophotometer was used to quantify the absorbance at a wavelength of 734 nm.
Antibacterial Activity in Vitro
The antibacterial activity of Atriplex halimus L. extracts was evaluated against four bacterial strains: Bacillus subtilis (ATCC 6633), Pseudomonas aeruginosa (ATCC 9027), Staphylococcus aureus (ATCC 6538), and Salmonella typhimurium (ATCC 14028). The assay was performed using the agar well diffusion method as described by Aouadi, et al 33 with slight modifications. Bacteria were cultured in nutrient broth for 24 h, and suspensions were adjusted to 0.5 McFarland standard. These suspensions were spread evenly onto Mueller–Hinton agar plates, and wells of 6 mm diameter were made with a sterile cork borer. Wells were filled with 100 µL of the extract at concentrations of 1, 4, 8, 12, and 16 mg/mL (prepared in DMSO). Concentrations were expressed in mg/mL instead of percentages for clarity. Ciprofloxacin was used as the positive control, and DMSO as the negative control. Plates were incubated at 37 °C for 24 h, after which inhibition zones were measured in millimeters
Statistical Analysis
Data were expressed as means ± standard deviation (n = 3). The statistical analysis was carried out using XLSTAT statistical software and a Tukey's Honestly Significant Difference (HSD) test was used to determine significant differences between the means (p < .05). Also, correlations between total phenolic and flavonoid contents and antioxidant activities was performed according to Bravais-Person correlation test.
Results
Effect of Extraction Conditions on Phytochemical Content and Antioxidant Activities
Effect of Solvent
Effect of Solvent Extraction on A. halimus Total Phenolic and Flavonoid Contents
This research used a variety of solvents to extract antioxidant phenolic components from A. halimus. The findings indicated that phenolic compounds were present in all extracts of A. halimus, with the concentration of these compounds differing according to the specific solvent used for extraction (Table 2 and Figure 1).

Effect of Solvent Type on the Total Phenolic (A) and Total Flavonoid (B) Contents of A. halimus. Bars are Mean Values ± Standard Error (n = 3). Bars with Different Letters are Significantly Different from Each Other (Tukey's Test, P < .05).
Effect of solvent extract on A. halimus total phenolic and total flavonoid contents, 1,1-diphenyl-2-picrylhydrazyl radical-scavenging ability, and the ferric power reduction.
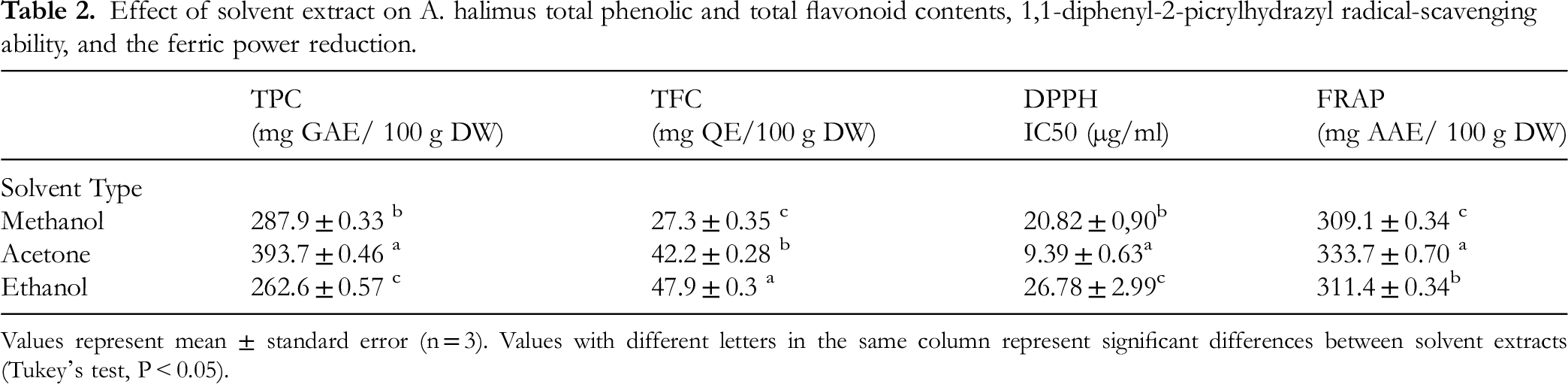
Values represent mean ± standard error (n = 3). Values with different letters in the same column represent significant differences between solvent extracts (Tukey's test, P < 0.05).
Acetone extract emerged as the most effective solvent for extracting total phenolic compounds (TPC) from A. halimus, yielding a significantly higher amount of TPC (393.7 ± 0.46 mg GAE/100 g DW, p < .0001) compared to ethanol extract (262.6 ± 0.57 mg GAE/100 g DW) and methanol extract (287.9 ± 0.33 mg GAE/100 g DW), which exhibited the lowest value. In this study, it was shown that ethanol extract had the highest efficacy as a solvent for the extraction of total flavonoid content (TFC) from A. halimus (p < .0001), resulting in a yield of 47.9 ± 0.3 mg QE/100 g DW. Acetone and methanol extract, on the other hand, yielded TFC values of 42.2 ± 0.28 mg QE/100 g DW and 27.3 ± 0.35 mg QE/100 g DW, respectively.
Effect of Solvent Extraction on Antioxidant Activity of A. halimus Extracts
The evaluation of the antioxidant potential of phenolic compounds derived from A. halimus was carried out using two well-established methodologies: the 1,1-diphenyl-2-picrylhydrazyl radical-scavenging ability (DPPH) and the ferric power reduction test (FRAP). The aforementioned techniques are often used for the assessment of the antioxidative properties shown by various extracts of plants and natural substances.
1,1-Diphenyl-2-picrylhydrazyl Radical Scavenging Activity
The assessment of antioxidant activity in extracts may be efficiently conducted by using the rapid method of scavenging stable DPPH free radicals. The absorbance of the DPPH radical decreases as it undergoes scavenging by an antioxidant, leading to the formation of a stable DPPH-H molecule, resulting in a color transition from violet to yellow. The molecule in question exhibited an absorbance at a wavelength of 515 nm in its radical state. This absorbance was shown to diminish with the acquisition of an electron or hydrogen from an antioxidant, resulting in the formation of a stable diamagnetic molecule.
