Abstract
Objective
In vitro fertilization-embryo transfer (IVF-ET) is a primary treatment method for infertility. It usually requires controlled ovarian hyperstimulation (COH) or repeated COH to obtain more superior oocytes, which could impair ovarian function, reduce embryo quality and limit pregnancy rates. Bushen Tiaojing Fang (BSTJF), our clinically effective prescription for kidney deficiency-related infertility over four decades, while its potential mechanisms to improve pregnancy rates and embryo quality remain unclear.
Methods
In this study, we focus on the approach of network pharmacology to predict key compounds, core targets, and potential mechanisms of BSTJF for blastocyst induced by repeated COH, mainly including drug-likeness (DL) and oral bioavailability (OB) screening, “herb-component-target-blastocyst” (HCTB) and protein-protein interaction (PPI) networks construction and analysis, GO and KEGG enrichment annotation, molecular docking and basic experiments validation.
Results
Network pharmacology analysis identified 29 key targets of BSTJF associated with blastocyst development. GO and KEGG enrichment revealed the key targets mainly involved regulating apoptosis to modulate the cell cycle, and demonstrated significant involvement in Cancer, PI3K-Akt, and HIF-1 signaling pathways. In vivo experiments suggested that BSTJF effectively improved the pregnancy rate of repeated COH mice, increased the number of blastocysts while enhancing the rate of high-quality blastocysts. Immunofluorescence (IF) results showed a significant downregulation of P21 and Bax proteins, and a significant upregulation of Cyclin B1 and Bcl-2 proteins in blastocyst of mice treated with BSTJF.
Conclusion
The study revealed the potential mechanism by which BSTJF could boost embryo quality in mice with repeated COH via activating the PI3K-Akt signaling pathway, which balances the proliferation and apoptosis of blastocyst cells, ultimately restoring normal cell cycle progression and offering new therapeutic strategies for IVF-ET.
Keywords
Introduction
Infertility poses a significant global challenge. According to the World Health Organization, it affects 15% of couples within reproductive age. 1 The rapid advancements in IVF-ET and associated technologies offer hope for those experiencing infertility. COH is crucial to the IVF-ET process, but due to failed transfer cycles, it often needs to be repeated. Our earlier studies have suggested that repeated COH can lead to ovarian dysfunction, 2 resulting in decreased oocyte quality,3,4 which in turn affects pregnancy rate 5 and may hinder early embryo development, reducing blastocyst formation rate and embryo quality. 6 Embryo quality is a key factor affecting IVF pregnancy outcome. 7 Therefore, embryo implantation failure caused by repeated COH is a major obstacle to pregnancy outcomes.
In Traditional Chinese Medicine (TCM), the kidney is considered the storage site for essence and a critical organ for reproduction and development. It is believed that essence transforms into qi, with kidney qi comprising both yin and yang aspects. Oocyte, sperm, and oosperm, considered congenital essence, are governed by the kidney; embryonic development also depends on the nourishment from essence and blood and the warming effect of yang qi. Repeated COH, driven by exogenous gonadotropins (Gn), stimulates the development and ovulation of multiple follicles simultaneously, rapidly depleting kidney essence. This depletion diminishes the developmental potential of oocytes, 8 leading to abnormal pre-implantation embryonic development, an increase in embryo apoptosis, and a decrease in blastocyst formation rate and implantation rate. 9
Bushen Tiaojing Fang, developed by Professor Huilan Du, a nationally renowned TCM doctor, is based on extensive clinical experience and the principle that ‘the kidney governs reproduction’. 9 Adjusted from Wuzi Yanzong Pill and Yangjing Zhongyu Decoction, this formula has shown significant clinical efficacy. Experimental studies have demonstrated that the formula can enhance glycolysis-related enzymes such as G6PDH, PKM2, and LDH, improving the energy metabolism of blastocysts and thus enhancing the quality of mouse blastocysts and promoting embryo implantation. 10 However, the active ingredients, target sites, and mechanisms through which this formula improves early embryo development remain unclear.
Network pharmacology, which embraces both integrity and systematicity, mirrors the holistic concept of traditional medicine and is a crucial method for exploring the mechanisms of TCM formulas.11–13 This study utilizes network pharmacology and molecular docking to explore the potential mechanisms by which BSTJF improves blastocyst quality. By simulating the repeated COH process in clinical practice, we established an animal model to verify the specific roles of key targets and classical pathways, aiming to provide new ideas for the clinical application of IVF-ET (Figure 1).

Flowchart of the Study.
