Abstract
Objective
The aim of this study was to determine the mechanism of action of Pithecellobium dulce aqueous extract on Jatropha oil-induced hemorrhoidal disease in rats.
Methods
36 Wistar rats were divided into 6 groups of 6 animals, including 3 controls (normal, negative and positive) and 3 tests groups. Hemorrhoidal disease was induced with Jatropha oil (100 μL) for 5 days in all animals except the normal control group. Recto-anal coefficient, hemorrhoidal permeability, hematological parameters, oxidative stress parameters and inflammatory cytokines were evaluated.
Results
Quantitative phytochemistry and in-vitro anti-inflammatory and anti-radical activity of the extract were determined. Extract of P. dulce at doses of 100, 200 and 400 mg/kg significantly (p ˂ 0.01) reduced recto-anal coefficient, prostaglandin E2 (PGE2), TNF-α, IL-6, nitric oxide (NO) and C-reactive protein (CRP) concentrations, accompanied by a significant (p < 0.001) increase of IL-5 and IL-10. Extract of P. dulce at different doses significantly reduced leukocyte and platelet counts and increased erythrocyte and hematocrit levels. Extract of P. dulce also significantly (p < 0.05, p < 0.01 and p < 0.001) increased superoxide dismutase (SOD), catalase (CAT) and reduced glutathione (GSH) activities and significantly (p < 0.001) decreased malondialdehyde (MDA) levels. Extract of P. dulce was able to trap DPPH (2,2-Diphenyl-2-picrylhydrazyl) radicals (64.9%) and ABTS (2,2'-azinobis-3-ethylbenzothiaoline-6-sulfonic acid) radicals (69.1%), as well as a high iron reducing power (FRAP) of 56.2% and the 50% inhibitory concentration (IC50) of the ABTS radical was 150 μg/mL compared to the butylhydroxytoluene (BHT) control (130μg/mL). In addition, extract of P. dulce showed high concentrations of polyphenols, flavonoids and saponins.
Conclusion
extract of P. dulce possesses anti-hemorrhoidal properties via mechanisms involving vasoconstriction, alongside enhancement of anti-inflammatory and antioxidant status.
Keywords
Introduction
Constipation is a major risk factor in the development of hemorrhoidal disease, which is characterized by an increased intra-abdominal pressure that directly impedes venous return, 1 due to stools applying strong forces to anal layers during defecation. 2 Damage can then occur to the epithelial barrier, leading to increased permeability, probably due to defective regulation of tight junctions. 3 In fact, the body's immune system reacts abnormally to alteration of hemorrhoidal mucosal cells due to the release of soluble factors involving inflammatory lipid metabolites, leading to an increase in the pro-inflammatory response, such as activation of monocytes, macrophages and cytokines. Inflammation of the mucosa of patients with hemorrhoidal disease (HD) results in extensive infiltration of lymphocytes, particularly CD4+ T cells.4,5 Activated CD4 T cells display increased cytotoxic activity 6 and secrete cytokines that enhance the inflammatory state, leading to tissue damage.7,8 Cytokine imbalance (Il-6 and TNF-α) and production of inflammatory mediators (NO, PGE2) by activated CD4+ T lymphocytes play an important role in the pathogenesis of HD. Type 2 T helper cells (Th2) and their cytokines, in particular interleukin IL-6, have been suggested to foster HD development. 9
The mechanism of inflammation involved in HD is characterized by the production of pro-inflammatory mediators such as nitric oxide (NO) by macrophages. iNOS is thought to be responsible for increased NO production in the epithelium and in inflammatory foci. iNOS-derived NO stimulates TNF-α production in hemorrhoidal tissue, which promotes neutrophil infiltration, leading to hemorrhoidal tissue damage. 6
Conventional treatment of hemorrhoidal disease involves the use of venotonics (Daflon, Veinamitol, and Veliten) and non-steroidal anti-inflammatory drugs (diclofenac).10,11 However, associated side effects can include digestive disorders such as nausea, vomiting or stomachache, accompanied by headaches, skin rashes and diarrhea. Because of this broad range of side effects, people are turning to herbal remedies as an alternative treatment for hemorrhoidal diseases.
Pithecellobium dulce is a plant of the Fabaceae family widely used in ethnomedicine for the treatment of hemorrhoidal diseases, diarrhea and high blood pressure. In addition, its numerous pharmacological properties such as analgesic, 12 anti-ulcerogenic, 13 antioxidant 14 and hepatic oxidative disorder reducing, 15 anti-Hymenolepisnana, 16 xanthine oxidase, 17 anti-ulcerogenic 18 activities have been evaluated. However, its anti-hemorrhoidal activity has not yet been scientifically investigated. Hence, the aim of this work was to determine the mechanism of action of Pithecellobium dulce aqueous extract on Jatropha oil-induced hemorrhoidal disease in rats.
Results and Discussion
Results
Qualitative and Quantitative Phytochemistry
Figure 1 presents the qualitative phytochemical analysis of the extract of P. dulce. The main classes of compounds present include alkaloids, flavonoids, terpenoids, glycosides, polyphenols, saponins and tannins. Total polyphenol, flavonoid, tannin, saponin, alkaloid and terpenoid contents are shown in Figure 1. The trapping capacity estimated by FRAP method was 56%, while the values found by DPPH and ABTS methods were 65% and 69%, respectively (Figure 2).
