Abstract
Introduction
Reactive oxygen species (ROS) were molecules or ions with reactive physical and chemical properties, including hydrogen peroxide (H2O2), hydroxyl radical (·OH) and superoxide anion radical (O2.−), etc 1 Reactive oxygen species was involved in various biological processes within living organisms. However, excess ROS causes cell damage, tissue damage, and oxidative stress, which can lead to cancer, cardiovascular disease and neurological diseases. 2
Antioxidants were substances that scavenge free radicals to prevent oxidative damage to cells and tissues or increasing the activity and number of antioxidant enzymes in organisms through a variety of pathways. 3 Antioxidants were divided into inartificial (eg, natural plants) 4 and synthetic (eg, Tert-butyl-p-cresol (BHT) and Metal chelators) antioxidants. 5 However, natural antioxidants were safer compared to synthetic antioxidants. 6 Now, antioxidants were increasingly being used to combat disease. 7
Lotus leaf belongs to the Nelumbo nucifera Gaertn, which is rich in resources in the world and has extensive medicinal values. Nuciferine was an aporphine alkaloid and one of the main components of lotus leaf.
8
It has the effect of reducing blood lipids,
9
anti-inflammatory
10
and antioxidant.
11
However, there has not been much research on the antioxidant capacity of nuciferine
Chemical antioxidant method refers to the method in which the antioxidant capacity of a substance was manifested in scavenging free radicals, inhibiting pro-oxidants and reducing power, etc It was currently the main means to assess the antioxidant activity of substances, as well as it has the advantages of simplicity and speed.
13
Cellular antioxidant method was an
Method
Reagents and Apparatus
Lotus leaf (Nelumbo nucifera Gaertn) were purchased from Lu’an Danbeier Biotechnology Co., Ltd, and the year of collection is 2020. Lotus leaf extract (containing nuciferine about 67%) was derived from “Optimizing Ultrasonic Extraction and Purification of nuciferine With Response Surface Method” (experimented, written, and published by the authors). The data for the purity characterization of lotus leaf extract were in Ref. 12
Nuciferine reference (98%) was purchased from Chengdu Lemeitian Pharmaceutical Technology Co., Ltd RAW264.7 cell (No. TCM13) were obtained from the Cell Bank of the Chinese Academy of Sciences. Additional reagents were placed in Supplementary Table 1 (Table S1). Preparation of all solutions was in Supplementary Table 2 (Table S2).
Quartz enzyme label plate was purchased from Shanghai Baiqian Biotechnology Co., Ltd Microplate reader (Molecular Devices iD5) was purchased from Annolen Biotechnology Co., Ltd The cell culture incubator was obtained from Shanghai Hengyi Scientific Instrument Co., Ltd The microscope was purchased from Olympus. Biosafety cabinet was purchased from BIOBASE. Cryogenic high-speed centrifuge was purchased from Eppendorf.
Chemical Antioxidant Method
Hydroxyl Radical (·OH) Scavenging Ability
The salicylic acid means can evaluate the scavenging ability of the target substance to ·OH free radical. 13 Different concentrations of lotus leaf extract and nuciferine reference solution (0.05-5.0 mg/mL) were made, and 50 μL of this solution was placed into a 96-well plate. Next, 50 μL 9 mmol/L FeSO4 (Iron(II) sulfate) solution was added, 50 μL 6 mmol/L H2O2 was added and left at normal temperature for 10 min, and 50 μL 9 mmol/L salicylic acid solution was kept at normal temperature for 30 min. The absorbance at 510 nm was surveyed with an enzyme-labeled instrument. L-ascorbic acid was applied as the control. Ultrapure water was used as the blank.