Materials and Methods
Network Pharmacological Analysis
Screening of Active Ingredients and Targets of BSTJF
Effective active components from 13 herbs in BSTJF were retrieved from the Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (TCMSP, https://www.tcmsp-e.com/). The screening criteria included OB ≥ 30% and DL ≥ 0.18. Target data was then verified and revised via the UniProt database (https://www.uniprot.org/), removing non-human genes and merging duplicates, ultimately obtaining standardized gene names.
Acquisition of Blastocyst-Related Targets
We set “blastocyst” as the keyword to collect blastocyst-related targets from the GeneCards database (https://www.genecards.org). These targets were processed by the UniProt database, and then saved for subsequent research.
Screening of Key Active Ingredients and Core Targets
Target genes linked to herbal components and blastocyst were uploaded to the VENNY 2.1 website (http://bioinfogp.cnb.csic.es/tools/venny/index.html),where Venn diagrams delineated overlapping genes, identified as potential targets for the formula's role in blastocyst development. We then inputted the overlapping targets to the STRING 12.0 website (http://cn.string-db.org), which is a free and public database to analyze the interactions among different proteins. On this online website, we set the organism as “Homo sapiens”, while kept other parameters at default settings, then the PPI network was presented. Subsequently, we logged into Cytoscape 3.7.2 software, imported the pre-compiled targets of herbs and blastocyst, one “herb-component-target-blastocyst” network diagram was built, and crucial active ingredients impacting blastocysts were identified based on their interaction degrees.
GO and KEGG Analysis
In order to clarify the biological functions and therapeutic mechanism of BSTJF in improving blastocyst development, we introduced the overlapping genes into the DAVID database (https://david.ncifcrf.gov) for functional annotation. Potential target proteins were delineated for GO functions across Biological Process (BP), Cellular Component (CC), and Molecular Function (MF), adopting a significance cutoff of p < 0.05. The principal 30 features from BP, CC, and MF categories were highlighted for further visualization. Moreover, KEGG pathway enrichment analysis was performed to assess these targets’ involvement in signaling pathways. Results were sorted by PValue, with the top 20 signaling pathways visualized. These targets were used to predict the gene function enrichment processes and signaling pathways in repeated COH treatment, offering a scientific foundation for understanding the mechanism of this formula.
Molecular Docking Validation of Key Components and Core Targets
In evaluating the interaction between primary active ingredients and intersecting targets, we identified five central targets from the PPI network and four pivotal active components from the HCTB network for molecular docking. The SDF files for these active components were sourced from the PubChem database (https://pubchem.ncbi.nlm.nih.gov), and pdb format 3D structures of the central target proteins were procured from the PDB database (https://www.rcsb.org). Target optimization was carried out using PyMOL 2.1.0 by removing water molecules and other small molecule ligands. AutoDock Tools 1.5.6 facilitated the addition of hydrogen and charge adjustments, storing the results in pdbqt format for subsequent molecular docking analysis conducted by Vina 2.0 in PyRx software, which computed binding energies, with values below −5.0 kCal/mol suggesting effective binding activity, and values below −7.0 kCal/mol indicating strong binding activity.
Animal Experiment Validation
Experimental Drugs
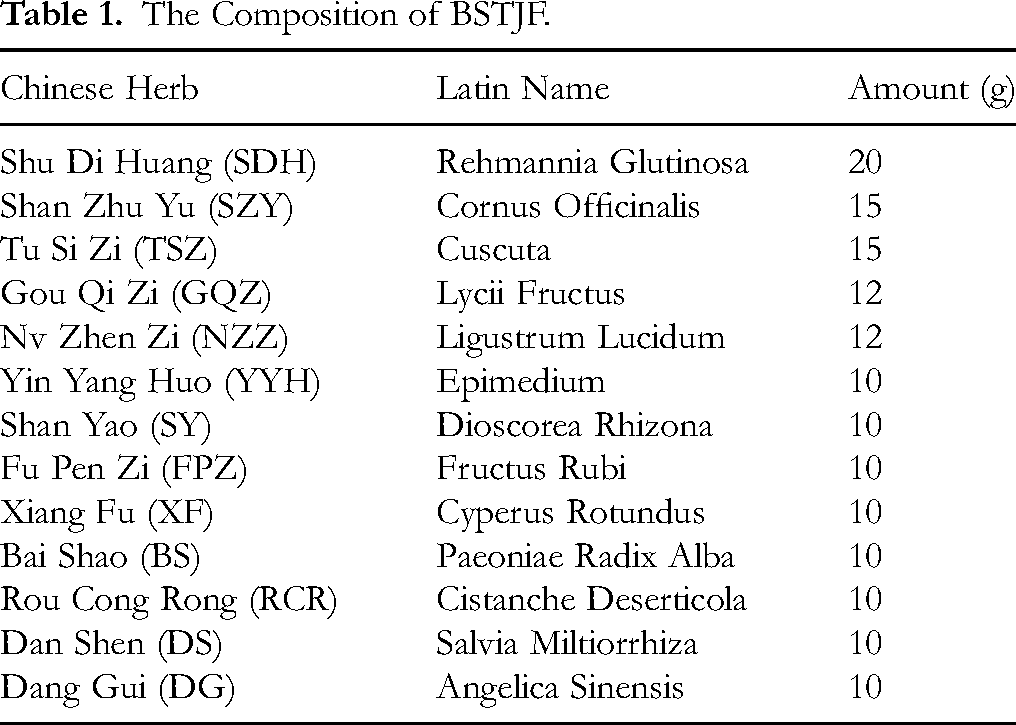
The composition of BSTJF and the amount of each herb are presented in Table 1. Herbal slices were sourced from Shijiazhuang Lerentang Pharmaceutical Co., Ltd After standard decoction, high and low doses were concentrated to 5.4 g/mL and 2.7 g/mL of crude drug, respectively.
