Abstract
Mortality due to microbial diseases continues to be a major problem in many developing countries. The present study aims to evaluate the antibacterial and antioxidant activities of the ethyl acetate extract and some isolated compounds from aerial parts of Echinops gracilis. The phytochemical study resulted in the isolation of a new flavonoid derivative named apigenin-7-O-(4″-feruloyl)-β-D-glucoside (
Infectious diseases caused by microbes are responsible for more deaths worldwide than any other single cause. Many microbes are developing new properties to resist drug treatments that once effectively destroyed them. Drug resistance has become a serious problem worldwide. In many regions affected by infectious diseases, local and indigenous plants are often the only available means of treating such infection. Plant natural products can also have antioxidant potential. These include phenolic compounds, alkaloids, terpenoids, and essential oils. Plant-based antioxidant compounds play a defensive role by preventing the generation of free radicals and hence are extremely beneficial to alleviate infectious diseases that generate free radicals as well as diseases caused by oxidative stress.
The genus Echinops,

Previous phytochemical studies on the roots of E. gracilis revealed the presence of erythrinasinate, vogelate, ferulic acid, p-coumaric acid, ursolic acid, oleanolic acid, and quercetin. Moreover, the anti-inflammatory activity of E. gracilis extracts and some isolated compounds has been established.
10
We herein report the isolation and structural elucidation of one new flavonoid derivative; apigenin-7-O-(4”-feruloyl)-β-D-glucoside (
Results and Discussion