The solvent with the greatest antioxidant activity was discovered to be acetone (Table 2 and Figure 2A). The acetone solvent had the lowest inhibitory concentration at 50% value of (IC50 = 9.39 ± 0.63 µg/ml), which was statistically more effective for trapping DPPH radical with the percents of superiority respective 54.89 and 64.94% compared to those of methanol (IC50 = 20.82 ± 0.90 µg/ml, p = .001) and ethanol (IC50 = 26.78 ± 2.99 µg/ml, p < .0001).
Ferric Reducing Antioxidant Power assay

Effect of Solvent Type on A. halimus DPPH (A) and Ferric Reducing (B) Antioxidant Power Assays. Bars are Mean Values ± Standard Error (n = 3). Bars with Different Letters are Significantly Different from Each Other (Tukey's Test, P < .05).
The evaluation of the ferric reduce antioxidant power (FRAP) of extract samples was conducted by the direct reduction of Fe3+(CN–)6 to Fe2+(CN–)6. The test solution undergoes a color transformation from yellow to different hues of green and blue, which is contingent upon the presence of reductants inside the sample. Therefore, the monitoring of Fe2+ may be achieved by quantifying the development of Perl's Prussian blue at a wavelength of 700 nm.
Similar to the DPPH method, it was noted that the acetone extract exhibited the maximum FRAP (333.7 ± 0.70 mg AAE/100 g), surpassing the values obtained for ethanol (311.4 ± 0.34 mg AAE/100 g) and methanol (309.1 ± 0.34 mg AAE/100 g) with 6.68 and 7.37%, successively (Table 2 and Figure 2B).
Correlation Between Phytochemical Contents and Antioxidant Capacities
The results of Pearson's correlation test (Table 3) indiquated that the DPPH radical inhibition showed a negative tight correlation with TPC (r = −0.96, p < .0001). In contrast, it was weakly and positively correlated with TFC (r = −0.099, p = .79). While, the FRAP values showed a positive correlation with TPC (r = 0.96, p < .0001) and FTC (r = 0.33, p = .378).
Pearson's Correlation Test Between Total Phenolic and Total Flavonoid Contents and Their DPPH and FRAP Antioxidant Properties of A. halimus Solvent Extracts.

p: Probability level. Correlation is signifcant at p < .05.
Effect of Solvent Acidity
The study's results as shown in Table 4 and Figure 3 revealed that the highest value of total phenolic was obtained with the introduction of hydrochloric acid (HCl) 1N in acetone extract solvent (411.8 ± 0.87 mg GAE/100 g DW). For flavonoid metabolits, a similar trend was detected; there was an increase in their amounts by the addition of 1N HCl, so that there was no significant diference between the extract solvents of 1N and 1.5N (p = .99), that recorded the highest values (48.1 ± 0.34 and 47.9 ± 0.27 mg QE/100 g DW, respectively). Concerning the antioxidant activity, as the same that the case of flavonoids, there is an increase in the antioxidant activity in the presence of HCl at 1 and 1.5 N without significant difference between them (8.31 ± 0.63 and 9.26 ± 0.22 µg/ml, p = .29) and (348.3 ± 0.23 and 347 ± 0.37 mg AAE/ 100 g DW, p = .59), respectively for DPPH and FRAP assays.

Effect of Solvent Acidity on A. halimus TPC, TFC, DPPH and FRAP Assays. Bars are Mean Values ± Standard Error (n = 3). Bars with Different Letters are Significantly Different from Each Other (Tukey's Test, P < .05).
Effect of Variations in Solvent Acidity, Extraction Time, and Extraction Tempperature on the Total Phenolic and Total Flavonoid Contents, 1,1-Diphenyl-2-Picrylhydrazyl Radical-Scavenging Ability, and the Ferric Power Reduction of A. halimus.

Values represent mean ± standard error (n = 3). Values with different letters in the same column and in the similar extraction conditions represent significant differences between each other (Tukey's test, P < .05).
Effect of Extraction Time
The duration of the extraction process plays a crucial role in optimizing energy consumption and reducing costs associated with extraction. The findings of our study indicate that the TPC and TFC, DPPH and FRAP exhibited an increase when the extraction period was extended from 30 min to 210 min. However, a drop in these parameters was seen at the 240-min mark, as shown in Table 4 and Figure 4. The extraction process yielded to the considerably high amounts of TPC of 408.1 ± 0.36 mg GAE/100 g and TFC of 47.5 ± 0.75 for A. halimus sample, with an antioxidant activity of 9.39 ± 0.34 µg/ml for DPPH test and 356.2 ± 0.22 mg AAE/100 g FRP method, for 210 min of extract time (Table 4).

Effect of Extraction Time on A. halimusTPC, TFC, DPPH and FRAP Assays. Bars are Mean Values ± Standard Error (n = 3). Bars with Different Letters are Significantly Different from Each Other (Tukey's Test, P < .05).
Effect of Extraction Temperature
The total polyphenol content, total flavonoid content, and the DPPH radical-scavenging capacity of A. halimus was seen to be strongly influenced by the use of 1N acetone as the solvent and an extraction duration of 210 min. Furthermore, an investigation was conducted to examine the influence of different extraction temperatures on these characteristics, as seen in Table 4 and Figure 5. The results revealed a significant decrease in TPC produced as the temperature increased from 37 °C to 70 °C, with recorded values ranging from 407.3 mg GAE/100 g to 310.1 mg GAE/100 g. The reduction in TPC equal to 23.88% upon exposure to 70 °C compared to the 37 °C . Likewise, an increase in the extraction temperature from 37 °C to 70 °C results in a concomitant decrease in the yield of TFC. More precisely, the yield exhibits a drop from 51.7 mg QE/100 g to 33.2 mg QE/100 g ie a reduction rate of 35.78%. The same behavior was recorded for the thermal effects on the reducing power of A. halimus extracts towards DPPH and FRAP radicals. Data demonstrated a significant dimunition in the reducing appility of extract samples when the extraction temperature was increased from 37 °C to 70 °C, leading to the dropp of 76.57 and 13.73% correspondingly.