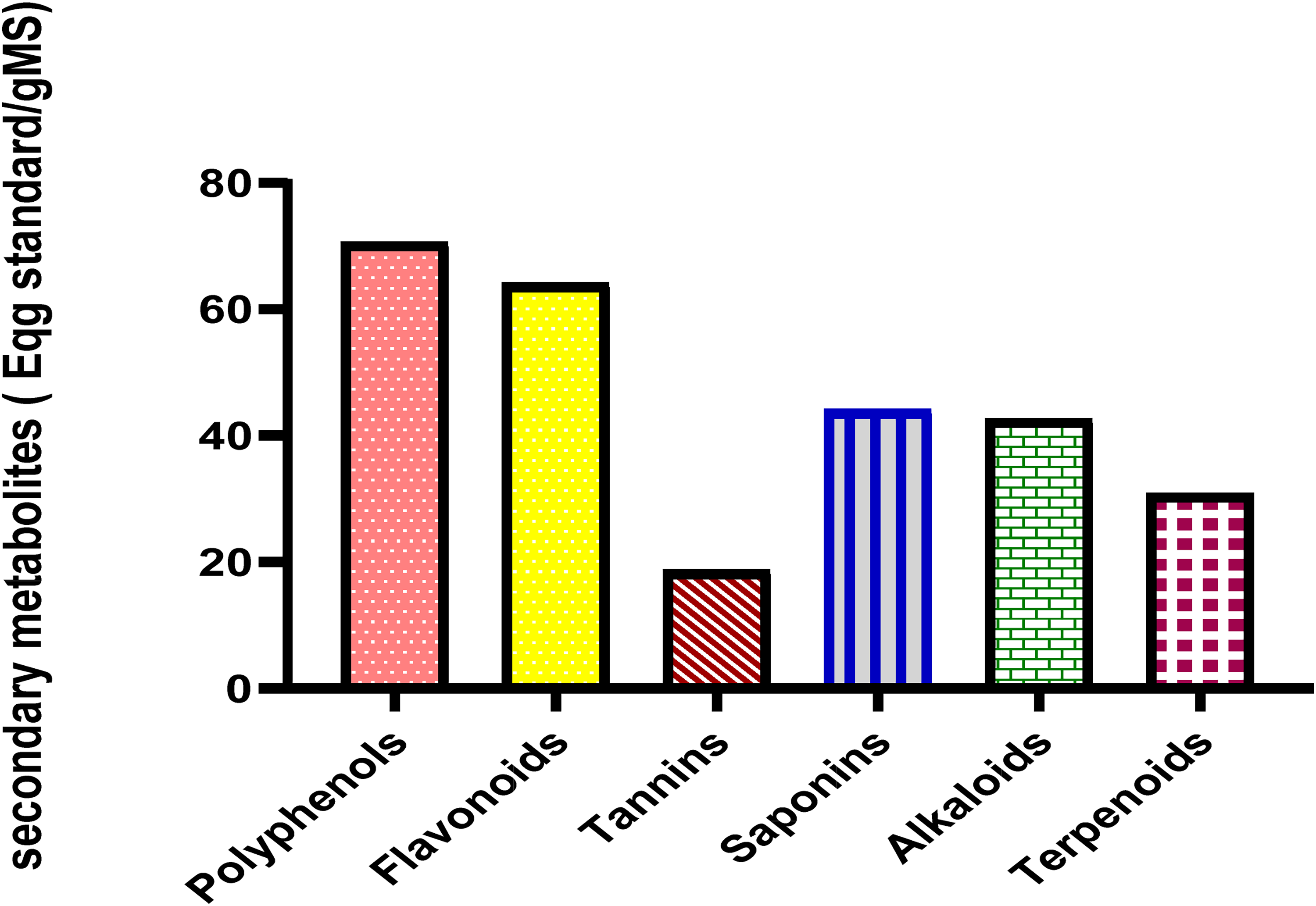
Total content of secondary metabolites of aqueous extract of P. dulce.
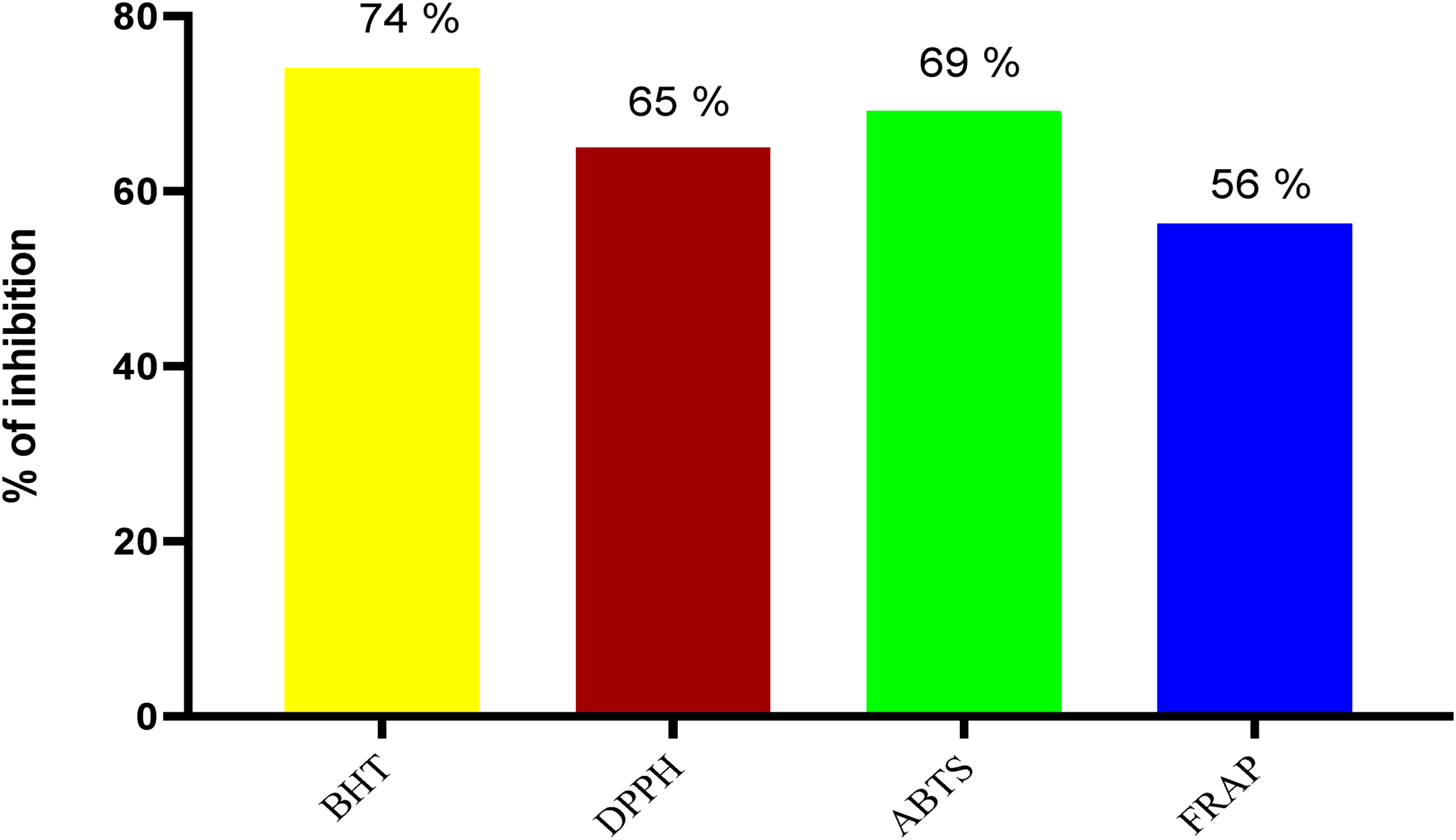
In vitro antioxidant activity of aqueous extract of P. dulce. BHT: butylated hydroxytoluene; DPPH: 2,2-diphenyl-2-picrylhydrazyl; ABTS: 2,2′-azinobis-3- ethylbenzothiazoline-6-sulfonic acid; FRAP: ferric reducing antioxidant potential.