The ethyl acetate fraction of the methanol extract of E. gracilis aerial parts was subjected to open column chromatography (CC) over silica gel to give compound
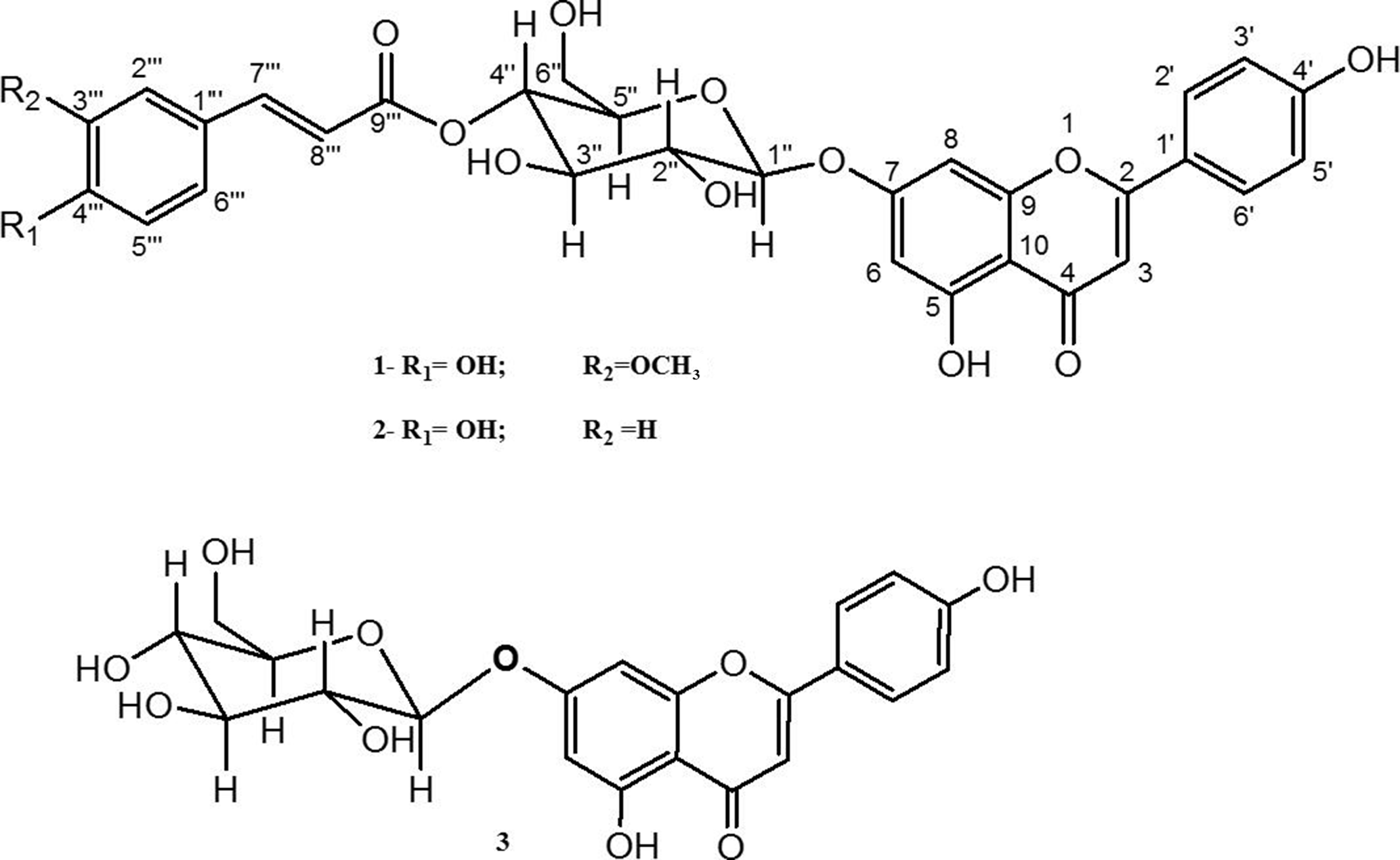
Structures of isolated compounds.
Compound
The 1H NMR signals at δ 12.99 and 9.70 ppm, in addition to their HMBC correlations, indicated the presence of chelated C-5 and free C-4″′ hydroxyl groups in the cinnamoyl moiety. The HMBC correlations unambiguously confirmed a linkage between the anomeric proton, H-1′′ of the glucose moiety and C-7 (δ 162.8 ppm) (Table 1, Figure 2). Furthermore, additional HMBC correlations between H-4″ (δ 4.79 ppm) and feruloyl carbonyl, C-9′″ at δ 166.0 ppm revealed that the feruloyl moiety is attached at position 4′′ (Figure 2). These correlations are further supported by the NOESY spectrum, which showed correlations between H-4′′ and the olefinic proton in the β position of carbonyl, C-9′″ (δ 166.0 ppm). All these NMR data are close to those of apigenin 7-O-(4″-trans-p-coumaroyl)-β-D-glucoside (
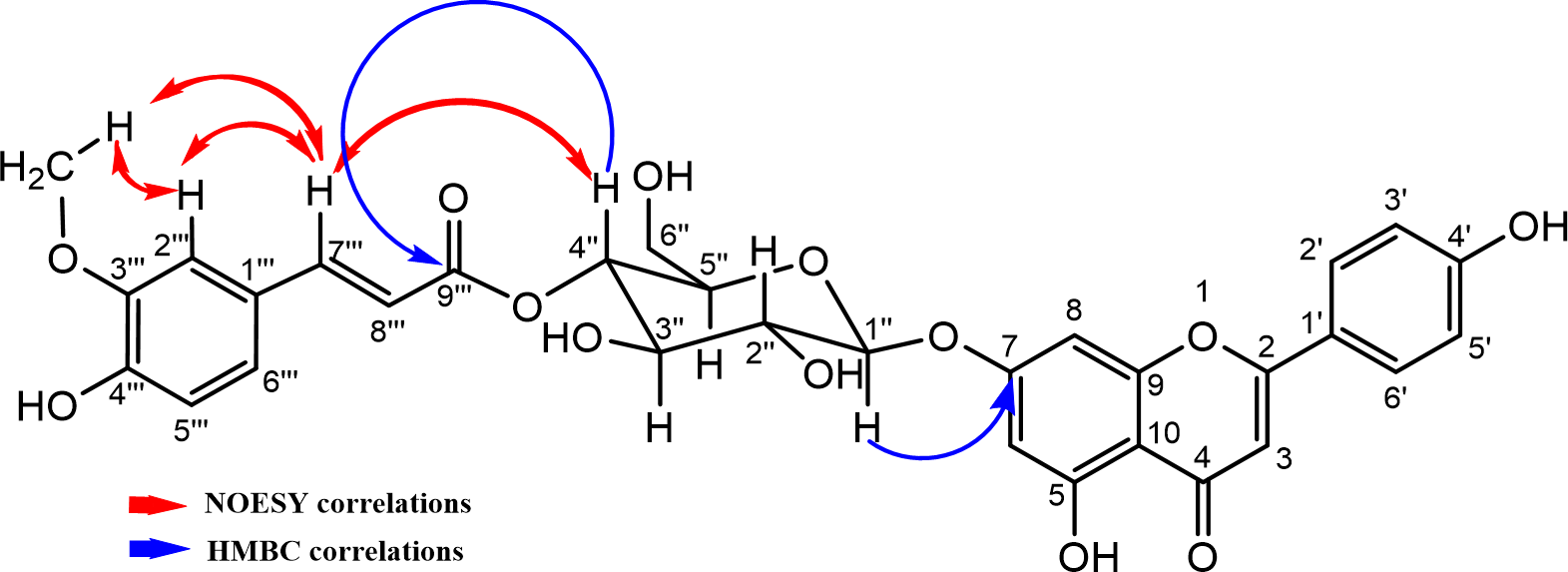
Some HMBC and NOESY correlations in compound 1.
13C NMR (100 MHz, DMSO-D6) and 1H NMR (400 MHz, DMSO-D6) Spectroscopic Data of Compound 1 (δ in ppm; J in Hz).

The antioxidant activities of the EtOAc extract and isolated compounds (
The ethyl acetate extract, along with compounds
Experimental
General Experimental Procedures
Electrospray ionization mass spectra (ESIMS) were recorded on a QSTARXL of AB Sciex Company, UV and visible spectra, recorded in MeOH at 25 °C, on a Kontron Uvikon spectrophotometer, IR spectra on a FT PerkinElmer 1750 FTIR spectrometer, and NMR spectra on a Bruker 400 MHz NMR Avance II spectrometer equipped with a cryoprobe, with TMS as internal reference. Chemical shifts were