Based on clearance curve, the scavenging ability of lotus leaf extract to ·OH free radicals was analyzed. Each sample was measured in triplicate copies. The scavenging rate formula was as follows: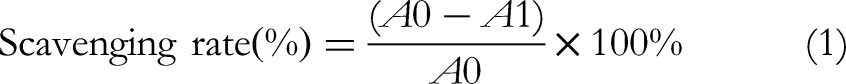
Among them,
Superoxide Anion Radical (O2.−) Scavenging Ability
Pyrogallol method was used to determine the scavenging ability of the target substance to O2.− free radical.15-17 Different concentrations of lotus leaf extract and nuciferine reference solution (0.05-5.0 mg/mL) were made, and 50 μL of this solution was placed into a 96-well plate. Then, 135 μL 50 mmol/L Tris-HCl (pH 8.2) buffer solution was added and reacted at 37 °C for 15 min. Next, 15 μL of 25 mmol/L pyrogallol solution was added, and the reaction was terminated with 50 μL of 8 mol/L HCl after 4 min at normal temperature. After standing for 30 min, the absorbance at 299 nm was surveyed with an enzyme-labeled instrument. The control and blank were the same as in “
The calculation and clearance curve were the same as “
DPPH Radical Scavenging Ability
The DPPH way can estimate the antioxidant activity of substances
According to the clearance curve, the scavenging ability of lotus leaf extract on DPPH radical was analyzed. Each sample was measured in triplicate copies. The scavenging rate formula was as follows:
Among them,
ABTS•+ Radical Scavenging Ability
The ABTS method can monitor the antioxidant capacity of a substance. 19 Different concentrations of lotus leaf extract and nuciferine reference solution (0.05-5.0 mg/mL) were made, and 50 μL of this solution was placed into a 96-well plate. Then, ABTS•+ and K2S2O8 (Potassium persulfate) mixtures was added, mixed well and left to stand for 15 min at normal temperature. Absorbance at 734 nm was determined with an enzyme-labeled instrument. L-ascorbic acid was applied as the control. Methanol added to ABTS•+ and K2S2O8 mixtures was blank 1, and methanol plus purified water was blank 2.
The calculation and clearance curve were the same as “
Determination of Ferric Reducing Ability of Plasma (FRAP)
FRAP was capable of analyzing the iron ion reduction capacity of a substance. 19 Different concentrations of lotus leaf extract and nuciferine reference solution (0.05-5.0 mg/mL) were made, and 20 μL of this solution was placed into a 96-well plate. Next, 200 μL FRAP mixtures was added, mixed well, and let stand at 37 °C for 20 min. The absorbance was determined with an enzyme-labeled instrument at 593 nm. BHT was the control. Methanol was blank.
According to FRAP curve, the reducing ability of lotus leaf extract was analyzed. Each sample was measured in triplicate copies. FeSO4 equivalent (FRAP value) was calculated based on a FeSO4 standard curve. The formula for FRAP value was as follows:
Among them,
The FeSO4 standard curve of concentration-absorbance was drew with the concentration (0.05-1.2 mmoL/L) and (
Cellular Antioxidant Method
Cell culture. 20
RAW264.7 cells were fostered with DMEM (adding 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin bispecific antibody) under conditions of 37 °C and 5% CO2. During cell culture, the cells were fed once every 24 h and passaged once every 48 h, and the experiment was carried out with the cells of logarithmic growth stage.
Establishment of Cell Damage Models Using H2O2
Based on relevant literature reports, an H2O2-induced cell injury model was established.
20
RAW264.7 cells were inoculated on a 96-well plate at a density of 2.0 × 105/mL and cultured under 37 °C and 5% CO2 for 24 h. The media were replaced with fresh DMEM including different concentrations of H2O2 (0-2000 μmol/L) (dissolved in DMSO) and incubation for 24 h. Then, the supernatant was discarded, 100 μL CCK8 solution was added and cultured for 40 min. Next, the cell culture was stopped, and the supernatant was removed to determine the absorbance at 450 nm. Each concentration was provided with 3 multiple holes. Cell viability was analyzed based on Formula 4 as follows:
Among,
Concentration Screening of Lotus Leaf Extract
The experimental process of screening was the same as “
Determination of MDA, Total GSH, SOD, and CAT Levels in Cells
Antioxidant enzymes were quantified with reference to relevant literature.20-22 2 mL RAW264.7 cells with density of 2.0 × 105/mL were added to each well of the 6-well plate and cultivated at 37 °C and 5% CO2 for 24 h. After discarding the culture medium, tests were carried out according to Table 1.
Experiment Operations.

Protein isolation refers to the isolation of proteins from experimental cells. The cells treated in Table 1 were placed in an ice bath and immediately added 250 μL RIPA lysis buffer for 10 min to completely lysate the cells. The fully lysed cells were then placed in a 4 °C centrifuge at a speed of 12 000 r/min for 15 min to obtain the isolated proteins. The protein was quantified by BCA protein quantitation kit. Activity of MDA, total GSH, SOD and CAT was determined according to the method of the corresponding kit.
