Abstract
Objective
This study aimed to evaluate the phenolic and flavonoid composition of the hydro-ethanolic extract of
Methods
Dried
Results
The results that the extract contained high levels of total phenolics and flavonoids, HPLC-DAD identified major compounds such as gallic acid and quercetin. The extract demonstrated potent antioxidant activity (IC50 = 17.24 ± 0.01 µg/mL), which was less effective than that of standard ascorbic acid. However, it showed superior α-amylase inhibitory activity compared to acarbose (IC50 = 168.32 ± 1.12 vs 299.26 ± 17.81 µg/mL). Additionally, the extract exhibited moderate anti-inflammatory activity (IC50 = 459.14 ± 27.49 µg/ml) compared to aspirin and moderate anti-hemolytic activity compared to vitamin E, antibacterial activity was more pronounced against Gram-positive strains.
Conclusion
The findings highlight the therapeutic potential of
Keywords
Introduction
Medicinal plants have constituted a cornerstone of human healthcare since antiquity, valued for their therapeutic efficacy, accessibility, and relatively low side effects. 1 In recent decades, scientific research has shown renewed interest in investigating the bioactive constituents of these plants and their associated biological activities. 2 These natural resources represent a vast reservoir of secondary metabolites, such as alkaloids, terpenes, phenolics, flavonoids, and essential oils, 3 many of which exhibit a broad spectrum of pharmacological properties, including antidiabetic, anti-inflammatory, anticancer, antioxidant, and antimicrobial effects. 4 This highlights the necessity of systematic scientific studies to elucidate their chemical profiles and harness their potential for developing safe and effective pharmaceuticals. 5
The health-promoting properties of
The antioxidant potential of phenolic compounds is intrinsically linked to their structural features, particularly the number and position of hydroxyl (-OH) groups on aromatic rings. These molecules act as hydrogen or electron donors to neutralize reactive oxygen species (ROS), such as superoxide and hydroxyl radicals), thereby stabilizing their structures and reducing oxidative stress, a major factor in the pathgenesis of cancer and hypertension.10–12 The chemical profile of plant-derived extracts can vary considerably depending on factors such as species origin, extraction method (eg, hydro-ethanolic extraction), temperature, and solvent concentration. 13
Hydro-ethanolic extraction (ethanol-water mixture) is widely recognized as one of the most effective techniques for isolating polyphenols and flavonoids given its ability to solubilize both polar and semi-polar constituents.
14
Several studies have demonstrated that hydro-ethanolic extraction of
Analytical investigations employing high-performance liquid chromatography coupled with diode-array detection (HPLC-DAD) have revealed considerable variation in the polyphenolic profile of ethanolic,
16
hydroethanolic,14–17 methanolic,
18
and aqueous,
9
extracts of
The present study aims to investigate the biological potential of
Materials and Methods
Chemicals and Reagents
This study employed various analytical-grade chemicals and reagents, used without further purification. The material includes ascorbic acid (AA), 2,2-diphenyl-1-picrylhydrazyl (DPPH•), absolute ethanol, ethanol 96%, Folin–Ciocalteu reagent, sodium carbonate (Na2CO3), aluminum chloride (AlCl3), methanol, ethylene diamine tetra acetic acid (EDTA), phosphate-buffered saline (PBS), quercetin (Q), petroleum ether, gallic acid (GA), formic acid, phosphate buffer, sulfuric acid (H2SO4), double-distilled water (DD), ammonium molybdate, sucrose solution, hydrogen peroxide (H2O2), acetonitrile, vitamin E (tocopherol), gentamicin, bovine serum albumin (BSA), distilled water, sodium phosphate, dimethyl sulfoxide (DMSO), aspirin, α-amylase, hydrochloric acid (HCl), sodium chloride (NaCl), acarbose, iodine solution, and starch.
Collection and Preparation of Psidium guajava Leaves
Leaves of
Preparation of Hydro-Ethanolic Extract
A total of 20 g of powdered plant material was accurately weighed to initiate the extraction process with a hydro-ethanolic solvent. The sample was first defatted by immersion in petroleum ether under magnetic stirring for 24 h at ambient temperature. The suspension was then filtered using Whatman No. 1 filter paper . The filtrate was discarded, while the solid residue was retained for further use. The defatted plant residue was subsequently macerated in 200 mL of an ethanol–water mixture (1:1, v/v) and subjected to constant agitation at room temperature for an additional 24 h. Filtration was performed again using Whatman No. 1 filter paper. This extraction procedure was performed twice using fresh solvent each time.
All resulting filtrates were combined and concentrated using a rotary evaporator. The concentrated extract was subsequently dried in a hot-air oven at 45 °C overnight. Finally, the dried material was preserved in a refrigerator maintained at 4 °C until further use.
The employed extraction method produced a yield of 29.258% relative to the dry leaf powder weight, calculated as follows:
HPLC Analysis of Phenolic Compounds
The detection and characterization of phytochemical constituents were carried out using HPLC-DAD on an Agilent 1200 series system outfitted with a Kinetex Evo C18 analytical column. Initially, standard compounds were first injected to determine their retention times under the specified chromatographic settings. Subsequently, a 20 µL aliquot of the plant extract was analyzed under the same chromatographic conditions, including a mobile phase flow rate set at 1 mL/min, a detection wavelength fixed at 324 nm, and a column temperature maintained at 30 °C. The chromatographic peaks were compared with those of the reference standards, enabling both qualitative and quantitative assessment of phenolic constituents present in the sample, as presented in Table 1. 21
Experimental Conditions for the Separation of Phenolic Compounds in HPLC-DAD Analysis.