Effect of Extract of P. dulce on Inhibition of Protein Denaturation
Figure 3 shows that the inhibition rate of protein denaturation increased with extract concentrations of Daflon and Diclofenac. At 10 μg/mL of extract of P. dulce, an anti-denaturation activity of 53.5% was observed compared to Daflon (53%) and Diclofenac (47%).

Effect of extract of P. dulce on inhibition of protein denaturation.
Macroscopic Appearance of the Anus After Induction of Hemorrhoidal Disease with Jatropha Oil
Macroscopic observation of the anus after application of Jatropha oil in rats showed a linear development of edema in rats of the negative group (Figure 4). Whereas in the groups treated with Daflon (10 mg/kg) and extract of P. dulce at different doses (100, 200 and 400 mg/kg) in B, C, D and E respectively. We observed significant protection of the anal canal structure compared to the negative control group.

Macroscopic appearance of the anus.
Effect of Pithecellobium dulce Aqueous Extract on Body Weight
Figure 5 shows the effects of the extract of P. dulce on body weight change throughout the treatment. In contrast to rats in the normal control group, there was a significant reduction (p < 0.001 and p < 0.01) in the weight of rats in the negative control group from the fourth day-to-day eighteen of the experiment. On the other hand, daily administration of Daflon and extract of P. dulce at doses of 100, 200 and 400 mg/kg resulted in a significant increase in body weight, especially from the tenth day onwards.

Effect of extract of P. dulce on body weight variation in rats. Values are represented as means ± SEM (n = 6). The significant difference compared with the control groups is presented as follows: θθ p < 0.01 and θθθ p < 0.001 compared to the normal control; **p < 0.01 and ***p < 0.001 compared to the negative control.
Effect of the Extract of P. dulce on the Recto-Anal Coefficient in Rats
Figure 6 shows the effect of Pithecellobium dulce aqueous extract on the recto-anal coefficient (RAC). Application of Jatropha oil to the recto-anal area of rats induced alterations characterized by a significant increase (p ˂0.01) of the RAC of the negative control group compared with the normal control. On the other hand, there was a significant decrease (p ˂0.05 and p ˂0.01) in RAC in the positive control group and those given the extract at different doses (100, 200 and 400 mg/kg) compared with the negative control.
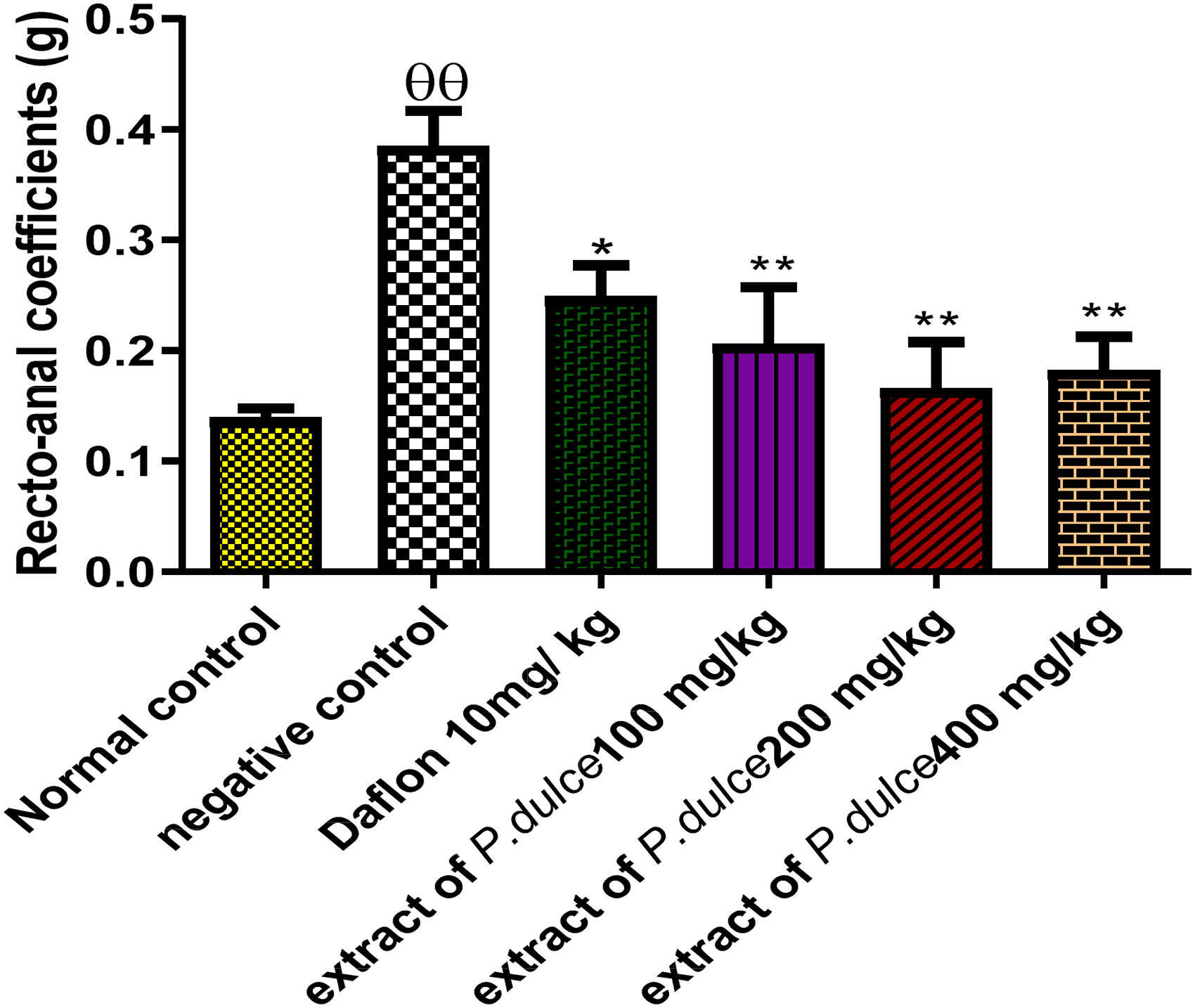
Effect of extract of P. dulce on recto-anal coefficient in rats. Values are represented as means ± SEM (n = 6). The significant difference compared with the control groups is presented as follows: θθ p < 0.01 compared to the normal control; * p < 0.05 and ** p < 0.01 compared to the negative control.