Statistical Methods
ANOVA was used for between-group differences. ANOVA for all cell data was provided in Supplementary Table 3 (Table S3). The average ± SEM of the three independent experiments was applied to represent the experimental data. The
Results
Chemical Antioxidant Activity
Hydroxyl Radical (·OH) Scavenging Activity
The results of scavenging ability of lotus leaf extract, nuciferine reference and l-ascorbic acid to ·OH radical were shown in Figure 1A. As the concentrations of lotus leaf extract, nuciferine reference and l-ascorbic acid increase, their scavenging activity gradually fortifies until a plateau was reached. The scavenging activity of lotus leaf extract and nuciferine reference to ·OH radicals was above l-ascorbic acid in the range of 0-1.0 mg/mL, but below it in the range of 1.5-5.0 mg/mL. This illustrates that lotus leaf extract and nuciferine reference have good ·OH radical scavenging capacity in the range of 0-5.0 mg/mL.

Chemical Antioxidant Activity. (A) Scavenging Activity of ·OH Radical. (B) Scavenging Activity of O2.− Radical. (C) Scavenging Activity of DPPH Radical. (D) Scavenging Activity of ABTS•+ Radical. (E) FeSO4 Standard Curve (0.05, 0.10, 0.20, 0.40, 0.60, 0.80, 1.20 mmol/L). (F) Ferric Reducing Ability of Plasma. The Concentration of lotus Leaf Extract, Nuciferine Referencer, l-Ascorbic Acid and BHT was all 0.05-5.0 mg/mL (0.05, 0.1, 0.25, 0.5, 1.0, 1.5, 2.0, 2.5, 5.0 mg/mL). The Average ± SEM of the Three Independent Experiments was Used to Represent the Experimental Data.
Superoxide Anion Radical (O2.−) Scavenging Activity
As shown in Figure 1B, the scavenging capacity of lotus leaf extract and nuciferine reference on O2.− radical was lower than that of l-ascorbic acid. With the increase of the concentrations of lotus leaf extract, nuciferine referencer and l-ascorbic acid, their scavenging rate rised until they reached a plateau. This illustrates that the O2.− radical scavenging ability of lotus leaf extract and nuciferine referencer was better, and it was close to l-ascorbic acid.
DPPH Radical Scavenging Activity
In Figure 1C, the DPPH radical scavenging capacity of l-ascorbic acid was better than that of lotus leaf extract and nuciferine referencer. Scavenging activity of lotus leaf extract, nuciferine referencer and l-ascorbic acid against DPPH radical augments with increasing concentration. Their scavenging activity was almost equivalent at a concentration of 5 mg/mL. The above results indicate that lotus leaf extract and nuciferine referencer have good DPPH radical scavenging ability.
ABTS•+ Radical Scavenging Activity
From Figure 1D, we can obtain results that the scavenging ability of lotus leaf extract and nuciferine referencer on ABTS•+ radical was weaker than that of l-ascorbic acid. With the increase of concentration, the scavenging rate of lotus leaf extract, nuciferine referencer and l-ascorbic acid rises until they reach a plateau of 2.5 mg/mL, at which they had almost the same scavenging activity on ABTS•+ radical. This illustrates that the ABTS•+ radical clearance capacity of lotus leaf extract and nuciferine referencer was better and close to l-ascorbic acid.
Ferric Reducing Ability of Plasma (FRAP)
Different concentrations of FeSO4 solution were configured. The standard curve obtained was shown in Figure 1E. The linear equation was y = 0. 66116x − 0.00173 and the correlation coefficient was 0.9997 (R = 0.9997).
As shown in Figure 1F, the FRAP value of lotus leaf extract and nuciferine referencer was significantly lower than that of BHT. With the augment of concentration, the FRAP value in lotus leaf extract and nuciferine referencer hardly increases, but BHT augments substantially. This indicates that the FRAP of lotus leaf extract and nuciferine referencer was weak.
Cellular Antioxidant Activity
600 μmol/L H2O2 as the Modeling Concentration of Cell Damage
H2O2 can induce oxidative stress in cells, so it was used to construct oxidative damage models in RAW264.7 cells. In Figure 2A, cell viability gradually decreases as the concentration of H2O2 increases. Different concentrations of H2O2 inhibited cell activity. If the concentration of H2O2 was too high, it will cause a large number of cell death, and if it is too low, the cell damage will not be obvious. Therefore, the concentration of H2O2, which has a cell viability of about 50%, was selected for the construction of the oxidative damage model. In this experiment, 600 μmol/L H2O2 (cell activity 57.65%) was selected as the modeling concentration.