The Composition of BSTJF.
The intraperitoneal injection of drugs included pregnant mare serum gonadotropin (PMSG), 1000 IU/vial, was obtained from Hangzhou Animal Pharmaceutical Factory, batch number: 110204564; human chorionic gonadotropin (hCG), 1000 IU/vial, was obtained from Ningbo Second Hormone Factory, batch number: 110251282.
Experimental Animals
The experimental cohort consisted of 152 female ICR mice, aged 4–5 weeks and weighing between 22–28 g, alongside 76 male counterparts, aged 8–10 weeks and weighing 25–30 g, all procured from SBF (Beijing) Biotechnology Co., Ltd under the license SCXK (Beijing) 2024–0001.
These mice were accommodated at the Experimental Animal Center of Hebei University of Chinese Medicine, undergoing a one-week acclimatization period. Female mice underwent vaginal cytology testing twice daily at 9
The model was established following the methods of Lijie Fan
14
and Mengrui Liu.
10
Both the normal and model groups received distilled water by gavage at 9
We can collect the blastocyst cells at 4
Inhibition of PI3K
The embryos were cultured in vitro starting from the 2-cell stage, and normal embryonic development should reach the blastocyst stage by the third day. We collected 2-cell stage embryos at 10
Morphological Observation
When collecting the samples, we randomly selected 10 mice from each group to observe their morphology and count the total number of blastocysts. Meanwhile, we counted the high-quality blastocysts based on the Gardner scoring system and calculated the high-quality blastocyst rate.15,16 The Gardner scoring system was used for blastocyst scoring, classifying the inner cell mass and trophectoderm cells into three grades: Grade A (numerous, tightly arranged cells), Grade B (fewer, loosely arranged cells), and Grade C (very few cells). Per the consensus of the Reproductive Medicine Branch of the Chinese Medical Association, blastocysts scoring greater than or equal to 3BB were classified as high-quality.
RT-qPCR
For each group, three tubes containing frozen blastocyst cells were processed for RNA extraction using the Pico Pure RNA Isolation Kit. RNA purity was assessed using a spectrophotometer, and cDNA was synthesized from mRNA following the Nearshore Protein Kit's three-step protocol. PCR amplification was performed utilizing Servare 2×SYBR Green qPCR Master Mix (Low ROX) to evaluate the expression levels of specific genes. Primers, designed and synthesized by Servare Biotechnology Co., Ltd, and the sequences are shown in Table 2. The relative expression of these genes was quantified using the equation RQ = 2−ΔΔCt, employing GAPDH as the reference gene for subsequent statistical evaluations.
Primer Sequence.

Note: TP53, tumour protein 53; EGFR, epidermal growth factor receptor; STAT3, signal transducer and activator of transcription 3; IL-6, interleukin 6; PI3K, phosphatidylinositol 3-kinase; AKT1: protein kinase B; P21: cyclin-dependent kinase inhibitor 1A; Bcl-2: B cell lymphoma 2; Bax: Bcl2 associated X protein.
IF
For the experimental procedure, 30 blastocysts per group were fixed in 4% paraformaldehyde at room temperature for 30 min. Subsequent washes, each lasting 5 min, were performed twice with PBS. The blastocyst cells then underwent permeabilization with 1% Triton X-100 for 15 min. Following two additional PBS washes, the cells were blocked in 10% goat serum at 37 °C for 10 min. The primary antibody was applied and incubated overnight at 4 °C. The next day, after two PBS washes, cells were incubated with a secondary antibody (1:400) at 37 °C in the dark for 10 min. Finally, after the last washes, the blastocysts were mounted on adhesive slides with a DAPI-contained mounting medium, incubated for 10 min at room temperature, and covered with a cover slip. They were dried in a fume hood before being imaged using an automated live cell imaging system. Fluorescence images were semi-quantitatively analyzed for protein expression using Image-J software.