recorded in δ (ppm) and the coupling constants (J) are in Hertz. Silica gel 60 F254 (70-230; Merck; Darmstadt, Germany) was used for column chromatography. Precoated silica gel Kieselgel 60 F254 plates (0.25 mm thick) were used for TLC, and compounds were detected by spraying with 50% H2SO4 followed by heating at 100 °C. All solvents were distilled before use. Optical density values were determined on a Thermo-Fisher-Scientific: Evolution 300 UV-VIS.
Plant Material
Aerial parts of Echinops gracilis were collected from the Fongo-Tongo Leweh neighborhood in the West Region of Cameroon during February, 2014. A voucher specimen (No 66943/HNC Cam), authenticated by ethnobotanist Dr. Tsabang Nole, is located at the Cameroon National Herbarium, Yaoundé.
Extraction and Isolation
Dried plant powder (3 kg) was soaked in 12 L of methanol (MeOH) for 72 hours at room temperature to yield the crude extract (190 g), after evaporation under vacuum. This extract was subjected to liquid-liquid separation with a mixture of n-hexane (0.5 L × 4) and MeOH 80% (1L). The phase made up of 80% MeOH was treated with ethyl acetate (EtOAc) (0.5 L × 4) to afford 19.5 g of EtOAc extract. The extract was subjected to open column chromatography (CC) over silica gel, eluting with a DCM-MeOH mixture of increasing polarity (from 30:1 to 1:1). Eighty-eight fractions (250 ml each) were collected and grouped into 5 major fractions [
ABTS Radical Cation Decolorization Assay
The method used to determine the antioxidant activities is based on following the discoloration kinetics of the ABTS+
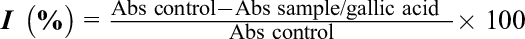
where, Abscontrol is the absorbance of control and Abssample/gallic acid is the absorbance of the sample or gallic acid.