Effect of Extraction Temperature on A. halimus TPC, TFC, DPPH and FRAP Assay Assays. Bars are Mean Values ± Standard Error (n = 3). Bars with Different Letters are Significantly Different from Each Other (Tukey's Test, P < .05).
Correlation Analysis Between Total Phenolic and Flavonoid Contents and Antioxidant Activity at Different Extract Conditions
The findings summarized in Table 5 shown that the TP and TF contents determined through each extraction conditions gaves successively a higher significantly negative and positive interrelationship with results of IC50 against DPPH radical and AAE values of FRAP assay. The obtained Pearson's correlation coefficients ranged respectively from 0.90≥ |r| ≥ 0.98 and 0.50≥ |r| ≥ 0.96, for DPPH test and between 0.62≥ |r| ≥ 0.90 and 0.59≥|r| ≥ 0.97, for FRAP method. The high correlations confirm the role of phenolic compounds as the main contributor to the antioxidant activities of A. halimus extracts.
Pearson's Correlation Test Between Metabolite Contents in A. halimus Extract and Their Antioxidant Properties.
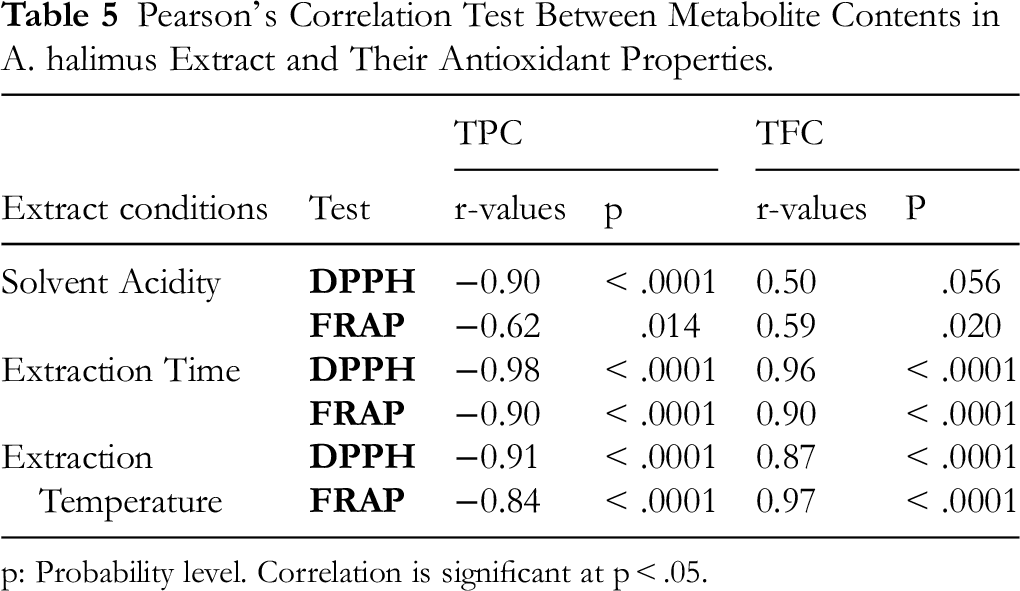
p: Probability level. Correlation is significant at p < .05.
Linoleic Acid / β-Carotene Bleaching Test
Based on the findings illustrate in Figure 1, the A. halimus extracts acquired via the utilization of acetone as a solvent, an acidity level of 1N, an extraction duration of 210 min, and a temperature of 37 °C showed a dose dependent potent inhibition of linoleic acid/ β–carotene system. The increase in the concentration of sample extract resulted in an increase in the linoleic acid/b-carotene scavenging ability (Figure 6). The IC50 value of the extract was comparable to that of the reference compound butylated hydroxyanisole (BHA), with the IC50 values of 0.46 ± 0.02 and 0.30 ± 0.01 mg/ml, respectively for A. halimus extract and BHA (Table 6).

Linoleic Acid/β-Carotene Bleaching Assay of Atriplex halimus Extracts at Different Concentrations. C1: 0.1 mg/mL; C2: 0.2 mg/mL; C3: 0.3 mg/mL; C4: 0.4 mg/mL; C5: 0.5 mg/mL; BHA: Butylated Hydroxyanisole (Positive Control).
Influence of A. halimus Extract on Linoleic Acid/ β-Carotene Bleaching, Hydroxyl Radical, Superoxide, Hydrogen Peroxide and ABTS Scavenging Activities IC50 Values (mg/mL).

Values represent mean ± standard error (n = 3). Values with different letters in the same column present significant differences between the results obtained for A. halimus extract and those for standards (Tukey's test, P < .05).
Hydroxyl Radical Scavenging Assay
A restricted quantity of free radicals serves an inevitable function in the regulation of homeostasis and the facilitation of stress responses inside living organisms. Nevertheless, an overabundance of free radicals may result in the oxidative degradation of proteins, lipids, and DNA molecules, hence contributing to the onset and progression of several degenerative ailments. Therefore, exogenous antioxidants are of significant importance in safeguarding against several maladies, such as Alzheimer's disease, cardiovascular disease, and some types of cancer. The inhibitory effects of hydroxyl radicals in A. halimus were quantified by calculating the percentage of scavenging activity. The data shown in Figure 7 illustrates the impact of A. halimus extract concentrations and durations of reaction on the scavenging activities against hydroxyl radical generated. The results indicate that higher concentrations of the substance led to a notable enhancement in antioxidant activity. Similarly, the antioxidant activity exhibited an increase when the response time was prolonged, eventually reaching a stable state within the timeframe of 20 to 25 min across various doses. The IC50 value of A. halimus extract was 0.28 ± 0.01 mg/ml, which twice as significantly lower than that of the standard ascorbic acid BHA (IC50 = 0.14 ± 0.02 mg/ml, p = .000) (Table 6).