Statistical Analysis
Data were processed using SPSS 27.0 software. Data adhering to a normal distribution were represented as mean ± standard deviation (
Results
Network Pharmacology Results
Screening of Common Targets Associated with BSTJF and Blastocyst, and Construction of PPI and HCTB Networks
After processing through the TCMSP database, 144 active ingredients of the BSTJF were identified, sourced from various herbs including Salvia miltiorrhiza (65), Goji berry (45), and others. Following conversion, merging, and deduplication through the Uniprot database, 156 effective component targets were obtained (Supplementary Table 1).
By querying the GeneCards database and deduplicating, 436 blastocyst-related genes were identified. The blastocyst-related genes were combined with the BSTJF-associated targets on the VENNY 2.1 online database, resulting in 29 common targets, visualized in a Venn diagram. We analyzed the interaction relationships among the intersection target proteins through STRING 12.0 database, and established a PPI network consisting of 29 nodes, 170 edges, and with an average node degree of 11.7. We then visualized it via the Cytoscape 3.7.2 software based on the degree values (Figure 2A). Notably, high-degree nodes such as TP53, EGFR, STAT3, IL6, and AKT1 are hypothesized as core targets of BSTJF in influencing blastocyst cells development.

Analysis of the Targets Associated with BSTJF and Blastocyst. (A) Venn Diagram of the Overlapping Targets Between BSTJF and Blastocyst, and PPI Network of Overlapping Targets. (B) Network Diagram of “Herb-Component-Target-Blastocyst”. (C) GO Enrichment Analysis. (D) KEGG Pathway Enrichment Analysis.
Regarding the study's computational analysis of key components and core targets, we compiled the “network.xlsx” and “type.xlsx” files to facilitate a HCTB network diagram with Cytoscape 3.7.2 software, displaying 149 nodes and 764 edges. As shown in Figure 2B, the network spotlighted quercetin, kaempferol, luteolin, and beta-sitosterol as principal active components crucial for enhancing blastocyst development.
Analysis of GO Functional Annotation and KEGG Pathway Enrichment
GO enrichment analysis revealed 300 significant entries (P < 0.05), encompassing BP, CC and MF. The top 30 annotation results were visually represented and analyzed based on their p-values (Figure 2C). These core target proteins primarily involve proliferation and apoptosis processes. Additionally, KEGG pathway analysis enriched 109 pathways (P < 0.05), with the top 20 sorted by PValue and visualized (Figure 2D), highlighting pathways such as the PI3K-Akt signaling pathway. Based on the enrichment results of GO and KEGG, we speculate the BSTJF may regulate the dynamic balance of proliferation and apoptosis by acting on the PI3K-Akt signaling pathway, ultimately improving the quality of blastocyst cells induced by repeated COH.
Molecular Docking Verification
Molecular docking revealed that key active components like quercetin, kaempferol, luteolin, and beta-sitosterol bind effectively to core targets including TP53, EGFR, STAT3, IL6, and AKT1 (Supplementary Table 2). These interactions potentially stabilize through forces such as hydrogen bonds and hydrophobic interactions, notably showing strong binding energies with AKT1 across all four components (Figure 3A-D). It is proposed that these interactions facilitate the formula's impact on blastocysts of repeated COH mice.

Molecular Docking Diagram of key Active Compounds and Core Targets. (A) Beta Sitosterol and AKT1. (B) Quercetin and AKT1. (C) Luteolin and AKT1. (D) Kaempferol and AKT1.
Analysis of Experimental Results
Impact of BSTJF on Pregnancy Rate and Blastocyst Development
As presented in Figure 4A, the pregnancy rate in the model group is significantly lower than that in the normal group, while the pregnancy rates in groups treated with BSTJF have increased significantly. Moreover, the pregnancy rate in BSTJFH is slightly higher than that in BSTJFL.

Effect of BSTJF on Pregnancy Rate and Blastocyst Development in Repeated COH Mice. (A) the Positive Effect of BSTJF on the Pregnancy Rate. (B) the Positive Effect of BSTJF on the Morphological Development; a) Normal Group; b) Model Group; c) BSTJFL Group; d) BSTJFH Group. the red Arrows Indicate Broken Blastocysts. (C) the Number of Blastocysts Collected by Each Group. (D) the Positive Effect of BSTJF on the High-Quality Rate. *P < 0.05, **P < 0.01, ***P < 0.001.