Ferric Ions (Fe3+) Reducing Antioxidant Power Assay
The ferric reducing antioxidant power assay (FRAP) is based on the reduction of the tripyridyltriazine ferric complex (Fe3+-TPTZ) to the tripyridyltriazine ferrous complex (Fe2+-TPTZ) in the presence of an antioxidant. To 2000 µL of FRAP in test tubes were added 75 µL of either extracts or gallic acid at different concentrations. 21 The tests were performed in triplicate, and the mixture was incubated for 30 minutes. The optical density was measured at 593 nm. The FRAP solution (pH 3.6) was prepared as follows: 14.1 mg of TPTZ was diluted in 9 ml 40 mM HCl, then ferric chloride (FeCl2 20 mM) and acetate buffer (300 mM, pH = 3.6) were mixed in the ratio of 1:1:10, respectively to form the FRAP solution.
In Vitro Antibacterial Activity
Clinical isolated
Solutions of the ethyl acetate extract and the isolated compounds were prepared in concentrations of 30 mg/mL and 1 mg/mL, respectively, in sterile distilled water to final volumes of 1 ml. Each solution was homogenized until clear solutions were obtained.
A microplate containing 96 wells was used for this study. 100 μL of Muller Hinton liquid medium supplemented with 10% glucose was introduced into each well. 100 μL of the stock solution of the ethyl acetate extract to be tested, prepared at 30 mg/mL, was introduced into the first term wells. An identical volume of the 1 mg/mL isolated compounds/levofloxacin solution was used. Final testing concentrations ranges were 7500 µg/mL to 7.32 µg/mL (for ethyl acetate extract) and 250 µg/mL to 0.488 µg/mL (for isolated compounds and levofloxacin). A volume of 100 µL of concentrated bacterial inoculum at 1.5 × 108 CFU/mL was introduced into each well to a final density of 7.5 × 107 CFU/mL. Thus, the final volume was 200 µL per well and all the tests were conducted in triplicate. In parallel, a third series of wells was used as a negative control containing only the culture medium and the sample, and other wells were used as a positive growth control and containing the culture medium and inoculum. The microplate was incubated at 37 °C for 18 to 24 hours. The color change was developed by adding a few drops of Alamar blue (Centre Pasteur; Cameroon) to each well and then incubating for 30 minutes. The change in coloration of the wells from blue to pink indicated bacterial growth. The MIC is defined as the lowest concentration of extract that inhibited bacterial growth visible to the naked eye. 22
For MBC, 50 µL from each well with a concentration greater or equal to the MIC was taken and added to 150 µL of broth. The microplate was incubated for 18 to 24 hours and then developed with Alamar blue. The MBC was considered to be the cup with the lowest concentration of extract that did not change color. 22
Supplemental Material
Supplementary Material 1 - Supplemental material for Antibacterial and Antioxidant Activities of the Extract and Some Flavonoids From Aerial Parts of Echinops Gracilis O. Hoffm. (Asteraceae)
Supplemental material, Supplementary Material 1, for Antibacterial and Antioxidant Activities of the Extract and Some Flavonoids From Aerial Parts of Echinops Gracilis O. Hoffm. (Asteraceae) by Fidèle Castro Weyepe Lah, Marius Balemaken Missi, Natasha October, Patrick Herve Betote Didoue, Nalova Hermia Nalova Ikome, Jean Pierre Abdou, Theodora Kopa Kowa, Gabriel Agbor, Alembert Tiabou Tchinda and Etienne Dongo in Natural Product Communications
Footnotes
Acknowledgments
The authors are grateful to TWAS-ICCBS for the Sandwich Postgraduate fellowship offered to AJP (FR No 3240275059) at the International Center for Chemical and Biological Sciences (ICCBS) of the University of Karachi, Karachi-75270, Pakistan. We also gratefully acknowledge the Belgian National Fund for Scientific Research (FNRS) for a fellowship to A.T.T.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