Effects of H2O2 and Lotus Leaf Extract on the Activity of RAW264.7 Cells. (A) The Effect of Different Concentrations of H2O2 (0, 200, 400, 600, 800, 1000, 1200 μmol/L) on the Activity of RAW264.7 cells. (B) The Effect of Different Concentrations of Lotus Leaf Extract (1, 10, 25, 50, 100, 150, 200, 500, 1000, 2000, 5000 μg/mL) on the Activity of RAW264.7 Cells. The Average ± SEM of the Three Independent Experiments was Used to Represent the Experimental Data.
Screening Results of Concentration of lotus Leaf Extract
Figure 2B shows that cell viability first increases and then decreases as the concentration of lotus leaf extract rises. The concentration of lotus leaf extract at 1-200 μg/mL promotes cell growth and inhibits cell growth at 200-5000 μg/mL. Therefore, concentrations that significantly promoted cell growth were selected for the low-, medium- and high-dose. The concentrations in the low-, medium- and high-dose were 50, 100, and 150 μg/mL, respectively. Due to the two-way regulatory effect of lotus leaf extract, its EC50 was 78.4 μg/mL (95%CI 65.2-94.1) and its IC50 was 1240 μg/mL (95%CI 980-1570) (n = 3).
Evaluation of Lipid Peroxidation and Antioxidant Enzyme Activity of lotus Leaf Extract
To study the antioxidant activity of lotus leaf extract

Evaluation of Lipid Peroxidation and Antioxidant Enzyme Activity of lotus Leaf Extract. (A) The Effect of lotus Leaf Extract on the Cellular Concentration of MDA. (B) The Effect of lotus Leaf Extract on the Activity of Total GSH. (C) The Effect of lotus Leaf Extract on the Activity of SOD. (D) The Effect of lotus Leaf Extract on the Activity of CAT. The Average ± SEM of the Three Independent Experiments was Used to Represent the Experimental Data. *
In Figure 3A, there was a significant difference in MDA levels between the blank and model, suggesting that ROS caused lipid peroxidation to occur dramatically (
Figure 3B shows that there was a significant difference in total GSH content between the blank and the model, indicating that ROS inhibits the production of antioxidant enzymes (
As shown in Figure 3C, there was a significant difference in SOD levels between the blank and model (
As we can see from Figure 3D, there was a significant difference in CAT content between the blank and model. Activity of CAT was significantly increased in the low-, medium- and high-dose. Among them, CAT activity was almost identical in the medium- and high-dose. The CAT level in the dose was slightly lower than in the control. The above results suggest that lotus leaf extract protects cells from ROS damage by augmenting the activity of antioxidant enzymes (total GSH, SOD, and CAT).
Discussion
The antioxidant activity of lotus leaf extract (containing nuciferine about 67%)
·OH radical was reactive oxygen species that interact with salicylic acid to produce a substance with strong UV absorption at 510 nm. When antioxidant was present, this colored substance decreases due to the disappearance of ·OH radical. 13 In this paper, the scavenging activity of lotus leaf extract and nuciferine reference against ·OH radical was significant, and the scavenging activity fortified with the increase of concentration in the range of 0.05-5.0 mg/mL. O2.− radical was also a reactive oxygen species. Pyrogallone produces O2.− radical under weakly alkaline conditions, which promotes the formation of a substance with strong UV absorption at 299 nm. When antioxidant was added, it hinders the formation of this colored substance. 15 In this study, lotus leaf extract and nuciferine reference had a better clearance of O2.− radical and were close to l-ascorbic acid. The DPPH means was often applied to analyze the antioxidant activity of a substance. There were lone electron pairs in DPPH radical, and there was strong UV absorption at 517 nm. When an antioxidant was present, it binds to lone electron pairs to attenuating UV absorption. 19 In this experiment, lotus leaf extract and nuciferine reference had good DPPH radical scavenging capacity, and the scavenging activity was close to that of l-ascorbic acid. The ABTS•+ means was applied to analyze the antioxidant activity of substances. The ABTS•+ radical have strong UV absorption at 734 nm. As antioxidant were added, their interaction with ABTS•+ weakens UV absorption. 19 In this study, lotus leaf extract and nuciferine reference were highly effective in scavenging ABTS•+ radical, and had almost the same scavenging capacity as l-ascorbic acid in the range of 2.0-5.0 mg/mL. The FRAP way can be used to analyze the reducing capacity of antioxidant. This method was based on Fe2+ and Tripyridyltriazine (TPTZ) to produce colored substances with UV absorption at 593 nm. Antioxidant can reduce these colored substances to reduce UV absorption. 19 In this experiment, the reducing ability of lotus leaf extract and nuciferine reference was weaker and significantly weaker than that of BHT.