The HPLC method was validated, demonstrating excellent linearity (R2 > 0.999) over the tested concentration range, high repeatability (RSD < 5%), and recovery rates of 80–90%, confirming analytical accuracy and reliability of the method.
Determination of Total Phenolic Content (TPC)
The TPC was quantified employing the Folin–Ciocalteu colorimetric assay with slight modifications based on the procedure reported by Chihi et al 22 Briefly, 1 mL of each appropriately diluted extract was combined with 0.5 mL of Folin–Ciocalteu reagent, previously diluted 1:10 with distilled water. After allowing the mixture to react for 5 min, 2 mL of sodium carbonate solution (7.5%, w/v) was added. The mixture was then incubated in the dark at room temperature for 30 min. Absorbance was subsequently measured at 765 nm using a UV-Vis spectrophotometer. A calibration curve was established using gallic acid in the concentration range of 0.006 to 0.05 mg/mL. The phenolic content of the extracts was quantified as milligrams of gallic acid equivalents per gram of extract (mg GAE/g extract).
Determination of Total Flavonoid Content (TFC)
The TFC was assessed following a modified version of the aluminum chloride colorimetric method according to the procedure reported by Oliveira et al 23 In brief, 0.5 mL of each diluted plant extract was combined with an equal volume (0.5 mL) of 2% (w/v) aluminum chloride solution prepared in absolute ethanol. The reaction mixture was allowed to stand in the dark for 45 min at ambient temperature. The absorbance was measured at 415 nm using a spectrophotometer. Quercetin served as the standard reference compound over a concentration range of 0.005–0.035 mg/mL. Results are presented as milligrams of quercetin equivalents (mg QE/g extract).
Antioxidant Activity
DPPH Assay
The antioxidant capacity of the hydro-ethanolic extract was evaluated using the DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging assay following the method described by Aqil et al 24 with minor modification. A stock solution of DPPH was freshly prepared by dissolving 4 mg of the violet crystalline compound in 50 mL of methanol, ensuring complete dissolution to obtain a uniform deep-purple solution. Ascorbic acid served as the positive control.
Serial dilutions were prepared for both the reference standard (2-20 µg/mL) and the plant extract (5-35 µg/mL) using methanol as the solvent. For each tested concentration, 0.5 mL of solution was placed in test tubes, then combined with 1 mL of the DPPH reagent. The tubes were gently shaken and incubated in the dark at room temperature for 30 min; absorbance was then recorded at 517 nanometers using a UV-Visible spectrophotometer.
The percentage of radical scavenging activity was determined by employing the following equation:
Where:
The antioxidant potential was determined by calculating the IC50 value, which refers to the required extract concentration (µg/mL) to neutralize 50% of DPPH free radicals.
Total Antioxidant Contents (TAC)