Morphological observations indicated a significant increase in the number of blastocysts in mice subjected to repeated COH; however, the quality of high-quality blastocysts declined, with a large amount of debris visible inside the blastocyst cell. The mice treated with BSTJF showed a significant improvement in the quantity and quality of high-quality blastocysts, demonstrating enhanced blastocyst development (Figure 4B-D).
Impact of BSTJF on mRNA Expression and PI3K-Akt Signaling Pathway
We employed the RT-qPCR method to analyze the mRNA expression levels of key targets in blastocyst cells of four groups. As shown in Figure 5A, in the model group, there is a clearly downward trend in the levels of EGFR, STAT3, PI3K and AKT1, while P53 and IL-6 show an upward trend. For the BSTJFL and BSTJFH groups, the tread of the aforementioned key targets is reversed, and their expression levels are close to the normal group. Furthermore, there is no significant difference between BSTJFL and BSTJFH. These results suggest that the formula's therapeutic effects may be linked to the modulation of these genes’ expression.

Effect of BSTJF on mRNA Expression and PI3K-Akt Signaling Pathway in Repeated COH Mice. (A) the Regulating Effect of BSTJF on the mRNA Expression of TP53, EGFR, STAT3, IL-6, PI3K and AKT1. (B) the Improved Effect of BSTJF on the Protein Levels of p-PI3K, p-AKT1 and p-STAT3. *P < 0.05, **P < 0.01, ***P < 0.001.
PI3K-Akt signaling pathway is one of the most pathways enriched in our studies. 17 To determine the effect of BSTJF on this essential signaling pathway in the development of blastocysts, we measured the phosphorylation levels of PI3K, AKT1, and STAT3 by IF method. As illustrated in Figure 5B, compared to the normal group, phosphorylation levels of three proteins are reduced in the model group; however, the formula's administration significantly enhance the phosphorylation of PI3K, AKT1, and STAT3, thus activating the PI3K-Akt signaling pathway. Given the vital role of the PI3K-Akt signaling pathway in maintaining the cell cycle, we hypothesize that BSTJF might inhibit cell apoptosis and promote proliferation of cell growth factors by activating the PI3K-Akt signaling pathway, thereby improving embryo quality.
Impact of BSTJF on Proliferation and Apoptosis
To verify the effect of BSTJF on the cell cycle operation of blastocysts, we randomly selected 10 blastocysts from each group to count the number of internal cells, and then evenly divided these blastocysts to detect the proliferation rate and apoptosis rate by means of EDU and TUNEL methods. Although there is no significant difference in the number of internal cells among the four groups (Figure 6A), the apoptosis rate is significantly increased, and the proliferation rate is significantly decreased in the model group; however, the BSTJF groups show an opposite trend compared to the model group (Figure 6B-C).

Effect of BSTJF on Proliferation and Apoptosis in Repeated COH Mice. (A) the Number of Internal Cell Spheres of Blastocysts in Each Group. (B) the Rising Effect of BSTJF on the EDU Positive Rate. (C) the Falling Effect of BSTJF on the Apoptosis Rate. (D) the Regulating Effect of BSTJF on the mRNA Expression of P21, Cyclin B1, Bcl-2 and Bax. (E-F) the Regulating Effect of BSTJF on the Protein Levels of P21, Cyclin B1, Bcl-2 and Bax. *P < 0.05, **P < 0.01, ***P < 0.001.
Subsequently, we analyzed the expression levels of the factors closely related to proliferation and apoptosis. The data visualization results revealed that the protein and mRNA levels of P21 and Bax were elevated in the model group, while those of CyclinB1 and Bcl-2 were reduced. Interestingly, the treatment with BSTJF was able to largely reverse the expression levels of these factors. In addition, the BSTJFH group demonstrated a greater advantage than BSTJFL group (Figure 6D-F). The above results indicated that repeated COH may disrupt the balance of proliferation and apoptosis in blastocysts. Importantly, the intervention of BSTJF can restore this balance.
BSTJF Activates the PI3K-Akt Signaling Pathway to Balance Proliferation and Apoptosis
In terms of embryo development, the addition of PI3K inhibitors leads to the stagnation of in vitro embryo development and a decline in embryo quality in repeated COH mice. As depicted in Figure 7A, compared with 0ummol/L, the blastocyst formation rates at 50ummol/L, 100ummol/L, and 200ummol/L interventions are significantly reduced. Moreover, as the concentration increases, the blastocyst formation rate gradually decreases. Considering economic and safety, we determine the concentration of the inhibitor at 50ummol/L for subsequent research.