Hydroxyl Radical Scavenging Activity from Different Concentration of A. halimus.
Hydrogen Peroxide Scavenging Activity
The hydroxyl radicals (H2O2) are considered to be the most reactive species due to its capacity to effectively traverse cellular membranes and ability to initiate the peroxidation process of the cell membrane. The lipid radical, when generated, has the potential to launch a chain reaction in the presence of O2, resulting in the production of lipid peroxide. Subsequently, the breakdown of lipid peroxide leads to the production of aldehydes, including malondialdehyde. The data shown in Table 6 and Figure 8 mentionnes the relationship between concentration and time in relation to the scavenging activities of hydrogen peroxide for A. halimus extracts. It is evident that increasing the concentration resulted in a favourable impact on the antioxidant activity, as did the duration of time. Upon the prolongation of response time, a notable increase in antioxidant activity was seen, which eventually reached a steady state within the time frame of 20 to 25 min across almost all doses. This suggests that the reaction reaches near completion within a time frame of around 20 to 30 min. The IC50 value of the ascorbic acid against hydrogen peroxide radical recorded after a reaction duration of 30 min as 0.28 ± 0.07 mg/ml, which 42.36% was more significantly higher than that recorded for sample extract (0.48 ± 0.02 mg/ml, p = .01) for the same times of interaction (Table 6). The results of this this research suggest that the utilization of A. halimus extract in the scavenging of hydrogen peroxide is very efficient, particularly when using elevated concentrations and a reaction duration of 30 min. Hence, the scavenging of hydrogen peroxide exhibits a clear correlation with both its concentration and the duration of the process.

Hydrogen Peroxide Scavenging Activity from Different Concentration of A. halimus.
Scavenging Test for Superoxide Anions
A comparative investigation was undertaken to examine the influence of the concentration of A. halimus plant extract and the length of the reaction on the scavenging of superoxide radicals. The scavenging activity exhibited a positive correlation with the rise in concentration as seen in Table 6 and Figure 9. Regarding the variable of reaction time, it was evident that a rise in response time corresponded to an increase in scavenging activity. However, the reaction reached a state of equilibrium after around 25 min. Hence, it is apparent that the present moment presents an opportune period for the scavenging of superoxide radicals. The IC50 values (Table 5) of the extract and standard ascorbic acid in this assay were 0.17 ± 0.002 and 0.06 ± 0.014 mg/ ml, respectively. The IC50 value of the extract was about 3 times significantly less than that of the standard.
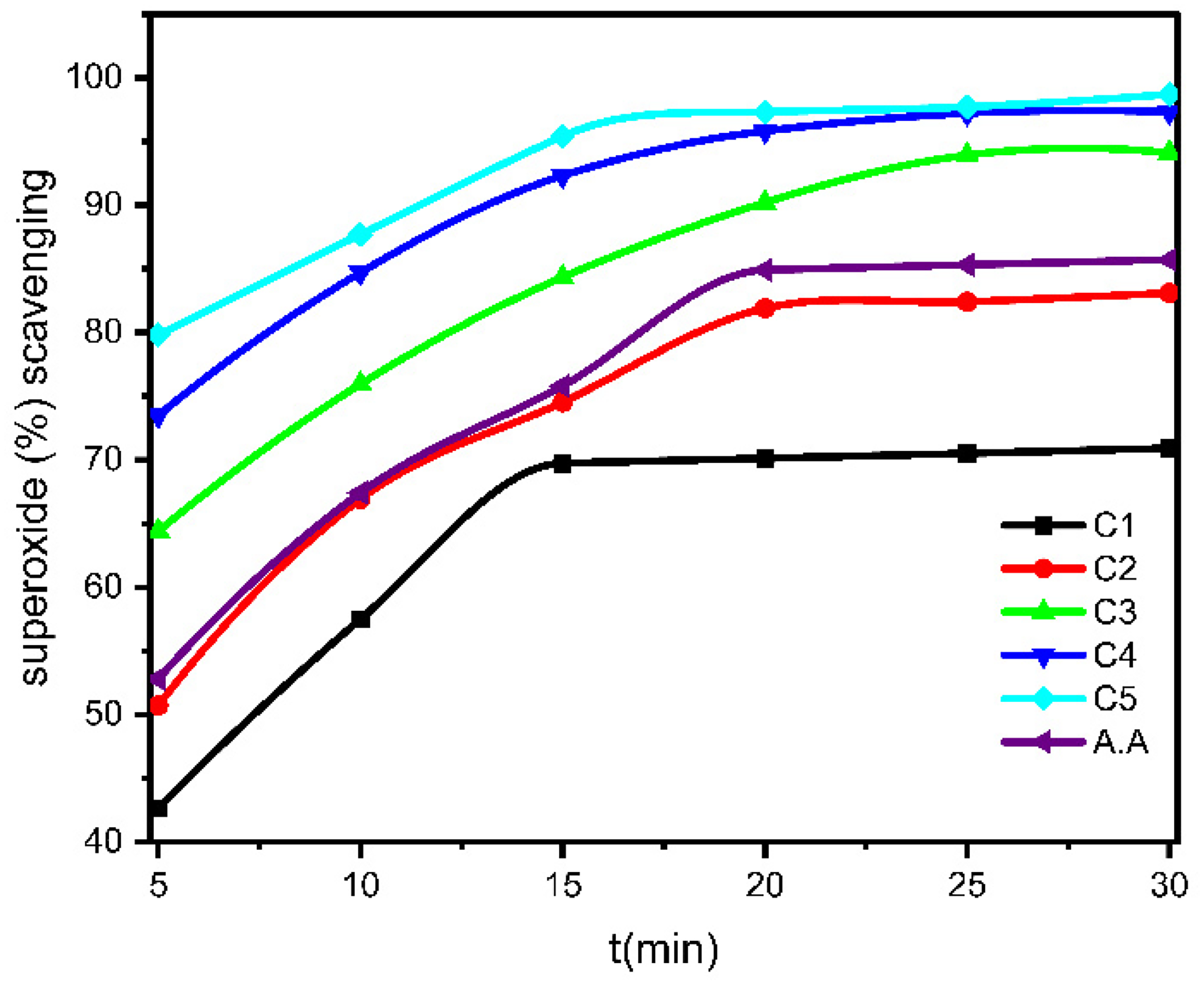
Hydrogen Peroxide Scavenging Activity from Different Concentration of A. halimus.