The total antioxidant capacity (TAC) was determined using the phosphomolybdenum method, which is based on the reduction of molybdenum(VI) ions (Mo6+) to molybdenum(V) ions (Mo5+) by the antioxidants present in the extract, leading to the formation of a green- colored phosphomolybdenum complex under acidic conditions. 25 For the assay, 0.2 mL of the diluted extract was mixed with 2 mL of the reagent solution, which contained a mixture of 0.6 M sulfuric acid, 28 mM sodium phosphate, and 4 mM ammonium molybdate prepared in 100 mL of distilled water. The mixtures were incubated in a water bath at 95 °C for 90 min, then cooled to room temperature before measuring the absorbance at 695 nm using a spectrophotometer. Gallic acid, at concentrations ranging from 0.01 to 0.14 mg/mL, was employed to generate a standard calibration curve. The antioxidant capacity of the extracts was expressed based on the linear regression equation correlating gallic acid concentrations with their corresponding absorbance values. 26
Anti-Hemolytic Activity Ex Vivo
The anti-hemolytic activity of
Plant extracts were prepared in PBS at final concentrations ranging from 10 to 40 mg/mL. In each well of a microplate, 20 µL of a red blood cell suspension was mixed with 40 µL of the extract solution. Hemolysis was then induced by adding 40 µL of hydrogen peroxide (H2O2) prepared in PBS. The plates underwent incubation at 37 °C for 3 h. To terminate the reaction, 80 µL of PBS and 30 µL of a 20% sucrose solution were added to each well. Finally, the degree of hemolysis was assessed by measuring the absorbance at 540 nm using a microplate reader.
A positive control was prepared by treating the erythrocyte suspension with H2O2 alone to represent 100% hemolysis, while PBS alone served as the negative control. α-Tocopherol (vitamin E), prepared at the same concentration range (10-40 mg/mL), was used as a reference antioxidant and subjected to identical experimental conditions. All experiments were performed in triplicate, and the anti-hemolytic activity was determined as a percentage using the following formula:
Evaluation of the Antimicrobial Activity of the Hydro-Ethanolic Extract of P. guajava
To evaluate the antimicrobial therapeutic potential of the hydro-ethanolic extract of
Agar Diffusion Method (Well Method)
Petri dishes containing Mueller–Hinton agar (for bacterial testing) were aseptically seeded with microbial suspensions standardized to 106 CFU/mL, derived from freshly cultured bacterial or yeast isolates. The inoculation was carried out evenly across the agar surface using sterile cotton swabs. After seeding, the plates were left to dry briefly to allow proper absorption before further processing. Wells were created at the center of each plate using the upper tip of a sterile Pasteur pipette. Each well was then filled with approximately 50 µL of the extract dissolved in water at concentrations of 60, 30, 15, and 5 mg/mL. Gentamicin (30 µg/disc) served as the positive control.
The plates were incubated at 37 °C for 24 h for bacterial cultures and 48 h for yeasts. The antimicrobial activity was assessed by observing the formation of clear inhibition zones around the wells. The diameters of these zones were measured in millimeters to evaluate the efficacy of the extract.29,30 (see Figure 1)