Alkaloids in lotus leaves have been shown to protect against ethanol-induced liver injury by scavenging ROS.23,24 In this experiment, lotus leaf extract in the low and medium dose could clear ROS, but the scavenging effect of ROS in the high dose was weaker than that in the medium dose. MDA was a lipid peroxidation product that can reflect the degree of lipid peroxidation. 25 The level of MDA can be significantly decreased in low, medium and high dose. GSH removes O2.− radical and H2O2 to reduce oxidative stress. 26 SOD was capable of converting O2.− radical to molecular oxygen (O2) or H2O2. 27 CAT can dissociate H2O2 into molecular oxygen (O2) and water. 28 In this paper, antioxidant enzyme (total GSH, SOD, and CAT) activity can be significantly elevated in the low-, medium-, and high-dose. Among them, the middle-dose had the strongest antioxidant capacity compared with the low- and high-dose. In this paper, we report a dose-dependent stimulation effect at low concentrations (1-200 μg/mL) (EC50 = 78.4 μg/mL) and inhibition at high concentrations (>200 μg/mL) (IC50 = 1240 μg/mL). This bidirectional regulatory pattern suggests that lotus leaf extract may affect target cell activity through different mechanisms. However, the current calculation of the IC50 is based on a four-parameter model of the inhibition phase, and the bidirectional effect may lead to the IC50 not being fully applicable. Therefore, future studies may need to detect cell cycle arrest at 200 μg/mL turning points and expand the concentration gradient (50-500 μg/mL) to improve accuracy.
This study evaluated the
Comparison of the Antioxidant Capacity of Different lotus Leaf Extracts.

The antioxidant capacity of lotus leaf extract was compared with other plants, and the results are shown in Table 3. Compared with the other plants in Table 3, there was a lack of reference substance in this paper. We used only l-ascorbic acid and BHT as control experiments. However, the effects of the extract on the activity of various free radicals and antioxidant enzymes are studied in detail in this paper. Although lotus leaf extract did not exhibit better antioxidant capacity than the control, its non-toxic and harmless properties were also advantages. In the world, lotus leaf resources are extensive, and it can become a potential object of industrial production.
Comparison of the Antioxidant Capacity of Different Plants.

Lotus leaves are abundant in Asia, providing a foundation for industrial applications. Based on this study on the
The limitations of this article are that it does not investigate the effects of different concentrations of nuciferine and lotus leaf extracts on free radical scavenging capacity and cellular antioxidant capacity. The sample size of this study was insufficient. This study only studied ·OH and O2.− radical, and did not fully consider the effects on other oxygen free radicals (such as hydrogen peroxide, singlet oxygen, and peroxide lipids, etc). Furthermore, the article does not analyze the specific components contained in the lotus leaf extract, and only nuciferine was quantified by high performance liquid chromatography. These will be the focus of subsequent research.
Conclusion
This study systematically evaluated the
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251349923 - Supplemental material for Study on Antioxidant Activity of Nuciferine of Lotus Leaf in Vitro
Supplemental material, sj-docx-1-npx-10.1177_1934578X251349923 for Study on Antioxidant Activity of Nuciferine of Lotus Leaf
Footnotes
Acknowledgements
We gratefully acknowledge the financial support from the National Natural Science Fund of China (No.82273687). We also extend our sincere appreciation to Shanghai University of Traditional Chinese Medicine and Shanghai University of Medicine & Health Sciences for their academic nurturing. Special thanks to Professor Hongzhi Pan for his invaluable guidance.
Author Contributions
Mengdie Wu was in charge of experiments, collecting data and writing manuscripts. Tao Han was responsible for drawing, data analysis and reviewing the manuscript. Hongmin Gao, Yang Zhou and Huan Zhu were responsible for data analysis and investigation. Hongzhi Pan was responsible for reviewing, supervising, managing projects, and providing funding. All authors support the publication of the article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by National Natural Science Fund of China (No.82273687).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