ABTS Scavenging Activity
Figure 6 shows that the free radical scavenging capacity of plant extract was a concentration-dependent. The observed augmentation in response time had favorable outcomes, as the scavenging activity exhibited an upward trend in conjunction with the elongation of the scavenging duration as well as evolution of the concentration, eventually reaching a plateau between 20 and 25 min across various concentrations. The calculated IC50 of A. halimus extract was 0.28 ± 0.004 mg/ml, which was significantly higher than that of the reference compound ascorbic acid (IC50 = 0.15 ± 0.005 mg/ml) (Table 6 and Figure 10), indicating that the plant extract was about 45.93% less potent to scavenger ABTS radical than BHA.

ABTS (%) Scavenging Activity from Different Concentration of A. halimus.
Hemolysis Inhibition Assay
A comparative analysis was done to look at the effects of reaction time and A. halimus plant extract concentrations on the percentage of erythrocyte hemolysis. The increase in concentrations was positively correlated with the hemolysis inhibition percentage. Regarding the variable of reaction time, it was evident that a rise in response time corresponded to an increase in anti-hemolytic activity. However, the reaction reached a state of equilibrium after around 20- 25 min across almost all doses. The half-effective concentration value EC50 estimated for the investigated sample was higher to the highest dose tested
The protection percentage from hemolysis is directly related to the antioxidant activity of A. halimus extract. Highest inhibition percentage indicate higher antioxidant activity, as seen in (Table 6 and Figure 11)

Hemolysis Inhibition (%) of Different Concentration of A. halimus.
Antibacterial Activity (in Vitro)
Table 7 lists the results of antibacterial activity against two Gram (+) bacteria (Staphylococcus aureus and Bacillus subtilis) and two Gram (-) bacteria (Pseudomonas aeruginosa and Salmonella typhimurium).
Zone of Inhibition of A. halimus Extract at Varied Doses Against Different bacteria.

Values represent mean ± standard error (n = 3). Values with different letters in the same row present significant differences between the bacteria strains (Tukey's test, P < .05)
The A. halimus extracts and reference positive (ciprofloxacin (CIP-5)) have an inhibitory action against both Gram positive and Gram negative bacteria. Obtained data demonstrate that when the concentrations of A. halimus extract and ciprofloxacin (CIP-5) rise, the recorded inhibitory zones were significantly widened. However, the Gram-positive tested strains are significantly the most sensitive to the tested samples, particularly S. aureus, with the inhibitory diameters varying from 16.83 ± 0.76 to 25.5 ± 1.32 mm for A. halimus extract and from 19.00 ± 1.5 to 26.83 ± 0.76 for ciprofloxacin (CIP-5).
Discussion
Effect of Extraction Conditions on Phytochemical Content and Antioxidant Activities
Effect of Solvent
Effect of Solvent Extraction on A. halimus Total Phenolic and Flavonoid Contents
Historically, it was conventional to isolate phenols from plants by liquid extraction methods utilizing solvents of varying temperatures, such as hot or cold solvents. The suitable solvents for this particular application include aqueous solutions including ethanol, methanol, and acetone. 34 The differences observed between solvents can be explained by their polarity, which determines the type of compounds extracted. Methanol, being highly polar, is more effective in extracting simple phenolics and low-molecular-weight flavonoids. Ethanol, with intermediate polarity, preferentially extracts flavonoid glycosides and moderately polar phenolic compounds. Acetone, which is less polar, is well known for its efficiency in extracting condensed tannins and proanthocyanidins. These observations are consistent with previous studies on solvent-dependent recovery of plant polyphenols. 35
Acetone is widely acknowledged as the most efficient solvent for the extraction of polyphenols, whereas ethanol is known for its excellent extraction of flavonoids. The aforementioned observations are consistent with the relationship between the solvent polarity used in the extraction procedure and the solubility of phenolic compounds in these solvents, as shown by the polarity values of acetone (0.355), ethanol (0.654), and methanol (0.762). 36 Therefore, it is apparent that no one solvent has the potential to extract all types of phenolic chemicals from a particular sample simultaneously. The solvent properties of acetone have been shown to be particularly effective in extracting phenolic substances with a significant molecular mass, such as condensed tannins. A commonly held concept posits that the polarity of a solvent decreases as its molecular weight increases, hence enhancing the extraction of molecules with comparable molecular weights. This phenomenon may be explained by the concepts of “like dissolves like” or “polarity versus polarity” as neither acetone and tannins exhibit significant molecular masses. Additionally, acetone exhibits a weakest polarity among the substances under consideration, while simultaneously possessing the highest recorded value for total phenolic compounds.37,38 The reason for the higher efficiency of acetone in extracting phenolics from A. halimus may be attributed to the prevalence of flavanols, which constitute the primary class of phenolic compounds in this plant.39,40 The performance of acetone in extracting antioxidant phenolic compounds from different source materials has been shown to be superior when compared to other organic solvents, including berries and apples, 41 fig, 42 buckwheat, 43 beach peas, 44 star fruits, 45 strawberries, 46 pistachio byproducts, 47 onions, 48 barley seeds, 49 eggplant byproducts, 50 fenugreek, 51 and soybean. 52
Effect of Solvent Extraction on Antioxidant Activity of A. halimus Extracts
The results of antioxidant investigation are consistent with the research conducted by González-Montelongon et al, 53 which indicated that acetone exhibited superior ability as a solvent for extracting phenolic components from banana peels.