Effect of Extract of P. dulce on serum CRP Levels in Rats
Figure 7 shows that there was a significant increase (θθθ p < 0.001) in serum CRP levels in the negative control compared with the normal control. Daily treatment of animals with extract (100, 200 and 400 mg/kg) and Daflon (10 mg/kg) significantly (*** p < 0.001) reduced CRP levels compared with the negative control.

Effect of extract of P.dulce on serum CRP levels in rats. Values are represented as means ± SEM (n = 6). Significant difference from control groups is presented as follows: θθθ p < 0.001 compared to the normal control; *** p < 0.001 compared to the negative control, CRP: C-reactive protein.
Effect of the Extract of P. dulce on Hematological Parameters
Figure 8 shows the effect of the extract of P. dulce on hematological parameters 18 days after induction of hemorrhoidal disease by Jatropha oil. There was a significant increase of platelets and white blood cells (p < 0.001), significantly reduced levels of red blood cells (θ p < 0.05) and hematocrit (θθ p < 0.01) in the negative group compared with the normal control rats. However, treatment of rats with Daflon (10 mg/kg) and extract of P. dulce (100, 200 and 400 mg/kg) significantly reduced (*** p < 0.001 and ** p < 0.01) white blood cell counts (BCC). In addition, treatment with the extract of P. dulce (100, 200 and 400 mg/kg) significantly increased (*** p < 0.001, ** p < 0.01 and * p < 0.05) hematocrit levels compared with the negative control.

Effect of extract of P. dulce on hematological parameters. Values are represented as means ± SEM, (n = 6). The significant difference compared to the control groups is shown as follows: θ p < 0.05; θθ p < 0.01 and θθθ p < 0.001 compared to the normal control; * p < 0.05;** p < 0.01 and*** p < 0.001; compared to the negative control. HTC: hematocrit and PLT: platelet.
Effect of the Extract of P. dulce on NO Levels in Rats
Assessment of plasma nitric oxide (Figure 9) showed a significant increase (θθ p < 0.01) in the negative control compared with the normal control. In contrast, Daflon (10 mg/kg) and extract of P. dulce (100, 200 and 400 mg/kg) significantly (** p < 0.01 and*** p < 0.001) reduced NO levels compared with the negative control.

Effect of extract of P. dulce on nitric oxide levels in anal tissue. Values are expressed in terms of mean ± SEM (n = 6). **p < 0.01, ***p < 0.001): comparison to the negative control group. θθ p < 0.01 : comparison to the normal control group. NO: nitric oxide.
Effect of the Extract of P. dulce on Evans Blue Dye Levels in Rats
Application of Jatropha oil to the recto-anal region of diseased rats resulted in a significant increase (p < 0.001) in the extravasation of Evan's Blue (Figure 10) dye compared with the normal control. However, treatment with the extract of P. dulce (100, 200 and 400 mg/kg) maintained significant protection (*** p < 0.001) against increased extravasation of EB dye.

Effect of extract of P. dulce on extravasation of Evans blue dye into recto-anal tissue. Values are represented as means ± SEM, (n = 6). Significant difference from control groups is shown as follows: θθθ p < 0.001 compared to the normal control;** p < 0.01 and *** p < 0.001; compared to the negative control. EB: Evan Blue.
Effect of the Extract of P. dulce on PGE2 Level in Rats
Figure 11 shows the effect of the extract of P. dulce on PGE2 levels. Rats in the negative control group showed a significant increase (θθθ p < 0.001) in PGE2 concentration compared with the normal control. On the other hand, rats treated with Daflon (10 mg/kg) and extract at doses of 100 and 400 mg/kg showed a significant decrease (***p < 0.001 and **p < 0.01) in PGE2 levels compared with the negative control.

Effect of extract of P. dulce on serum PGE2 levels. Values are represented as means ± SEM, (n = 6). Significant difference from control groups is presented as follows: θθθ p < 0.001 compared to the normal control;** p < 0.01 and*** p < 0.001; compared with negative control. PGE2: prostaglandin E2.
Effect of the Extract of P. dulce on TNF-α and IL-6 Levels in Rats
Figure 12 shows the effect of the extract of P. dulce on pro-inflammatory cytokines during Jatropha oil-induced acute inflammation. These results show significantly elevated expression (θθθ p < 0.001) of TNF-α and IL-6 in the negative control group compared with the normal control. Treatment with Daflon (10 mg/kg) and extract of P. dulce (100, 200 and 400 mg/kg) significantly (p < 0.001 and p < 0.01) decreased these cytokines levels.

Effect of extract of P. dulce on TNF-α and IL-6 levels in rats. Values are represented as means ± SEM, (n = 6). Significant difference from control groups is shown as follows: θθθ p < 0.001 compared to the normal control;** p < 0.01 and*** p < 0.001; compared to the negative control. TNF-α tumor necrosis factor-alpha, IL-6: interleukin-6.
Effect of the Extract of P. dulce on IL-5 and 10 Levels in Rats
Figure 13 shows the effect of the extract of P. dulce on serum levels of anti-inflammatory cytokines, revealing a significant decrease (p < 0.001) in serum levels of IL-5 and IL-10 in untreated diseased rats compared with normal control rats. Treatment of rats with extract of P. dulce (100, 200 and 400 mg/kg) and Daflon (10 mg/kg) significantly increased (p < 0.001 and p < 0.01) IL-5 and IL-10 levels compared with the negative control.

Effect of extract of P. dulce on IL-5 and 10 levels in rats. Values are represented as means ± SEM, (n = 6). Significant difference from control groups is presented as follows: θθθ p < 0.001 compared to the normal control;** p < 0.01 and*** p < 0.001; compared to the negative control. IL-5: interleukin-5, IL-10: interleukin-10.
Effects of the Extract of P. dulce on Some Oxidative Stress Parameters in Liver and Anal Tissues
Table 1 shows the results of oxidative stress markers in liver and anal homogenates.
Effect of Aqueous Extract of P. dulce Seeds on Some Oxidative Stress Parameters.

Values are represented as means ± SEM, (n = 6). The significant difference compared to the control groups is presented as follows: θθp < 0.01 and θθθ p < 0.001 compared to the normal control;*p < 0.05, **p < 0.01 and ***p < 0.001; compared to the negative control. MDA: Malondialdehyde, SOD: superoxide dismutase, CAT: catalase, GSH: Reduced glutathione.
Malondialdehyde (MDA) levels were significantly (p < 0.001) elevated in the liver and anus homogenates of animals in the negative control group compared with those in the normal group. In the liver and anus homogenates of animals treated with extract of P. dulce and Daflon, the level of MDA was significantly (p < 0.01 and p < 0.001) lower than in the negative control group.