Mechanism of BSTJF on Promoting Blastocyst Development in Repeated COH Mice. (A) The effects of different concentrations of PI3K inhibitor on the blastocyst formation rate in vitro. Compared to 0ummol/L, *P < 0.05, **P < 0.01, ***P < 0.001; Compared to 25ummol/L, #P < 0.05, ##P < 0.01, ###P < 0.001; Compared to 50ummol/L, △P < 0.05, △△P < 0.01. (B) The negative effect of PI3K inhibitor on the morphological development. (C-D) Inhibition of PI3K could reserve the therapeutic effects of BSTJF in blastocyst induced by repeated COH. *P < 0.05, **P < 0.01, ***P < 0.001.
The PI3K-Akt signaling pathway is a well-known mediator of cell growth, proliferation, apoptosis, and survival signals.18–20 To verify whether the BSTJF improves embryonic development by activating the PI3K-Akt signaling pathway, we further confirmed through cell experiments. We collected the 2-cell embryos for in vitro culture. Among them, the BSTJF group and the BSTJF + LY294002 group were given drug-containing serum obtained from rats that were gavaged with high-dose formula. The development of blastocysts in each group was observed on the third day. It is evident from the Figure 7B that the normal group and the BSTJF group have more regular blastocyst morphology, better development, and less fragmentation, while the model group and the BSTJF + LY294002 group have poorer blastocyst development, irregular cell morphology, and more fragmentation. Further analysis of the IF results shows that compared with the normal group, the levels of P21 and Bax proteins in the model group are significantly increased, while the protein levels of Cyclin B1 and Bcl-2 are decreased, and these changes are effectively improved in the BSTJF group. However, after combinational intervention with PI3K inhibition, the improvement effects are reversed (Figure 7C-D).
Discussion
IVF-ET is one of the most mature and common techniques for treating infertility. 21 COH is a considerable method in IVF-ET, 22 which can increase the number of mature oocytes per cycle and generate more embryos for transplantation. However, more and more studies confirm that COH, especially repeated COH, can reduce the developmental potential of early embryos.23–25 The quality of embryos before transplantation is one of the key factors determining IVF-ET pregnancy outcomes.26,27 Currently, modern medicine mainly employs symptomatic prevention and treatment methods to maintain the growth of early embryos. However, these methods are limited by various clinical factors and lack large-sample randomized controlled trials as well as evidence-based medical support. TCM believes that the COH will deplete a large amount of kidney essence in a short period, and repeated COH further aggravates this depletion, leading to a decline in the developmental potential of oocytes and causing early embryonic maldevelopment. Moreover, the more the number of COH occurrences, the more severe the embryo development abnormalities will be. 28 Therefore, tonifying the kidney and enriching kidney essence is the key to treating repeated COH.
BSTJF is an empirical formula based on the mentor's clinical experience of over 40 years, which has been clinically proven to increase the number of oocytes retrieved, fertilization rate, high-quality embryo rate, and clinical pregnancy rate in patients undergoing repeated COH.29–32 It can also improve the oxidative stress of ovarian granulosa cells in patients with repeated COH, 33 thereby improving pregnancy outcomes. This formula incorporates multiple herbs aimed at nourishing yin, enriching blood, and enhancing essence. Rehmannia glutinosa, the principal herb, supports yin and enriches blood. Cornus officinalis and Lycii fructus target the liver and kidney yin through the thoroughfare and conception vessels. Dioscorea rhizona and Ligustrum lucidum, alongside principal herbs, fortify the kidney and essence, while Fructus rubi, Epimedium, Cuscuta, and Cistanche deserticola serve as ministerial drugs, warming the kidney and enriching essence. Cyperus rotundus addresses qi disorders. Paeoniae radix alba, Salvia miltiorrhiza, and Angelica sinensis act as assistant and guiding drugs, regulating qi and blood and maintaining yin-yang balance. This synergistic combination promotes the abundance of congenital essence and enhances physiological endowment.
Network pharmacology approaches pinpointed quercetin, kaempferol, luteolin, and beta-sitosterol as the primary active compounds in the formula, exerting substantial effects on blastocyst development in repeated COH mice. Quercetin has been shown to affect embryonic development by influencing oocyte apoptosis during the fetal period and by promoting embryo survival by inhibiting apoptosis.34,35 Kaempferol enhances embryonic development by regulating autophagy, activating the PI3K/Akt/AMPK signaling pathway to support primordial follicle development and ovarian cell proliferation.36–38 During the implantation window, kaempferol may enhance endometrial receptivity by modulating autophagy levels, thus fostering successful embryo implantation and development.39–41 Luteolin, a natural flavonoid, regulates the cell cycle and influences cell proliferation. It can finely adjust the cell cycle to promote or inhibit cell proliferation at specific developmental stages, ensuring normal embryo development.42–44 Luteolin also enhances endometrial receptivity by activating autophagy-related signaling pathways like PI3K/Akt/mTOR, thus supporting embryo implantation and early development.45–47 Additionally, luteolin positively impacts the decidualization of the endometrium, essential for successful embryo implantation. 48 Beta-sitosterol, recognized for its role in embryonic development, may regulate embryonic cell proliferation and apoptosis through cell cycle-related signaling pathways, influencing gene expression via pathways like JAK2/STAT3, thus supporting normal embryonic development.49–51 In conclusion, the active components in the BSTJF exhibit various properties that promote cell proliferation, inhibit apoptosis and inflammation, thus improving blastocyst development in mice induced by repeated COH.