The observed disparities in the antioxidant activity, as measured by DPPH and FRP, of extracts from A. halimus may be attributed to variations in the amount and quality of phenolic compounds found in the distinct extracts. The results indicate that acetone, as a solvent, demonstrated the most favourable outcome in terms of both total phenolic content (TPC) and antioxidant activities (DPPH and FRAP). This suggests that the phenolic compounds responsible for the overall antioxidant activity of the A. halimus extract are likely to be those with higher molecular weight and lower polarity, in accordance with the principle of “like dissolves like”.
Correlation Between Phytochemical Contents and Antioxidant Capacities
The negative correlation can be explained by that the lower IC50 value corresponds the higher antioxidant potenial. The antioxidant capacity of our extracts increased proportional to the TPC. In agreement with report of, 54 who annunced that the polyphenol and favonoid compositions of Searsia tripartita and Limoniastrum guyonianum correlated well with antioxidant proprietes. Also, 54 established a positive statistical correlation between the total phenolic contents and the antioxidant activities of Ziziphus lotus L. (Desf.) extracts, both for DPPH and FRAP assays. This may be associated with the chemical structure of phenolic compounds and the availability of phenolic hydroxyl groups which have the capacity to donate their electron or hydrogen, there by forming stable end products. 55
The extracts prepared by the use of acetone demonstrated a higher level of total phenolic compounds and antioxidant activity. Hence, the selection of acetone as the optimal solvent for assessing the influence of variation in each of the following conditions: solvent acidity, extraction time, and temperature.
Effect of Solvent Acidity
The addition of acid in the extraction solvent may potentially enhance the stability of phenolic compounds, such as anthocyanins, and facilitate their dissolution. The chemicals, which are first integrated into polymers or linked to other constituents of the cell wall by a hydrolysis process, facilitate the degradation of cell walls. Consequently, this particular procedure enhances the solubility and dispersion of phenolic chemicals derived from the botanical specimen. 56 However, it is important to acknowledge that past research which has shown that a high concentration of strong acid might potentially lead to partial hydrolysis and destruction of anthocyanin. 57 The observed decline in the FRSA of A. halimus extracts might be elucidated by the augmentation of acid content. Therefore, a solvent consisting of acetone with a concentration of 1N HCl was used for the subsequent procedures.
Effect of Extraction Time
Following a duration of 210 min, it was observed that extending the extraction period did not provide any significant improvements in the recoveries. The phenomenon in question may be elucidated by the use of Fick's second law of diffusion. The concept of equilibrium, seen in chemical systems, entails the anticipation of an ultimate state when the concentrations of solution in the matrix of solids and in the surrounding solution reach an equilibrium after a certain duration. Therefore, it is not vital to extend the length of phenolics extraction. 58 The results of our investigation are consistent with the studies done by Yap et al 59 and Chan et al, 60 which demonstrated that a time of 180 min resulted in the most effective extraction of phenolic compounds from star fruit (Averrhoa carambola L.) and Citrus hystrix peels, respectively.
In addition, it has been shown that extended extraction durations might lead to a higher likelihood of phenolics oxidation. 61 The prevention of oxidation in the solvent system might potentially be achieved by the incorporation of reducing substances. 62 The best extraction time for the recovery of phenolic components and antioxidant activity from A. halimus was found to be 210 min, based on the data available.
Effect of Extraction Temperature
Several investigations have shown that the use of heat has been seen to enhance the efficiency of extracting phenolic components and elevating the antioxidant activity of phenolic extracts.63,64 The behavior under observation may be ascribed to many causes, such as the increased solubility of phenolic compounds, the faster rate of diffusion, the improved efficiency of mass transfer and extraction, and the reduced viscosity and surface tension of the solvent. 65 The use of heat has the capacity to initiate the softening of plant tissue, resulting in a reduction in the intensity of phenol peptide and hydroxyl-polysaccharide interactions. As a result, it may be anticipated that a larger amount of polyphenols would translocate into the solvent. 66
In a research done by Guihua Xu et al. 67 It was shown that subjecting orange peel to heat treatment resulted in an elevation in the unbound fraction of phenolic acids, thereby leading to an augmentation in the overall antioxidant activity of the extract. However, it is crucial to recognize that raising the temperature extraction beyond specific thresholds could potentially lead to the concurrent degradation of phenolic compounds that were previously released at lower temperatures, or even the decomposition of residual phenolics within the plant matrix. In addition, it should be noted that elevated temperatures have the potential to increase the evaporation of solvents, hence increasing the overall cost of the extraction process from an industrial perspective. 60 Juan and Giuseppe 68 conducted a research on the black currants whereby they noticed that elevating the temperature above the range of 20–25 °C led to the degradation of anthocyanins.
Correlation Analysis Between Total Phenolic and Flavonoid Contents and Antiox-Idant Activity at Different Extract Conditions
The strong correlations observed between total phenolic and flavonoid contents and antioxidant activities across the different extraction conditions clearly underscore the pivotal role of these secondary metabolites in determining the bioactivity of Atriplex halimus. The negative correlation between TPC and IC50 values in the DPPH assay indicates that higher phenolic levels directly enhance radical scavenging capacity, as lower IC50 values reflect stronger antioxidant efficiency. Conversely, the positive association of TPC and TFC with FRAP values highlights the reducing power of these compounds, consistent with their ability to donate electrons or hydrogen atoms to neutralize reactive species. These findings align with earlier studies on other medicinal plants, such as Searsia tripartita and Limoniastrum guyonianum, 69 and Ziziphus lotus, 70 where phenolic abundance was also shown to strongly correlate with antioxidant performance. Collectively, the data confirm that polyphenols and flavonoids are not merely ancillary phytochemicals but act as the principal contributors to the antioxidant defense system of A. halimus, reinforcing its potential as a valuable natural source of bioactive compounds for pharmaceutical and nutraceutical applications.