Diagram Illustrating the Well Diffusion Method on Agar Medium. 31
Broth Microdilution Method for MIC and MBC Determination
The antimicrobial efficacy of
A stock solution of the extract was prepared at a concentration of 80 mg/mL by dissolving the dry plant extract in dimethyl sulfoxide (DMSO). Serial dilutions were made to obtain final concentrations between 80 and 2.5 mg/mL. In the microplate assay, 100 μL of each extract dilution was added to the wells, followed by 50 μL of the standardized bacterial suspension. Each assay included a growth control (without extract or antibiotic) and a sterility control (broth only). Plates were incubated at 37 °C for 24 h.
The MIC was determined as the lowest concentration of the extract that completely inhibited visible bacterial growth. 33 To determine the MBC, 10 μL from each well showing no visible growth was subcultured on MHA plates free of the extract and incubated under the same conditions. The MBC was defined as the minimum concentration at which bacterial growth was completely inhibited on the agar surface. All experiments were performed in triplicate to ensure reproducibility of the results. 34
Anti-Inflammatory Activity In Vitro
The anti-inflammatory potential of the hydro-ethanolic extract was evaluated based on its ability to inhibit heat-induced denaturation of BSA following a previously established protocol with minor modifications.35,36
Various concentrations of the extract and the standard anti-inflammatory agent aspirin were prepared in distilled water. For each treatment, 50 µL of the test extract was combined with 450 µL of a 0.5% (w/v) aqueous BSA solution. The mixtures were incubated at 37 °C for 20 min, followed by heat treatment at 57 °C for 5 min to induce protein denaturation. A control sample was prepared by replacing the extract with double-distilled water (DD). After cooling at room temperature, the absorbance of each sample was measured at 255 nm using a UV–Visible spectrophotometer. Aspirin (acetylsalicylic acid) served as the positive control. All experiments were carried out in triplicate to ensure the reproducibility and reliability of the results. The percentage inhibition of protein denaturation was calculated using the following equation:
Vt: Absorbance of the test sample; Vc: Absorbance of the control. The IC50 value, which corresponds to the concentration of the extract required to inhibit 50% of protein denaturation, was determined from the dose–response curve.
Evaluation of The Inhibitory Activity of P. guajava Extract Against α-amylase Enzyme
The inhibitory potential of the hydro-ethanolic extract of
The experiment included various controls: a blank sample (extract replaced by phosphate buffer), acarbose as a standard inhibitor, an enzyme-free control, and a starch-only control. All measurements were carried out in triplicate to ensure result accuracy. The percentage of inhibition was calculated using the following formula:
The IC50 value was determined through regression analysis.
Statistical Analysis
Most experiments were repeated three times (n = 3), and results were recorded as mean ± standard deviation. All statistical data and IC50 values were calculated using origin pro 2018 and Microsoft Excel 2016 software.
Results
Determination of Total Flavonoid and Phenolic Contents
The total phenolic and flavonoid contents of the
Phenolic and Flavonoid Contents of