In this study, we have confirmed TP53, EGFR, STAT3, IL6, and AKT1 are the core targets involved in blastocyst development through PPI analysis. During early embryonic development, appropriate TP53 expression regulates the cell cycle, fosters cell differentiation, and ensures normal embryonic progression. 52 EGFR influences cell proliferation, differentiation, and migration by activating the PI3K/Akt signaling pathway,53,54 which is vital for ensuring cell function and positioning during early embryo development. As a transcription factor, STAT3 is essential in cell signal transduction.55,56 Its activation supports cell growth and survival, which is vital for early embryonic and organ development.57–59 IL-6, a key cytokine, regulates immune and inflammatory responses and significantly contributes to early embryonic development by modulating intercellular signaling. 60 AKT1, a pivotal element of the PI3K/Akt signaling pathway, plays a crucial role in a range of biological functions including cell proliferation, metabolism, and survival, primarily through its phosphorylation and dephosphorylation activities.61–63
This study's GO functions show that the biological processes by which the BSTJF promotes blastocyst development mainly include positive regulation of gene expression, apoptotic process, and negative regulation of autophagy. KEGG analysis results indicate that the PI3K-Akt signaling pathway is one of the crucial pathways through which the BSTJF improves repeated COH blastocyst, and this pathway is related to cell proliferation, apoptosis, and immune regulation. What's more, the molecular docking results have showed that the core targets, especially AKT1, exhibit strong binding energy with the active components, this means the core targets have excellent affinity with the key components. Therefore, we reasonably believe that BSTJF can effectively improve the development of blastocysts induced by repeated COH, and its mechanism may be related to the regulation of the PI3K-Akt signaling pathway.
To further determine the results of network pharmacology analysis, we established a repeated COH model in mice and collected blastocyst cells for evaluation and detection. Experimental data revealed that the BSTJF significantly improved the morphology of blastocyst cells in the repeated COH model mice, activated the PI3K-Akt signaling pathway, and promoted the proliferation of blastocysts while inhibiting apoptosis by upregulating Cyclin B1 and Bcl-2 levels, and downregulating P21 and Bax levels. Among these regulatory factors, P21 and Cyclin B1 are proliferation-related factors, with P21 inhibiting proliferation and Cyclin B1 promoting proliferation; Bcl-2 and Bax are apoptosis-related factors, with Bcl-2 inhibiting apoptosis and Bax promoting apoptosis.
In summary, this study preliminarily reveals the main active components, core targets and potential molecular mechanisms of BSTHF in treating blastocysts induced by repeated COH through network pharmacology analysis combined with in vivo and in vitro experiments, providing a scientific basis for the clinical application of the formula to improve IVF-ET blastocyst quality. However, this study still has several limitations. For example, we screened the active components and targets of herbs from public databases, which may differ from the actual efficacy; in addition, due to time constraints and difficulties in obtaining embryonic specimens, we only observed the development of blastocysts, and we only used IF and RT-qPCR for experimental validations. In future research, we will attempt to use ultra-high-performance liquid chromatography-tandem mass spectrometry for quality control analysis of BSTJF and expand the concentration gradient of the experimental formula to find the optimal clinically effective dose. Furthermore, we will also focus on the research of embryos at all stages before implantation, improve sampling methods, and optimize experimental procedures, such as Western blotting, single-cell RNA sequencing, to explore other mechanisms by which the BSTJF improves early embryonic development.
Conclusion
In this research, we have provided evidence that BSTJF improves embryo quality by activating the PI3K-Akt signaling pathway, which can restore normal cell cycle progression by promoting blastocyst proliferation and inhibiting blastocyst apoptosis. The theoretical and experimental evidence offers new therapeutic strategies for IVF-ET.