Linoleic Acid / β-Carotene Bleaching Test
The β-carotene/linoleic acid bleaching assay further substantiates the potent antioxidant potential of Atriplex halimus extracts obtained under optimized conditions. The dose-dependent inhibition pattern demonstrates that increasing extract concentration proportionally enhances the ability to prevent oxidative degradation of β-carotene, reflecting efficient neutralization of linoleic acid–derived free radicals. 71 The IC50 value (0.46 ± 0.02 mg/mL), being comparable to that of the synthetic antioxidant BHA (0.30 ± 0.01 mg/mL) is particularly significant, as it suggests that natural phenolic constituents of A. halimus can rival the efficacy of widely used synthetic stabilizers. This finding not only highlights the plant's strong capacity to inhibit lipid peroxidation but also supports its potential as a natural alternative to synthetic antioxidants in food and pharmaceutical formulations. Similar outcomes have been reported for other phenolic-rich species, where natural extracts exhibited comparable or superior β-carotene bleaching inhibition relative to synthetic antioxidants, reinforcing the relevance of A. halimus as a sustainable and safe source of bioactive compounds. 72
Hydroxyl Radical Scavenging Assay
The hydroxyl radical scavenging activity of Atriplex halimus extracts provides compelling evidence of their ability to mitigate one of the most reactive and deleterious species in biological systems. The observed concentration- and time-dependent increase in activity underscores the efficiency of the extract in neutralizing hydroxyl radicals, with scavenging capacity stabilizing after approximately 20–25 min. 73 Although the IC50 value of A. halimus extract (0.28 ± 0.01 mg/mL) was moderately higher than that of ascorbic acid (0.14 ± 0.02 mg/mL), the extract still demonstrated a strong inhibitory effect, confirming the contribution of phenolic constituents to hydroxyl radical neutralization. Given that hydroxyl radicals are directly implicated in oxidative damage to lipids, proteins, and nucleic acids, the ability of A. halimus to effectively quench these species highlights its therapeutic relevance in preventing oxidative stress–related degenerative diseases such as neurodegeneration, cardiovascular disorders, and cancer. 74 These results are in line with previous findings for other polyphenol-rich plants, where phenolics were consistently identified as the principal contributors to hydroxyl radical scavenging activity. Collectively, this positions A. halimus as a promising candidate for the development of natural antioxidant formulations aimed at counteracting hydroxyl radical–mediated cellular damage.75,76
Hydrogen Peroxide Scavenging Activity
The ability of Atriplex halimus extracts to scavenge hydrogen peroxide further supports their broad-spectrum antioxidant potential. Hydrogen peroxide, although less reactive than hydroxyl radicals, is a key precursor of highly damaging oxidative species through the Fenton reaction and its capacity to permeate biological membranes makes it particularly dangerous. The concentration- and time-dependent scavenging pattern observed indicates that A. halimus extracts can efficiently neutralize H2O2, with activity reaching equilibrium within 20-30 min. 77 While the extract displayed a higher IC50 value (0.48 ± 0.02 mg/mL) compared with ascorbic acid (0.28 ± 0.07 mg/mL), its activity remains notable, especially given that plant-derived antioxidants often function through synergistic interactions of multiple phenolic compounds rather than the potency of a single molecule. 78 These findings are consistent with prior reports showing that polyphenol-rich plant extracts attenuate hydrogen peroxide–induced oxidative stress by stabilizing radical intermediates and interrupting lipid peroxidation cascades. Therefore, the capacity of A. halimus to quench H2O2 highlights its potential to provide cellular protection against oxidative stress–mediated damage, further reinforcing its relevance as a candidate for nutraceutical and therapeutic applications. 79
Scavenging Test for Superoxide Anions
The superoxide radical is widely acknowledged as a prominent biological factor in the production of reactive oxygen species. 80 Although the superoxide anion is considered a relatively weak oxidant, it has the ability to produce very strong and dangerous hydroxyl radicals, along with singlet oxygen. The involvement of these reactive species is of considerable importance in the progression of oxidative stress. 81 Antioxidant enzymes have a crucial role in maintaining physiological levels of superoxide, hence influencing the development of rheumatoid arthritis, reperfusion damage, cardiovascular disease, immunological injury, and cancer. 82 In addition, it is worth noting that tissue damage in several situations may be attributed to the presence of reactive oxygen species, namely the superoxide anion, which is generated by neutrophils. 83
The superoxide radical scavenging assay revealed that Atriplex halimus extracts possess a remarkable capacity to mitigate one of the most biologically relevant reactive oxygen species. The concentration- and time-dependent enhancement of scavenging activity, reflects the efficiency of the extract in neutralizing superoxide radicals before they undergo dismutation to form more harmful oxidants, including hydroxyl radicals and singlet oxygen. 84 Although the IC50 value of the extract (0.17 ± 0.002 mg/mL) was higher than that of ascorbic acid (0.06 ± 0.014 mg/mL), the activity remains significant, demonstrating the contribution of polyphenols and flavonoids to free radical detoxification. The ability of A. halimus to quench superoxide radicals is particularly relevant given the established role of superoxide in the initiation of oxidative stress–mediated tissue injury, such as in cardiovascular disorders, rheumatoid arthritis, and cancer progression. 85 These findings are consistent with previous studies reporting strong correlations between phenolic abundance and superoxide radical inhibition in other medicinal plants, highlighting the therapeutic promise of A. halimus as a natural antioxidant source for preventing superoxide-driven oxidative damage. 86
ABTS Scavenging Activity
Many study research have investigate the inhibitory and scavenging capabilities of plant antioxidants towards the ABTS radical. 87 The evaluation of antioxidant efficacy may be conducted by assessing their capacity to take electrons or hydrogen radicals, therefore achieving molecular stability. 88