HPLC Analysis of Phenolic Compounds
HPLC analysis of the hydro-ethanolic extract from

HPLC Chromatogram of Phenolic Compounds in Hydro-Ethanol Extract of
Concentrations of Phenolic Compounds in the Hydro-Ethanolic Extract and Their Retention Time as Determined by HPLC-DAD (n = 3).

Antioxidant Activity
In Vitro
Table 4 summarizes the antioxidant properties of hydro-ethanolic extract, revealing significant antioxidant capacity. The TAC reached 491.95 ± 2.45 mg GAE/g extract, as determined by the phosphomolybdenum method. DPPH radical scavenging assays demonstrated potent activity, with the extract showing an IC50 of 17.24 ± 0.01 μg/mL - comparable to ascorbic acid (reference standard). These results indicate that lower IC50 values correlate with enhanced antioxidant efficacy, confirming the extract's strong free radical neutralization potential.
Total Antioxidant Capacity (TAC) and IC50 Values for DPPH Test of
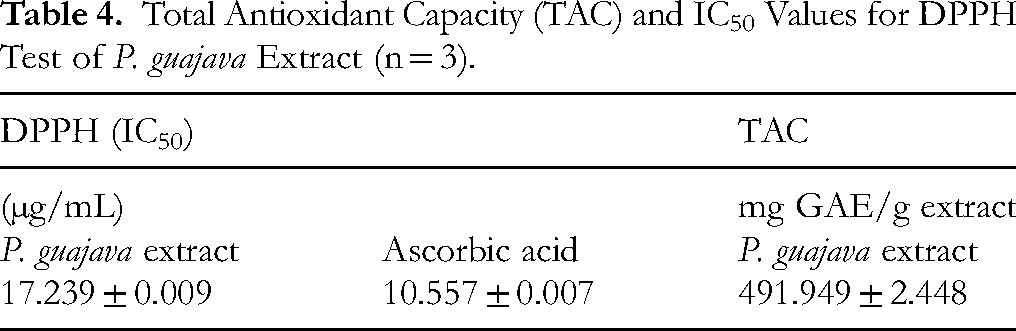
IC50: Half Maximal Inhibitory Concentration.
Anti-Hemolytic Activity Ex Vivo
The protective capacity of the

Anti-Hemolytic Inhibitory Activity of
Percentage Inhibition of the Anti-Hemolytic Activity of

Antimicrobial Activity
Table 6 (Figure 4) presents the antibacterial activity of the hydro-ethanolic extract of

Antibacterial Assay by the Wells Diffusion Method for Hydro-Ethanolic Extract on Agar Plate.
Results of Antibacterial Activity of

NI = No Inhibition, CN = Gentamicin (CN) 30ug Discs.
Using the broth microdilution method, the minimum concentrations required to inhibit growth (MIC) of the

Photo of Broth Microdilution Method Used for Antimicrobial Activity of Hydro-Ethanolic Extract Against Bacteria Strains.
MIC and MBC of P. Guajava Extract Against Different Bacteria and MBC/MIC Ratio.

The experimental results for both tests are shown in Figure 4 and Figure 5, respectively.
In Vitro Denaturation of Bovine Serum Albumin (BSA)
The anti-inflammatory potential of the hydro-ethanolic extract was evaluated based on its ability to inhibit the heat-induced denaturation of BSA. At a concentration of 1000 µg/mL, the extract exhibited significant inhibitory activity (67.70 ± 0.66%). In comparison, the reference compound aspirin showed a superior inhibitory effect (94.13 ± 2.20%) at a lower concentration (500 µg/mL), which represented the maximum observed effect. The calculated IC50 values were 459.14 ± 27.49 µg/mL for the extract and 7.86 ± 0.15 µg/mL for aspirin, highlighting the markedly higher potency of aspirin. A dose-dependent increase in inhibition was observed for both the extract and the standard compound, as presented in Table 8. These findings indicate that the hydro-ethanolic extract exhibits moderate inhibitory activity against BSA denaturation compared to aspirin.
In Vitr