Treatment of animals with extract of P. dulce (100, 200 and 400 mg/kg) and Daflon (10 mg/kg) for 18 days significantly (p < 0.001) increased superoxide dismutase activity in liver and anus homogenates compared to untreated animals. SOD activity significantly decreased (p < 0.001) in the negative control group, compared to the normal control group.
Catalase activity was significantly decreased in the liver (p < 0.001) and anus (p < 0.01) homogenates of the negative control group compared to animals in the normal control group. Treatment with extract of P. dulce (100, 200 and 400 mg/kg) and Daflon (10 mg/kg) significantly increased catalase activity in liver (p < 0.01) and anus homogenates (p < 0.05, p < 0.01 and p < 0.001) compared with the negative control.
Reduced glutathione levels were significantly (p < 0.01) reduced in liver and anus homogenates from the negative control group compared with normal control animals. In the liver and anus homogenates of animals treated with extract of P. dulce (100, 200 and 400 mg/kg) and Daflon (10 mg/kg) the level of reduced glutathione was maintained close to the normal value.
Effects of the Extract of P. dulce on the Ultrastructure of Anal Tissue in Rats
Figure 14 below shows the effects of Pithecelobium dulce aqueous extract on the structure of anal tissue in rats. Animals in the normal group showed normal anal tissue parenchyma with mucosal, muscular and submuscular layers and a distinct lumen. The negative controls, on the other hand, showed leucocytic infiltration of the mucosal layer. Compared with the negative controls, the diseased animals receiving the plant extract at different doses and the satellite animals showed normal anal tissue parenchyma.
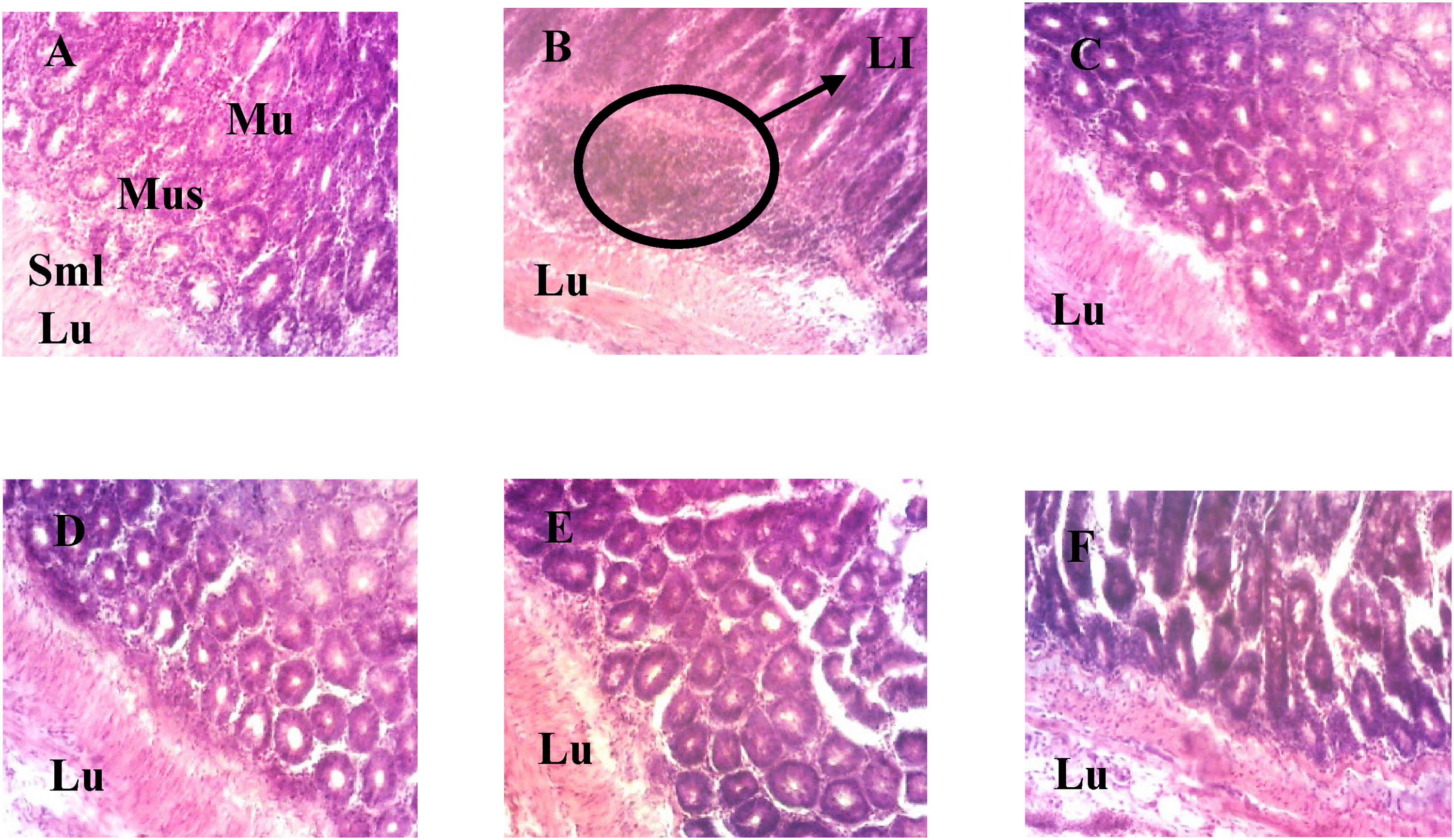
Microphotograph of histological sections of the anal canal (H-E, 100X). Mus: Muscularis, Mu: Mucosa, Lu: Lumen, LI: Leukocyte infiltration, Smus: Sub muscularis. A: Normal control; B: Negative control; C: Positive control; D, E, F: Test batches treated with Pithecellobium dulce extract at different doses.