The ABTS radical cation assay confirmed the antioxidant potential of Atriplex halimus extracts. Although the extract displayed an IC50 value of 0.28 ± 0.004 mg/mL, which was higher than that of ascorbic acid (0.15 ± 0.005 mg/mL), the observed activity remains notable, particularly considering the complex mixture of phenolic constituents that likely act synergistically. The relatively lower potency compared to the standard antioxidant does not undermine the significance of the findings, as plant-derived extracts often provide broader bioactivity due to the combined effects of multiple phenolic and flavonoid compounds rather than a single active molecule. These results are consistent with reports on other medicinal plants where polyphenol-rich extracts demonstrated robust ABTS radical scavenging ability, albeit with IC50 values generally higher than those of pure reference antioxidants. The ability of A. halimus to effectively scavenge ABTS radicals thus reinforces its potential as a natural source of antioxidant molecules capable of complementing or replacing synthetic antioxidants in therapeutic and nutraceutical applications. 89
Hemolysis Inhibition Assay
The objective of this experiment is to evaluate the potential protective impact of the extracts on erythrocytes, specifically their ability to mitigate damage caused by oxidative stress. This will be achieved by quantifying the fraction of degenerated erythrocytes after exposure to an explosive event. 90
The hemolysis inhibition assay highlights the protective role of Atriplex halimus extracts against oxidative damage in erythrocytes, demonstrating both concentration- and time-dependent activity that plateaued after approximately 20-25 min. 91 The positive correlation between extract concentration and hemolysis inhibition percentage underscores the capacity of bioactive compounds, particularly phenolics and flavonoids, to stabilize cell membranes and prevent oxidative lysis induced by free radicals. 92 Although the IC50 value was relatively high, the overall trend indicates that higher concentrations of the extract provide substantial protection, which is consistent with its strong antioxidant profile observed in radical scavenging assays. This suggests that the anti-hemolytic effect is mechanistically linked to the radical-scavenging and reducing power of the phenolic constituents. 93 Comparable findings have been reported for other medicinal plants, where phenolic-rich extracts mitigated erythrocyte damage by enhancing membrane resilience and neutralizing reactive oxygen species. Therefore, the ability of A. halimus to inhibit hemolysis further reinforces its therapeutic promise in protecting biological systems from oxidative stress–related cellular injury, with potential applications in managing hemolytic and degenerative disorders. 94
Antibacterial Activity (in Vitro)
This characteristic is likely due to the presence of hydrophobic lipopolysaccharide molecules in the membrane of Gram (-) germs, which hinders the diffusion of hydrophobic compounds.95,96 Furthermore, the periplasm contains enzymes that can eliminate foreign molecules introduced from outside, making these bacteria less susceptible to plant extracts than Gram + bacteria and that provides their protection against different agents.97,98 This confirms the work of, 99 which investigated the antibacterial activity of methanolic extracts of the aerial parts (stem and leaf) of A. halimus vis-à-vis sixteen bacterial strains (Staphylococcus MRSA, Staphylococcus aureus, Staphylococcus ATCC 00, Streptococcus Sp., Enterococci Sp., Enterococci feacalis ATCC 12, Salmonella, E. coli ESBL+, Klebsiella pn Marseille, KpC+, Kp ETP R/IMP R, Serratia Sp., Serratia environmental, Pseudomonas aerogenosa ATCC 53, Pseudomonas VIM 2, Bacillus). The findings show a remarkable antibacterial activity against Gram + bacteria compared to Gram- bacteria, the most sensitive germ is the Staphylococcus MRSA with diameters of 27.1 and 18.2 mm for the dilutions of the leaf extract 1/8 and 1/2 mg/ml respectively; followed by Staphylococcus ATCC 00 with diameters ranging from 10.2 to 15.2 mm for the range of dilutions of leaf extract 1/2 to 1/8 mg/ml.
Limitations of the Study
This study was limited to in-vitro evaluations and focused mainly on crude extracts of Atriplex halimus L. without identifying individual compounds. In addition, only a few bacterial strains were tested. While these aspects provide a solid first insight into the plant's bioactivity, further research should include compound isolation, in vivo studies, and a wider range of microbial targets to confirm and extend the present findings.
Conclusions
In conclusion, this study demonstrated that Atriplex halimus L. possesses strong antioxidant, antibacterial, and anti-hemolytic properties. Among the different extraction conditions tested, acetone acidified with 1 N HCl, an extraction time of 210 min, and a temperature of 37 °C proved to be optimal, yielding the highest phenolic and flavonoid contents as well as the strongest bioactivity. These optimized extracts also exhibited marked antibacterial effects, particularly against Gram-positive strains. Overall, the findings provide updated evidence supporting A. halimus as a promising natural source of bioactive compounds with potential applications in the pharmaceutical and nutraceutical fields.
Footnotes
Acknowledgments
The authors express their gratitude to all those who contributed to this work.
Author Contributions
Conceptualization: Abdelatif Aouadi; Methodology: Abdelatif Aouadi, Fatma Mohamed Abd El-Mordy, Djamila Hamada Saoud, Yasir A. A. Mustafa, Hakim Belkhalifa, Omar Berkane, Ayachi Amar Fatma Zohra, Soumia Hadjadj, and Mohammed Messaoudi;
Software: Abdelatif Aouadi, Djamila Hamada Saoud, Fatma Mohamed Abd El-Mordy, Omar Berkane, Soumia Hadjadj, Yasir A. A. Mustafa, and Mohammed Messaoudi;
Validation: Abdelatif Aouadi, Fatma Mohamed Abd El-Mordy, Djamila Hamada Saoud, Yasir A. A. Mustafa, Hakim Belkhalifa, Omar Berkane, Ayachi Amar Fatma Zohra, Soumia Hadjadj, Ayomide Victor Atoki, Amar Djemoui and Mohammed Messaoudi;
Writing – Original Draft: Abdelatif Aouadi, Fatma Mohamed Abd El-Mordy, Djamila Hamada Saoud, Yasir A. A. Mustafa, Hakim Belkhalifa, Omar Berkane, Ayachi Amar Fatma Zohra, Soumia Hadjadj, and Mohammed Messaoudi;
Writing – Review & Editing: Abdelatif Aouadi, Fatma Mohamed Abd El-Mordy, Djamila Hamada Saoud, Yasir A. A. Mustafa, Hakim Belkhalifa, Omar Berkane, Ayachi Amar Fatma Zohra, Soumia Hadjadj, Mohammed Messaoudi and Samira Choula.
All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Competing Interests
The authors declare no competing interests.
Institutional Review Board Statement
Not applicable.
Declaration of Conflicting Interests
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Data Availability Statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