In Vitro α-amylase Inhibitory Activity
The hydro-ethanolic extract of

Discussion
In the present study, the extraction yield obtained using 50% hydro-ethanolic solvent infusion was approximately 29.258%, which is considered satisfactory. A comparable study by Park et al 39 using the same solvent system reported a yield of 25.8 ± 2.54%. Hydro-ethanol (50%) is widely regarded as one of the most effective solvents for phytochemical extraction, offering an optimal balance between polarity and solubility, and generally outperforming absolute ethanol or water alone. Park et al further demonstrated that ethanol 30% achieved the highest yield (35.8%), while the yield decreased significantly with ethanol 70% (17.1%), suggesting that the intermediate solvent concentration (50%) offers excellent quantitative and qualitative extraction advantages. Other reports also indicate that medium hydro-ethanol concentrations improve total phenol recovery and antioxidant activities compared to other solvents. 15
The antioxidant capacity of
Previous studies (Nursanty et al;
42
Huynh et al,
19
) have identified a broad range of phenolic compounds—including gallic acid, chlorogenic acid, isorhamnetin, and catechin derivatives, quinic acid, epigallocatechin, and flavonoids such as quercetin, apigenin-7-β-D-glucuronopyranoside and naringenin-4'-O-glucopyranoside—in
Leaf maturation stages, harvest season, drying, and storage significantly influence chemical composition, as confirmed by multiple seasonal studies. 46 However, the lack of mass spectrometry (MS) in our study limits precise compound identification, necessitating the application of advanced analytical techniques such as LC-MS in future research.
Regarding bioactivity, quantitative estimates in this study revealed high levels of total phenolic (268.304 ± 0.659 mg GAE/g) and flavonoids (16.959 ± 0.099 mg QE/g), reflecting notable antioxidant capacity. The extract exhibited strong DPPH radical scavenging activity (IC50 = 17.239 ± 0.009 µg/mL) and a TAC of 491.949 ± 2.448 mg GAE/g.
The extract also demonstrated 43% anti-hemolytic activity (43% inhibition at 47 mg/mL compared to 82% for vitamin E), likely due to the ability of flavonoids and polyphenols to stabilize erythrocyte membranes and reduce lipid peroxidation. 48
In terms of antimicrobial properties, the extract showed selective efficacy against Gram-positive bacteria such as
The extract also displayed moderate anti-inflammatory activity, as evidenced by its inhibition of BSA denaturation. This effect may be attributed to compounds such as gallic acid, caffeic acid, chlorogenic acid, p-coumaric acid, sinapic acid, quercetin, and apigenin, known for their anti-inflammatory properties. 50 However, while this assay is preliminarily acceptable for assessing anti-inflammatory activity, it is insufficient alone to explain the mechanism of action, necessitating more advanced models (eg, COX-2 or TNF-α inhibition). 51
Finally, the extract showed significant α-amylase inhibitory activity (IC50 = 168.3 ± 1.12 µg/mL), surpassing the pharmaceutical standard acarbose (IC50 = 299.2 µg/mL), This suggests potential antidiabetic properties, although the current data are limited to
The novelty of this study lies in its comprehensive in vitro evaluation of multiple bioactivities of
However, this study has several limitations. All bioassays were conducted in vitro, and no in vivo or clinical validation was performed. and the plant material was from a single geographic location, which may not capture chemotypic diversity. In addition, although the activities observed are promising, the exact mechanisms of action remain to be elucidated.
Overall, the findings support further research on
Conclusion
The findings of this study indicate that the hydro-ethanolic extract of
While further
Abbreviations
To enhance accuracy and readability, all abbreviations have been systematically reviewed and defined as follows
Total Phenolic Content
Total Flavonoid Content
High-Performance Liquid Chromatography coupled with Diode-Array Detection.
Total Antioxidant Contents
Reactive Oxygen Species
Phosphate-Buffered Saline
Bovine Serum Albumin
Minimum Inhibitory Concentration
minimum bactericidal concentration
Milligrams of Gallic Acid Equivalents per Gram of sample extract
Milligrams of Rutin Equivalents per Gram of sample extract
Hydrolysable Polyphenols
Condensed Tannins
An inducible enzyme involved in prostaglandin synthesis, playing a key role in inflammation and pain.
A pro-inflammatory cytokine that regulates immune responses and is implicated in various inflammatory and autoimmune diseases.
Dimethyl Sulfoxide.
Footnotes
Acknowledgments
We would like to thank the editor and the anonymous reviewers for their thoughtful and accurate comments on this manuscript, as well as everyone who helped us complete and proofread this manuscript.
Ethical Approval
This study is not applicable to both human and/or animal studies that require ethical approval.
Author Contributions
All authors contributed to the study.
Funding
The authors declare that no funding was received for this study.
Competing Interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data Availability
The data that supports the findings of this study are available within the article.