Discussion
Hemorrhoidal disease (HD) is a pathological condition often characterized by vasodilation and inflammation in the recto-anal region, resulting in increased vascular permeability and extravasation of fluid accompanied by migration of large numbers of inflammatory cells into interstitial spaces, causing oedema. 19 In the present study, hemorrhoidal disease was induced with Jatropha oil. Jatropha oil causes inflammation due to the release of inflammatory lipid metabolites. These lipid factors, individually or in combination, regulate the activation of fibroblasts, endothelial cells, macrophages, monocytes, lymphocytes, neutrophils and newly recruited eosinophils, resulting in inflammation. Inflammation of the mucosa of patients with hemorrhoidal disease leads to significant infiltration of lymphocytes, particularly CD4+ T cells.4,5 Activated CD4 T cells display increased cytotoxic activity 6 and secrete cytokines which improve the inflammatory state leading to tissue damage.7,8 Type 1 (Th1) T-helper cells and their cytokines, in particular interleukin IL-6, have been suggested to enhance the development of HD. 9 In addition, the mechanism of inflammation involved in HD is characterized by the production of pro-inflammatory mediators such as nitric oxide (NO) by macrophages. iNOS is thought to be responsible for greatly increased NO production in the epithelium and in inflammatory foci. The NO derived from iNOS stimulates the production of TNF-α in hemorrhoidal tissue, which promotes neutrophil infiltration, leading to hemorrhoidal tissue damage. 6 The application of Jatropha oil to the recto-anal region caused a significant increase in TNF-α, IL-6 and NO in negative controls, thus confirming the establishment of our HD model. Administration of the extract of P. dulce at different doses favored recovery from this inflammatory state through a significant decrease in TNF-α, IL-6, RAC and Evan Blue dye exudation. Furthermore, Th2 cells secrete anti-inflammatory cytokines, such as IL-5 and IL-10, that also play an important role in regulating the levels of endogenous pro-inflammatory cytokines during HD. 20 IL-5 blocks Th1 cell production and supports Th2 cell formation. IL-10 inhibits the synthesis of cytokines generated by Th1 cells (TNF-α and IL-6). 21 Administration of the extract stimulated the expression of these anti-inflammatory cytokines (IL-5 and IL-10) in treated rats. These results suggest that the extract of P. dulce would have anti-inflammatory properties.
The mechanism of the inflammation process involved in HD is characterized by the production of pro-inflammatory mediators such as nitric oxide (NO). In this work, the anti-hemorrhoidal effects of the extract of P. dulce could be achieved in part by inhibiting pro-inflammatory cytokines and stimulating anti-inflammatory cytokines in rats with hemorrhoidal disease induced by Jatropha oil. During inflammatory processes, the precursor of NO is L-arginine via inducible nitrite oxide synthase (iNOS) in monocytes and macrophages. 22 At excessively high concentrations (in the case of animals in the negative group), this NO combines with a superoxide anion to form peroxynitrite, which induces nitration of nucleic acids, proteins and lipids, deforming their structures and altering their function. 23 The extract led to a reduction in NO levels in diseased animals, and the in-vitro anti-inflammatory test showed a high inhibition percentage of protein denaturation, which confirms the in vivo anti-inflammatory capacity of the extract.
BCC was performed to quantify the level of leukocytes, mainly monocytes, neutrophils and lymphocytes, in the blood of experimental animals. During Jatropha oil-induced inflammation, monocytes, leukocytes and neutrophils are recruited from the blood to the site of the disorder to perform their functions. 24 This explains the increase in these monocytes, leukocytes and neutrophils in the negative control. The decrease in these parameters reflects a reduction of inflammation in animals. Extract administration reduced leucocyte (WBC) levels thanks to the phenols, flavonoids and tannins contained in the extract, which are known to be better inhibitors of leucocyte migration. 25 Similarly, the BCC indicated that the treated groups with extract of P. dulce and Daflon had a platelet count very close to the normal control, with this drop thought to be linked to a reduction in the production of pro-inflammatory molecules. In addition to their main function of platelet aggregation during blood coagulation, thrombocytes also have the ability to recognize foreign bodies and to initiate and modulate inflammatory responses. 26 During inflammatory phenomena, the mediators released lead to increased platelet production. 27 extract of P. dulce therefore acts on these mediators by inhibiting their syntheses, which explains the significant difference in platelet levels between treated and untreated hemorrhoidal animals.
Assessment of the concentration of C-reactive protein shows the degree of tissue damage. CRP binds to phosphocholine expressed on the surface of dead or dying cells and certain bacteria. This activates the complement system and phagocytosis by macrophages, which eliminate necrotic and apoptotic cells and bacteria. 28 Blood serum from negative animals showed a marked positive agglutination, indicating an increased release of CRP following tissue injury. This acute phase reaction results from an increase in the concentration of interleukin 6, produced by macrophages and adipocytes. Administration of the extract of P. dulce caused a significant reduction in CRP levels, so the extract may have acted to limit inflammation by increasing the expression of cell adhesion (L-selectin) and production of superoxide by neutrophils. Similar results were obtained by Xue-Yun Liang et al, 29 who suggested that Danshen (Salvia miltiorrhiza) aqueous extract limited agglutination in the serum of test animals, thus possessing anti-inflammatory activity.
Body weight was another parameter monitored in this study. In this study, a significant loss (p < 0.001) in body weight was observed in untreated sick rats compared with rats in the normal control group. This was probably due to the lack of mobility of the rats as a result of the inflammation of the anal canal, which limited their access to food. On the other hand, the inflammatory reaction caused by the application of Jatropha oil stimulates the production of leptin, a cytokine-like hormone, resulting in a reduction in food intake and consequent weight loss. 30 Similar results have been reported by some authors, showing that weight loss often accompanies hemorrhoidal disease due to the systemic and local action of inflammatory cytokines such as TNF-α and IL-6 produced mainly by monocytes and macrophages that stimulate muscle degeneration. 31 Furthermore, a significant weight gain (p < 0.01 and p < 0.001) was observed in rats treated with Daflon (10 mg/kg) and extract of P. dulce (100, 200 and 400 mg/kg). This is probably linked to their anti-inflammatory power, which reverses the effect of the disease thanks to the bioactive molecules.
Elevated PGE2 concentrations are associated with erythema, pain, vasodilation, fluid drainage and bone loss. 32 Inflammatory cytokines evoke COX2 expression and indirectly increase PGE2 concentration in anal vessels. 33 extract of P. dulce would have limited membrane permeability through reduced PGE2 expression in animals.
Lipid peroxidation is the process of oxidative degradation of polyunsaturated fatty acids in biological membranes that causes impairment of membrane function, deterioration of structural integrity, decreased fluidity and inactivation of a number of protein receptors and membrane-bound enzymes. 34 During this process, a large number of by-products are formed that affect a site far from the area of their generation, among these toxic derivatives, MDA is the most widely used marker in lipid peroxidation that has a longer half-life than free radicals. It diffuses easily and can form bonds with DNA bases, and is itself mutagenic. 34 Carol and Timothy demonstrated 35 that intra-anal administration of 100 μl of Jatropha oil is followed by oxidative stress. It is therefore possible that Jatropha oil administered into the anal tract is degraded and then phagocytosed by macrophages. Macrophages also play a major role in the production of reactive oxygen species, leading to lipid peroxidation and an imbalance in antioxidant status. 36 SOD, CAT and GSH are enzymes involved in protecting the anal canal from free radical damage. 37 SOD converts the superoxide anion to H2O2, which is degraded to H2O and O2 by catalase. GSH has been known as a transition metal chelator, vitamin E and C regenerator. 38 It reacts with O2−, OH, H2O2. Extract of P. dulce led to a decrease in MDA levels, which was associated with a significant increase in the activity of antioxidant enzymes such as SOD, CAT and GSH at different doses of the extract (100, 200 and 400 mg/kg). The reduction in lipid peroxidation by the extract suggests the in vivo antioxidant capacity of the extract of P. dulce. Our results are similar to those obtained by Atsang et al, 39 who showed that the aqueous extract of Pithecellobium dulce reduced the level of MDA in anal tissue and therefore lipid peroxidation. In addition, the in-vitro antioxidant test showed a strong capacity to trap ABTS and DPPH radicals and to reduce ferric iron to ferrous iron, confirming the extract's in-vivo antioxidant capacity. This observed activity could be due to the extract's high content of polyphenols, flavonoids and tannins.