Supplemental Material
sj-xlsx-1-npx-10.1177_1934578X251385769 - Supplemental material for Potential Mechanism of Bushen Tiaojing Fang on Development of Blastocysts in Mice Induced by Repeated Controlled Ovarian Hyperstimulation: Comprehensive Evidence from Network Pharmacology, Molecular Docking and Experimental Validation
Supplemental material, sj-xlsx-1-npx-10.1177_1934578X251385769 for Potential Mechanism of Bushen Tiaojing Fang on Development of Blastocysts in Mice Induced by Repeated Controlled Ovarian Hyperstimulation: Comprehensive Evidence from Network Pharmacology, Molecular Docking and Experimental Validation by Beibei Shi, Lulu Shen, Wenyan Xiong, Yajing Song, Ruigan Shi and Huilan Du in Natural Product Communications
Supplemental Material
sj-xlsx-2-npx-10.1177_1934578X251385769 - Supplemental material for Potential Mechanism of Bushen Tiaojing Fang on Development of Blastocysts in Mice Induced by Repeated Controlled Ovarian Hyperstimulation: Comprehensive Evidence from Network Pharmacology, Molecular Docking and Experimental Validation
Supplemental material, sj-xlsx-2-npx-10.1177_1934578X251385769 for Potential Mechanism of Bushen Tiaojing Fang on Development of Blastocysts in Mice Induced by Repeated Controlled Ovarian Hyperstimulation: Comprehensive Evidence from Network Pharmacology, Molecular Docking and Experimental Validation by Beibei Shi, Lulu Shen, Wenyan Xiong, Yajing Song, Ruigan Shi and Huilan Du in Natural Product Communications
Supplemental Material
sj-docx-3-npx-10.1177_1934578X251385769 - Supplemental material for Potential Mechanism of Bushen Tiaojing Fang on Development of Blastocysts in Mice Induced by Repeated Controlled Ovarian Hyperstimulation: Comprehensive Evidence from Network Pharmacology, Molecular Docking and Experimental Validation
Supplemental material, sj-docx-3-npx-10.1177_1934578X251385769 for Potential Mechanism of Bushen Tiaojing Fang on Development of Blastocysts in Mice Induced by Repeated Controlled Ovarian Hyperstimulation: Comprehensive Evidence from Network Pharmacology, Molecular Docking and Experimental Validation by Beibei Shi, Lulu Shen, Wenyan Xiong, Yajing Song, Ruigan Shi and Huilan Du in Natural Product Communications
Supplemental Material
sj-tif-4-npx-10.1177_1934578X251385769 - Supplemental material for Potential Mechanism of Bushen Tiaojing Fang on Development of Blastocysts in Mice Induced by Repeated Controlled Ovarian Hyperstimulation: Comprehensive Evidence from Network Pharmacology, Molecular Docking and Experimental Validation
Supplemental material, sj-tif-4-npx-10.1177_1934578X251385769 for Potential Mechanism of Bushen Tiaojing Fang on Development of Blastocysts in Mice Induced by Repeated Controlled Ovarian Hyperstimulation: Comprehensive Evidence from Network Pharmacology, Molecular Docking and Experimental Validation by Beibei Shi, Lulu Shen, Wenyan Xiong, Yajing Song, Ruigan Shi and Huilan Du in Natural Product Communications
Supplemental Material
sj-tif-5-npx-10.1177_1934578X251385769 - Supplemental material for Potential Mechanism of Bushen Tiaojing Fang on Development of Blastocysts in Mice Induced by Repeated Controlled Ovarian Hyperstimulation: Comprehensive Evidence from Network Pharmacology, Molecular Docking and Experimental Validation
Supplemental material, sj-tif-5-npx-10.1177_1934578X251385769 for Potential Mechanism of Bushen Tiaojing Fang on Development of Blastocysts in Mice Induced by Repeated Controlled Ovarian Hyperstimulation: Comprehensive Evidence from Network Pharmacology, Molecular Docking and Experimental Validation by Beibei Shi, Lulu Shen, Wenyan Xiong, Yajing Song, Ruigan Shi and Huilan Du in Natural Product Communications
Footnotes
Acknowledgements
The authors would like to thank Lijie Fan for the technical assistance in the establishment of the animal model. The progress would not have been possible, without her expertise and enthusiasm.
Ethical Considerations
Ethics Approval to report this research was obtained from Hebei University of Chinese Medicine (APPROVAL NUMBER: DWLL2020032).
Author Contributions
The authors confirm contribution to the paper as follows: study conception and design: Beibei Shi and Huilan Du; data collection: Beibei Shi, Lulu Shen and Wenyan Xiong; analysis data: Yajing Song and Ruigan Shi; draft manuscript preparation: Beibei Shi and Huilan Du. All authors reviewed the results and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded partially by the National Natural Science Foundation of China (82074486), and the Innovative Funding for Graduate Students in Hebei Province project (XCXZZBS2024013).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
All data generated or analyzed during this study are included in this published article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