Histopathological examination revealed that there was a significant difference in ARC and severity score in the normal control and treated groups compared with the negative control rats. Animals in the normal group showed normal cytoarchitecture of the recto-anal region. In addition, isolated tissues were observed to determine severity, with the results revealing no severity in the normal control group. Interestingly, treatment with the extract of P. dulce (400 mg/kg) remarkably improved the recto-anal lesions caused by Jatropha oil compared with the negative control group. Treatment with Daflon (10 mg/kg) also reduced recto-anal damage caused by Jatropha oil compared with the negative control group. Histopathological examination of the group treated with extract of P. dulce (400 mg/kg) and Daflon (10 mg/kg) revealed a marked reduction in the extent of vasodilation, the level of haemorrhage and necrosis. It should be noted that treatment with extract of P. dulce (400 mg/kg) restored near-normal architecture of the recto-anal region.
Limitations of the Study
The study was based on the evaluation of the anti-hemorrhoidal activity of Pithecellobium dulce extract. Pithecellobium dulce extract has a complex mixture of compounds and needs to be fractionated to arrive at potentially chemically active substances. This would enable us to identify the chemical constituent responsible for the most active anti-hemorrhoidal activity of Pithecellobium dulce extract and elucidate its possible mechanism of action.
Conclusion
Extract of P. dulce has anti-hemorrhoidal properties via its anti-inflammatory, antioxidant and vasoconstrictor properties. The anti-inflammatory effect of the extract of P. dulce shows the ability to attenuate the inflammation of the anal canal induced by Jatropha oil by reducing the RAC and the production of TNF-α and interleukin-6. Extract of P. dulce shows an ability to improve the antioxidant balance by strengthening the antioxidant capacities (SOD, CAT and GSH) responsible for inhibiting lipid peroxidation. The vasoconstrictor effect of the extract of P. dulce shows its ability to reduce hemorrhoidal permeability through a mechanism involving both the reduction in nitric oxide and prostaglandin E2. These effects attributed to the extract of P. dulce are due to the presence of pharmacologically active phytoconstituents. The results obtained from this study justify its use in ethnopharmacology for the treatment of hemorrhoidal disease.
Materials and Methods
Animal Material
For this study, Wistar rats of both sexes weighing 150 ± 10 grams and aged 13 ± 1 weeks were used. These animals were raised at the Animal House of the Laboratory of Biological Sciences of the University of Maroua, Cameroon. They were housed in cages covered with wire mesh and maintained at room temperature with a natural light/dark cycle. They received a standard diet with unlimited access to tap water daily. Prior authorization for the use of laboratory animals in this study has been obtained from the Cameroon National Ethics Committee (Reg. N. FWA-IRB 00001954). The use, handling, and care of animals were done in adherence to the European convention for the protection of vertebrate animals used for experimental and other purposes. 40
Plant Material
The plant material consisted of Pithecellobium dulce seeds collected at the fruiting stage in Maroua (N10°34’23.56968’’ ; E14°16’33.834’’). The plant was authenticated in the Cameroon national herbarium by comparison with existing material registered under No. 33821/HNC. After harvesting, the seeds were shade-dried for 7 days. They were then ground to a fine powder using a traditional mortar and sieve. The resulting powder was stored in hermetically sealed glass bottles. The powder obtained was used to prepare the extract.
Preparation of Pithecellobium dulce Aqueous Extract
Three hundred (300) grams of P. dulce powder were boiled in two (2 L) liters of distilled water for 15 min. After cooling, the mixture was filtered through Whatman No. 4 paper and the filtrate obtained was evaporated in an oven at 50 °C for 24 h, yielding 42.6 g of extract (15.4% yield).
Phytochemical Analysis
Qualitative Phytochemical Analysis
Phytochemical testing for the main secondary metabolites of the extract of P. dulce was carried out. Biologically active compounds such as alkaloids and glycosides, 41 phenols and flavonoids, 42 terpenoids, 43 saponins, 44 tannins and quinones were tested. 45
Quantitative Phytochemical Analysis
Determination of Total Polyphenols
Total polyphenols were determined using the Folin-Ciocalteu method. 46 The reagent, a mixture of phosphotungstic acid and phosphomolybdic acid, is reduced to a mixture of tungsten oxide and molybdenum oxide during phenol oxidation. The blue coloration produced is proportional to the total phenol content. 47
Determination of Flavonoids
In an acidic medium and in the presence of AlCl3, flavonoids give a red coloration with a maximal absorption at 430 nm. Total flavonoids were determined using the aluminum trichloride (AlCl3) method Mimica-Duckic. 48
Determination of Tannins
In an acidic medium, tannins react with vanillin to form a complex with maximal absorption at 500 nm. The assay consisted of taking up a volume of 0.2 mL of methanolic extract or standard solution with 2 mL of reagent (1 g vanallin/100 mL concentrated HCl), shaking the mixture and incubating at 30 °C for 5 min, then reading the optical density (OD) at 500 nm. Flavonoid concentrations in the sample are determined from a calibration curve plotted with catechin (0-50 μg/mL). 49
Saponin Assay
Saponin quantification was carried out according to the method described by Brunner in 1984. 50 Absorbance was measured using a spectrophotometer at 530 nm. A calibration curve was plotted using galactose (0-250 μg/mL) and results were expressed as milligrams of galactose equivalents per hundred grams of extract (mEqG / 100 g dry matter). 51
Alkaloid Assay
The alkaloid content of extract P. dulce was measured using the bromocresol green calorimetric method. 52 In the presence of bromocresol green, the alkaloids form a yellow complex that is easily extractable with chloroform at pH 4.7, with an absorption at 470 nm.
Terpenoid Assay
The terpenoid content of the extract of P. dulce was measured using the chloroform calorimetric method. 53 In acidic media, terpenoids react with chloroform to form a reddish-brown complex that shows an absorption maximum at 538 nm.
Evaluation of the in-Vitro Antioxidant Activity of the Aqueous Extract of P. dulce
FRAP Test
The reducing power of the extract is correlated to its antioxidant power. This technique was developed to measure the ability of the extracts tested to reduce ferric iron (Fe3+) present in the TPTZ complex (Fe(III)-2, 4, 6-Tri (2-pyridyl)-striazine) to ferrous iron (Fe2+). Fe3+ is involved in the formation of the hydroxyl radical via the Fenton reaction. The absorbance of the reaction medium is determined at 593 nm. 54 An increase in absorbance corresponds to an increase in the reducing power of tested extracts.
Anti-Radical Activity of ABTS
Activity of ABTS was measured according to the method described by Pellegrini et al 55 Fifty microliters (50 µL) of extract or standard were added to 150 μL of ABTS+ (7 mM). The mixture was stirred and incubated at room temperature for 40 min. The absorbance was read using a spectrophotometer at 745 nm. The antioxidant capacity of the sample was determined from the calibration curve established with Trolox (0-125 μg/mL). Percentage inhibition was calculated using the following formula: I % = ((Ac - At) /Ac) - 100 with (Ac: absorbance of control, At: absorbance of test). From this percentage, the concentration of extract inhibiting 50% (IC50) of the ABTS radical was determined. 51
Antiradical Activity of DPPH
DPPH is a stable purplish-colored free radical that absorbs at 517 nm. In the presence of free radical scavengers, the DPPH
Evaluation of the in-vitro Anti-Inflammatory Activity of the Aqueous Extract of P. dulce
The in vitro anti-inflammatory activity of Pithecellobium dulce seed aqueous extract was evaluated using the protein denaturation inhibition method.
57
The reaction mixture consisted of 0.05 mL (aqueous extract of Pithecellobium dulce seeds at different concentrations (10, 50, 100, 300, 500 and 1000 μg/mL) for the test solution, distilled water for the control solution, Diclofenac sodium and Daflon for the standard test solution at different concentrations than the extract) and 0.45 mL aqueous bovine serum albumin (BSA) solution. All the above solutions were adjusted to pH 6.3 with HCl (1 N), and the samples were incubated at 37 °C for 20 min, after which the temperature was increased to maintain the samples at 57 °C for 3 min. After cooling the tubes, 2.5 mL of saline phosphate buffer (0.1 M; pH 6.3) was added to the above solutions, and absorbance was measured using a UV-visible spectrometer at 416 nm. The percentage inhibition (% inh) of protein denaturation was calculated as follows :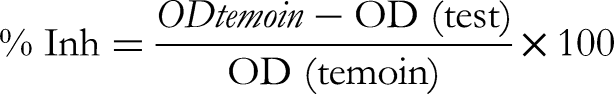
Evaluation of Antihemorrhoidal Activity Induced by Jatropha Oil
Antihemorrhoidal activity was evaluated in thirty-six (36) rats of both sexes. These animals were divided into 6 groups of 6 animals each, including 3 control groups (normal, negative and positive) and 3 test groups. Hemorrhoidal disease was induced in all animals except the normal control by insertion into the anus (recto-anal portion, 20 mm from the anal opening) of sterile cotton swabs (4 mm in diameter) soaked in Jatropha oil (100 μL) once a day for five days while restraining the animal for 10 s. On day 5 of induction, Evans Blue (EB) dye (30 mg/kg) was administered by injection into the animals’ tail veins, 30 min before application of the Jatropha oil preparation to quantify plasma exudation. From the sixth day, the animals received 10 mL/kg of distilled water daily for 18 days (normal and negative control); 10 mg/kg of Daflon (positive control) and the extract of P. dulce at doses of 100, 200 and 400 mg/kg (for the 3 test groups). Body weight was assessed daily in all animals. On the eighteenth day, 1 h after treatment, all animals were fasted for 24 h. On the nineteenth day, all animals were sacrificed by cervical dislocation. Blood samples were taken from the jugular vein in EDTA tubes for BCC and in dry tubes for the determination of inflammatory cytokines and CRP. Recto-anal tissues were harvested and weighed ; a portion was fixed in 10% formalin for histological examination
58
and another portion was used for oxidative stress marker assays. The liver was also harvested and used for stress marker assays.
58
The RAC was calculated using the formula: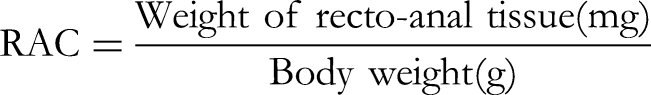
Hematological Analysis
The BCC was performed on a multiparameter automated system. Red blood cell, white blood cell, hematocrit and platelet counts were the hematological parameters of interest in our study. 58
CRP Assay
The CRP assay was performed using the immunoturbidimetric test by Chetana. 59
Determination of Cytokines and PGE2
The determination of IL-5, IL-6, IL-10, TNF-α and PGE2 proteins was performed by ELISA (Enzyme Linked Immunosorbent Assay) using the Quantikine kit and following the supplier's instructions.
In Vivo Antioxidant Activity of the Extract of P. dulce on Hemorrhoidal Disease
The in vivo antioxidant capacity of the extract was assessed in liver and anus homogenates. MDA levels were determined according to the protocol of Wilbur et al 60 Superoxide dismutase (SOD), catalase (CAT) and reduced glutathione (GSH) activity were determined according to the protocol of Misra and Fridovich. 61
Determination of Nitrites
Nitrites were measured with Griess reagent according to the method described by. 35 Absorption of the chromophore upon deionization of nitrites by sulfanilamide coupled to phenaphthylenediamine (NED) was read at 546 nm. The product obtained was proportional to the amount of nitrite present in the sample. The level of nitric oxide was determined from the calibration curve established using different concentrations of NaNO2. 51
Evan Blue Dye Assay
EB dye present in the tissue was extracted using 1 ml formamide. Sample absorbance was recorded using Synergy HT (multimode microplate reader, BioTek) at 620 nm and quantified using the EB dye standard curve. 62
Histological Analysis
The histological study was carried out according to the technique described by Smith and Bruton. 63
Statistical Analysis
Statistical analysis was performed using GraphPad Prism 8.01. ANOVA test followed by a Tukey multiple comparison test was performed and results were expressed as mean ± SEM (n = 6). A probability level of P < 0.05 was accepted as a significant difference.
Footnotes
Acknowledgements
Not applicable
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval to report this case was obtained from Cameroon National Ethics Committee (Reg. N. FWA-IRB 00001954).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
Prior authorization for the use of laboratory animals and all procedures in this study have been obtained from Cameroon National Ethics Committee (Reg. N. FWA-IRB 00001954).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
